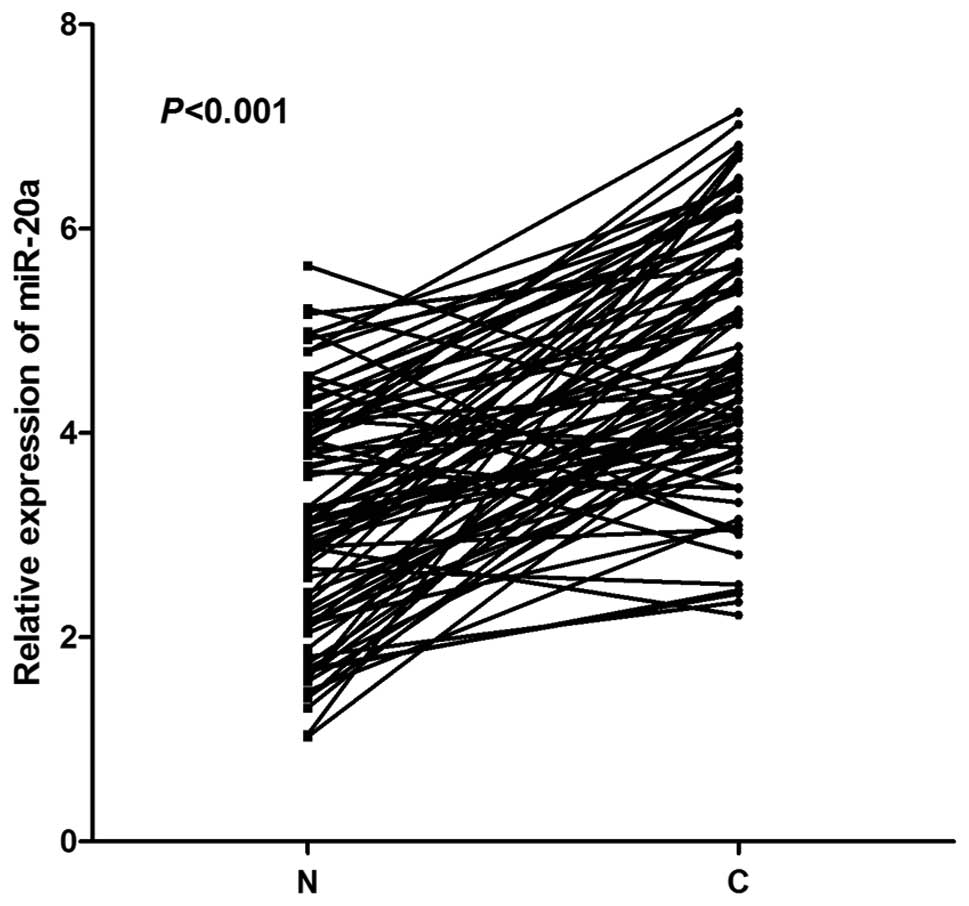
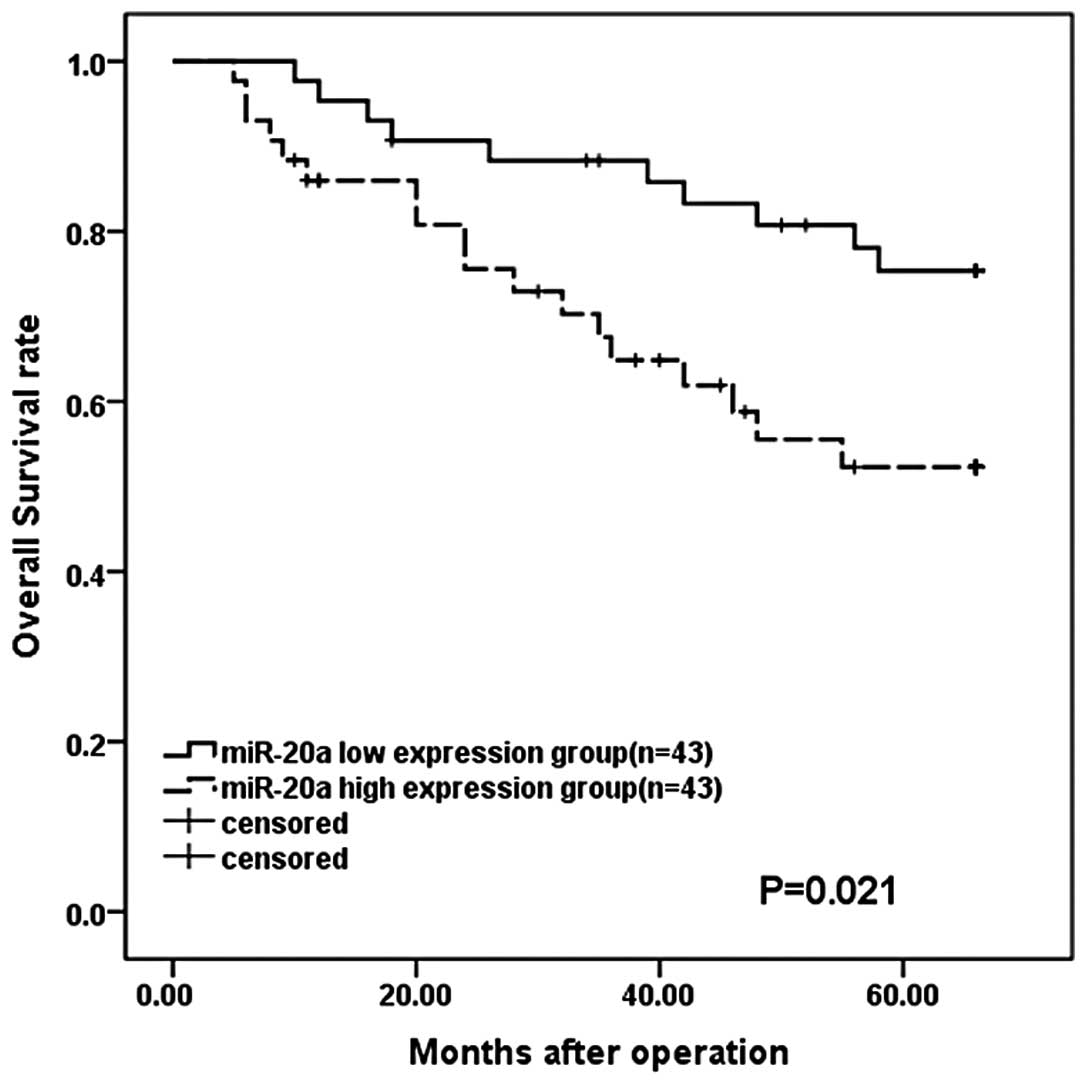
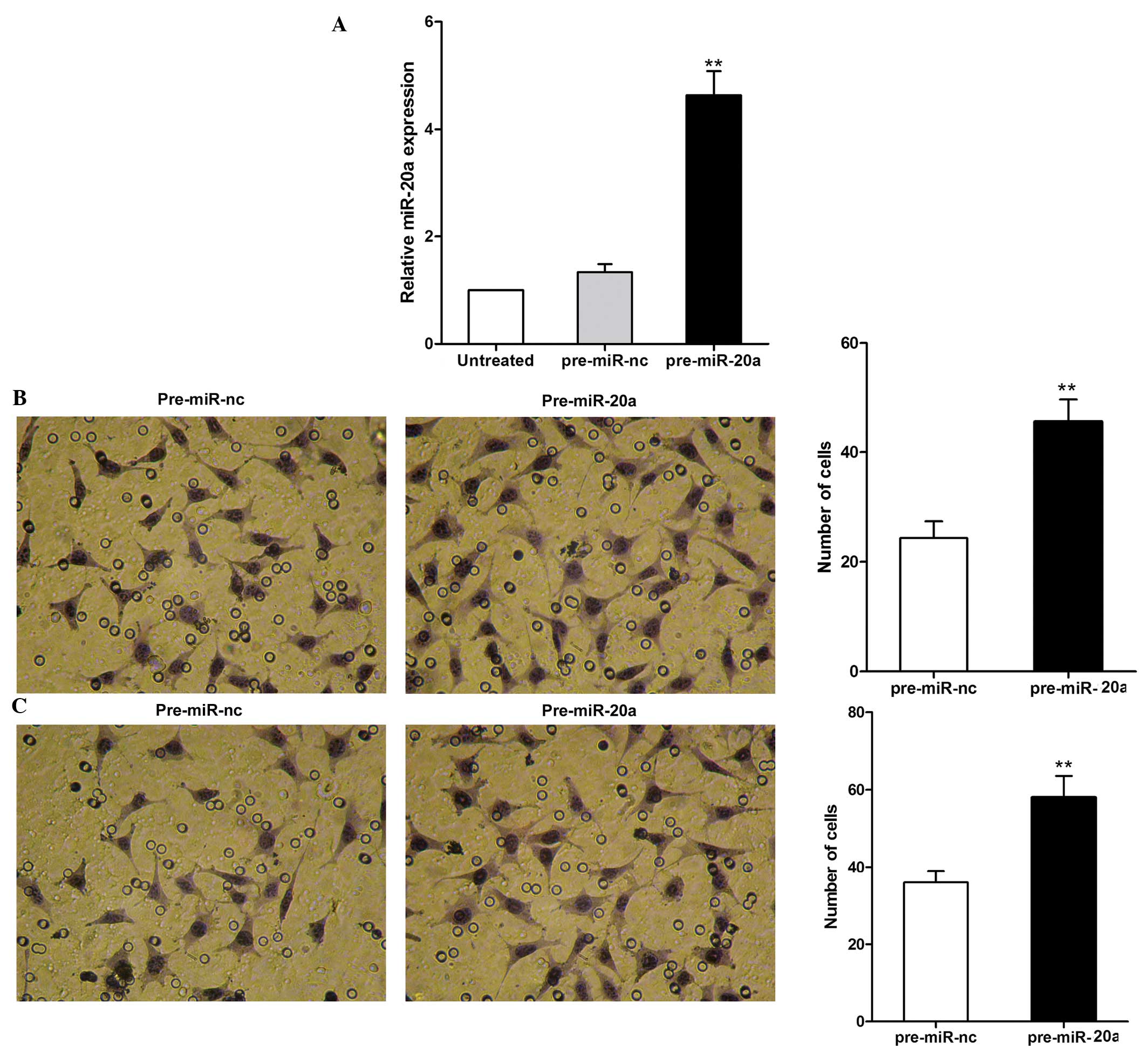
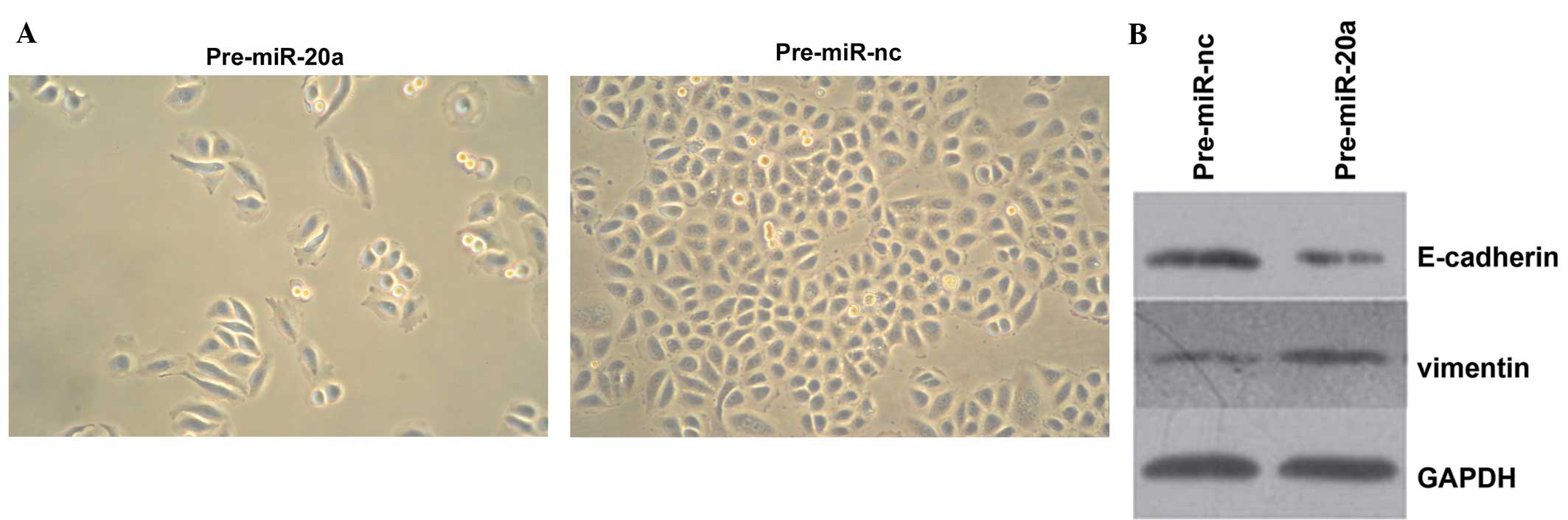
|
1
|
Tenesa A and Dunlop MG: New insights into
the aetiology of colorectal cancer from genome-wide association
studies. Nat Rev Genet. 10:353–358. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yang IP, Tsai HL, Hou MF, et al:
MicroRNA-93 inhibits tumor growth and early relapse of human
colorectal cancer by affecting genes involved in the cell cycle.
Carcinogenesis. 33:1522–1530. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kalluri R and Weinberg RA: The basics of
epithelial-mesenchymal transition. J Clin Invest. 119:1420–1428.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Thiery JP and Sleeman JP: Complex networks
orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell
Biol. 7:131–142. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Voulgari A and Pintzas A:
Epithelial-mesenchymal transition in cancer metastasis: mechanisms,
markers and strategies to overcome drug resistance in the clinic.
Biochim Biophys Acta. 1796:75–90. 2009.PubMed/NCBI
|
|
6
|
Du C, Zhang C, Hassan S, et al: Protein
kinase D1 suppresses epithelial-to-mesenchymal transition through
phosphorylation of snail. Cancer Res. 70:7810–7819. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Spaderna S, Schmalhofer O, Hlubek F, et
al: A transient, EMT-linked loss of basement membranes indicates
metastasis and poor survival in colorectal cancer.
Gastroenterology. 131:830–840. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yao D, Dai C and Peng S: Mechanism of the
mesenchymal-epithelial transition and its relationship with
metastatic tumor formation. Mol Cancer Res. 9:1608–1620. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lee Y, Jeon K, Lee JT, et al: MicroRNA
maturation: stepwise processing and subcellular localization. EMBO
J. 21:4663–4670. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lee Y, Ahn C, Han J, et al: The nuclear
RNase III Drosha initiates microRNA processing. Nature.
425:415–419. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lund E, Güttinger S, Calado A, et al:
Nuclear export of microRNA precursors. Science. 303:95–98. 2004.
View Article : Google Scholar
|
|
12
|
Esquela-Kerscher A and Slack FJ: Oncomirs
- microRNAs with a role in cancer. Nat Rev Cancer. 6:259–269. 2006.
View Article : Google Scholar
|
|
13
|
Li C, Nguyen HT, Zhuang Y, et al:
Comparative profiling of miRNA expression of lung adenocarcinoma
cells in two-dimensional and three-dimensional cultures. Gene.
511:143–150. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Moriyama T, Ohuchida K, Mizumoto K, et al:
MicroRNA-21 modulates biological functions of pancreatic cancer
cells including their proliferation, invasion, and chemoresistance.
Mol Cancer Ther. 8:1067–1074. 2009. View Article : Google Scholar
|
|
15
|
Bhaumik D, Scott GK, Schokrpur S, et al:
Expression of microRNA-146 suppresses NF-kappaB activity with
reduction of metastatic potential in breast cancer cells. Oncogene.
27:5643–5647. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang G, Xia S, Tian H, et al: Clinical
significance of miR-22 expression in patients with colorectal
cancer. Med Oncol. 29:3108–3812. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Paterson EL, Kazenwadel J, Bert AG, et al:
Down-regulation of the miRNA-200 family at the invasive front of
colorectal cancers with degraded basement membrane indicates EMT is
involved in cancer progression. Neoplasia. 15:180–191.
2013.PubMed/NCBI
|
|
18
|
Zhang GJ, Xiao HX, Tian HP, et al:
Upregulation of microRNA-155 promotes the migration and invasion of
colorectal cancer cells through the regulation of claudin-1
expression. Int J Mol Med. 31:1375–1380. 2013.PubMed/NCBI
|
|
19
|
Fan MQ, Huang CB, Gu Y, et al: Decrease
expression of microRNA-20a promotes cancer cell proliferation and
predicts poor survival of hepatocellular carcinoma. J Exp Clin
Cancer Res. 32:212013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chang Y, Liu C, Yang J, et al: MiR-20a
triggers metastasis of gallbladder carcinoma. J Hepatol.
59:518–527. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Fan X, Liu Y, Jiang J, et al: miR-20a
promotes proliferation and invasion by targeting APP in human
ovarian cancer cells. Acta Biochim Biophys Sin (Shanghai).
42:318–324. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Huang G, Nishimoto K, Zhou Z, et al:
miR-20a encoded by the miR-17–92 cluster increases the metastatic
potential of osteosarcoma cells by regulating Fas expression.
Cancer Res. 72:908–916. 2012.
|
|
23
|
Li JY, Zhang Y, Zhang WH, et al:
Differential distribution of miR-20a and miR-20b may underly
metastatic heterogeneity of breast cancers. Asian Pac J Cancer
Prev. 13:1901–1906. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yan H, Wu J, Liu W, et al: MicroRNA-20a
overexpression inhibited proliferation and metastasis of pancreatic
carcinoma cells. Hum Gene Ther. 21:1723–1734. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chai H, Liu M, Tian R, et al: miR-20a
targets BNIP2 and contributes chemotherapeutic resistance in
colorectal adenocarcinoma SW480 and SW620 cell lines. Acta Biochim
Biophys Sin (Shanghai). 43:217–225. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yu G, Tang JQ, Tian ML, et al: Prognostic
values of the miR-17-92 cluster and its paralogs in colon cancer. J
Surg Oncol. 106:232–237. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Motoyama K, Inoue H, Takatsuno Y, et al:
Over- and under-expressed microRNAs in human colorectal cancer. Int
J Oncol. 34:1069–1075. 2009.PubMed/NCBI
|
|
28
|
Massagué J and Chen YG: Controlling
TGF-beta signaling. Genes Dev. 14:627–644. 2000.PubMed/NCBI
|
|
29
|
Wang LH, Kim SH, Lee JH, et al:
Inactivation of SMAD4 tumor suppressor gene during gastric
carcinoma progression. Clin Cancer Res. 13:102–110. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Powell SM, Harper JC, Hamilton SR, et al:
Inactivation of Smad4 in gastric carcinomas. Cancer Res.
57:4221–4224. 1997.PubMed/NCBI
|
|
31
|
Miyaki M, Iijima T, Konishi M, et al:
Higher frequency of Smad4 gene mutation in human colorectal cancer
with distant metastasis. Oncogene. 18:3098–3103. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Maitra A, Molberg K, Albores-Saavedra J
and Lindberg G: Loss of Dpc4 expression in colonic adenocarcinomas
correlates with the presence of metastatic disease. Am J Pathol.
157:1105–1111. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Riggins GJ, Kinzler KW, Vogelstein B and
Thiagalingam S: Frequency of Smad gene mutations in human cancers.
Cancer Res. 57:2578–2580. 1997.PubMed/NCBI
|
|
34
|
Deckers M, van Dinther M, Buijs J, et al:
The tumor suppressor Smad4 is required for transforming growth
factor beta-induced epithelial to mesenchymal transition and bone
metastasis of breast cancer cells. Cancer Res. 66:2202–2209. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Valcourt U, Kowanetz M, Niimi H, et al:
TGF-beta and the Smad signaling pathway support transcriptomic
reprogramming during epithelial-mesenchymal cell transition. Mol
Biol Cell. 16:1987–2002. 2005. View Article : Google Scholar
|
|
36
|
Levy L and Hill CS: Smad4 dependency
defines two classes of transforming growth factor {beta}
(TGF-{beta}) target genes and distinguishes TGF-{beta}-induced
epithelial-mesenchymal transition from its antiproliferative and
migratory responses. Mol Cell Biol. 25:8108–8125. 2005.PubMed/NCBI
|
|
37
|
Müller N, Reinacher-Schick A, Baldus S, et
al: Smad4 induces the tumor suppressor E-cadherin and P-cadherin in
colon carcinoma cells. Oncogene. 21:6049–6058. 2002.PubMed/NCBI
|
|
38
|
Pohl M, Radacz Y, Pawlik N, et al: SMAD4
mediates mesenchymal-epithelial reversion in SW480 colon carcinoma
cells. Anticancer Res. 30:2603–2613. 2010.PubMed/NCBI
|