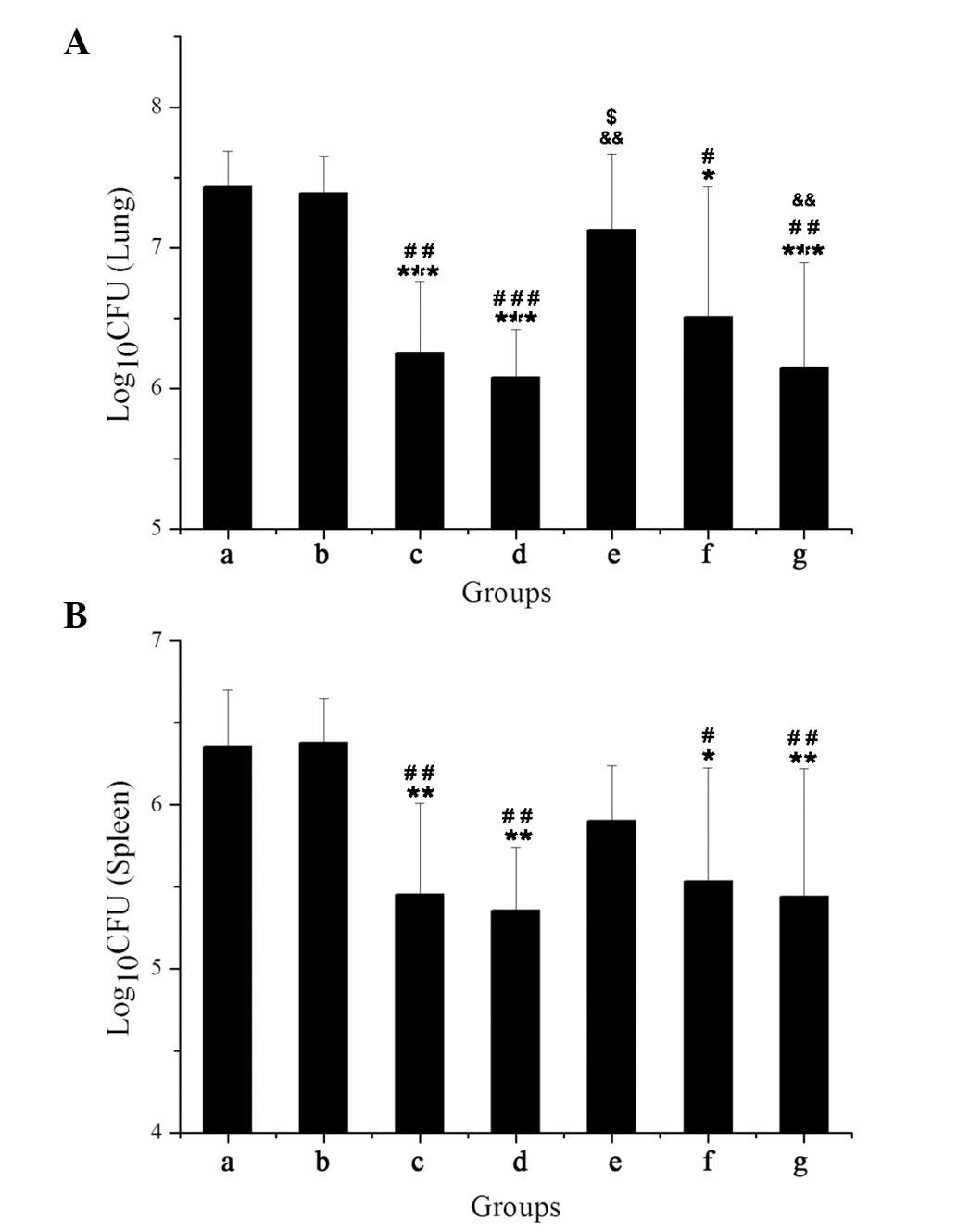
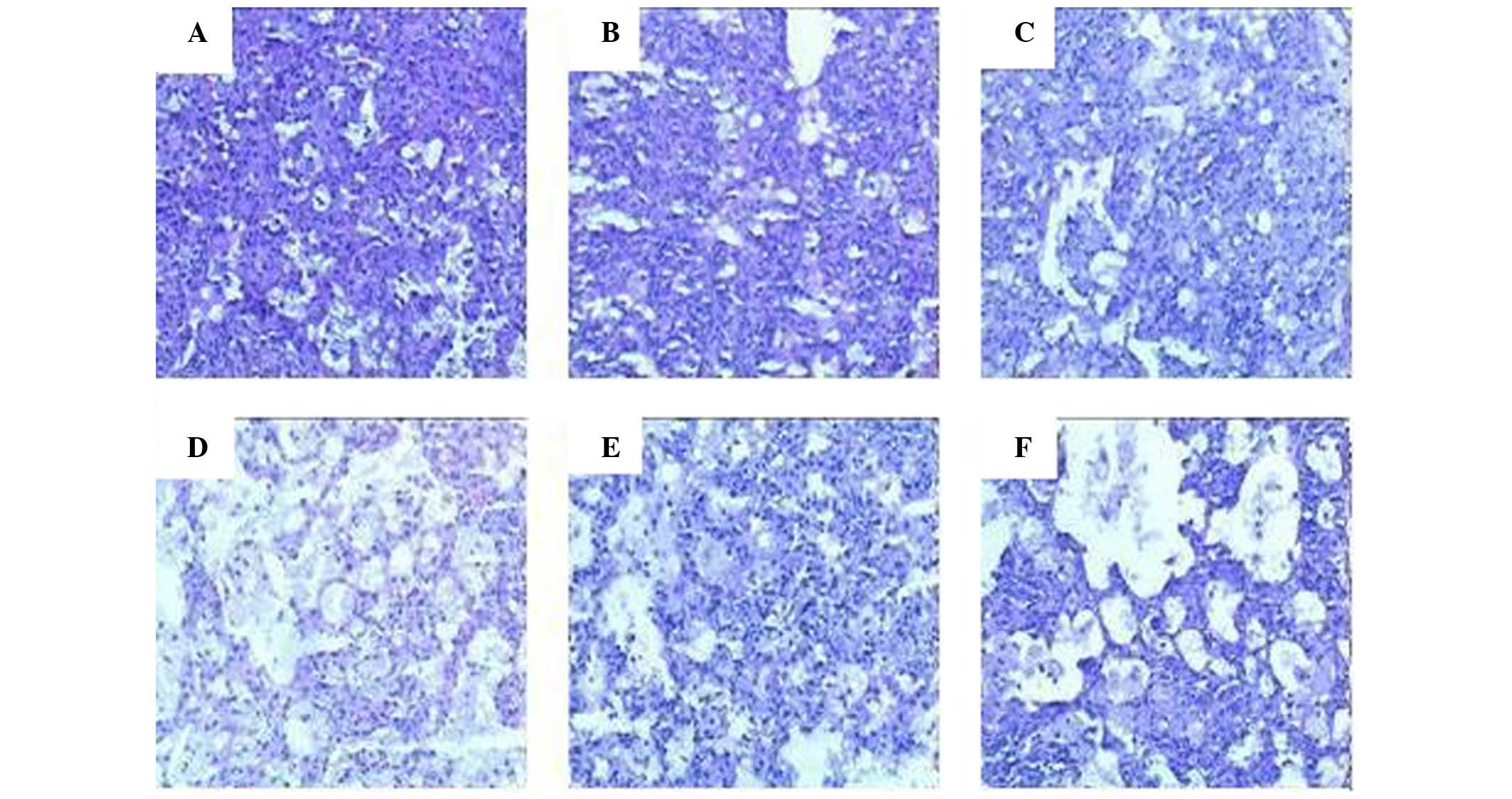
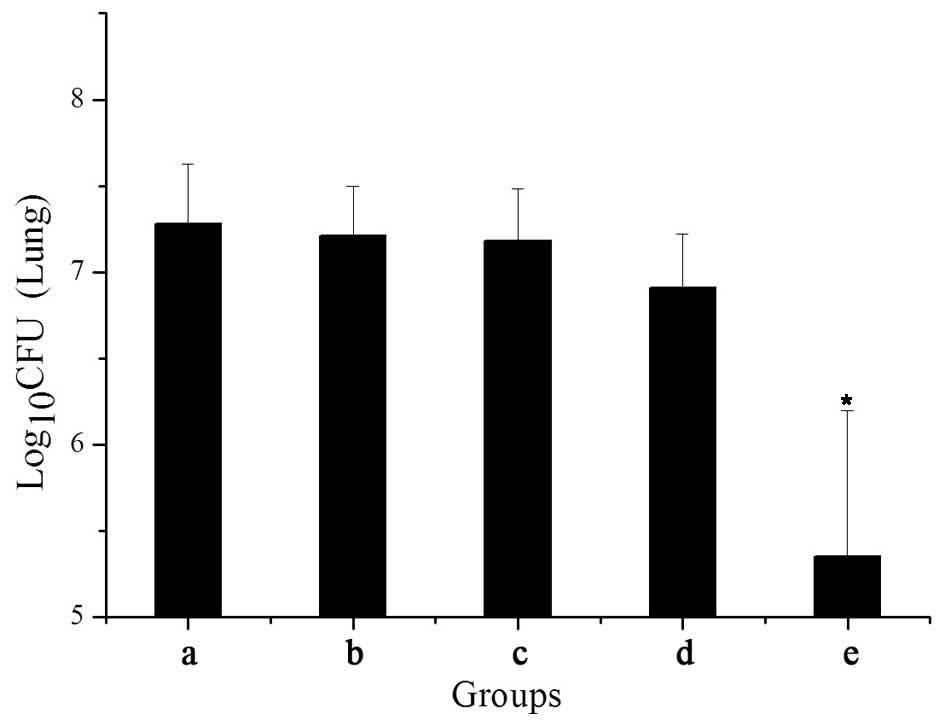
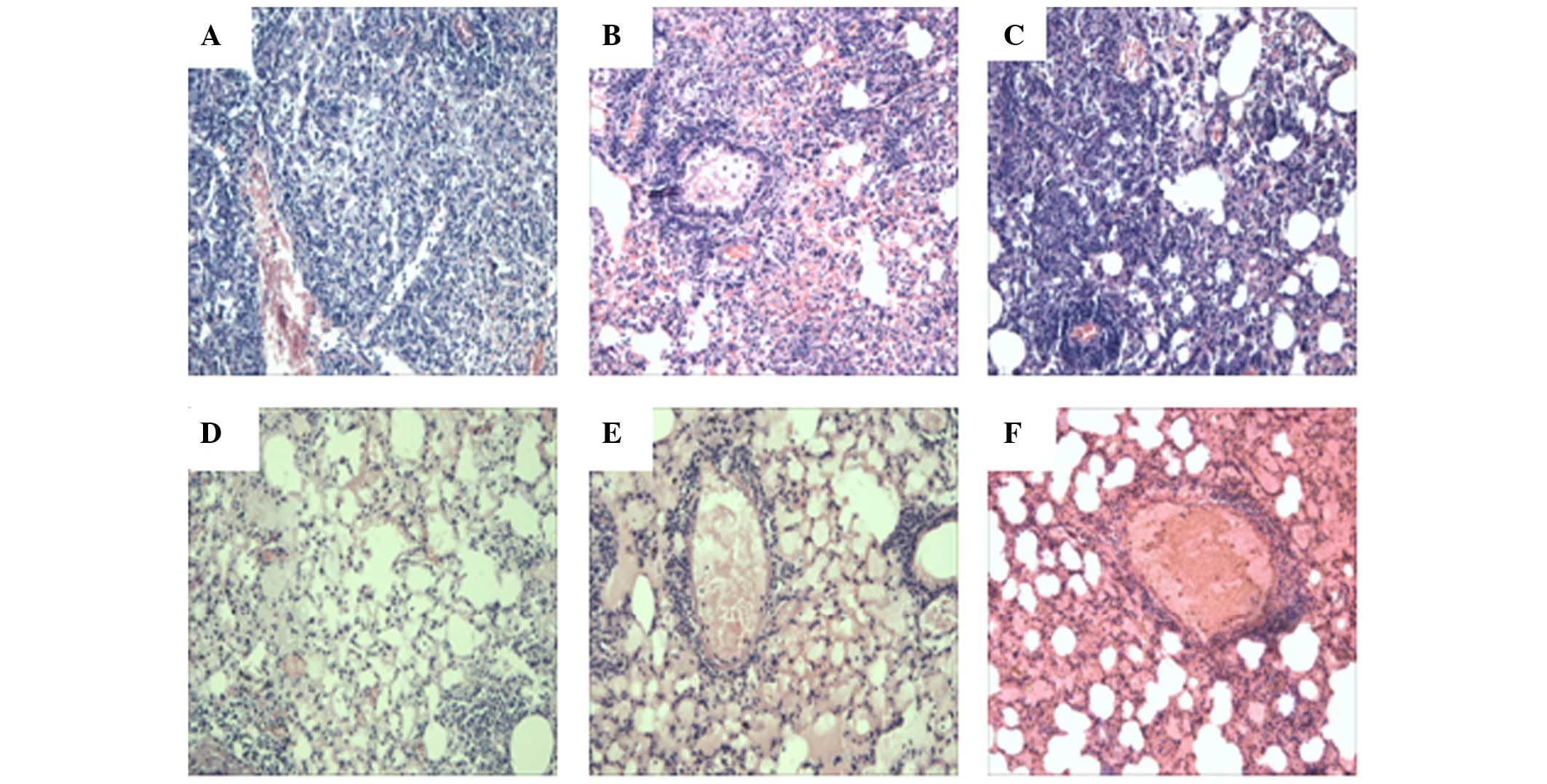
|
1
|
Glaziou P, Falzon D, Floyd K and
Raviglione M: Global epidemiology of tuberculosis. Semin Respir
Crit Care Med. 34:3–16. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
WHO/IUATLD Global Project on
Anti-Tuberculosis Drug Resistance Surveillance: Anti-tuberculosis
drug resistance in the world. (Third global report/the WHO/IUATLD
Global Project on Anti-Tuberculosis Drug Resistance Surveillance).
1999–2002
|
|
3
|
Mori T: MDR-TB - its characteristics and
control in Asia-Pacific rim symposium in USJCMSP 10th international
conference on emerging infectious diseases in the Pacific rim.
Tuberculosis (Edinb). 87(Suppl 1): S5–S9. 2007. View Article : Google Scholar
|
|
4
|
The Office of the Fifth National TB
Epidemiological Survey, Technical Guidance Group of the Fifth
National TB Epidemiological Survey: The Fifth National Tuberculosis
Epidemiological Survey in 2010. Chin J Antitubere. 34:485–508.
2010, 2012.
|
|
5
|
Ha SJ, Jeon BY, Youn JI, Kim SC, Cho SN
and Sung YC: Protective effect of DNA vaccine during chemotherapy
on reactivation and reinfection of Mycobacterium tuberculosis. Gene
Ther. 12:634–638. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Silva CL, Bonato VL, Coelho-Castelo AA, De
Souza AO, Santos SA, Lima KM, Faccioli LH and Rodrigues JM:
Immunotherapy with plasmid DNA encoding mycobacterial hsp65 in
association with chemotherapy is a more rapid and efficient form of
treatment for tuberculosis in mice. Gene Ther. 12:281–287. 2005.
View Article : Google Scholar
|
|
7
|
Ha SJ, Jeon BY, Kim SC, Kim DJ, Song MK,
Sung YC and Cho SN: Therapeutic effect of DNA vaccines combined
with chemotherapy in a latent infection model after aerosol
infection of mice with Mycobacterium tuberculosis. Gene Ther.
10:1592–1599. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhu D, Jiang S and Luo X: Therapeutic
effects of Ag85B and MPT64 DNA vaccines in a murine model of
Mycobacterium tuberculosis. Vaccine. 23:4619–4624. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cooper AM, Dalton DK, Stewart TA, Griffin
JP, Russell DG and Orme IM: Disseminated tuberculosis in interferon
gamma gene-disrupted mice. J Exp Med. 178:2243–2247. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Flynn JL, Chan J, Triebold KJ, Dalton DK,
Stewart TA and Bloom BR: An essential role for interferon gamma in
resistance to Mycobacterium tuberculosis infection. J Exp Med.
178:2249–2254. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lowrie DB, Tascon RE, Bonato VL, Lima VM,
Faccioli LH, Stavropoulos E, Colston MJ, Hewinson RG, Moelling K
and Silva CL: Therapy of tuberculosis in mice by DNA vaccination.
Nature. 400:269–271. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fan X, Gao Q and Fu R: DNA vaccine
encoding ESAT-6 enhances the protective efficacy of BCG against
Mycobacterium tuberculosis infection in mice. Scand J Immunol.
66:523–528. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Harboe M, Oettinger T, Wiker HG,
Rosenkrands I and Andersen P: Evidence for occurrence of the ESAT-6
protein in Mycobacterium tuberculosis and virulent Mycobacterium
bovis and for its absence in Mycobacterium bovis BCG. Infect Immun.
64:16–22. 1996.PubMed/NCBI
|
|
14
|
Brandt L, Oettinger T, Holm A, Andersen AB
and Andersen P: Key epitopes on the ESAT-6 antigen recognized in
mice during the recall of protective immunity to Mycobacterium
tuberculosis. J Immunol. 157:3527–3533. 1996.PubMed/NCBI
|
|
15
|
Andersen P and Heron I: Specificity of a
protective memory immune response against Mycobacterium
tuberculosis. Infect Immun. 61:844–851. 1993.PubMed/NCBI
|
|
16
|
Langermans JA, Doherty TM, Vervenne RA,
van der Laan T, Lyashchenko K, Greenwald R, Agger EM, Aagaard C,
Weiler H, van Soolingen D, et al: Protection of macaques against
Mycobacterium tuberculosis infection by a subunit vaccine based on
a fusion protein of antigen 85B and ESAT-6. Vaccine. 23:2740–2750.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pathan AA, Minassian AM, Sander CR,
Rowland R, Porter DW, Poulton ID, Hill AV, Fletcher HA and McShane
H: Effect of vaccine dose on the safety and immunogenicity of a
candidate TB vaccine, MVA85A, in BCG vaccinated UK adults. Vaccine.
30:5616–5624. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bryder K, Sbai H, Nielsen HV, Corbet S,
Nielsen C, Whalen RG and Fomsgaard A: Improved immunogenicity of
HIV-1 epitopes in HBsAg chimeric DNA vaccine plasmids by structural
mutations of HbsAg. DNA Cell Biol. 18:219–225. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li Z, Song D, Zhang H, He W, Fan X, Zhang
Y, Huang J, Wang X, Liu Q and Xiong S: Improved humoral immunity
against tuberculosis ESAT-6 antigen by chimeric DNA prime and
protein boost strategy. DNA Cell Biol. 25:25–30. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liang Y, Wu X, Zhang J, Li N, Yu Q, Yang
Y, Bai X, Liu C, Shi Y, Liu Q, et al: The treatment of mice
infected with multi-drug-resistant Mycobacterium tuberculosis using
DNA vaccines or in combination with rifampin. Vaccine.
26:4536–4540. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chinese Antituberculosis Association:
Chinese laboratory science procedure of diagnostic bacteriology in
tuberculosis. Wang SM: China Education Culture Press; Beijing: pp.
46–61. 2006
|
|
22
|
Skjøt RL, Oettinger T, Rosenkrands I, Ravn
P, Brock I, Jacobsen S and Andersen P: Comparative evaluation of
low-molecular-mass proteins from Mycobacterium tuberculosis
identifies members of the ESAT-6 family as immunodominant T-cell
antigens. Infect Immun. 68:214–220. 2000. View Article : Google Scholar
|
|
23
|
Wang QM, Sun SH, Hu ZL, Yin M, Xiao CJ and
Zhang JC: Improved immunogenicity of a tuberculosis DNA vaccine
encoding ESAT6 by DNA priming and protein boosting. Vaccine.
22:3622–3627. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Humber DP, Nsanzumuhire H, Aluoch JA,
Webster AD, Aber VR, Mitchison DA, Girling DJ and Nunn AJ:
Controlled double-blind study of the effect of rifampin on humoral
and cellular immune responses in patients with pulmonary
tuberculosis and tuberculosis contacts. Am Rev Respir Dis.
122:425–436. 1980.PubMed/NCBI
|
|
25
|
Ziglam HM, Daniels I and Finch RG:
Immunomodulating activity of rifampicin. J Chemother. 16:357–361.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Chanwong S, Maneekarn N, Makonkawkeyoon L
and Makonkawkeyoon S: Intracellular growth and drug susceptibility
of Mycobacterium tuberculosis in macrophages. Tuberculosis.
87:130–133. 2007. View Article : Google Scholar
|
|
27
|
van Dissel JT, Arend SM, Prins C, Bang P,
Tingskov PN, Lingnau K, Nouta J, Klein MR, Rosenkrands I, Ottenhoff
TH, et al: Ag85B-ESAT-6 adjuvanted with IC31 promotes strong and
long-lived Mycobacterium tuberculosis specific T cell responses in
naïve human volunteers. Vaccine. 28:3571–3581. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Smith J, Manoranjan J, Pan M, Bohsali A,
Xu J, Liu J, McDonald KL, Szyk A, LaRonde-LeBlanc N and Gao LY:
Evidence for pore formation in host cell membranes by
ESX-1-secreted ESAT-6 and its role in Mycobacterium marinum escape
from the vacuole. Infect Immun. 76:5478–5487. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Derrick SC and Morris SL: The ESAT6
protein of Mycobacterium tuberculosis induces apoptosis of
macrophages by activating caspase expression. Cell Microbiol.
9:1547–1555. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
van der Wel N, Hava D, Houben D, Fluitsma
D, van Zon M, Pierson J, Brenner M and Peters PJ: M. tuberculosis
and M. leprae translocate from the phagolysosome to the cytosol in
myeloid cells. Cell. 129:1287–1298. 2007. View Article : Google Scholar : PubMed/NCBI
|