Introduction
Sepsis is a systemic inflammatory response syndrome
associated with a high rate of mortality (1). The incidence of sepsis has been
increasing over the last two decades. This increase has been more
marked in developed countries, where 6–30% of patients in Intensive
Care Units suffer from sepsis (2).
Sepsis remains the leading cause of mortality in patients in
Intensive Care Units (3) and the
associated burden of care incurs financial burden. However, the
pathogenesis of sepsis remains to be fully delineated and effective
therapies are lacking. A state of immunosuppression induced by
sepsis has been demonstrated in clinical and experimental sepsis
(3), with the majority of patients
who have succumbed to sepsis-associated mortality showing evidence
of immunosuppression and unresolved septic foci (4).
Polymorphonuclear neutrophils (PMNs) are an
essential component of the innate immune system, involved in the
clearance of extracellular pathogens (5). As the most abundant subset of
leukocytes, the involvement of PMN in sepsis is significant
(3,6,7). The
migration of PMNs is regulated by chemoattractants and chemokine
gradients (3,7,8).
CXC-chemokine receptor (CXCR)1 and CXCR2 are the major chemokine
receptors on PMNs, with interleukin (IL)-8 acting as a ligand of
these receptors (9). Severe sepsis
is associated with the failure of PMNs to migrate (10). In a previous clinical study,
patients with suppression of PMN receptors were predisposed to
inflammatory response syndrome (11). Previous in vitro
investigations have demonstrated that CXCR2 is downregulated upon
stimulation with tumor necrosis factor-α (TNF-α) (12). The activation of Toll-like receptor
4 (TLR4) also suppresses the expression of CXCR2 (13). In an experimental mouse model of
sepsis, the failure of PMNs to migrate was shown to result in a
high rate of mortality (1).
Tancevski et al and Van Zee et al reported that
promoting the recruitment of PMNs ameliorates sepsis and attenuates
sepsis-related injury and infection, respectively (14,15).
Therefore, in order to improve treatment of inflammatory disorders,
including sepsis, the promotion of PMN chemotaxis is an attractive
target (6).
As an ancient Chinese herb, Folium isatidis
is considered to have detoxification properties in traditional
Chinese medicine. In addition to being used to cure febrile
diseases, including dipsosis, fever, jaundice and hematemesis,
F. isatidis is also deployed against diseases, including
mumps, viral hepatitis, influenza and epidemic encephalitis B
(16–18). It was reported to be effective
during the 2003 severe acute respiratory syndrome flu outbreak
(19). These antimicrobial and
anti-endotoxic properties indicate its potential to be developed
into a natural antibiotic (20,21).
In our previous study (22), the
main chemical components of F. isatidis were identified
using high-performance liquid chromatography. It was also
demonstrated that F. isatidis increased the survival of
septic mice, and ameliorated lung injury by inhibiting the
production of inflammatory cytokines TNF-α and IL-6 through the
myeloid differentiation primary response gene 88 and nuclear
factor-κB pathways (22). Although
the beneficial immunomodulatory effects of F. isatidis have
been the focus of previous investigations, whether this herb has an
effect on chemokine receptors remains to be elucidated. The present
study aimed to determine whether F. isatidis affects the
migration of PMNs.
Materials and methods
Isolation of neutrophils
The present study was approved by the Ethics
Committee of The Second Hospital of Wenzhou Medical University
(Wenzhou, China), and informed consent was obtained. Peripheral
blood was collected from four 20-30-year-old female healthy
volunteers between August and December 2015, who had been referred
to The Second Affiliated Hospital and Yuying Children's Hospital of
Wenzhou Medical University and who provided informed, written
consent. The blood was transferred into heparin lithium-containing
tubes (BD Bioscience, San Jose, CA, USA). The samples were
processed within 1 h of collection. Whole blood was incubated with
3% dextran T-500 in the dark at room temperature for 20 min for
sedimentation of red blood cells (RBCs). Following sedimentation,
the white blood cell-enriched upper phase was layered over a
Ficoll-Paque solution (GE Healthcare Life Science, Uppsala,
Sweden). Centrifugation was performed at 427 × g at room
temperature for 20 min without breaks, resulting in four distinct
layers. The first three layers were discarded, and the remaining
layer containing granulocytes and remnant RBCs was diluted in ACK
lysis buffer (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA,
USA). The cells were then washed with PBS, and the freshly isolated
neutrophils were resuspended in RPMI 1640 (Gibco; Thermo Fisher
Scientific, Inc.). The purity of the isolated neutrophils was
>90% based on Wright's staining and differential counts.
Flow cytometry
The isolated neutrophils were diluted to 1×106
cells/ml with RPMI 1640 (Gibco; Thermo Fisher Scientific, Inc.).
The cells were then incubated with either vehicle (0.01% DMSO) or
increasing concentrations of n-butanol extract from Folium isatidis
(100, 250 and 500 µg/ml) at 37°C and 5% CO2 for 2 h. The specific
method to obtain n-butanol extract from F. isatidis was described
in previous study (22). The cells
were then stimulated with 0.5 µg/ml lipopolysaccharide (LPS; Sigma;
Merck Millipore, Darmstadt, Germany) for 4 h at 37°C. The cells
were washed with ice-cold PBS and then resuspended in 50 µl FACS
buffer. The cells were then incubated with a 1:50 dilution of the
following antibodies (the concentrations suggested by the
manufacturer) for 30 min at 4°C in the dark: FITC-conjugated
anti-human CD181 (CXCR1; cat. no 11-1819-42), PE-conjugated
anti-human CD182 (CXCR2; cat. no 12-1829-42) and APC-conjugated
anti-human CD11b (cat no. 17-0118-42) or FITC-conjugated anti-human
L-selectin (CD62L; cat. no 11-0629-42), PE-conjugated anti-human
TLR4 (cat no. 12-9917-41) and APC conjugated anti-human TLR2 (cat
no. 17-9922-42) (all from eBioscience, San Diego, CA, USA).
Antibodies of the same isotype were used as negative controls. The
cells were analyzed using a FACS calibur system (BD Biosciences).
The mean fluorescence intensity (MFI) for 10,000 cells in each
sample was determined using CellQuest software, version 5.2 (BD
Biosciences).
Chemotaxis assays
The isolated neutrophils were pretreated as
described above for 2 h and then stimulated with LPS (0.5 µg/ml)
for 4 h. The cells were then washed twice with PBS, and resuspended
in RPMI 1640 at 1×106 cells/ml. The chemotaxis assays were
performed using Transwell inserts (24 wells, 5 µm pore size;
Corning, Inc., Corning, NY, USA), where 10,000 cells in 100 µl were
loaded in the upper inserts and 600 µl of IL-8 (100 ng/ml;
eBioscience; Thermo Fisher Scientific, Inc.) containing RPMI 1640
was added to the lower wells. Following co-incubation for 2 h at
37°C and a humidified 5% CO2 atmosphere, the inserts were removed,
and the migrated neutrophils in the lower wells were collected and
quantified according to myeloperoxidase (MPO) activity using a
myeloperoxidase assay kit (Nanjing Jiancheng Bioengineering
Institute, Nanjing, China).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) analysis
Total RNA was isolated from cells using TRIzol
reagent (Invitrogen; Thermo Fisher Scientific, Inc.). RT-qPCR
analysis was performed using an M-MLV Platinum SYBR Green qPCR
SuperMix-UDG kit (Invitrogen; Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol. A total of 1 µg RNA was
used for the determination. qPCR was performed in a 10 µl reaction
volume containing 2 µl cDNA target, 2.25 µl SYBR Green (Bio-Rad
Laboratories, Inc., Hercules, CA, USA), 0.25 µl sense primers, 0.25
µl anti-sense primers and 5.25 µl ddH2O. An Eppendorf Mastercycler
realplex detection system (Eppendorf, Hamburg, Germany) was used
for RT-qPCR analysis: The reaction consisted of the following
conditions: 95°C for 3 min, followed by 95°C for 15 sec, 60°C for
30 sec, and 70°C for 30 sec (40 cycles). The primers of the genes
were synthesized by Invitrogen (Thermo Fisher Scientific, Inc.) and
the sequences were as follows: Human CXCR1, sense
5′-GCAGCTCCTACTGTTGGACA-3′ and antisense
5′-GGGCATAGGCGATGATCACA-3′; human CXCR2, sense
5′-CCCATCTTCATTCTTCGGAC-3′, and antisense
5′-GACAATGTTGTAGGGAAGCCAG-3′; human TLR2, sense
5′-ATGCCTACTGGGTGGAGAAC-3′ and antisense
5′-TGCACCACTCAGTCTTCACA-3′; human TLR4, sense
5′-CAGCTCTTGGTGGAAGTTGA-3′ and antisense
5′-GCAAGAAGCATCAGGTGAAA-3′; human CD62L, sense
5′-CTCCTTGCCAGCCAAATGATAA-3′ and antisense
5′-CCTCTTCATTCCAGTGGCAGTC-3′; human CD11b, sense
5′-GGACCTCGGGCTCAAGTAAT-3′ and antisense
5′-GCCTGTAATGCCAGCACTTT-3′; human IL-8, sense
5′-CTGGCCGTGGCTCTCTTG-3′ and antisense 5′-CCTTGGCAAAACTGCACCTT-3′;
human β-actin, sense 5′-CCTGGCACCCAGCACAAT' and antisense
5′-GCCGATCCACACGGAGTACT-3′. The result of real-time PCR was
expressed as the threshold cycle (CT). The CT represents the PCR
cycle at which the reported fluorescence rises above a set baseline
threshold when the DNA amplicon is replicating exponentially
(12). Results were normalized to
β-actin as plotted as relative expression to the average of CON or
DMSO, which were set as 100.
Quantification of IL-8
The neutrophils were pretreated for 2 h with the
extract as described above, and then stimulated with LPS (0.5
µg/ml) for 1 or 4 h. The supernatant and cells were collected, and
the levels of IL-8 in the supernatant were determined using
enzyme-linked immunosorbent assay (ELISA) kits (cat no. 88-8086-88;
eBioscience; Thermo Fisher Scientific, Inc.) according to the
manufacturer's protocol. The total quantity of IL-8 in the
supernatant was normalized to that of total protein in the viable
cell pellets.
Statistical analysis
All the experiments were performed in triplicate.
Data are expressed as the mean ± standard error of the mean. All
the data were statistically analyzed using Student's t test.
P<0.05 was considered to indicate a statistically significant
difference. GraphPad Prism 5.0 software (GraphPad Software, Inc.,
La Jolla, CA, USA) was used for statistical analysis.
Results
LPS downregulates neutrophil
expression of CXCR1, CXCR2 and CD62L
TLR2 and TLR4/myeloid differentiation factor 2 are
vital in the recognition of LPS in the host, and the triggering of
these receptors can lead to neutrophil recruitment and migration
via CXCR1 and CXCR2. CD11b and CD62L, which are expressed on the
surface of neutrophils, are critical for the occurrence of sepsis.
LPS, also known as endotoxin, is a component of the outer membrane
of Gram-negative bacteria, and is known to induce septic shock when
present in high quantities. Therefore, the present study measured
the expression of neutrophil chemokine receptors under LPS
stimulation. The isolated neutrophils were treated with either
vehicle or increasing concentrations (10, 100 and 1,000 ng/ml) of
LPS for 4 h, and the expression levels of CXCR1, CXCR2, TLR2, TLR4,
CD11b and CD62L were measured using flow cytometry. The results
showed that LPS treatment resulted in decreased expression levels
of CXCR1, CXCR2 and CD62L in a dose-dependent manner (Fig. 1A-C), which was in accordance with
the results of a previous study (23). However, no significant changes were
observed in the expression levels of TLR2, TLR4 or CD11b in the
LPS-treated neutrophils, compared with the vehicle-treated
neutrophils (Fig. 1D-F).
Therefore, the expression levels of TLR2, TLR4 and CD11b were not
examined in the remainder of the experiments.
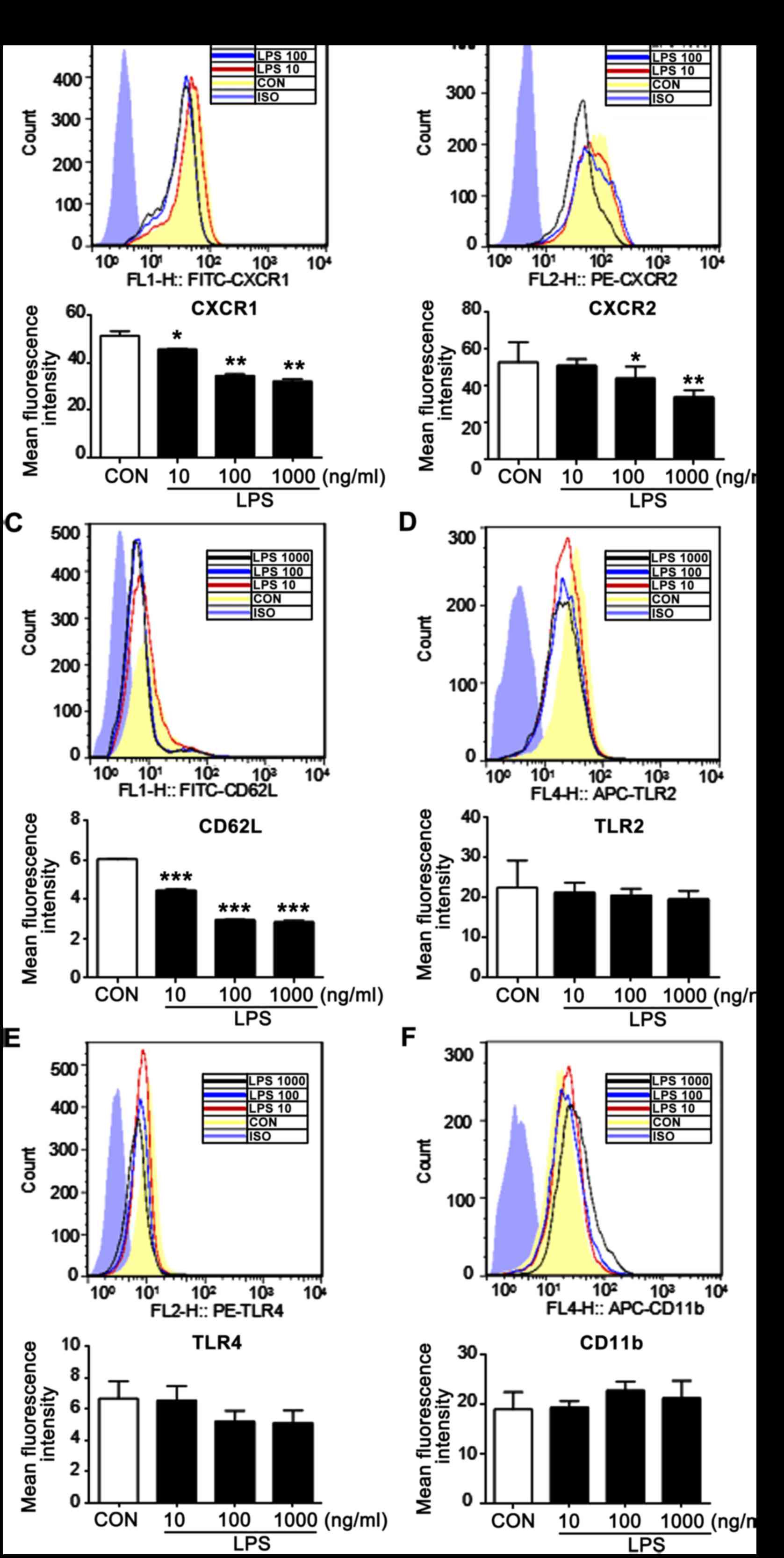 | Figure 1.LPS induces a decrease in the
neutrophilic expression of CXCR1, CXCR2 and CD62L. Isolated
neutrophils were incubated with LPS for 4 h. Levels of (A) CXCR1,
(B) CXCR2, (C) CD62L, (D) TLR2, (E) TLR4 and (F) CD11b were
measured using flow cytometry. The corresponding mean fluorescence
intensity was calculated. Each bar represents the mean ± standard
error of the mean of three independent experiments. Statistical
significance relative to the CON group was determined. *P<0.05,
**P<0.01 and ***P<0.001. LPS, lipopolysaccharide; CXCR1,
CXC-chemokine receptor 1; CXCR2, CXC-chemokine receptor 2; CD62L,
L-selectin; TLR2, toll-like receptor 2; TLR4, toll-like receptor 4;
CON, control. |
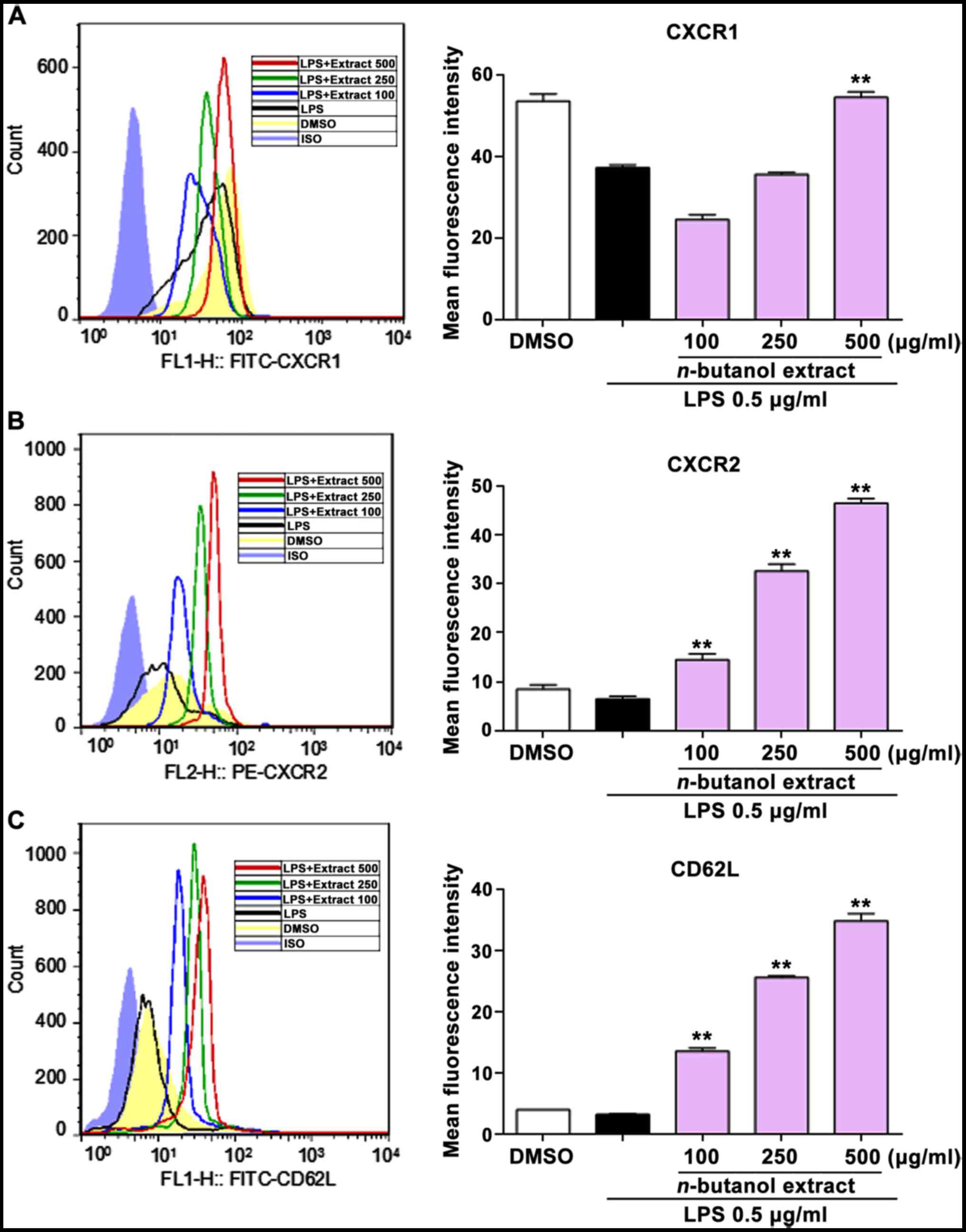
n-butanol extract from Folium isatidis
prevents the LPS-induced downregulation of CXCR1, CXCR2 and
CD62L
CXCR1, CXCR2 and CD62L may be important in sepsis,
however, whether n-butanol extract affects these chemokine
receptors remains to be elucidated. Therefore, the present study
assessed the effect of the extract on the expression of CXCR1,
CXCR2 and CD62L. The isolated neutrophils were pre-incubated with
either vehicle or increasing concentrations of extract (100, 250
and 500 µg/ml) for 2 h, and were subsequently treated with LPS (0.5
µg/ml) for 4 h. Subsequent analysis of the cells using flow
cytometry indicated that the extract inhibited the LPS-induced
downregulation of CXCR1, CXCR2 and CD62L (Fig. 2A-C) in a dose-dependent manner.
Effect of n-butanol extract from
Folium isatidis on the gene expression levels of CXCR1, CXCR2 and
CD62L
As the n-butanol extract obtained from F.
isatidis had a significant effect on chemokine receptor protein
levels, the present study investigated the gene expression levels
of these receptors. The isolated neutrophils were pretreated with
extract and then treated with LPS (0.5 µg/ml) for 1 or 4 h
(Fig. 3A-F). The extract increased
the gene expression levels of CXCR1, CXCR2 and CD62L in a
dose-dependent manner.
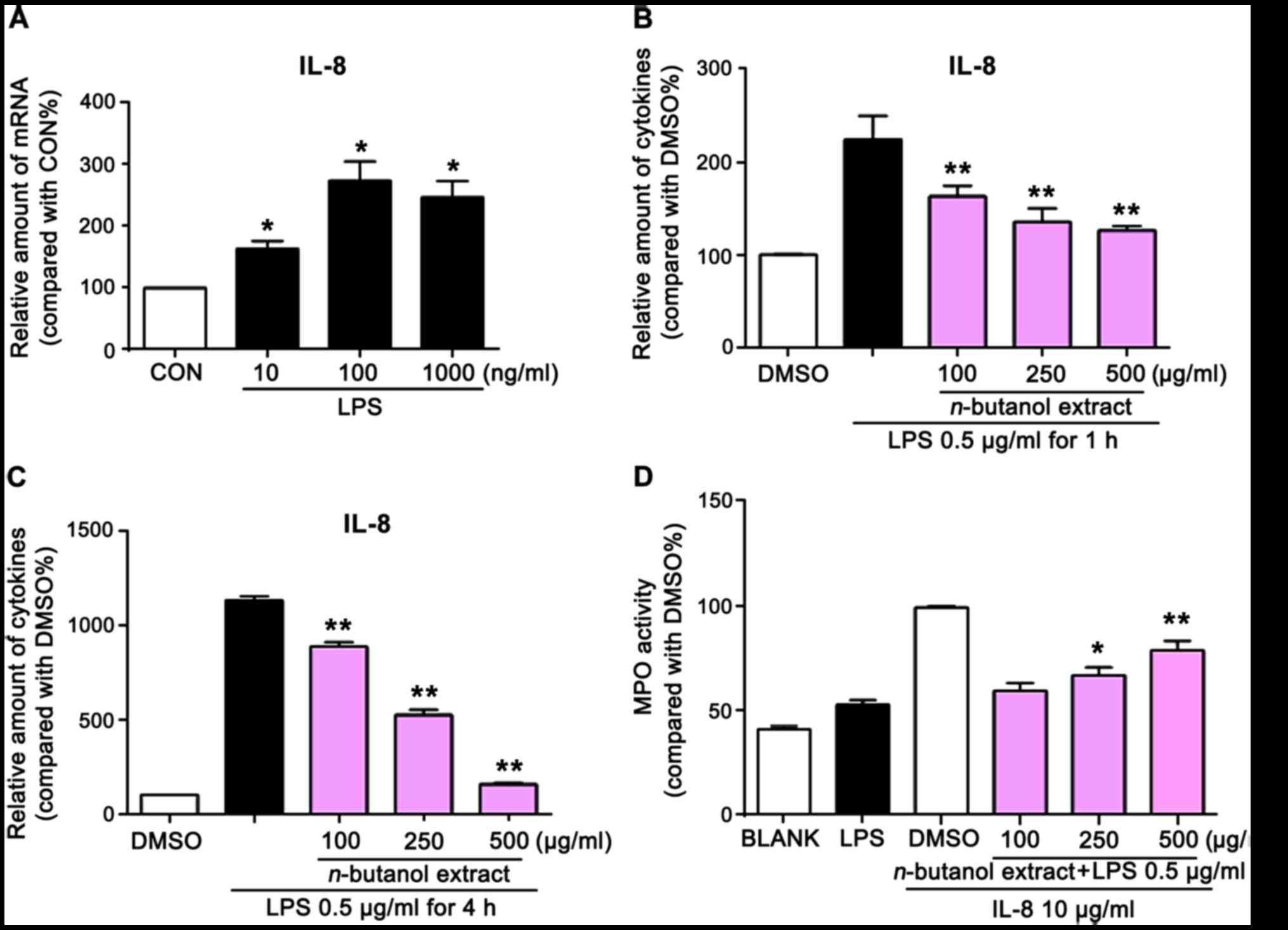
n-butanol extract from Folium isatidis
decreases the expression of IL-8 and increases the activity of
MPO
IL-8, as the ligand of CXCR1 and CXCR2, is important
in the regulation of neutrophil migration. Following exposure of
the neutrophils to LPS for 4 h, the mRNA expression of IL-8 was
significantly increased (Fig. 4A).
Therefore, the present study examined whether n-butanol
extract affected the production of IL-8. The isolated neutrophils
were pretreated with extract and then incubated with LPS (0.5
µg/ml) for 1 h (Fig. 4B) or 4 h
(Fig. 4C). The expression levels
of IL-8 were quantified using ELISA. A decrease in IL-8 was
observed following exposure to LPS for 1 and 4 h when pretreated
with the extract. Specifically, following exposure to LPS for 4 h,
the cytokine expression of IL-8 was decreased >5-fold when the
cells were treated with the extract at a dose of 500 µg/ml,
compared with LPS stimulation (Fig.
4C). MPO levels are a measure of neutrophil migration.
Therefore, the present study examined the effect of the extract on
the activity of MPO. Neutrophils were pretreated with
n-butanol extract for 2 h followed by stimulation with LPS
for 4 h. The activity of MPO increased in a dose-dependent manner
following treatment with extract, compared with the control
(Fig. 4D), suggesting that
n-butanol extract promoted the function of neutrophils.
Discussion
Sepsis remains a threat to human health, primarily
due to a lack of effective therapies (3). With current treatment options, the
mortality rate of patients with sepsis remains high at ~30%, and
mortality rates increase directly with disease severity (24). A previous worldwide survey of
relevant pathogens in intensive care units found that the majority
of patients with sepsis had blood cultures positive for
Gram-negative bacteria (25). LPS,
a component of the outer leaflet of the outer membrane of
Gram-negative bacteria, is a key molecule in the induction of
sepsis. Therefore, the present study used LPS to simulate sepsis
induced by Gram-negative bacteria infection.
As previously reported, the present study observed
that LPS stimulation decreased the expression of CXCR1, CXCR2 and
CD62L on neutrophils. In addition to being effector cells involved
in the acute phase of the inflammatory response, neutrophils are
also important in the resolution of inflammation. The failure of
neutrophils to migrate can lead to an inability to control
infection due to weakened neutrophil phagocytic and bactericidal
abilities (26). CXCR1 and CXCR2
are the major chemokine receptors on neutrophils. Chemokine
receptors regulate the migration of neutrophils to the site of
infection to assist in controlling invading pathogens and
protecting the body. Duerschmied et al (27) demonstrated that survival following
LPS-induced endotoxic shock improved upon the promotion of
neutrophil recruitment during acute inflammation. n-butanol
extract was found to have significant antiseptic abilities in our
previous study. However, whether the extract affected neutrophil
migration remained unclear. In the present study, it was
demonstrated that n-butanol extract prevented the
downregulation of CXCR1, CXCR2 and CD62L.
In our previous study, it was demonstrated that
n-butanol extract from Folium isatidis significantly
inhibited the activation of TLR4 and its signaling pathways
(22). It has been reported that
the systemic activation of TLRs and high levels of TNF-α are
involved in the reduction of neutrophil recruitment through the
downregulation of CXCR2 in neutrophils (28). Therefore, it was hypothesized that
n-butanol extract from F. isatidis prevents the
downregulation of CXCR1 and CXCR2 through the activation of TLR4
and secretion of TNF-α.
The primary function of CD62L, a vascular adhesion
molecule, is directing neutrophil migration. In addition to CXCR1
and CXCR2, neutrophil migration requires the assistance of CD62L.
In the present study, the expression of CD62L decreased following
incubation with LPS for 4 h (Fig.
1C), whereas n-butanol extract promoted the expression
of CD62L in a dose-dependent manner (Fig. 2C). This suggested that the extract
promoted the migration of neutrophils by inhibiting the
sepsis-induced downregulation of CD62L.
IL-8 is a ligand of CXCR1 and CXCR2, and is rapidly
secreted upon cell stimulation. The mRNA expression of IL-8
significantly increased upon stimulation with LPS (Fig. 4A). However, n-butanol
extract decreased the expression of IL-8 in a dose-dependent manner
(Fig. 4B and C), and prevented the
decreases in the expression of CXCR1 and CXCR2. This suggested that
the extract increased the activity of neutrophils and upregulated
the expression of chemokine receptors, including CXCR1 and CXCR2,
rendering neutrophils more sensitive to the ligand IL-8. The
present study quantified MPO activity as a measure of neutrophil
migration. It was found the extract promoted the activity of MPO.
The extract also promoted the ability of neutrophils to migrate by
increasing the expression of CXCR1 and CXCR2 on the surface of
neutrophils.
To date, the treatment of sepsis consists primarily
of supportive measures and experimental therapeutic approaches
(29). Therefore, novel
pharmacological strategies are urgently required to promote the
treatment of sepsis (30). With
the ability to promote the expression of CXCR1, CXCR2 and CD62L,
n-butanol extract itself is a potential candidate for the
treatment of LPS-induced sepsis. However, due to the presence of
multiple bioactive components in the extract and limited approaches
for preparing extracts from F. isatidis, it is difficult to
identify the precise agent inhibiting LPS-induced chemokine
receptor downregulation. Although the main active chemical
components have been identified, there are more than eight major
compounds in n-butanol extract (22). Further investigation is required to
identify the active compound in F. isatidis.
In conclusion, the present study demonstrated that
n-butanol extract obtained from F. isatidis prevented
the LPS-induced downregulation of CXCR1, CXCR2 and CD62L on
neutrophils. The extract promoted neutrophil migration in response
to IL-8. These results indicated that n-butanol extract from
F. isatidis, which targets neutrophil chemotaxis during
LPS-induced sepsis, is a potential candidate for the treatment of
sepsis.
Acknowledgements
This study was supported by the Provincial Natural
Science Foundation of Zhejiang (grant no. Y2100530).
References
|
1
|
Lee SK, Kim SD, Kook M, Lee HY, Ghim J,
Choi Y, Zabel BA, Ryu SH and Bae YS: Phospholipase D2 drives
mortality in sepsis by inhibiting neutrophil extracellular trap
formation and down-regulating CXCR2. J Exp Med. 212:1381–1390.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Prucha M, Bellingan G and Zazula R: Sepsis
biomarkers. Clin Chim Acta. 440:97–103. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Drifte G, Dunn-Siegrist I, Tissières P and
Pugin J: Innate immune functions of immature neutrophils in
patients with sepsis and severe systemic inflammatory response
syndrome. Crit Care Med. 41:820–832. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Silva SC, Baggio-Zappia GL, Brunialti MK,
Assunçao MS, Azevedo LC, Machado FR and Salomao R: Evaluation of
Toll-like, chemokine, and integrin receptors on monocytes and
neutrophils from peripheral blood of septic patients and their
correlation with clinical outcomes. Braz J Med Biol Res.
47:384–393. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Seeley EJ, Matthay MA and Wolters PJ:
Inflection points in sepsis biology: From local defense to systemic
organ injury. Am J Physiol Lung Cell Mol Physiol. 303:L355–L363.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Boppana NB, Devarajan A, Gopal K, Barathan
M, Bakar SA, Shankar EM, Ebrahim AS and Farooq SM: Blockade of
CXCR2 signalling: A potential therapeutic target for preventing
neutrophil-mediated inflammatory diseases. Exp Biol Med (Maywood).
239:509–518. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jaillon S, Galdiero MR, Del Prete D,
Cassatella MA, Garlanda C and Mantovani A: Neutrophils in innate
and adaptive immunity. Semin Immunopathol. 35:377–394. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Park DW, Jiang S, Tadie JM, Stigler WS,
Gao Y, Deshane J, Abraham E and Zmijewski JW: Activation of AMPK
enhances neutrophil chemotaxis and bacterial killing. Mol Med.
19:387–398. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Takahashi M, Ishiko T, Kamohara H, Hidaka
H, Ikeda O, Ogawa M and Baba H: Curcumin
(1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione)
blocks the chemotaxis of neutrophils by inhibiting signal
transduction through IL-8 receptors. Mediators Inflamm.
2007:107672007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ishii M, Asano K, Namkoong H, Tasaka S,
Mizoguchi K, Asami T, Kamata H, Kimizuka Y, Fujiwara H, Funatsu Y,
et al: CRTH2 is a critical regulator of neutrophil migration and
resistance to polymicrobial sepsis. J Immunol. 188:5655–5664. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tarlowe MH, Duffy A, Kannan KB, Itagaki K,
Lavery RF, Livingston DH, Bankey P and Hauser CJ: Prospective study
of neutrophil chemokine responses in trauma patients at risk for
pneumonia. Am J Respir Crit Care Med. 171:753–759. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tikhonov I, Doroshenko T, Chaly Y,
Smolnikova V, Pauza CD and Voitenok N: Down-regulation of CXCR1 and
CXCR2 expression on human neutrophils upon activation of whole
blood by S. aureus is mediated by TNF-alpha. Clin Exp Immunol.
125:414–422. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Deng M, Ma T, Yan Z, Zettel KR, Scott MJ,
Liao H, Frank A, Morelli AE, Sodhi CP, Hackam DJ and Billiar TR:
Toll-like receptor 4 signaling on dendritic cells suppresses
polymorphonuclear leukocyte CXCR2 expression and trafficking via
interleukin 10 during intra-abdominal sepsis. J Infect Dis.
213:1280–1288. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tancevski I, Nairz M, Duwensee K, Auer K,
Schroll A, Heim C, Feistritzer C, Hoefer J, Gerner RR, Moschen AR,
et al: Fibrates ameliorate the course of bacterial sepsis by
promoting neutrophil recruitment via CXCR2. EMBO Mol Med.
6:810–820. 2014.PubMed/NCBI
|
|
15
|
Van Zee KJ, DeForge LE, Fischer E, Marano
MA, Kenney JS, Remick DG, Lowry SF and Moldawer LL: IL-8 in septic
shock, endotoxemia, and after IL-1 administration. J Immunol.
146:3478–3482. 1991.PubMed/NCBI
|
|
16
|
Deng YP, Liu YY, Liu Z, Li J, Zhao LM,
Xiao H, Ding XH and Yang ZQ: Antiviral activity of Folium isatidis
derived extracts in vitro and in vivo. Am J Chin Med. 41:957–969.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Liu Z, Yang ZQ and Xiao H: Antiviral
activity of the effective monomers from Folium Isatidis against
influenza virus in vivo. Virol Sin. 25:445–451. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fang JG, Hu Y, Tang J, Wang WQ and Yang
ZQ: Antiviral effect of Folium Isatidis on herpes simplex virus
type I. Zhongguo Zhong Yao Za Zhi. 30:1343–1346. 2005.(In Chinese).
PubMed/NCBI
|
|
19
|
Lau TF, Leung PC, Wong EL, Fong C, Cheng
KF, Zhang SC, Lam CW, Wong V, Choy KM and Ko WM: Using herbal
medicine as a means of prevention experience during the SARS
crisis. Am J Chin Med. 33:345–356. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liu CS, Cham TM, Yang CH, Chang HW, Chen
CH and Chuang LY: Antibacterial properties of Chinese herbal
medicines against nosocomial antibiotic resistant strains of
Pseudomonas aeruginosa in Taiwan. Am J Chin Med. 35:1047–1060.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Liau BC, Jong TT, Lee MR and Chen SS:
LC-APCI-MS method for detection and analysis of tryptanthrin,
indigo and indirubin in daqingye and banlangen. J Pharm Biomed
Anal. 43:346–351. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jiang L, Lu Y, Jin J, Dong L, Xu F, Chen
S, Wang Z, Liang G and Shan X: n-Butanol extract from Folium
isatidis inhibits lipopolysaccharide-induced inflammatory cytokine
production in macrophages and protects mice against
lipopolysaccharide-induced endotoxic shock. Drug Des Devel Ther.
9:5601–5609. 2015.PubMed/NCBI
|
|
23
|
Khandaker MH, Xu L, Rahimpour R, Mitchell
G, DeVries ME, Pickering JG, Singhal SK, Feldman RD and Kelvin DJ:
CXCR1 and CXCR2 are rapidly down-modulated by bacterial endotoxin
through a unique agonist-independent, tyrosine kinase-dependent
mechanism. J Immunol. 161:1930–1938. 1998.PubMed/NCBI
|
|
24
|
Iskander KN, Craciun FL, Stepien DM, Duffy
ER, Kim J, Moitra R, Vaickus LJ, Osuchowski MF and Remick DG: Cecal
ligation and puncture-induced murine sepsis does not cause lung
injury. Crit Care Med. 41:159–170. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Vincent JL, Rello J, Marshall J, Silva E,
Anzueto A, Martin CD, Moreno R, Lipman J, Gomersall C, Sakr Y, et
al: International study of the prevalence and outcomes of infection
in intensive care units. JAMA. 302:2323–2329. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kipnis E: Neutrophils in sepsis: Battle of
the bands. Crit Care Med. 41:925–926. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Duerschmied D, Suidan GL, Demers M, Herr
N, Carbo C, Brill A, Cifuni SM, Mauler M, Cicko S, Bader M, et al:
Platelet serotonin promotes the recruitment of neutrophils to sites
of acute inflammation in mice. Blood. 121:1008–1015. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sônego F, Alves-Filho JC and Cunha FQ:
Targeting neutrophils in sepsis. Expert Rev Clin Immunol.
10:1019–1028. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Rivers E, Nguyen B, Havstad S, Ressler J,
Muzzin A, Knoblich B, Peterson E and Tomlanovich M: Early
Goal-Directed Therapy Collaborative Group: Early goal-directed
therapy in the treatment of severe sepsis and septic shock. N Engl
J Med. 345:1368–1377. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Czepielewski RS, Porto BN, Rizzo LB,
Roesler R, Abujamra AL, Pinto LG, Schwartsmann G, Fde Cunha Q and
Bonorino C: Gastrin-releasing peptide receptor (GRPR) mediates
chemotaxis in neutrophils. Proc Natl Acad Sci USA. 109:547–552.
2012. View Article : Google Scholar : PubMed/NCBI
|