Introduction
Osteoarthritis (OA) is the most common type of
arthritis, characterized by pain and loss of joint function
(1). Knee joint OA has been
reported to affect 3.64% of the population worldwide in 2010
(2). Currently, there is no
effective disease-modifying therapy for OA. Pain management and
joint replacement are the options for end-stage OA (3,4). The
pathogenesis of OA remains unclear and an improved understanding is
required in order to target the prevention and effective
intervention for early-stage OA (5).
The transforming growth factor-β (TGF-β) superfamily
consists of >35 members, including TGF-β1, activins and bone
morphogenetic proteins (6). They
have an active role in the development and homeostasis of various
tissues. Previous in vivo and ex vivo studies have
reported that TGF-β1 is crucial for the formation of articular
cartilage during development. It may increase the production of
proteoglycan and collagen type II (Col II). Lack of TGF-β1 may
induce a normal joint into the OA-phenotype (7,8).
However, previous studies also determined that TGF-β also exerts
deleterious effect in adult joints. Massicotte et al noticed
increased TGF-β level in human osteoarthritic osteoblasts compared
with normal cells (9).
Additionally, high levels of TGF-β were found in OA cartilage when
compared with normal ones in humans (10). Moldovan et al reported that
TGF-β may lead to result elevated levels of matrix metallopeptidase
(MMP)-13 in normal cartilage, which mimicked the in situ
distribution that observed in arthritic cartilage (11). Multiple intra-articular injections
of TGF-β1in normal adult mice joints may induce inflammation and
osteophyte formation, leading to the onset of OA (12,13).
Halofuginone (HF) is an analogue of febrifugine,
which is an alkaloid originally isolated from the plant Dichroa
febrifuga (14). HF is used in
commercial poultry production worldwide (15). Increased attention has been paid to
this small molecule for its beneficial biological activity. HF has
been identified as crucial in dealing with fibrosis and fibroblasts
to myofibroblasts transition by inhibiting SMAD family member 2/3
(Smad2/3) phosphorylation downstream of the TGF-β signaling pathway
(16,17). HF has been demonstrated to be
effective in treating muscle fibrosis in Duchenne muscular
dystrophy and chronic graft-versus host disease (18,19).
The present study aimed to investigate whether HF
could attenuate the progression of OA by preventing articular
cartilage degeneration, which may add to its potential clinical
application.
Materials and methods
Cell culture
The ATDC5 murine chondrogenic cell line (Riken Cell
Bank, Tsukuba, Japan), were plated at a density of
1.2×104 cells/cm2 in 6-well plate (Corning,
Inc., Corning, NY, USA). The cells were cultured with maintenance
medium, which was Dulbecco's modified Eagle's medium/F12
(11320–033; Gibco; Thermo Fisher Scientific, Inc., Waltham, MA,
USA) containing 5% fetal bovine serum (10099–141; Gibco; Thermo
Fisher Scientific, Inc.) and 1% penicillin-streptomycin (SV30010;
GE Healthcare Life Sciences, Logan, UT, USA), at 37°C in a
humidified atmosphere of 5% CO2. The maintenance medium
and it was exchanged every other day. The differentiation medium,
where 1% Insulin-Transferrin-Selenium (41400–045; Gibco; Thermo
Fisher Scientific, Inc.) was added to the maintenance medium, which
was used when the cells became confluent. The differentiation
medium was changed every other day.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
ATDC5 cells were cultured with differentiation
medium for 7, 14 and 21 days. Total RNA was extracted using TRIzol
reagent (15596026, Invitrogen; Thermo Fisher Scientific, Inc.) and
cDNA was synthesized using PrimeScript RT Master mix (RR036A;
Takara Bio, Inc., Otsu, Japan) Three or more different samples were
used. qPCR reactions were performed using SYBR Fast qPCR mix
(RR430A, Takara Bio, Inc.) according to the manufacturer's
protocol. Amplification curves of samples were converted into
relative expression values according to the curves of standard
controls. Relative quantification of each gene was normalized
against GAPDH. The 2−∆∆Cq method was used to calculate
relative gene expression levels (20). The sequences of the following
primers were used: GAPDH forward (F) 5′-AGCTTCGGCACATATTTCATCTG-3′
and reverse (R) 5′-CGTTCACTCCCATGACAAACA-3′; Col II F
5′-ACGAAGCGGCTGGCAACCTCA-3 and R 5′-CCCTCGGCCCTCATCTCTACATCA-3; Col
X F 5′-TGCCCGTGTCTGCTTTTACTGTCA-3 and R
5′-TCAAATGGGATGGGGGCACCTACT-3. Amplification cycle was set as 94°C
for 30 sec, followed by 40 cycles at 95°C for 5 sec, 60°C for 10
sec, and finally a melt curve was inserted with 65°C to 95°C.
Cell Counting Kit-8 (CCK-8)
analysis
The toxicity of HF on chondrocytic ATDC5 cells was
observed using the CCK-8 method. After 14 days of chondrogenic
differentiation, ATDC5 cells were seeded in 96-well plates with a
density of 4×103 cells/well and incubated for 24 h. The
cells were treated with different concentrations of HF (0, 6.25,
12.5, 25, 50, 100 and 200 ng/ml). CCK-8 (CK04; Dojindo Molecular
Technologies, Inc., Kumamoto, Japan) was applied to monitor the
cell viability after 24, 48 and 72 h and the percentage of cell
viability was calculated based on the absorbance at 450 nm after 3
h incubation using a microplate reader (51119300; Thermo Fisher
Scientific, Inc.).
Western blot analysis
After 14 days of chondrogenic differentiation, ATDC5
cells were treated with HF (17395–31-2; Watson Noke Scientific
Ltd., Kunshan, China) at different doses 0, 6.25, 12.5 and 25 ng/ml
for 6 h, and were treated with 2 ng/ml rhTGF-β1 (240-B-002; R&D
Systems, Inc., Minneapolis, MN, USA) for 30 min and processed for
western blotting. Then the time-course experiment was performed.
After 14 days of chondrogenic differentiation, ATDC5 cells were
treated without HF for 0 h, or with 25 ng/ml HF for 4, 8 and 24 h.
Then cells were treated with either 2 ng/ml rhTGF-β1 or the reagent
used to reconstitute rhTGF-β1 (sterile 4 mM HCl containing 1 mg/ml
bovine serum albumin) in an equal volume for 30 min and processed
for western blotting. Samples (20 µg) were subjected to 10%
SDS-PAGE (4561083; Bio-Rad Laboratories Inc., Hercules, CA, USA)
and transferred onto polyvinylidene fluoride (PVDF) membranes
(ISEQ00010; Merck KGaA, Darmstadt, Germany). Membranes were blocked
in 5% bovine serum albumin (AD0023; Sangon Biotech Co., Ltd.,
Shanghai, China) for 1 h, incubated with primary antibodies against
Smad2/3 (1:1,000; cat. no. 3102S; Cell Signaling Technology Inc.,
Danvers, MA, USA), phosphorylated (p)-Smad2 (1:1,000; cat. no.
3108; Santa Cruz Biotechnology Inc., Dallas, TX, USA) and GAPDH
(1:1,000; cat. no. 2118S; Cell Signaling Technology Inc.) at 4°C
overnight, followed by incubation with secondary antibody
(peroxidase-conjugated AffiniPure goat anti-rabbit IgG (H+L);
1:5,000; cat no. ZB-2301; OriGene Technologies, Inc., Beijing,
China) for 2 h. Protein was visualized using Pierce Fast Western
Blot kit, enhanced chemiluminescence substrate (cat. no. 35055;
Thermo Fisher Scientific, Inc.).
Experimental animals
C57BL/6 male mice (3-month old, 24–25 g, 22 in
total) were purchased from Beijing Vital River Laboratory Animal
Technology Co., Ltd. (Beijing, China). The mice were maintained in
an animal room on a 12-h light/dark cycle with a temperature and
humidity of 25±2°C and 55%, respectively. The mice were provided
with food and water ad libitum. The anterior cruciate ligament of
the right knee was transected to establish a destabilized OA animal
model. Sham operation was performed on independent mice by opening
the knee joint capsule and suturing the incision in the right knee
joint. Mice were divided into Sham, vehicle-treated anterior
cruciate ligament transection (ACLT) group and HF-treated ACLT
group (n=6–8 per group). Either HF (0.25 mg/kg) or distilled water
of equivalent volume (0.2 ml) was administered by oral gavage every
other day for 30 days since the second day post-operation. Mice
were sacrificed at day 30 post-operatively. All animal experiment
protocols were reviewed and approved by the Institutional Animal
Care and Use Committee of First Affiliated Hospital of Xinjiang
Medical University (Xinjiang, China).
Histochemistry, immunohistochemistry
and histomorphometry
When the mice were sacrificed, the right knee joints
were resected and fixed in 10% buffered formalin for 24 h. The
specimen was decalcified in 10% EDTA (pH 7.3) for 21 days and
embedded in paraffin sagittally. Serial sections at 4-µm thickness
of the medial compartment of the knee joint were processed for
Safranin O and Fast Green staining. The sections were incubated
with primary antibodies against MMP-13 (1:100; cat. no. ab39012;
Abcam; Cambridge, UK), Col X (1:100; cat. no. ab58632; Abcam),
TGF-β1 (1:100; cat. no. ab92486; Abcam) and p-Smad2/3 (1:40; cat.
no. sc-11769; Santa Cruz Biotechnology Inc.) overnight at 4°C.
Subsequently a horseradish peroxidase-streptavidin detection system
(1:1,000; cat no. ZB-2301 and ZB-2306; OriGene Technologies, Inc.,
Beijing, China) was used to detect the immunoactivity at room
temperature for 2 h, followed by counterstaining with hematoxylin
(OriGene Technologies, Inc.). The number of all chondrocytes and
positively stained ones were calculated by eye in the entire
articular cartilage using an optical microscope (version
510_UMA_cellSens19-Krishna-ch_00_01August 2013, Olympus
Corporation, Tokyo, Japan). The Osteoarthritis Research Society
International (OARSI)-Modified Mankin score was calculated as
previously described by Pritzker et al (21).
Statistical analysis
Data are presented as the mean ± standard deviation.
One-way analysis of variance followed by the Least-Significant
Difference post hoc test was used to determine if the difference
among different groups was statistically significant. SPSS version
22.0 (IBM Corporation, Armonk, NY, USA) was used for data analysis.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Chondrogenic differentiation of ATDC5
cells
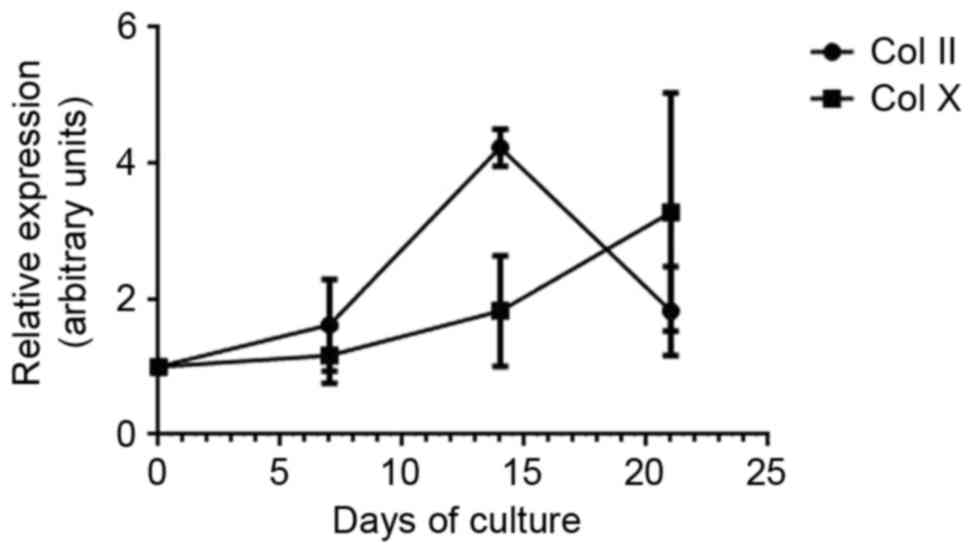
To examine the chondrogenic differentiation of ATDC5
cells, expression of Col II and Col X were analyzed by RT-qPCR.
Expression of Col II increased from day 1 and peaked at day 14,
indicating the early-stage differentiation of chondrocytes. Col X
expression gradually increased from day 7 to day 21, which
indicated hypertrophic and calcified chondrocytes (Fig. 1).
Toxicity of HF on chondrocytic ATDC5
cells
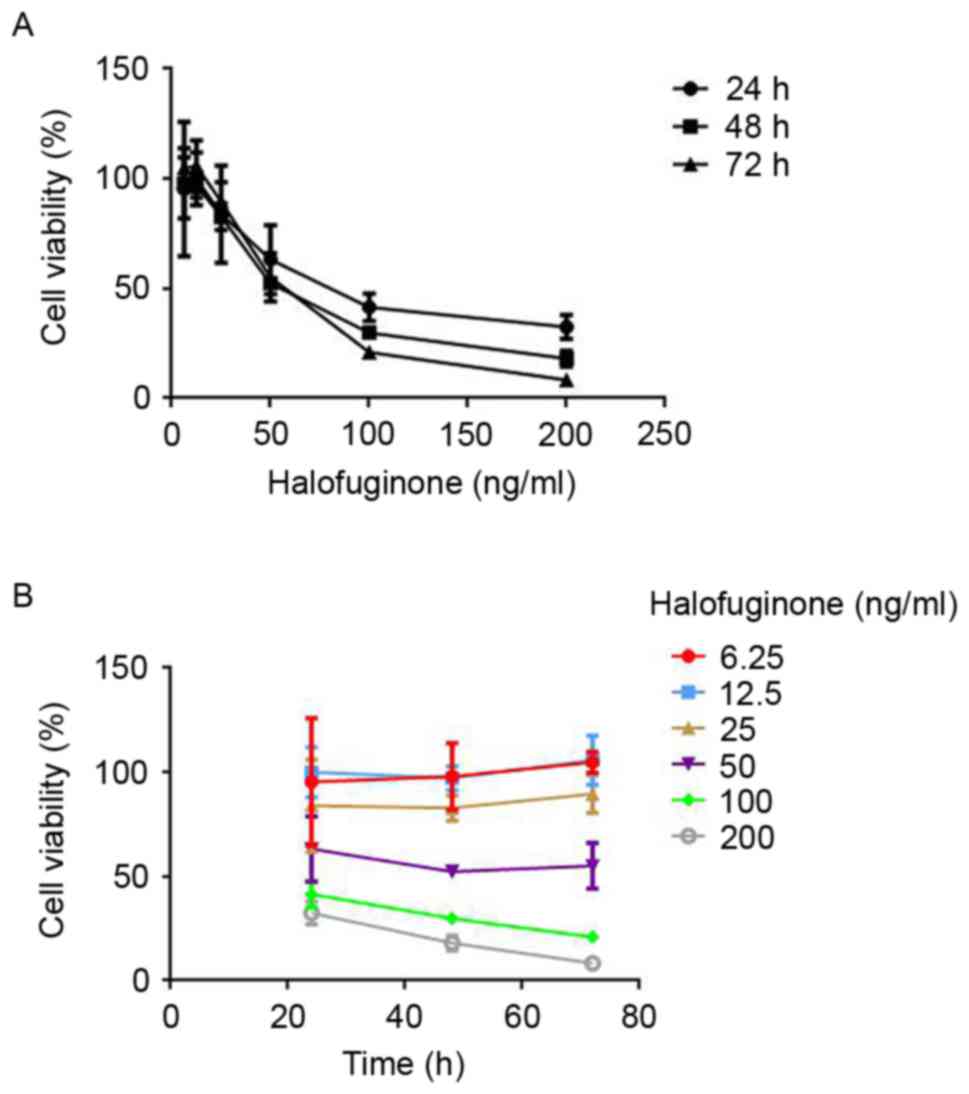
In order to determine the appropriate dosage of HF
for the subsequent in vitro study, toxicity of HF on
chondrocytic ATDC5 cells was tested using CCK-8. Following
incubation with HF of different concentrations for 24, 48 and 72 h,
a dose-dependent and time-dependent decrease in cell viability was
observed. Cell viability was >84% after exposure to HF of 6.25,
12.5 and 25 ng/ml for 24 h. However, it was reduced to 63%
following treatment with 50 ng/ml HF and it was even lower in
groups treated with 100 and 200 ng/ml HF (Fig. 2A). From 24 to 72 h of exposure,
6.25, 12.5 and 25 ng/ml HF had a negligible effect on the cell
viability. As for HF concentrations of 50, 100 and 200 ng/ml, the
cell viability decreased in a time-dependent manner (Fig. 2B). These findings suggested that HF
of 6.25, 12.5 and 25 ng/ml did not affect chondrocytic ATDC5 cell
viability.
Halofuginone inhibited TGF-β1
signaling in chondrocytic ATDC5 cells
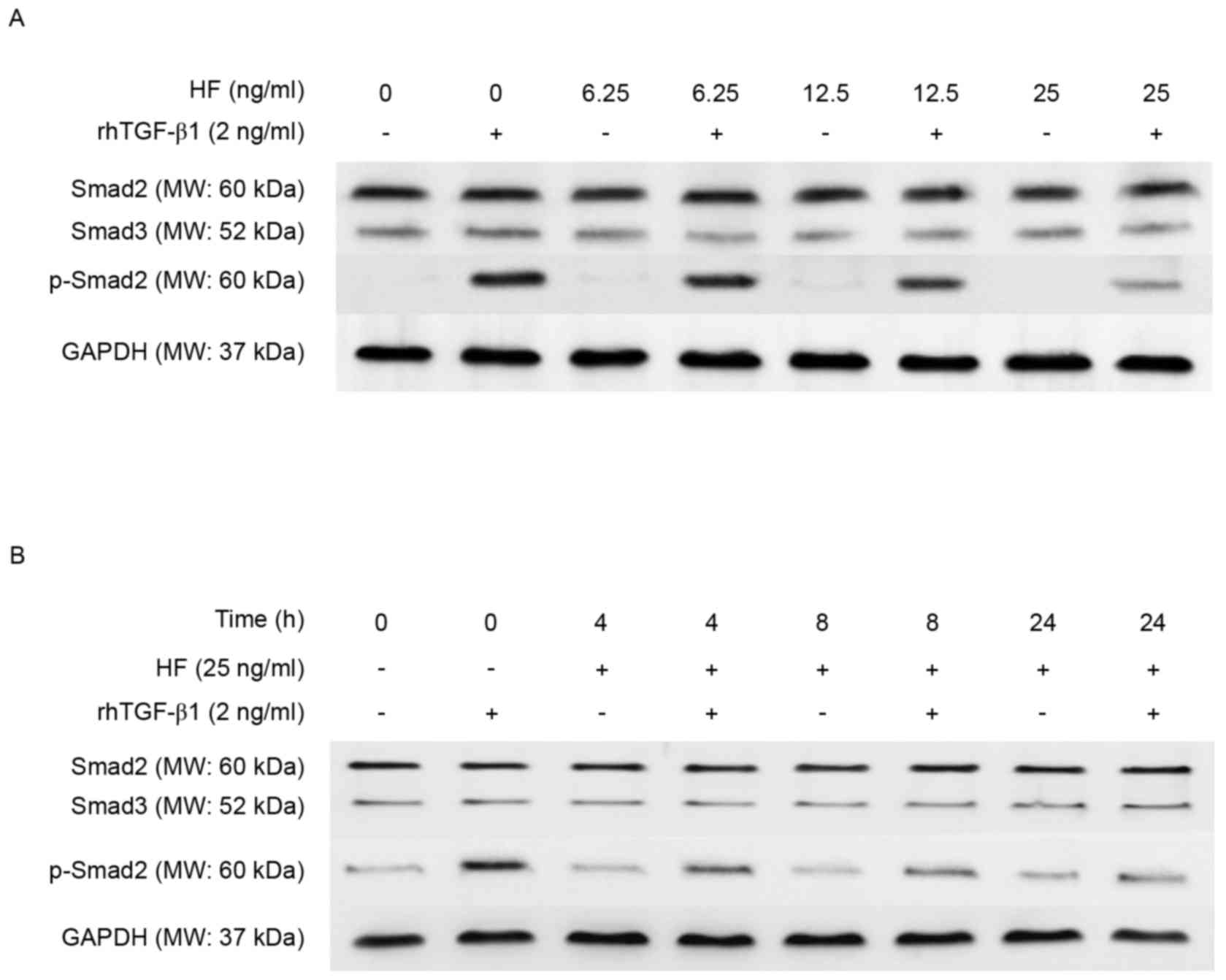
To determine whether HF has a role in regulating
TGF-β1 signaling in chondrocytic ATDC5 cells, ATDC5 cells that
underwent differentiation for 14 days were used. Dose and
time-dependent experiments were performed followed by western
blotting. The level of p-Smad2 protein was reduced in a does
dependent manner, whereas the level of total Smad2/3 remained
unchanged (Fig. 3A). Next, the
present study determined the effect of HF pretreatment of
chondrocytic ATDC5 cells at three different time points (4, 8 and
24 h) on the phosphorylation of Smad2 protein in response to
TGF-β1. The level of p-Smad2 was reduced with time, whereas the
level of total Smad2/3 remained unchanged (Fig. 3B).
Halofuginone prevented articular
cartilage degeneration by inhibiting elevated TGF-β1 signaling
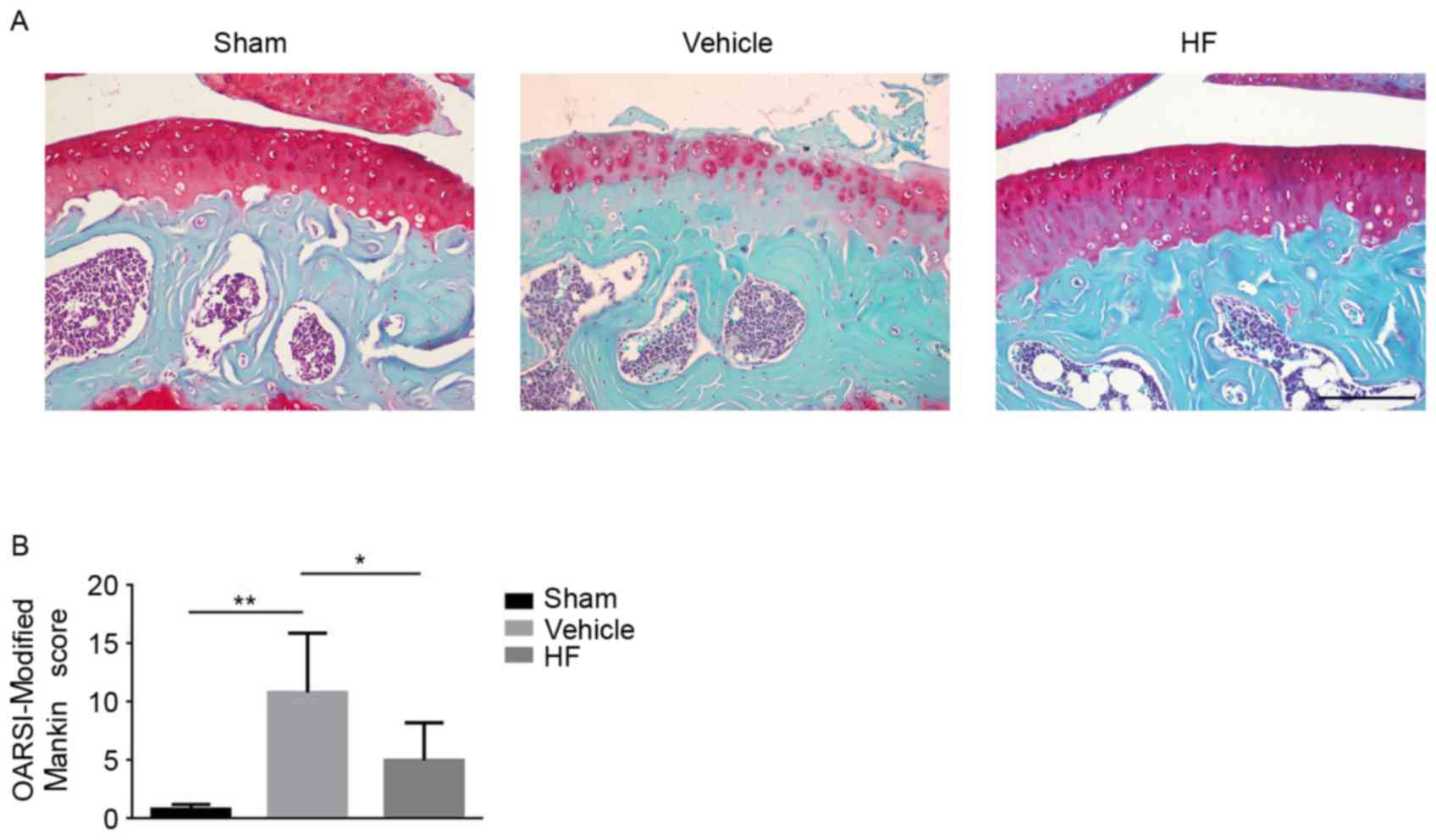
To investigate whether HF can attenuate OA
progression by way of oral gavage an ACLT mice model was
established and HF was administered every other day. Safranin O
Fast Green staining was applied to assess the severity of articular
cartilage degeneration. Deparaffinization of the slides was
performed in xylene three times for 5 min, followed by hydration in
100% alcohol twice for 5 min, 95% alcohol for 5 min and 80% alcohol
for 5 min. Hematoxylin was added to slides for 1 min prior to
hydrating the slides gently in running water for 10 min. Slides
were then stained with 0.2% Fast Green for 5 min, and then
subjected to 1% acetic acid for 10 sec, 0.1% Safranin O for 2 min
and rinsing in water for 30 sec. Slides were hydrated in 95%
alcohol for 15 sec, 100% alcohol twice for 15 sec, followed by 3
changes in xylene prior to cover-slipping the slides. Proteoglycan
loss in HF-treated group was reduced compared with the
vehicle-treated group (Fig. 4A).
Furthermore, the severity of articular cartilage degeneration was
quantitatively evaluated using OARSI-Modified Mankin score.
HF-treated group had a lower score compared with the
vehicle-treated group, indicating reduced articular cartilage
damage (Fig. 4B). Additionally,
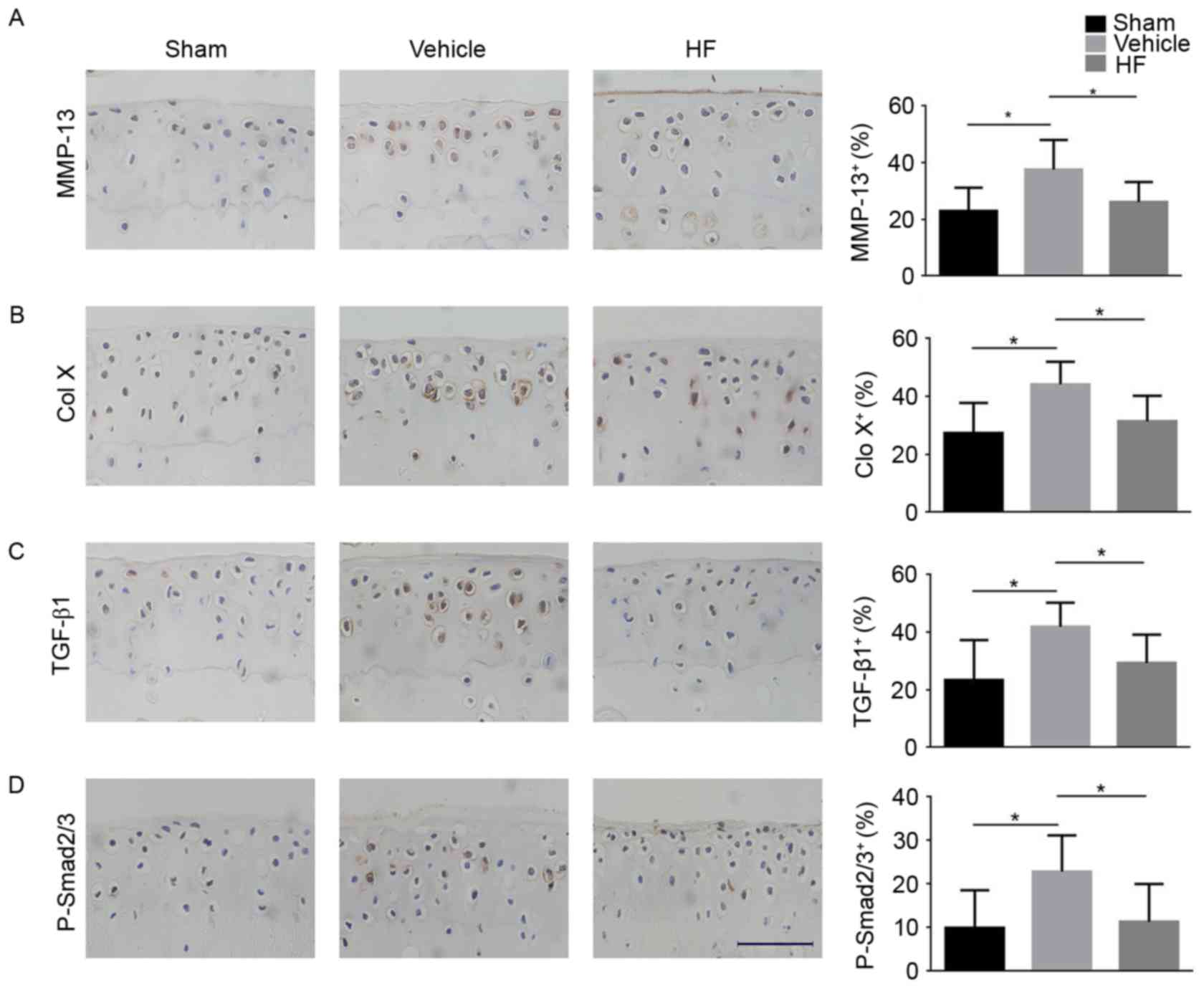
the expression of MMP-13 and Col X was lower in the HF-treated
group when compared with the vehicle-treated group (Fig. 5A and B), indicating that HF had a
protective role in articular cartilage degeneration. The present
study aimed to investigate whether TGF-β1 had a role in the onset
of OA. It was determined that the protein expression of TGF-β1 in
articular cartilage was higher in the vehicle-treated group
compared with the Sham group, and HF reduced its expression
(Fig. 5C). Additionally, similar
results were obtained for the expression of p-Smad2/3 in articular
cartilage. HF-treated group had reduced expression of p-Smad2/3
compared with the vehicle-treated group (Fig. 5D). Altogether, these findings
suggest HF attenuated articular cartilage degeneration by
inhibiting elevated TGF-β1 signaling in articular cartilage.
Discussion
The present study transected the anterior cruciate
ligament of C57BL/6 male mice to generate an unstable mechanical
loading OA model. Compared with vehicle-treated group, HF
administration attenuated OA progression by preserving articular
cartilage. The underlying mechanisms of this phenomenon may be
associated with its inhibitory effects on the elevated
Smad2/3-dependent TGF-β1 pathway in articular cartilage during the
onset of OA.
OA is a joint disorder characterized by articular
cartilage degeneration, subchondral bone sclerosis, inflammation
and osteophyte formation (22,23).
Articular cartilage has a very limited reparative capacity, which
makes it important to prevent cartilage damage at early stage.
Articular cartilage is highly specialized connective tissue that
transfers loads during weight bearing and joint motion (24,25).
It primarily consists of extracellular matrix and chondrocytes.
Chondrocytes are embedded in the extracellular matrix, producing a
large number of collagenous extracellular matrix, including
proteoglycan and Col II. Proteoglycan and Col II maintain the
homeostasis and integrity of articular cartilage. During OA, the
chondrocytes become hypertrophic and synthesize proinflammatory
cytokines that contribute to their own destruction (26,27).
Col X is a classic marker of hypertrophic differentiation of
chondrocytes (28). MMP-13 is the
primary collagenase synthesized by chondrocytes to damage aggrecan
and Col II during OA. The OARSI-Modified Mankin scoring system is
used to assess the severity of cartilage damage (21). The present study identified the
that HF treatment reduced the expression of Col X and MMP-13 in
cartilage, lowered the OARSI score when compared with the
vehicle-treated group. These findings demonstrated that HF improved
the homeostasis and integrity of articular cartilage.
The role of TGF-β1 in articular cartilage is
controversial. The reason for the conflicting role of TGF-β1 in
articular cartilage remains to be elucidated. It has been
previously reported that TGF-β1 is critical for the development of
the knee joint. Serra et al used transgenic mice where the
type II TGF-β receptor was truncated (29). They identified the onset of early
stage OA in the transgenic mice. At the age of 4 and 8 weeks,
higher expression levels of Col X and lower levels of proteoglycan
were detected in the transgenic mice when compared with their wild
type littermates (29). Yang et
al noticed OA characteristics in Smad3-deficient mice,
including loss of articular cartilage, osteophyte formation and
elevated expression of Col X (30). These previous studies suggested
that TGF-β1 is important for the chondrocyte hypertrophic
differentiation in immature articular cartilage. Data from in
vitro studies revealed that TGF-β1 may inhibit hypertrophic
differentiation of chondrocytes through the regulation of articular
cartilage matrix proteins and metalloproteases (31,32).
These investigations support the view that TGF-β1 is a necessary
protector against the onset of OA during the development of knee
joints. Nevertheless, TGF-β1 may also contribute to the joint
destruction. High level of TGF-β1 was detected in the articular
cartilage of OA patients and mice OA models (33,34).
The association between elevated TGF-β1 level and the onset of OA
remains to be elucidated. The present study revealed that HF may
inhibit Smad2/3-dependent TGF-β1 signaling in chondrocytes in
vitro. In adult mice, compared with sham group, elevated levels
of TGF-β1 and p-Smad2/3 were noticed in the vehicle-treated group
and this was alleviated in the group treated with HF. The
aforementioned findings suggested that HF prevented the cartilage
damage during early-stage OA by inhibition of the elevated TGF-β1
signaling in articular cartilage.
HF has been used in a clinical trial for the
treatment of chronic graft-versus host diseases and solid tumors,
and had safe therapeutic effectiveness through oral application
(35,36). The findings of the present study
may add to its clinical application. To the best of our knowledge,
the present study is the first to indicate that HF, by way of oral
gavage, improved the homeostasis and integrity of articular
cartilage. The underlying mechanism may be due to the
downregulation of abnormally elevated TGF-β1 signaling in the
articular cartilage. These findings indicated that HF, administered
by oral gavage and may be an effective strategy to prevent the
onset of OA.
Acknowledgements
The present study was supported by grants from Major
Science and Technology Projects in Xinjiang Uygur Autonomous Region
(grant no. 201430123-3) and Joint Funds of the National Natural
Science Foundation of China (grant no. U1503221).
References
|
1
|
McAlindon TE, Bannuru RR, Sullivan MC,
Arden NK, Berenbaum F, Bierma-Zeinstra SM, Hawker GA, Henrotin Y,
Hunter DJ, Kawaguchi H, et al: OARSI guidelines for the
non-surgical management of knee osteoarthritis. Osteoarthritis
Cartilage. 22:363–388. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Vos T, Flaxman AD, Naghavi M, Lozano R,
Michaud C, Ezzati M, Shibuya K, Salomon JA, Abdalla S, Aboyans V,
et al: Years lived with disability (YLDs) for 1160 sequelae of 289
diseases and injuries 1990–2010: A systematic analysis for the
global burden of disease study 2010. Lancet. 380:2163–2196. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Carr AJ, Robertsson O, Graves S, Price AJ,
Arden NK, Judge A and Beard DJ: Knee replacement. Lancet.
379:1331–1340. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bijlsma JW, Berenbaum F and Lafeber FP:
Osteoarthritis: An update wit relevance for clinical practice.
Lancet. 377:2115–2126. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Van den Berg WB: Osteoarthritis year 2010
in review: Pathomechanisms. Osteoarthritis Cartilage. 19:338–341.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
de Caestecker M: The transforming growth
factor-beta superfamily of receptors. Cytokine Growth Factor Rev.
15:1–11. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Galera P, Vivien D, Pronost S, Bonaventure
J, Redini F, Loyau G and Pujol JP: Transforming growth factor-beta
1 (TGFbeta 1) up-regulation of collagen type II in primary cultures
of rabbit articular chondrocytes (RAC) involves increased mRNA
levels without affecting mRNA stability and procollagen processing.
J Cell Physiol. 153:596–606. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shen J, Li J, Wang B, Jin H, Wang M, Zhang
Y, Yang Y, Im HJ, O'Keefe R and Chen D: Deletion of the
transforming growth factor β receptor type II gene in articular
chondrocytes leads to a progressive osteoarthritis-like phenotype
in mice. Arthritis Rheum. 65:3107–3119. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Massicotte F, Lajeunesse D, Benderdour M,
Pelletier JP, Hilal G, Duval N and Martel-Pelletier J: Can altered
production of interleukin-1β, interleukin-6, transforming growth
factor-β and prostaglandin E (2) by isolated human subchondral
osteoblasts identify two subgroups of osteoarthritic patients.
Osteoarthritis Cartilage. 10:491–500. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Pombo-Suarez M, Castano-Oreja MT, Calaza
M, Gomez-Reino J and Gonzalez A: Differential upregulation of the
three transforming growth factor beta isoforms in human
osteoarthritic cartilage. Ann Rheum Dis. 68:568–571. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Moldovan F, Pelletier JP, Hambor J,
Cloutier JM and Martel-Pelletier J: Collagenase-3 (matrix
metalloprotease 13) is preferentially localized in the deep layer
of human arthritic cartilage in situ: In vitro mimicking effect by
transforming growth factor beta. Arthritis Rheum. 40:1653–1661.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
van Beuningen HM, Van Der Kraan PM, Arntz
OJ and van den Berg WB: Transforming growth factor-beta 1
stimulates articular chondrocyte proteoglycan synthesis and induces
osteophyte formation in the murine knee joint. Lab Invest.
71:279–290. 1994.PubMed/NCBI
|
|
13
|
van Beuningen HM, Glansbeek HL, van der
Kraan PM and van den Berg WB: Osteoarthritis-like changes in the
murine knee joint resulting from intra-articular transforming
growth factor-β injections. Osteoarthris Cartilage. 8:25–33. 2000.
View Article : Google Scholar
|
|
14
|
Pines M and Nagler A: Halofuginone: A
novel antifibrotic therapy. Gen Pharmacol. 30:445–450. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pinion JL, Bilgili SF, Eckman MK and Hess
JB: The effects of halofuginone and salinomycin, alone and in
combination, on live performance and skin characteristics of
broilers. Poult Sci. 74:391–397. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pines M: Targeting TGFβ signaling to
inhibit fibroblasts activation as a therapy for fibrosis and
cancer: Effect of halofuginone. Expert Opin Drug Discov. 3:11–20.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pines M: Halofuginone for fibrosis,
regeneration and cancer in the gastrointestinal tract. World J
Gastroenterol. 20:14778–14786. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
McLoon LK: Focusing on fibrosis:
Halofuginone-induced functional improvement in the mdx mouse model
of Duchenne muscular dystrophy. Am J Physiol Heart Circ Physiol.
294:H1505–H1507. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pines M, Snyder D, Yarkoni S and Nagler A:
Halofuginone to treat fibrosis in chronic graft-versus-host disease
and scleroderma. Biol Blood Marrow Transplant. 9:417–425. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Pritzker KP, Gay S, Jimenez SA, Ostergaard
K, Pelletier JP, Revell PA, Salter D and van den Berg WB:
Osteoarthritis cartilage histopathology: Grading and staging.
Osteoarthritis Cartilage. 14:13–29. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Robinson WH, Lepus CM, Wang Q, Raghu H,
Mao R, Lindstrom TM and Sokolove J: Low-grade inflammation as a key
mediator of the pathogenesis of osteoarthritis. Nat Rev Rheumatol.
12:580–592. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Goldring SR and Golding MB: Changes in the
osteochondral unit during osteoarthritis: Structure, function and
cartilage-bone crosstalk. Nat Rev Rheumatol. 12:632–644. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lories RJ and Luyten FP: The
bone-cartilage unit in osteoarthritis. Nat Rev Rheumatol. 7:43–49.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Madry H, van Dijk CN and Mueller-Gerbl M:
The basic science of the subchondral bone. Knee Surg Sports
Traumatol Arthrosc. 18:419–433. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Luyten FP, Lories RJ, Verschueren P, de
Vlam K and Westhovens R: Contemporaty concepts of inflammation,
damage and repair in rheumatic disease. Best Pract Res Clin
Rheumatol. 20:829–848. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Goldring MB and Goldring SR: Articular
cartilage and subchondral bone in the pathogenesis of
osteoarthritis. Ann N Y Acad Sci. 1192:230–237. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
von der Mark K, Kirsch T, Nerlich A, Kuss
A, Weseloh G, Gluckert K and Stoss H: Type X collagen synthesis in
human osteoarthritic cartilage. Indication of chondrocyte
hypertrophy. Arthritis Rheum. 35:806–811. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Serra R, Johnson M, Filvaroff EH, LaBorde
J, Sheehan DM, Derynck R and Moses HL: Expression of a truncted,
kinase-defective TGF-beta type II receptor in mouse skeletal tissue
promotes terminal chondrocyte differentiation and osteoarthritis. J
Cell Biol. 139:541–552. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yang X, Chen L, Xu X, Li C, Huang C and
Deng CX: TGF-beta/Smad3 signals repress chondrocyte hypertrophic
differentiation and are required for maintaining articular
cartilage. J Cell Biol. 153:35–46. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ballock RT, Heydemann A, Wakefield LM,
Flanders KC, Roberts AB and Sporn MB: TGF-beta 1 prevents
hypertrophy of epiphyseal chondrocytes: Regulation of gene
expression for cartilage matrix proteins and metalloproteases. Dev
Biol. 158:414–429. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Dreier R: Hypertrophic differentiation of
chondrocytes in osteoarthritis: The development aspect of
degenerative joint disorders. Arthritis Res Ther. 12:2162010.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Schlaak JF, Pfers I, Zum Buschenfelde KH
Meyer and Marker-Hermann E: Different cytokine profiles in the
synovial fluid of patients with osteoarthritis, rheumatoid
arthritis and seronegative spondylarthropathies. Clin Exp
Rheumatol. 14:155–162. 1996.PubMed/NCBI
|
|
34
|
Xu L, Golshirazian I, Asbury BJ and Li Y:
Induction of high temperature requirement A1, a serine protease, by
TGF-beta1 in articular chondrocytes of mouse models of OA. Histol
Histopathol. 29:609–618. 2014.PubMed/NCBI
|
|
35
|
Nagler A and Pines M: Topical treatment of
cutaneous chronic graft versus host disease (cGvHD) with
halofuginone: A novel inhibitor of collagen type I synthesis.
Transplantation. 68:1806–1809. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
De Jonge MJ, Dumez H, Verweij J, Yarkoni
S, Snyder D, Lacombe D, Marreaud S, Yamaguchi T, Punt CJ and van
Oosterom A: EORTC New Drug Development Group (NDDG): Phase I and
pharmacokinetic study of halofuginone, an oral quinazolinone
derivative in patients with advanced solid tumours. Eur J Cancer.
42:1768–1774. 2006. View Article : Google Scholar : PubMed/NCBI
|