Introduction
Acute respiratory distress syndrome (ARDS) is an
extreme manifestation of acute injury to the lung, characterized by
extensive lung inflammation, profound hypoxemia and non-cardiogenic
pulmonary edema formation (1,2). In
addition, ARDS is a serious complication resulting from sepsis and
associated with multiple organ failure, and mortality and morbidity
(3). It has been estimated that
~200,000 cases of ARDS occurred annually, with a high mortality
rate of ~40% in the United States (4). The outcome in ARDS is complicated as
the syndrome is influenced by multiple factors, such as the nature
of the precipitating factors and the extent of the subsequent
multiorgan failure (5).
The pathogenesis of ARDS involves inflammatory
injury to the lung endothelium and epithelium (6), which is accompanied by an influx of
neutrophils into the pulmonary interstitium and bronchoalveolar
space (7). The activated
neutrophils may damage endothelial and epithelial cells and are
considered to be a crucial factor in pathogenesis and progression
of ARDS (8). Endothelial injury
may result in the effusion of protein-rich fluid into alveolar
airspace and the damage to alveolar epithelium may increase the
entry of fluid into the alveolar lumen (2).
Previous studies have performed proteomic and
genomic analyses to determine the pathogenesis of lung diseases
(9–11), an increasing number of studies have
focused on the role of small molecules, such as microRNAs (miRNAs)
in the pathogenesis of the diseases (12–14).
miRNAs are a class of non-coding small RNAs ~22
nucleotides in length. miRNAs negatively regulate gene expression
through translational repression or mRNA degradation by binding to
3′-untranslated region (3′-UTR) of the target mRNA transcripts
(14,15). miRNAs have been reported to be
extensively involved in many biological processes and associated
with various diseases (16,17).
A considerable number of miRNAs have been observed to participate
in the regulation of physiological lung function, including miRNA
(miR)-17, miR-92a and miR-127 (18–20).
However, how miRNAs are involved in the pathogenesis and
progression of ARDS remains to be elucidated.
The present study performed miRNA microarray
analysis of the GSE57223 dataset downloaded from Gene Expression
Omnibus (GEO) database, and aimed to identify the potential
regulatory roles of miRNAs in the pathogenesis and progression of
ARDS in order to provide novel insights into the regulatory
mechanisms of the ARDS etiology.
Materials and methods
miRNA microarray data
The datasets of GSE57223 (2) deposited in the GEO database
(https://www.ncbi.nlm.nih.gov/geo) were
downloaded. The data set contains miRNA expression profiles of 12
samples from 6 control normal lung tissues and 6 ARDS lung tissue
from a rat model, which has previously been described (2). The platform was GPL18654 (Rat
microRNA array).
Data preprocessing
The probe numbers were converted into the
corresponding gene symbols. The average probe value was calculated
as the expression value of the gene when multiple probes
corresponded to one gene. For probes with a missing value, the
k-nearest neighbor (KNN) averaging method from the impute package
in R (21) was applied to fill the
vacancy, with k in the default parameter of 10. Subsequently,
quantile normalization was conducted using the preprocessCore
package in R (22).
Identification of differentially
expressed miRNAs
Following normalization of the data, the limma
package in R (23) was used to
identify differentially expressed miRNAs between the ARDS lung
tissues and the control tissues, using the t-test method. The
P-value was adjusted using the Benjamini-Hochberg method (24). Significant differentially expressed
miRNAs with P<0.05 and |log2FC|>1 were identified.
Construction of miRNA-miRNA network
with co-regulated target genes
A gene that may be targeted by more than one miRNA
was termed the co-regulated target gene of these miRNAs. The
miRWalk database (25), which
provides 10 different types of prediction algorithms, including
DIANAmT, miRanda, miRDB, miRWalk, RNAhybrid, PICTAR4, PICTAR5,
PITA, RNA22 and TargetScan, was used to construct the regulatory
network of miRNAs and their target genes. The database was searched
to construct the regulatory network of miRNAs and their target
genes. Each method may produce a prediction result. In the present
study, the target genes were predicted by at least 4 different
algorithms to construct the miRNA-miRNA network with co-regulated
target genes, which was visualized utilizing Cytoscape software
version 3.4.0 (26).
Synergistic function analysis of
miRNAs and biological function analysis of their target genes
The gene symbol of target genes was transformed into
gene ID and clusterProfiler package in R (27) was used to perform Gene Ontology
(GO) enrichment analysis, particularly for the Biological Process
(BP) term. A functional synergistic miRNA-miRNA interaction pair
was defined when their co-regulated target genes were enriched in
at least one BP term. The threshold criteria were P<0.05 and
q<0.05 for significant BP terms. The miRNA interaction pairs
having common target genes were filtered out to establish the
miRNA-miRNA functional synergistic network (MFSN) (15).
In the MFSN, the co-regulated target genes of the
significant miRNA-miRNA interaction pairs were screened for further
analysis. GO and pathway analyses were conducted using the Database
for Annotation, Visualization and Integrated Discovery online
software (28) and the threshold
for significant GO and pathway terms were P-<0.05. Additionally,
the interacted protein-protein pairs with combined score >0.04
were selected to establish the protein-protein-interaction (PPI)
network for the co-regulated target genes using STRING version 9.1
(29), which was visualized with
Cytoscape software (26). In
addition, hub proteins were screened out through the analysis of
topological characteristics of the PPI network.
Results
miRNAs differentially expressed
between ARDS lung tissues and normal tissues
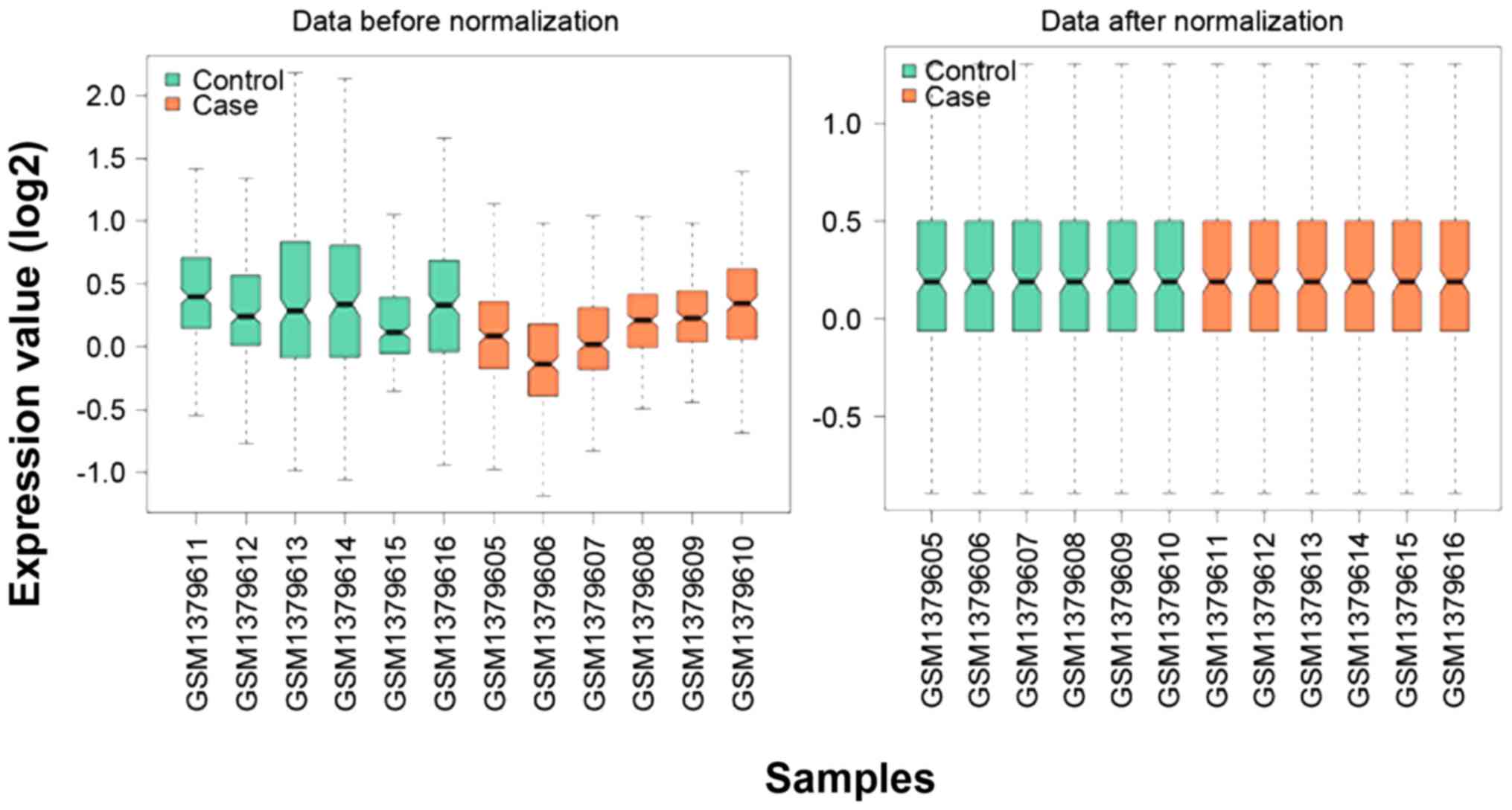
Normalization of the microarray data was performed
and the medians of miRNA expression values are presented in
Fig. 1. According to the defined
threshold criteria, a total of 19 significant differentially
expressed miRNAs were identified between ARDS lung tissues and
normal tissues, with 6 upregulated miRNAs, including rno-miR-30e,
rno-mir-129, rno-miR-199a, rno-miR-34b and rno-miR-103, and 13
downregulated miRNAs, namely rno-miR-122a, rno-miR-290,
rno-miR-324-5p, rno-miR-35, rno-miR-223, rno-let-7f, rno-miR-26a,
rno-miR-24, rno-miR-143, rno-let-7b, rno-let-7c, rno-let-7a and
rno-miR-126.
miRNA-miRNA network with co-regulated
target genes
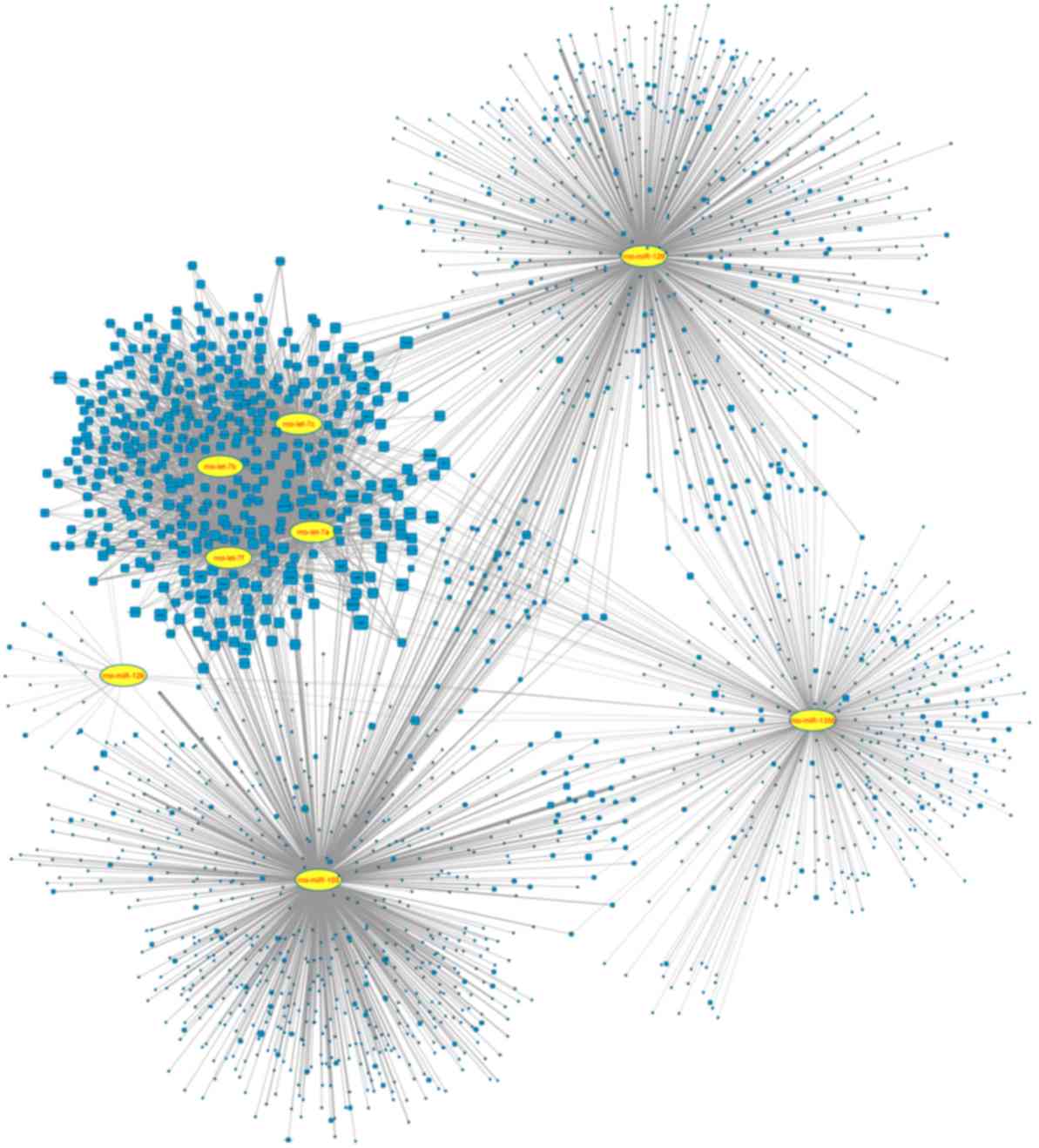
Based on the prediction of mirWalk databases and the
selection criterion, the target genes of eight miRNAs were screened
out. Notably, rno-miR-103 had the highest number (n=757) of target
genes and the 4 rno-let-7 miRNAs also had high numbers (n=409) of
target genes (Table I). To detect
the associations of the miRNAs and their target genes, the
regulatory network for the 8 miRNAs with their target genes was
constructed, comprising of 3590 edges and 2152 nodes (Fig. 2). Furthermore, a miRNA-miRNA
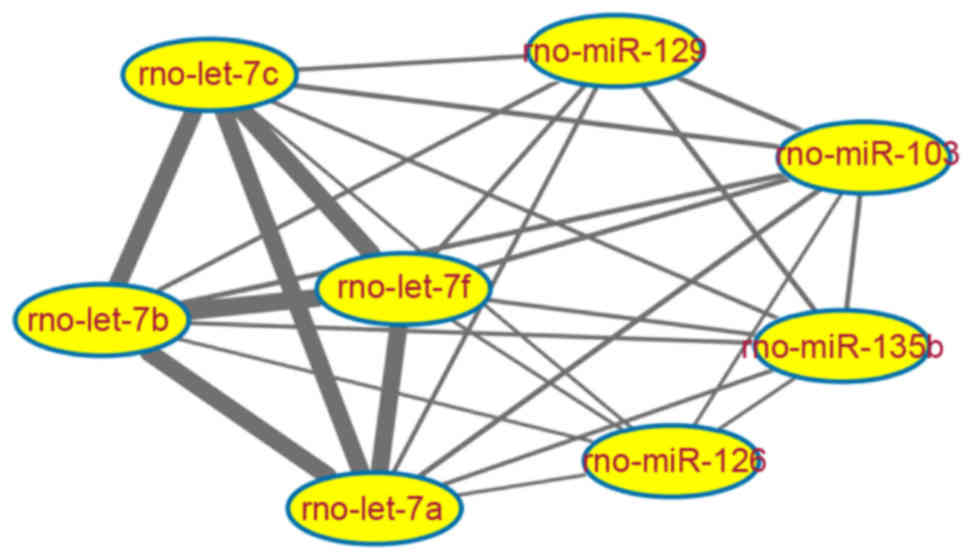
network with co-regulated target genes was established, consisting
of 27 miRNA-miRNA interaction pairs. As presented in Fig. 3 and Table I, the four downregulated miRNAs
from the rno-let-7 family had high numbers (n=409) of target
genes.
 | Table I.Predicted target genes of
differentially expressed miRNAs based on the mirWalk database. |
Table I.
Predicted target genes of
differentially expressed miRNAs based on the mirWalk database.
| miRNA | Target genes | Regulation |
|---|
| rno-miR-126 | 28 | Down |
| rno-miR-129 | 674 | Up |
| rno-let-7a | 409 | Down |
| rno-let-7b | 409 | Down |
| rno-miR-135b | 495 | Down |
| rno-let-7c | 409 | Down |
| rno-miR-103 | 757 | Up |
| rno-let-7f | 409 | Down |
MFSN and biological functions of the
target genes
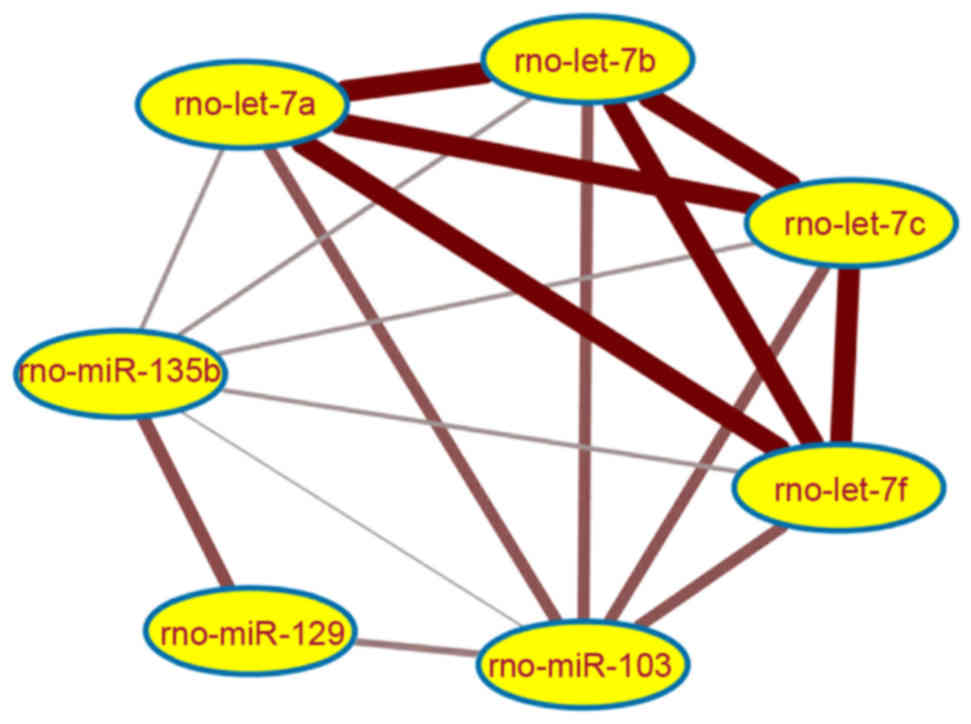
In order to investigate the synergic functions
between miRNAs, the MFSN was established, consisting of 17
miRNA-miRNA functional synergistic pairs, of the 27 identified
interaction pairs with co-regulated target genes, involving a total
of 7 miRNAs. As presented in Fig.
4, the 4 rno-let-7 family members were detected with synergic
functions. Further functional analysis indicated that 409 target
genes of the 4 rno-let-7 family miRNAs were significantly enriched
in BP terms, such as response to wounding and inflammatory response
and cellular component terms, including cell fraction and insoluble
fraction, and molecular function terms, such as manganese ion
binding and activin receptor activity. In addition, these target
genes were significantly enriched in the chronic myeloid leukemia
and Graft-vs.-host disease pathways (Table II). The PPI network for these
target genes was constructed and had 176 edges and 153 nodes
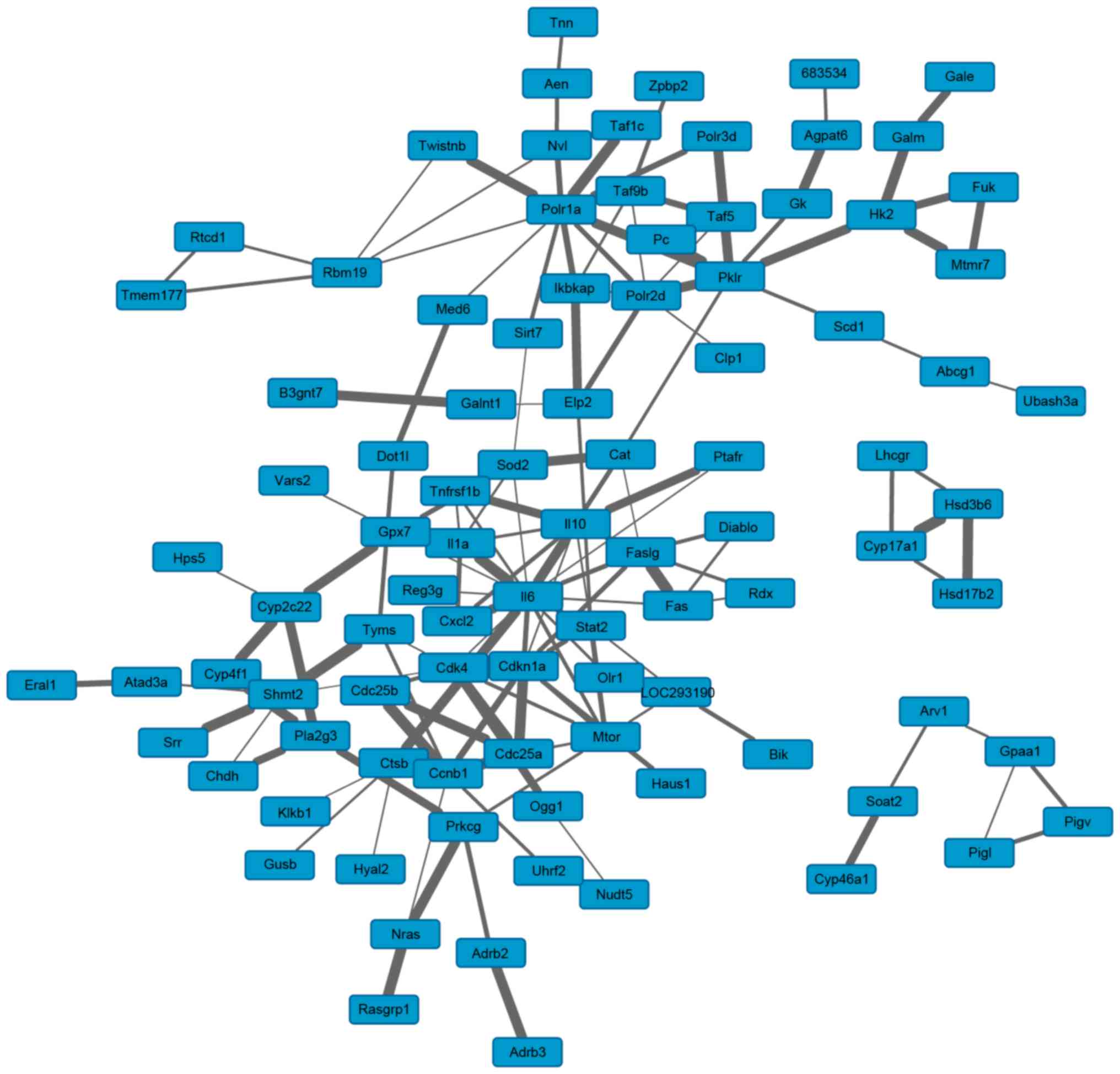
(Fig. 5). Based on the topological
characteristics of the PPI network, the hub proteins with high
degrees were identified, including interleukin (IL)-6 (degree, 17),
RNA polymerase I subunit A (degree, 11), Fas cell surface death
receptor (degree, 10), IL-10 and cyclin-dependent kinase-inhibitor
1 (degree, both 9).
 | Table II.Top 5 significant GO and KEGG pathway
terms for the co-regulated target genes of 4 rno-let-7
microRNAs. |
Table II.
Top 5 significant GO and KEGG pathway
terms for the co-regulated target genes of 4 rno-let-7
microRNAs.
| A, Biological
processes |
|---|
|
|---|
| Term | Description | Count | P-value |
|---|
| GO:0010033 | Response to organic
substance | 29 |
1.66×10−5 |
| GO:0009611 | Response to
wounding | 17 |
1.08×10−4 |
| GO:0006954 | Inflammatory
response | 12 |
1.09×10−4 |
| GO:0032496 | Response to
lipopolysaccharide | 9 |
1.27×10−4 |
| GO:0009991 | Response to
extracellular stimulus | 14 |
1.36×10−4 |
|
| B, Cellular
component |
|
| Term | Description | Count | P-value |
|
| GO:0000267 | Cell fraction | 33 |
6.66×10−6 |
| GO:0005626 | Insoluble
fraction | 28 |
9.99×10−6 |
| GO:0005624 | Membrane
fraction | 27 |
1.14×10−5 |
| GO:0031090 | Organelle
membrane | 31 |
2.81×10−5 |
| GO:0043235 | Receptor
complex | 9 |
1.37×10−4 |
|
| C, Molecular
function |
|
| Term | Description | Count | P-value |
|
| GO:0048037 | Cofactor
binding | 13 |
4.29×10−4 |
| GO:0030145 | Manganese ion
binding | 7 | 0.001334536 |
| GO:0017002 | Activin receptor
activity | 3 | 0.001543724 |
| GO:0050662 | Coenzyme
binding | 10 | 0.002201207 |
| GO:0008289 | Lipid binding | 13 | 0.002212067 |
|
| D, KEGG
pathway |
|
| Term | Description | Count | P-value |
|
| rno05220 | Chronic myeloid
leukemia | 6 | 0.010215393 |
| rno05332 | Graft-versus-host
disease | 5 | 0.01224159 |
| rno04142 | Lysosome | 7 | 0.017086633 |
| rno00260 | Glycine, serine and
threonine metabolism | 4 | 0.018428521 |
| rno04060 | Cytokine-cytokine
receptor interaction | 9 | 0.022832763 |
Discussion
ARDS is a severe syndrome of acute respiratory
failure associated with multiple symptoms and influenced by
numerous factors (2,6). The expression of miRNAs is critically
associated with cancer initiation, development and metastasis
(30). The present study
identified a total of 19 significant differentially expressed
miRNAs between ARDS rat lung tissues and normal rat lung tissues.
Notably, 4 downregulated let-7 miRNA family members (rno-let-7a,
rno-let-7b, rno-let-7c and rno-let-7f) were observed to have a high
number of co-regulated target genes and were recognized as
miRNA-miRNA interactions with synergistic functions. Furthermore,
the target genes of the miRNAs were significantly involved in the
wounding and inflammatory response BP functions. In addition, IL-6
was identified as a vital hub protein based on the PPI network.
The highly conserved let-7 miRNA family members are
downregulated in multiple types of cancer and the low expression of
let-7 is associated with poor prognosis (31,32).
By directly targeting oncogenes and inhibiting their expression
levels, let-7 can act as a tumor suppressor (33). In addition, let-7 miRNA may
directly limit cancer growth in the lung and the loss of its
function enhances lung tumor formation (34,35).
In humans, let-7 miRNAs consist of 12 family members (let-7a-1, −2,
−3; let-7b; let-7c; let-7d; let-7e; let-7f-1, −2; let-7g; let-7i;
miR-98), which are located at 8 different chromosomal loci and the
coordinate downregulation of multiple let-7 miRNAs has been
identified in various tumors (32,36).
In the present study, the 4 rno-let-7 family members (rno-let-7a,
rno-let-7b, rno-let-7c and rno-let-7f) were all downregulated in
ARDS rat lung tissues and had synergistic functions, suggesting
that they may synergistically suppress tumor growth associated with
ARDS.
Reduced expression levels of let-7 were detected in
lung cancer and were determined to be involved in the pathogenesis
of this fatal disease (37).
Through directly binding to the 3′-UTR of their target genes, the
let-7 family members are involved in the regulation of inflammatory
pathways and processes. A previous study indicated that let-7
participated in airway inflammation via the direct regulation of
IL-13, a cytokine that was associated with the mediation of
allergic inflammation (38,39).
Another cytokine, IL-6 was also reported to be targeted by let-7
and an inverse association between lower expression levels of
let-7a and higher levels of IL-6 was established in various cancer
types compared with normal breast, prostate, hepatocellular and
lung tissues (40). A feedback
loop involving nuclear factor-kB, Lin28 homolog A, let-7 and IL-6
is a critical part of the transient inflammatory signal pathway
(33). IL-6 is a cytokine protein
that contributes to inflammation and it has been demonstrated that
elevated IL-6 expression is closely associated with increased
mortality in critically ill patients with ARDS (41). IL-6 has been identified as a
potential predictor for the development of ARDS in adults with
severe blunt trauma (42). In
addition, the high expression levels of cytokines, such as IL-6 and
IL-8, were determined to be reliable markers for predicting the
development and the degree of severity of the systemic inflammatory
response, which may result in ARDS (43). The findings of the present study
indicated that IL-6, which was significantly enriched in the
inflammatory response BP, is a vital target gene of let-7 miRNAs.
Therefore, let-7 miRNAs may be involved in the inflammatory process
via the direct regulation of IL-6 during the progression of
ARDS.
In conclusion, the downregulated rno-let-7 miRNAs,
including rno-let-7a, rno-let-7b, rno-let-7c and rno-let-7f, may
have important roles in the inflammatory process during the
pathogenesis and progression of ARDS through the synergistic
regulation of their target genes, such as IL-6. These findings
provided a novel insight into the roles of miRNAs in the mediation
of ARDS development. However, further experimental experiments are
required to verify the association between IL-6 and rno-let-7
miRNAs, as the current findings were based only on microarray
data.
References
|
1
|
Udobi KF, Childs E and Touijer K: Acute
respiratory distress syndrome. Am Fam Physician. 67:315–322.
2003.PubMed/NCBI
|
|
2
|
Huang C, Xiao X, Chintagari NR, Breshears
M, Wang Y and Liu L: MicroRNA and mRNA expression profiling in rat
acute respiratory distress syndrome. BMC Med Genomics. 7:462014.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mikkelsen ME, Shah CV, Meyer NJ, Gaieski
DF, Lyon S, Miltiades AN, Goyal M, Fuchs BD, Bellamy SL and
Christie JD: The epidemiology of acute respiratory distress
syndrome in patients presenting to the emergency department with
severe sepsis. Shock. 40:375–381. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rubenfeld GD, Caldwell E, Peabody E,
Weaver J, Martin DP, Neff M, Stern EJ and Hudson LD: Incidence and
outcomes of acute lung injury. N Engl J Med. 353:1685–1693. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wyncoll DL and Evans TW: Acute respiratory
distress syndrome. Lancet. 354:497–501. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Matthay MA and Zemans RL: The acute
respiratory distress syndrome: Pathogenesis and treatment. Annu Rev
Pathol. 6:147–163. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Abraham E: Neutrophils and acute lung
injury. Crit Care Med. 31:(Suppl 4). S195–S199. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Grommes J and Soehnlein O: Contribution of
neutrophils to acute lung injury. Mol Med. 17:293–307. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Magi B, Bargagli E, Bini L and Rottoli P:
Proteome analysis of bronchoalveolar lavage in lung diseases.
Proteomics. 6:6354–6369. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Amos CI, Wu X, Broderick P, Gorlov IP, Gu
J, Eisen T, Dong Q, Zhang Q, Gu X, Vijayakrishnan J, et al:
Genome-wide association scan of tag SNPs identifies a
susceptibility locus for lung cancer at 15q25. 1. Nat Genetics.
40:616–622. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Potti A, Mukherjee S, Petersen R, Dressman
HK, Bild A, Koontz J, Kratzke R, Watson MA, Kelley M, Ginsburg GS,
et al: A genomic strategy to refine prognosis in early-stage
non-small-cell lung cancer. N Engl J Med. 355:570–580. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nana-Sinkam SP, Hunter MG, Nuovo GJ,
Schmittgen TD, Gelinas R, Galas D and Marsh CB: Integrating the
MicroRNome into the study of lung disease. Am J Respir Crit Care
Med. 179:4–10. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yanaihara N, Caplen N, Bowman E, Seike M,
Kumamoto K, Yi M, Stephens RM, Okamoto A, Yokota J, Tanaka T, et
al: Unique microRNA molecular profiles in lung cancer diagnosis and
prognosis. Cancer Cell. 9:189–198. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Angulo M, Lecuona E and Sznajder JI: Role
of MicroRNAs in lung disease. Arch Bronconeumol. 48:325–330.
2012.(In English, Spanish). View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Xu J, Li CX, Li YS, Lv JY, Ma Y, Shao TT,
Xu LD, Wang YY, Du L, Zhang YP, et al: MiRNA-miRNA synergistic
network: Construction via co-regulating functional modules and
disease miRNA topological features. Nucleic Acids Res. 39:825–836.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ha TY: MicroRNAs in human diseases: From
lung, liver and kidney diseases to infectious disease, sickle cell
disease and endometrium disease. Immune Netw. 11:309–323. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Small EM and Olson EN: Pervasive roles of
microRNAs in cardiovascular biology. Nature. 469:336–342. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Carraro G, El-Hashash A, Guidolin D,
Tiozzo C, Turcatel G, Young BM, De Langhe SP, Bellusci S, Shi W,
Parnigotto PP and Warburton D: miR-17 family of microRNAs controls
FGF10-mediated embryonic lung epithelial branching morphogenesis
through MAPK14 and STAT3 regulation of E-Cadherin distribution. Dev
Biol. 333:238–250. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lin HY, Chiang CH and Hung WC: STAT3
upregulates miR-92a to inhibit RECK expression and to promote
invasiveness of lung cancer cells. Br J Cancer. 109:731–738. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bhaskaran M, Wang Y, Zhang H, Weng T,
Baviskar P, Guo Y, Gou D and Liu L: MicroRNA-127 modulates fetal
lung development. Physiol Genomics. 37:268–278. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hastie T, Tibshirani R, Narasimhan B and
Chu G: Impute: Imputation for microarray data. R package. version
1.44.0. 2001.
|
|
22
|
Bolstad B: PreprocessCore: A collection of
pre-processing functions. R package. version 1. 2013.
|
|
23
|
Smyth GK: Limma: Linear Models for
Microarray DataBioinformatics and Computational Biology Solutions
Using R and Bioconductor. Springer; New York, NY: pp. 397–420.
2005, View Article : Google Scholar
|
|
24
|
Hochberg Y and Benjamini Y: More powerful
procedures for multiple significance testing. Stat Med. 9:811–818.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Dweep H, Sticht C, Pandey P and Gretz N:
miRWalk-database: Prediction of possible miRNA binding sites by
‘walking’ the genes of three genomes. J Biomed Inform. 44:839–847.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shannon P, Markiel A, Ozier O, Baliga NS,
Wang JT, Ramage D, Amin N, Schwikowski B and Ideker T: Cytoscape: A
software environment for integrated models of biomolecular
interaction networks. Genome Res. 13:2498–2504. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yu G, Wang LG, Han Y and He QY:
clusterProfiler: An R package for comparing biological themes among
gene clusters. OMICS. 16:284–287. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dennis G Jr, Sherman BT, Hosack DA, Yang
J, Gao W, Lane HC and Lempicki RA: DAVID: Database for annotation,
visualization, and integrated discovery. Genome Biol. 4:P32003.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Franceschini A, Szklarczyk D, Frankild S,
Kuhn M, Simonovic M, Roth A, Lin J, Minguez P, Bork P, von Mering C
and Jensen LJ: STRING v9. 1: Protein-protein interaction networks,
with increased coverage and integration. Nucleic Acids Res.
41:D808–D815. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Di Leva G and Croce CM: Roles of small
RNAs in tumor formation. Trends Mol Med. 16:257–267. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Roush S and Slack FJ: The let-7 family of
microRNAs. Trends Cell Biol. 18:505–516. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Piskounova E, Polytarchou C, Thornton JE,
LaPierre RJ, Pothoulakis C, Hagan JP, Iliopoulos D and Gregory RI:
Lin28A and Lin28B inhibit let-7 microRNA biogenesis by distinct
mechanisms. Cell. 147:1066–1079. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Iliopoulos D, Jaeger SA, Hirsch HA, Bulyk
ML and Struhl K: STAT3 activation of miR-21 and miR-181b-1 via PTEN
and CYLD are part of the epigenetic switch linking inflammation to
cancer. Mol Cell. 39:493–506. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Trang P, Medina PP, Wiggins JF, Ruffino L,
Kelnar K, Omotola M, Homer R, Brown D, Bader AG, Weidhaas JB and
Slack FJ: Regression of murine lung tumors by the let-7 microRNA.
Oncogene. 29:1580–1587. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Esquela-Kerscher A, Trang P, Wiggins JF,
Patrawala L, Cheng A, Ford L, Weidhaas JB, Brown D, Bader AG and
Slack FJ: The let-7 microRNA reduces tumor growth in mouse models
of lung cancer. Cell Cycle. 7:759–764. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Shell S, Park SM, Radjabi AR, Schickel R,
Kistner EO, Jewell DA, Feig C, Lengyel E and Peter ME: Let-7
expression defines two differentiation stages of cancer. Proc Natl
Acad Sci USA. 104:11400–11405. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Takamizawa J, Konishi H, Yanagisawa K,
Tomida S, Osada H, Endoh H, Harano T, Yatabe Y, Nagino M, Mitsudomi
T and Takahashi T: Reduced expression of the let-7 microRNAs in
human lung cancers in association with shortened postoperative
survival. Cancer Res. 64:3753–3756. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhu Z, Zheng T, Homer RJ, Kim YK, Chen NY,
Cohn L, Hamid Q and Elias JA: Acidic mammalian chitinase in
asthmatic Th2 inflammation and IL-13 pathway activation. Science.
304:1678–1682. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kumar M, Ahmad T, Sharma A, Mabalirajan U,
Kulshreshtha A, Agrawal A and Ghosh B: Let-7 microRNA-mediated
regulation of IL-13 and allergic airway inflammation. J Allergy
Clin Immunol. 128:1077–1085, e1-e10. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Iliopoulos D, Hirsch HA and Struhl K: An
epigenetic switch involving NF-kappaB, Lin28, Let-7 MicroRNA, and
IL6 links inflammation to cell transformation. Cell. 139:693–706.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Marshall RP, Webb S, Hill MR, Humphries SE
and Laurent GJ: Genetic polymorphisms associated with
susceptibility and outcome in ARDS. Chest. 121:(3 Suppl). 68S–69S.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Kasotakis G, Kromer M, Narsule C, Sideris
A, Klein E, Tompkins R, Velmahos G and Burke P: 349: Interleukin-6
predicts acute respiratory distress syndrome in adults with severe
blunt trauma. Crit Care Med. 41:A822013. View Article : Google Scholar
|
|
43
|
Volpin G, Cohen M, Assaf M, Meir T, Katz R
and Pollack S: Cytokine levels (IL-4, IL-6, IL-8 and TGFβ) as
potential biomarkers of systemic inflammatory response in trauma
patients. Int Orthop. 38:1303–1309. 2014. View Article : Google Scholar : PubMed/NCBI
|