Introduction
Glycosphingolipids (GSLs), consisting of a
hydrophobic ceramide backbone and a hydrophilic carbohydrate
residue, are an important type of glycolipid found in all animal
cell surface membranes. GSLs are important in a variety of normal
physiological processes and pathological conditions, including
embryogenesis, immune responses, signal transduction as well as
tumor initiation and progression (1–6).
However, the mechanisms underlying the regulation of GSL
biosynthesis remain to be elucidated.
Previous studies have demonstrated that
epithelial-mesenchymal transition (EMT) is an important process in
disease development, particularly in tumor metastasis. During EMT,
cells undergo a morphological change from epithelial cells,
arranged in a cobblestone-like monolayer, to dispersed mesenchymal
cells, which are spindle-shaped. They also exhibit a reduction in
the expression of epithelial cell marker molecules, including
E-cadherin (E-cad) and an increase in the expression of mesenchymal
cell marker molecules, including fibronectin, N-cadherin (N-cad)
and vimentin. Furthermore, they exhibit enhanced motility, enabling
them to invade neighboring tissues through the extracellular matrix
(7,8). Transforming growth factor-β (TGFβ) is
commonly used to induce EMT in cancer cell models. Our previous
study demonstrated a decrease in the expression of
gangliotetraosylceramide (Gg4) and in the transcription of the
β1,3-galactosyltransferase-4 (β3GalT4) gene during a
TGFβ-induced EMT process in normal murine mammary gland (NMuMG)
cells (9). The expression of Gg4
and the key epithelial markers E-cad and β-catenin reduced with a
similar time course during EMT. Immunoprecipitation assays
confirmed the interaction between Gg4, E-cad and β-catenin and
suggested that the EMT process is modulated by Gg4 through its
interaction with E-cad and β-catenin at the NMuMG cell surface
(9,10). However, the molecular mechanism
underlying the reduced gene transcription of β3GalT4 during
TGFβ-induced EMT remains to be elucidated.
TGFβ signal transmission involves numerous
biochemical pathways, including the TGFβ/Smads signaling pathway
(11). In this pathway, TGFβ
activates receptor-regulated SMAD (R-Smads) proteins, Smad2 and
Smad3, through receptor-induced phosphorylation, thereby reducing
the affinity of R-Smads for cytoplasmic anchors and increasing
their accumulation in the nucleus (11–13).
R-Smads and Smad4 located in the nucleus recruit
co-activators/repressors and bind to the promoters of target genes
to regulate their transcription (14). The Smad3/4 complex can bind
directly to a DNA sequence (5′-GTCT-3′) termed the Smad-binding
element (SBE) (15,16). Analysis of the β3GalT4
promoter has revealed a number of potential binding sites for
transcription factors, including a Smad4-binding site
(5′-GTCTAGAC-3′) (17). However,
to the best of our knowledge, there is no experimental evidence
that the reduced gene expression of β3GalT4 by TGFβ is
regulated via Smads in general or in particular via the Smad3/4
complex.
In the present study, the transcription levels of
the β3GalT4 gene was detected in patients with breast
cancer, and the association between Smad3/4 and Gg4 during the EMT
process was further studied.
Materials and methods
Cell lines, culture and samples
NMuMG epithelial cells were obtained from the
American Type Culture Collection (Manassas, VA, USA) and cultured
in Dulbecco’s modified Eagle’s medium (DMEM; HyClone, Logan, UT,
USA) containing 10% fetal bovine serum (HyClone), 10 μg/ml insulin
(Sigma-Aldrich, St. Louis, MO, USA) and 1X penicillin/streptomycin
(Gibco-BRL, Carlsbad, CA, USA) at 37°C in 5% CO2.
All samples from the patients with breast cancer and
the healthy controls were collected at the First Affiliated
Hospital of Xi’an, Jiaotong University (Xi’an, China). The patient
characteristics are shown in Table
I. The present study followed the tenets of the Declaration of
Helsinki for the Use of Human Subjects. The present study was
approved by the Ethics Committee of Jiangnan University (Wuxi,
Jiangsu, China) and written informed consent was obtained from all
patients and healthy donors.
 | Table IPatient/disease characteristics and
level of β3GalT4 mRNA in patients with breast cancer. |
Table I
Patient/disease characteristics and
level of β3GalT4 mRNA in patients with breast cancer.
| Sample no. | Age (years) | Tumor size
(cm) | TNMa classification | β3GalT4
expression (relative fold change) |
|---|
| Healthy donors |
| Normal 1 | 49 | - | - | 1 |
| Normal 2 | 54 | - | - | 1.00 ± 0.237 |
| Normal 3 | 35 | - | - | 1.26 ± 0.174 |
| Cancer samples |
| 1 | 54 | multifocal | T2N3M0 IIIc | 0.04± 0.005c |
| 2 | 39 | 1.6 | T1N0M0 I | 0.26±0.055b |
| 3 | 35 | 2.3 | T2N1M0 IIb | 0.09±0.005c |
| 4 | 60 | 6.0 | T4N1M0 IIIb | 0.18±0.006c |
| 5 | 57 | 2.5 | T2N0M0 IIa | 0.04±0.007c |
| 6 | 33 | 2.0 | T1N1M0 IIa | 0.55±0.053b |
| 7 | 42 | 2.5 | T2N1M0 IIb | 0.02±0.003c |
| 8 | 50 | 2.0 | T1N3M0 IIIc | 0.17±0.045c |
Antibodies and reagents
Antibodies
Mouse anti-Gg4 immunoglobulin (Ig)M monoclonal
antibody (mAb) TKH7 (Kjeldsen and Hakomor, unpublished data) was
donated by Dr S. Hakomori (Biomembrane Institute, Seattle, WA,
USA). The primary antibodies used were mouse anti-E-cad IgG2a
monoclonal antibody and mouse-anti-β catenin IgG1 monoclonal
antibody (BD Biosciences, San Jose, CA, USA), mouse anti-N-cad IgG1
and rabbit anti-RNA polymerase II (Pol II) IgG polyclonal antibody
(Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA), mouse
anti-vimentin IgG1 monoclonal antibody and anti-β tubulin IgG1
monoclonal antibody (Sigma-Aldrich), rabbit anti-Smad3 mAb IgG
monoclonal antibody and anti-Smad4 polyclonal antibody (Cell
Signaling Technology, Boston, MA, USA). The secondary antibodies
used were horseradish peroxidase (HRP)-labeled goat anti-mouse IgG
and HRP-labeled goat anti-rabbit IgG (Beyotime Institute of
Biotechnology, Haimen, China) and HRP-labeled goat anti-mouse IgM
(Southern Biotech, Birmingham, Alabama, USA).
Reagents
TGFβ was obtained from BD Biosciences and
Asialo-ganglio-N-tetraosylceramide (asialo-GM1; Gg4), GM1, GM2, GM3
and GM4 were obtained from Matreya Inc. (Pleasant Gap, PA, USA).
Other reagents used were obtained from Sigma-Aldrich, unless
described otherwise.
Expression of recombinant hexahistidine
(His6)-tagged Smad3 and Smad4 proteins in Escherichia
coli (E. coli)
The following primers were used to amplify the gene
for mouse Smad3: Smad3p28, forward
5′-GGAATTTCATATTCGTCCATCCTGCCC-3′, NdeI and Smad3cDNA,
reverse 5′-CCGCTCGAGACCCGCTCCCTTTACTCCTA-3′, XhoI. The
following primers were used were used to amplify the gene for mouse
Smad4: Smad4p28, forward
5′-GGAATTTCATATGGACAATATGTCTATAACAAATAC-3′, NdeI and
Smad4cDNA, reverse 5′-GGAATTCCTGAGATCTCAGTCTAAAGGCT-3′,
EcoRI. The polymerase chain reaction (PCR) fragments were
inserted into the expression vector pET-28a (+) to generate
pET28-Smad3 and pET28-Smad4 and introduced into E. coli BL21
(DE3) for protein expression. The recombinant
His6-tagged Smad3 and Smad4 proteins produced by
isopropyl β-D-1-thiogalactopyranoside induction were purified on a
Ni2+-NTA spin column (Roche Diagnostics, Basel,
Switzerland).
Electrophoretic mobility shift assay
(EMSA)
EMSAs were performed using a 2nd Generation DIG Gel
Shift kit (Roche Diagnostics) according to the manufacturer’s
instructions. The probes were amplified using PCR and labeled with
digoxigenin (DIG) at the 3′-terminal end. The probes, proteins and
poly(deoxyinosinic-deoxycytidylic) in binding buffer [100 mM Hepes,
pH 7.6; 5 mM EDTA; 50 mM
(NH4)2S04; 5 mM DTT; Tween 20, 1%
(w/v); 150 mM KCl; Roche Diagnostics, Basel, Switzerland] were
mixed and incubated at 25°C for 30 min prior to adding 5 μl loading
buffer. The protein-DNA complex and free DNA were separated on
native 5% polyacrylamide gels and transferred onto nylon membranes.
The membranes were treated according to the manufacturer’s
instructions and the bands on the membranes were detected using a
Chemi Doc XRS chemiluminescent imaging system (Bio-Rad, Hercules,
CA, USA).
Chromatin immunoprecipitation (ChIP)
assay
ChIP assays were performed, as described previously
(18). In brief, NMuMG cells were
grown in regular medium (DMEM medium containing 10% fetal bovine
serum) for 24 h, following which the medium was replaced by regular
medium containing 2 ng/ml TGFβ and the cells were incubated for 48
h at 37°C and then fixed in DMEM containing 1% formaldehyde. ChIP
was performed using the anti-Smad4 antibody. Anti-Pol II antibody
immunoprecipitated with a Pol II-actin promoter complex was used as
a positive control. DNA was extracted and pellets were purified
using a Quick DNA purification kit (Beijing CoWin Biotech. Co.,
Beijing, China). The DNA template (2 μl) was used for PCR with the
following primers: β3GalT4pL, forward 5′-GGTGTGTTAGGGGACTGGT-3′ and
reverse 5′-TGGACTGTGCAGCCTGAT-3′ for the β3GalT4 promoter;
pNon, forward 5′-CTGAGGGTCTTGAGGGTGAG-3′ and reverse
5′-CTCTTCCTCCTGGGAAAACC-3′ for the nonspecific DNA fragment and
pactinp forward 5′-TCAATCTCGCTTTCTCTCTCG-3′ and reverse 5′-CAA
CGAAGGAGCTGCAAAG-3′ for the actin promoter.
Cell transfection
The protein coding regions of the genes were
amplified by PCR using the following primers: Smad3cDNA, forward
‘5-CCCAAGCTTGCCACCATGTCGTCCATCCTGCCC-3′, HindIII, and
Smad3cDNA, reverse for the smad3 gene. Smad4cDNA, forward
‘5-GGGGTACCCCCTTGAACAAATGGACAATATGT-3′, KpnI, and Smad4cDNA,
reverse for the smad4 gene. The products were digested and
ligated into vector pcDNA3.1 (Invitrogen Life Technologies,
Carlsbad, CA, USA). The constructed plasmids were transfected into
NMuMG cells using Lipofectamine 2000 (Invitrogen Life
Technologies). Stable transfectants were selected by screening with
the antibiotic G418 and confirmed by western blot analysis.
Western blot analysis
Cells were harvested and lysed in
radioimmunoprecipitation assay buffer. Protein lysates were
analyzed by SDS-PAGE and western blot analysis, as described
previously (10).
Semiquantitative and quantitative reverse
transcription (RT)-PCR analysis
RNA was extracted using an RNApure Tissue kit
(Beijing CoWin Biotech, Co.). RNA samples were treated with DNase I
and assessed by PCR to rule out chromosomal DNA contamination. Each
RNA sample was reverse transcribed using ReverTra
Ace-α-® (Toyobo, Shanghai, China). Semiquantitative and
quantitative RT-PCR analysis were performed to determine the
transcription levels of various genes using the following primers:
β3GalT4real, forward 5′-CTCTTCCTCCTGGGAAAACC-3′ and reverse
5′-CTGAGGGTCTTGAGGGTGAG-3′ for the β3GalT4 gene and
Tubulinreal, forward 5′-ATCTACCTGTCGGAGCATGG-3′ and reverse
5′-GCCTCCCGATCTATGATGTC-3′ for the Tubulin gene. The
following thermocycler conditions were used for RT-qPCR: 95°C for
10 min, 40 cycles of 95°C for 10 sec and 60°C for 1 min in a 15 μl
reaction system using an UltraSYBR mixture (Beijing CoWin Biotech,
Co.). The DNA products were analyzed using CFX manager software
(version 3.0.1224.1015; Bio-Rad).
GSL extraction, analysis and
immunostaining
GSL extraction, high performance thin layer
chromatography (HPTLC) analysis and immunostaining were performed,
as described previously (19).
Cells were harvested following washing with PBS, extracted with 2
ml isopropanol/hexane/water (55:25:20) by sonication for 30 min and
centrifuged at 1082 × g for 5 min. The extracts were dried in a
nitrogen stream. Redissolved GSLs were incubated in 0.1 M NaOH in
methanol at 40°C for 2 h to hydrolyze the phospholipids and then
neutralized with 1 M HCl. The hydrolyzed phospholipids were removed
by hexane and the remaining solution was evaporated and dissolved
in 1 ml distilled water. The solution was applied to a Sep-Pak C18
cartridge (Varian Medical Systems, Palo Alto, CA, USA) and washed
with water. The total GSLs were eluted with 2 ml
chloroform/methanol (2:1) and stained using 0.5% orcinol in 2 M
sulfuric acid. Gg4 bands were then immunostained using mAb TKH7 on
HPTLC plates (EMD Biosciences, Inc., San Diego, CA, USA).
Statistical analysis
Data were analyzed using a two-tailed two-sample
t-test assuming equal variance using GraphPad Prism 5 software
(GraphPad Software Inc., La Jolla, CA, USA). P≤0.05 was considered
to indicate a statistically significant difference.
Results
Expression of β3GalT4 at the mRNA level
in breast cancer and healthy control subjects
Our previous study demonstrated that the
transcription levels of β3GalT4 were decreased during
TGFβ-induced EMT in NMuMG cells (9). To determine the expression of
β3GalT4 in breast cancer, the expression of β3GalT4
at the mRNA level in different clinical samples was determined
using RT-qPCR. As shown in Table
I, a significant reduction in the expression of β3GalT4
was detected in the patients with breast cancer compared with the
healthy control subjects. This suggested that decreased expression
of the β3GalT4 gene is often present during breast cancer
progression.
Interaction between Smad3/4 and the
β3GalT4 promoter
The Smad3 and Smad4 proteins have a ‘Mad-homology 1’
domain, a DNA-binding module stabilized by a bound zinc atom, at
the N-terminus (11). Smad3 and
Smad4 can bind to DNA directly via the SBE (16). A previous study of the
β3GalT4 promoter (17)
revealed a Smad4 binding site between positions -788 and -795
(5′-GTCTAGAC-3′) relative to the β3GalT4 transcriptional
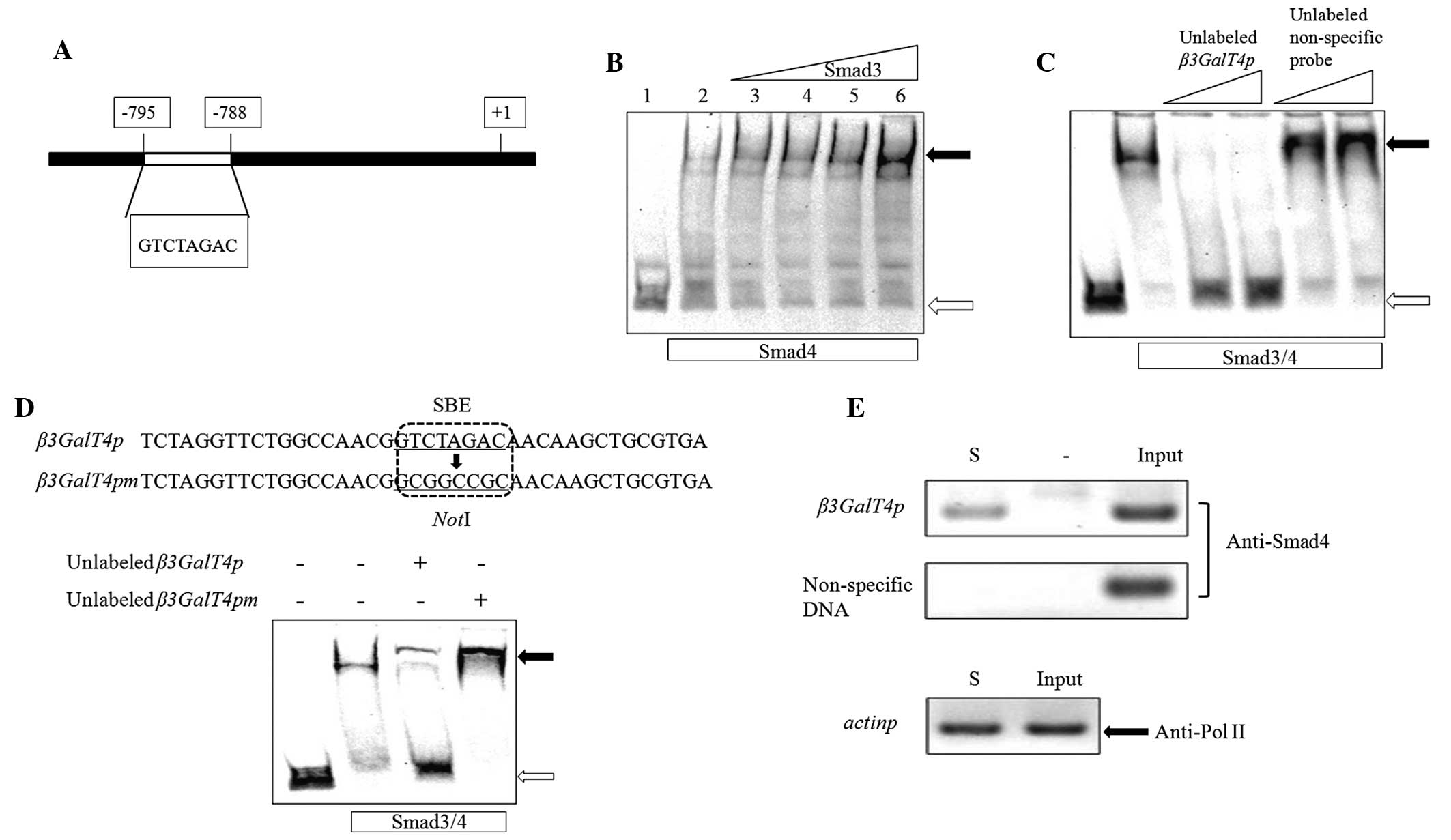
start point (Fig. 1A). In the
present study, EMSAs and ChIP assays were performed to identify the
interactions between Smads and the β3GalT4 promoter.
EMSAs were performed using full-length recombinant
His6-Smad3 and -Smad4 proteins expressed in E.
coli. The probe used was a 237 bp DNA fragment of the
β3GalT4 promoter, amplified using the forward and reverse
β3GalT4pL primers and labeled with DIG. The probe was clearly
retarded on the EMSA gel by the Smad3/4 complex and became more
notable as the quantity of Smad3 increased (Fig. 1B). This finding indicated that
Smad4 alone bound to the probe with low affinity, whereas the
Smad3/4 complex enhanced the retardation more specifically. To
eliminate nonspecific binding, unlabeled probes were used in a
competitive EMSA assay. The retarded band was eliminated completely
by a 100 or 200-fold excess of unlabeled β3GalT4 promoter
probe, whereas an unlabeled nonspecific probe, amplified by the
forward and reverse pNon primers had no effect (Fig. 1C). Xia et al (17) reported that a DNA fragment
(5′-GTCTAGAC-3′) of the β3GalT4 promoter is a Smad4 binding
site. To assess the relative contribution of the SBE, EMSAs were
performed using an unlabeled probe containing either the intact SBE
(β3GalT4p) or a mutated sequence lacking SBE
(β3GalT4pm). The affinity of Smad3/4 for β3GalT4pm
was eliminated completely compared with β3GalT4p (Fig. 1D). These findings indicated that
the SBE is essential for Smad3/4 binding activity.
ChIP assays are commonly used to determine
DNA-protein binding regions. In the present study, NMuMG cells were
treated with TGFβ and formaldehyde was used to fix the
cross-linking between the Smad3/4 proteins and their DNA target.
The cross-linked DNA was extracted and fragmented by sonication and
an anti-Smad4 antibody was used to screen the DNA fragments
attached to the Smad4 protein. The forward and reverse β3GalT4pL
primers used in the above EMSAs were also used in the ChIP assays.
PCR products of the correct size were obtained from the input DNA
and the immunoprecipitated DNA, whereas no such PCR bands were
detected in the control experiments, which used the same DNA
fragment as the template, but without specific antibody addition.
Negative control bands were amplified only with input DNA using the
non-specific primers used in the EMSAs (Fig. 1E). Antibodies directed to Pol II,
an enzyme responsible for transcription of protein-coding genes,
were used as positive controls to ensure the accuracy of the ChIP
results. Forward and reverse pactinp primers were used to detect
the PCR band. Taken together, the findings from the EMSAs and ChIP
assays indicated that the Smad3/4 complex bound specifically to the
β3GalT4 promoter directly through the SBE.
Morphological alterations resulting from
overexpression of Smad3 or Smad4
Our previous study demonstrated that the
β3GalT4 gene, responsible for the expression of Gg4, is
downregulated during the EMT process in NMuMG cells (9). Smads are key factors in this process.
In the present study, to clarify the association between Smad3/4
and β3GalT4, Smad3 and Smad4 overexpression cells were
constructed. NMuMG cells containing empty vector pcDNA3.1 were
described as ‘mock’.
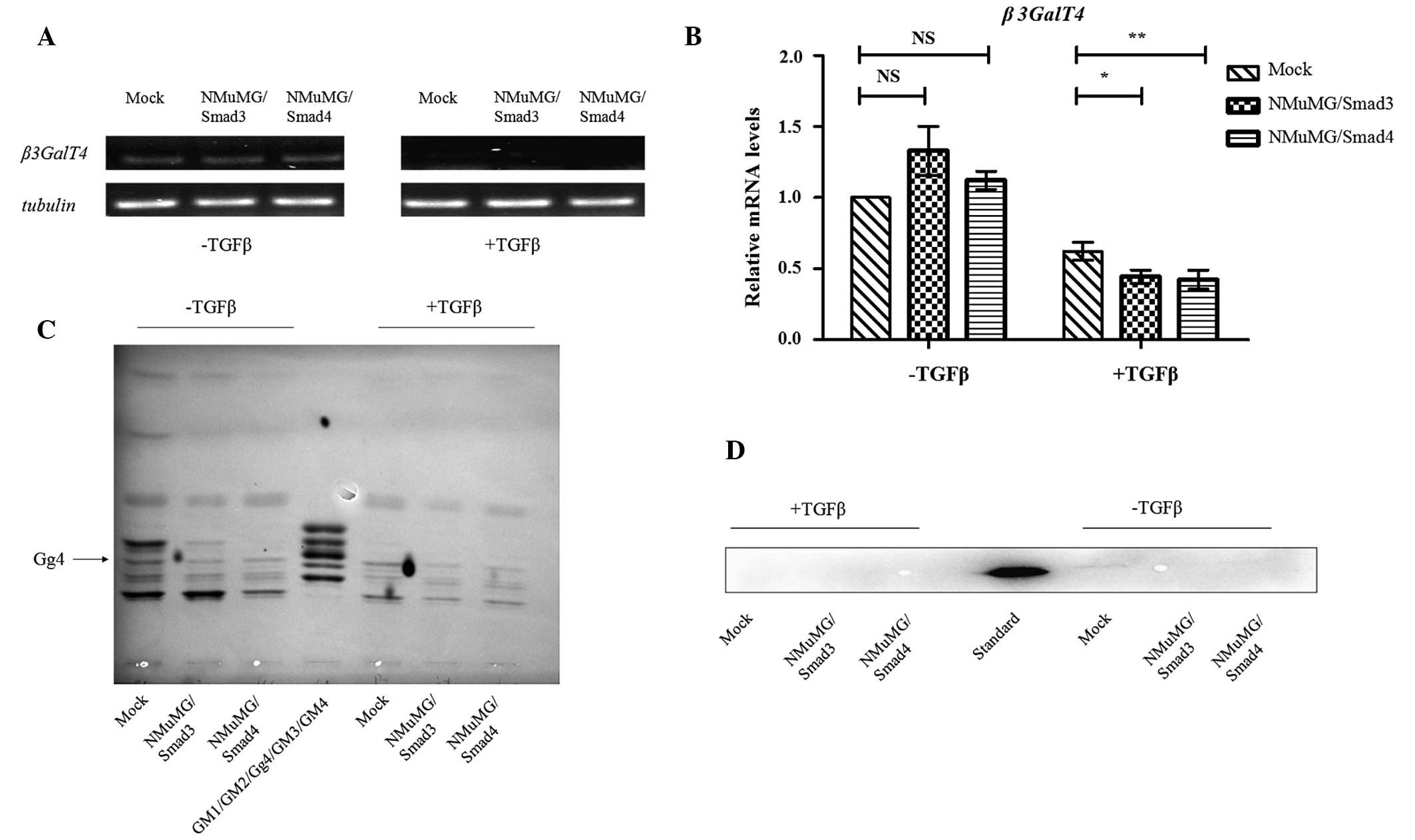
The two transfectants, when cultured in normal DMEM,
exhibited a flattened epithelial morphology similar to that of the
mock (Fig. 2A), indicating that
neither Smad3 nor Smad4 affected the expression of genes that
control cell morphology. Exogenously added TGFβ activated the
TGFβ/Smads signaling pathway and altered cell shape, with the
transfectants and mock cells converted to a fibroblastic morphology
(Fig. 2A).
Reduced expression of the epithelial marker E-cad
and increased expression of the mesenchymal markers N-cad and
vimentin are characteristic processes of EMT. Cells overexpressing
Smad3 and Smad4 demonstrated a markedly enhanced expression of
vimentin and N-cad and a reduced expression of E-cad and β-catenin,
which forms a complex with E-cad stabilizing the cell-cell junction
(Fig. 2B and C). TGFβ treatment
intensified the enhanced and reduced expression of the EMT markers
mentioned above. These findings suggested that the genes
controlling these markers are regulated by Smads and involved in
the TGFβ/Smads pathway via different regulatory mechanisms.
Level of β3GalT4 transcription in
transfectants cells
To determine whether the β3GalT4 gene is
positively or negatively regulated by Smad3/4, the transcription
levels of β3GalT4 in two transfectants, as above, and mock
were evaluated by semiquantitative and quantitative RT-PCR using
the forward and reverse β3GalT4real primers. TGFβ treatment caused
a reduction in β3GalT4 expression (Fig. 3A and B) and the degree of reduction
was greater in the transfectants compared with the mock (Fig. 3B). These findings suggested that,
following activation of the TGFβ/Smads signaling pathway by TGFβ,
the Smads complex is translocated into the nucleus, binds to the
β3GalT4 promoter and downregulates its transcription.
Gg4 expression in transfectants
cells
In our previous study, the mRNA levels of Gg4 and
β3GalT4 were reduced during TGFβ-induced EMT and the
reduction in Gg4 was associated with decreased expression of E-cad
and β-catenin (9). In the present
study, total GSL fractions from transfectants and mock, with or
without TGFβ treatment, were prepared and analyzed using HPTLC. The
level of Gg4 expression decreased in parallel with the levels of
E-cad and β-catenin (Fig. 3C and
D). These findings suggested that the Smad3/4 complex affects
the expression of Gg4 by suppressing mRNA levels of β3GalT4
and that Gg4 interacts closely with the E-cad/β-catenin complex to
stabilize the cell-cell junction.
Discussion
The enzyme β3GalT4 is responsible for the synthesis
of Gg4 in the ganglioside biosynthetic pathway. The decreasing
expression of its gene, β3GalT4, in the TGFβ-induced EMT of
NMuMG cells and in human breast cancer samples indicated its
relevance to the formation and development of breast cancer
(Table I). Therefore, the present
study investigated the molecular mechanism underlying the reduced
transcription of the β3GalT4 gene during TGFβ-induced EMT. A
1.4 kb promoter sequence upstream of the β3GalT4 gene
appears to include a number of potential binding sites for
transcription factors, including Smad4 (17). In the present study, NMuMG cells,
which undergo EMT when treated with TGFβ, were used as a model to
investigate the involvement of the Smad3/4 complex in the
regulation of β3GalT4 expression. The results of EMSAs and
ChIP assays demonstrated that the Smad3/4 complex bound directly to
the β3GalT4 promoter (Fig. 1B,
C and E). Sequence analysis by Xia et al revealed an SBE
site (5′-GTCTAGAC-3′) between positions -788 and -795 relative to
the β3GalT4 transcriptional start point (17). In order to evaluate the role of
this sequence in the interaction between Smads and DNA, an SBE
mutated probe was generated for competitive EMSA (Fig. 1D). The results demonstrated that
the SBE is important in Smads-DNA affinity binding.
Although Smads are key in the EMT process, NMuMG
cells overexpressing either Smad3 or Smad4 did not exhibit notable
morphological changes. These Smad3/4-overexpressing cells were
highly sensitive to TGFβ treatment. Previous studies demonstrated
that R-Smads and Smad4 constantly shuttle between the nucleus and
cytoplasm, regardless of the presence or absence of a signal
(20–22). An activated Smads complex is
typically expressed at a low level to maintain normal physiological
functions, and R-Smads are present in an unphosphorylated form
without sensing signals (23).
These findings may explain why the overexpression of Smads in NMuMG
cells in the present study did not cause notable phenotypic changes
(Fig. 2A). The overexpression of
Smad3/4 did cause enhanced expression of the mesenchymal markers
vimentin and N-cad and reduced the expression of the epithelial
marker E-cad and the intracellular signal transducer β-catenin
(Fig. 2B and C). It appeared that
EMT markers were regulated by the Smad transcription factors, as
also indicated by previous studies (24–26).
The direct or indirect mechanisms whereby Smad3/4 affect EMT
markers remain to be elucidated.
Our previous study demonstrated that, during
TGFβ-induced EMT in NMuMG cells, the expression of the
β3GalT4 gene and Gg4 are downregulated and Gg4 acts as an
inhibitor of EMT, possibly by interacting with E-cad and β-catenin
(9,10). In the present study, the activated
Smad3/4 complex was found to downregulate the expression of
β3GalT4 and the associated levels of mRNA in TGFβ-treated
cells (Fig. 3A and B). Consistent
with this finding, the expression of Gg4, E-cad and β-catenin were
all reduced in the TGFβ-treated cells (Fig. 3C and D). Exogenous TGFβ presumably
activated the Smads to form a complex and stimulated the
translocation of this complex into the nucleus, where it bound to
target genes, including β3GalT4, in the TGFβ/Smads signaling
pathway.
In conclusion, the present study demonstrated that
the activated Smad3/4 complex downregulated the expression of the
β3GalT4 gene responsible for ganglioside expression through
translocation into the nucleus and binding to the β3GalT4
promoter. The findings of the present study suggest that Gg4 is
important in the TGFβ/Smads signaling pathway. Subsequent studies
are required to assess this hypothesis and to elucidate the
mechanisms whereby Gg4 and other GSLs are involved in the
TGFβ-mediated EMT processes.
Acknowledgements
This study was supported by the National Science
Foundation for Young Scientists of China (grant no. 81201572), the
Natural Science Foundation of Jiangsu Province, China (grant no.
BK2012113), the Fundamental Research Funds for the Central
Universities (grant no. JUSRP51319B) and Jiangsu Planned Projects
for Postdoctoral Research Funds (grant no. 1201011C). The authors
would like to thank Dr S. Anderson for editing the English of the
manuscript.
References
|
1
|
Yamashita T, Wada R, Sasaki T, et al: A
vital role for glycosphingolipid synthesis during development and
differentiation. Proc Natl Acad Sci USA. 96:9142–9147. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zeng G, Gao L, Birkle S and Yu RK:
Suppression of ganglioside GD3 expression in a rat F-11 tumor cell
line reduces tumor growth, angiogenesis, and vascular endothelial
growth factor production. Cancer Res. 60:6670–6676. 2000.PubMed/NCBI
|
|
3
|
Zeng G, Gao L and Yu RK: Reduced cell
migration, tumor growth and experimental metastasis of rat F-11
cells whose expression of GD3-synthase is suppressed. Int J Cancer.
88:53–57. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hakomori S: Tumor malignancy defined by
aberrant glycosylation and sphingo(glyco)lipid metabolism. Cancer
Res. 56:5309–5318. 1996.PubMed/NCBI
|
|
5
|
Hakomori S: Glycosylation defining cancer
malignancy: new wine in an old bottle. Proc Natl Acad Sci USA.
99:10231–10233. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Allende ML and Proia RL: Lubricating cell
signaling pathways with gangliosides. Curr Opin Struct Biol.
12:587–592. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial-mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Quaggin SE and Kapus A: Scar wars: mapping
the fate of epithelial-mesenchymal-myofibroblast transition. Kidney
Int. 80:41–50. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Guan F, Schaffer L, Handa K and Hakomori
SI: Functional role of gangliotetraosylceramide in
epithelial-to-mesenchymal transition process induced by hypoxia and
by TGF-(beta). FASEB J. 24:4889–4903. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Guan F, Handa K and Hakomori SI: Specific
glycosphingolipids mediate epithelial-to-mesenchymal transition of
human and mouse epithelial cell lines. Proc Natl Acad Sci USA.
106:7461–7466. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shi Y and Massagué J: Mechanisms of
TGF-beta signaling from cell membrane to the nucleus. Cell.
113:685–700. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xu L and Massagué J: Nucleocytoplasmic
shuttling of signal transducers. Nat Rev Mol Cell Biol. 5:209–219.
2004. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kang JS, Liu C and Derynck R: New
regulatory mechanisms of TGF-beta receptor function. Trends Cell
Biol. 19:385–394. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Derynck R and Zhang YE: Smad-dependent and
Smad-independent pathways in TGF-beta family signalling. Nature.
425:577–584. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zawel L, Dai JL, Buckhaults P, et al:
Human Smad3 and Smad4 are sequence-specific transcription
activators. Mol Cell. 1:611–617. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Shi Y, Wang YF, Jayaraman L, Yang H,
Massagué J and Pavletich NP: Crystal structure of a Smad MH1 domain
bound to DNA: insights on DNA binding in TGF-beta signaling. Cell.
94:585–594. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xia T, Gao L, Yu RK and Zeng G:
Characterization of the promoter and the transcription factors for
the mouse UDP-Gal:betaGlcNAc beta1,3-galactosyltransferase gene.
Gene. 309:117–123. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Nelson EA, Walker SR, Alvarez JV and Frank
DA: Isolation of unique STAT5 targets by chromatin
immunoprecipitation-based gene identification. J Biol Chem.
279:54724–54730. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Todeschini AR, Dos Santos JN, Handa K and
Hakomori SI: Ganglioside GM2-tetraspanin CD82 complex inhibits met
and its cross-talk with integrins, providing a basis for control of
cell motility through glycosynapse. J Biol Chem. 282:8123–8133.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pierreux CE, Nicolás FJ and Hill CS:
Transforming growth factor beta-independent shuttling of Smad4
between the cytoplasm and nucleus. Mol Cell Biol. 20:9041–9054.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Schmierer B and Hill CS: Kinetic analysis
of Smad nucleocytoplasmic shuttling reveals a mechanism for
transforming growth factor beta-dependent nuclear accumulation of
Smads. Mol Cell Biol. 25:9845–9858. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hill CS: Nucleocytoplasmic shuttling of
Smad proteins. Cell Res. 19:36–46. 2009. View Article : Google Scholar
|
|
23
|
Inman GJ, Nicolas FJ and Hill CS:
Nucleocytoplasmic shuttling of Smads 2, 3, and 4 permits sensing of
TGF-beta receptor activity. Mol Cell. 10:283–294. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Nieman MT, Prudoff RS, Johnson KR and
Wheelock MJ: N-cadherin promotes motility in human breast cancer
cells regardless of their E-cadherin expression. J Cell Biol.
147:631–644. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Conacci-Sorrell M, Zhurinsky J and
Ben-Ze’ev A: The cadherin-catenin adhesion system in signaling and
cancer. J Clin Invest. 109:987–991. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Peinado H, Portillo F and Cano A:
Transcriptional regulation of cadherins during development and
carcinogenesis. Int J Dev Biol. 48:365–375. 2004. View Article : Google Scholar : PubMed/NCBI
|