Introduction
Diabetes mellitus (DM) is characterized by elevated
blood glucose levels (hyperglycemia) and has been identified as the
most widespread metabolic disease worldwide (1). It is estimated that >300 million
people are suffering from DM and this number is expected to reach
600 million by 2030 (2). The
rapidly increasing incidence of DM is one of the most challenging
threats to public health in the 21st century. Despite some
achievements for DM treatment, a variety of complications persist,
such as endothelial dysfunction and retinopathy (3,4).
Therefore, a better understanding of the mechanisms of DM
pathogenesis is of great clinical significance.
Pancreatic β cell dysfunction has been reported to
be the key process in the development and progression of DM; these
cells are particularly sensitive to the glucose concentration under
diabetic conditions (5–7). Hyperglycemia serves a direct role in
inducing pancreatic β cell apoptosis and decreasing β cell
proliferation (8). Dysfunctional β
cells result in low insulin secretion and subsequently accelerated
development of DM (9). Therefore,
it is vital to maintain a normal status of β cells and to protect β
cell function, which will be important therapeutic targets for the
treatment of DM (10,11). Previous studies have focused on how
to protect and improve functional β cells to regulate glucose
homeostasis in diabetes (12,13);
however, the exact mechanisms underlying glucose-induced pancreatic
β cell apoptosis and dysfunction remain largely unknown.
MicroRNAs (miRNAs) are a large class of small,
non-coding single-stranded RNAs that have been demonstrated to be
involved in the pathogenesis of many diseases, including cancers
(14), bacterial and viral
infections (15,16) and diabetes (17). In addition, alterations in miRNA
expression exert regulatory functions in posttranscriptional
modification or degradation of their target genes by binding to
complementary regions in the 3′-untranslated region (UTR) of their
target mRNA transcripts (18,19).
Previous studies have demonstrated that miRNAs play a crucial role
in pancreatic development and function. For example, miRNA (miR)-7
was the first to be reported to negatively regulate insulin
secretion by β cells (20), and
miR-375 was revealed to negatively regulate glucose-stimulated
insulin secretion by regulating the expression of myotrophin and
phosphoinositide-dependent protein kinase 1 (21,22).
In addition, miR-96 was demonstrated to inhibit insulin secretion
by targeting the expression of the Rab GTPase effector Noc2
(23). Other miRNAs, such as
miR-7a, were also reported to potentially contribute to β cell
expansion during pancreatic organogenesis (24). In addition to these miRNAs, miR-433
expression was revealed to be downregulated in the visceral adipose
tissues of patients with non-alcoholic steatohepatitis (25) and may act as a suppressor in human
gastric carcinoma (26).
Furthermore, miRNA profiling revealed miR-433 was downregulated in
insulin 2-mutant diabetic Akita mice (27), suggesting that it may be closely
related with diabetes pathogenesis.
The present study aimed to elucidate the biological
functions and molecular target of miR-433 in pancreatic β cells and
to examine its role in a high-glucose environment. The results
indicated that miR-433 may be able to protect mouse pancreatic β
cells against high-glucose-induced impairment of cell viability by
directly targeting cyclooxygenase 2 [COX2; also known as
prostaglandin-endoperoxide synthase 2 (PTGS2)] transcripts.
Restoration of miR-433 expression may provide preliminary evidence
for the clinical management of DM.
Materials and methods
Cell culture and glucose
treatment
Min-6 mouse pancreatic β cells and HEK293T cells
were purchased from the Cell Bank of Type Culture Collection of the
Chinese Academy of Sciences (Shanghai, China). All cells were
cultured in Dulbecco's Modified Eagle's Medium (DMEM; HyClone; GE
Healthcare Life Sciences, Logan, UT, USA) supplemented with 10%
fetal bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA). Cells were incubated in a humidified incubator
at 37°C under 5% CO2 atmosphere. The medium was replaced
every other day.
Min-6 cells were treated with either a low
concentration of glucose (5 mmol/l; Sigma-Aldrich; Merck KGaA,
Darmstadt, Germany) or a high concentration of glucose (11 mmol/l;
the high-glucose group) for 24 h. The high-glucose group was
further divided for subsequent miR-433 overexpression analysis, as
detailed below.
Transient transfection with miRNA
mimics or siRNA
Mouse miR-433 mimics and negative controls (NC), and
COX2 short interfering (si)RNA (siCOX) and siRNA negative control
were obtained from Shanghai GenePharma Co., Ltd. (Shanghai, China).
The sequences were as follows: miR-433 mimic, sense
5′-AUCAUGAUGGGCUCCUCGGUGU-3′; siCOX, sense
5′-GACCCAAGCAUGUUAUAAAUU-3′; siRNA-NC and mimics-NC,
5′-CAGUACUUUGUGUAGUACAA-3′. For transfection, Min-6 cells
(2×105 cells/well) were cultured in a 6-well plate and
transiently transfected at ~80% confluence with 50 nM siCOX and NC
for 48 h at 37°C using Lipofectamine 2000 reagent (cat. no. 52887;
Invitrogen; Thermo Fisher Scientific, Inc.), according to the
manufacturer's protocol.
In addition, according to the manufacturer's
protocol, Min-6 cells (2×105 cells/well) seeded in
6-well plates were transfected with 200 µl mature miR-433 mimic
(100 nM) and NC (100 nM) for 48 h at 37°C using Lipofectamine 2000
reagent (Invitrogen; Thermo Fisher Scientific, Inc.) for 72 h.
Successful transfection was confirmed using reverse
transcription-quantitative polymerase chain reaction (RT-qPCR). The
primers used were as follows: miR-433, forward
5′-GGGATCATGATGGGCTCCTCG-3′ and reverse
5′-CTCAACTGGTGTCGTGGA-3′.
RNA isolation and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was isolated from cells (2×106)
using the mirVana miRNA Isolation kit (Ambion; Thermo Fisher
Scientific, Inc.), according to the manufacture's protocol. RNA
purity was measured photometrically using a NanoDrop
spectrophotometer (Thermo Fisher Scientific, Inc., Wilmington, DE,
USA). The optical density (OD)260/280 ratio was used as an
indicator for RNA purity. A ratio >1.8 was assumed as suitable
for gene expression measurements. Total RNA was reverse transcribed
into cDNA using the RevertAid First Strand cDNA Synthesis kit (cat
no. K1622; Thermo Fisher Scientific, Inc.) according to the
manufacturer's protocol. For miRNA expression analysis, 200 ng of
total RNA were isolated from Min-6 and HEK293T cells and reverse
transcribed into cDNA using the RevertAid First Strand cDNA
Synthesis kit (Thermo Fisher Scientific, Inc.). qPCR was performed
using SYBR Green PCR Master Mix (Applied Biosystems; Thermo Fisher
Scientific, Inc.) on the on the ABI HT9600 system (Applied
Biosystems; Thermo Fisher Scientific, Inc.). The reaction volume
was 10 µl and contained the following: 5 µl SYBR-Green PCR Master
Mix, 10 µM (0.4 µl) of each forward and reverse primer, 0.2 µl ROX
reference dye, 1 µl cDNA template and 3 µl ddH2O.
Thermocycling conditions were as follows: Initial 1 step at 95°C
for 10 min, followed by 40 cycles at 95°C for 15 sec and at 60°C
for 60 sec. Relative gene expression was calculated using the
comparative Cq method (28), and
miR-433, miR-199-5p, miR-30a and miR-22 expression was normalized
to U6, whereas COX2 expression was normalized to β-actin. The
following primers were in the present study: miR-433, forward
5′-GGGATCATGATGGGCTCCTCG-3′; miR-199-5p, forward
5′-CTGGGCCCAGTGTTCAGACTACCT-3′; miR-30a, forward
5′-GCTGGGTGTAAACATCCTCG-3′; and miR-22, forward
5′-GCTGGGAAGCTGCCAGTTGAAG-3′; each forward primer was used in
combination with the universal miRNA reverse primer,
5′-CTCAACTGGTGTCGTGGA-3′; COX2, forward 5′-AGATGACTGCCCAACTCCCA-3′,
reverse 5′-TGAACCCAGGTCCTCGCTTA-3′; U6, forward
5′-CTCGCTTCGGCAGCACA-3′, reverse 5′-AACGCTTCACGAATTTGCGT-3′; and
β-actin, forward 5′-CATTGCTGACAGGATGCAGA-3′ and reverse
5′-CTGCTGGAAGGTGGACAGTGA-3′.
Western blot analysis
Total proteins were isolated from cells
(2×106) using radioimmunoprecipitation assay lysis
buffer (Sigma-Aldrich; Merck KGaA) supplemented with a protease
inhibitor cocktail (cat no. P8340; Sigma-Aldrich; Merck KGaA) for 5
min on ice, and then centrifuged at 13,000 × g for 20 min at 4°C.
Protein concentration was determined using a bicinchoninic acid
assay kit (Bio-Rad Laboratories, Inc., Hercules, CA, USA),
according to the manufacturer's protocol. Equal amounts of
extracted protein samples (30 µg) were separated by 10% SDS-PAGE
and transferred onto polyvinylidene difluoride membranes, which
were then blocked for 2 h with 5% non-fat dry milk and incubated at
4°C overnight with primary antibodies against COX2 (cat no. 12282;
1:1,000; Cell Signaling Technology, Inc., Danvers, MA, USA) or
GAPDH (cat no. 5174; 1:1,000; Cell Signaling Technology, Inc.).
Membranes were then incubated for 1 h at room temperature with the
following horseradish peroxidase-conjugated secondary antibodies:
goat anti-mouse immunoglobulin (Ig) G (cat no. SC-2005; 1:1,000;
Santa Cruz Biotechnology, Inc., Dallas, TX, USA) and goat
anti-rabbit IgG (cat no. SC-2004; 1:1,000; Santa Cruz
Biotechnology, Inc.). Protein bands were visualized using the
Enhanced Chemiluminescence Western Blotting Analysis System
(Pierce; Thermo Fisher Scientific, Inc.). Data were analyzed using
the Image Lab software version 4.1 (Bio-Rad Laboratories,
Inc.).
Dual luciferase assays
StarBASE software version 2.0 (http://starbase.sysu.edu.cn/panCancer.php) was used to
identify potential target genes for miR-433; results indicated that
the COX2 gene contained a potential miR-433 binding site in its
3′-UTR. To confirm whether miR-433 directly binds to COX2 3′-UTR, a
dual luciferase assay was performed. Genomic DNA was extracted
using the QIAamp DNA Mini kit (Qiagen, Inc., Valencia, CA, USA)
according to the manufacturer's protocol. The COX2 3′-UTR sequence
(length, 779 bp) was amplified by PCR from genomic DNA using the
GoTaq® Long PCR Master Mix (Promega Corporation,
Madison, WI, USA). The following primers were used: COX2 3′UTR,
forward 5′-CCCTCGAGTCATTCCTCTACATAAGCCAGTG-3′ and reverse
5′-ATTTGCGGCCGCGAGTACCAGGCCAGCACAA-3′. Thermocycling conditions
were as follows: Pre-denaturation at 95°C for 2 min, followed by 30
cycles of denaturation at 95°C for 30 sec, annealing at 60°C for 30
sec and extension at 72°C for 45 sec, and a final extension step at
72°C for 5 min. COX2 3′-UTR mutants were generated using the
Site-Directed Mutagenesis kit (Yeasen Biological Technology Co.,
Ltd., Shanghai, China), according to the manufacturer's protocol.
Following amplification, the PCR products were digested with XbaI
and KpnI restriction enzymes (New England BioLabs, Inc., Ipswich,
MA, USA) for at least 3 h and purified using the QIAquick PCR
purification kit (Qiagen, Inc.) according to the manufacturer's
protocol. The purified sequences were then inserted into a
psiCHECK-2 reporter plasmid (Guangzhou RiboBio Co., Ltd.,
Guangzhou, China). The purified vector and PCR products were
ligated overnight at 16°C using T4 DNA ligase (1 µl, 400 U/µl; cat
no. EL0012; Thermo Fisher Scientific, Inc.), at a ratio of 1:3 in 2
µl 10X ligation buffer. The mutation in the miR-433 binding site of
the COX2 3′-UTR was confirmed by Shanghai Shenggong Biology
Engineering Technology Service, Ltd., Shanghai, China). For the
luciferase assay, HEK293T cells at a density of 5×104
cells/well were seeded in 24-well plates and transfected with the
luciferase reporter plasmid (0.4 µg/µl) along with miR-433 mimics
(50 nM) or negative control (50 nM) and a Renilla luciferase
control reporter vector (0.218 µg/µl; pRL-SV40; Promega
Corporation) using Lipofectamine 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.). Following 24 h of transfection, luciferase
activities were measured using a Luciferase Assay kit (Promega
Corporation) and a GloMax® 96 microplate luminometer
(Promega Corporation), according to the manufacturer's
protocol.
Cell viability assay
The Cell Counting kit 8 (CCK8) assay was used to
determine the viability of the Min-6 cells in the following
treatment groups: 5 mmol/l glucose, 11 mmol/l glucose, 11 mmol/l
glucose and siCOX2, 11 mmol/l glucose and mimics, and 11 mmol/l
glucose and NC. Briefly, cells (3×103 cells/well) were
seeded in a 96-well plate and incubated in 10% CCK8 solution
(Shanghai 7Sea PharmTech Co. Ltd., Shanghai, China) solution for 2
h at 37°C. Proliferation rates were determined at 0, 12, 24, 48,
and 72 h post-treatment. The absorbance of each well was measured
at 490 nm using using a Synergy HTX Multi-Mode Microplate Reader
(BioTek Instruments, Inc., Winooski, VT, USA). Each experiment was
performed in triplicate.
Colony formation assay
Min-6 cells (2×103 cells) from different
treatment groups were seeded in a 10-cm cell culture dish and
cultured in DMEM (HyClone; GE Healthcare Life Sciences)
supplemented with 10% FBS (Gibco; Thermo Fisher Scientific, Inc.)
for 10 days at 37°C and 5% CO2 until the colonies were
visible. Min-6 cells were treated with a low concentration of
glucose (5 mmol/l group) or a high concentration of glucose (11
mmol/l group); high glucose-treated cells were treated with miR-433
mimic (11 mmol/l + mimics group), COX2 siRNA (11 mmol/l + siCOX2
group) or NC siRNA (11 mmol/l + siNC group). Following incubation
for 24 h, the medium was removed and the plates were washed twice
in PBS. Colonies were fixed in 95% ethanol for 10 min at room
temperature, air-dried for 10 min at room temperature and stained
with 0.1% crystal violet solution for 10 min at room temperature.
Colonies containing >50 cells were directly identified by their
size and counted. The experiment was performed three times.
Flow cytometric analysis
Min-6 cells were treated with 5 mmol/l glucose, 11
mmol/l glucose, 11 mmol/l glucose and COX2 siRNA (11 mmol/l +
siCOX2 group), 11 mmol/l glucose and NC (11 mmol/l + NC group), or
11 mmol/l glucose and miR-433 mimic (11 mmol/l + mimics group) for
24 h. Min-6 cells (1×106 cells/ml) from the different
treatment groups were used for apoptosis and cell cycle analysis
with the Dead Cell Apoptosis Kit with Annexin V, fluorescein
isothiocyanate (FITC) and propidium iodide (PI), for flow cytometry
(Invitrogen, U.S.A; Thermo Fisher Scientific, Inc.), according to
the manufacturer's protocol. Briefly, cells were harvested by
trypsinization, resuspended in 500 µl binding buffer and stained
with 5 µl Annexin V-FITC and 5 µl PI solution for 30 min at room
temperature in the dark. Samples were immediately analyzed for
apoptosis using a FACSCalibur flow cytometer (BD Biosciences,
Franklin Lakes, NJ, USA). For cell cycle analysis, the cells were
fixed in ice cold 70% (v/v) ethanol overnight, resuspended in 500
µl PBS containing 50 mg/ml RNase (Sigma-Aldrich; Merck KGaA) and
incubated at 37°C for 30 min. Following staining with 50 mg/ml PI
(Sigma-Aldrich; Merck KGaA) at 4°C in the dark for 30 min, cell
cycle distribution was analyzed using a FACSCalibur flow cytometer
(BD Biosciences). Each sample was analyzed using Flowing Software
version 2.5.0 (http://flowingsoftware.btk.fi/index.php?page=1) in
triplicate.
Statistical analysis
Data were analyzed using SPSS software, version 13.0
(SPSS Inc., Chicago, IL, USA) and expressed as the mean ± standard
deviation of >3 samples. The statistical significance of the
differences between groups was assessed using one-way analysis of
variance followed by a post hoc Bonferroni test for multiple
comparisons. P<0.05 was considered to indicate a statistically
significant difference.
Results
miR-433 expression is inhibited under
high glucose conditions, and miR-433 directly targets COX2 in
pancreatic β cells
As shown in Fig.
1A, Min-6 cell viability was significantly inhibited in
high-glucose condition (11 mmol/l) compared with low-glucose
condition (5 mml/l; P<0.001). miRNAs, including miR-433,
miR-199-5p, miR-30a and miR-22, have been reported to serve an
important role in hyperglycemia (29,30).
Therefore, the expression levels of several miRNAs were examined in
Min-6 cells cultured in high-glucose or low-glucose. Notably, the
expression level of miR-433 was significantly lower in
high-glucose-induced Min-6 cells, whereas the expression level of
miR-22 was significantly higher in high-glucose-induced Min-6 cells
(P<0.001 and P<0.01, respectively; Fig. 1B). Since the change in miR-433
expression appeared to be the most profound, miR-433 was further
investigated. Bioinformatics analysis was used to identify
potential target genes for miR-433, which indicated that COX2
contained a potential miR-433 binding site in the 3′-UTR (Fig. 1C). To further confirm whether COX2
was directly regulated by miR-433, a wild-type and a mutated
version of the COX2 3′-UTR was cloned and inserted into luciferase
reporter constructs. Co-transfection with miR-433 mimics
significantly reduced luciferase activity in reporter constructs
containing the wild-type COX 2 3′-UTR (P<0.01; Fig. 1D), while there was no change in
luciferase activity following mutation of the potential miR-433
binding site.
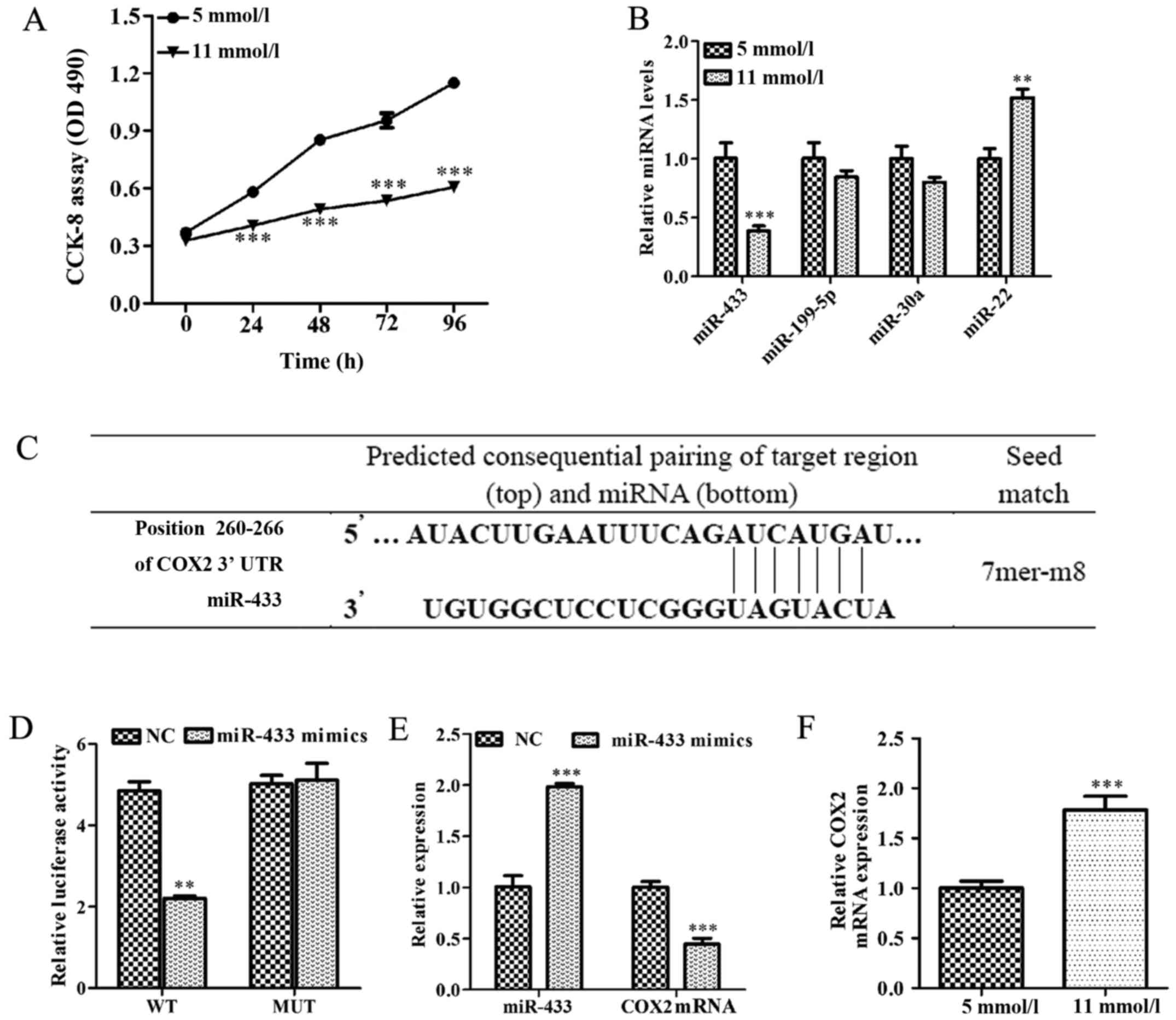 | Figure 1.miR-433 is associated with cell growth
inhibition induced by high glucose, and miR-433 negatively
regulates COX2 expression in Min-6 pancreatic β cells. (A) CCK-8
assay was used to determine cell viability in Min-6 cells treated
with either a low (5 mmol/l) or a high (11 mmol/l) concentration of
glucose for 24 h. (B) Expression levels of miR-433, miR-199-5p,
miR-30a and miR-22 were analyzed by RT-qPCR in Min-6 cells treated
with either 5 mmol/l or 11 mmol/l glucose. (C) Bioinformatics
prediction of miR-433 binding site in the 3′-UTR of COX2 mRNA. (D)
HEK293T cells were co-transfected with a WT or a MUT COX 3′-UTR
reporter plasmid along with miR-433 mimics or NC mimics. Activity
was measured by dual luciferase reporter assay and presented as
relative luciferase activity. (E) COX2 mRNA expression levels were
detected by RT-qPCR in Min-6 cells cultured in high glucose
conditions following transfection with miR-433 mimics. (F) mRNA
levels of COX2 were determined by RT-qPCR in Min-6 cells treated
with 5 mmol/l or 11 mmol/l glucose. Data are expressed as the mean
± standard deviation; **P<0.01 and ***P<0.001 vs. low-glucose
treatment or NC. CCK-8, Cell Counting Kit-8; COX2, cyclooxygenase
2; miR, microRNA; miRNA, microRNA; Mmu, Mus musculus; MUT,
mutant; RT-qPCR, reverse transcription-quantitative polymerase
chain reaction; UTR, untranslated region; WT, wild-type. |
The effects of miR-433 overexpression on endogenous
COX2 expression were also examined in Min-6 cells under normal
glucose conditions (5 mmol/l); compared with the control cells,
endogenous COX2 mRNA levels were significantly reduced in Min-6
cells transfected with miR-433 mimics (P<0.001; Fig. 1E). The levels of COX2 mRNA
expression were significantly increased in high-glucose-induced
Min-6 cells compared with cells in the low-glucose treatment group
(P<0.001; Fig. 1F). These
results suggested that COX2 was overexpressed in Min-6 cell
cultured in high-glucose condition and that COX2 may be a target of
miR-433.
miR-433 stimulates pancreatic β cell
proliferation
To investigate the potential roles of miR-433 on
Min-6 cell proliferation, Min-6 cells were first transfected with
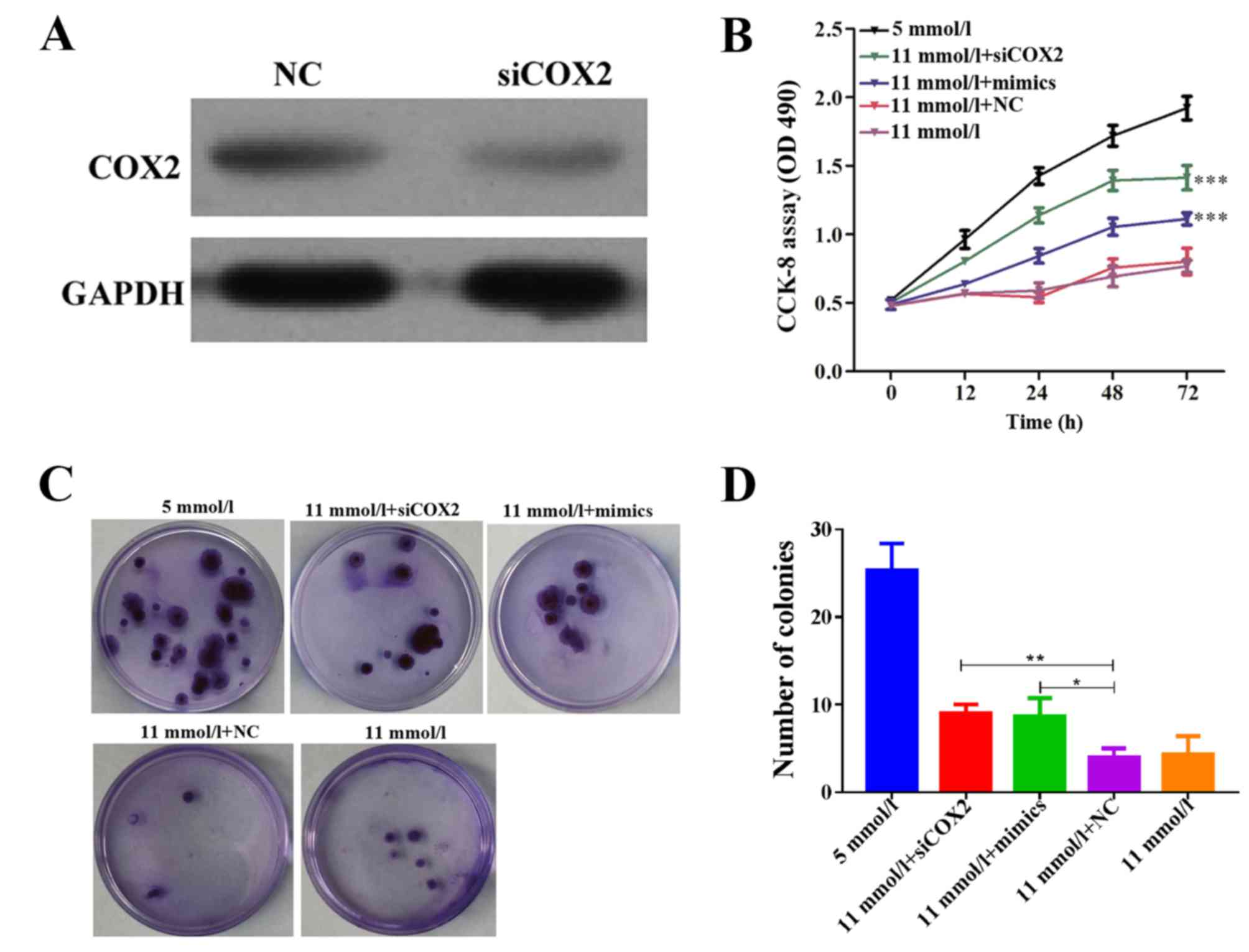
miR-433 mimics. Endogenous COX2 protein expression was reduced in
Min-6 cells treated with siCOX2, as determined by western blot
analysis (Fig. 2A). Results from
the CCK-8 assay indicated that both miR-433 mimics and siCOX2
treatments alleviated cell growth inhibition in Min-6 cells
cultured in high-glucose conditions (Fig. 2B; P<0.001). Consistently, the
colony formation capabilities of Min-6 cells were inhibited in
high-glucose conditions, whereas treatment with either miR-433
mimics or siCOX2 significantly enhanced the colony forming ability
in Min-6 cells cultured in high-glucose conditions (Fig. 2C and D; P<0.001). These results
suggested that the glucose-induced reduction in Min-6 cell
proliferation may be reversed following COX2 inhibition or miR-433
overexpression. Therefore, these results suggested that miR-433
overexpression may be able to promote pancreatic β cell
proliferation in Min-6 cells grown in a high-glucose
environment.
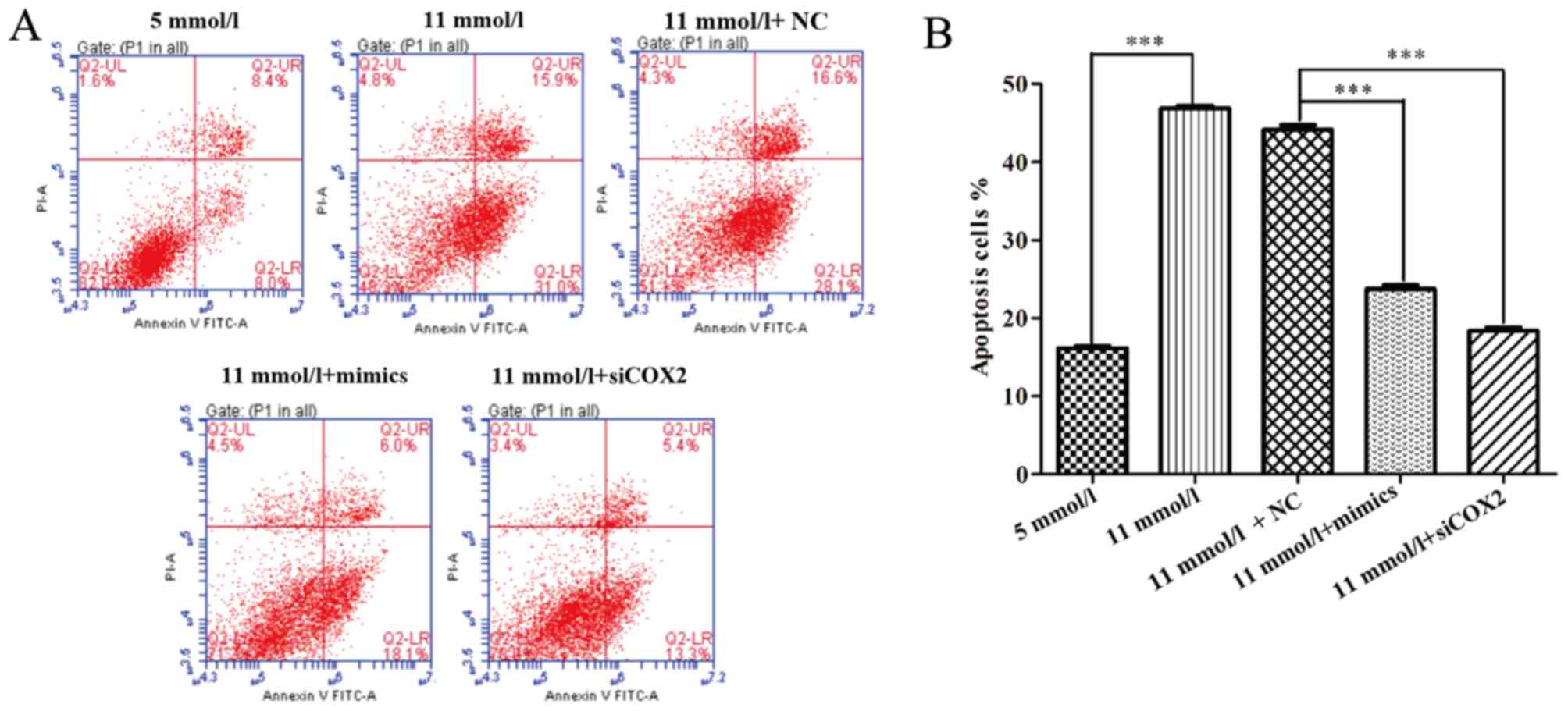
miR-433 alleviated apoptosis and cell
cycle arrest induced by high-glucose
To further uncover the mechanisms underlying the
improved cell proliferation induced by miR-433 in pancreatic β
cells treated with high-glucose, the effects of miR-433 mimics on
apoptosis were examined using a flow cytometry apoptosis assay.
Both miR-433 mimics and siCOX2 treatments caused significant
changes in the profiles of Annexin V-FITC/PI-stained cell
populations. The apoptotic rate of the miR-433 mimics and siCOX2
groups appears to be much lower compared with the NC group
(Fig. 3A) Statistical analysis
further demonstrated that both miR-433 mimics and siCOX2 treatments
significantly reduced the level of high-glucose-induced apoptosis
in Min-6 cells (P<0.001; Fig.
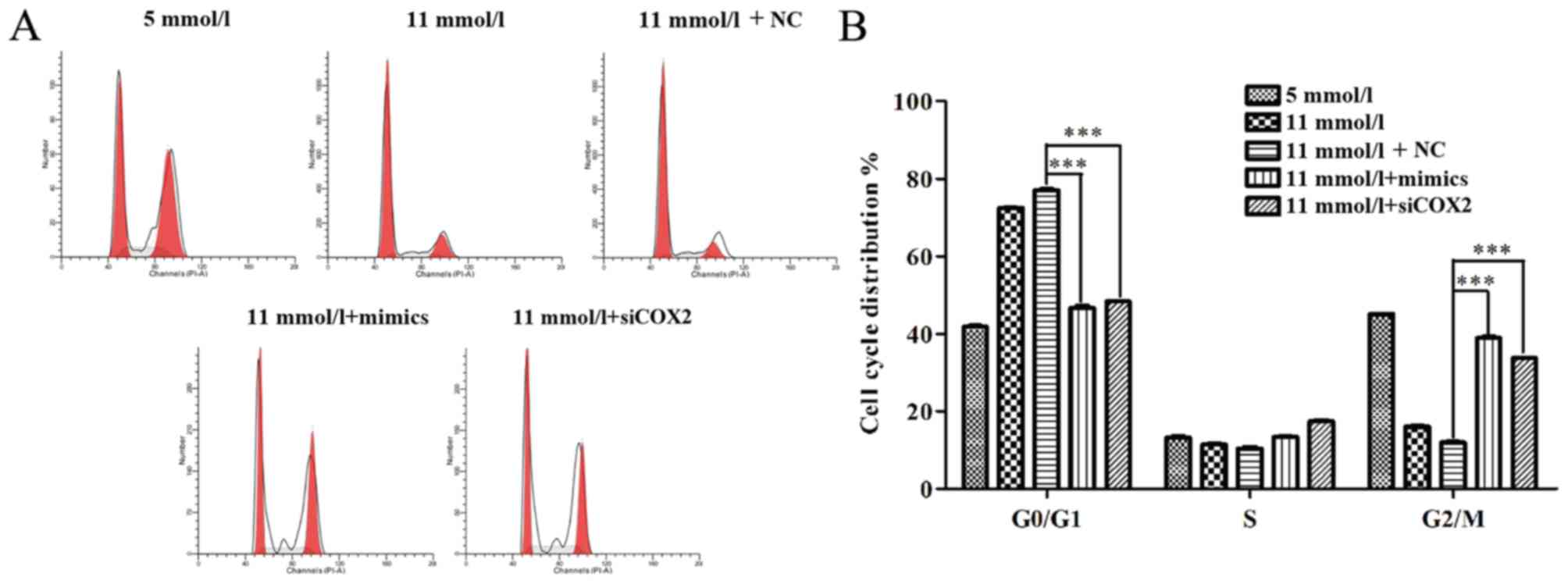
3B). In addition, flow cytometric analysis was used to
characterize cell cycle behavior of Min-6 cells treated with siCOX2
or miR-433 mimics. The distribution of cells in the phases of the
cell cycle (G0/G1, S and G2/M) varied between the different
treatment groups (Fig. 4A).
Compared with the NC group, miR-433 overexpression and treatment
with siCOX2 led to a significant increase in the percentage of
cells in G2/M phase and a reduction in G0/G1 phase cells
(P<0.001; Fig. 4B). Based on
these results, the present study speculated that miR-433 could
alleviate high-glucose-induced growth inhibition in pancreatic β
cells through regulating cell cycle and apoptosis.
Discussion
The proliferative and regenerative capacity of
pancreatic β cells is known to be closely associated with the
quantity of insulin released; there is a feedback loop between
insulin secretion and the concentration of glucose in the blood
(31). In this regard, it is
important to improve pancreatic β cell function and survival under
proapoptotic conditions to design innovative therapeutics for DM.
In the present study, Min-6 cells grown in high-glucose medium
exhibited more suppressive effects on β cell viability and
proliferation compared with cells grown in low-glucose conditions,
which is consistent with a previous study (8). In addition, significant increases in
apoptosis and cell cycle arrest at G2/M phase were noted in β cells
cultured with high-glucose compared with those grown in low-glucose
media.
To further investigate the mechanisms underlying the
high-glucose-induced β cell growth inhibition, several
concentrations of glucose in β cells were screened. The expression
of miR-433 was revealed to be downregulated in high-glucose-induced
Min-6 cells, whereas the expression of miR-22 was upregulated.
However, further studies are required to validate the results
regarding miR-22 expression and to investigate its effects in
pancreatic β cells.
In the present study, miR-433 expression was
demonstrated to be downregulated in high-glucose treatments and its
overexpression by miRNA-433 mimics transfection significantly
promoted cell viability and proliferation in high-glucose
conditions by suppressing apoptosis and enhancing cell cycle
progression. Previous studies have reported that miR-433 is
downregulated in various cancer tissues, such as bladder cancer and
oral squamous cell carcinomas (32,33),
which indicated that miR-433 may have a tumor-suppressor function
in carcinomas. Notably, the expression levels of miR-433 were
revealed to be reduced in renal tissues of pancreatic
kininogenase-treated patients with diabetic nephropathy (34). miR-433 may negatively regulate the
expression of thymidylate synthase in HeLa cells (35), suggesting a role in metabolism
regulation, thus possibly modulating the glucose metabolism.
Furthermore, the present study identified and
validated COX2 as a potential target of miR-433, which suggested
that miR-433 may participate in β cell biology by regulating COX2
expression. COX2 is an important enzyme involved in the production
of prostaglandin E2 (PGE2) and arachidonic acid (36). PGE2, as a COX2-generated
metabolite, protects β cells from apoptosis (37). Results from the present study
indicated that COX2 was overexpressed in Min-6 cells cultured in
high-glucose compared with cells grown in low-glucose conditions.
Knockdown of COX2 by siRNA was demonstrated to alleviate the
high-glucose-induced inhibition of cell proliferation by
suppressing apoptosis and promoting cell cycle progression.
According to a recent report, COX2 was enhanced by the
overexpression of p50 following exposure to an inflammatory
mediator in DM (38). Moreover,
inhibition of COX2 has been demonstrated to be a promising
antitumor therapy in renal cell carcinoma (39).
In summary, the expression level of miR-433
decreased, whereas the level of COX2 mRNA increased in Min-6 mouse
pancreatic β cells cultured in high-glucose medium; results from
in vitro analyses indicated that miR-433 may promote
pancreatic β cell growth by regulating COX2 expression. Results
from the present study may provide information regarding novel
therapeutic targets for DM.
References
|
1
|
Abdulazeez S Sheik: Diabetes treatment: A
rapid review of the current and future scope of stem cell research.
Saudi Pharm J. 23:333–340. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Scully T: Diabetes in numbers. Nature.
485:S2–S3. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sikorska D, Pawlaczyk K, Olewicz-Gawlik A,
Czepulis N, Posnik B, Baum E, Wanic-Kossowska M, Lindholm B and Oko
A: The importance of residual renal function in peritoneal
dialysis. Int Urol Nephrol. 48:2101–2108. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Abbasakoor NO, Healy ML, O'Shea D, Maguire
D, Muldoon C, Sheahan K and O'Toole D: Metastatic insulinoma in a
patient with type 2 diabetes mellitus: Case report and review of
the literature. Int J Endocrinol. 2011:1240782011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
American Diabetes Association, . Diagnosis
and classification of diabetes mellitus. Diabetes care. 37:(Suppl
1). S81–S90. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Prentice KJ, Luu L, Allister EM, Liu Y,
Jun LS, Sloop KW, Hardy AB, Wei L, Jia W, Fantus IG, et al: The
furan fatty acid metabolite CMPF is elevated in diabetes and
induces β cell dysfunction. Cell Metab. 19:653–666. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rutter GA, Pullen TJ, Hodson DJ and
Martinez-Sanchez A: Pancreatic β-cell identity, glucose sensing and
the control of insulin secretion. Biochem J. 466:203–218. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang S, Yang Z, Gao Y, Li Q, Su Y, Wang Y,
Zhang Y, Man H and Liu H: Pyruvate kinase, muscle isoform 2
promotes proliferation and insulin secretion of pancreatic β-cells
via activating Wnt/CTNNB1 signaling. Int J Clin Exp Pathol.
8:14441–14448. 2015.PubMed/NCBI
|
|
9
|
Tangvarasittichai S: Oxidative stress,
insulin resistance, dyslipidemia and type 2 diabetes mellitus.
World J Diabetes. 6:456–480. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Del Prato S, Bianchi C and Marchetti P:
beta-cell function and anti-diabetic pharmacotherapy. Diabetes
Metab Res Rev. 23:518–527. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vetere A, Choudhary A, Burns SM and Wagner
BK: Targeting the pancreatic β-cell to treat diabetes. Nat Rev Drug
Discov. 13:278–289. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Santulli G, Pagano G, Sardu C, Xie W,
Reiken S, D'Ascia SL, Cannone M, Marziliano N, Trimarco B, Guise
TA, et al: Calcium release channel RyR2 regulates insulin release
and glucose homeostasis. J Clin Invest. 125:43162015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Prabhakar PK and Doble M: Mechanism of
action of natural products used in the treatment of diabetes
mellitus. Chin J Integr Med. 17:563–574. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hwang HW and Mendell JT: MicroRNAs in cell
proliferation, cell death, and tumorigenesis. Br J Cancer.
94:776–780. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sullivan CS and Ganem D: MicroRNAs and
viral infection. Mol Cell. 20:3–7. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Staedel C and Darfeuille F: MicroRNAs and
bacterial infection. Cell Microbiol. 15:1496–1507. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kato M, Castro NE and Natarajan R:
MicroRNAs: potential mediators and biomarkers of diabetic
complications. Free Radic Biol Med. 64:85–94. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
He L and Hannon GJ: MicroRNAs: Small RNAs
with a big role in gene regulation. Nat Rev Genet. 5:522–531. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Filipowicz W: RNAi: The nuts and bolts of
the RISC machine. Cell. 122:17–20. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Latreille M, Hausser J, Stutzer I, Stützer
I, Zhang Q, Hastoy B, Gargani S, Kerr-Conte J, Pattou F, Zavolan M,
et al: MicroRNA-7a regulates pancreatic β cell function. J Clin
Invest. 124:2722–2735. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Poy MN, Eliasson L, Krutzfeldt J, Kuwajima
S, Ma X, Macdonald PE, Pfeffer S, Tuschl T, Rajewsky N, Rorsman P
and Stoffel M: A pancreatic islet-specific microRNA regulates
insulin secretion. Nature. 432:226–230. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
El Ouaamari A, Baroukh N, Martens GA,
Lebrun P, Pipeleers D and van Obberghen E: miR-375 targets
3′-phosphoinositide-dependent protein kinase-1 and regulates
glucose-induced biological responses in pancreatic beta-cells.
Diabetes. 57:2708–2717. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lovis P, Gattesco S and Regazzi R:
Regulation of the expression of components of the exocytotic
machinery of insulin-secreting cells by microRNAs. Biol Chem.
389:305–312. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Correa-Medina M, Bravo-Egana V, Rosero S,
Ricordi C, Edlund H, Diez J and Pastori RL: MicroRNA miR-7 is
preferentially expressed in endocrine cells of the developing and
adult human pancreas. Gene Expr Patterns. 9:193–199. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Estep M, Armistead D, Hossain N, Elarainy
H, Goodman Z, Baranova A, Chandhoke V and Younossi ZM: Differential
expression of miRNAs in the visceral adipose tissue of patients
with non-alcoholic fatty liver disease. Aliment Pharmacol Ther.
32:487–497. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Luo H, Zhang H, Zhang Z, Zhang X, Ning B,
Guo J, Nie N, Liu B and Wu X: Down-regulated miR-9 and miR-433 in
human gastric carcinoma. J Exp Clin Cancer Res. 28:822009.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chavali V, Tyagi SC and Mishra PK:
Differential expression of dicer, miRNAs, and inflammatory markers
in diabetic Ins2+/− Akita hearts. Cell Biochem Biophys. 68:25–35.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jansen F, Wang H, Przybilla D, Franklin
BS, Dolf A, Pfeifer P, Schmitz T, Flender A, Endl E, Nickenig G and
Werner N: Vascular endothelial microparticles-incorporated
microRNAs are altered in patients with diabetes mellitus.
Cardiovasc Diabetol. 15:492016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang JM and Zhang K: Microarray analysis
of microRNA expression in bone marrow-derived progenitor cells from
mice with type 2 diabetes. Genom Data. 7:86–87. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Weir GC and Bonner-Weir S: Five stages of
evolving beta-cell dysfunction during progression to diabetes.
Diabetes. 53:(Suppl 3). S16–S21. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xu X, Zhu Y, Liang Z, Li S, Xu X, Wang X,
Wu J, Hu Z, Meng S, Liu B, et al: c-Met and CREB1 are involved in
miR-433-mediated inhibition of the epithelial-mesenchymal
transition in bladder cancer by regulating Akt/GSK-3β/Snail
signaling. Cell Death Dis. 7:e20882016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wang XC, Ma Y, Meng PS, Han JL, Yu HY and
Bi LJ: miR-433 inhibits oral squamous cell carcinoma (OSCC) cell
growth and metastasis by targeting HDAC6. Oral Oncol. 51:674–682.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhu D, Zhang L, Cheng L, Ren L, Tang J and
Sun D: Pancreatic kininogenase ameliorates renal fibrosis in
streptozotocin induced-diabetic nephropathy rat. Kidney Blood Press
Res. 41:9–17. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gotanda K, Hirota T, Matsumoto N and Ieiri
I: MicroRNA-433 negatively regulates the expression of thymidylate
synthase (TYMS) responsible for 5-fluorouracil sensitivity in HeLa
cells. BMC Cancer. 13:3692013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Greenhough A, Smartt HJ, Moore AE, Roberts
HR, Williams AC, Paraskeva C and Kaidi A: The COX-2/PGE2 pathway:
Key roles in the hallmarks of cancer and adaptation to the tumour
microenvironment. Carcinogenesis. 30:377–386. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Papadimitriou A, King AJ, Jones PM and
Persaud SJ: Anti-apoptotic effects of arachidonic acid and
prostaglandin E2 in pancreatic beta-cells. Cell Physiol Biochem.
20:607–616. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Burke SJ, Karlstad MD, Regal KM, Sparer
TE, Lu D, Elks CM, Grant RW, Stephens JM, Burk DH and Collier JJ:
CCL20 is elevated during obesity and differentially regulated by
NF-κB subunits in pancreatic β-cells. Biochim Biophys Acta.
1849:637–652. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Tabriz HM, Mirzaalizadeh M, Gooran S, Niki
F and Jabri M: COX-2 expression in renal cell carcinoma and
correlations with tumor grade, stage and patient prognosis. Asian
Pac J Cancer Prev. 17:535–538. 2016. View Article : Google Scholar : PubMed/NCBI
|