Introduction
Pancreatic carcinoma is a malignant tumor of the
digestive system and has an extremely poor prognosis. The average
survival time following diagnosis is <6 months and the 5-year
survival rate is <5% (1). In
China, the incidence of pancreatic cancer is increasing every year
and only a small number of patients are able to undergo surgical
resection. However, there remains a high recurrence rate and a poor
prognosis (1,2). Invasion and metastasis are the key
factors impacting the recurrence and mortality of patients with
pancreatic carcinoma. Therefore, identifying effective molecular
targets to prevent or reduce pancreatic cancer invasion and
metastasis is critical for improving the treatment efficacy and
prognosis.
Peroxisome proliferator-activated receptor-γ (PPARγ)
belongs to a class of ligand-activated nuclear transcription
factors and is a member of the type II nuclear receptor superfamily
(3). PPARγ ligands are divided
into two categories as natural ligands, including 15-deoxy
prostaglandin J2 (15d-PGJ2), and synthetic ligands, including
mainly thiazolidinedione compounds, such as rosiglitazone (RGZ),
pioglitazone (PGZ) and troglitazone (TGZ). Among the synthetic
ligands, RGZ has the highest bioavailability and efficacy and the
least adverse effects. Previous in vivo experiments
demonstrated that following activation with ligands, PPARγ can
inhibit tumor cell growth, induce apoptosis of tumor cells and
exert other biological effects (3,4).
Sasaki et al (5) detected
the expression of PPARγ in human pancreatic cancer cells and human
pancreatic tissues using western blot analysis and reverse
transcription polymerase chain reaction (RT-PCR) methods. The
authors found that PPARγ was expressed in all human pancreatic
cancer cells and 71% of human pancreatic cancer tissues, while no
PPARγ expression was observed in the adjacent normal pancreatic
tissues. Farrow et al (6)
used RGZ to activate PPARγ in the pancreatic cancer cell line
AsPC-1 and thereby suppressed the invasiveness of AsPC-1 cells via
reducing the secretion of a tumor cell invasion factor i.e. tissue
plasminogen activator. Thus, PPARγ activation can inhibit the
invasion of pancreatic cancer, though the exact mechanism remains
to be elucidated.
Hashimoto et al (7) found that the PPARγ natural ligand
15d-PGJ2 can suppress the expression of MMP-2 and MMP-9 and inhibit
pancreatic cancer cell invasion. MMPs are a group of calcium- and
zinc-dependent proteases, and together with their inhibitor, tissue
inhibitor of metalloproteinases (TIMPs), regulate tumor invasion
and metastasis. Among them, MMP-2 and MMP-9 were found to be
overexpressed in pancreatic cancer tissue and closely associated
with tumor invasion and metastasis (8,9). In
addition, Farrow et al (10) reported that the selective PPARγ
agonist RGZ enhanced the expression of phosphatase and tensin
homolog (PTEN) in pancreatic cancer AsPC-1 cells and inhibited AKT
phosphorylation, while application of the PPARγ inhibitor GW9662
suppressed PTEN expression, suggesting that PPARγ ligands attenuate
pancreatic cancer cell growth via inhibiting phosphoinositide
3-kinase (PI3K) activity through upregulating the expression of the
tumor suppressor gene PTEN. PTEN was identified in 1997 as a tumor
suppressor gene closely associated with cancer (11) w. It is important in tumor cell
apoptosis and migration and is the main component of signaling
pathways that control cell cycle initiation and exit, promote
differentiation, initiate repair and regulate apoptosis (11). Therefore, PPARγ activation may
possibly regulate the expression of MMPs through PTEN and thereby
inhibit the invasion of pancreatic cancer cells. These aspects were
examined in the present study.
Materials and methods
Cell culture
Human pancreatic carcinoma PANC-1 cell lines were
purchased from the American Type Culture Collection and were
maintained in Dulbecco's modified Eagle's medium (Gibco-BRL,
Carlsbad, CA, USA) supplemented with 10% fetal bovine serum
(Gibco-BRL), 100 U/ml penicillin and 100 µg/ml streptomycin.
Cells were maintained at 37°C in a humidified atmosphere containing
5% CO2.
Overexpression vector and small
interfering RNA (siRNA) transfection
The following primers were used for PTEN and MMP-2
CDS amplification: PTEN-CDS, forward 5′-CGGAATTCGCCACCATGACAGCCATCATCAAAGAG-3′
and reverse 5′-CCGCTCGAGTCAGACTTTTGTAATTTGTGTATGC-3′;
MMP2-CDS, forward 5′-CGGAATTCGCCACCATGGAGGCGCTAATGGCCCG-3′
and reverse 5′-CCGCTCGAGTCAGCAGCCTAGCCAGTCGG-3′.
Underlined letters represent the restriction enzyme sites, and bold
letters represent the Kozak sequence. The products were digested
with EcoRI and XhoI (Takara Bio, Inc., Otsu, Japan),
cloned into pcDNA3.1 vectors, sequenced and verified. PTEN and
negative control (NC) siRNAs were obtained from Santa Cruz
Biotechnology, Inc. (Shanghai, China). The cells were seeded into
6-well plates at 1×105 cells/ml and incubated for 24 h.
When the cell confluence reached ~70%, plasmid and siRNA
transfection were performed according to the manufacturer's
instructions of Lipofectamine 2000 (Invitrogen Life Technologies,
Carlsbad, CA, USA). The medium was changed within 4–6 h after
transfection. After 24 h of transfection, either RGZ (20 µM)
or GW9662 (20 µM) was added and the cells were cultured for
24–48 h.
Western blot analysis
Total cellular proteins were extracted by incubating
cells in lysis buffer. The protein concentrations in the cell
lysates were determined by bicinchoninic acid assay (Pierce
Biotechnology Inc., Rockford, IL, USA). SDS-PAGE was performed in
8% glycine gels (Bio-Rad Laboratories Inc., Hercules, CA, USA)
loading equal quantities of proteins per lane. Following
electrophoresis, separated proteins were transferred onto
nitrocellulose membranes and blocked with 5% non-fat milk in
Tris-buffered saline and Tween 20 for 1 h. Following this, the
membranes were incubated with rabbit anti-human polyclonal MMP-2
(cat. no. 4022; Cell Signaling Technology, Inc., Beverly, MA, USA;
1:1,000), rabbit anti-human polyclonal MMP-9 (cat. no. 3852; Cell
Signaling Technology, Inc.; 1:1,000), rabbit anti-human monoclonal
PTEN (cat. no. 9552; Cell Signaling Technology Inc.; 1:600) and
mouse anti-human monoclonal GAPDH (cat. no. NBP1-47339; Novus
Biologicals, San Diego, CA, USA; 1:1,000) overnight at 4°C, and
then secondary antibodies conjugated with horseradish peroxidase
(anti-rabbit IgG; cat. no. 7074; Cell Signaling Technology, Inc.)
at 1:7,000–8,000 dilution were incubated with membranes for 1 h at
room temperature. Protein bands were detected using the West Femto
System (Pierce Biotechnology, Inc.).
Wound healing assay
Cells (1×106 per well) were seeded in
6-well plates and allowed to grow until 70% confluence. The cells
were pretreated with Mitomycin C (Roche Diagnostics, Hong Kong,
China), which inhibits cell division, so that the difference in
motility was not affected by differences in cell proliferation
rates. The cells were treated as above and monolayers were then
wounded with a pipette tip. Following being washed with phosphate
buffered-saline (PBS) three times, the Olympus IX-70 microscope
(Olympus, Tokyo, Japan) was used to capture images immediately
following scratching for 0, 6, 24 and 48 h in the same marked
location of the dish. Data were obtained from three independent
experiments.
Transwell matrigel invasion assay
Invasion of cells was evaluated by Transwell
matrigel invasion assay. Briefly, 200 µl of cells following
transfection (1×106 cells/ml) and 600 µl of the
complete medium were added to the upper and lower compartments of
the chamber, respectively. Following incubation for 48 h, cells
migrating to the lower side of the filter were fixed with 4%
paraformaldehyde for 15 min at room temperature, washed with PBS,
stained with crystal violet and then observed under an Olympus
Fluoview 500 IX71 confocal microscope (Olympus).
Statistical analysis
The experiments were performed at least in
triplicate and results are expressed as the mean ± standard
deviation. SPSS statistical package (SPSS 13.0 for Windows; SPSS,
Inc., Chicago, IL, USA) was used for statistical analysis. The
difference between two groups was analyzed by two-tailed Student's
t-test and that between three or more groups was analyzed by
one-way analysis of variance multiple comparisons. P<0.05 and
P<0.01 were considered to indicate a statistically significant
difference.
Results
PPARγ activation by RGZ inhibits the
migration of pancreatic cancer cells
The majority of current studies suggest that PPARγ
activation can inhibit the migration and invasion of tumor cells,
however, few relevant studies have focused on pancreatic cancer.
The present study initially demonstrated the effect of RGZ on the
in vitro migration ability of the pancreatic cancer cell
line Panc-1 using a scratch wound assay. The cells were subjected
to scratch tests following RGZ treatment and the scratch wound was
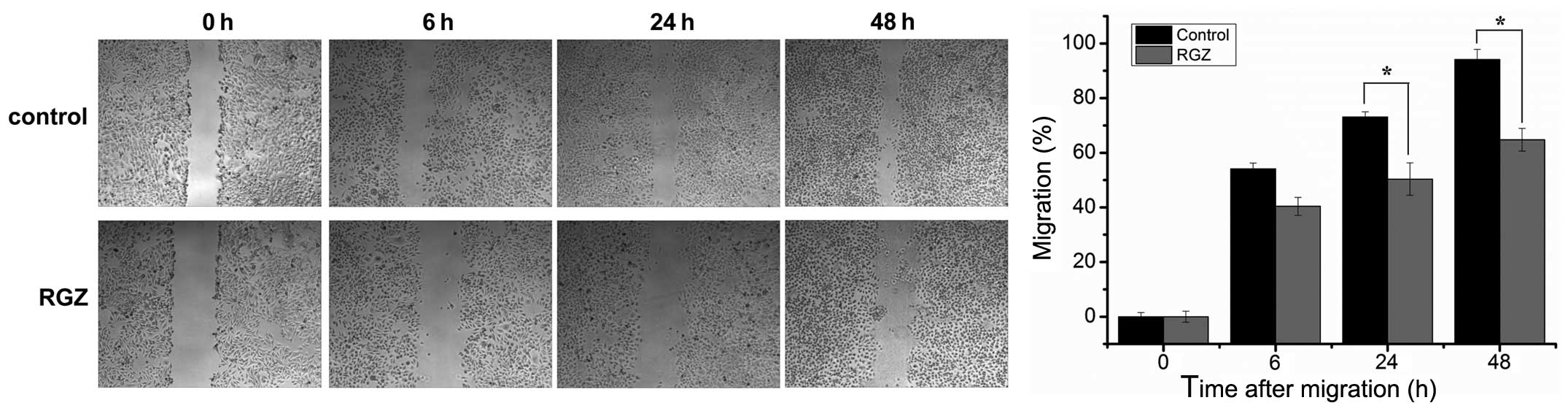
monitored following different periods of time. The results shown in
Fig. 1 indicated that the
migration of RGZ-treated cells was markedly slower than that of the
control cells at the indicated time points. These results suggested
that in pancreatic cancer cells, RGZ activation of PPARγ has the
ability to inhibit migration.
PPARγ activation regulates MMP-2
expression in pancreatic cancer cells
During the process of invasion and metastasis, tumor
cells must overcome a series of natural barriers, including the
extracellular matrix (ECM). MMPs are the main direct factors
involved in the destruction of the ECM; among them, type II
collagenases, including MMP-2 and MMP-9, are most closely
associated with tumor invasion and metastasis (12). Therefore, the present study further
examined the association between PPARγ activation and the
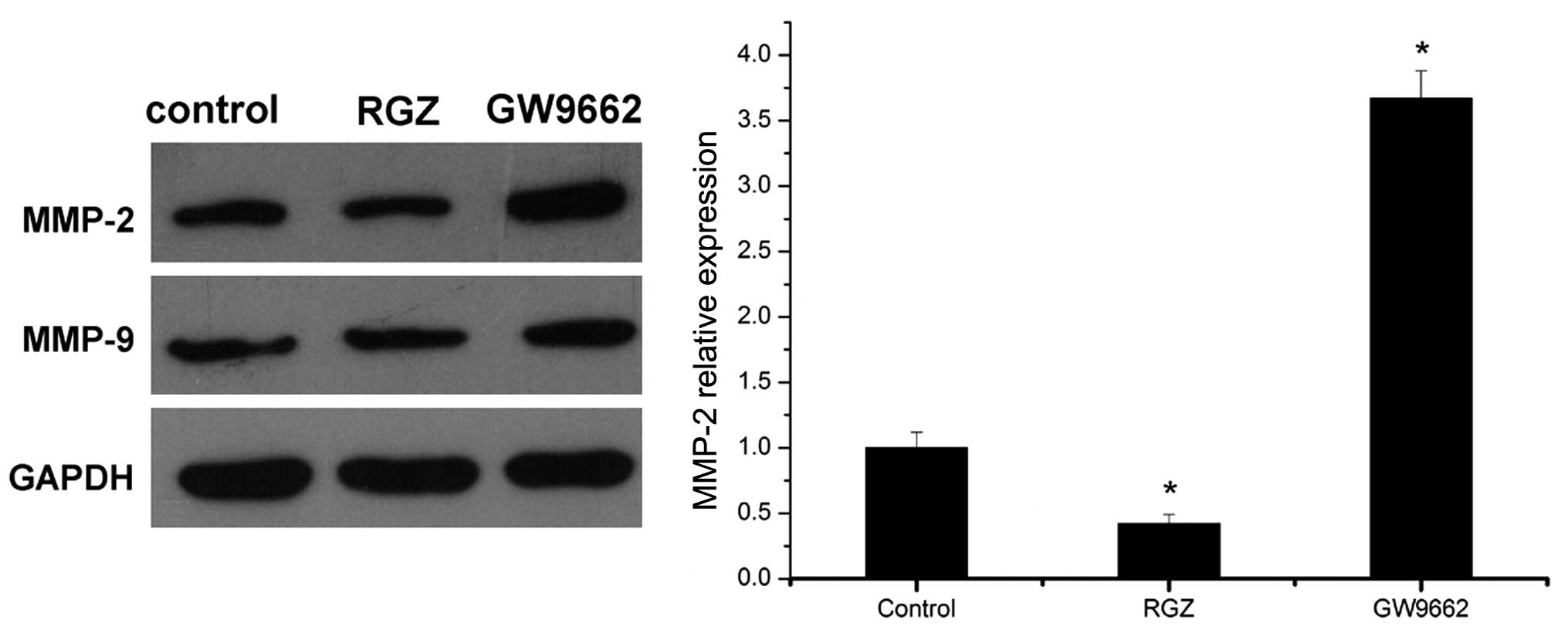
expression of MMP-2 and MMP-9. The results demonstrated that RGZ
treatment significantly inhibited MMP-2 expression, whereas
addition of the PPARγ-specific inhibitor GW9662 significantly
upregulated the expression of MMP-2. However, neither RGZ nor
GW9662 significantly altered the expression of MMP-9 (Fig. 2), suggesting that in pancreatic
cancer cells, PPARγ activation predominantly affected the
expression of MMP-2, while MMP-9 expression may be regulated by
other factors.
PPARγ activation regulates MMP-2
expression through PTEN
Existing studies have demonstrated that PPARγ
activation exerts an inhibitory effect on tumor growth by
activating the tumor suppressor gene PTEN (10,11).
In addition, several studies have also indicated that PTEN has a
tumor suppressor role via regulating a series of factors, including
MMP-2 (11,13,14).
Therefore, PPARγ, PTEN and MMP-2 are closely associated with each
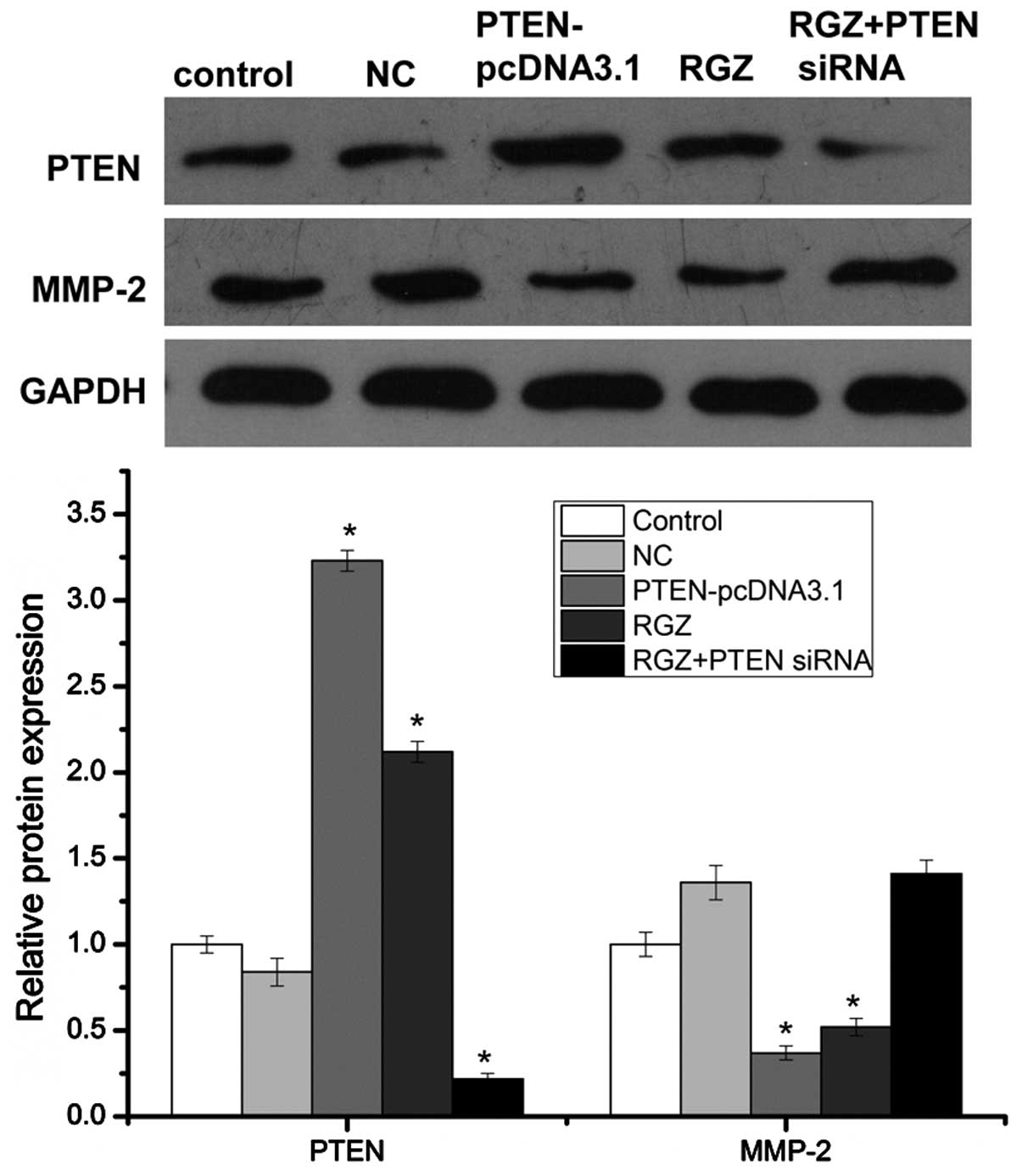
other. Following transfection of a PTEN-overexpressing vector into
cells, the expression of MMP-2 was downregulated. When treating the
pancreatic cancer cells with RGZ, upregulation of PTEN expression
occurred and the expression of MMP-2 was simultaneously
downregulated. However, following transfection of PTEN siRNA, the
RGZ-induced upregulation of PTEN expression was inhibited and the
inhibitory effect of RGZ on MMP-2 expression was also reversed
(Fig. 3). These results indicated
that PPARγ activation inhibits MMP-2 expression via activating
PTEN.
PPARγ activation affects pancreatic
cancer cell invasion by regulating the expression of MMP-2
PPARγ activation affects the invasion and metastasis
of pancreatic cancer cells, and MMP-2 is closely associated with
tumor cell invasion and metastasis. Therefore, in order to
determine whether PPARγ activation affects the invasion of
pancreatic cancer cells via regulating MMP-2 expression, further
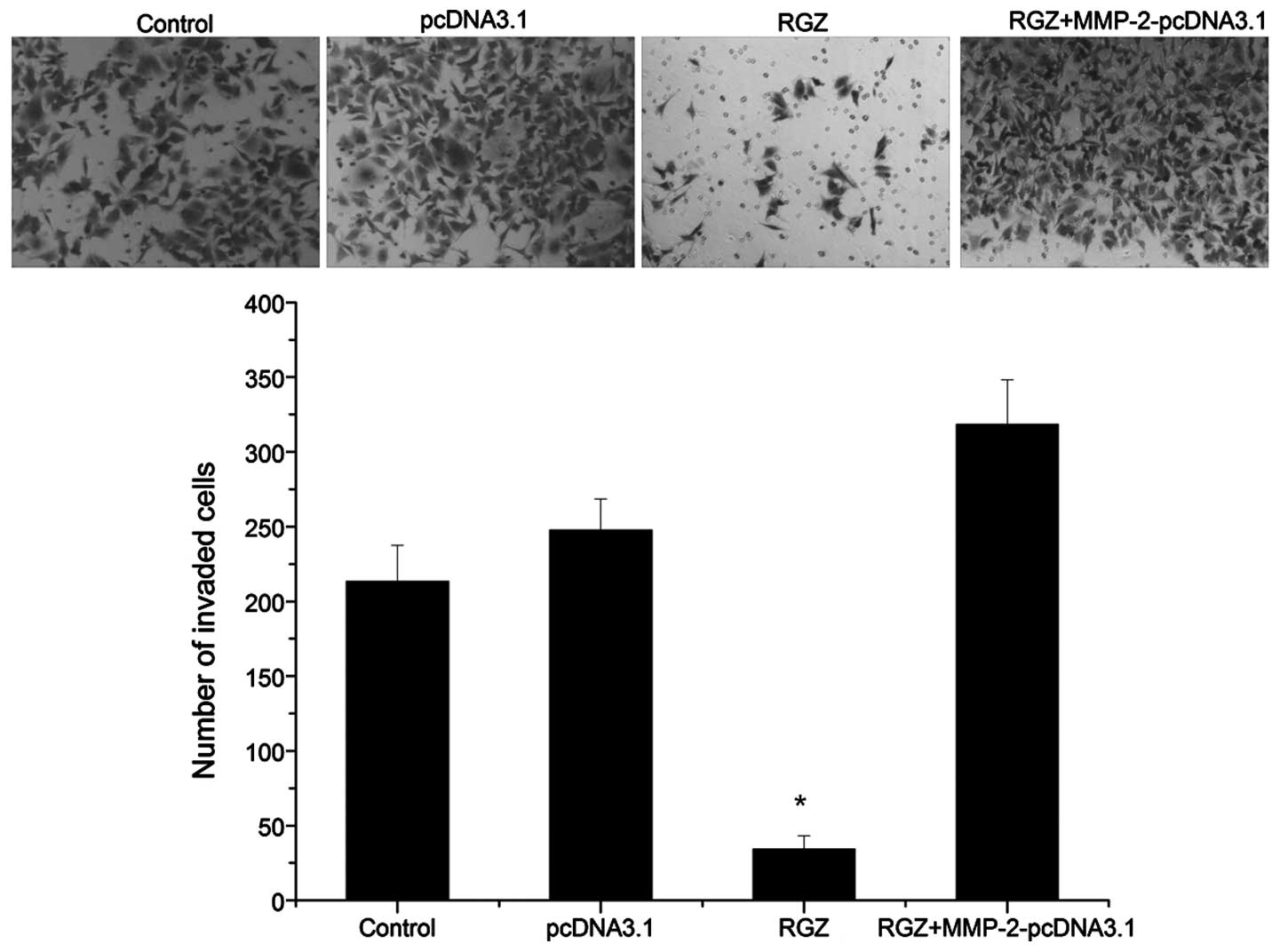
investigation was conducted. The results of the transwell
experiment demonstrated that RGZ significantly inhibited the
invasiveness of pancreatic cancer cells. However, transfection with
a vector overexpressing MMP-2 completely suppressed the inhibitory
effect of RGZ on pancreatic cancer cell invasion, and the
invasiveness of pancreatic cancer cells exhibited an increasing
trend (Fig. 4), indicating that
PPARγ activation can affect the invasion and metastasis of
pancreatic cancer cells via regulating MMP-2 expression.
Discussion
In previous years, the incidence of pancreatic
cancer has exhibited a rising trend globally. It has a latent onset
and rapid progression, but there is are few effective methods for
an early diagnosis and systemic treatment. Due to the late
diagnosis and poor treatment level of pancreatic cancer there is an
urgent requirement for new and effective molecular targets for the
treatment of pancreatic cancer (15).
PPARs belong to the nuclear receptor gene
superfamily, containing three subtypes: α, β (or δ) and γ. Among
them, PPARγ is the most frequently investigated and is closely
associated with tumors. Previous studies have demonstrated that
upon activation with specific ligands, PPARγ can inhibit the growth
of human tumor cells in vitro and in vivo (10,11,16).
PPARγ and its ligands have thus become one of the main focuses of
basic tumor research. Currently, the results of studies on PPARγ in
pancreatic cancer have demonstrated that PPARγ is highly expressed
in pancreatic tumor tissues (17)
and PPARγ activation by ligands inhibits the growth and arrests the
cell cycle of human pancreatic cancer cells at the G1 phase through
various pathways (5,18–20).
Itami et al (20) found
that overexpression of PPARγ in the pancreatic cancer cell line
KMP-3 led to lipid accumulation, while treatment with TGZ increased
the level of p27Kip1 and thereby arrested the cell cycle at G1
phase. However, few studies have focused on the association between
PPARγ and pancreatic cancer cell invasion and metastasis. A scratch
wound assay in the present study demonstrated that following
treatment with RGZ, pancreatic cancer cells exhibited decreased
movement and migration, which is consistent with current findings
in other cell lines. For example, the in vitro and in
vivo experiments of Yu et al (21) identified that RGZ inhibited the
invasion and metastasis of human hepatocellular carcinoma cells
MHCC97-L and BEL-7404. Therefore, similarly, RGZ activation of
PPARγ can inhibit the migration of pancreatic cancer cells.
The invasion and metastasis of pancreatic cancer
cells is a continuous multi-step process, an important step of
which is the detachment of tumor cells from the primary site by
degrading the basal membrane (BM) and ECM through secretion of
destructive enzymes; the tumor cells then migrate through the ECM,
invade the circulation, and form metastatic lesions. The MMPs and
TIMPs together regulate the integrity of the BM and ECM (12,22).
Several studies have demonstrated that compared with normal
pancreatic tissue, subcutaneously-implanted hamster pancreatic
ductal carcinoma cells overexpress MMP-2 and MMP-9, and the serum
levels of MMP-2 and MMP-9 also increased significantly. The growth
of pancreatic cancer was significantly correlated with serum MMP
levels (23). Another study
demonstrated that among 32 pancreatic resection specimens, 31 were
found with venous invasion. Compared with patients without hepatic
metastasis, cases with hepatic metastasis demonstrated a
significantly increased invasion of large and medium vessels, which
was correlated with the overexpression of MMP-2 and MMP-9 (24). Therefore, MMP-2 and MMP-9 has an
important role in the invasion and metastatic process of pancreatic
cancer. The present study found that RGZ significantly
downregulated the expression of MMP-2 in pancreatic cancer cells,
whereas the PPARγ specific inhibitor GW9662 was able to promote
MMP-2 expression. However, the effect of RGZ and GW9662 on MMP-9
expression was not significant. These results suggested that in
pancreatic cancer cells, PPARγ activation predominantly affects the
expression of MMP-2, thereby affecting invasion and metastasis,
which was further confirmed by a subsequent transwell test. The
results of the transwell experiment demonstrated that RGZ
significantly inhibited the invasiveness of pancreatic cancer
cells. However, transfection with a vector overexpressing MMP-2
completely suppressed the inhibitory effect of RGZ on pancreatic
cancer cell invasion, and the invasiveness of pancreatic cancer
cells exhibited an increasing trend, indicating that PPARγ
activation can affect the invasion and metastasis of pancreatic
cancer cells via regulating MMP-2 expression. Using immunoblotting
and RT-PCR methods, Sasaki et al (5) demonstrated that the PPARγ ligand
15d-PGJ2 significantly inhibited the infiltration of pancreatic
cancer cells, which was associated with a reduced activity and
expression of MMP-2 and MMP-9.
In addition, a previous study has also demonstrated
that PPARγ activation increases the expression of PTEN, the first
tumor suppressor gene identified with phosphatase activity, and can
antagonize the activation of protein kinase B by PI3K, inhibit the
Akt/PI3K signaling pathway, regulate cell proliferation and the
cell cycle and inhibit cell growth, thus having a role in tumor
cell metastasis (25). In previous
years, with increasing studies on tumor pathogenesis, it has been
revealed that PPARγ and PTEN are closely associated with the
occurrence and development of pancreatic cancer (20,21).
Therefore, whether PPARγ activation regulates the expression of
MMP-2 via PTEN was investigated and the data confirmed that PPARγ
activation inhibited the expression of MMP-2 through activating
PTEN. Galli et al (26)
found that thiazolidinediones inhibited the invasiveness of
pancreatic cancer cells, affecting gelatinolytic and fibrinolytic
activity with a mechanism involving MMP-2 and PAI-1 expression.
In conclusion, RGZ activates PPARγ leading to
increased PTEN expression and consequently inhibits the expression
of MMP-2, thereby inhibiting the invasion and metastasis of
pancreatic cancer cells. The results further illustrated the
mechanism underlying the inhibitory effect of PPARγ activation on
the invasion and metastasis of pancreatic cancer cells, and hence
provided sufficient scientific basis to establish the PPARγ
signaling pathway as a new target for pancreatic cancer
therapy.
Acknowledgments
This study was supported by research grants from the
Science and Technology Project in Guangdong Province (grant no.
2012B061700047) and the Science and Technology Project in Guangzhou
City-Zhujiang Science and Technology New Star (grant no.
2012J2200039). The authors would like to thank Guang Zhou Ying Sen
Biotech Co., Ltd.
References
|
1
|
Yachida S and Iacobuzio-Donahue CA: The
pathology and genetics of metastatic pancreatic cancer. Arch Pathol
Lab Med. 133:413–422. 2009.PubMed/NCBI
|
|
2
|
Huguet F, Girard N, Guerche CS, Hennequin
C, Mornex F and Azria D: Chemoradiotherapy in the management of
locally advanced pancreatic carcinoma: A qualitative systematic
review. J Clin Oncol. 27:2269–2277. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Koeffler HP: Peroxisome
proliferator-activated receptor gamma and cancers. Clin Cancer Res.
9:1–9. 2003.PubMed/NCBI
|
|
4
|
Rumi MA, Sato H, Ishihara S, Kawashima K,
Hamamoto S, Kazumori H, Okuyama T, Fukuda R, Nagasue N and
Kinoshita Y: Peroxisome proliferator-activated receptor gamma
ligand-induced growth inhibition of human hepatocellular carcinoma.
Br J Cancer. 84:1640–1647. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sasaki T, Fujimoto Y, Tsuchida A, Kawasaki
Y, Kuwada Y and Chayama K: Activation of peroxisome
proliferator-activated receptor gamma inhibits the growth of human
pancreatic cancer. Pathobiology. 69:258–265. 2001. View Article : Google Scholar
|
|
6
|
Farrow B, O'Connor KL, Hashimoto K,
Iwamura T and Evers BM: Selective activation of PPARgamma inhibits
pancreatic cancer invasion and decreases expression of tissue
plasminogen activator. Surgery. 134:206–212. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hashimoto K, Ethridge RT and Evers BM:
Peroxisome proliferator-activated receptor gamma ligand inhibits
cell growth and invasion of human pancreatic cancer cells. Int J
Gastrointest Cancer. 32:7–22. 2002. View Article : Google Scholar
|
|
8
|
Koshiba T, Hosotani R, Wada M, Fujimoto K,
Lee JU, Doi R, Arii S and Imamura M: Detection of matrix
metalloproteinase activity in human pancreatic cancer. Surg Today.
27:302–304. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bramhall SR, Neoptolemos JP, Stamp GW and
Lemoine NR: Imbalance of expression of matrix metalloproteinases
(MMPs) and tissue inhibitors of the matrix metalloproteinases
(TIMPs) in human pancreatic carcinoma. J Pathol. 182:347–355. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Farrow B and Evers BM: Activation of
PPARgamma increases PTEN expression in pancreatic cancer cells.
Biochem Biophys Res Commun. 301:50–53. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen JS, Wang Q, Fu XH, Huang XH, Chen XL,
Cao LQ, Chen LZ, Tan HX, Li W, Bi J, et al: Involvement of
PI3K/PTEN/AKT/mTOR pathway in invasion and metastasis in
hepatocellular carcinoma: Association with MMP-9. Hepatol Res.
39:177–186. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nagase H and Woessner JF Jr: Matrix
metalloproteinases. J Biol Chem. 274:21491–21494. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li X, Yang Z, Song W, Zhou L, Li Q, Tao K,
Zhou J, Wang X, Zheng Z, You N, Dou K and Li H: Overexpression of
Bmi-1 contributes to the invasion and metastasis of hepatocellular
carcinoma by increasing the expression of matrix metalloproteinase
(MMP)-2, MMP-9 and vascular endothelial growth factor via the
PTEN/PI3K/Akt pathway. Int J Oncol. 43:793–802. 2013.PubMed/NCBI
|
|
14
|
Tian T, Nan KJ, Guo H, Wang WJ, Ruan ZP,
Wang SH, Liang X and Lu CX: PTEN inhibits the migration and
invasion of HepG2 cells by coordinately decreasing MMP expression
via the PI3K/Akt pathway. Oncol Rep. 23:1593–600. 2010.PubMed/NCBI
|
|
15
|
Goggins M: Molecular markers of early
pancreatic cancer. J Clin Oncol. 23:4524–4531. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Michalik L, Desvergne B and Wahli W:
Peroxisome-proliferator-activated receptors and cancers: Complex
stories. Nat Rev Cancer. 4:61–70. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kristiansen G, Jacob J, Buckendahl AC,
Grützmann R, Alldinger I, Sipos B, Klöppel G, Bahra M, Langrehr JM,
Neuhaus P, et al: Peroxisome proliferator-activated receptor gamma
is highly expressed in pancreatic cancer and is associated with
shorter overall survival times. Clin Cancer Res. 12:6444–6451.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Toyota M, Miyazaki Y, Kitamura S, Nagasawa
Y, Kiyohara T, Shinomura Y and Matsuzawa Y: Peroxisome
proliferator-activated receptor gamma reduces the growth rate of
pancreatic cancer cells through the reduction of cyclin D1. Life
Sci. 70:1565–1575. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kawa S, Nikaido T, Unno H, Usuda N,
Nakayama K and Kiyosawa K: Growth inhibition and differentiation of
pancreatic cancer cell lines by PPAR gamma ligand troglitazone.
Pancreas. 24:1–7. 2002. View Article : Google Scholar
|
|
20
|
Itami A, Watanabe G, Shimada Y, Hashimoto
Y, Kawamura J, Kato M, Hosotani R and Imamura M: Ligands for
peroxisome proliferator-activated receptor gamma inhibit growth of
pancreatic cancers both in vitro and in vivo. Int J Cancer.
94:370–376. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yu J, Shen B, Chu ES, Teoh N, Cheung KF,
Wu CW, Wang S, Lam CN, Feng H, Zhao J, et al: Inhibitory role of
peroxisome proliferator-activated receptor gamma in
hepatocarcinogenesis in mice and in vitro. Hepatology.
51:2008–2019. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gomez DE, Alonso DF, Yoshiji H and
Thorgeirsson UP: Tissue inhibitors of metalloproteinases:
Structure, regulation and biological functions. Eur J Cell Biol.
74:111–122. 1997.PubMed/NCBI
|
|
23
|
Iki K, Takeo T, Kubozoe T, Aoki S, Hayashi
J and Tsunoda T: Detection of serum MMPs in tumor-bearing hamsters.
J Hepatobiliary Pancreat Surg. 9:478–484. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Nagakawa Y, Aoki T, Kasuya K, Tsuchida A
and Koyanagi Y: Histologic features of venous invasion, expression
of vascular endothelial growth factor and matrix
metalloproteinase-2 and matrix metalloproteinase-9 and the relation
with liver metastasis in pancreatic cancer. Pancreas. 24:169–178.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jones G, Machado J Jr, Tolnay M and Merlo
A: PTEN-independent induction of caspase-mediated cell death and
reduced invasion by the focal adhesion targeting domain (FAT) in
human astrocytic brain tumors which highly express focal adhesion
kinase (FAK). Cancer Res. 61:5688–5691. 2001.PubMed/NCBI
|
|
26
|
Galli A, Ceni E, Crabb DW, Mello T,
Salzano R, Grappone C, Milani S, Surrenti E, Surrenti C and Casini
A: Antidiabetic thiazolidinediones inhibit invasiveness of
pancreatic cancer cells via PPARgamma independent mechanisms. Gut.
53:1688–1697. 2004. View Article : Google Scholar : PubMed/NCBI
|