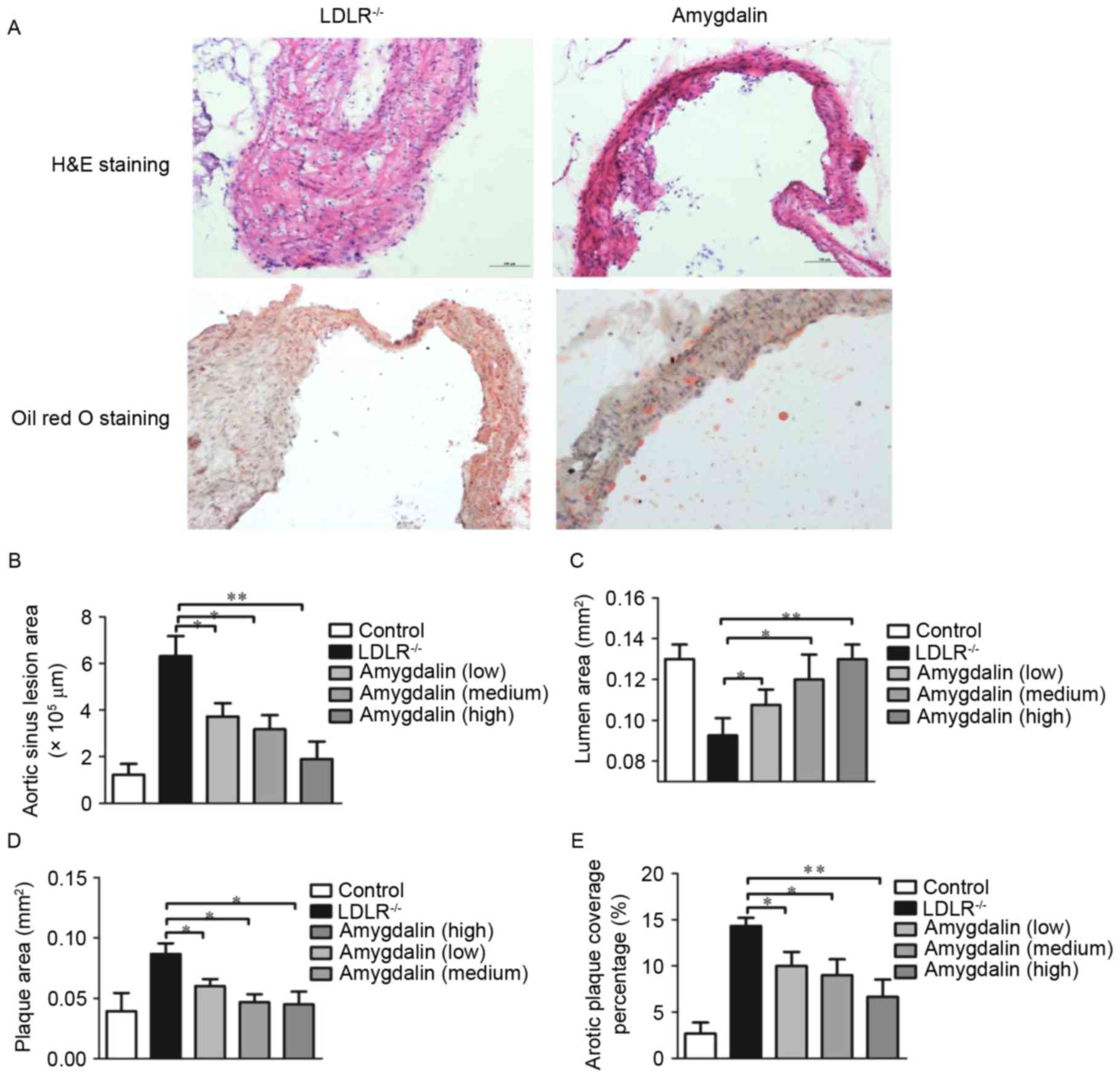
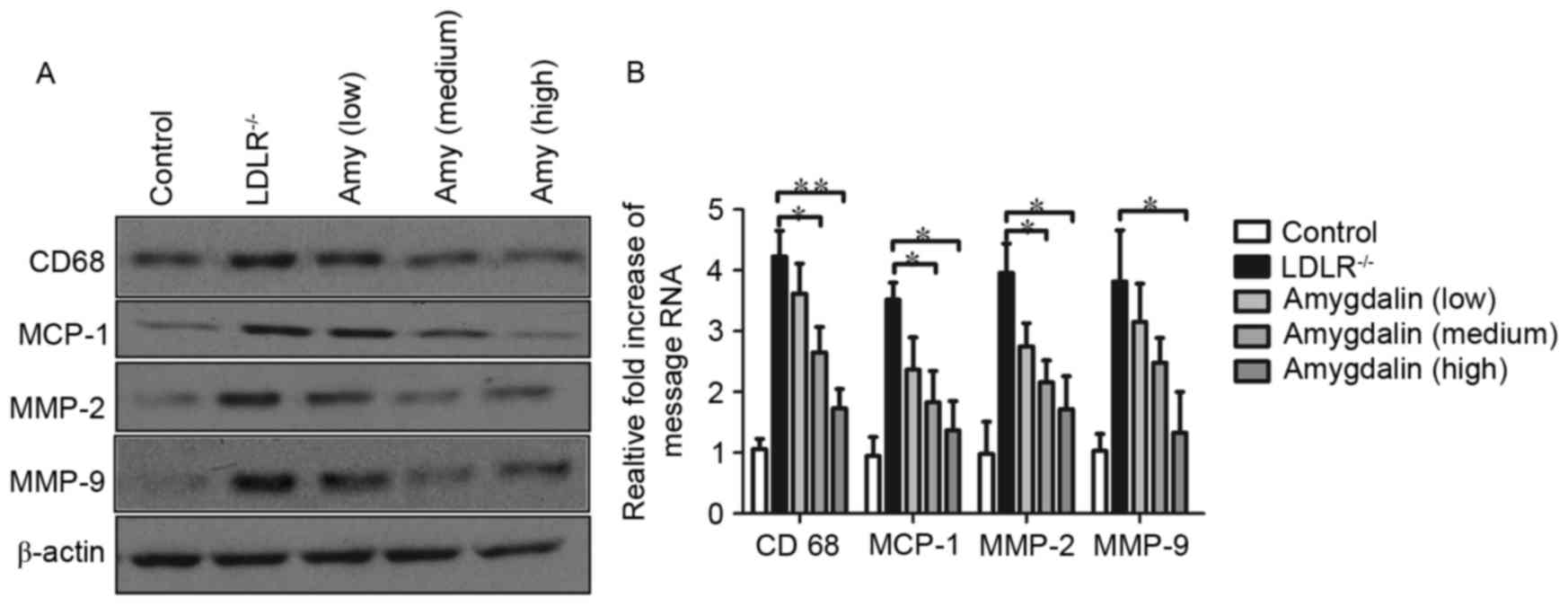
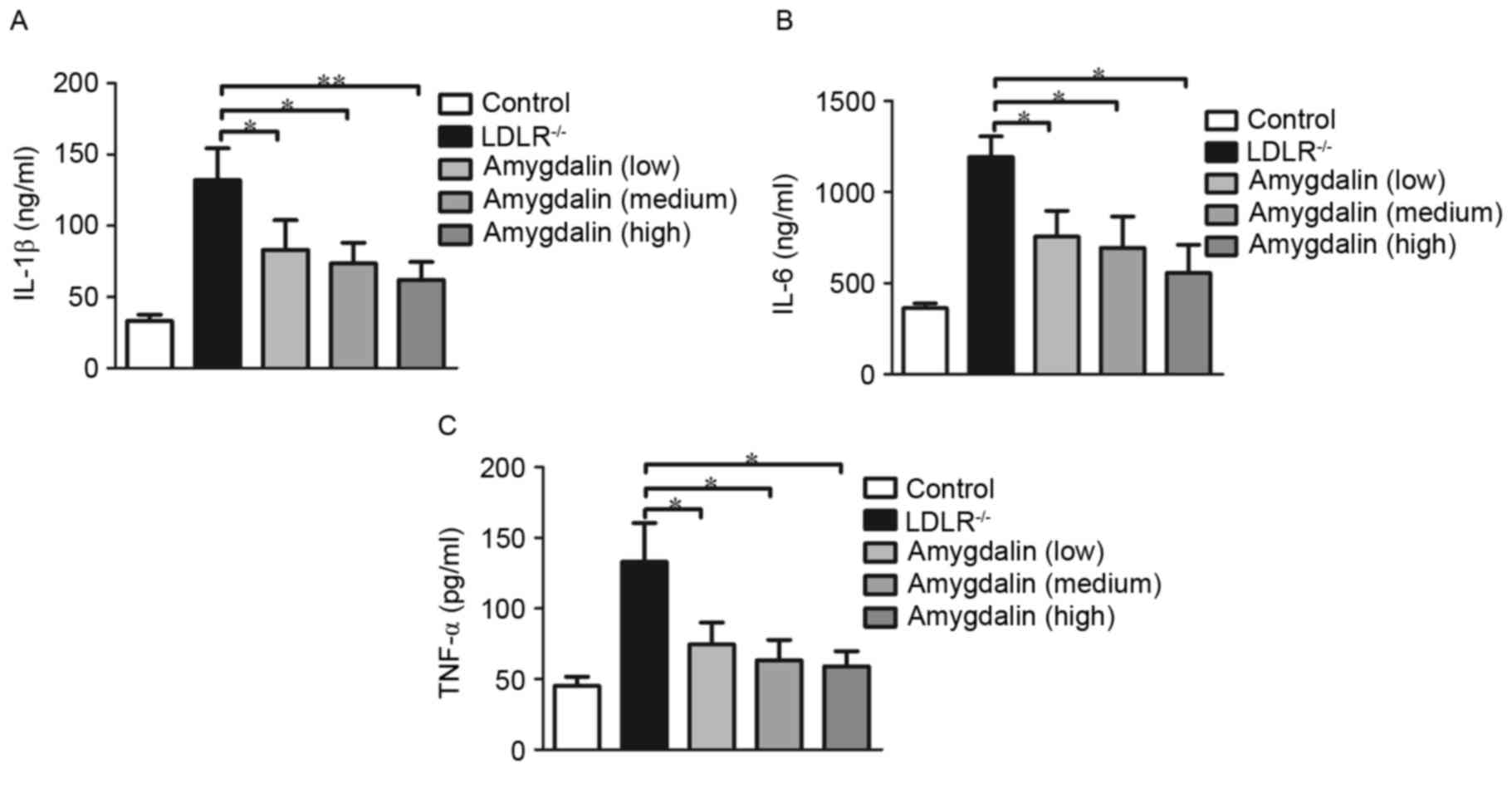
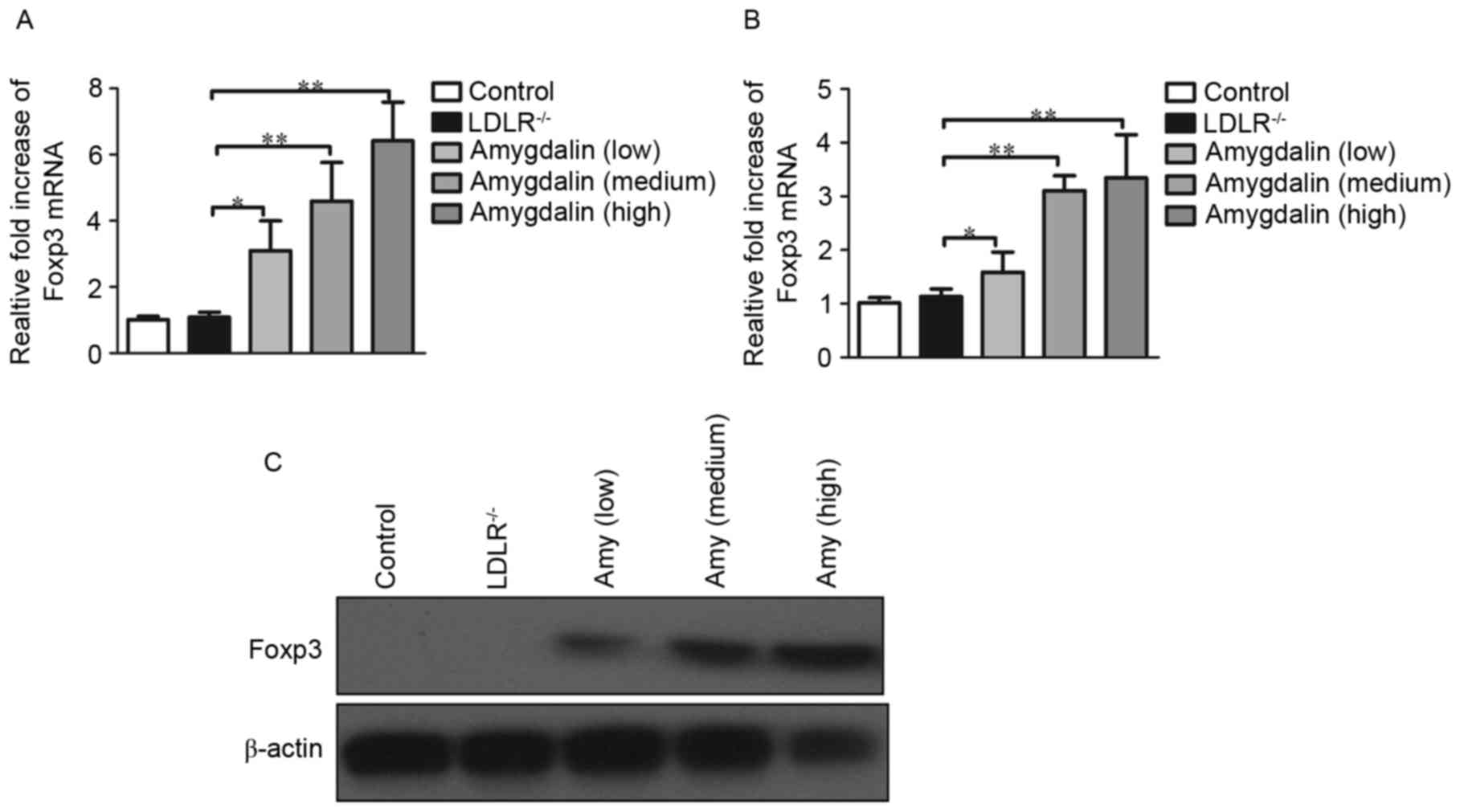
|
1
|
Glass CK and Witztum JL: Atherosclerosis.
The road ahead. Cell. 104:503–516. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hansson GK: Inflammation, atherosclerosis,
and coronary artery disease. N Engl J Med. 352:1685–1695. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ross R and Harker L: Hyperlipidemia and
atherosclerosis. Science. 193:1094–1100. 1976. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tell GS, Crouse JR and Furberg CD:
Relation between blood lipids, lipoproteins, and cerebrovascular
atherosclerosis. A review. Stroke. 19:423–430. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Napoli C, D'Armiento FP, Mancini FP,
Postiglione A, Witztum JL, Palumbo G and Palinski W: Fatty streak
formation occurs in human fetal aortas and is greatly enhanced by
maternal hypercholesterolemia. Intimal accumulation of low density
lipoprotein and its oxidation precede monocyte recruitment into
early atherosclerotic lesions. J Clin Invest. 100:2680–2690. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ross R: Atherosclerosis-an inflammatory
disease. N Engl J Med. 340:115–126. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tedgui A and Mallat Z: Cytokines in
atherosclerosis: Pathogenic and regulatory pathways. Physiol Rev.
86:515–581. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Binder CJ, Chang MK, Shaw PX, Miller YI,
Hartvigsen K, Dewan A and Witztum JL: Innate and acquired immunity
in atherogenesis. Nat Med. 8:1218–1226. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Whitman SC, Ravisankar P, Elam H and
Daugherty A: Exogenous interferon-gamma enhances atherosclerosis in
apolipoprotein E-/- mice. Am J Pathol. 157:1819–1824. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Frostegård J, Ulfgren AK, Nyberg P, Hedin
U, Swedenborg J, Andersson U and Hansson GK: Cytokine expression in
advanced human atherosclerotic plaques: Dominance of
pro-inflammatory (Th1) and macrophage-stimulating cytokines.
Atherosclerosis. 145:33–43. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mallat Z, Gojova A, Brun V, Esposito B,
Fournier N, Cottrez F, Tedgui A and Groux H: Induction of a
regulatory T cell type 1 response reduces the development of
atherosclerosis in apolipoprotein E-knockout mice. Circulation.
108:1232–1237. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jiagang D, Li C, Wang H, Hao E, Du Z, Bao
C, Lv J and Wang Y: Amygdalin mediates relieved atherosclerosis in
apolipoprotein E deficient mice through the induction of regulatory
T cells. Biochem Biophys Res Commun. 411:523–529. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sluijter JP, Pulskens WP, Schoneveld AH,
Velema E, Strijder CF, Moll F, de Vries JP, Verheijen J,
Hanemaaijer R, de Kleijn DP and Pasterkamp G: Matrix
metalloproteinase 2 is associated with stable and matrix
metalloproteinases 8 and 9 with vulnerable carotid atherosclerotic
lesions: A study in human endarterectomy specimen pointing to a
role for different extracellular matrix metalloproteinase inducer
glycosylation forms. Stroke. 37:235–239. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
George J, Harats D, Gilburd B, Afek A,
Shaish A, Kopolovic J and Shoenfeld Y: Adoptive transfer of
beta(2)-glycoprotein I-reactive lymphocytes enhances early
atherosclerosis in LDL receptor-deficient mice. Circulation.
102:1822–1827. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
George J, Afek A, Gilburd B, Shoenfeld Y
and Harats D: Cellular and humoral immune responses to heat shock
protein 65 are both involved in promoting fatty-streak formation in
LDL-receptor deficient mice. J Am Coll Cardiol. 38:900–905. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cobbold SP, Castejon R, Adams E, Zelenika
D, Graca L, Humm S and Waldmann H: Induction of Foxp3+
regulatory T cells in the periphery of T cell receptor transgenic
mice tolerized to transplants. J Immunol. 172:6003–6010. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yang L, Chu Y, Wang Y, Zhao X, Xu W, Zhang
P, Liu X, Dong S, He W and Gao C: siRNA-mediated silencing of Wnt5a
regulates inflammatory responses in atherosclerosis through the
MAPK/NF-kB pathways. Int J Mol Med. 34:1147–1152. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Huang G, Lv J, Li T, Huai G, Li X, Xiang
S, Wang L, Qin Z, Pang J, Zou B and Wang Y: Notoginsenoside R1
ameliorates podocyte injury in rats with diabetic nephropathy by
activating the PI3K/Akt signaling pathway. Int J Mol Med.
38:1179–1189. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Brattelid T, Winer LH, Levy FO, Liestøl K,
Sejersted OM and Andersson KB: Reference gene alternatives to Gapdh
in rodent and human heart failure gene expression studies. BMC Mol
Biol. 11:222010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tsai BY, Suen JL and Chiang BL:
Lentiviral-mediated Foxp3 RNAi suppresses tumor growth of
regulatory T cell-like leukemia in a murine tumor model. Gene Ther.
17:972–979. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ziegler SF: FOXP3: Of mice and men. Annu
Rev Immunol. 24:209–226. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
van Puijvelde GH, Hauer AD, de Vos P, van
den Heuvel R, van Herwijnen MJ, Van der Zee R, van Eden W, van
Berkel TJ and Kuiper J: Induction of oral tolerance to oxidized
low-density lipoprotein ameliorates atherosclerosis. Circulation.
114:1968–1976. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Skoog T, Dichtl W, Boquist S,
Skoglund-Andersson C, Karpe F, Tang R, Bond MG, de Faire U, Nilsson
J, Eriksson P and Hamsten A: Plasma tumour necrosis factor-alpha
and early carotid atherosclerosis in healthy middle-aged men. Eur
Heart J. 23:376–383. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bruunsgaard H, Skinhøj P, Pedersen AN,
Schroll M and Pedersen BK: Ageing, tumour necrosis factor-alpha
(TNF-alpha) and atherosclerosis. Clin Exp Immunol. 121:255–260.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ridker PM, Rifai N, Stampfer MJ and
Hennekens CH: Plasma concentration of interleukin-6 and the risk of
future myocardial infarction among apparently healthy men.
Circulation. 101:1767–1772. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ridker PM, Rifai N, Pfeffer M, Sacks F,
Lepage S and Braunwald E: Elevation of tumor necrosis factor-alpha
and increased risk of recurrent coronary events after myocardial
infarction. Circulation. 101:2149–2153. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Huber SA, Sakkinen P, Conze D, Hardin N
and Tracy R: Interleukin-6 exacerbates early atherosclerosis in
mice. Arterioscler Thromb Vasc Biol. 19:2364–2367. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kirii H, Niwa T, Yamada Y, Wada H, Saito
K, Iwakura Y, Asano M, Moriwaki H and Seishima M: Lack of
interleukin-1beta decreases the severity of atherosclerosis in
ApoE-deficient mice. Arterioscler Thromb Vasc Biol. 23:656–660.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Libby P, Ridker PM and Maseri A:
Inflammation and atherosclerosis. Circulation. 105:1135–1143. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhou X, Nicoletti A, Elhage R and Hansson
GK: Transfer of CD4(+) T cells aggravates atherosclerosis in
immunodeficient apolipoprotein E knockout mice. Circulation.
102:2919–2922. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ochs HD, Ziegler SF and Torgerson TR:
FOXP3 acts as a rheostat of the immune response. Immunol Rev.
203:156–164. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mor A, Luboshits G, Planer D, Keren G and
George J: Altered status of CD4(+)CD25(+) regulatory T cells in
patients with acute coronary syndromes. Eur Heart J. 27:2530–2537.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Brunkow ME, Jeffery EW, Hjerrild KA,
Paeper B, Clark LB, Yasayko SA, Wilkinson JE, Galas D, Ziegler SF
and Ramsdell F: Disruption of a new forkhead/winged-helix protein,
scurfin, results in the fatal lymphoproliferative disorder of the
scurfy mouse. Nat Genet. 27:68–73. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Gu L, Okada Y, Clinton SK, Gerard C,
Sukhova GK, Libby P and Rollins BJ: Absence of monocyte
chemoattractant protein-1 reduces atherosclerosis in low density
lipoprotein receptor-deficient mice. Mol Cell. 2:275–281. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Harrington JR: The role of MCP-1 in
atherosclerosis. Stem Cells. 18:65–66. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Pasterkamp G, Schoneveld AH, Hijnen DJ, de
Kleijn DP, Teepen H, van der Wal AC and Borst C: Atherosclerotic
arterial remodeling and the localization of macrophages and matrix
metalloproteases 1, 2 and 9 in the human coronary artery.
Atherosclerosis. 150:245–253. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Gough PJ, Gomez IG, Wille PT and Raines
EW: Macrophage expression of active MMP-9 induces acute plaque
disruption in apoE-deficient mice. J Clin Invest. 116:59–69. 2006.
View Article : Google Scholar : PubMed/NCBI
|