Introduction
Orexins or hypocretins are peptide neurotransmitters
expressed in a specific neuron population within the lateral
hypothalamus of the mammalian brain (1,2).
Orexins exert multiple effects in the central nervous system and
modulate several important behaviors, including arousal, food
intake, reward and the sleep-wake cycle (3–7).
This neurotransmitter is part of a complex system, which receives
and integrates multiple signals from metabolic, hormonal, neuronal
and molecular origins (4,6,8–12).
Two orexin isoforms, orexin-A and orexin-B, are
produced from a common precursor protein, prepro-orexin, through
proteolysis (6). Prepro-orexin is
encoded by the Hcrt gene, which contains 2 exons and 1
intron (13). This gene shows high
sequence homology in mouse, rat and human genomes (95% homology
between rat and mouse gene sequences and 82% homology between the
mouse and human gene sequences) (14).
The region upstream of the transcriptional start
site contains essential elements for correct hypothalamic
expression (12,15–17)
and for the binding of several transcription factors, including
forkhead box A2 (Foxa2), insulin-like growth factor binding protein
3 and the nuclear receptors, nuclear receptor subfamily 4 group A
member 1 (Nr4a1, also known as Nur77) and Nr6a1, which promote or
repress the expression of the Hcrt gene (12,18,19).
Within this upstream region, two phylogenetically
conserved elements (OE1 and OE2) direct the expression of the
Hcrt gene in the lateral hypothalamic area (LHA) and repress
it outside this region of the nervous system (16). It has been suggested that the
proximal OE1 element, which is located 278 bp upstream of the
putative transcriptional start site, is required to restrict the
expression of Hcrt to the LHA, as this gene can be expressed
in other areas of the hypothalamus in transgenic animals carrying
mutated versions of this OE1 sequence (16). However, the regulation of orexin
biosynthesis remains to be fully elucidated. There is an interest
in understanding the key molecular elements in orexin regulation as
these may serve to define potential targets to generate novel
therapies for the treatment of eating and sleep disorders, and
other disorders associated with motivation behaviors, including
addictive behavior.
Our previous studies identified two putative
olf-1 sequences within the OE1 element of the Hcrt
gene promoter (20). These
olf-1 sequences are targets for the ebf family of
transcription factors (21). Our
previous studies also revealed that mice carrying null mutations of
the ebf2 locus show a narcoleptic phenotype, characterized
by direct to rapid eye movement transitions from the wake state and
cataplexy episodes (20). This
phenotype is due to the loss of 80% of the orexinergic neurons in
the LHA and decreased expression levels of orexin A in the remnant
cells. Taken together, these results suggest that ebf2 is central
in the correct development, expression and/or differentiation of
the orexinergic circuit in the LHA (20).
Ebf transcription factors are also members of the
basic helix-loop-helix protein family. Members of this family are
known to be master regulators of neural differentiation and
migration, and are involved in establishing particular gene
expression profiles within the central nervous system, which lead
to neural specialization. In particular, ebf2 regulates
neurogenesis, the initiation of neural differentiation, migration
and neural specification in the olfactory system, cerebral cortex
and cerebellum (22–25).
The present study further analyzed the modulation of
the expression of Hcrt by defining a minimal Hcrt
gene promoter containing the proximal OE1 element, and examining
the effects of mutating the olf-1-like elements present in
this minimal promoter. In addition, the present study examined the
effects of overexpressing ebf2 in 293 cells on the reporter gene
expression driven by this minimal promoter.
Materials and methods
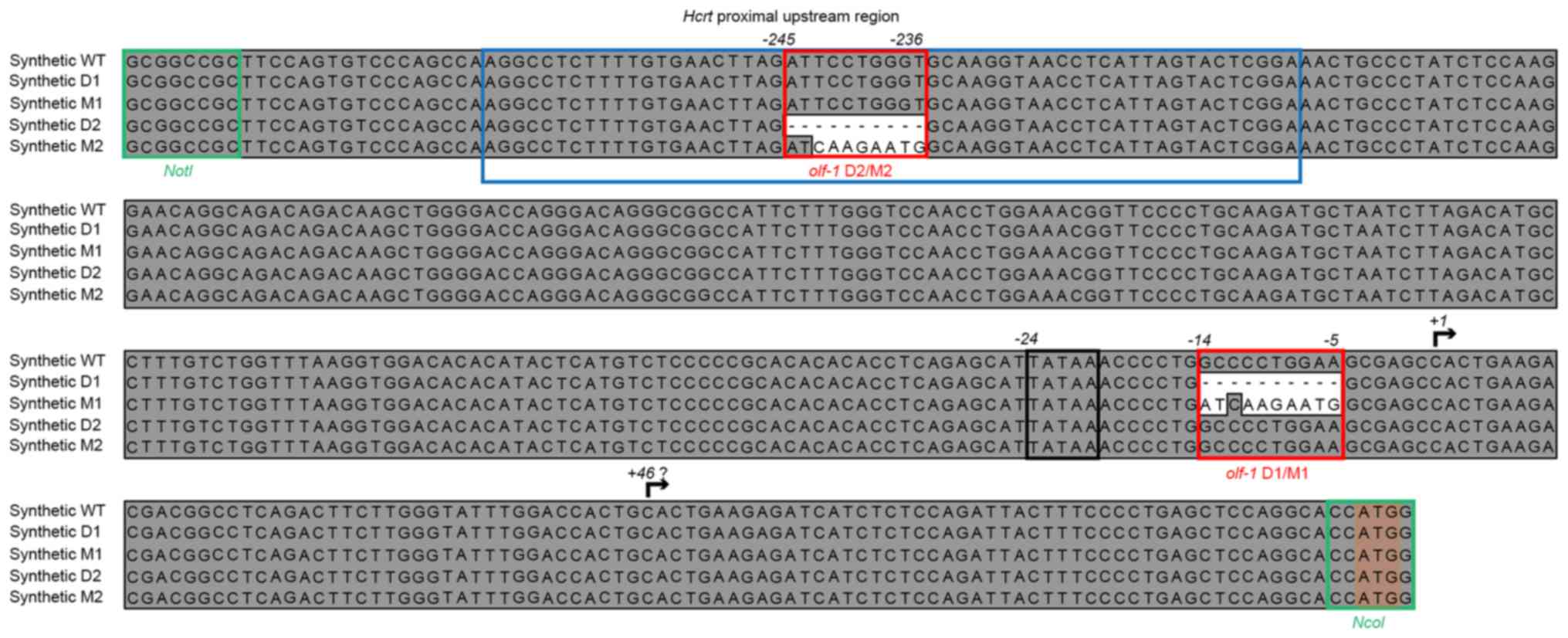
Sequence design and synthesis
The proximal upstream sequence of the murine
Hcrt gene was obtained from the Ensembl database (14). A 390 bp fragment upstream of the
transcription start site was synthetically assembled (Integrated
DNA Technologies, Coralville, IA, USA), in order to generate
constructs containing the wild-type (WT) sequence and several
different deletions and mutations in one or both putative
olf1-like sites (Fig. 1).
The putative transcription and translation start sites from the
Hcrt gene were preserved, by introducing NcoI at the
3′-end of the sequence for cloning purposes. Finally, a NotI
site was added at the 5′-end of the synthetic constructs for
promoter replacement in the reporter gene vector constructs.
Reporter plasmids and expression
vectors
The synthetic Hcrt promoter sequences were
subcloned into the pNiFty3-Luc expression vector (InvivoGen, San
Diego, CA, USA). This reporter plasmid encodes a
coelenterazine-utilizing secreted luciferase and carries resistance
to the antibiotic zeocin. The original interferon (IFN)-β minimal
promoter of the vector was replaced and the synthetic Hcrt
promoter sequences were inserted as NotI-NcoI
fragments. The plasmids were purified through anion-exchange
columns (Qiagen, Venlo, The Netherlands) and all vector constructs
were quantified using a fluorescence method with the Quant-iT
PicoGreen dsDNA Assay kit (Thermo Fisher Scientific, Inc., Waltham,
MA, USA).
Cell culture
The 293 cells were obtained from America Type
Culture Collection (ATCC; Manassas, VA, USA). 293/ebf2 cells were
generated by GenScript (Piscataway, NJ, USA) through lentiviral
transduction of the murine ebf2 cDNA sequence. The cell
lines were grown in Dulbecco's modified Eagle's medium (DMEM;
Caisson Laboratories, Inc., North Logan, UT, USA) supplemented with
10% fetal bovine serum (Gibco; Thermo Fisher Scientific, Inc.) at
37°C and 5% CO2. Puromycin (0.5 µg/ml) and zeocin (0.4
mg/ml; InvivoGen) were added to the ebf2-overexpressing and
pNifty3-carrying semi-stable cells, respectively, as selection
agents.
Transfection and luciferase reporter
assays
The cells were seeded in 96-well plates 1 day prior
to transfection. The cells were transfected with Lipofectamine
reagent (Thermo Fisher Scientific, Inc.) and 100 ng of the reporter
plasmids. Luciferase signal was measured 48 h following
transfection using a Glo-Max multidetection system (Promega
Corporation, Madison, WI, USA) with QUANTI-Luc luminescence assay
reagent (InvivoGen). Semi-stable transfected cells were generated
from these initial transient transfections by selecting the
cultures under 0.4 mg/ml zeocin for 1 month, prior to assaying
luciferase output. All luminescence signals were normalized by the
number of cells present in each well during the assays. The viable
cell number in each well was determined using an ATP-dependent kit
(CellTiter-Glo; Promega Corporation). The concentration of secreted
Lucia luciferase was determined using purified recombinant Lucia
protein (InvivoGen) as a standard for the determination of
enzymatic activity.
RNA isolation and relative expression
analysis
Total RNA was isolated from the cell lines using
PureZol RNA isolation reagent (Bio-Rad Laboratories, Inc.,
Hercules, CA, USA) followed by RNA purification using the RNAeasy
MinElute Cleanup kit (Qiagen) and quantification by ultraviolet
spectrophotometry. Reverse transcription-quantitative polymerase
chain reaction (RT-qPCR) analysis was performed with 1,000 ng of
total RNA in 20 µl of GoScript master mix buffer via GoTaq Probe
2-Step RT-qPCR (Promega Corporation). The amplification of Ebf2 and
GAPDH mRNA was performed with validated, species-specific
hydrolysis probes labeled with FAM and HEX, according to the
manufacturer's instructions (Integrated DNA Technologies). The
amplification of Lucia luciferase mRNA was performed with two sets
of amplification primers and a common hydrolysis probe (Integrated
DNA Technologies), designed using the qPCR designer tool in
MacVector software version 15.5.4 (MacVector, Inc., Apex, NC, USA),
to determine the relative mRNA levels of luciferase starting at two
different nucleotides. All primer and hydrolysis probe sequences
are listed in Table I.
 | Table I.Oligonucleotide sequences. |
Table I.
Oligonucleotide sequences.
| Gene (species) | Forward primer
(5′-3′) | Reverse primer
(5′-3′) | Hydrolysis probe
(5′-3′) |
|---|
| qPCR |
|
GAPDH (H. sapiens) |
TGTAGTTGAGGTCAATGAAGGG |
ACATCGCTCAGACACCATG |
AAGGTCGGAGTCAACGGATTTGGTC |
| EBF2
(H. sapiens) |
AGTCGATGCTGCGAAAAGAAAAG |
CTTCTTGCTCTCCGTCCATGCTTGG |
CGTCCTGGCTGTTTCTGACA |
| Ebf2
(M. musculus) |
AGTCGATGCTGTGAGAAGAAGAG |
CTTCTTGCTCTCCTTCCATGCTTAG |
TGTGCTGGCTGTTTCTGACA |
|
pHcrtWT-Lucia 5′ (+1) TSS |
CATCTCTCCAGATTACTTTCCCCTG |
CCTTATCCTTGAAGCCAGGAATCTC |
AACACAGATGCTGACAGGGG |
|
pHcrtWT-Lucia 3′ (+46)
TSS |
CATCTCTCCAGATTACTTTCCCCTG |
CCTTATCCTTGAAGCCAGGAATCTC |
AACACAGATGCTGACAGGGG |
| EMSA duplex |
| OE1
top |
GCCTCTTTTGTGAACTTAGATTCCTG |
|
|
|
|
GGTGCAAGGTAACCTCATTAGTAC |
|
|
| OE1
bottom |
GTACTAATGAGGTTACCTTGCACCCAG |
|
|
|
|
GAATCTAAGTTCACAAAAGAGGC |
|
|
All PCR amplifications were performed using 2 µl of
the previously synthesized cDNA (equivalent to 100 ng of the total
RNA input sample) in 50 µl of GoTaq PCR master mix, following a
2-step amplification protocol: A starting denaturing step for 2 min
at 95°C, followed by 40 cycles of denaturation for 15 sec at 96°C,
and then annealing and extension for 1 min at 60°C using a
LightCycler 480 Instrument II (Roche Applied Science, Branford, CT,
USA). GAPDH was used as reference gene. The results were analyzed
according to the 2−ΔΔCq method (26) and reported as the number of copies
of the mRNA of interest per copies of the reference GAPDH mRNA.
Electrophoretic mobility shift assay
(EMSA)
A biotinylated 50-bp duplex oligonucleotide was
incubated with nuclear protein extracts from either parental 293 or
293-ebf2 cells with the LightShift Chemiluminescent EMSA kit
according to the manufacturer's instructions (Thermo Fisher
Scientific, Inc.). Nuclear proteins were obtained using NE-PER
Nuclear and Cytoplasmic Extractions Reagents (Thermo Fisher
Scientific, Inc.). The binding reactions, containing 3 µg of
nuclear extract protein and 2 fmol of the biotinylated
oligonucleotide in 20 µl of the recommended binding buffer, were
run on a 12% polyacrylamide gel, and transferred onto a nylon
membrane for 30 min at 15 V using a Transblot transfer SD cell
(Bio-Rad Laboratories, Inc.) and detected using the ChemiDoc MP
system (Bio-Rad Laboratories, Inc.).
Statistical analysis
All data are shown as box plots, with the limits of
the box indicating the 25 and 75% percentiles of the data
distribution, the line in the center of the box indicating the
median of the population, and the error bars indicating the 5 and
95% distribution percentiles. These percentiles overlapped with the
25 and 75% percentiles in certain figures due to the scales of the
horizontal axes. Statistical comparison was performed by analysis
of variance and post hoc Holm-Sidak tests using Sigmastat version
3.5 (Systat Software Inc., San Jose, CA, USA). P<0.01 was
considered to indicate a statistically significant difference.
Results
Proximal 390-base-pair upstream
sequence from the mouse Hcrt gene drives the expression of a
reporter gene in 293 cells
In the present study, a reporter gene vector,
(pHcrtWT-Lucia) carrying ~390 bp of the proximal upstream sequence
of the WT murine Hcrt gene promoter, was transfected into
293 cells. As shown in Fig. 2A (WT
vs. IFN boxes), this small fragment of the Hcrt gene
promoter induced ~100-fold higher levels of luciferase activity,
compared with the IFN-β minimal promoter of the parental vector
(pNifty3-Lucia).
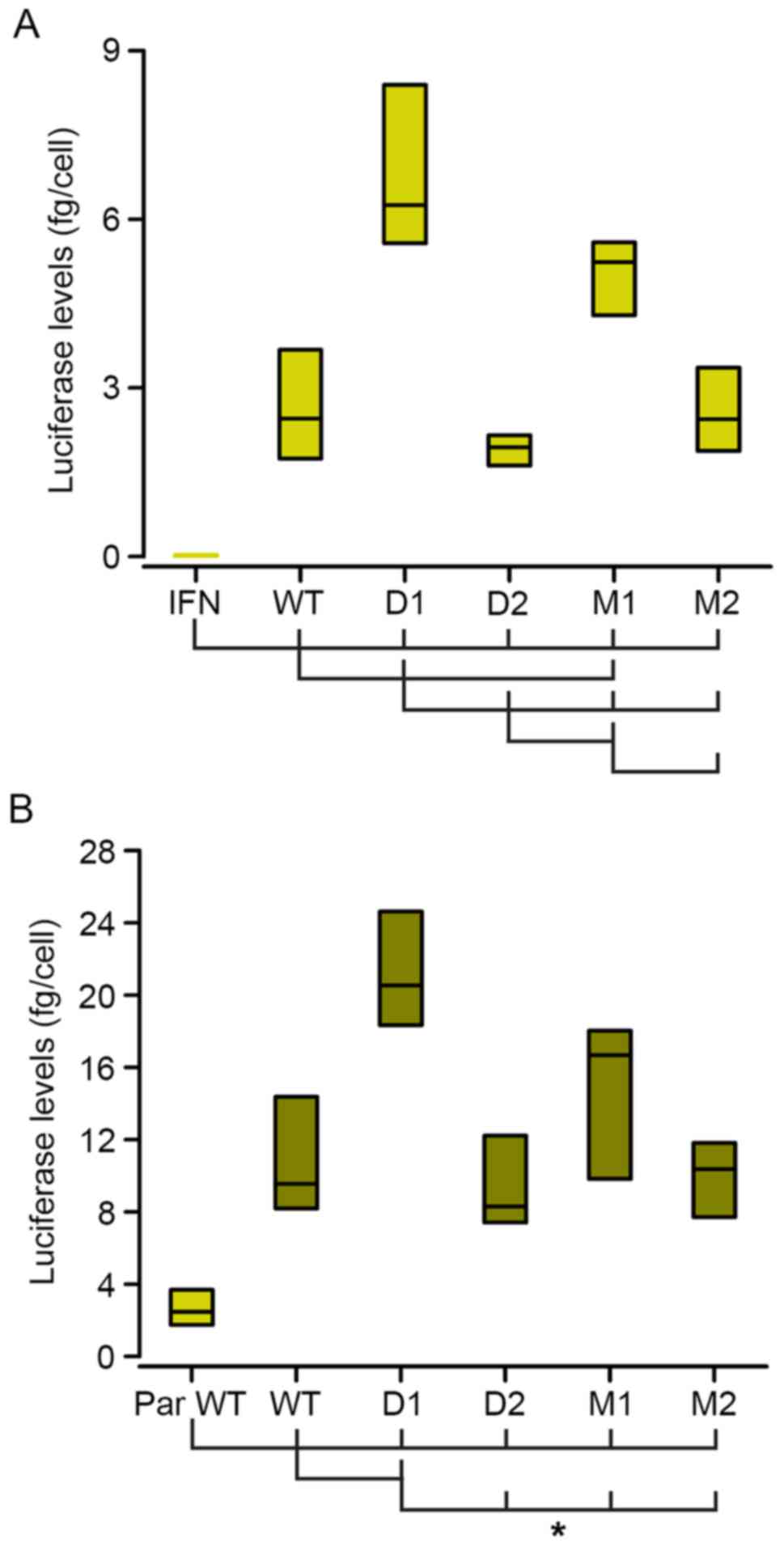 | Figure 2.Expression of the luciferase reporter
gene is driven by the different derivatives of the minimal
Hcrt promoter. Luciferase signals from transiently
transfected parental 293 (A, light green bars) and
ebf2-lentiviral-transduced 293 cells (293-ebf2, B, dark green bars)
showed that the expression of luciferase was driven by the
different Hcrt promoter sequence derivatives. (B)
Overexpression of ebf2 increased the activity of secreted
luciferase. Mutations introduced to the proximal olf-1 site
(D1 and M1 groups) increased, whereas mutations of the distal
olf-1 site (D2 and M2) decreased the expression of
luciferase, compared with that in the WT promoter sequence in
parental 293 cells. In 293-ebf2 cells, deletion of the proximal
site increased luciferase expression, whereas the other mutations
caused no statistically significant changes, although trends
similar to those observed in parental 293 cells were observed.
*P<0.01 (analysis of variance and Holm-Sidak post-hoc
test) vs. groups indicated by joining lines. n=8 experiments/group.
WT, wild-type; D, deletion; M, mutation; ebf2, early B-cell factor
2; IFN, interferon; PAR, parental. |
Perturbation of olf-1 sites in the
minimal Hcrt promoter affects reporter gene expression
The effects of deleting or mutating the two putative
olf-1 sites within the minimal Hcrt promoter sequence
were then analyzed. The deletions consisted of eliminating a 10-bp
fragment spanning the central CCTGG motif of the olf-1
sites. In the mutant promoter constructs, this central motif was
replaced with an AAGAA sequence (Fig.
1).
Deletion or mutation of the proximal olf-1
site (D1 and M1 constructs) increased luciferase expression in the
293 cells, whereas the deletion of the distal olf-1 site (D2
construct) induced a marginal decrease in luciferase levels,
although without statistical significance. Mutation of this distal
site (M2 construct) did not alter luciferase activity, compared
with that observed in the cells transfected with the WT construct
(Fig. 2A).
Overexpression of ebf2 increases
Hcrt-driven reporter gene expression
As the olf-1 sites are putative binding sites
for ebf2, the present study analyzed the expression of this
transcription factor in 293 cells using RT-qPCR probes. The
contribution of the ebf2 transcription factor to Hcrt gene
regulation was analyzed by inducing the overexpression of ebf2 in
293 cells through lentiviral transduction of the murine ebf2
cDNA.
The transduced 293-ebf2 cells expressed high mRNA
levels of ebf2 (400 copies/1,000 copies of GAPDH mRNA) and the
overexpression of ebf2 in the 293 cells increased the luciferase
levels driven by the minimal Hcrt-gene promoter by almost
4-fold, compared with the parental 293 cell line (Fig. 2B). In these 293-ebf2 cells,
deletion of the proximal olf-1 site (D1 construct) increased
luciferase activity by almost 2-fold. Mutation of this proximal
site induced an increase in luciferase expression, although this
was not statistically significant (Fig. 2B).
Deletion or mutation of the distal olf-1 site
(D2 and M2 constructs) did not significantly alter the expression
of luciferase, although the luciferase levels tended to be lower,
compared with those observed with the WT construct in 293-ebf2
cells (Fig. 2B).
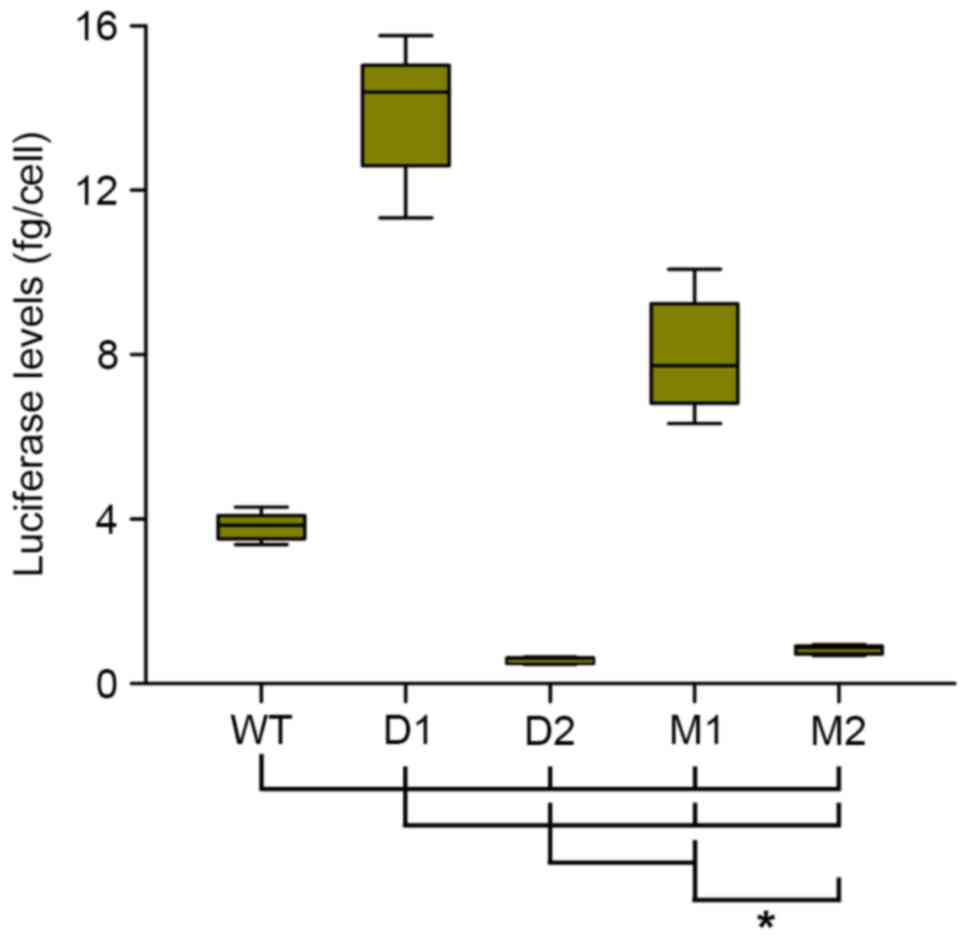
Semi-stable transfections demonstrate
the different roles of the distal and proximal olf-1 sites of the
minimal Hcrt promoter
In order to determine whether the observed
variations in luciferase expression were due to different
transfection rates for each construct, semi-stable cells lines were
generated, which carried the different variant constructs of the
minimal Hcrt-gene promoter, by selecting the transfected
cultures under zeocin for 1 month prior to analyzing luciferase
expression.
In these semi-stable cells lines, deletion (D1) of
the proximal olf-1 site of the Hcrt promoter
increased the levels of luciferase activity almost 4-fold, whereas
the mutation of this proximal site (M1 construct) increased
luciferase expression by 2-fold, compared with those observed with
the WT promoter (Fig. 3). Deletion
(D2) or mutation (M2) of the distal site decreased luciferase
expression by 85 and 80%, respectively, compared with the WT
promoter. These results indicated that the distal olf-1 site
was a positive regulator of the activity of the minimal
Hcrt-gene promoter, as its mutation led to decreased
expression. The proximal olf-1 site was a negative regulator
of this promoter activity.
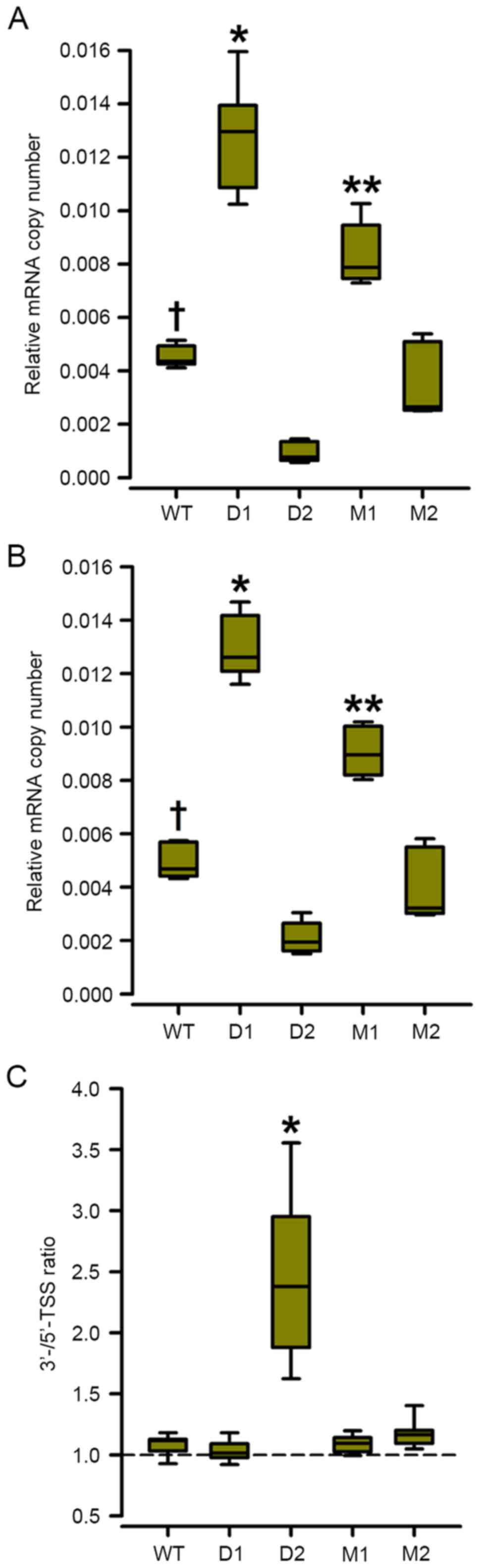
mRNA levels of luciferase correlate
with levels of luciferase secreted by the semi-stable Hcrt-promoter
derivative cell lines
In order to analyze whether the levels of the
secreted luciferase protein were correlated with the levels of
transcription, the steady-state levels of mRNA coding for the
luciferase reporter were analyzed. This involved assaying two sets
of primers and hydrolysis probes, designed to amplify luciferase
mRNA transcripts starting at the consensus transcription starting
site (TSS) at position +1 of the Hcrt gene or at position
+46, which is a sequence identical to the consensus TSS present
downstream in this promoter (Fig.
1). If the transcripts start at position +1, the ratio of the
qPCR products are expected to approach 1, whereas if position +46
is used as the TSS, the 3′-TSS/5′-TSS ratios are expected to
increase.
Deletion or mutation of the proximal olf-1
site (D1 and M1 constructs) increased the steady-state levels of
luciferase-coding mRNA ~2-fold in the semi-stable 293-ebf2 cells
(Fig. 4A and B) and all the
transcripts appeared to start at the consensus TSS, as there were
no changes in the ratio of the levels of the qPCR products
containing the 3′ vs. the 5′ TSS (Fig.
4C).
By contrast, deletion of the distal olf-1
site (D2 construct) decreased the mRNA levels of luciferase
starting the position +1 TSS by ~80%, whereas the mRNA levels
starting at the +46 TSS decreased by ~60% (Fig. 4A). The ratio of
3′-TSS-containing/5′-TSS-containing qPCR increased almost 2.3-fold,
indicating that deletion of the distal olf-1 site shifted
the TSS downstream (Fig. 4C).
Mutation of the distal olf-1 site (M2 construct) induced a
marginal, non-significant decrease in mRNA levels of luciferase and
a shift towards the 3′ TSS (Fig. 4A
and B).
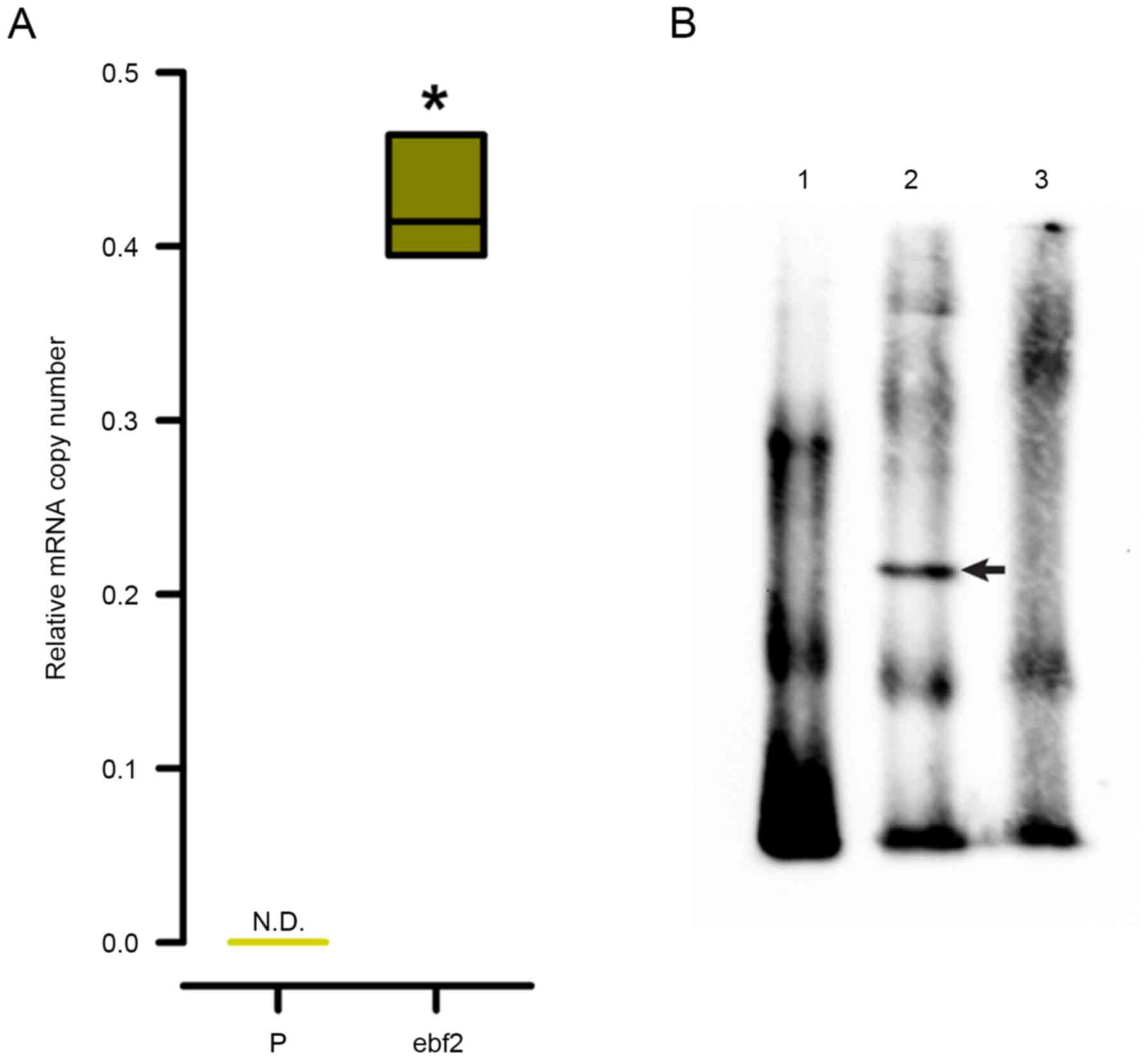
Distal olf-1 sequence of the Hcrt
minimal promoter is a binding site for ebf2
The present study also evaluated the capability of
ebf2 to bind to the distal olf-1 sequence present in the
minimal Hcrt promoter, by performing an EMSA using a
biotinylated 50-bp duplex oligonucleotide corresponding to the OE1
element of the Hcrt gene promoter containing this distal
olf-1 site (Table I). As
shown in Fig. 5A, the mRNA
expression of ebf2 was not detected in the 293 cells, therefore,
the contribution of ebf2 was analyzed by inducing the
overexpression of ebf2 in 293 cells through lentiviral transduction
of the murine ebf2 cDNA. Nuclear protein extracts from the 293-ebf2
cells, but not from the parental 293 cells, induced a shift in the
mobility of the duplex (Fig. 5B).
This shifted band disappeared when the EMSA reaction included a
200-fold molar excess of unlabeled oligonucleotide (Fig. 5B, lane 3).
Discussion
Understanding the mechanisms regulating the
expression of orexin is key to generating novel therapeutic
approaches for disorders of motivation, including feeding and
addictive behavior and sleep behavior, including narcolepsy and
insomnia. The levels of orexin in the brain vary during the day
according to the physical activity of animals. However, whether
these variations are due to changes in the activity of the
orexin-secreting neurons or to changes in orexin biosynthesis,
either at the transcriptional or translational level, remain to be
fully elucidated (3,27–32).
These regulatory mechanisms are also involved in the developmental
determination of the orexinergic phenotype of hypothalamic neurons,
and the in vitro generation of such neurons may lead to
cell-based therapies for orexin replacement.
The promoter region of the Hcrt gene is a key
element in regulating the expression of orexin in the target neural
population within the hypothalamus. This region contains different
control sites, which specify and restrict the expression of
prepro-orexin within the LHA, including orexin regulatory elements
1 and 2 (OE1 and OE2). The OE1 site lies proximal to the putative
transcriptional start site of Hcrt mRNA and it has been
suggested that the OE1 site prevents the gene expression of
Hcrt outside the LHA, as constructs containing a 400-bp
upstream fragment of the Hcrt gene driving a nuclear
β-galactosidase reporter gene were poorly expressed in the brains
of transgenic mice (16). It is
suggested that this restrictive role is mediated by nuclear
receptor response elements (NurREs), which lie in the close
upstream vicinity of the OE1 element (12).
The inhibitory activity of the OE1 element has also
been suggested as the cause of poor expression of reporter genes
driven by the Hcrt promoter in cell lines in vitro
(12,17,19,33,34).
The results of the present study showed that a minimal fragment of
the mouse Hcrt gene, containing just up to the OE1 element,
was able to drive the expression of a secreted luciferase reporter
gene, to levels ~100-fold higher than those driven by the minimal
IFN-β promoter (Fig. 2) in human
embryonic 293 cells.
The differences in the expression between the
constructs in the present study and those of other groups can be
attributed to several factors. By using artificial gene synthesis,
the present study limited the length of the minimal promoter to 17
bp upstream of the OE1 sequence, thus avoiding the inclusion of
NurREs described previously (12,17).
The constructs used in the present study preserved the context of
the transcription and translation start sites of the Hcrt
gene (Fig. 1), and the distance
between the transcription start site and the initial AUG codon of
the Hcrt transcript. Constructs generated in other studies,
driving the expression of reporter genes by using a longer
Hcrt gene promoter, exchange the context of the Hcrt
transcript with those of the reporter genes (12,16).
Overexpression of the transcription factor ebf2 in
the 293 cells increased the Hcrt-driven luciferase
expression by almost 4-fold (Fig.
2B). To the best of our knowledge, this is the first report of
a transcription factor upregulating Hcrt promoter-driven
gene expression in 293 cells. Other transcription factors,
including Foxa2 and Lim-homeobox 9 (Lhx9), have been suggested to
increase in vivo expression at the Hcrt locus
(19,33) and it has been shown that Foxa2
increases Hcrt-driven reporter gene expression in a
hepatocyte cell line (19).
In our previous study, it was demonstrated that the
loss of ebf2 activity in knockout mice led to narcoleptic traits
and a marked decrease in orexinergic neuron numbers (20). The findings of the present study
suggested that this effect is attributed to direct regulation at
the level of the Hcrt locus (Fig. 5B). In addition, ebf2 may contribute
to the cell-type specification process of the orexinergic phenotype
in the LHA through this direct effect and indirect effects on other
regulators of the expression of Hcrt.
It may be that ebf2 is a key regulator of
orexinergic neuron development, joining a network of transcription
factors, including Lhx9 (33,34)
and sonic hedgehog (35), which
may be involved in establishing the mature phenotype of the
orexinergic population in the LHA. The order in which these
transcription factors are involved during development remains to be
elucidated.
The analysis of the proximal Hcrt gene
promoter region in the present study revealed novel features of its
regulatory elements. Mutation of a 10-bp segment with a putative
olf-1-like sequence 7 bp downstream of the TATAA box of the
minimal Hcrt promoter, either by deletion (D1 construct) or
nucleotide substitution (M1 construct), increased the mRNA levels
of the reporter gene (Figs. 2 and
3) and luciferase (Fig. 4) by almost 3-fold. Taken together,
these results suggested that the olf-1-like sequence
downstream of the TATAA box was a negative element, which
controlled the expression of orexin through downregulation.
Perturbations of the distal olf-1-like site
at position-236 (D2 and M2 constructs; Fig. 1), which lies within the OE-1
element, decreased reporter gene expression at the enzymatic and
mRNA levels, particularly in the semi-stable 293-ebf2 derivative
cell lines (Figs. 3 and 4). The OE-1 element has been previously
reported as a restrictive element of the expression of Hcrt,
as deletions within the OE-1 sequence prevent the expression of a
transgenic reporter gene in LHA orexinergic neurons, and direct
expression to a limited small population of hypothalamic neurons
populations outside the LHA (12,16).
Taken together, the previous observations and those of the present
study showed that integrity of the OE-1 element may be necessary to
drive high levels of expression by the Hcrt gene
promoter.
The mutations introduced to the distal olf-1
site appeared to shift the TSS of the reporter gene mRNA to a
downstream position at +46, as the level of mRNA containing this
+46-sequence increased above that containing the TSS sequence at +1
in the semi-stable 293-ebf2 cells carrying the D2 and M2
constructs, particularly when the distal olf-1 site was
deleted (Fig. 4B). This shift
appeared to lead to lower rates of translation of luciferase mRNA,
as the enzymatic activity levels in these semi-stable cells were
lower than the corresponding mRNA levels, compared with those in
cells carrying the WT construct (Figs.
3 and 4).
The distal olf-1 sequence was identified as a
binding site for ebf2 as nuclear extracts from 293-ebf2 cells, but
not from parental 293 cells, induced a shift in electrophoretic
mobility, which was competed by an unlabeled oligonucleotide
contacting this olf-1 sequence (Fig. 5B). This result suggested that ebf2
was a direct regulator of Hcrt-gene promoter driven
expression in vitro, however, the relevance of this effect
during development or the adult stage in vivo remains to be
elucidated.
The data obtained in the present study demonstrated
that a minimal sequence derived from the murine Hcrt gene
was able to drive high levels of expression in a heterologous
system and that ebf2 is a transcription factor, which increased the
activity of this Hcrt gene minimal promoter in vitro.
The identification of this minimal promoter may lead to improved
vectors for gene therapy, where the expression of orexin is
required, and the identification of ebf2 as a direct regulator of
the expression of orexin may lead to the development of
orexin-expressing cells for cell therapies in the treatment of
narcolepsy.
Acknowledgements
This study was supported by grants from Consejo
Nacional de Ciencia y Tecnología, Mexico (grant no. 220342 to R.
Vidaltamayo and grant no. 220006 to V. Zomosa-Signoret) and
Vicerrectoría Académica-Universidad de Monterrey (grant nos. 14011,
15027 and 17559 to Román Vidaltamayo). Authors would like to thank
Mr. Alejandro Trejo and Mr. Alejandro Treviño for their technical
assistance.
References
|
1
|
de Lecea L, Kilduff TS, Peyron C, Gao X,
Foye PE, Danielson PE, Fukuhara C, Battenberg EL, Gautvik VT,
Bartlett FS II, et al: The hypocretins: Hypothalamus-specific
peptides with neuroexcitatory activity. Proc Natl Acad Sci USA.
95:pp. 322–327. 1998; View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sakurai T, Amemiya A, Ishii M, Matsuzaki
I, Chemelli RM, Tanaka H, Williams SC, Richarson JA, Kozlowski GP,
Wilson S, et al: Orexins and orexin receptors: A family of
hypothalamic neuropeptides and G protein-coupled receptors that
regulate feeding behavior. Cell. 92:573–585. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Routh VH, Hao L, Santiago AM, Sheng Z and
Zhou C: Hypothalamic glucose sensing: Making ends meet. Front Syst
Neurosci. 8:2362014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tsujino N and Sakurai T: Role of orexin in
modulating arousal, feeding, and motivation. Front Behav Neurosci.
7:282013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nuñez A, Rodrigo-Angulo ML, Andrés ID and
Garzón M: Hypocretin/orexin neuropeptides: Participation in the
control of sleep-wakefulness cycle and energy homeostasis. Curr
Neuropharmacol. 7:50–59. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ohno K and Sakurai T: Orexin neuronal
circuitry: Role in the regulation of sleep and wakefulness. Front
Neuroendocrinol. 29:70–87. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Matsuki T and Sakurai T: Orexins and
orexin receptors: From molecules to integrative physiology. Results
Probl Cell Differ. 46:27–55. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Cai XJ, Widdowson PS, Harrold J, Wilson S,
Buckingham RE, Arch JR, Tadayyon M, Clapham JC, Wilding J and
Williams G: Hypothalamic orexin expression: Modulation by blood
glucose and feeding. Diabetes. 48:2132–2137. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Griffond B, Risold PY, Jacquemard C,
Colard C and Fellmann D: Insulin-induced hypoglycemia increases
preprohypocretin (orexin) mRNA in the rat lateral hypothalamic
area. Neurosci Lett. 262:77–80. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wagner D, Salin-Pascual R, Greco MA and
Shiromani PJ: Distribution of hypocretin-containing neurons in the
lateral hypothalamus and C-fos-immunoreactive neurons in the VLPO.
Sleep Res Online. 3:35–42. 2000.PubMed/NCBI
|
|
11
|
Sakurai T, Nagata R, Yamanaka A, Kawamura
H, Tsujino N, Muraki Y, Kageyama H, Kunita S, Takahashi S, Goto K,
et al: Input of orexin/hypocretin neurons revealed by a genetically
encoded tracer in mice. Neuron. 46:297–308. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tanaka S, Kodama T, Nonaka T, Toyoda H,
Arai M, Fukazawa M, Honda Y, Honda M and Mignot E: Transcriptional
regulation of the hypocretin/orexin gene by NR6A1. Biochem Biophys
Res Commun. 403:178–183. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sakurai T, Moriguchi T, Furuya K, Kajiwara
N, Nakamura T, Yanagisawa M and Goto K: Structure and function of
human prepro-orexin gene. J Biol Chem. 274:17771–17776. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cunningham F, Amode MR, Barrell D, Beal K,
Billis K, Brent S, Carvalho-Silva D, Clapham P, Coates G,
Fitzgerald S, et al: Ensembl 2015. Nucleic Acids Res. 43(Database
issue): D662–D669. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Waleh NS, Apte-Deshpande A, Terao A, Ding
J and Kilduff TS: Modulation of the promoter region of
prepro-hypocretin by alpha-interferon. Gene. 262:123–128. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Moriguchi T, Sakurai T, Takahashi S, Goto
K and Yamamoto M: The human prepro-orexin gene regulatory region
that activates gene expression in the lateral region and represses
it in the medial regions of the hypothalamus. J Biol Chem.
277:16985–16992. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tanaka S: Transcriptional regulation of
the hypocretin/orexin gene. Vitam Horm. 89:75–90. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Honda M, Eriksson KS, Zhang S, Tanaka S,
Lin L, Salehi A, Hesla PE, Maehlen J, Gaus SE, Yanagisawa M, et al:
IGFBP3 colocalizes with and regulates hypocretin (orexin). PLoS
One. 4:e42542009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Silva JP, von Meyenn F, Howell J, Thorens
B, Wolfrum C and Stoffel M: Regulation of adaptive behaviour during
fasting by hypothalamic Foxa2. Nature. 462:646–650. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
De La Herrán-Arita AK, Zomosa-Signoret VC,
Millán-Aldaco DA, Palomero-Rivero M, Guerra-Crespo M, Drucker-Colín
R and Vidaltamayo R: Aspects of the narcolepsy-cataplexy syndrome
in O/E3-null mutant mice. Neuroscience. 183:134–143. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang SS, Lewcock JW, Feinstein P,
Mombaerts P and Reed RR: Genetic disruptions of O/E2 and O/E3 genes
reveal involvement in olfactory receptor neuron projection.
Development. 131:1377–1388. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Garcia-Dominguez M, Poquet C, Garel S and
Charnay P: Ebf gene function is required for coupling neuronal
differentiation and cell cycle exit. Development. 130:6013–6025.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Croci L, Chung SH, Masserdotti G, Gianola
S, Bizzoca A, Gennarini G, Corradi A, Rossi F, Hawkes R and
Consalez GG: A key role for the HLH transcription factor
EBF2COE2,O/E-3 in Purkinje neuron migration and cerebellar cortical
topography. Development. 133:2719–2729. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chuang SM, Wang Y, Wang Q, Liu KM and Shen
Q: Ebf2 marks early cortical neurogenesis and regulates the
generation of cajal-retzius neurons in the developing cerebral
cortex. Dev Neurosci. 33:479–493. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Roby YA, Bushey MA, Cheng LE, Kulaga HM,
Lee SJ and Reed RR: Zfp423/OAZ mutation reveals the importance of
Olf/EBF transcription activity in olfactory neuronal maturation. J
Neurosci. 32:13679–13688. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Burt J, Alberto CO, Parsons MP and
Hirasawa M: Local network regulation of orexin neurons in the
lateral hypothalamus. Am J Physiol Regul Integr Comp Physiol.
301:R572–R580. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tanno S, Terao A, Okamatsu-Ogura Y and
Kimura K: Hypothalamic prepro-orexin mRNA level is inversely
correlated to the non-rapid eye movement sleep level in high-fat
diet-induced obese mice. Obes Res Clin Pract. 7:e251–e257. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Arafat AM, Kaczmarek P, Skrzypski M,
Pruszyńska-Oszmałek E, Kołodziejski P, Adamidou A, Ruhla S,
Szczepankiewicz D, Sassek M, Billert M, et al: Glucagon regulates
orexin A secretion in humans and rodents. Diabetologia.
57:2108–2116. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Iwasa T, Matsuzaki T, Munkhzaya M,
Tungalagsuvd A, Kuwahara A, Yasui T and Irahara M: Developmental
changes in the hypothalamic mRNA levels of prepro-orexin and orexin
receptors and their sensitivity to fasting in male and female rats.
Int J Dev Neurosci. 46:51–54. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Justinussen JL, Holm A and Kornum BR: An
optimized method for measuring hypocretin-1 peptide in the mouse
brain reveals differential circadian regulation of hypocretin-1
levels rostral and caudal to the hypothalamus. Neuroscience.
310:354–361. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhou Y, Leri F, Cummins E and Kreek MJ:
Individual differences in gene expression of vasopressin, D2
receptor, POMC and orexin: Vulnerability to relapse to
heroin-seeking in rats. Physiol Behav. 139:127–135. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dalal J, Roh JH, Maloney SE, Akuffo A,
Shah S, Yuan H, Wamsley B, Jones WB, de Guzman Strong C, Gray PA,
et al: Translational profiling of hypocretin neurons identifies
candidate molecules for sleep regulation. Genes Dev. 27:565–578.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liu J, Merkle FT, Gandhi AV, Gagnon JA,
Woods IG, Chiu CN, Shimogori T, Schier AF and Prober DA:
Evolutionarily conserved regulation of hypocretin neuron
specification by Lhx9. Development. 142:1113–1124. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Szabó NE, Zhao T, Cankaya M, Theil T, Zhou
X and Alvarez-Bolado G: Role of neuroepithelial Sonic hedgehog in
hypothalamic patterning. J Neurosci. 29:6989–7002. 2009. View Article : Google Scholar : PubMed/NCBI
|