Introduction
Cardiac remodeling post myocardial infarction (MI)
results in ventricular dysfunction and heart failure, which
contribute to a poor outcome and high mortality rate (1,2).
Previous studies have reported that the inflammatory response and
cardiomyocyte apoptosis were the underlying mechanisms in the
development of chronic heart failure (3–5). MI
causes local inflammation and apoptosis, which can result in
cardiomyocyte damage (5).
Proinflammatory mediators are upregulated in cardiac dysfunction
and, in particular, elevated levels of inflammatory cytokines,
including tumor necrosis factor-α (TNF-α) and interleukin (IL)-1β
in the infarcted myocardium contribute to cardiomyocyte apoptosis
(2,6,7).
Cell apoptosis may also persist in infarcted areas (8,9). The
nuclear factor-κB (NF-κB) signaling pathway, involved in
inflammation, is also active in the apoptotic pathway. Several
approaches to reduce the activation of NF-κB have been demonstrated
to reduce myocardial inflammation and apoptosis (10–12).
Therefore, inhibition of inflammation and apoptosis may be an
effective therapeutic strategy for MI.
The NF-κB family is a key factor regulating the
inflammatory response by promoting the expression of
proinflammatory factors (13). A
member of the NF-κB family, p65, forms homo or heterodimers that
are bound to inhibitory (IκB) proteins in the cytosol (14). Degradation of IκB releases NF-κB
dimers and enables the translocation of NF-κB into the nucleus,
where it can initiate transcription of target genes. NF-κB
activation and subsequent augmented inflammatory responses largely
contribute to maladaptive left ventricular remodeling and cardiac
dysfunction post-MI (15).
Lycopene is a type of carotenoid, present predominantly in
tomatoes. Several epidemiological studies have confirmed that high
levels of plasma lycopene could reduce the risk of cardiovascular
disease, neurodegenerative disease and cancer (16,17).
In addition, it was demonstrated that lycopene inhibited
inflammatory cell infiltration and pathologic histological
alterations following MI induced by isoprenaline (18,19).
A previous study also reported that lycopene inhibited the
expression of interleukin (IL)-8 and the activation of NF-κB in the
lungs induced by cigarette smoke (20) and reduced the expression of
proinflammatory cytokines in adipose tissue of an animal model of
obesity (21). However, the role
and mechanism of lycopene in the inhibition of inflammation and
apoptosis in MI are yet to be elucidated.
In the present study, it was hypothesized that
lycopene inhibited NF-κB activation, which mediates the
inflammatory response and cardiomyocyte apoptosis in post-MI
remodeling. Therefore, MI mice models were constructed by ligation
of the left anterior descending coronary artery (LAD) to examine
whether lycopene can inhibit NF-κB activation and thus reduce
inflammation and apoptosis in the process of post-MI
remodeling.
Materials and methods
Materials
Primary antibodies against NF-κB, p65 (cat no. 8242)
and phospho-NF-κB p65 (cat no. 3039) were purchased from Cell
Signaling Technology, Inc. (Danvers, MA, USA). Primary antibodies
against caspase-3, (cat no. ab4051) -8 (cat no. ab25901) and -9
(cat no. ab32539) were purchased from Abcam (Cambridge, MA, USA).
Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was purchased from
Bioworld Technology (St. Louis Park, MN, USA). The bicinchoninic
acid protein assay kit was purchased from Pierce Biotechnology,
Inc. (Rockford, IL, USA). TRIzol (cat no. 15596018) was purchased
from Invitrogen Life Technologies (Carlsbad, CA, USA).
Animals and animal model
The present study was conducted in accordance with
the Guide for the Local Care and Use of Laboratory Animals and was
approved by the Ethics Committee of The Central Hospital of Wuhan
(Wuhan, China). All surgery and subsequent analyses were performed
in a blinded manner. Male BALA/c mice (18–25 g; 10–12 weeks)
purchased from the Experimental Animal Center of Hubei Province
(Wuhan, China), were randomized to undergo LAD ligation or sham
surgery. The animals were anesthetized with an intraperitoneal
injection of 1% pentobarbital sodium (30 mg/kg) and local injection
of 1 ml 1% lidocaine. Thoracotomy was performed via the third left
intercostal space. The pericardium was opened and prolene (8-0) was
used to ligate the LAD. Visible blanching and hypokinesis of the
anterior wall of the left ventricle and swelling of the left atrium
were indicative of successful ligation. Sham surgery consisted of
the same procedure, however, the suture was passed through the
myocardium beneath the LAD without ligation. Animals were divided
into several groups. Group I mice received the sham surgery. Group
II mice received LAD ligation. Group III mice received LAD ligation
and normal saline and group IV mice received LAD ligation and
lycopene (10 mg/kg/day, intragastrically for 4 weeks).
Terminal deoxynucleotidyl transferase
dUTP nick end labeling (TUNEL) staining
Hearts were perfused with 0.1 ml phosphate-buffered
saline (PBS) via the left coronary artery and then dissected and
fixed in 4% paraformaldehyde in PBS (pH 7.4) for 24 h at room
temperature. Fixed tissues were embedded in paraffin and five
sections of 6 mm thickness were cut from each tissue block. TUNEL
staining was performed according to the manufacturer’s instructions
(Roche Diagnostics, Mannheim, Germany). Total nuclei were stained
with DAPI (Vector Laboratories Inc., Burlingame, CA, USA).
Additionally, cardiac tissue was specifically labeled with the
mouse monoclonal α-actinin antibody. (ab9465, Abcam). The apoptotic
index (the number of positively stained nuclei/total number of
nuclei counted × 100%) was determined in a blinded manner.
Western blot analysis
Protein (50 μg) was extracted from myocardial tissue
and lysed in radioimmunoprecipitation assay lysis buffer (Cell
Signaling Technology, Inc.) for use in SDS-PAGE (Invitrogen Life
Technologies). The proteins were then transferred onto
nitrocellulose membranes and inhibited with 5% nonfat dry milk in
tris-buffered saline (TBS; Cell Signaling Technology, Inc.) for 90
min at room temperature. Membranes were probed with several primary
antibodies against caspase-3, -8, -9, NF-κB p65 and phospho-NF-κB
p65 overnight. The next day, it was washed using 1X TBS with
Tween-20 (TBST) and incubated for 1 h with horseradish
peroxidase-labeled polyclonal mouse anti rabbit antibody (1:2,000)
and anti-avidin antibodies (1:1,000) (Jackson Immunoresearch, West
Grove, PA, USA) in double anti-TBST fluid. The membrane was washed
three times and the film was placed in 10 ml LumiGLO solution (Cell
Signaling Technology, Inc.) for 1 min. Following development, the
images were put into an automatic image analyzer to determine the
function of proteins and the reference gray scale values.
Monoclonal GAPDH was used separately as a loading control.
Quantitative polymerase chain reaction
(qPCR) analysis
Total mRNA was extracted from the myocardial tissue
using TRIzol according to the manufacturer’s instructions and cDNA
was synthesized using oligo (dT) primers with the Transcriptor
First Strand cDNA synthesis kit (Roche Diagnostics). Selected gene
differences were confirmed by qPCR using SYBR green (Roche
Diagnostics) and results were normalized against GAPDH gene
expression. The sequences of all primers used were as follows:
Transforming growth factor-β1 (TGF-β1), forward
5′-AACAACGCCATCTATGAG-3′ and reverse 5′-TATTCCGTCTCCTTGGTT-3′;
collagen I, forward 5′-AGGCTTCAGTGGTTTGGATG-3′ and reverse
5′-CACCAACAGCACCATCGTTA-3′; collagen III, forward 5′-CCCAACCCAGAGA
TCCCATT-3′ and reverse 5′-GAAGCACAGGAGCAGGTGTAGA-3′; TNF-α, forward
5′-CATCT TCTCAAAATTCGAGTGACAA-3′ and reverse
5′-TGGGAGTAGACAAGGTACAACCC-3′; IL-1β, forward
5′-CCGTGGACCTTCCAGGATGA-3′ and reverse 5′-GGGAACGTCACA CACCAGCA-3′;
GAPDH, forward 5′-ACTCCACTCACGGCAAATTC-3′ and reverse 5′-TCTC
CATGGTGGTGAAGACA-3′.
Statistical analysis
Data are presented as the mean ± standard deviation.
Comparisons between four groups were performed using one-way
analysis of variance. P<0.05 was considered to indicate a
statistically significant difference.
Results
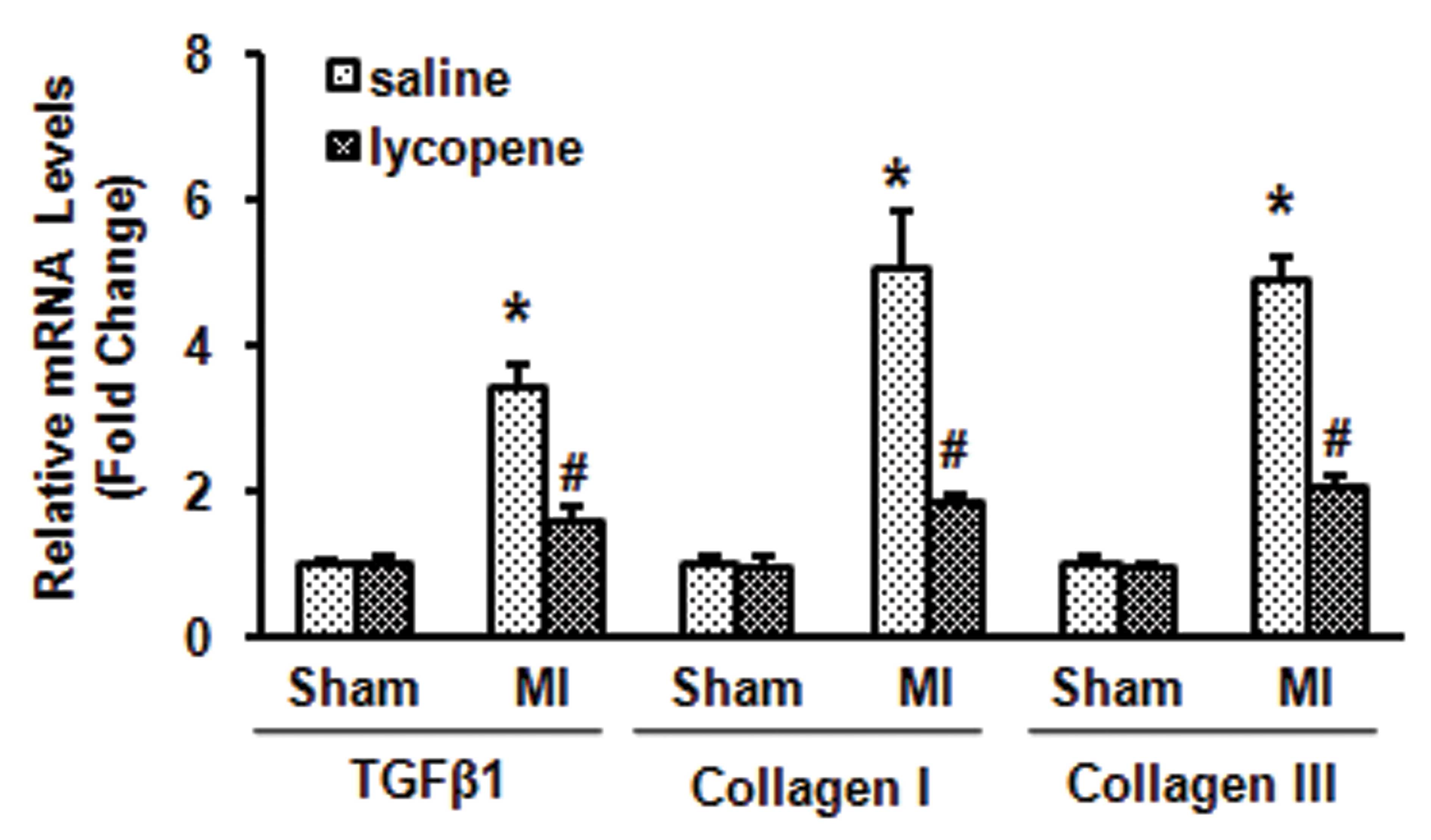
Lycopene improves cardiac remodeling in
MI mice
A model of MI was established by ligating the left
coronary artery. Cardiac remodeling was estimated by detecting the
mRNA expression of fibrosis markers, including TGF-β1, collagen I
and III. Compared with the MI group adminstered with saline,
administration of lycopene (10 mg/kg/day) reduced the mRNA
expression of TGF-β1, collagen I and III (P<0.05; Fig. 1). These results demonstrated that
lycopene protected the mice from MI.
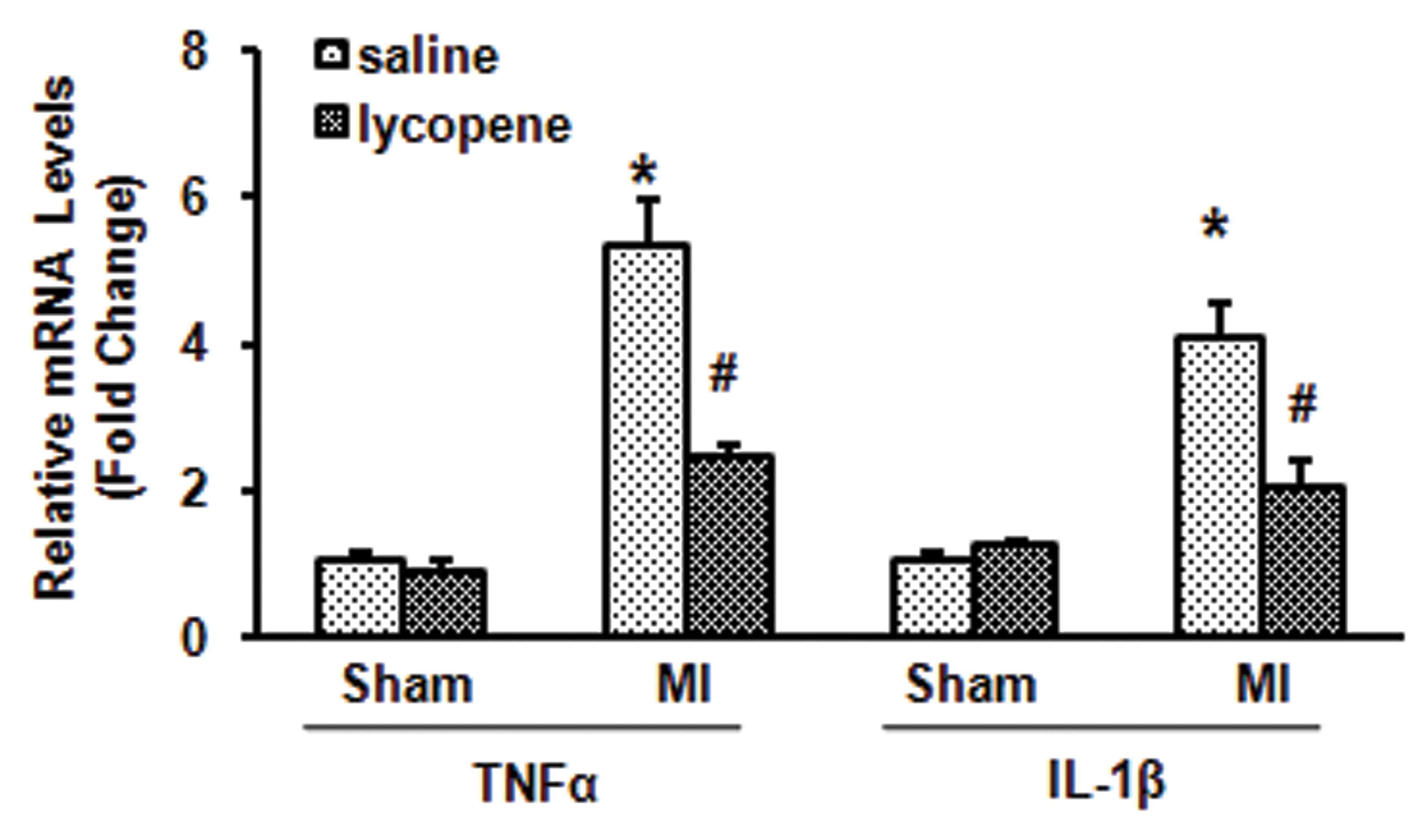
Lycopene reduces inflammatory responses
in MI mice
The inflammatory cytokines TNF-α and IL-1β were
increased in mice 4 weeks after MI induction by LAD ligation.
However, treatment with 10 mg/kg/day lycopene significantly reduced
the mRNA expression of TNF-α and IL-1β in infarcted myocardium
compared with the normal saline-treated group (P<0.05; Fig. 2). These results suggested that
lycopene exerted an anti-inflammatory effect on MI mice.
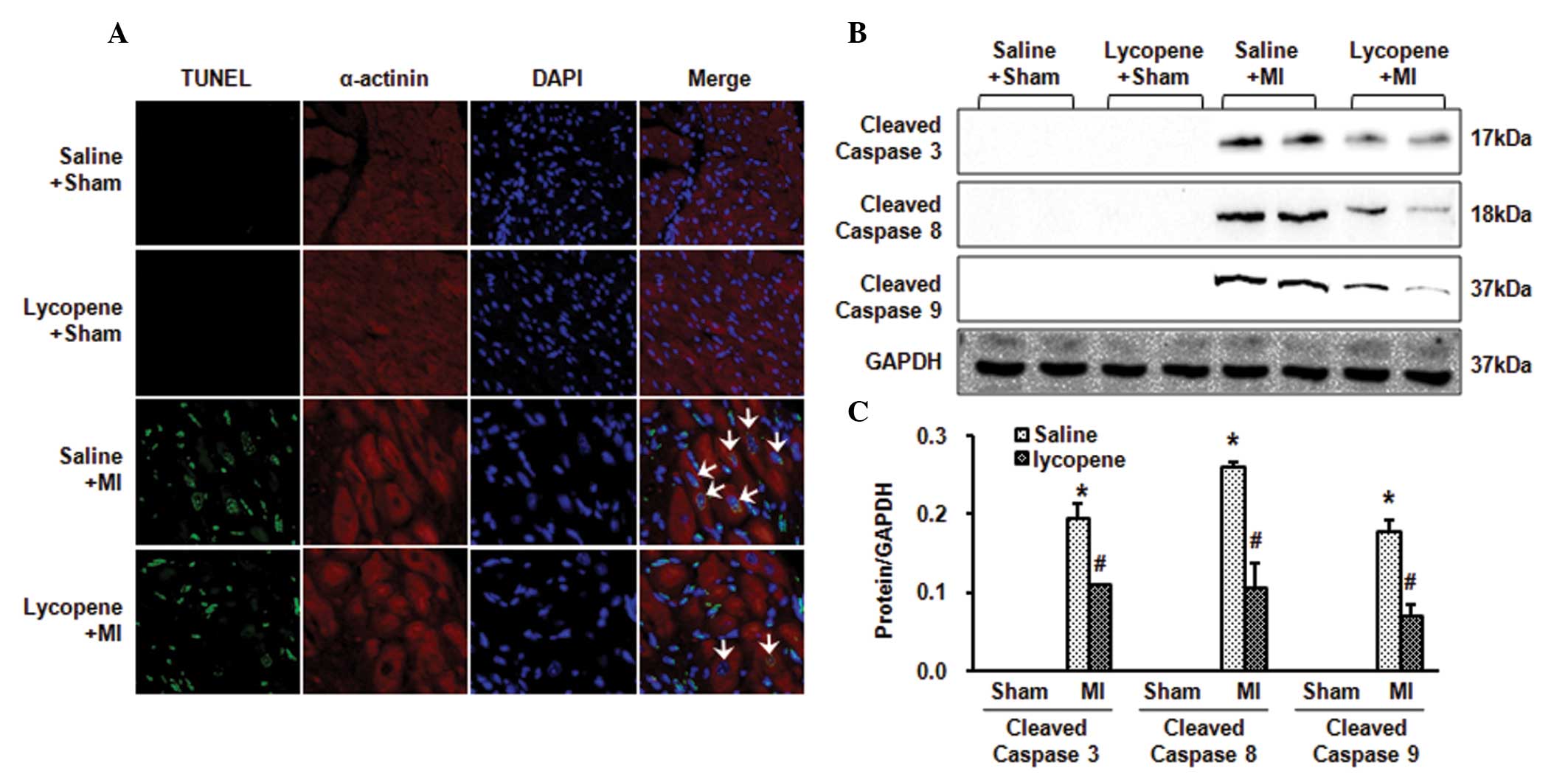
Lycopene attenuates cardiomyocyte
apoptosis in MI mice
A marked decrease in TUNEL-positive nuclear staining
was observed in the sectioned left ventricular myocardium following
treatment with lycopene (10 mg/kg/day) compared with saline at 4
weeks post-MI (P<0.05; Fig.
3A). TUNEL-positive nuclear staining was not observed in the
sham group. The level of apoptosis, assessed by detecting the
protein expression of cleaved caspase-3, -8 and -9 was
significantly higher 4 weeks post-MI compared with the sham group.
However, the expression of caspase-3, -8 and -9 were markedly
decreased following treatment with lycopene (10 mg/kg/day) compared
with saline (P<0.05; Fig. 3B and
C). These data indicated that lycopene inhibited myocardial
cell apoptosis in mice subjected to MI.
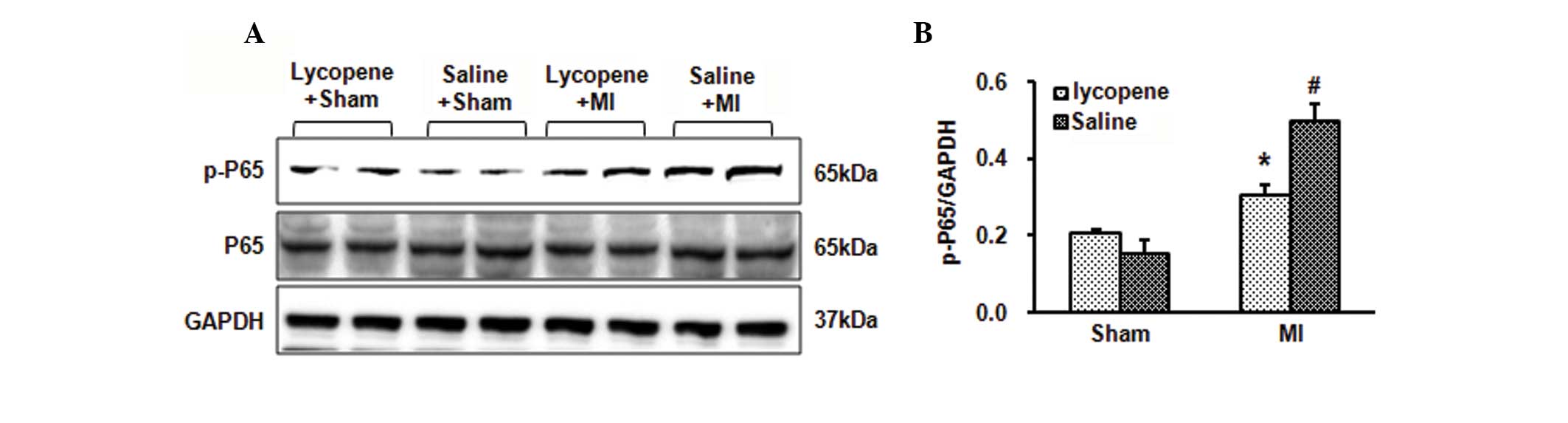
Lycopene inhibits activation of the NF-κB
signaling pathway in MI mice
In order to identify the mechanism involved in the
inhibition of the inflammatory response and cell apoptosis by
lycopene in MI mice, the activation of NF-κB in the ischemic
myocardium was detected. Western blot analysis of nuclear and
cytoplasmic extracts confirmed that 10 mg/kg/day lycopene decreased
the nuclear localization of NF-κB p65 4 weeks post-MI (Fig. 4A and B). In the ischemic
myocardium, the level of NF-κB p65 phosphorylation was markedly
induced 4 weeks post-MI. NF-κB p65 phosphorylation was reduced in
the lycopene treatment group compared with the MI group
administered with saline (Fig. 4A and
B). These results suggest that lycopene may exert an
anti-inflammatory and anti-apoptotic effect by suppressing NF-κB
activation in MI mice.
Discussion
Coronary heart disease is expected to become the
leading worldwide cause of mortality by 2020 (22). The primary pathological
manifestation of coronary artery disease is myocardial damage,
which most likely develops into cardiac remodeling. Cardiac
remodeling post-MI involves a complex set of events that
paradoxically causes an increase in tissue injury characterized by
inflammation, apoptosis, infarction and fibrosis (23–25).
MI causes inflammatory cell infiltration into the infarct zone and
increases the production of proinflammatory cytokines (26). Therefore, inhibition of
inflammatory cell infiltration could reduce cardiac damage and
attenuate the development of MI. Lycopene could be useful in
treating various inflammatory diseases (27–29).
The results of the present study indicated that lycopene offers
protection from myocardial cell injury and reduces the production
of proinflammatory cytokines, including TNF-α and IL-1β in
infarcted myocardium.
Cardiac apoptosis is a key pathological feature of
MI (30,31). Two major pathways that lead to
apoptosis have been identified, including the mitochondrial pathway
and the death-receptor pathway (32,33).
These pathways lead to the activation of caspase-3, -8 and -9, the
central mediators of the cascade (34). Cascade protein levels are increased
in myocardial samples from patients with right ventricular
dysplasia and heart failure (35,36).
In mice, cardiomyocyte apoptosis may occur transiently during the
first few days following MI in the ischemic area, whereas left
ventricular remodeling following MI is associated with apoptosis in
the myocardium remote from the infarct. Previous studies have also
demonstrated that the amplification of apoptosis was sufficient to
increase infarct size, indicating a critical role for cardiomyocyte
apoptosis in myocardial infarct expansion (37,38).
The present study demonstrated that myocardial cell apoptosis,
measured via TUNEL assay and caspase-3, -8 and -9 protein
expression levels, was inhibited by lycopene treatment, implying an
antiapoptotic role for lycopene and correlating with protection
from myocardial cell injury. Therefore, it was hypothesized that
lycopene treatment may reduce ventricular remodeling by decreasing
cardiomyocyte apoptosis in the infarct and peri-infarct
regions.
In addition, lycopene decreased the nuclear
localization of nuclear NF-κB p65 and markedly suppressed NF-κB
activation. NF-κB is a transcription factor that regulates several
inflammatory genes and is a key regulator of the inflammatory
response (39). Activation of
NF-κB induces the transcription of pro-inflammatory genes,
including TNF-α and IL-1β. It has been demonstrated that NF-κB is
activated by myocardial injury, with cardiac myocytes and
interstitial cells being important sources of NF-κB (40,41).
Proinflammatory cytokines exert their actions on target cells by
transactivating NF-κB, thus cells, including cardiomyocytes,
respond to proinflammatory cytokines by activating NF-κB. The
results of the present study suggest that the cardioprotective
effect of lycopene may be due to reduced production of
proinflammatory cytokines via inhibition of the NF-κB activity
induced by local myocardial injury. Therefore, this suggests that
lycopene may have a cardioprotective effect associated with the
inhibition of local myocardial inflammation and apoptosis.
The present study provided evidence of a novel
cardioprotective effect of lycopene in a mouse model of MI. This
protection was conferred, at least partially, by its
anti-inflammatory and antiapoptotic actions following myocardial
injury. These findings may provide a basis for a new therapeutic
application of lycopene in the prevention of myocardial cell injury
following MI.
Acknowledgements
The present study was supported by grants from the
National Natural Science Foundation of China (no. 81300214).
References
|
1
|
Sutton MG and Sharpe N: Left ventricular
remodeling after myocardial infarction: pathophysiology and
therapy. Circulation. 101:2981–2988. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Frangogiannis NG, Smith CW and Entman ML:
The inflammatory response in myocardial infarction. Cardiovasc Res.
53:31–47. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dorn GW 2nd: Apoptotic and non-apoptotic
programmed cardiomyocyte death in ventricular remodelling.
Cardiovasc Res. 1:465–473. 2009.PubMed/NCBI
|
|
4
|
Frantz S, Bauersachs J and Ertl G:
Post-infarct remodelling: contribution of wound healing and
inflammation. Cardiovasc Res. 81:474–481. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nian M, Lee P, Khaper N and Liu P:
Inflammatory cytokines and postmyocardial infarction remodeling.
Circ Res. 94:1543–1553. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Koyama M, Kelly TR and Watanabe KA: Novel
type of potential anticancer agents derived from chrysophanol and
emodin. Some structure-activity relationship studies. J Med Chem.
31:283–284. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sun M, Dawood F, Wen WH, Chen M, Dixon I,
Kirshenbaum LA and Liu PP: Excessive tumor necrosis factor
activation after infarction contributes to susceptibility of
myocardial rupture and left ventricular dysfunction. Circulation.
110:3221–3228. 2004.
|
|
8
|
Baldi A, Abbate A, Bussani R, Patti G,
Melfi R, Angelini A, Dobrina A, Rossiello R, Silvestri F, Baldi F
and Di Sciascio G: Apoptosis and post-infarction left ventricular
remodeling. J Mol Cell Cardiol. 34:165–174. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Palojoki E, Saraste A, Eriksson A, Pulkki
K, Kallajoki M, Voipio-Pulkki LM and Tikkanen I: Cardiomyocyte
apoptosis and ventricular remodeling after myocardial infarction in
rats. Am J Physiol Heart Circ Physiol. 280:H2726–H2731.
2001.PubMed/NCBI
|
|
10
|
Yeh CH, Chen TP, Wu YC, Lin YM and Jing
Lin P: Inhibition of NFkappaB activation with curcumin attenuates
plasma inflammatory cytokines surge and cardiomyocytic apoptosis
following cardiac ischemia/reperfusion. J Surg Res. 125:109–116.
2005. View Article : Google Scholar
|
|
11
|
Yeh CH, Chen TP, Lee CH, Wu YC, Lin YM and
Lin PJ: Cardiomyocytic apoptosis following global cardiac ischemia
and reperfusion can be attenuated by peroxisome
proliferator-activated receptor alpha but not gamma activators.
Shock. 26:262–270. 2006. View Article : Google Scholar
|
|
12
|
Wang M, Tsai BM, Reiger KM, Brown JW and
Meldrum DR: 17-beta-Estradiol decreases p38 MAPK-mediated
myocardial inflammation and dysfunction following acute ischemia. J
Mol Cell Cardiol. 40:202–212. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kopp EB and Ghosh S: NF-kappa B and rel
proteins in innate immunity. Adv Immunol. 58:1–27. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kanno T, Franzoso G and Siebenlist U:
Human T-cell leukemia virus type I Tax-protein-mediated activation
of NF-kappa B from p100 (NF-kappa B2)-inhibited cytoplasmic
reservoirs. Proc Natl Acad Sci USA. 91:12634–12638. 1994.
View Article : Google Scholar
|
|
15
|
Onai Y, Suzuki J, Maejima Y, Haraguchi G,
Muto S, Itai A and Isobe M: Inhibition of NF-{kappa}B improves left
ventricular remodeling and cardiac dysfunction after myocardial
infarction. Am J Physiol Heart Circ Physiol. 292:H530–H538. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Karppi J, Kurl S, Nurmi T, Rissanen TH,
Pukkala E and Nyyssönen K: Serum lycopene and the risk of cancer:
the Kuopio Ischaemic Heart Disease Risk Factor (KIHD) study. Ann
Epidemiol. 19:512–518. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Riccioni G, Mancini B, Di Ilio E,
Bucciarelli T and D’Orazio N: Protective effect of lycopene in
cardiovascular disease. Eur Rev Med Pharmacol Sci. 12:183–190.
2008.PubMed/NCBI
|
|
18
|
Parvin R and Akhter N: Protective effect
of tomato against adrenaline-induced myocardial infarction in rats.
Bangladesh Med Res Counc Bull. 34:104–108. 2008.PubMed/NCBI
|
|
19
|
Upaganlawar A, Gandhi H and Balaraman R:
Effect of vitamin E alone and in combination with lycopene on
biochemical and histopathological alterations in
isoproterenol-induced myocardial infarction in rats. J Pharmacol
Pharmacother. 1:24–31. 2010. View Article : Google Scholar
|
|
20
|
Simone RE, Russo M, Catalano A, Monego G,
Froehlich K, Boehm V and Palozza P: Lycopene inhibits
NF-κB-mediated IL-8 expression and changes redox and PPARγ
signalling in cigarette smoke-stimulated macrophages. PLoS One.
6:e196522011.
|
|
21
|
Gouranton E, Thabuis C, Riollet C,
Malezet-Desmoulins C, El Yazidi C, Amiot MJ, Borel P and Landrier
JF: Lycopene inhibits proinflammatory cytokine and chemokine
expression in adipose tissue. J Nutr Biochem. 22:642–648. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Rajadurai M and Stanely Mainzen Prince P:
Preventive effect of naringin on cardiac markers,
electrocardiographic patterns and lysosomal hydrolases in normal
and isoproterenol-induced myocardial infarction in Wistar rats.
Toxicology. 230:178–188. 2007. View Article : Google Scholar
|
|
23
|
Lujan HL and DiCarlo SE: Mimicking the
endogenous current of injury improves post-infarct cardiac
remodeling. Med Hypotheses. 81:521–523. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Meloni M, Marchetti M, Garner K,
Littlejohns B, Sala-Newby G, Xenophontos N, Floris I, Suleiman MS,
Madeddu P, Caporali A and Emanueli C: Local inhibition of
microRNA-24 improves reparative angiogenesis and left ventricle
remodeling and function in mice with myocardial infarction. Mol
Ther. 21:1390–1402. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kanno Y, Watanabe R, Zempo H, Ogawa M,
Suzuki J and Isobe M: Chlorogenic acid attenuates ventricular
remodeling after myocardial infarction in mice. Int Heart J.
54:176–180. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lei J, Xue S, Wu W, Zhou S, Zhang Y, Yuan
G and Wang J: Sdc1 overexpression inhibits the p38 MAPK pathway and
lessens fibrotic ventricular remodeling in MI rats. Inflammation.
36:603–615. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Morgia G, Cimino S, Favilla V, Russo GI,
Squadrito F, Mucciardi G, Masieri L, Minutoli L, Grosso G and
Castelli T: Effects of Serenoa repens, selenium and lycopene
(Profluss®) on chronic inflammation associated with
benign prostatic hyperplasia: results of ‘FLOG’ (Flogosis and
Profluss in Prostatic and Genital Disease), a multicentre Italian
study. Int Braz J Urol. 39:214–221. 2013.
|
|
28
|
Renju GL, Muraleedhara Kurup G and Saritha
Kumari CH: Anti-inflammatory activity of lycopene isolated from
Chlorella marina on type II collagen induced arthritis in
Sprague Dawley rats. Immunopharmacol Immunotoxicol. 35:282–291.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hadad N and Levy R: The synergistic
anti-inflammatory effects of lycopene, lutein, β-carotene and
carnosic acid combinations via redox-based inhibition of NF-κB
signaling. Free Radic Biol Med. 53:1381–1391. 2012.PubMed/NCBI
|
|
30
|
Abbate A and Narula J: Role of apoptosis
in adverse ventricular remodeling. Heart Fail Clin. 8:79–86. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Singh SS and Kang PM: Mechanisms and
inhibitors of apoptosis in cardiovascular diseases. Curr Pharm Des.
17:1783–1793. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sinha K, Das J, Pal PB and Sil PC:
Oxidative stress: the mitochondria-dependent and
mitochondria-independent pathways of apoptosis. Arch Toxicol.
87:1157–1180. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Delmas D, Solary E and Latruffe N:
Resveratrol, a phytochemical inducer of multiple cell death
pathways: apoptosis, autophagy and mitotic catastrophe. Curr Med
Chem. 18:1100–1121. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
MacKenzie SH and Clark AC: Death by
caspase dimerization. Adv Exp Med Biol. 747:55–73. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Moorjani N, Westaby S, Narula J, Catarino
PA, Brittin R, Kemp TJ, Narula N and Sugden PH: Effects of left
ventricular volume overload on mitochondrial and
death-receptor-mediated apoptotic pathways in the transition to
heart failure. Am J Cardiol. 103:1261–1268. 2009.PubMed/NCBI
|
|
36
|
Satoh M, Minami Y, Takahashi Y and
Nakamura M: Immune modulation: role of the inflammatory cytokine
cascade in the failing human heart. Curr Heart Fail Rep. 5:69–74.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Gao Y, Li T, Wu C, Bittle GJ, Chen S, Wu
ZJ and Griffith BP: Pim-1 mediated signaling during the process of
cardiac remodeling following myocardial infarction in ovine hearts.
J Mol Cell Cardiol. 63:89–97. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Echtermeyer F, Harendza T, Hubrich S,
Lorenz A, Herzog C, Mueller M, Schmitz M, Grund A, Larmann J,
Stypmann J, Schieffer B, Lichtinghagen R, Hilfiker-Kleiner D,
Wollert KC, Heineke J and Theilmeier G: Syndecan-4 signalling
inhibits apoptosis and controls NFAT activity during myocardial
damage and remodelling. Cardiovasc Res. 92:123–131. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hoesel B and Schmid JA: The complexity of
NF-κB signaling in inflammation and cancer. Mol Cancer.
12:862013.
|
|
40
|
González A, Ravassa S, Beaumont J, López B
and Díez J: New targets to treat the structural remodeling of the
myocardium. J Am Coll Cardiol. 58:1833–1843. 2011.PubMed/NCBI
|
|
41
|
Gordon JW, Shaw JA and Kirshenbaum LA:
Multiple facets of NF-κB in the heart: to be or not to NF-κB. Circ
Res. 108:1122–1132. 2011.
|