Introduction
Renal cell carcinoma (RCC) is a type of highly
metastasized tumor that accounts for 3% of all malignancies in
adults. RCC has the highest rate of lethality out of all urological
malignancies (1,2). In the United States, there are
~65,000 new cases of RCC and ~14,000 cases of RCC-associated
mortality annually (3). Despite
the development of novel therapeutic drugs, it remains difficult to
treat patients with metastatic RCC and prognostic improvements are
rarely accomplished (4).
Therefore, the development of a more effective therapy for RCC is
required.
The long non-coding RNA (lncRNA) cancer
susceptibility candidate 2 (CASC2), which is located at chromosome
10q26, was initially reported to be downregulated in endometrial
cancer, where it acted as a tumor suppressor gene (5). Previous studies have demonstrated
that exogenous expression of CASC2 significantly inhibits the
growth of undifferentiated endometrial cancer cells and suppresses
glioma cell metastasis (6,7). However, little is currently known
regarding the expression and function of CASC2 in RCC.
MicroRNAs (miRNAs) are non-coding RNA molecules
(length, ~22 nucleotides) which serve important regulatory roles in
various biological processes, including proliferation, migration,
invasion, apoptosis and cell cycle distribution (8). Emerging evidence suggests that miRNAs
are aberrantly expressed in various types of human cancer, and are
involved in cancer initiation, development and metastasis (9). miRNAs may suppress translation or
induce mRNA cleavage by binding to the 3′-untranslated region (UTR)
of target mRNA. As well as protein-coding genes, lncRNAs are novel
targets of miRNAs (10).
The current study demonstrated that CASC2 is lowly
expressed in RCC tissues and cell lines, suggesting that CASC2 acts
as a tumor suppressor gene that may inhibit RCC cell proliferation
and migration. Additionally, miRNA (miR)-21 decreased CASC2
expression in a sequence-specific manner. Thus, the downregulation
of CASC2 by miR-21 may account for CASC2-mediated promotion of RCC
cell proliferation and migration.
Materials and methods
Patient samples
A total of 32 RCC tissues and paired normal tissues
were collected from the Third Affiliated Hospital of Soochow
University (Changzhou, China). Informed consent was obtained from
all patients. The present study was approved by the Institutional
Review Board of the Third Affiliated Hospital of Soochow
University. All samples were frozen in liquid nitrogen immediately
after surgery. The clinical information and pathological
characteristics of the 32 patients with RCC are presented in
Table I.
 | Table IClinical characteristics of 32
patients with renal cell carcinoma. |
Table I
Clinical characteristics of 32
patients with renal cell carcinoma.
| Characteristic | Number |
|---|
| Age (years) |
| ≥50 | 18 |
| <50 | 14 |
| Gender |
| Male | 23 |
| Female | 9 |
| Histological
type |
| Clear cell renal
cell carcinoma | 25 |
| Papillary renal cell
carcinoma | 7 |
| Stage |
| T1a | 8 |
| T1b | 12 |
| T2 | 9 |
| T3 | 2 |
| T4 | 1 |
| Fuhrman grade |
| G1 | 3 (T1a), 4 (T1b) |
| G2 | 2 (T1a), 5 (T1b), 6
(T2) |
| G3 | 3 (T1a), 3 (T1b), 3
(T2), 2 (T3), 1 (T4) |
| G4 | 0 |
Cell culture
The 786-O and A498 human RCC cell lines, and human
embryonic kidney (HEK) 293 cells were obtained from the American
Type Culture Collection (Manassas, VA, USA). Cells were cultured in
Dulbecco's modified Eagle's medium (Invitrogen; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) supplemented with 10% fetal
bovine serum (Invitrogen; Thermo Fisher Scientific, Inc.), 1% 100
U/ml penicillin and 1% 100 mg/ml streptomycin sulfate
(Sigma-Aldrich, St. Louis, MO, USA) at 37°C in a humidified
incubator containing 5% CO2.
Cell transfection
Human CASC2 gene (NR_026939) was cloned into a
pcDNA3.1(+) vector (Thermo Fisher Scientific, Inc.). CASC2 was
amplified by polymerase chain reaction (PCR), then the PCR products
were double-digested by restriction endonucleases of PmeI
and NotI (Takara Bio, Inc., Otsu, Japan). Subsequently, the
digested products were sub-cloned into the PmeI and
NotI sites of the psiCHECK-2 luciferase vector. An empty
vector served as a negative control (NC). The miR-21 mimics,
5′-UAGCUUAUCAGACUGAUGUUGA-3′, and NC mimics,
5′-CAGUACUUUUGUGUAGUACAA-3′ were obtained from Shanghai GenePharma
Co., Ltd. (Shanghai, China). pcDNA3.1(+)-CASC2 over-expression
vector (2 µg) or 2 µg pcDNA3.1(+) empty vector was
transfected into 786-O or A498 cells, and 200 pmol miR-21 mimics or
NC mimics were transfected into 786-O or A498 cells. The 786-O and
A498 cells were transfected using Opti-MEM and Lipofectamine 2000
reagents (Invitrogen; Thermo Fisher Scientific, Inc.) once the
cells reached 50–70% confluence, according to the manufacturer's
protocols.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA of each sample (tissues and cells) was
extracted with TRIzol Reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocols.
Synthesis of cDNA with reverse transcriptase was performed with the
PrimeScript RT Enzyme Mix I kit (Takara Bio, Inc.). The reaction
mixtures were incubated at 37°C for 60 min, 95°C for 5 min and then
held at 4°C. MiScript SYBR Green PCR kit (Qiagen, Inc., Valencia,
CA, USA) was used to conduct a qPCR analysis. The sequences of the
primers (Invitrogen; Thermo Fisher Scientific, Inc.) used were as
follows: miR-21, forward 5′-TTGACTGTTGAATCTCATGGCAA-3′, reverse
primers were provided by the miScript SYBR Green PCR kit; CASC2,
forward 5′-TACAGGACAGTCAGTGGTGGTA-3′, reverse
5′-ACATCTAGCTTAGGAATGTGGC-3′; and human GAPDH (which served as an
internal control), forward 5′-TCAAGAAGGTGGTGAAGCA-3′ and reverse
5′-AGGTGGAGGAGTGGGTGT-3′. The qPCR reaction was set up in a total
volume of 20 µl, consisting of 10 µl MiScript SYBR
Green PCR Mix, 2 µl primers (forward and reverse), 1
µl cDNA template and 7 µl RNase-free water. The
reactions were performed as follows: 95°C for 15 min, followed by
40 cycles at 95°C for 10 sec, 60°C for 30 sec and 72°C for 20 sec,
using an Applied Biosystems 7900 HT PRISM Real-time PCR system
(Applied Biosystems; Thermo Fisher Scientific, Inc.). The PCR
results were calculated using the ΔΔCq method (11).
In vitro cell proliferation assay
Proliferation of 786-O and A498 cells was measured
using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium
bromide (MTT) assay (Sigma-Aldrich). The cells (~2×104)
were seeded into a 96-well culture plate 24 h prior to
transfection. After 0, 24, 48 or 72 h of transfection, 20 µl
MTT (5 mg/ml) was added to each well, and the plates were incubated
for 3 h at 37°C. Subsequently, the precipitate was solubilized with
150 µl dimethyl sulfoxide (Sigma-Aldrich), and absorbance
was measured at a wavelength of 450 nm using an enzyme-linked
immunosorbent assay microplate reader (model 3550; Bio-Rad
Laboratories, Inc., Hercules, CA, USA). Experiments were repeated
at least three times.
In vitro cell migration scratch
assay
786-O and A498 cells were transfected with
pcDNA3.1(+)-CASC2 vector or pcDNA3.1(+) empty vector, and miR-21
mimics or NC mimics. A total of 6 h post-transfection, a
cell-scratch spatula was used to make a scratch in the cell
monolayer. The cells were then washed three times with
phosphate-buffered saline and were incubated at 37°C for 24 h.
Images of the scratches were captured using a digital camera system
(Leica DC 350FX; Leica Microsystems, Wetzlar, Germany) after the
cells had been incubated for 0 and 24 h. The software program
MIAS-2000 (Leica Microsystems) was used to determine the distance
of migration. The experiments were performed in triplicate,
repeated at least three times, and were analyzed in a double-blind
fashion by at least two observers.
Bioinformatics prediction
The potential miR-21 binding sites of CASC2
predicted by computer-aided algorithms were obtained from miRanda
(http://www.microrna.org) and miRcode (http://www.mircode.org).
Dual-luciferase reporter assay
The RNA sequence of wild-type CASC2 (CASC2-WT) was
amplified by PCR, and was inserted between the PmeI and
NotI restriction sites in the 3′-UTR of the hRluc gene in
the psiCHECK-2 luciferase vector (Promega Corporation, Madison, WI,
USA). The primer sequences for CASC2 (Invitrogen; Thermo Fisher
Scientific, Inc.) were as follows: Forward
5′-AGCGGGCTGCAGGGCTGCGGGCGCT-3′ and reverse
5′-TTGATTTAAAGTAATTAGCACATTC-3′. CASC2 RNA sequence with mutations
in the putative binding site (CASC2-MUT) was also inserted into the
hRluc gene in the psiCHECK-2 luciferase vector. CASC2-MUT was
chemically synthesized by Shanghai GenePharma Co., Ltd. All
recombinant plasmids were verified by DNA sequencing analysis.
For the dual-luciferase reporter assay, 786-O cells
were seeded into 24-well plates, and were co-transfected with 0.8
µg CASC2-WT or 0.8 µg CASC2-MUT plasmids and 80 pmol
miR-21 mimics or 80 pmol NC mimics using Lipofectamine 2000. A
total of 24 h post-transfection, firefly and Renilla
luciferase activities were detected using the
GloMax®-Multi Jr Single Tube Multimode Reader (Promega
Corporation), according to the Dual-Luciferase Reporter Assay
system (Promega Corporation). All experiments were performed in
triplicate and repeated at least three times.
Statistical analysis
Experimental data are presented as the mean ±
standard deviation, and were analyzed using SPSS 17.0 software
(SPSS Inc., Chicago, IL, USA). Data were analyzed using Student's
t-test or one-way analysis of variance and the Student-Newman-Keuls
method. P<0.05 was considered to indicate a statistically
significant difference.
Results
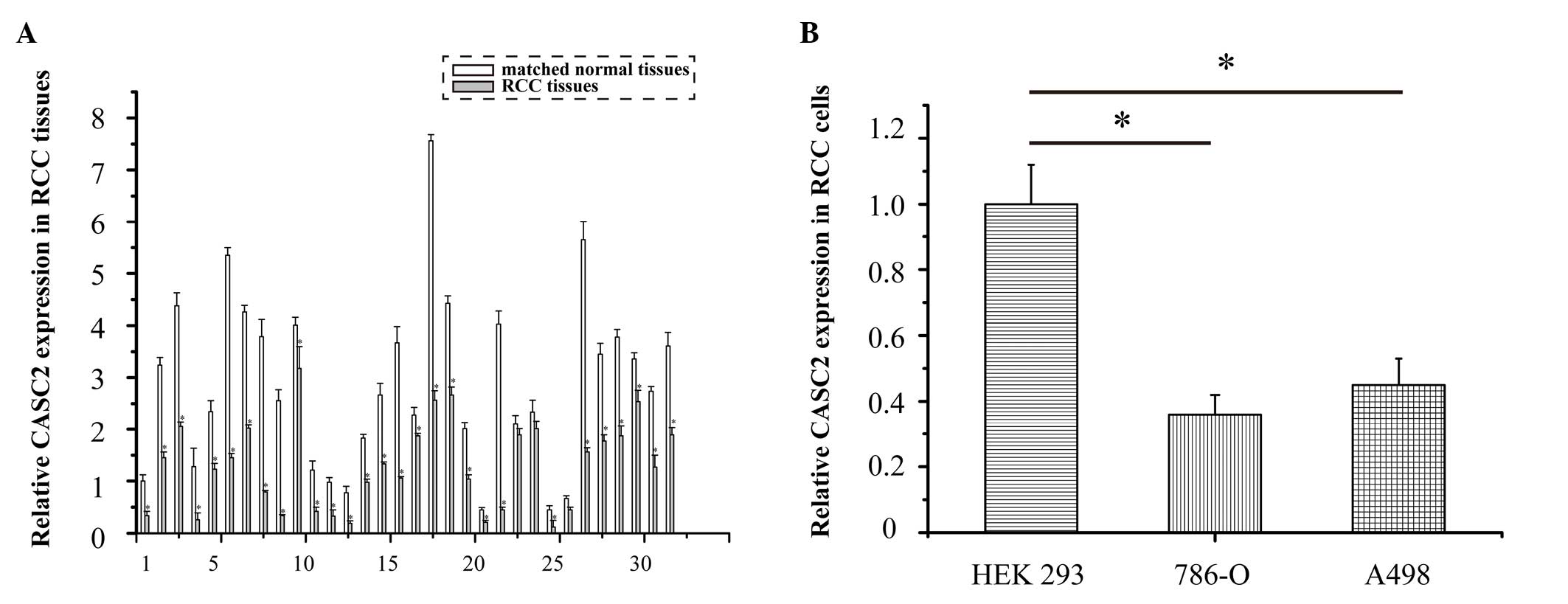
CASC2 is markedly downregulated in RCC
tissues and cell lines, as determined by qPCR analysis
The expression levels of CASC2 were detected in 32
RCC specimens using qPCR. The expression levels of CASC2 were
significantly downregulated in the 32 RCC tissues compared with in
the adjacent normal tissues (Fig.
1A; P<0.05). The expression levels of CASC2 were also lower
in the 786-O and A498 cell lines compared with in the human
embryonic kidney HEK 293 cells (Fig.
1B; P=0.017 and P=0.033, respectively). These data suggest that
CASC2 may function as a tumor suppressor gene in RCC
development.
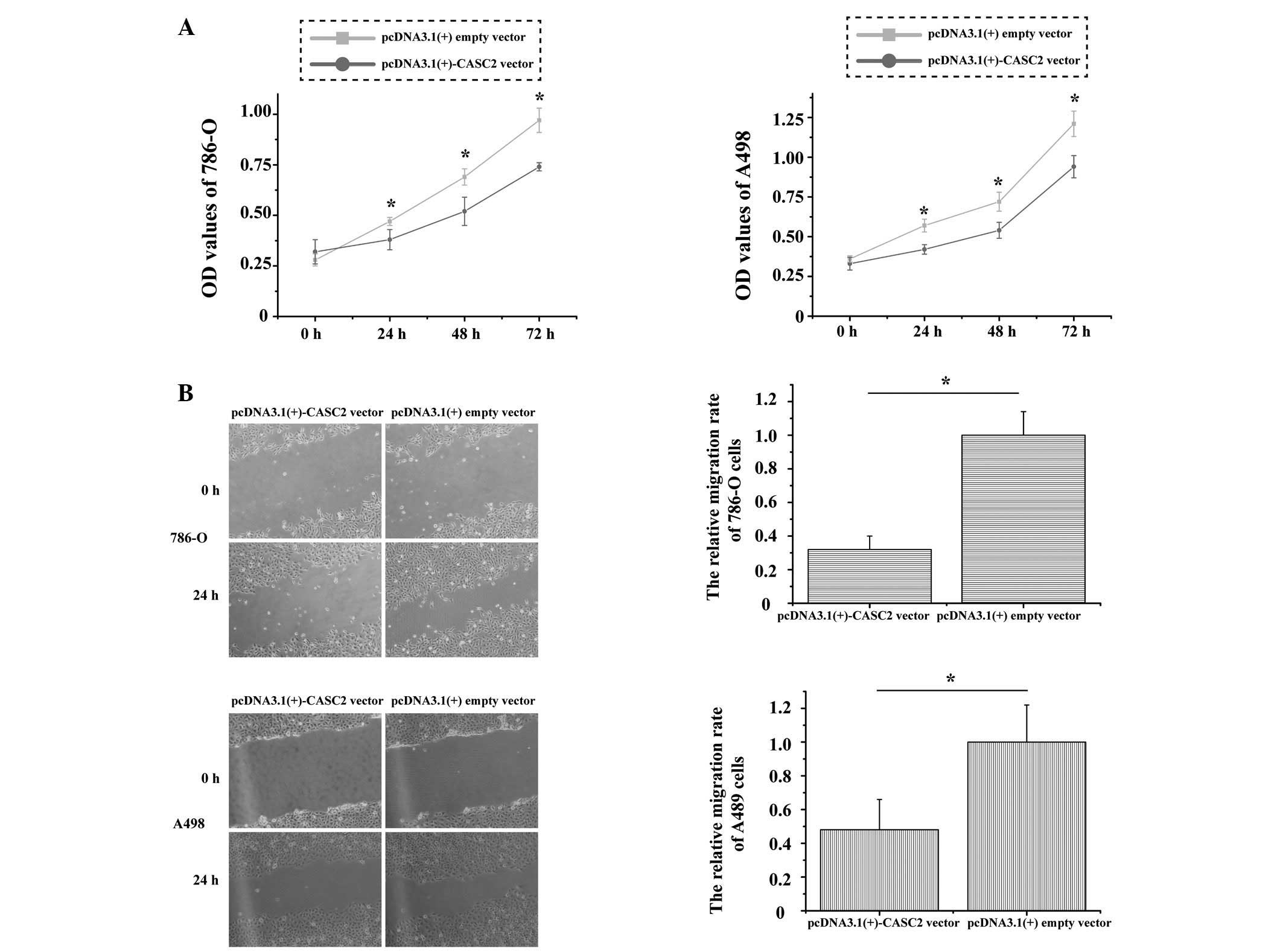
CASC2 inhibits 786-O and A498 cell
proliferation and migration in vitro
To explore the function of CASC2 in RCC cell
proliferation and migration, MTT and wound scratch assays were
performed in 786-O and A498 cell lines. Post-transfection with the
pcDNA3.1(+)-CASC2 overexpression vector, the optical density (OD)
of 786-O cells revealed that relative cell proliferation was
significantly decreased by 8.21% at 24 h, 14.22% at 48 h and 21.1%
at 72 h (P=0.001); the OD of A498 cells also demonstrated that
proliferation was decreased by 6.36% at 24 h, 12.46% at 48 h and
16.32% at 72 h (P=0.005; Fig. 2A).
Images of the wound scratches were captured at 0 and 24 h
post-transfection; overexpression of CASC2 markedly inhibited the
migration of 786-O and A498 cells. The distance of migration is
presented in Fig. 2B. The relative
rates of migration were 32% in 786-O cells and 47% in A498 cells
(P=0.007 and P=0.009, respectively). These results indicate that
CASC2 inhibits the proliferation and migration of RCC cells.
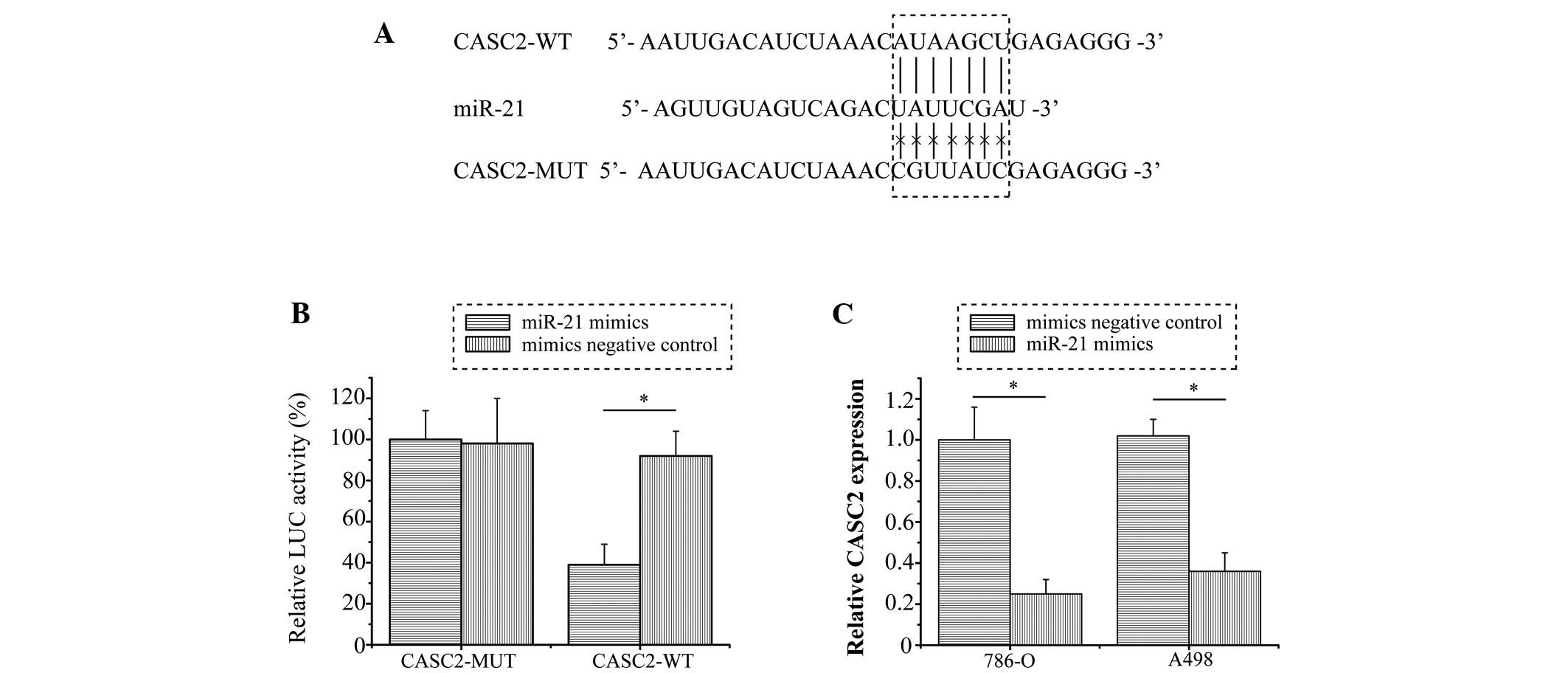
CASC2 is a direct target gene of miR-21
in RCC cells
Previous studies have reported that lncRNAs may act
as competing endogenous RNAs or as molecular sponges that modulate
the concentration and biological function of miRNAs (12,13).
Bioinformatics analysis was performed in order to identify the
potential targeted miRNA of CASC2. The results revealed that miR-21
binding sites were present in CASC2 (Fig. 3A). To further confirm whether CASC2
was a direct target of miR-21, a dual-luciferase reporter assay was
conducted in 786-O cells. Consistent with expectations, the
relative luciferase activity was significantly inhibited by miR-21
in the CASC2-WT vector-transfected cells (P=0.008), whereas miR-21
exhibited no inhibitory effect in cells transfected with the
CASC2-MUT vector (Fig. 3B).
Furthermore, as shown in Fig. 3C,
the RNA expression levels of CASC2 were significantly downregulated
in the cells transfected with miR-21 mimics compared with in the NC
mimics group (P<0.001 and P=0.005 in 786-O and A498 cells,
respectively). These results suggest that CASC2 may be directly
targeted by miR-21 in RCC.
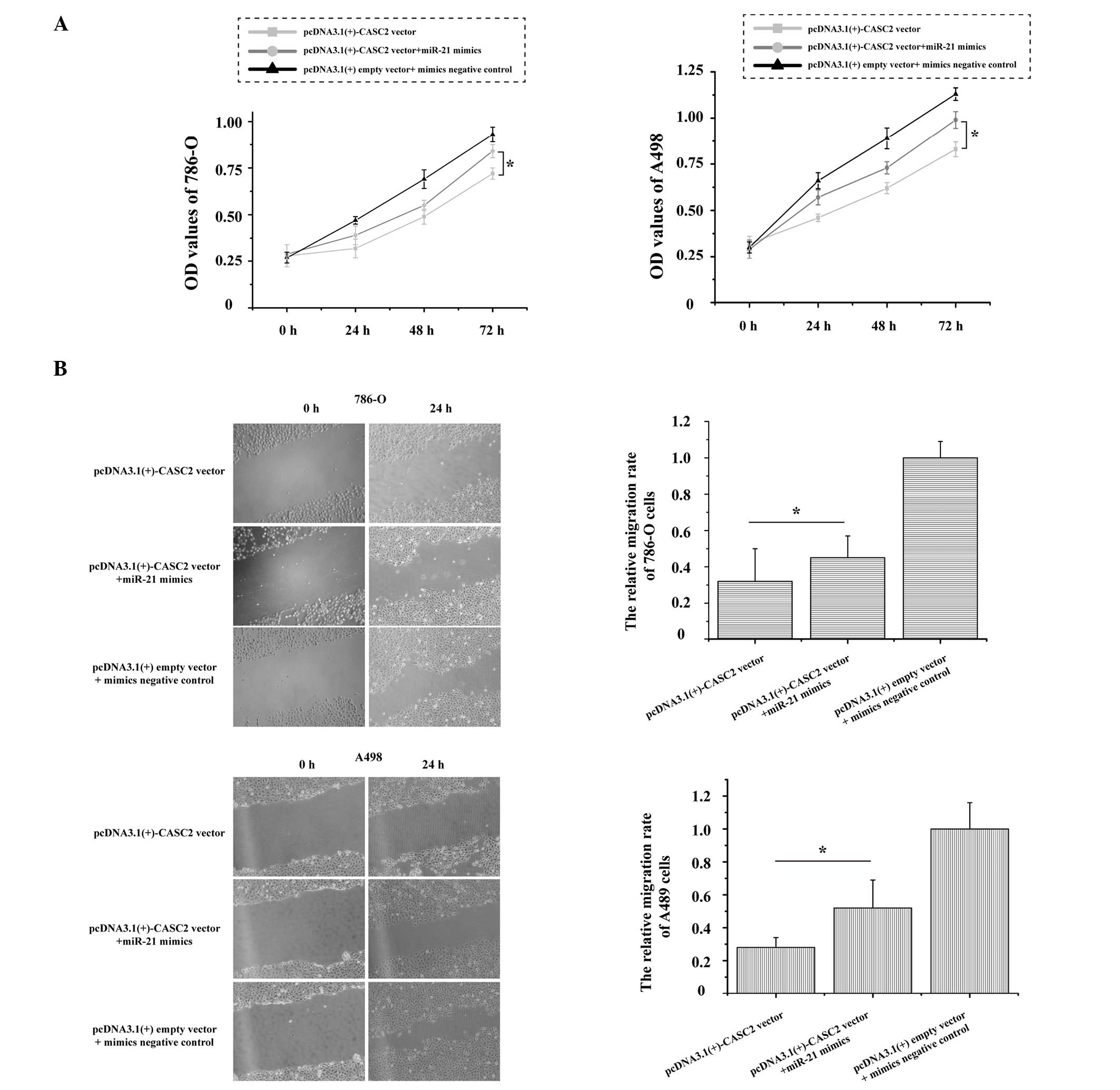
Over-expression of miR-21 partly
abrogates CASC2-induced inhibitory effects on RCC cells
CASC2 was identified as a direct target of miR-21 in
RCC, as demonstrated by dual-luciferase reporter assays. The role
of miR-21 in CASC2-induced inhibition on RCC cells remains unknown.
As presented in Fig. 4A, the
growth of 786-O and A498 cells was significantly increased (P=0.017
and P=0.011, respectively) following transfection with the
pcDNA3.1(+)-CASC2 vector + miR-21 mimics as compared with the
pcDNA3.1(+)-CASC2 vector. Additionally, over-expression of miR-21
and CASC2 could promote 786-O (P=0.042) and A498 (P=0.025) cell
migration when compared with over-expression of CASC2 (Fig. 4B). These results strongly indicate
that over-expression of miR-21 in RCC cells partially reverses the
inhibitory effects of CASC2 on cell proliferation and
migration.
Discussion
Previous studies have demonstrated that the
molecular mechanisms underlying carcinogenesis are not only
associated with protein-coding genes but also with non-coding
regulatory RNAs (14–16). Various lncRNAs have been identified
to have a crucial role in regulating the development of cancer
(17–19). Previous studies have revealed that
several lncRNAs are aberrantly expressed in RCC (20,21).
Furthermore, several lncRNAs have been reported to be involved in
regulating RCC metastasis, including proliferation, migration,
invasion and apoptosis (20,22).
Previous studies have reported that the
downregulation of the lncRNA, CASC2 is associated with numerous
types of carcinoma, including human endometrial cancer and gliomas
(5–7). As a common type of urological cancer,
the expression and function of CASC2 in RCC has not yet, to the
best of our knowledge, been reported. The present study illustrated
the expression and role of CASC2 in RCC development. Consistent
with our hypotheses, the results indicated that the expression
levels of CASC2 were significantly lower in RCC specimens and cell
lines, compared with that in adjacent normal tissue samples and
human HEK 293 cells. Furthermore, restoration of CASC2 expression
in 786-O and A498 cells inhibited cell proliferation and migration.
These results suggested that dysregulation of CASC2 exerts a
suppressive role in RCC development.
In a previous study, miR-21 was demonstrated to be
upregulated in RCC tissues and cell lines (23). miR-21 has been shown to promote
cell transformation, proliferation and metastasis in RCC by
targeting the tumor suppressor programmed cell death protein 4
(24). In addition, miR-21 may
influence RCC cell proliferation by regulating nuclear
factor-κB-mediated cyclin D1 expression (25). In the present study, the results of
the bioinformatics analysis and dual-luciferase reporter assay
confirmed that CASC2 was a direct target gene of miR-21.
Furthermore, upregulated miR-21 expression was able to suppress
CASC2 expression in 786-O and A498 cells, thus suggesting that
CASC2 is a direct target gene of miR-21 in RCC.
To further verify the speculation that miR-21
functions by targeting CASC2 in RCC, synthetic miR-21 mimics were
used to overexpress miR-21 in 786-O and A498 cells. miR-21 was able
to partially reverse pcDNA3.1(+)-CASC2 vector-induced cell
proliferation and migration inhibition. These results suggested
that miR-21 has a key role inhibiting CASC2.
In conclusion, CASC2 acts as a tumor suppressor gene
in RCC. CASC2 is directly targeted by miR-21, and ectopic CASC2
expression inhibits proliferation and migration of RCC cells. The
present study may provide an improved understanding regarding the
role of CASC2 in RCC development, which may be used to develop
lncRNA-directed diagnostics and therapeutics against RCC.
Acknowledgments
The present study was supported by the Changzhou
Health Research Program (grant no. CE20125025).
References
|
1
|
Rini BI, Campbell SC and Escudier B: Renal
cell carcinoma. Lancet. 373:1119–1132. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhang WB, Pan ZQ, Yang QS and Zheng XM:
Tumor suppressive miR-509-5p contributes to cell migration,
proliferation and antiapoptosis in renal cell carcinoma. Ir J Med
Sci. 182:621–627. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Redova M, Poprach A, Besse A, Iliev R,
Nekvindova J, Lakomy R, Radova L, Svoboda M, Dolezel J, Vyzula R
and Slaby O: MiR-210 expression in tumor tissue and in vitro
effects of its silencing in renal cell carcinoma. Tumour Biol.
34:481–491. 2013. View Article : Google Scholar
|
|
5
|
Baldinu P, Cossu A, Manca A, Satta MP,
Sini MC, Rozzo C, Dessole S, Cherchi P, Gianfrancesco F, Pintus A,
et al: Identification of a novel candidate gene, CASC2, in a region
of common allelic loss at chromosome 10q26 in human endometrial
cancer. Hum Mutat. 23:318–326. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Baldinu P, Cossu A, Manca A, Satta MP,
Sini MC, Palomba G, Dessole S, Cherchi P, Mara L, Tanda F and
Palmieri G: CASC2a gene is down-regulated in endometrial cancer.
Anticancer Res. 27:235–243. 2007.PubMed/NCBI
|
|
7
|
Wang P, Liu YH, Yao YL, Li Z, Li ZQ, Ma J
and Xue YX: Long non-coding RNA CASC2 suppresses malignancy in
human gliomas by miR-21. Cell Signal. 27:275–282. 2015. View Article : Google Scholar
|
|
8
|
Yu Z, Chen D, Su Z, Li Y, Yu W, Zhang Q,
Yang L, Li C, Yang S, Ni L, et al: MiR-886-3p upregulation in clear
cell renal cell carcinoma regulates cell migration, proliferation
and apoptosis by targeting PITX1. Int J Mol Med. 34:1409–1416.
2014.PubMed/NCBI
|
|
9
|
Ha TY: MicroRNAs in human diseases: From
cancer to cardiovascular disease. Immune Netw. 11:135–154. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Paraskevopoulou MD, Georgakilas G,
Kostoulas N, Reczko M, Maragkakis M, Dalamagas TM and Hatzigeorgiou
AG: DIANA-LncBase: Experimentally verified and computationally
predicted microRNA targets on long non-coding RNAs. Nucleic Acids
Res. 41(Database Issue): D239–D245. 2013. View Article : Google Scholar :
|
|
11
|
Chen C, Ridzon DA, Broomer AJ, Zhou Z, Lee
DH, Nguyen JT, Barbisin M, Xu NL, Mahuvakar VR, Andersen MR, et al:
Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic
Acids Res. 33:e1792005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cesana M, Cacchiarelli D, Legnini I,
Santini T, Sthandier O, Chinappi M, Tramontano A and Bozzoni I: A
long noncoding RNA controls muscle differentiation by functioning
as a competing endogenous RNA. Cell. 147:358–369. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu XH, Sun M, Nie FQ, Ge YB, Zhang EB,
Yin DD, Kong R, Xia R, Lu KH, Li JH, et al: Lnc RNA HOTAIR
functions as a competing endogenous RNA to regulate HER2 expression
by sponging miR-331-3p in gastric cancer. Mol Cancer. 13:922014.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Qiao HP, Gao WS, Huo JX and Yang ZS: Long
non-coding RNA GAS5 functions as a tumor suppressor in renal cell
carcinoma. Asian Pac J Cancer Prev. 14:1077–1082. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Prensner JR and Chinnaiyan AM: The
emergence of lncRNAs in cancer biology. Cancer Discov. 1:391–407.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li Y and Wang X: Role of long noncoding
RNAs in malignant disease (Review). Mol Med Rep. 13:1463–1469.
2016.
|
|
17
|
Salameh A, Lee AK, Cardó-Vila M, Nunes DN,
Efstathiou E, Staquicini FI, Dobroff AS, Marchiò S, Navone NM,
Hosoya H, et al: PRUNE2 is a human prostate cancer suppressor
regulated by the intronic long noncoding RNA PCA3. Proc Natl Acad
Sci USA. 112:8403–8408. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li T, Xie J, Shen C, Cheng D, Shi Y, Wu Z,
Deng X, Chen H, Shen B, Peng C, et al: Upregulation of long
noncoding RNA ZEB1-AS1 promotes tumor metastasis and predicts poor
prognosis in hepatocellular carcinoma. Oncogene. 35:1575–1584.
2016. View Article : Google Scholar
|
|
19
|
Liang WC, Fu WM, Wong CW, Wang Y, Wang WM,
Hu GX, Zhang L, Xiao LJ, Wan DC, Zhang JF and Waye MM: The lncRNA
H19 promotes epithelial to mesenchymal transition by functioning as
miRNA sponges in colorectal cancer. Oncotarget. 6:22513–22525.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hirata H, Hinoda Y, Shahryari V, Deng G,
Nakajima K, Tabatabai ZL, Ishii N and Dahiya R: Long noncoding RNA
MALAT1 promotes aggressive renal cell carcinoma through Ezh2 and
interacts with miR-205. Cancer Res. 75:1322–1331. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Song S, Wu Z, Wang C, Liu B, Ye X, Chen J,
Yang Q, Ye H, Xu B and Wang L: RCCRT1 is correlated with prognosis
and promotes cell migration and invasion in renal cell carcinoma.
Urology. 84:730.e1–e7. 2014. View Article : Google Scholar
|
|
22
|
Wang L, Cai Y, Zhao X, Jia X, Zhang J, Liu
J, Zhen H, Wang T, Tang X, Liu Y and Wang J: Down-regulated long
non-coding RNA H19 inhibits carcinogenesis of renal cell carcinoma.
Neoplasma. 62:412–418. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lv L, Huang F, Mao H, Li M, Li X, Yang M
and Yu X: MicroRNA-21 is overexpressed in renal cell carcinoma. Int
J Biol Markers. 28:201–207. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li X, Xin S, He Z, Che X, Wang J, Xiao X,
Chen J and Song X: MicroRNA-21 (miR-21) post-transcriptionally
downregulates tumor suppressor PDCD4 and promotes cell
transformation, proliferation and metastasis in renal cell
carcinoma. Cell Physiol Biochem. 33:1631–1642. 2014. View Article : Google Scholar
|
|
25
|
Bera A, Ghosh-Choudhury N, Dey N, Das F,
Kasinath BS, Abboud HE and Choudhury GG: NFκB-mediated cyclin D1
expression by microRNA-21 influences renal cancer cell
proliferation. Cell Signal. 25:2575–2586. 2013. View Article : Google Scholar : PubMed/NCBI
|