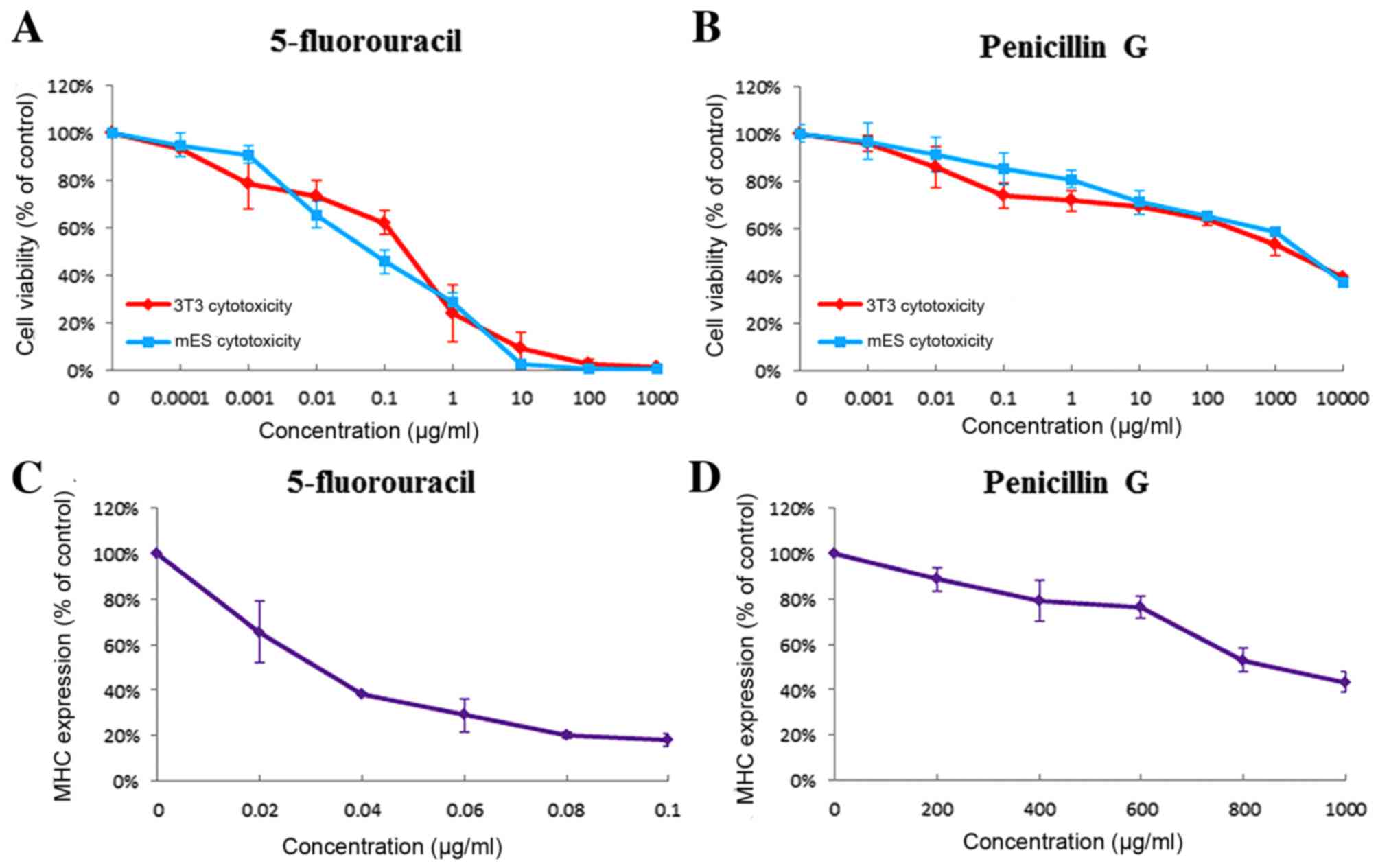
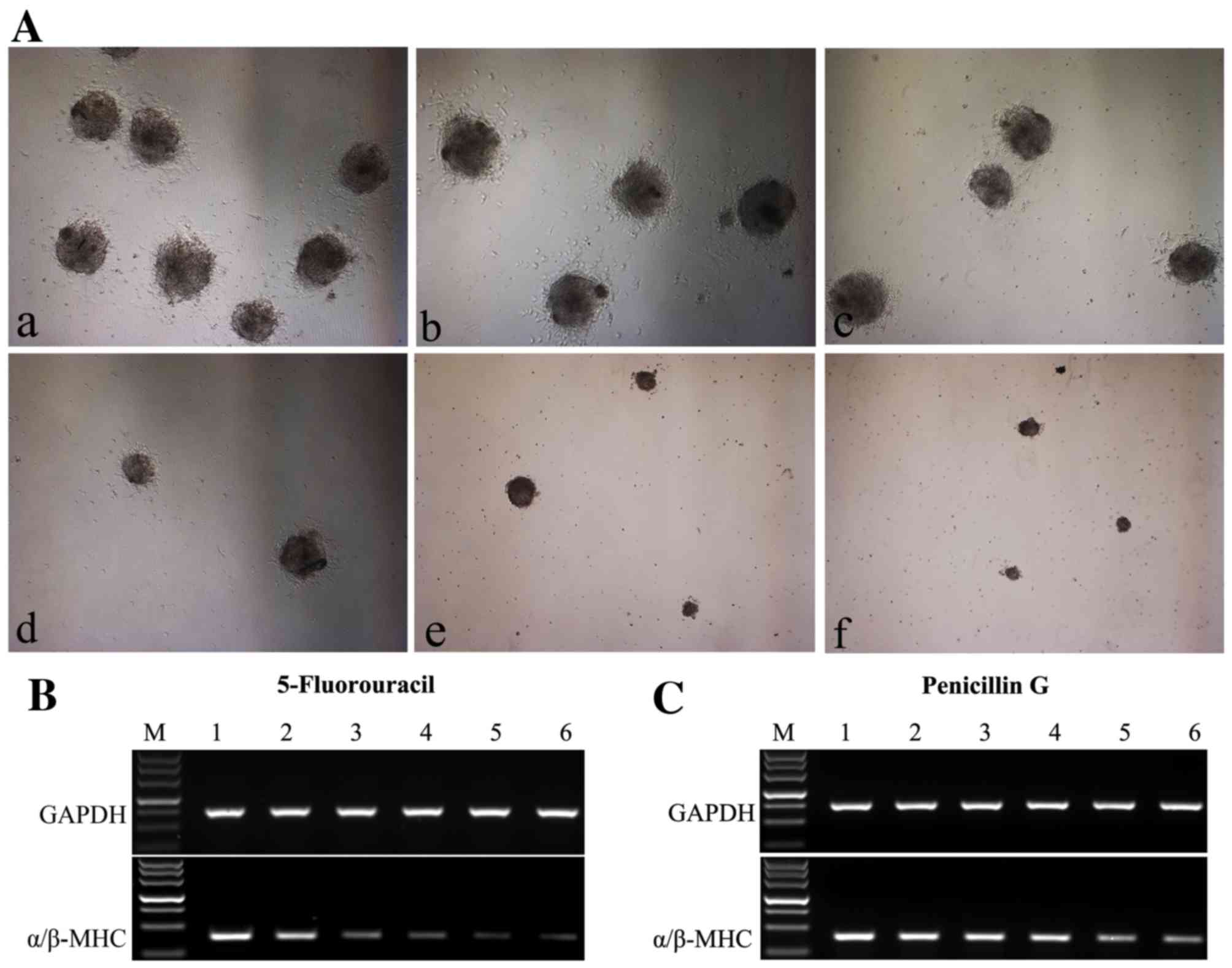
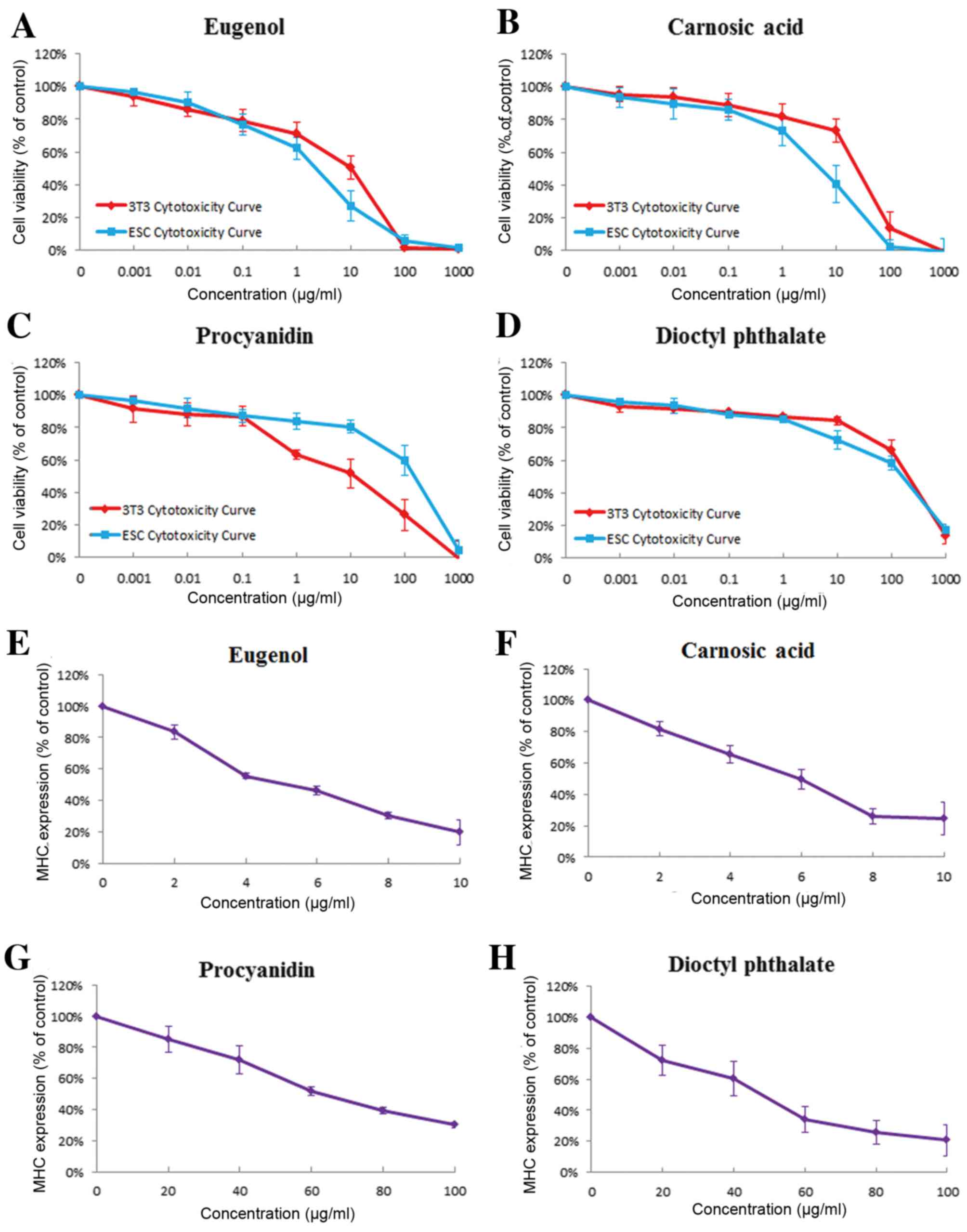
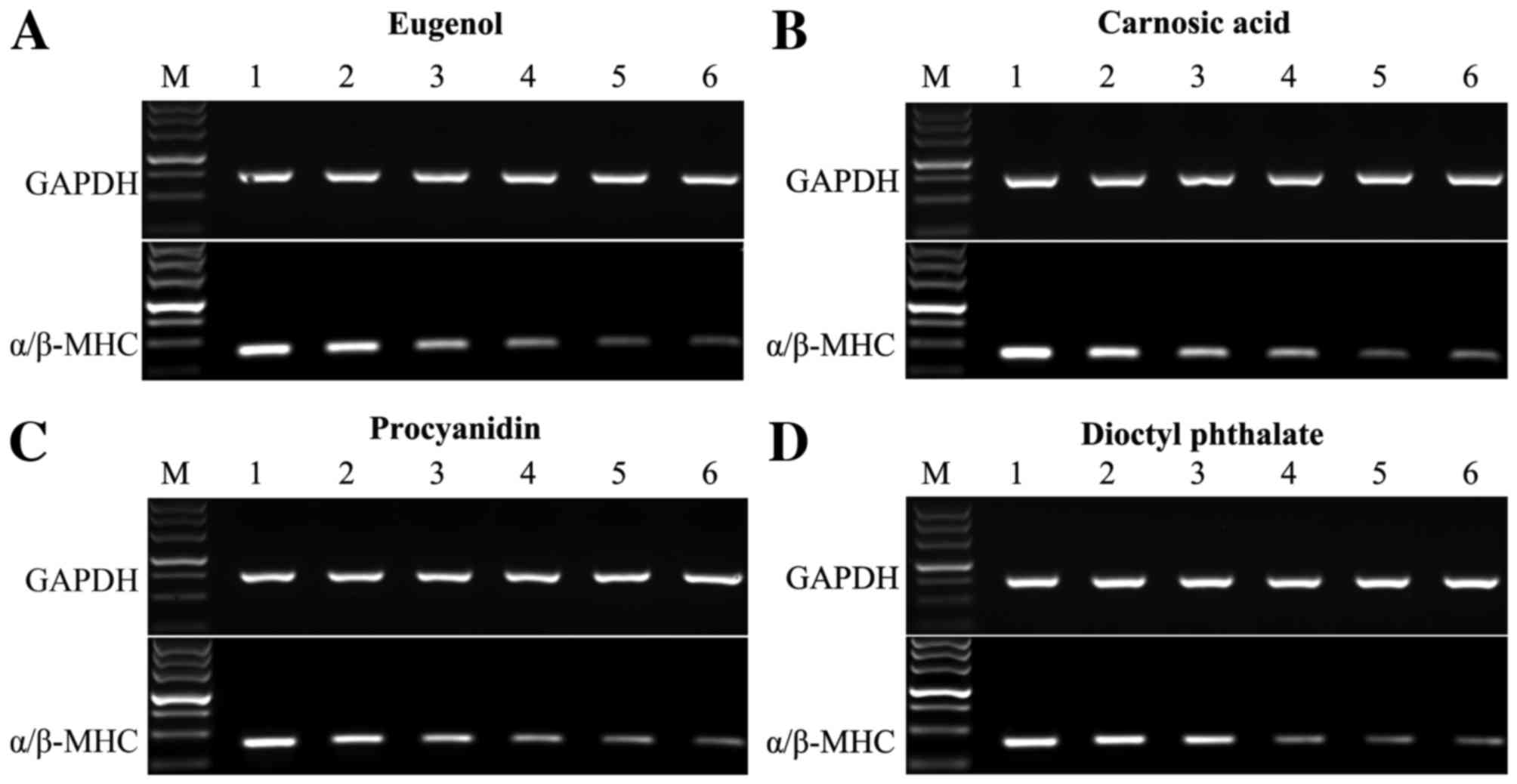
|
1
|
Scholz G, Genschow E, Pohl I, Bremer S,
Paparella M, Raabe H, Southee J and Spielmann H: Prevalidation of
the embryonic stem cell test (EST)-A new in vitro embryotoxicity
Test. Toxicol In Vitro. 13:675–681. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Genschow E, Spielmann H, Scholz G, Pohl I,
Seiler A, Clemann N, Bremer S and Becker K: Validation of the
embryonic stem cell test in the international ECVAM validation
study on three in vitro embryotoxicity tests. Altern Lab Anim.
32:209–244. 2004.PubMed/NCBI
|
|
3
|
Seiler AE and Spielmann H: The validated
embryonic stem cell test to predict embryotoxicity in vitro. Nat
Protoc. 6:961–978. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen R, Chen J, Cheng S, Qin J, Li W,
Zhang L, Jiao H, Yu X, Zhang X, Lahn BT and Xiang AP: Assessment of
embryotoxicity of compounds in cosmetics by the embryonic stem cell
test. Toxicol Mech Methods. 20:112–118. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Festag M, Viertel B, Steinberg P and
Sehner C: An in vitro embryotoxicity assay based on the disturbance
of the differentiation of murine embryonic stem cells into
endothelial cells. II. Testing of compounds. Toxicol In Vitro.
21:1631–1640. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li L, Zhang X, Wang L, Chai Z, Shen X,
Zhang Z and Liu C: A toxicology study to evaluate the
embryotoxicity of metformin compared with the hypoglycemic drugs,
the anticancer drug, the anti-epileptic drug, the antibiotic, and
the cyclo-oxygenase (COX)-2 inhibitor. J Diabetes. 7:839–849. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Paquette JA, Kumpf SW, Streck RD, Thomson
JJ, Chapin RE and Stedman DB: Assessment of the embryonic stem cell
test and application and use in the pharmaceutical industry. Birth
Defects Res B Dev Reprod Toxicol. 83:104–111. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen F, Cao F, Su Z, Li L, Huang A and Xu
H: Assessment of the developmental toxicity of epidermal growth
factor using embryonic stem cell test. Trop J Pharm Res.
13:6892014. View Article : Google Scholar
|
|
9
|
Fuegemann CJ, Samraj AK, Walsh S,
Fleischmann BK, Jovinge S and Breitbach M: Differentiation of mouse
embryonic stem cells into cardiomyocytes via the hanging-drop and
mass culture methods. Curr Protoc Stem Cell Biol Chapter. 1:Unit 1F
11. 2010. View Article : Google Scholar
|
|
10
|
Doetschman TC, Eistetter H, Katz M,
Schmidt W and Kemler R: The in vitro development of
blastocyst-derived embryonic stem cell lines: Formation of visceral
yolk sac, blood islands and myocardium. J Embryol Exp Morphol.
87:27–45. 1985.PubMed/NCBI
|
|
11
|
Sánchez A, Jones WK, Gulick J, Doetschman
T and Robbins J: Myosin heavy chain gene expression in mouse
embryoid bodies. An in vitro developmental study. J Biol Chem.
266:22419–22426. 1991.PubMed/NCBI
|
|
12
|
Robbins J, Gulick J, Sanchez A, Howles P
and Doetschman T: Mouse embryonic stem cells express the cardiac
myosin heavy chain genes during development in vitro. J Biol Chem.
265:11905–11909. 1990.PubMed/NCBI
|
|
13
|
de Jong E, Louisse J, Verwei M, Blaauboer
BJ, van de Sandt JJ, Woutersen RA, Rietjens IM and Piersma AH:
Relative developmental toxicity of glycol ether alkoxy acid
metabolites in the embryonic stem cell test as compared with the in
vivo potency of their parent compounds. Toxicol Sci. 110:117–124.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Deng SQ, Xu H, He Q, Jiang HX, Su BJ and
Zhang QH: Detecting the developmental toxicity of bFGF in the
embryonic stem cell test using differential gene expression of
differentiation-related genes. Toxicol Mech Methods. 24:323–331.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Schulpen SH, Pennings JL, Tonk EC and
Piersma AH: A statistical approach towards the derivation of
predictive gene sets for potency ranking of chemicals in the mouse
embryonic stem cell test. Toxicol Lett. 225:342–349. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
zur Nieden NI, Ruf LJ, Kempka G,
Hildebrand H and Ahr HJ: Molecular markers in embryonic stem cells.
Toxicol In Vitro. 15:455–461. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Nieden NI, Kempka G and Ahr HJ: Molecular
multiple endpoint embryonic stem cell test-a possible approach to
test for the teratogenic potential of compounds. Toxicol Appl
Pharmacol. 194:257–269. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Seiler A, Visan A, Buesen R, Genschow E
and Spielmann H: Improvement of an in vitro stem cell assay for
developmental toxicity: The use of molecular endpoints in the
embryonic stem cell test. Reprod Toxicol. 18:231–240. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Genschow E, Scholz G, Brown N, Piersma A,
Brady M, Clemann N, Huuskonen H, Paillard F, Bremer S, Becker K and
Spielmann H: Development of prediction models for three in vitro
embryotoxicity tests in an ECVAM validation study. In Vitr Mol
Toxicol. 13:51–66. 2000.PubMed/NCBI
|
|
20
|
Buesen R, Genschow E, Slawik B, Visan A,
Spielmann H, Luch A and Seiler A: Embryonic stem cell test
remastered: Comparison between the validated EST and the new
molecular FACS-EST for assessing developmental toxicity in vitro.
Toxicol Sci. 108:389–400. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jadhav BK, Khandelwal KR, Ketkar AR and
Pisal SS: Formulation and evaluation of mucoadhesive tablets
containing eugenol for the treatment of periodontal diseases. Drug
Dev Ind Pharm. 30:195–203. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chami N, Chami F, Bennis S, Trouillas J
and Remmal A: Antifungal treatment with carvacrol and eugenol of
oral candidiasis in immunosuppressed rats. Braz J Infect Dis.
8:217–226. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Dip EC, Pereira NA and Fernandes PD:
Ability of eugenol to reduce tongue edema induced by Dieffenbachia
picta Schott in mice. Toxicon. 43:729–735. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Fischedick JT, Standiford M, Johnson DA
and Johnson JA: Structure activity relationship of phenolic
diterpenes from Salvia officinalis as activators of the
nuclear factor E2-related factor 2 pathway. Bioorg Med Chem.
21:2618–2622. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bai N, He K, Roller M, Lai CS, Shao X, Pan
MH and Ho CT: Flavonoids and phenolic compounds from Rosmarinus
officinalis. J Agric Food Chem. 58:5363–5367. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yu YM, Lin CH, Chan HC and Tsai HD:
Carnosic acid reduces cytokine-induced adhesion molecules
expression and monocyte adhesion to endothelial cells. Eur J Nutr.
48:101–106. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mengoni ES, Vichera G, Rigano LA,
Rodriguez-Puebla ML, Galliano SR, Cafferata EE, Pivetta OH, Moreno
S and Vojnov AA: Suppression of COX-2, IL-1β and TNF-α expression
and leukocyte infiltration in inflamed skin by bioactive compounds
from Rosmarinus officinalis L. Fitoterapia. 82:414–421.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Xiang Q, Liu Z, Wang Y, Xiao H, Wu W, Xiao
C and Liu X: Carnosic acid attenuates lipopolysaccharide-induced
liver injury in rats via fortifying cellular antioxidant defense
system. Food Chem Toxicol. 53:1–9. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ibarra A, Cases J, Roller M, Chiralt-Boix
A, Coussaert A and Ripoll C: Carnosic acid-rich rosemary
(Rosmarinus officinalis L.)leaf extract limits weight gain
and improves cholesterol levels and glycaemia in mice on a high-fat
diet. Br J Nutr. 106:1182–1189. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Birtic S, Dussort P, Pierre FX, Bily AC
and Roller M: Carnosic acid. Phytochemistry. 115:9–19. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Santos-Buelga C and Scalbert A:
Proanthocyanidins and tannin-like compounds-nature, occurrence,
dietary intake and effects on nutrition and health. J Sci Food
Agriculture. 80:1094–1117. 2000. View Article : Google Scholar
|
|
32
|
Monagas M, Quintanilla-López JE,
Gómez-Cordovés C, Bartolomé B and Lebrón-Aguilar R: MALDI-TOF MS
analysis of plant proanthocyanidins. J Pharm Biomed Anal.
51:358–372. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yamakoshi J, Saito M, Kataoka S and
Kikuchi M: Safety evaluation of proanthocyanidin-rich extract from
grape seeds. Food Chem Toxicol. 40:599–607. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Corder R, Mullen W, Khan NQ, Marks SC,
Wood EG, Carrier MJ and Crozier A: Oenology: Red wine procyanidins
and vascular health. Nature. 444:5662006. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Benson R: Hazard to the developing male
reproductive system from cumulative exposure to phthalate
esters-dibutyl phthalate, diisobutyl phthalate, butylbenzyl
phthalate, diethylhexyl phthalate, dipentyl phthalate, and
diisononyl phthalate. Regul Toxicol Pharmacol. 53:90–101. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kavlock R, Boekelheide K, Chapin R,
Cunningham M, Faustman E, Foster P, Golub M, Henderson R, Hinberg
I, Little R, et al: NTP center for the evaluation of risks to human
reproduction: Phthalates expert panel report on the reproductive
and developmental toxicity of di(2-ethylhexyl) phthalate. Reprod
Toxicol. 16:529–653. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Helal MA: Celery oil modulates
DEHP-induced reproductive toxicity in male rats. Reprod Biol.
14:182–189. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Latini G: Monitoring phthalate exposure in
humans. Clin Chim Acta. 361:20–29. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Li G, Ren C, Shi J, Huang W, Liu H, Feng
X, Liu W, Zhu B, Zhang C, Wang L, et al: Identification, expression
and subcellular localization of ESRG. Biochem Biophys Res Commun.
435:160–164. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
van Dartel DA and Piersma AH: The
embryonic stem cell test combined with toxicogenomics as an
alternative testing model for the assessment of developmental
toxicity. Reprod Toxicol. 32:235–244. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hartung T: Toxicology for the twenty-first
century. Nature. 460:208–212. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Rovida C and Hartung T: Re-evaluation of
animal numbers and costs for in vivo tests to accomplish REACH
legislation requirements for chemicals-a report by the
transatlantic think tank for toxicology (t(4)). ALTEX. 26:187–208.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Genschow E, Spielmann H, Scholz G, Seiler
A, Brown N, Piersma A, Brady M, Clemann N, Huuskonen H, Paillard F,
et al: The ECVAM international validation study on in vitro
embryotoxicity tests: Results of the definitive phase and
evaluation of prediction models. European centre for the validation
of alternative methods. Altern Lab Anim. 30:151–176.
2002.PubMed/NCBI
|
|
44
|
Peters AK, Steemans M, Hansen E, Mesens N,
Verheyen GR and Vanparys P: Evaluation of the embryotoxic potency
of compounds in a newly revised high throughput embryonic stem cell
test. Toxicol Sci. 105:342–350. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Jones KL, Lacro RV, Johnson KA and Adams
J: Pattern of malformations in the children of women treated with
carbamazepine during pregnancy. N Engl J Med. 320:1661–1666. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Domaracky M, Rehák P, Juhás S and Koppel
J: Effects of selected plant essential oils on the growth and
development of mouse preimplantation embryos in vivo. Physiol Res.
56:97–104. 2007.PubMed/NCBI
|
|
47
|
George JD, Price CJ, Marr MC, Myers CB and
Jahnke GD: Evaluation of the developmental toxicity of isoeugenol
in Sprague-Dawley (CD) rats. Toxicol Sci. 60:112–120. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wang QL, Li H, Li XX, Cui CY, Wang R, Yu
NX and Chen LX: Acute and 30-day oral toxicity studies of
administered carnosic acid. Food Chem Toxicol. 50:4348–4355. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ray S, Bagchi D, Lim PM, Bagchi M, Gross
SM, Kothari SC, Preuss HG and Stohs SJ: Acute and long-term safety
evaluation of a novel IH636 grape seed proanthocyanidin extract.
Res Commun Mol Pathol Pharmacol. 109:165–197. 2001.PubMed/NCBI
|
|
50
|
Lluís L, Muñoz M, Nogués MR,
Sánchez-Martos V, Romeu M, Giralt M, Valls J and Solà R: Toxicology
evaluation of a procyanidin-rich extract from grape skins and
seeds. Food Chem Toxicol. 49:1450–1454. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Hazelton WI: A subchronic (4-week) dietary
oral toxicity study of di(2-ethylhexyl) phthalate in B6FC3F1 mice.
8D submission, microfische no. OTS0535433: Submited to US EPA.
Office of Toxic Substances, prepared for Eastman Kodak Company
Washington, DC: 1992
|
|
52
|
Poon R, Lecavalier P, Mueller R, Valli VE,
Procter BG and Chu I: Subchronic oral toxicity of di-n-octyl
phthalate and di(2-Ethylhexyl) phthalate in the rat. Food Chem
Toxicol. 35:225–239. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
David RM, Moore MR, Cifone MA, Finney DC
and Guest D: Chronic peroxisome proliferation and hepatomegaly
associated with the hepatocellular tumorigenesis of
di(2-ethylhexyl)phthalate and the effects of recovery. Toxicol Sci.
50:195–205. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Li SG, Huang X, Zhang XW and Xu SH:
Effects of diethylhexyl phthalate on lipid peroxidation and the
life-span in Drosophila melanogaster. Zhonghua Yu Fang Yi
Xue Za Zhi. 39:111–114. 2005.(In Chinese). PubMed/NCBI
|
|
55
|
Tyl R, Jones-Price C, Marr MC and Kimmel
CA: Teratological evaluation of diethylhexylphthalate (CAS no.
117-81-7) in CD-1 mice. National Center for Toxicological Research.
1984.
|
|
56
|
Tyl RW, Price CJ, Marr MC and Kimmel CA:
Developmental toxicity evaluation of dietary
di(2-ethylhexyl)phthalate in Fischer 344 rats and CD-1 mice. Fundam
Appl Toxicol. 10:395–412. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Peters JM, Taubeneck MW, Keen CL and
Gonzalez FJ: Di(2-ethylhexyl) phthalate induces a functional zinc
deficiency during pregnancy and teratogenesis that is independent
of peroxisome proliferator-activated receptor-alpha. Teratology.
56:311–316. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Dobrzynska MM, Tyrkiel EJ, Derezińska E,
Pachocki KA and Ludwicki JK: Two generation reproductive and
developmental toxicity following subchronic exposure of pubescent
male mice to di(2-ethylhexyl)phthalate. Ann Agric Environ Med.
19:31–37. 2012.PubMed/NCBI
|
|
59
|
Erkekoglu P, Zeybek ND, Giray B, Asan E,
Arnaud J and Hincal F: Reproductive toxicity of di(2-ethylhexyl)
phthalate in selenium-supplemented and selenium-deficient rats.
Drug Chem Toxicol. 34:379–389. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Zhang XF, Zhang LJ, Li L, Feng YN, Chen B,
Ma JM, Huynh E, Shi QH, De Felici M and Shen W: Diethylhexyl
phthalate exposure impairs follicular development and affects
oocyte maturation in the mouse. Environ Mol Mutagen. 54:354–361.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Zhang XF, Zhang T, Han Z, Liu JC, Liu YP,
Ma JY, Li L and Shen W: Transgenerational inheritance of ovarian
development deficiency induced by maternal diethylhexyl phthalate
exposure. Reprod Fertil Dev. 27:1213–1221. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Hauser R and Calafat AM: Phthalates and
human health. Occup Environ Med. 62:806–818. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Swan SH: Environmental phthalate exposure
in relation to reproductive outcomes and other health endpoints in
humans. Environ Res. 108:177–184. 2008. View Article : Google Scholar : PubMed/NCBI
|