Introduction
Breast cancer is the most common cancer and
continues to be a major cause of morbidity and mortality among
women worldwide (1). Global
statistics show that >1.15 million women each year are diagnosed
with breast cancer, which results in >502,000 mortalities
(2–4). This multifactorial disease occurs as a
result of several risk factors, including female gender, increasing
age, familial history of breast cancer, diet, obesity and life
behavior, such as smoking and alcohol consumption (5,6). Based
on the increased incidence of breast cancer (2), early detection will improve treatment
success. In this regard, investigating appropriate oncomarkers for
early detection, monitoring and treatment appears to be crucial.
Due to limited treatment options, resistances to current standard
therapies, the short duration of response and rapid relapse
(7,8), finding novel therapeutics is considered
as extremely important. MicroRNA (miR) may be one of these
therapeutic targets.
miR are class of endogenously expressed,
single-stranded, non-protein coding small RNA that bind to target
mRNA. miR have important roles in diverse biological and
pathological processes via both transcriptional and
post-transcriptional gene regulation, making them attractive
biomarkers in cancer (9,10). Research has demonstrated that miR
have important roles in regulating cell processes that are often
deregulated in cancer, including proliferation, differentiation and
apoptosis (11). The miR-29 family
has three main members, including hsa-miR-29a, hsa-miR-29b and
hsa-miR-29c (12). Research has
indicated that miR-29a negatively regulates the expression of
Myb-related protein B, resulting in inhibition of breast cancer
growth and arrest of cells in the G0/G1 phase (13). Downregulation of myeloid cell
leukemia 1, as an anti-apoptotic protein, by miR-29b in malignant
KMCH cholangiocarcinoma cells has been reported (14). Although the mir-29 family is involved
in regulating tumorigenesis and development of various types of
cancer, the role and the underlying mechanism of miR-29a in breast
cancer remains to be fully elucidated (15).
B-cell lymphoma 2 (BCL-2) is a prototypical member
of the BCL-2 family, which has been identified as a regulator of
apoptosis (16). This molecule
prevents the mitochondrial pathway of apoptosis (16,17). It
has been reported that the miR-15a and miR-16-1 region is deleted
in chronic lymphocytic leukemia, which leads to an aggressive state
(18). These miR naturally induce
apoptosis in normal cells through regulation of the anti-apoptotic,
proto-oncogene BCL-2 (18). BCL-2
expression may be downregulated by miR-34, which is compatible with
the role of miR-34 in apoptosis mediated by p53 expression
(19). p53 is a pleiotropic
regulator of cell fate, which may facilitate apoptosis (20,21). miR
have been identified to regulate p53 expression by controlling the
upstream regulation of p53 and its pro-apoptotic function, as
research has demonstrated that miR-125 negatively regulates p53
(22). Research has indicated that
the expression level of p53 protein was decreased and apoptosis was
stopped in human neuroblastoma and in lung fibroblasts (22). p21 is induced by p53-dependent and
-independent mechanisms implicated in both major functions of the
tumor suppressor-cell cycle arrest and apoptosis (23).
Survivin is overexpressed in the majority of human
malignancies and functions as a key regulator of mitosis and
programmed cell death (24,25). Research has demonstrated that miR-218
is involved in breast cancer evasion of apoptosis by targeting
survivin (26). This molecule has
been targeted for therapy in cancer based on its selective
expression in tumors, but not normal tissues (27,28). The
present study aimed to evaluate miR-29a knockdown on the expression
of important genes involved in cell apoptosis and proliferation of
breast carcinoma. In particular, it was proposed that knockdown of
miR-29a may alter the expression profile of p53, p21, BCL-2 and
survivin genes.
Materials and methods
Cell line
The MCF-7 human breast cancer line was obtained from
the Iranian Stem Cell Technology Research Center (Tehran, Iran) and
cultured in RPMI-1640 culture medium supplemented with 10% fetal
bovine serum and 1% penicillin and streptomycin (all Gibco; Thermo
Fisher Scientific, Inc., Waltham, MA, USA) at 37°C in a 5%
CO2 humidified incubator. At ~80% confluence, adherent
cultured cells were harvested following trypsin treatment at 37°C
for 3 min and used for transfection.
Reverse transfection of MCF-7 cells
with anti-miR-29a
Cells were transfected using Lipofectamine reagent
(Invitrogen; Thermo Fisher Scientific, Inc.) with anti-miR-29a
(MIMAT0000086; 5′-UAGCACCAUCUGAAAUCGGUUA-3′; Qiagen, Inc.,
Valencia, CA, USA), at a final concentration of 50 nM, and scramble
at a final concentration of 5 nM in opti-MEM medium (Invitrogen;
Thermo Fisher Scientific, Inc.), as described previously (29). Following transfection, MCF7 cells
were treated with Taxol [1, 10 and 50 ng/ml; concentrations
determined using MTT assay (data not shown)] to inhibit cell growth
and trigger apoptosis. Taxol (Paclitaxel) was purchased from Sobhan
Oncology Co., (Tehran, Iran). Cells were harvested after 24 h of
incubation with Taxol at 37°C in a 5% CO2 humidified
incubator.
Viability assessment
Studied cells were categorized into the nine
following groups: Non-treated; scramble; anti-miR-29a; anti-miR-29a
+ Taxol (1 ng/µl); anti-miR-29a + Taxol (10 ng/µl); anti-miR-29a +
Taxol (50 ng/µl); Taxol (1 ng/µl); Taxol (10 ng/µl); and Taxol (50
ng/µl). A total of 72 h after anti-miR-29a and Taxol treatment in a
5% humidified CO2 incubator, 100 µl MTT (5 mg/ml) was
added to the cells. Optical absorbance of generated formazan was
evaluated at a wavelength of 590 nm in each group 4 h following
replacement of supernatant by dimethyl sulfoxide. All experiments
were conducted three times.
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted from cultured cells in the
different groups using an easy-BLUE TM Total RNA Extraction kit
(iNtRON Biotechnology, Inc., Sungnam, Korea), according to the
manufacturer's instructions, 72 h after transfection and treatment
with Taxol. RNA quality and concentration were determined after
extraction using a Nanodrop spectrophotometer (Thermo Fisher
Scientific, Inc.) and electrophoresis on 2% agarose gel. cDNA was
generated using a cDNA Synthesis H Minus First Strand Revert Aid
kit (Thermo Fisher Scientific, Inc.), according to the
manufacturer's instructions. Sequences of specific primers for stem
loop RT-PCR cDNA synthesis of miR-29a (target gene) and U6 small
nuclear (sn)RNA (reference gene) were
5′-GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATCAGACTAACCGAT-3′ and
5′-AAAATATGGAACGCTTCACGAATTTG-3′, respectively. Using general
primers (random and oligo dT primers) and a Maxime RT premix cDNA
synthesis (iNtRON Biotechnology, Inc.) the cDNA for quantifying
p21, p53, BCL-2 and survivin were synthesized.
qPCR was performed using a 2X Real MOD Green PCR
Master Mix kit (Takara Bio, Inc., Otsu, Japan), according to the
manufacturer's instructions. Briefly, 2 µl cDNA product was diluted
in a final volume of 20 µl, containing 10 pmol of each primer, 10
µl 2× reaction mixture of SYBR Green and 7.4 µl sterile deionized
water. The cycling program was as follows: Initial denaturation at
95°C for 10 min, followed by 40 cycles of denaturation at 95°C for
10 sec, annealing and extension at 55°C for 40 sec. U6 snRNA and
hypoxanthine-guanine phosphoribosyltransferase (HGPRT) were used as
internal housekeeping controls. All reactions were performed in
triplicate and relative quantity of gene expression was analyzed
using the Pfaffl method (30).
Sequences of primers used for quantification of the desired genes
involved in apoptosis are presented in Table I.
 | Table I.Primers used for reverse
transcription-quantitative polymerase chain reaction. |
Table I.
Primers used for reverse
transcription-quantitative polymerase chain reaction.
| Gene | Direction | Primer
sequence |
|---|
| MicroRNA-29a | F |
5′-TGCGCTAGCACCATCTGAAA-3′ |
|
| R |
5′-CAGTGCAGGGTCCGAGGAT-3′ |
| U6 small nuclear
RNA | F |
5′-CTCGCTTCGGCAGCACATATAC-3′ |
|
| R |
5′-ACGCTTCACGAATTTGCGTGTC-3′ |
| p21 | F |
5′-GAGGCCGGGATGAGTTGGGAGGAG-3′ |
|
| R |
5′-CAGCCGGCGTTTGGAGTGGTAGAA-3′ |
| p53 | F |
5′-TAACAGTTCCTGCATGGGCGGC-3′ |
|
| R |
5′-AGGACAGGCACAAACACGCACC-3′ |
| B-cell lymphoma
2 | F |
5′-GGTGGGGTCATGTGTGTGG-3′ |
|
| R |
5′-CGGTTCAGGTACTCAGTCATCC-3′ |
| Survivin | F |
5′-AGAACTGGCCCTTCTTGGAGG-3′ |
|
| R |
5′-CTTTTTATGTTCCTCTATGGGGT-3′ |
|
Hypoxanthine-guanine
phosphoribosyltransferase | F |
5′-GGACAGGACTGAACGTCTTG-3′ |
|
| R |
5′-ATAGCCCCCCTTGAGCACAC-3′ |
Statistical analysis
Results were expressed as the mean ± standard
deviation and analyzed using SPSS v. 16.0 software (SPSS, Inc.,
Chicago, IL, USA). Differences between groups was evaluated by
Kruskal-Wallis tests, and comparison of studied groups for the
expression of the desired genes was assessed by Mann-Whitney U
tests. P<0.05 was considered to indicate a statistically
significant difference.
Results
Knockdown of the miR-29a gene in the
MCF-7 cell line
Downregulating the expression of miR-29a was the
initial aim of the present experiment. In order to achieve this,
cells were treated with anti-miR-29a and scramble. The cycle
threshold of miR-29a was normalized with U6 snRNA as a reference
gene and compared with the non-treated group as a calibrator. As
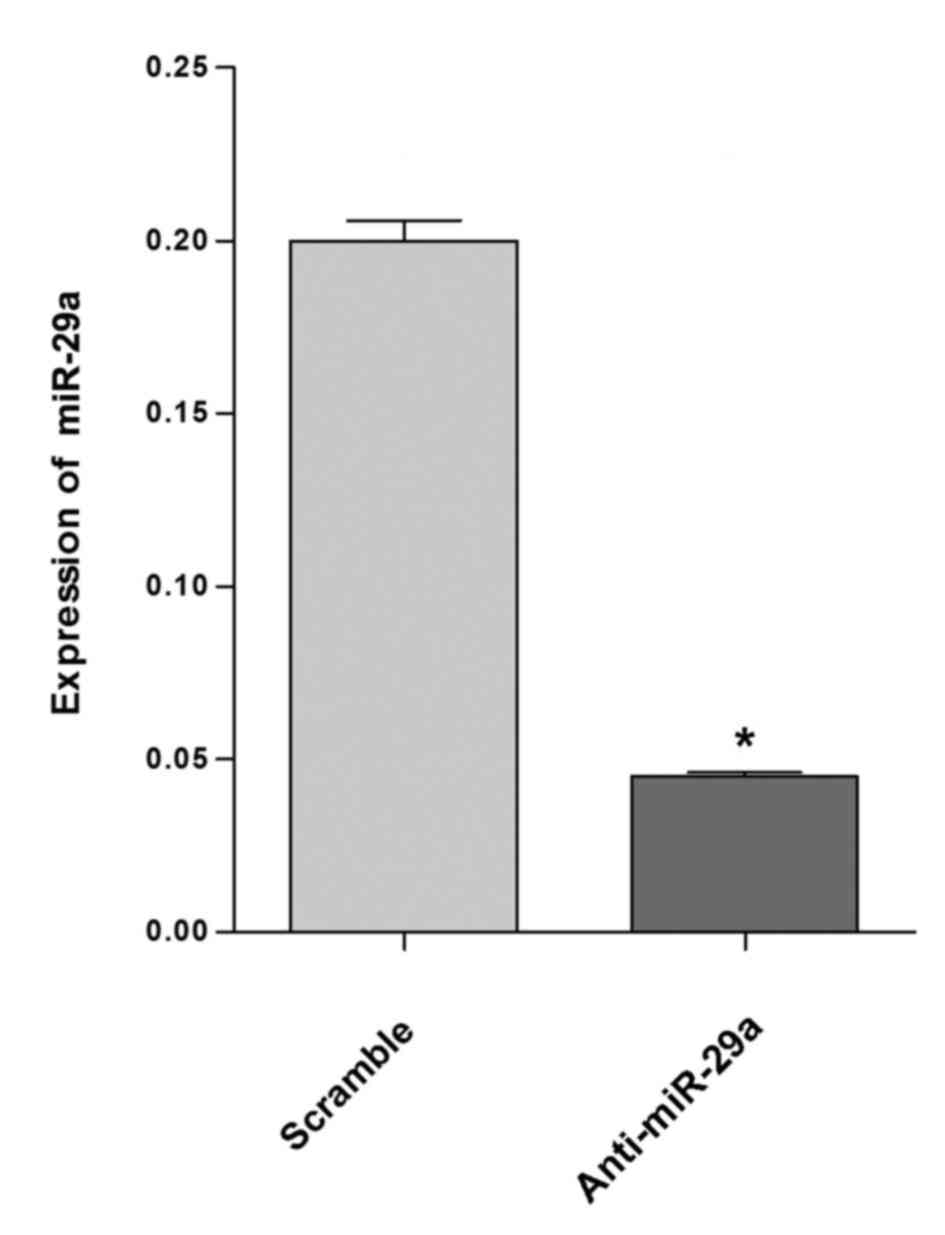
demonstrated in Fig. 1, a
significant reduction of miR-29a expression was observed following
anti-miR-29a treatment compared with the scramble treatment
(P<0.05). In cells transfected with anti-miR-29a, the expression
of miR-29a was decreased by up to 75% compared with the
scramble-treated group.
Effect of miR-29a suppression on cell
viability
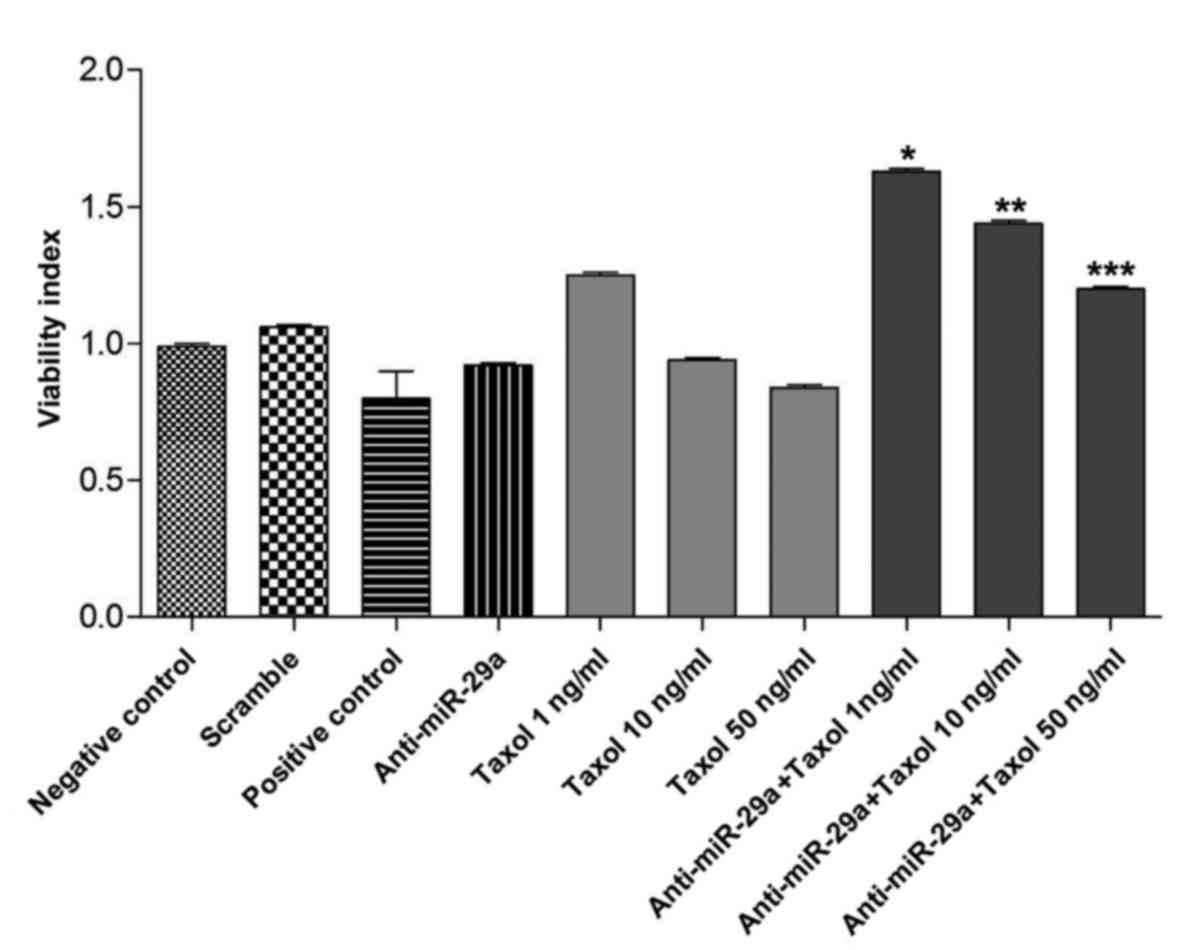
The viability of MCF-7 cells following knockdown of
miR-29a was evaluated 72 h post-transfection with anti-miR-29a. As
demonstrated in Fig. 2, the
viability of cells transfected with anti-miR-29a was reduced
compared with the scramble and negative control groups; however,
this difference was not significant (P>0.05). According to MTT
assay, 10 ng/ml Taxol was selected as the optimal concentration.
With increasing concentrations of Taxol, cell viability of MCF-7
cells decreased; however, transfection with anti-miR-29a and
treatment with Taxol simultaneously resulted in significant
increases in the viability of cells compared to treatment with
Taxol alone (P<0.05). Therefore, anti-miR-29a induces
modification of cell viability following treatment with Taxol.
BCL-2 gene expression decreases
following knockdown of miR-29a
Fold changes in the expression levels of genes,
including p21, p53, BCL-2 and survivin, are represented in Table II. The cycle threshold of target
genes was normalized against HGPRT as a reference gene. Fold
changes were obtained by dividing the cycle threshold of each
treatment group by the cycle threshold of the non-treated group.
Nine groups of MCF-7 cells for each gene were treated: Non-treated
(calibrator); treated by scramble; anti-miR-29a; anti-miR-29a +
Taxol 1 ng/ml; anti-miR-29a + Taxol 10 ng/ml; anti-miR-29a + Taxol
50 ng/ml; Taxol 1 ng/ml; Taxol 10 ng/ml; and Taxol 50 ng/ml.
Kruskal-Wallis and Mann-Whitney U non-parametric tests were used
for evaluating the differences in expression levels of the desired
genes between the experimental groups.
 | Table II.Fold changes in p21, p53, BCL-2 and
survivin gene expression in the experimental groups. |
Table II.
Fold changes in p21, p53, BCL-2 and
survivin gene expression in the experimental groups.
|
| Gene |
|---|
|
|
|
|---|
| Group | p21 | p53 | BCL-2 | Survivin |
|---|
| Scramble |
0.32±0.070 |
1.9±0.080 |
3.27±0.701 |
0.97±0.021 |
| Anti-miR-29a |
1.06±0.030 |
1.4±0.051 |
2.1±0.070 |
0.8±0.020 |
| Anti-miR-29a +
Taxol 1 ng/ml |
1.46±0.090 |
1.06±0.011 |
2.3±0.061 |
1.07±0.020 |
| Anti-miR-29a +
Taxol 10 ng/ml |
0.4±0.005 |
0.5±0.003 |
0.49±0.030 |
1.6±0.061 |
| Anti-miR-29a +
Taxol 50 ng/ml |
0.47±0.011 |
0.78±0.110 |
0.62±0.021 |
0.6±0.040 |
| Taxol 1 ng/ml |
0.2±0.006 |
0.42±0.070 |
0.16±0.003 |
1.2±0.090 |
| Taxol 10 ng/ml |
0.1±0.005 |
0.24±0.030 |
0.12±0.003 |
1.16±0.051 |
| Taxol 50 ng/ml |
0.19±0.020 |
0.14±0.011 |
0.43±0.008 |
1.28±0.010 |
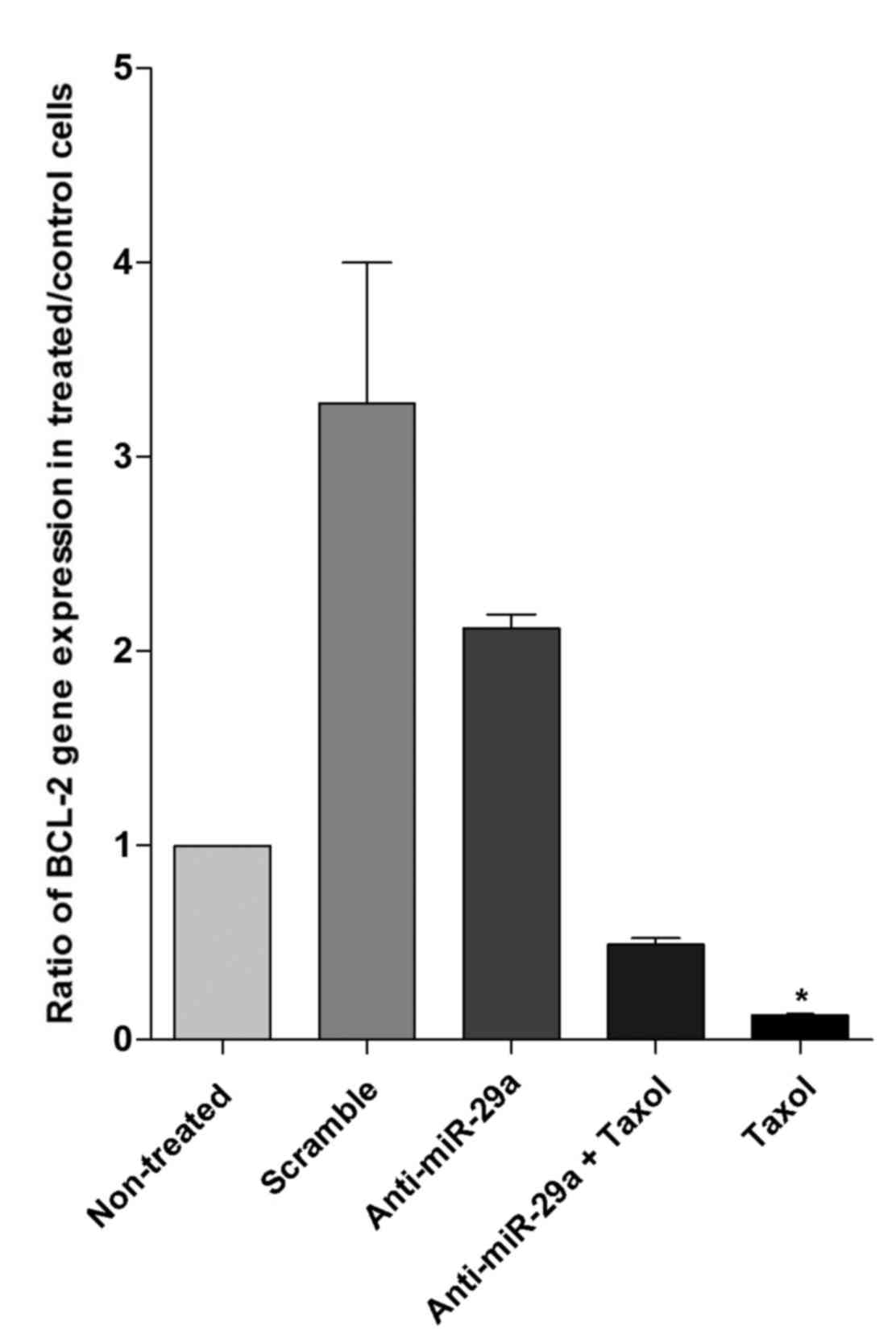
Results demonstrated that BCL-2 gene expression
decreased following knockdown of miR-29a. It has been indicated
that BCL-2 has a critical role in the pathogenesis of various types
of cancer by regulating apoptosis (31). Thus, the expression of BCL-2 was
measured in the different treatment groups. Based on RT-qPCR, it
was observed that in cells transfected with anti-miR-29a, the gene
expression of BCL-2 was downregulated by ~36% compared with the
scramble; however, this difference was not significant (P>0.05).
As demonstrated in Fig. 3,
transfection with anti-miR-29a + Taxol and Taxol alone caused 50
and 87% reduction, respectively, in expression levels of BCL-2
compared with the non-treated group; however, these differences
were not statistically significant. Thus, treatment with
anti-miR-29a + Taxol could reduce the expression of BCL-2; however,
it could not reduce the level to as low as that observed following
treatment with Taxol alone, which was significantly lower than the
expression level in the anti-miR-29a group (P<0.05).
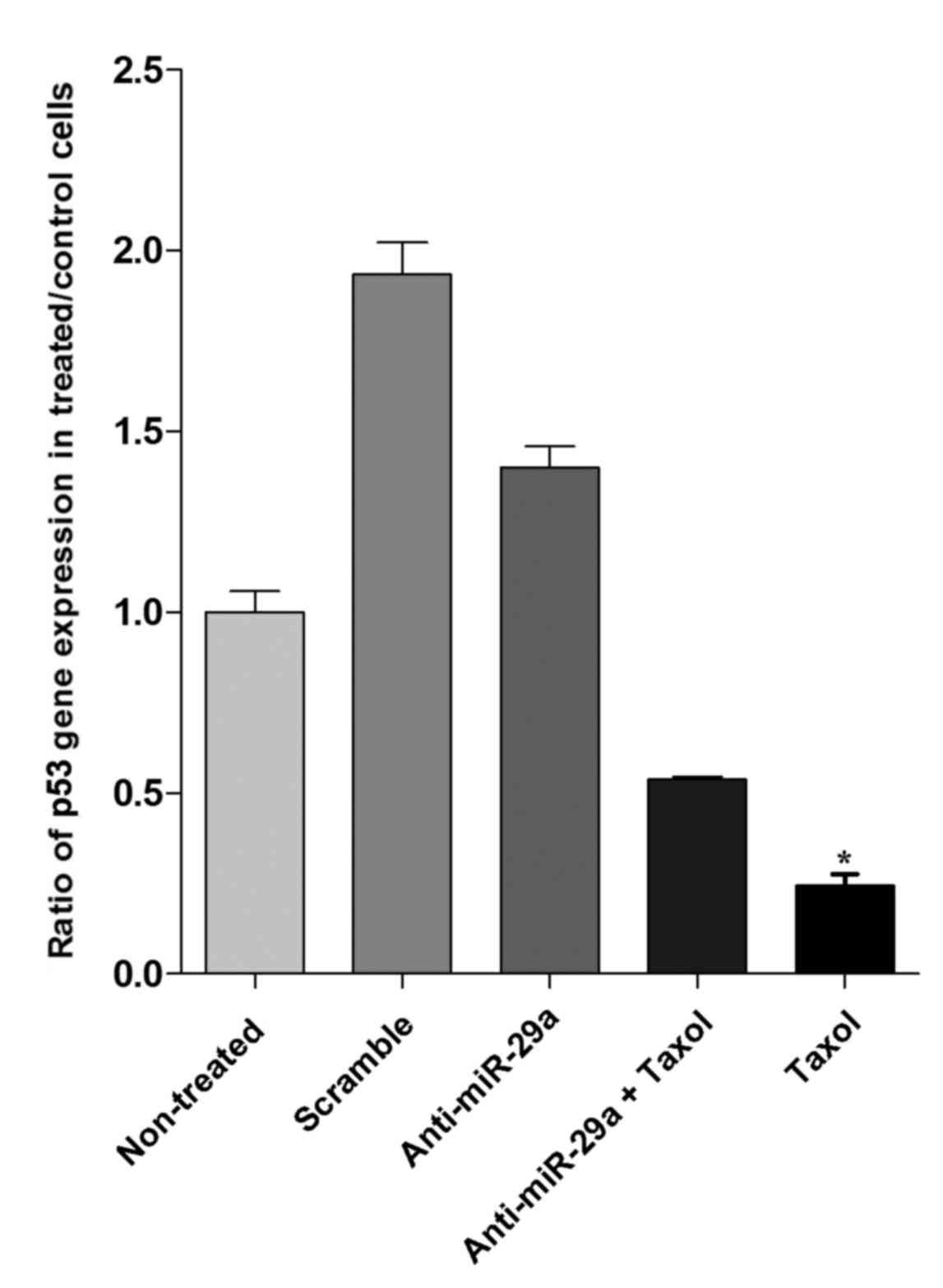
p53 gene expression increases in
anti-miR-29a-treated cells
To further understand the role of miR-29a in the
apoptosis of cancer cells, the expression of p53 as a regulatory
gene of apoptosis was analyzed by suppressing miR-29a with
anti-miR-29a. Based on RT-qPCR, expression levels of p53 increased
by ~40% in cells transfected with anti-miR-29a and scramble
treatment induced a ~1.9-fold increase in p53 gene expression
compared with the non-treated control group. Treatment with
anti-miR-29a + Taxol and Taxol alone caused a reduction of 46 and
70%, respectively, in p53 expression compared with the non-treated
group (Fig. 4). These results also
indicated that p53 gene expression in the anti-miR-29a-treated
group was significantly upregulated (5.8-fold higher) compared with
the Taxol-treated group (P<0.05).
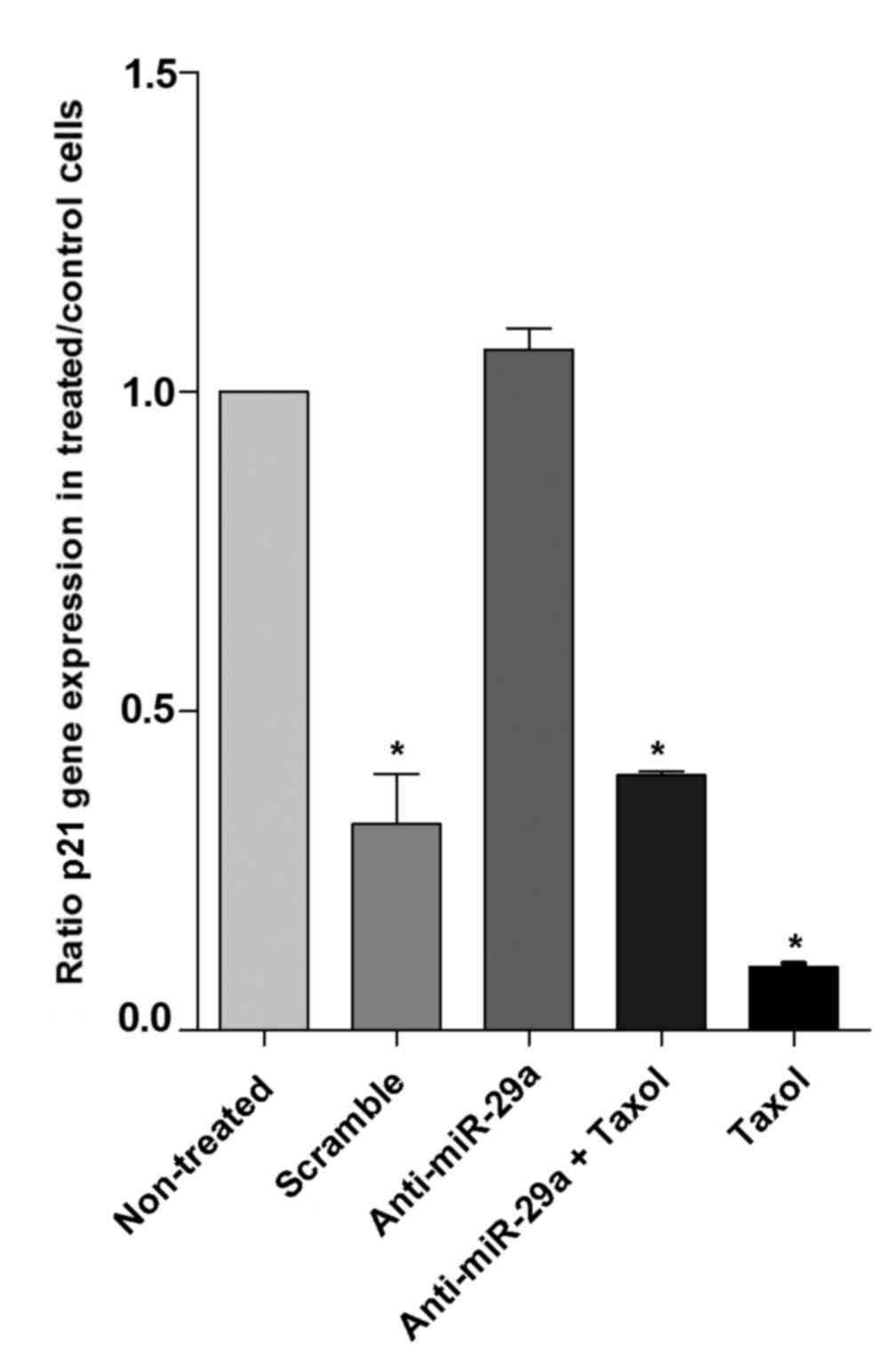
Inhibition of miR-29a has no effect on
the expression level of p21
The role of miR-29a in apoptosis of cancer cells was
explored through investigating the gene expression of p21 in MCF-7
cells. There were no significant differences between p21 gene
expression levels in cells transfected with anti-miR-29a and the
non-treated controls. As demonstrated in Fig. 5, transfection with scramble,
anti-miR-29a + Taxol and Taxol alone resulted in significant
reductions of 80, 60 and 90%, respectively, in gene expression of
p21 compared with cells transfected with anti-miR-29a (P<0.05).
The expression of p21 in Taxol-treated cells decreased by 10.5-fold
compared with cells treated with anti-miR-29a (P<0.05).
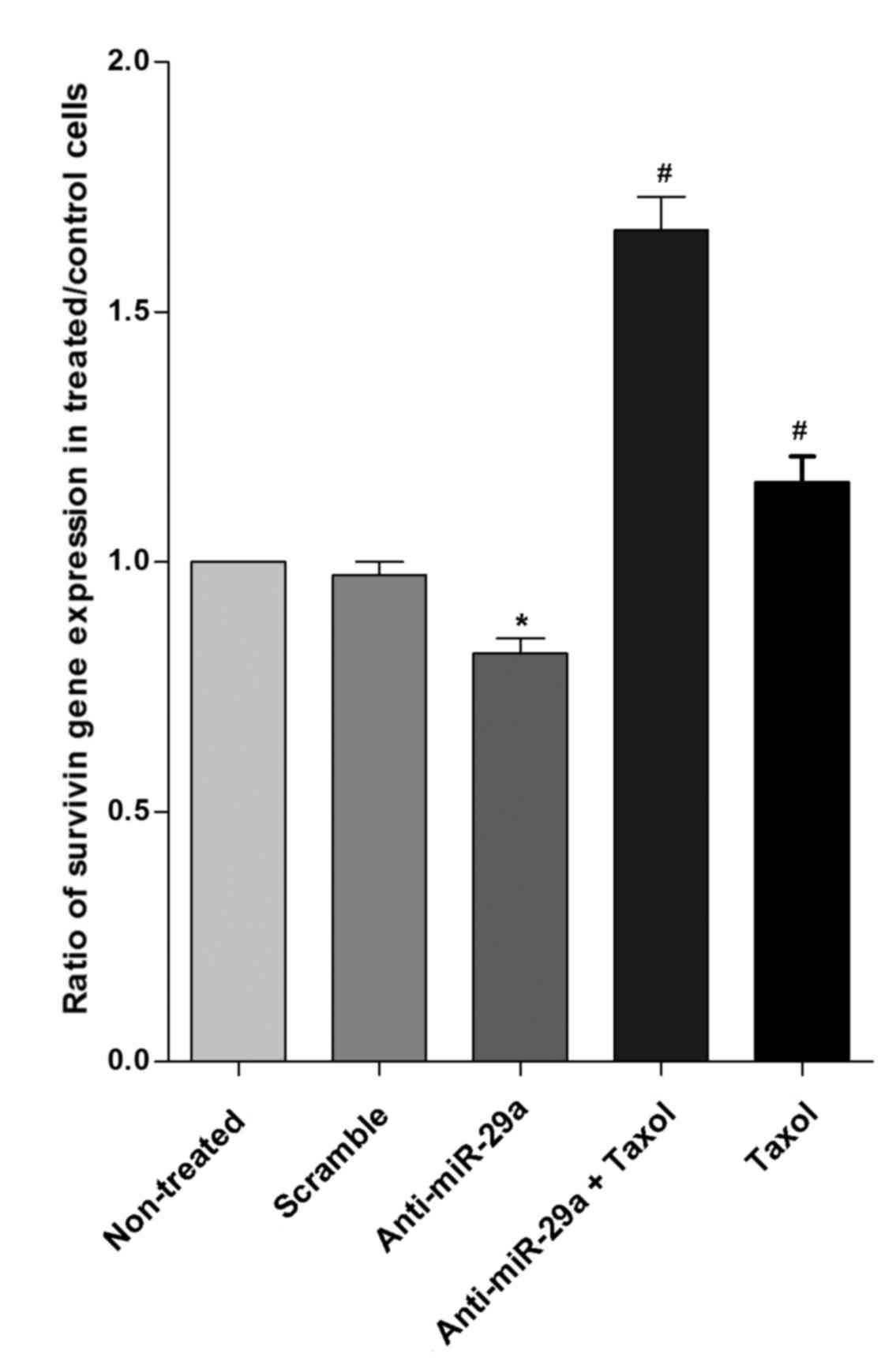
Survivin gene expression is reduced in
cells transfected with anti-miR-29a
Transfection of MCF-7 cells with anti-miR-29a caused
reduction of survivin expression by 17% compared with the
non-treated control group. However, there was no significant
difference in expression of survivin in cells transfected with
scramble and the non-treatment group. The present results
demonstrated that transfection with anti-miR-29a significantly
reduced the expression of survivin 1.43-fold lower than the
Taxol-treated group (P<0.05). As indicated in Fig. 6, survivin gene expression increased
significantly, by 1.7- and 1.2-fold, in cells transfected with
anti-miR29a + Taxol and Taxol alone compared with the scramble,
respectively (P<0.05).
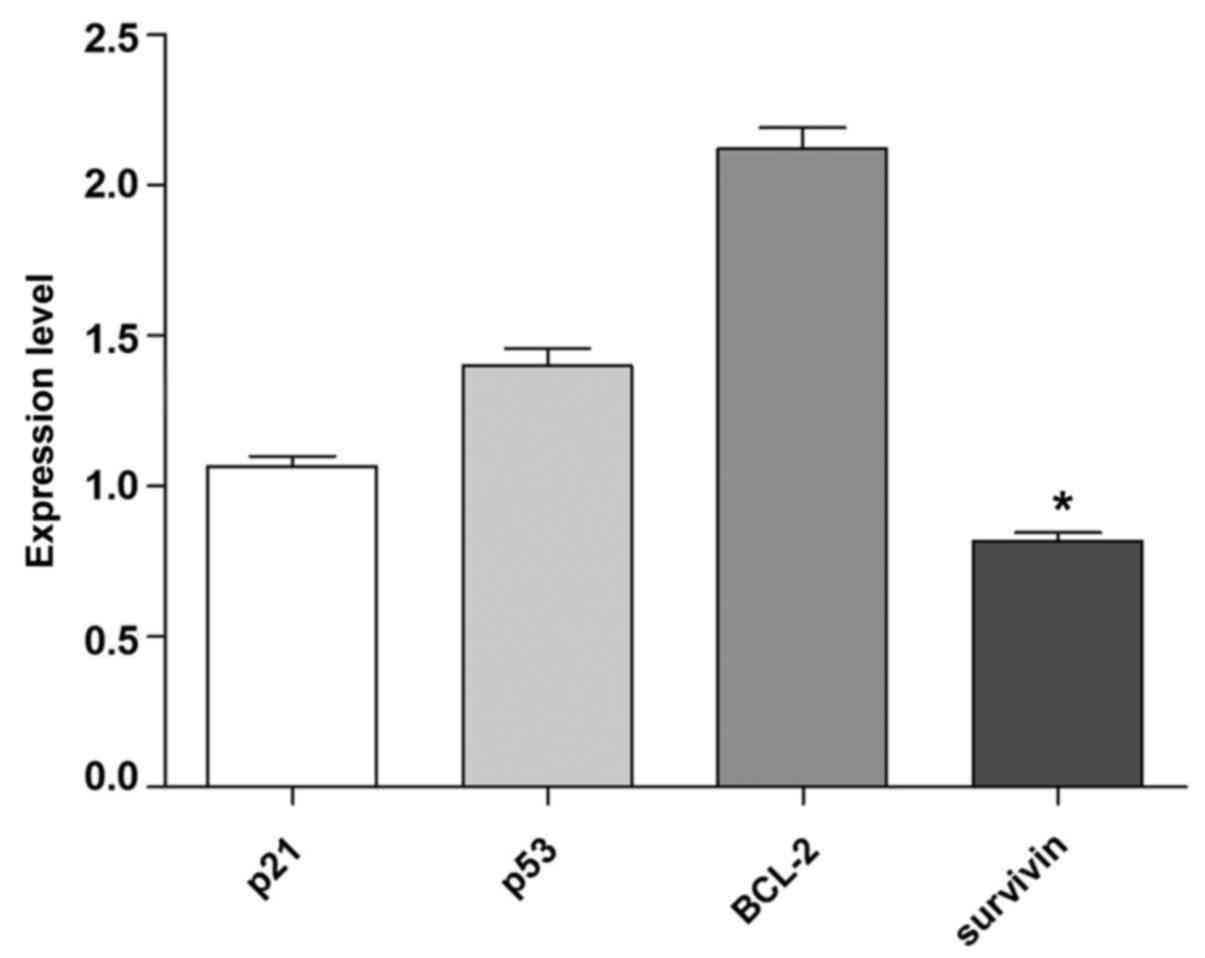
Expression of target genes following
transfection with anti-miR-29a
The present experiment demonstrated that inhibition
of miR-29a could alter the expression profiles of target genes. As
indicated in Fig. 7, following
transfection with anti-miR-29a, the expression of BCL-2 increased
2.6-fold higher and 2-fold higher than survivin and p21,
respectively. The difference between the expression levels of BCL-2
and survivin were significant (P<0.05). Also, the expression of
p21 and survivin decreased 1.3- and 1.7-fold lower than p53
expression, respectively; however, these differences were not
significant (P>0.05). Thus, transfection with anti-miR-29a
resulted in increases of p53 and BCL-2 gene expression compared to
p21 and survivin.
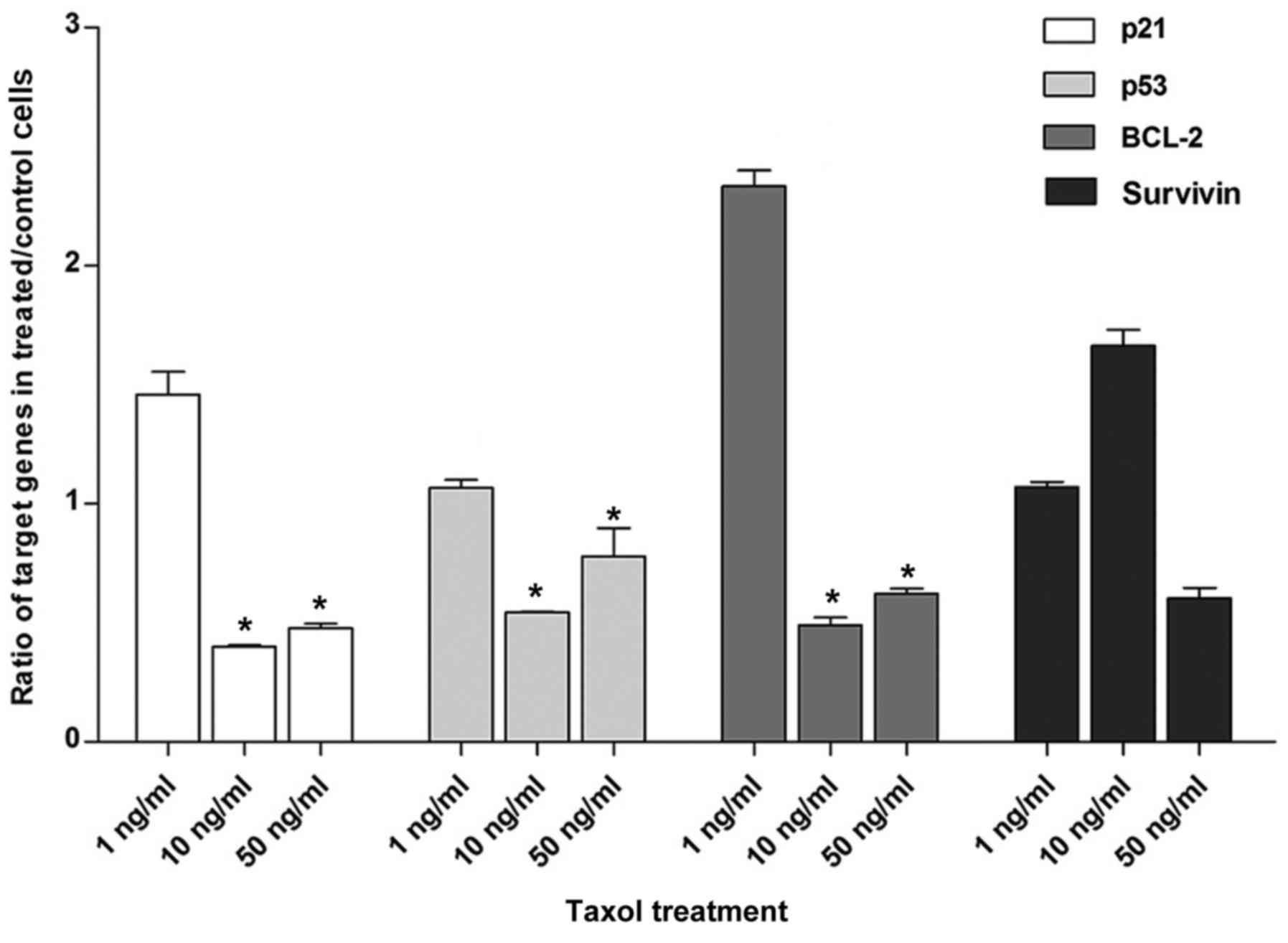
Anti-miR-29a transfection combined
with treatment with various concentrations of Taxol results in
different expression of target genes
To further analyze the expression of target genes
(p21, p53, BCL-2 and survivin), in anti-miR-29a + Taxol-treated
cells, it was hypothesized that various concentrations of Taxol
would have different effects on gene expression. Therefore,
transfection with anti-miR-29a was conducted, along with treatment
with 1, 10 or 50 ng/ml Taxol. Results demonstrated that treatment
with 1 ng/ml Taxol and anti-miR-29a transfection resulted in
increased expression of target genes p21, p53 and BCL-2, compared
with the other Taxol concentrations. As demonstrated in Fig. 8, cells treated with anti-miR-29a +
Taxol 1 ng/ml showed a significant 3.6-, 1.9- and 4.7-fold increase
in p21, p53 and BCL-2 expression, respectively, compared with cells
treated with anti-miR-29a + Taxol 10 ng/ml (P<0.05). p21, p53
and BCL-2 levels were also significantly lower in the anti-miR-29a
+ Taxol 50 ng/ml groups compared with the anti-miR-29a + Taxol 1
ng/ml (P<0.05). Also, in cells treated with anti-miR-29a + Taxol
1 ng/ml, the expression of survivin was reduced by 1.5-fold
compared with the expression levels in cells treated with
anti-miR-29a + Taxol 10 ng/ml; however, this difference was not
significant.
Discussion
Cellular expression of p21, p53, BCL-2 and survivin,
which are involved in apoptosis and proliferation, is tightly
regulated by miR-29a (32). High
expression of miR-29a in the MCF-7 human adenocarcinoma breast
cancer cell line and sera of patients with breast cancer disease
has been reported (33). As
described earlier, miR are key regulators in expression of genes
involved in the control of tumor development, proliferation,
apoptosis and the stress response (34). Aberrant expression of miR in various
types of cancer have been demonstrated; however, the molecular
mechanisms by which miR modulate the process of tumor genesis and
the behavior of cancer cells remains controversial (35). In present study, it was demonstrated
that the inhibition of miR-29a could efficiently alter the
expression profile of genes that are regulators of apoptosis in
MCF-7 cells. First, miR-29a expression was successfully
downregulated using anti-miR-29a. The viability of cells treated
with Taxol and anti-miR-29a simultaneously increased compared with
treatment of Taxol alone, suggesting the interference role of
anti-miR-29a in cancer cell apoptosis. Results indicated that
miR-29a expression was reduced by up to 75% in cells transfected
with anti-miR-29a compared with the non-treated control group.
miR-29 family members have been identified as
regulators of p53, leading to upregulation of p53 and apoptosis
induction (32). The effect of
miR-29a on breast cancer cell growth is largely dependent on
upregulation of p53, and the present study shed new insight on the
regulation of breast cancer cell apoptosis by miR-29a. The
expression of p53 as a tumor suppressor gene was upregulated by 50%
in absence of miR-29a, although Taxol treatment caused a 70%
reduction in p53 expression compared with non-treated controls.
This undesirable reduction in p53 was also observed following
treatment with anti-miR-29a + Taxol, with a 46% reduction compared
with that seen in cells transfected with the scramble control. p53
expression in anti-miR-29a-treated cells was 5.8-fold higher
compared with the Taxol-treated group. Therefore, inhibition of
miR-29a alone could increase p53 expression and result in cancer
cell cycle arrest and apoptosis.
Of the four genes examined in the present study, p21
was the only gene for which expression was not altered following
anti-miR-29a transfection compared with the non-treated control.
p21 is a downstream target of p53 and has a role in apoptosis
inhibition, which reflects the unwanted performance of this
molecule in the cancer process (36). Research has indicated that p21
activity may be regulated by multiple signal transduction pathways,
resulting in a stimulatory or inhibitory role of p21 in tumor
genesis depending on cellular status (37). The present study demonstrated that
treatment of miR-29a + Taxol reduced the p21 expression levels by
up to 60% compared with cells transfected with anti-miR-29a. This
reduction was 90% in treatment with Taxol alone. The negative
effect of Taxol on the expression of p21 was therefore moderated by
the use of anti-miR-29a and Taxol simultaneously. The expression of
p21 in the Taxol-treated group was reduced by 10.5-fold compared
with the anti-miR-29a group. Regarding the dual role of p21 in
tumor genesis, using Taxol to reduce p21 expression and resulting
in apoptosis of tumor cells appears to be useful; however, on the
other hand, this could help tumor progression.
Furthermore, the present study characterized BCL-2
as a functional target of miR-29a. BCL-2 acts as an apoptosis
regulator and it has been reported that BCL-2 expression is
associated with a good prognosis in patients with breast cancer
(38). It has been reported that in
breast cancer cells, silencing miR-15a and miR-16 restore BCL-2
expression (39). Also, due to the
activity of miR-15a and miR-16, BCL-2 downregulation results in
apoptosis (18). Accordingly, the
present results demonstrated that silencing miR-29a increased BCL-2
expression 2-fold higher than the control. Although, in Taxol and
anti-miR-29a + Taxol-treated groups, BCL-2 decreased by 87 and 50%,
respectively, compared with the non-treated control. It may be
concluded that silencing miR-29a could not exert a beneficial
effect against tumor genesis through BCL-2. On the other hand,
Taxol treatment through reduction of BCL-2 would have a positive
effect on apoptosis induction.
Survivin, a multi-functional protein, has an
important role in cell cycle regulation, inhibition of apoptosis
and stimulation the angiogenesis (40). Survivin expression is upregulated in
malignancies, which is associated with upregulation of human
epidermal growth factor receptor 2 and vascular endothelial growth
factor resulting in angiogenesis (41). It has been demonstrated that survivin
leads to resistance to chemotherapy (42). Therefore, inhibition of this molecule
could be an important strategy to overcome resistance to
chemotherapy and to remove the inhibitory effect of this molecule
on apoptosis. It has been reported that survivin expression could
be inhibited using deguelin, which results in restoration of the
sensitivity of tumor cells to chemotherapy (43). The results of the present study
demonstrated that inhibition of miR-29a resulted in the
downregulation of survivin by up to 17% compared with the
non-treated group. Expression of survivin as a unique inhibitor of
apoptosis was upregulated in cells transfected with Taxol alone and
in combination with anti-miR-29a compared with cells transfected
with anti-miR-29a alone. These findings demonstrated that reduction
of survivin expression was achieved through inhibition of miR-29a.
This may have a positive impact on apoptosis induction. However,
Taxol had negative effects in regards to the levels of
survivin.
The anticancer agent, Taxol, is used in chemotherapy
regimens against breast cancer and leads to cell cycle arrest and
apoptotic cell death (44). However,
its application is limited due to adverse effects on normal cells.
In the present study, it was demonstrated that lower doses of Taxol
in combination with anti-miR-29a treatment induced stronger effects
on the expression of target genes. In cells transfected with
anti-miR-29a + Taxol 1 ng/ml, the expression profile of p21, p53
and BCL-2 was upregulated significantly compared with anti-miR-29a
treatment combined with 10 or 50 ng/ml Taxol. Therefore,
anti-miR-29a transfection attracts much attention as an anticancer
treatment in the manner of boosting the effect of Taxol.
In conclusion, regarding the important role of
miR-29a inhibitors in regulating the apoptosis of breast cancer
cells that express a high level of miR-29a, they may be candidates
for chemotherapeutic agents. Unlike the adverse effects of Taxol on
normal cells and its infilltration throughout the body, as it is
not a specific agent, anti-miR-29a may be applied locally in the
tumor site. Due to the low stability of miR, the time duration of
their application should be considered important. Furthermore,
improving the stability of these molecules would be an important
research area. However, further investingation in cells and animal
models is required to verify the results of the present study.
Acknowledgements
We are grateful to the Research Deputy at Bushehr
University of Medical Sciences (Bushehr, Iran) for financially
supporting the present study (grant no. 2307-2013.07.16). We wish
to thank Bonyakhteh Research Center (Tehran, Iran).
References
|
1
|
Montazeri A, Vahdaninia M, Harirchi I,
Harirchi AM, Sajadian A, Khaleghi F, Ebrahimi M and Haghighat S:
Breast cancer in Iran: Need for greater women awareness of warning
signs and effective screening methods. Asia Pac Fam Med. 7:62008.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ferlay J: Cancer IAfRo: GLOBOCAN 2000:
Cancer incidence, mortality and prevalence worldwide. IARC press;
2001
|
|
3
|
Parkin DM, Bray F, Ferlay J and Pisani P:
Global cancer statistics, 2002. CA Cancer J Clin. 55:74–108. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Stewart B and Kleihues P: World cancer
report. Geneva: International Agency for Research on Cancer. World
Health Organization; 2003
|
|
5
|
Khatib OM and Modjtabai A: Guidelines for
the early detection and screening of breast cancer: World Health
Organization. Regional Office for the Eastern Mediterranean.
2006.
|
|
6
|
Network NCC: NCCN clinical practice
guidelines in oncology™ breast cancer: National Comprehensive
Cancer Network. 2007.
|
|
7
|
André F and Zielinski CC: Optimal
strategies for the treatment of metastatic triple-negative breast
cancer with currently approved agents. Ann Oncol. 23 suppl
6:vi46–vi51. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bauer KM and Hummon AB: Effects of the
miR-143/-145 microRNA cluster on the colon cancer proteome and
transcriptome. J Proteome Res. 11:4744–4754. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bartel DP: MicroRNAs: Target recognition
and regulatory functions. Cell. 136:215–233. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen K and Rajewsky N: The evolution of
gene regulation by transcription factors and microRNAs. Nat Rev
Genet. 8:93–103. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lu J, Getz G, Miska EA, Alvarez-Saavedra
E, Lamb J, Peck D, Sweet-Cordero A, Ebert BL, Mak RH, Ferrando AA,
et al: MicroRNA expression profiles classify human cancers. nature.
435:834–838. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chang TC, Yu D, Lee YS, Wentzel EA, Arking
DE, West KM, Dang CV, Thomas-Tikhonenko A and Mendell JT:
Widespread microRNA repression by Myc contributes to tumorigenesis.
Nat Genet. 40:43–50. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wu Z, Huang X, Huang X, Zou Q and Guo Y:
The inhibitory role of Mir-29 in growth of breast cancer cells. J
Exp Clin Cancer Res. 32:982013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mott JL, Kobayashi S, Bronk SF and Gores
GJ: mir-29 regulates Mcl-1 protein expression and apoptosis.
Oncogene. 26:6133–6140. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jiang H, Zhang G, Wu JH and Jiang CP:
Diverse roles of miR-29 in cancer (review). Oncol Rep.
31:1509–1516. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Letai A: Pharmacological manipulation of
Bcl-2 family members to control cell death. J Clin Invest.
115:2648–2655. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Plati J, Bucur O and Khosravi-Far R:
Dysregulation of apoptotic signaling in cancer: Molecular
mechanisms and therapeutic opportunities. J Cell Biochem.
104:1124–1149. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cimmino A, Calin GA, Fabbri M, Iorio MV,
Ferracin M, Shimizu M, Wojcik SE, Aqeilan RI, Zupo S, Dono M, et
al: miR-15 and miR-16 induce apoptosis by targeting BCL2. Proc Natl
Acad Sci USA. 102:pp. 13944–13949. 2005; View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bommer GT, Gerin I, Feng Y, Kaczorowski
AJ, Kuick R, Love RE, Zhai Y, Giordano TJ, Qin ZS, Moore BB, et al:
p53-mediated activation of miRNA34 candidate tumor-suppressor
genes. Curr Biol. 17:1298–1307. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Brown CJ, Lain S, Verma CS, Fersht AR and
Lane DP: Awakening guardian angels: Drugging the p53 pathway. Nat
Rev Cancer. 9:862–873. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Junttila MR and Evan GI: p53-a Jack of all
trades but master of none. Nat Rev Cancer. 9:821–829. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Le MT, Teh C, Shyh-Chang N, Xie H, Zhou B,
Korzh V, Lodish HF and Lim B: MicroRNA-125b is a novel negative
regulator of p53. Genes Dev. 23:862–876. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xia M, Knezevic D and Vassilev LT: p21
does not protect cancer cells from apoptosis induced by
nongenotoxic p53 activation. Oncogene. 30:346–355. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Altieri DC: Survivin in apoptosis control
and cell cycle regulation in cancer. Prog Cell Cycle Res.
5:447–452. 2003.PubMed/NCBI
|
|
25
|
Mita AC, Mita MM, Nawrocki ST and Giles
FJ: Survivin: Key regulator of mitosis and apoptosis and novel
target for cancer therapeutics. Clin Cancer Res. 14:5000–5005.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hu Y, Xu K and Yagüe E: miR-218 targets
survivin and regulates resistance to chemotherapeutics in breast
cancer. Breast Cancer Res Treat. 151:269–280. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Altieri DC: Survivin, cancer networks and
pathway-directed drug discovery. Nat Rev Cancer. 8:61–70. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kanwar JR, Kamalapuram SK and Kanwar RK:
Survivin signaling in clinical oncology: A multifaceted dragon. Med
Res Rev. 33:765–789. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Choghaei E, Khamisipour G, Falahati M,
Naeimi B, Mossahebi-Mohammadi M, Tahmasebi R, Hasanpour M, Shamsian
S and Hashemi ZS: Knockdown of microRNA-29a changes the expression
of heat shock proteins in breast carcinoma MCF-7 cells. Oncol Res.
23:69–78. 2016. View Article : Google Scholar
|
|
30
|
Jolla L: Quantification strategies in
real-time PCR Michael W. Pfaffl. 1–112. 2004.
|
|
31
|
Yip KW and Reed JC: Bcl-2 family proteins
and cancer. Oncogene. 27:6398–6406. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Park SY, Lee JH, Ha M, Nam JW and Kim VN:
miR-29 miRNAs activate p53 by targeting p85 alpha and CDC42. Nat
Struct Mol Biol. 16:23–29. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Michael MZ, O'Connor SM, van Holst
Pellekaan NG, Young GP and James RJ: Reduced accumulation of
specific MicroRNAs in colorectal neoplasia11note: Susan M. O'Connor
and Nicholas G. van holst pellekaan contributed equally to this
work. Mol Cancer Res. 1:882–891. 2003.PubMed/NCBI
|
|
34
|
Ha TY: MicroRNAs in human diseases: From
cancer to cardiovascular disease. Immune Netw. 11:135–454. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Iorio MV and Croce CM: MicroRNA
dysregulation in cancer: Diagnostics, monitoring and therapeutics.
A comprehensive review. EMBO Mol Med. 4:143–159. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Abbas T and Dutta A: p21 in cancer:
Intricate networks and multiple activities. Nat Rev Cancer.
9:400–414. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
37
|
Warfel NA and El-Deiry WS: p21WAF1 and
tumourigenesis: 20 years after. Curr Opin Oncol. 25:52–58. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Redondo M: Bcl-2, an antiapoptotic gene
indicator of good prognosis in breast cancer: The paradox. J
Carcinogene Mutagene. 4:135–139. 2013. View Article : Google Scholar
|
|
39
|
Yang J, Cao Y, Sun J and Zhang Y: Curcumin
reduces the expression of Bcl-2 by upregulating miR-15a and miR-16
in MCF-7 cells. Med Oncol. 27:1114–1118. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Schmidt SM, Schag K, Müller MR, Weck MM,
Appel S, Kanz L, Grünebach F and Brossart P: Survivin is a shared
tumor-associated antigen expressed in a broad variety of
malignancies and recognized by specific cytotoxic T cells. Blood.
102:571–576. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Asanuma H, Torigoe T, Kamiguchi K,
Hirohashi Y, Ohmura T, Hirata K, Sato M and Sato N: Survivin
expression is regulated by coexpression of human epidermal growth
factor receptor 2 and epidermal growth factor receptor via
phosphatidylinositol 3-kinase/AKT signaling pathway in breast
cancer cells. Cancer Res. 65:11018–11025. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Jha K, Shukla M and Pandey M: Survivin
expression and targeting in breast cancer. Surg Oncol. 21:125–131.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Ghanbari P, Mohseni M, Tabasinezhad M,
Yousefi B, Saei AA, Sharifi S, Rashidi MR and Samadi N: Inhibition
of survivin restores the sensitivity of breast cancer cells to
docetaxel and vinblastine. Appl Biochem Biotechnol. 174:667–681.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ofir R, Seidman R, Rabinski T, Krup M,
Yavelsky V, Weinstein Y and Wolfson M: Taxol-induced apoptosis in
human SKOV3 ovarian and MCF7 breast carcinoma cells is caspase-3
and caspase-9 independent. Cell Death Differ. 9:636–642. 2002.
View Article : Google Scholar : PubMed/NCBI
|