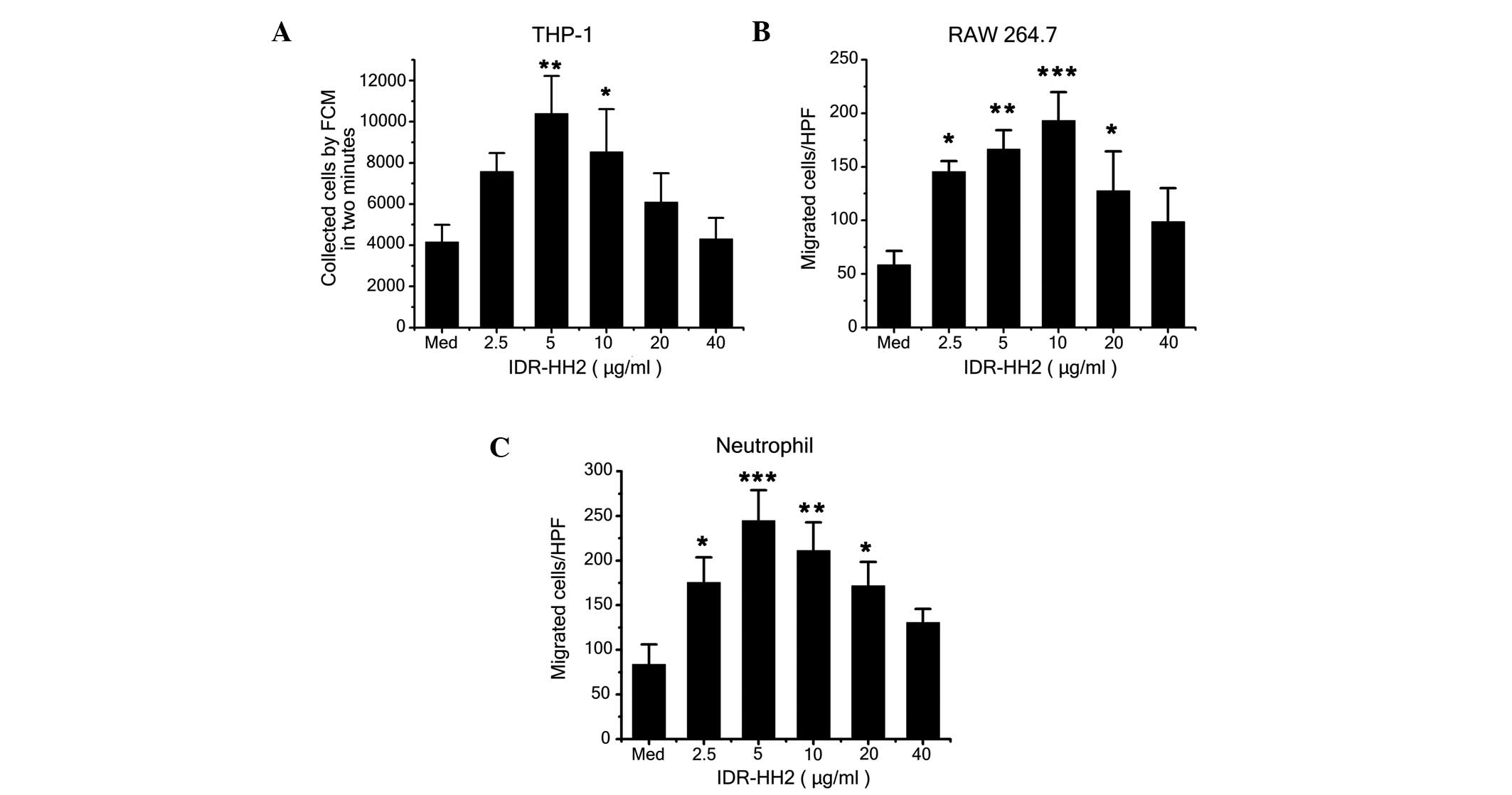
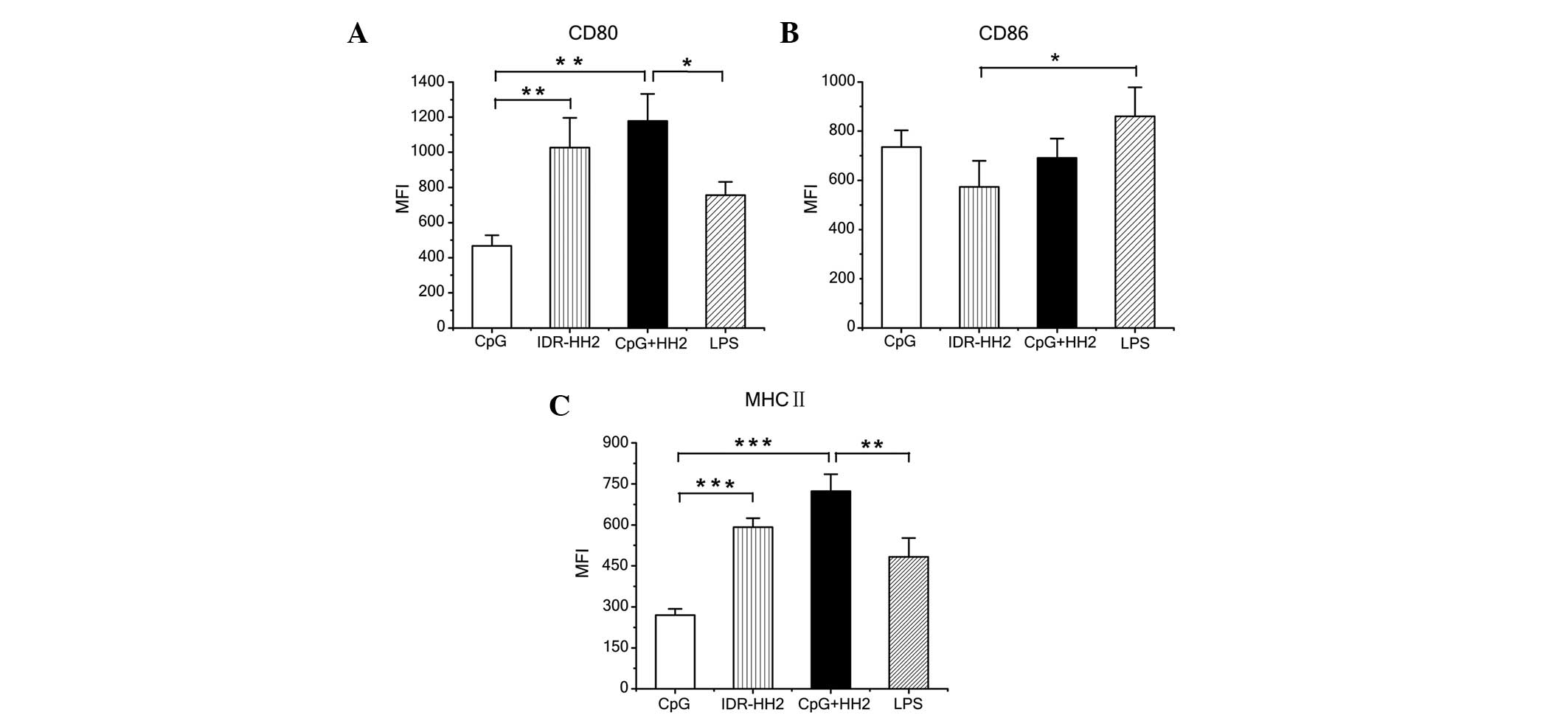
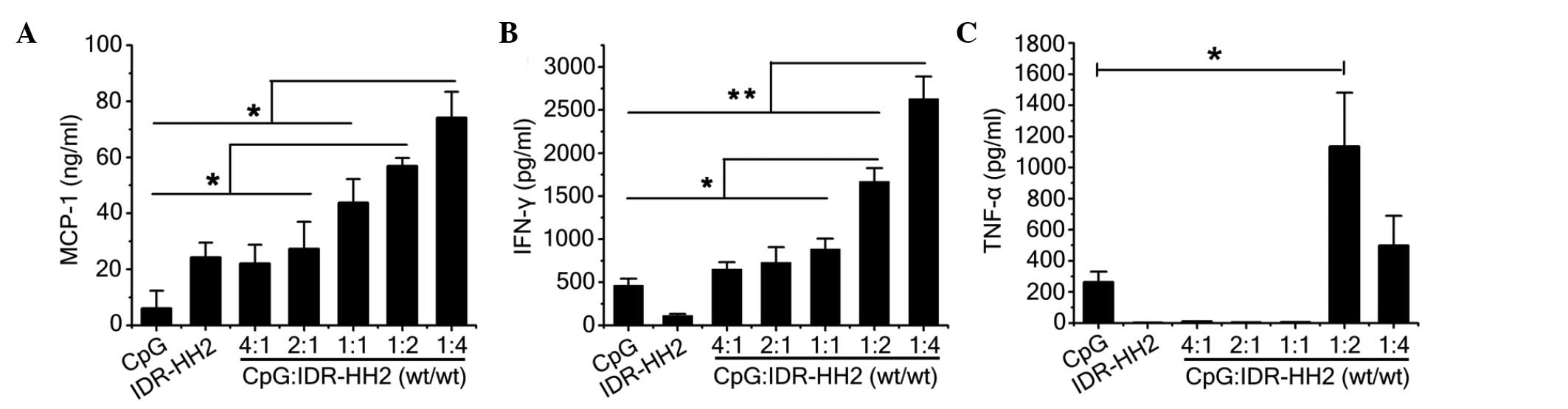
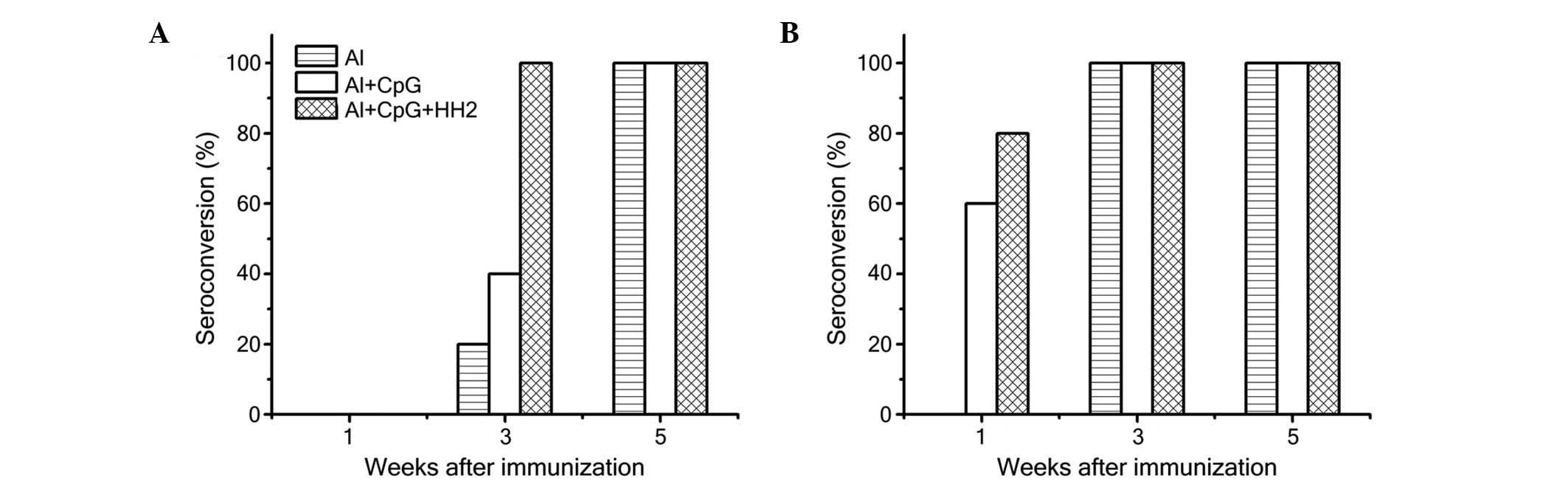
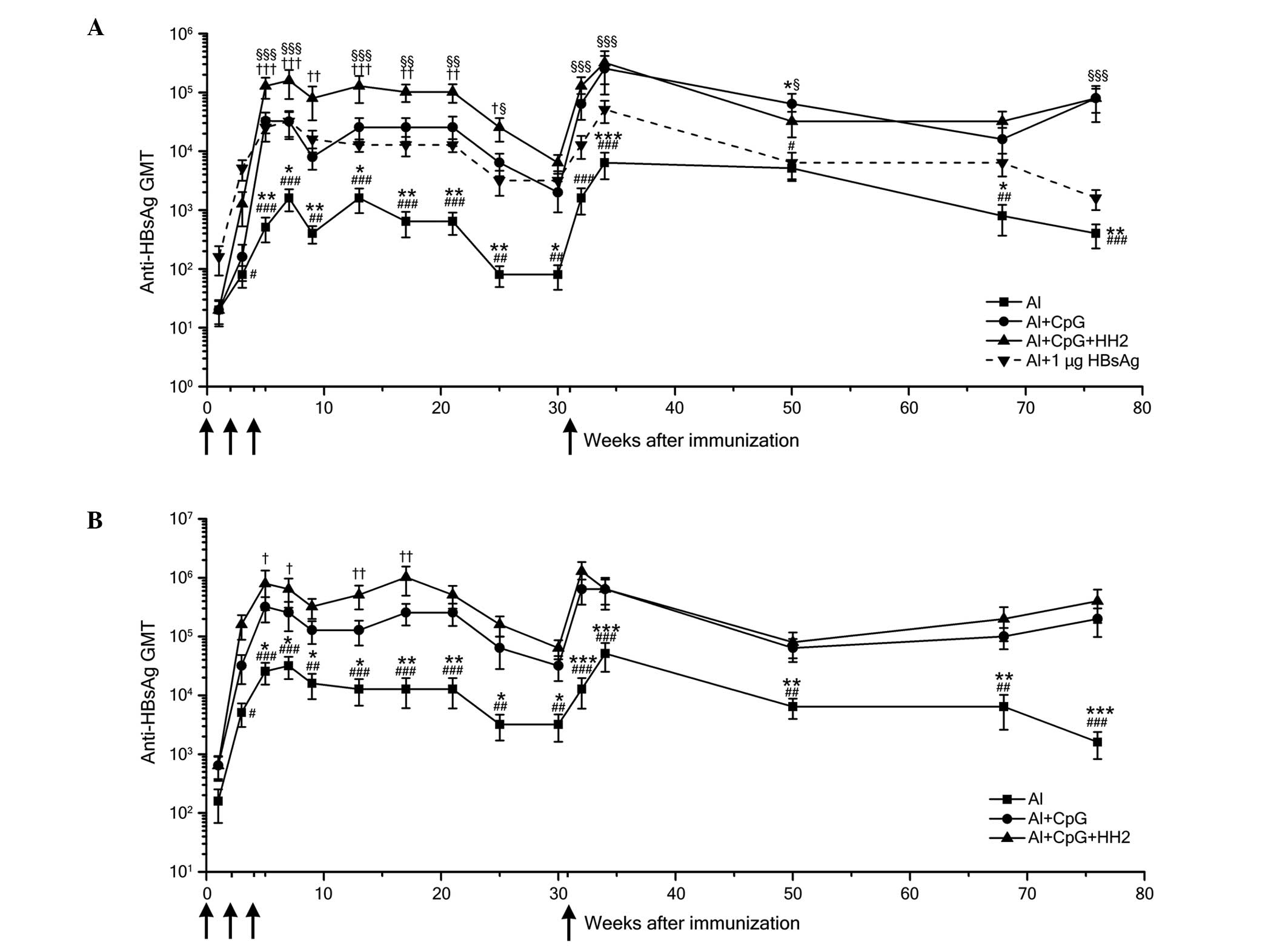
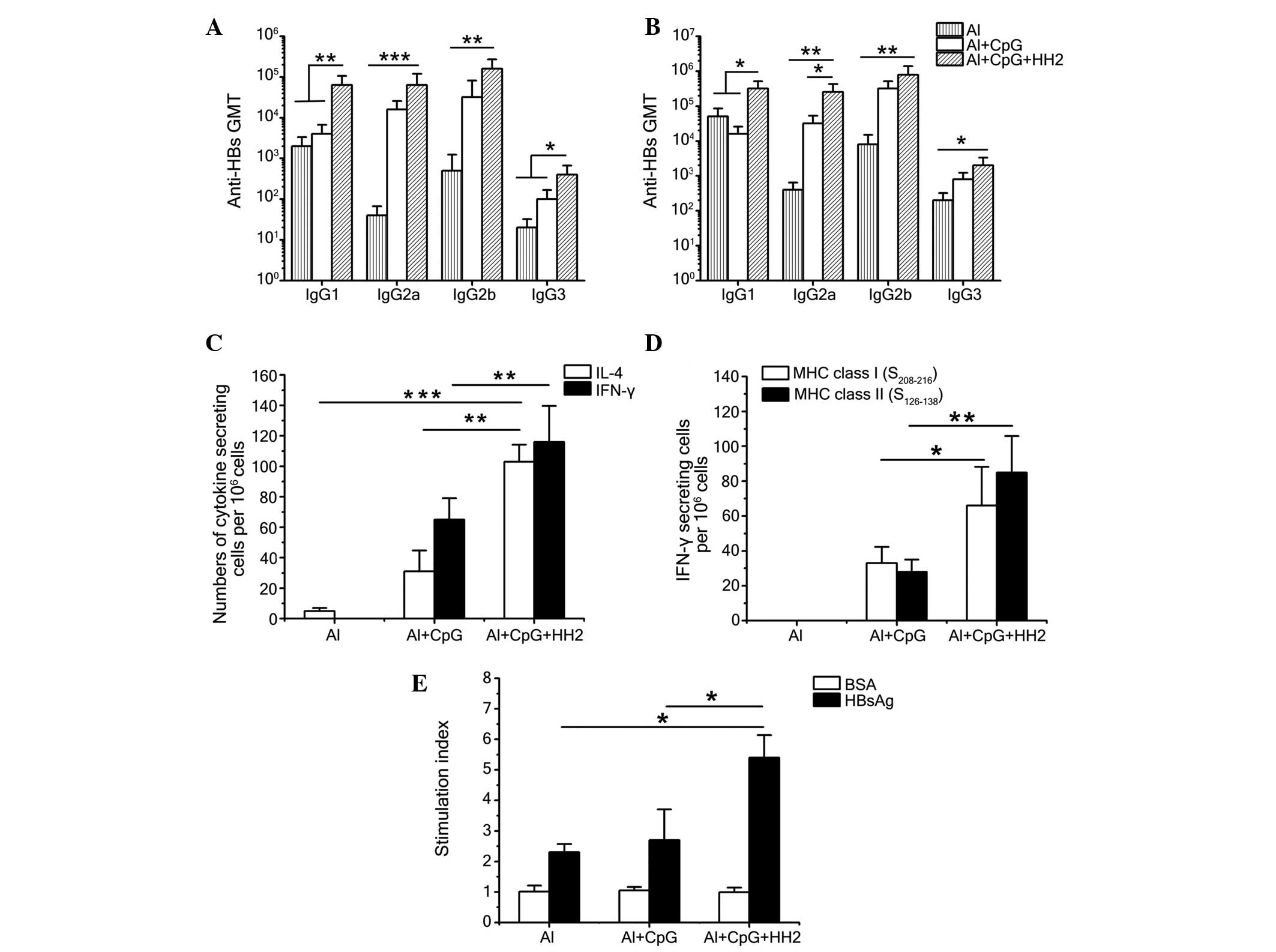
|
1
|
Reed SG, Orr MT and Fox CB: Key roles of
adjuvants in modern vaccines. Nat Med. 19:1597–1608. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mutwiri G, Gerdts V, van Drunen Littel-van
den Hurk S, Auray G, Eng N, Garlapati S, Babiuk LA and Potter A:
Combination adjuvants: The next generation of adjuvants? Expert Rev
Vaccines. 10:95–107. 2011. View Article : Google Scholar
|
|
3
|
Aebig JA, Mullen GE, Dobrescu G, Rausch K,
Lambert L, Ajose-Popoola O, Long CA, Saul A and Miles AP:
Formulation of vaccines containing CpG oligonucleotides and alum. J
Immunol Methods. 323:139–146. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kovacs-Nolan J, Latimer L, Landi A,
Jenssen H, Hancock RE, Babiuk LA and van Drunen Littel-van den Hurk
S: The novel adjuvant combination of CpG ODN, indolicidin and
polyphos-phazene induces potent antibody- and cell-mediated immune
responses in mice. Vaccine. 27:2055–2064. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cao D, Li H, Jiang Z, Xu C, Cheng Q, Yang
Z, Cao G and Zhang L: Synthetic innate defence regulator peptide
enhances in vivo immunostimulatory effects of CpG-ODN in newborn
piglets. Vaccine. 28:6006–6013. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Weeratna RD, Brazolot Millan CL, McCluskie
MJ and Davis HL: CpG ODN can re-direct the Th bias of established
Th2 immune responses in adult and young mice. FEMS Immunol Med
Microbiol. 32:65–71. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Thaker HD, Som A, Ayaz F, Lui D, Pan W,
Scott RW, Anguita J and Tew GN: Synthetic mimics of antimicrobial
peptides with immunomodulatory responses. J Am Chem Soc.
134:11088–11091. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hancock RE and Sahl HG: Antimicrobial and
host-defense peptides as new anti-infective therapeutic strategies.
Nat Biotechnol. 24:1551–1557. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li M, Shi H, Mu Y, Luo Z, Zhang H, Wan Y,
Zhang D, Lu L, Men K, Tian Y, et al: Effective inhibition of
melanoma tumorigenesis and growth via a new complex vaccine based
on NY-ESO-1-alum-polysaccharide-HH2. Mol Cancer. 13:1792014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wu X, Wang Z, Li X, Fan Y, He G, Wan Y, Yu
C, Tang J, Li M, Zhang X, et al: In vitro and in vivo activities of
antimicrobial peptides developed using an amino acid-based activity
prediction method. Antimicrob Agents Chemother. 58:5342–5349. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kindrachuk J, Jenssen H, Elliott M,
Townsend R, Nijnik A, Lee SF, Gerdts V, Babiuk LA, Halperin SA and
Hancock RE: A novel vaccine adjuvant comprised of a synthetic
innate defence regulator peptide and CpG oligonucleotide links
innate and adaptive immunity. Vaccine. 27:4662–4671. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Niyonsaba F, Madera L, Afacan N, Okumura
K, Ogawa H and Hancock RE: The innate defense regulator peptides
IDR-HH2, IDR-1002 and IDR-1018 modulate human neutrophil functions.
J Leukoc Biol. 94:159–170. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Klinman DM: CpG DNA as a vaccine adjuvant.
Expert Rev Vaccines. 2:305–315. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Nierkens S, den Brok MH, Garcia Z, Togher
S, Wagenaars J, Wassink M, Boon L, Ruers TJ, Figdor CG,
Schoenberger SP, et al: Immune adjuvant efficacy of CpG
oligonucleotide in cancer treatment is founded specifically upon
TLR9 function in plasma-cytoid dendritic cells. Cancer Res.
71:6428–6437. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cooper CL, Davis HL, Morris ML, Efler SM,
Adhami MA, Krieg AM, Cameron DW and Heathcote J: CPG 7909, an
immu-nostimulatory TLR9 agonist oligodeoxynucleotide, as adjuvant
to Engerix-B HBV vaccine in healthy adults: A double-blind phase
I/II study. J Clin Immunol. 24:693–701. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lee CC, Avalos AM and Ploegh HL: Accessory
molecules for Toll-like receptors and their function. Nat Rev
Immunol. 12:168–179. 2012.PubMed/NCBI
|
|
17
|
Mendez S, Tabbara K, Belkaid Y, Bertholet
S, Verthelyi D, Klinman D, Seder RA and Sacks DL: Coinjection with
CpG-containing immunostimulatory oligodeoxynucleotides reduces the
pathogenicity of a live vaccine against cutaneous Leishmaniasis but
maintains its potency and durability. Infect Immun. 71:5121–5129.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Brown TH, David J, Acosta-Ramirez E, Moore
JM, Lee S, Zhong G, Hancock RE, Xing Z, Halperin SA and Wang J:
Comparison of immune responses and protective efficacy of
intranasal prime-boost immunization regimens using adenovirus-based
and CpG/HH2 adjuvanted-subunit vaccines against genital Chlamydia
muridarum infection. Vaccine. 30:350–360. 2012. View Article : Google Scholar
|
|
19
|
Kumar S, Jones TR, Oakley MS, Zheng H,
Kuppusamy SP, Taye A, Krieg AM, Stowers AW, Kaslow DC and Hoffman
SL: CpG oligodeoxynucleotide and Montanide ISA 51 adjuvant
combination enhanced the protective efficacy of a subunit malaria
vaccine. Infect Immun. 72:949–957. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chow YH, Chiang BL, Lee YL, Chi WK, Lin
WC, Chen YT and Tao MH: Development of Th1 and Th2 populations and
the nature of immune responses to hepatitis B virus DNA vaccines
can be modulated by codelivery of various cytokine genes. J
Immunol. 160:1320–1329. 1998.PubMed/NCBI
|
|
21
|
Scott MG, Dullaghan E, Mookherjee N,
Glavas N, Waldbrook M, Thompson A, Wang A, Lee K, Doria S, Hamill
P, et al: An anti-infective peptide that selectively modulates the
innate immune response. Nat Biotechnol. 25:465–472. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ishida T, Oyama T, Carbone DP and
Gabrilovich DI: Defective function of Langerhans cells in
tumor-bearing animals is the result of defective maturation from
hemopoietic progenitors. J Immunol. 161:4842–4851. 1998.PubMed/NCBI
|
|
23
|
Mackay CR: Chemokines: Immunology's high
impact factors. Nat Immunol. 2:95–101. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Billeskov R, Elvang TT, Andersen PL and
Dietrich J: The HyVac4 subunit vaccine efficiently boosts BCG-
primed anti- mycobac-terial protective immunity. PLoS One.
7:e399092012. View Article : Google Scholar
|
|
25
|
Milich DR, Thornton GB, Neurath AR, Kent
SB, Michel ML, Tiollais P and Chisari FV: Enhanced immunogenicity
of the pre-S region of hepatitis B surface antigen. Science.
228:1195–1199. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liljeqvist S and Stahl S: Production of
recombinant subunit vaccines: Protein immunogens, live delivery
systems and nucleic acid vaccines. J Biotechnol. 73:1–33. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Flach TL, Ng G, Hari A, Desrosiers MD,
Zhang P, Ward SM, Seamone ME, Vilaysane A, Mucsi AD, Fong Y, et al:
Alum interaction with dendritic cell membrane lipids is essential
for its adjuvanticity. Nat Med. 17:479–487. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Fritz JH, Brunner S, Birnstiel ML, Buschle
M, Gabain Av, Mattner F and Zauner W: The artificial antimicrobial
peptide KLKLLLLLKLK induces predominantly a TH2-type immune
response to co-injected antigens. Vaccine. 22:3274–3284. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Huang W, Seo J, Willingham SB, Czyzewski
AM, Gonzalgo ML, Weissman IL and Barron AE: Learning from
host-defense peptides: Cationic, amphipathic peptoids with potent
anticancer activity. PLoS One. 9:e903972014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ren SX, Cheng AS, To KF, Tong JH, Li MS,
Shen J, Wong CC, Zhang L, Chan RL, Wang XJ, et al: Host immune
defense peptide LL-37 activates caspase-independent apoptosis and
suppresses colon cancer. Cancer Res. 72:6512–6523. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hurtado P and Peh CA: LL-37 promotes rapid
sensing of CpG oligodeoxynucleotides by B lymphocytes and
plasmacytoid dendritic cells. J Immunol. 184:1425–1435. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Soehnlein O, Kai-Larsen Y, Frithiof R,
Sorensen OE, Kenne E, Scharffetter-Kochanek K, Eriksson EE, Herwald
H, Agerberth B and Lindbom L: Neutrophil primary granule proteins
HBP and HNP1-3 boost bacterial phagocytosis by human and murine
macrophages. J Clin Invest. 118:3491–3502. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hilchie AL, Wuerth K and Hancock RE:
Immune modulation by multifaceted cationic host defense
(antimicrobial) peptides. Nat Chem Biol. 9:761–768. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Harder J, Bartels J, Christophers E and
Schröder JM: A peptide antibiotic from human skin. Nature.
387:8611997. View
Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bode C, Zhao G, Steinhagen F, Kinjo T and
Klinman DM: CpG DNA as a vaccine adjuvant. Expert Rev Vaccines.
10:499–511. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Klinman DM, Yi AK, Beaucage SL, Conover J
and Krieg AM: CpG motifs present in bacteria DNA rapidly induce
lymphocytes to secrete interleukin 6, interleukin 12 and interferon
gamma. Proc Natl Acad Sci USA. 93:2879–2883. 1996. View Article : Google Scholar
|
|
37
|
Klinman DM: Immunotherapeutic uses of CpG
oligodeoxy-nucleotides. Nat Rev Immunol. 4:249–258. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Weeratna RD, McCluskie MJ, Xu Y and Davis
HL: CpG DNA induces stronger immune responses with less toxicity
than other adjuvants. Vaccine. 18:1755–1762. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Brazolot Millan CL, Weeratna R, Krieg AM,
Siegrist CA and Davis HL: CpG DNA can induce strong Th1 humoral and
cell-mediated immune responses against hepatitis B surface antigen
in young mice. Proc Natl Acad Sci USA. 95:15553–15558. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chedid MG, Deulofeut H, Yunis DE,
Lara-Marquez ML, Salazar M, Deulofeut R, Awdeh Z, Alper CA and
Yunis EJ: Defect in Th1-like cells of nonresponders to hepatitis B
vaccine. Hum Immunol. 58:42–51. 1997. View Article : Google Scholar
|
|
41
|
Larsen CE, Xu J, Lee S, Dubey DP, Uko G,
Yunis EJ and Alper CA: Complex cytokine responses to hepatitis B
surface antigen and tetanus toxoid in responders, nonresponders and
subjects naive to hepatitis B surface antigen. Vaccine.
18:3021–3030. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Goncalves L, Albarran B, Salmen S, Borges
L, Fields H, Montes H, Soyano A, Diaz Y and Berrueta L: The
nonresponse to hepatitis B vaccination is associated with impaired
lymphocyte activation. Virology. 326:20–28. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chiou SS, Yamauchi K, Nakanishi T and
Obata H: Nature of immunological non-responsiveness to hepatitis B
vaccine in healthy individuals. Immunology. 64:545–550.
1988.PubMed/NCBI
|
|
44
|
Shokrgozar MA and Shokri F: Enumeration of
hepatitis B surface antigen- specific B lymphocytes in responder
and non- responder normal individuals vaccinated with recombinant
hepatitis B surface antigen. Immunology. 104:75–79. 2001.
View Article : Google Scholar : PubMed/NCBI
|