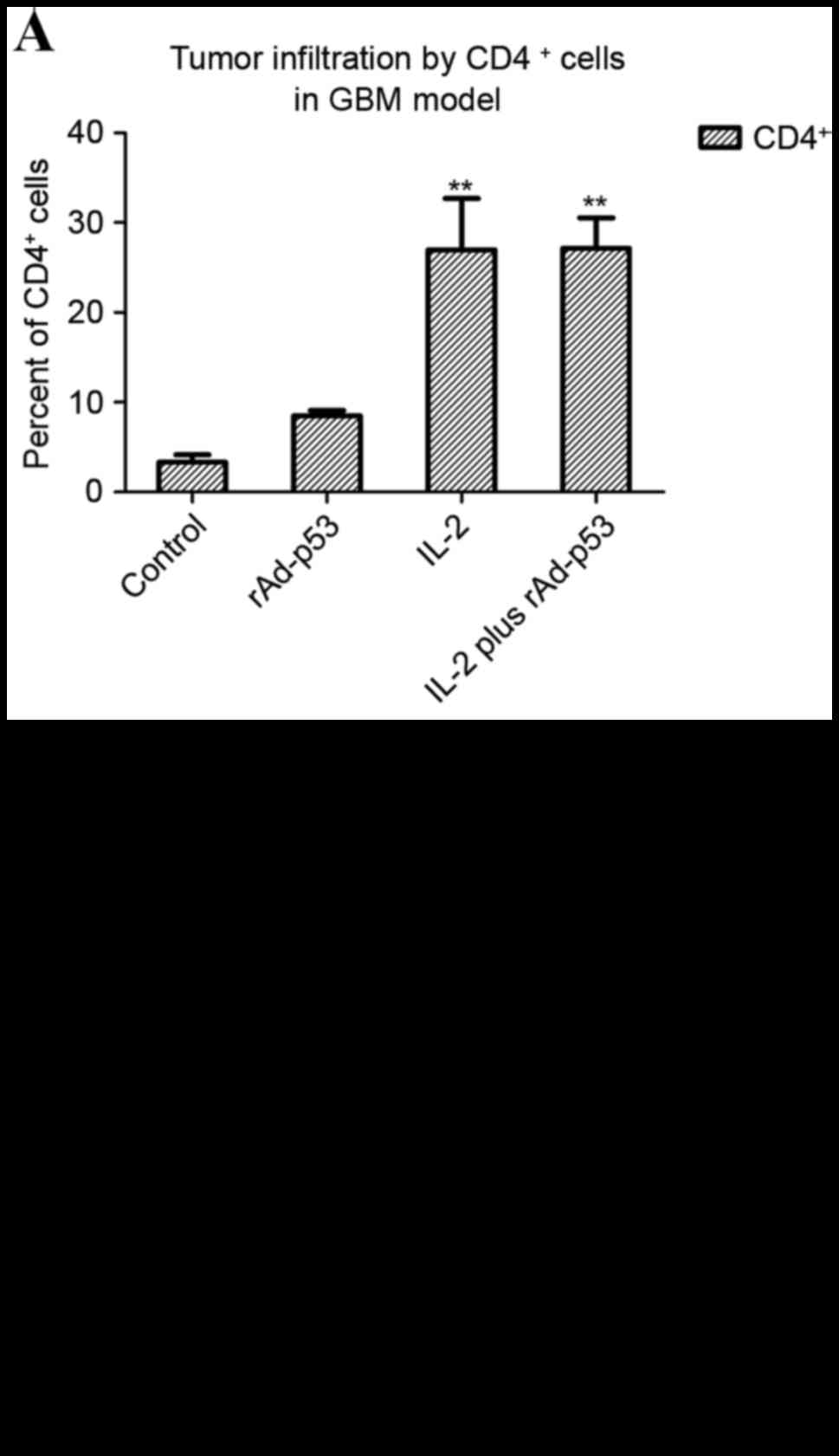
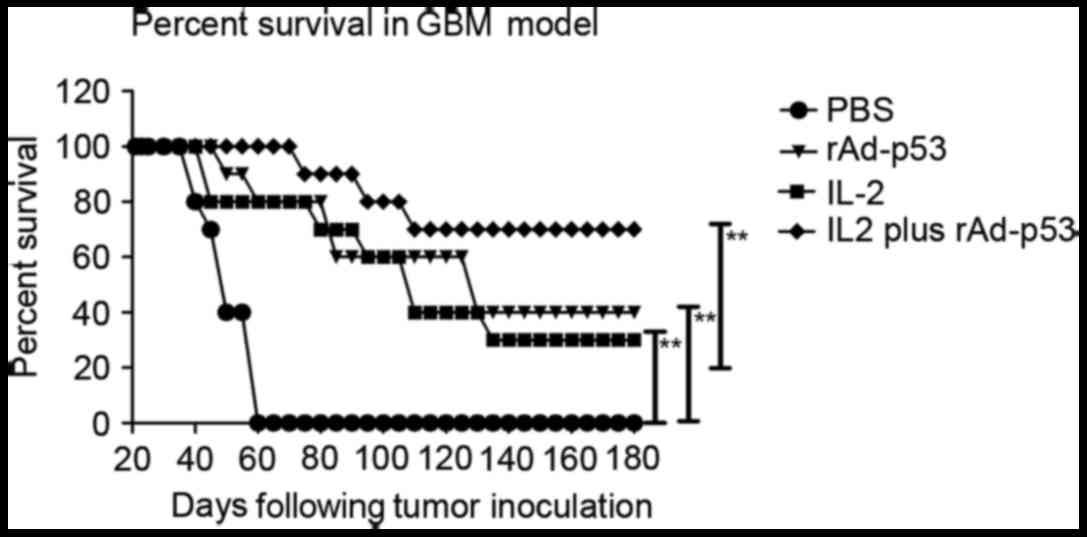
|
1
|
Lu M, Zhang X, Zhang M, Chen H, Dou W, Li
S and Dai J: Non-model segmentation of brain glioma tissues with
the combination of DWI and fMRI signals. Biomed Mater Eng. 26 Suppl
1:S1315, 2015–S1324. 2015.
|
|
2
|
Chow KK, Naik S, Kakarla S, Brawley VS,
Shaffer DR, Yi Z, Rainusso N, Wu MF, Liu H, Kew Y, et al: T cells
redirected to EphA2 for the immunotherapy of glioblastoma. Mol
Ther. 21:629–637. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Koekkoek JA, Postma TJ, Heimans JJ,
Reijneveld JC and Taphoorn MJ: Antiepileptic drug treatment in the
end-of-life phase of glioma patients: A feasibility study. Support
Care Cancer. 24:1633–1638. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bai FL, Tian H, Yu QZ, Renl GP and Li DS:
Expressing foreign genes by Newcastle disease virus for cancer
therapy. Mol Biol (Mosk). 49:195–204. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Snyder A, Zamarin D and Wolchok JD:
Immunotherapy of Melanoma. Prog Tumor Res. 42:22–29.
2015.PubMed/NCBI
|
|
6
|
Bai FL, Yu YH, Tian H, Ren GP, Wang H,
Zhou B, Han XH, Yu QZ and Li DS: Genetically engineered Newcastle
disease virus expressing interleukin-2 and TNF-related
apoptosis-inducing ligand for cancer therapy. Cancer Biol Ther.
15:1226–1238. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Shi L, Zhou Q, Wu J, Ji M, Li G, Jiang J
and Wu C: Efficacy of adjuvant immunotherapy with cytokine-induced
killer cells in patients with locally advanced gastric cancer.
Cancer Immunol Immunother. 61:2251–2259. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen Y, Guo ZQ, Shi CM, Zhou ZF, Ye YB and
Chen Q: Efficacy of adjuvant chemotherapy combined with
immunotherapy with cytokine-induced killer cells for gastric cancer
after d2 gastrectomy. Int J Clin Exp Med. 8:7728–7736.
2015.PubMed/NCBI
|
|
9
|
Lippitz BE: Cytokine patterns in patients
with cancer: A systematic review. Lancet Oncol. 14:e218–e228. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Karsy M, Neil JA, Guan J, Mahan MA, Colman
H and Jensen RL: A practical review of prognostic correlations of
molecular biomarkers in glioblastoma. Neurosurg Focus. 38:E42015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yamanishi Y, Boyle DL, Green DR, Keystone
EC, Connor A, Zollman S and Firestein GS: p53 tumor suppressor gene
mutations in fibroblast-like synoviocytes from erosion synovium and
non-erosion synovium in rheumatoid arthritis. Arthritis Res Ther.
7:R12–R18. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ohgaki H, Eibl RH, Schwab M, Reichel MB,
Mariani L, Gehring M, Petersen I, Höll T, Wiestler OD and Kleihues
P: Mutations of the p53 tumor suppressor gene in neoplasms of the
human nervous system. Mol Carcinog. 8:74–80. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sakai E, Rikimaru K, Ueda M, Matsumoto Y,
Ishii N, Enomoto S, Yamamoto H and Tsuchida N: The p53
tumor-suppressor gene and ras oncogene mutations in oral
squamous-cell carcinoma. Int J Cancer. 52:867–872. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Greenblatt MS, Bennett WP, Hollstein M and
Harris CC: Mutations in the p53 tumor suppressor gene: Clues to
cancer etiology and molecular pathogenesis. Cancer Res.
54:4855–4878. 1994.PubMed/NCBI
|
|
15
|
Rivlin N, Brosh R, Oren M and Rotter V:
Mutations in the p53 tumor suppressor gene: Important milestones at
the various steps of tumorigenesis. Genes Cancer. 2:466–474. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yue Q, Yulong G, Liting Q, Shuai Y, Delong
L, Yubao L, Lili J, Sidang L and Xiaomei W: Mutations in and
expression of the tumor suppressor gene p53 in egg-type chickens
infected with subgroup J avian leukosis virus. Vet Pathol.
52:1052–1066. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hollstein M, Sidransky D, Vogelstein B and
Harris CC: p53 mutations in human cancers. Science. 253:49–53.
1991. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hamzehloie T, Mojarrad M, Hasanzadeh
Nazarabadi M and Shekouhi S: The role of tumor protein 53 mutations
in common human cancers and targeting the murine double minute
2-p53 interaction for cancer therapy. Iran J Med Sci. 37:3–8.
2012.PubMed/NCBI
|
|
19
|
Fagin JA: Tumor suppressor genes in human
thyroid neoplasms: p53 mutations are associated undifferentiated
thyroid cancers. J Endocrinol Invest. 18:140–142. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Harris CC and Hollstein M: Clinical
implications of the p53 tumor-suppressor gene. N Engl J Med.
329:1318–1327. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Casson AG, Evans SC, Gillis A, Porter GA,
Veugelers P, Darnton SJ, Guernsey DL and Hainaut P: Clinical
implications of p53 tumor suppressor gene mutation and protein
expression in esophageal adenocarcinomas: Results of a ten-year
prospective study. J Thorac Cardiovasc Surg. 125:1121–1131. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kuczyk MA, Serth J, Hervatin C, Arndt H,
Derendorf L, Thon WF and Jonas U: Detection of p53
tumor-suppressor-gene protein in bladder tumors and prostate
cancer: Possible clinical implications. World J Urol. 12:345–351.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chen GX, Zheng LH, Liu SY and He XH:
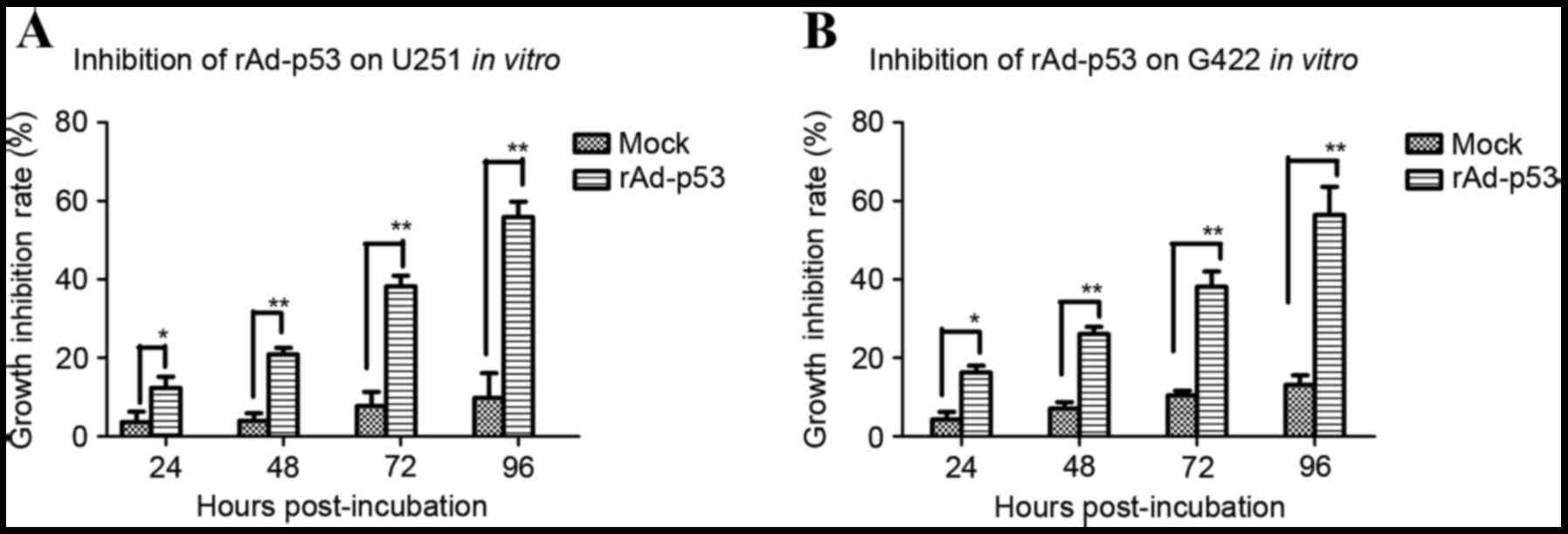
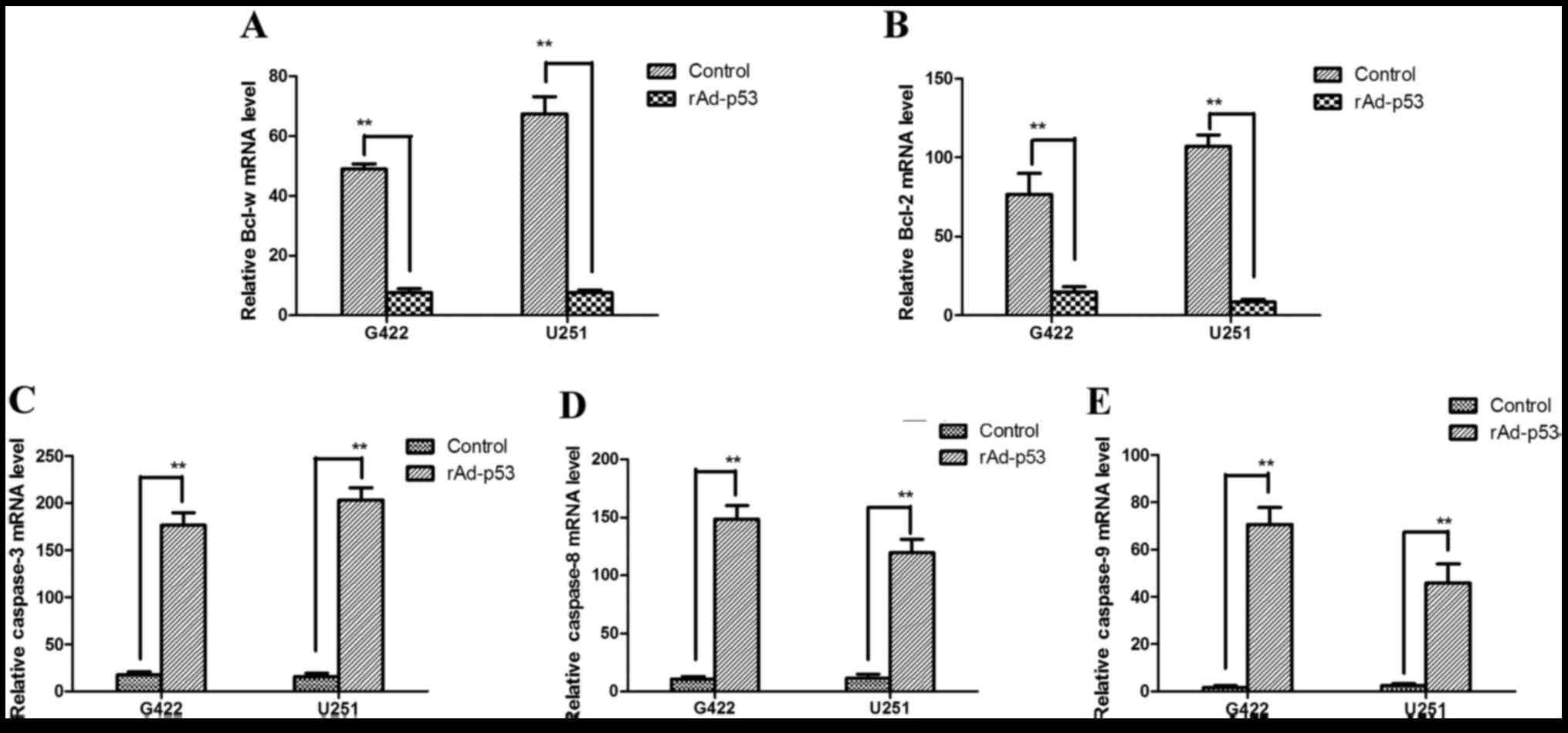
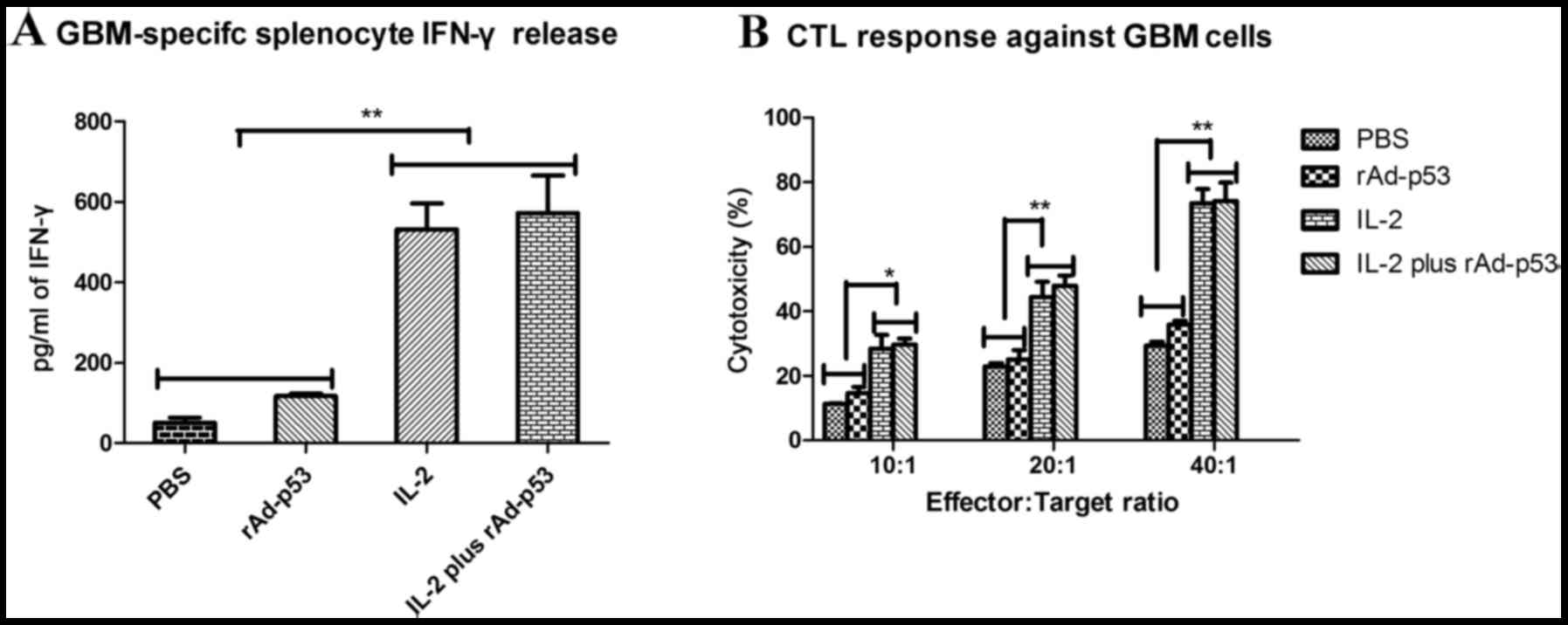
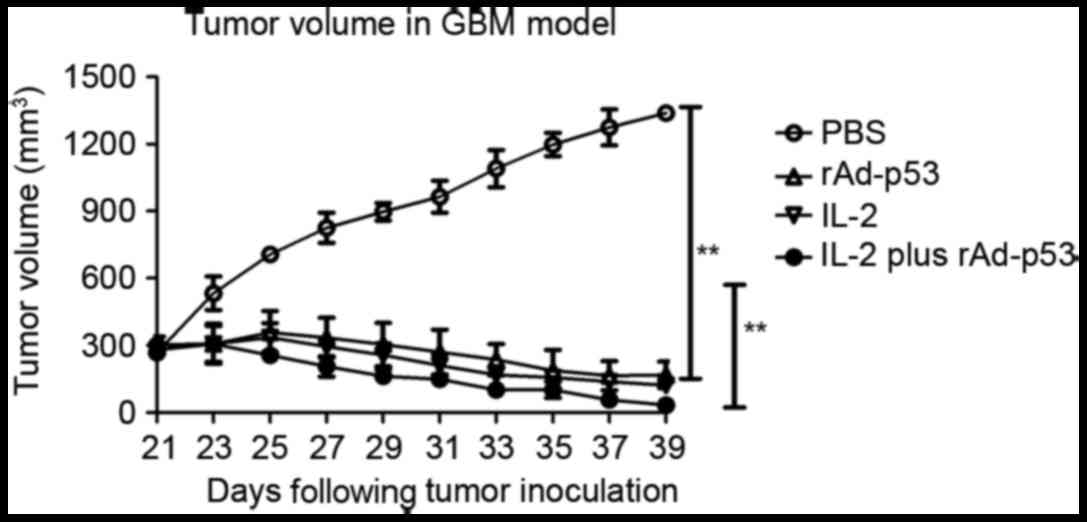
rAd-p53 enhances the sensitivity of human gastric cancer cells to
chemotherapy. World J Gastroenterol. 17:4289–4297. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Xie Q, Liang BL, Wu YH, Zhang J, Chen MW,
Liu HY, Gu XF and Xu J: Synergistic anticancer effect of rAd/P53
combined with 5-fluorouracil or iodized oil in the early
therapeutic response of human colon cancer in vivo. Gene.
499:303–308. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
National Research Council (US) Committee
for the Update of the Guide for the Care and Use of Laboratory
Animals: Guide for the Care and Use of Laboratory Animals. 8th.
National Academies Press (US); Washington, DC: 2011
|
|
26
|
Greaves MF and Brown G: Purification of
human T and B lymphocytes. J Immunol. 112:420–423. 1974.PubMed/NCBI
|
|
27
|
Zamarin D, Vigil A, Kelly K, Garcia-Sastre
A and Fong Y: Genetically engineered Newcastle disease virus for
malignant melanoma therapy. Gene Ther. 16:796–804. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Thomas AA, Ernstoff MS and Fadul CE:
Immunotherapy for the treatment of glioblastoma. Cancer J.
18:59–68. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Larsen CJ: Cellular immunotherapy and
glioblastoma: A hopeful treatment? Bull Cancer.
98:4572011.PubMed/NCBI
|
|
31
|
Varghese S, Rabkin SD, Nielsen GP,
MacGarvey U, Liu R and Martuza RL: Systemic therapy of spontaneous
prostate cancer in transgenic mice with oncolytic herpes simplex
viruses. Cancer Res. 67:9371–9379. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Husain SR, Behari N, Kreitman RJ, Pastan I
and Puri RK: Complete regression of established human glioblastoma
tumor xenograft by interleukin-4 toxin therapy. Cancer Res.
58:3649–3653. 1998.PubMed/NCBI
|
|
33
|
Debinski W, Gibo DM, Obiri NI, Kealiher A
and Puri RK: Novel anti-brain tumor cytotoxins specific for cancer
cells. Nat Biotechnol. 16:449–453. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Bera TK, Viner J, Brinkmann E and Pastan
I: Pharmacokinetics and antitumor activity of a bivalent
disulfide-stabilized Fv immunotoxin with improved antigen binding
to erbB2. Cancer Res. 59:4018–4022. 1999.PubMed/NCBI
|
|
35
|
Ghetie MA, Richardson J, Tucker T, Jones
D, Uhr JW and Vitetta ES: Antitumor activity of Fab' and
IgG-anti-CD22 immunotoxins in disseminated human B lymphoma grown
in mice with severe combined immunodeficiency disease: Effect on
tumor cells in extranodal sites. Cancer Res. 51:5876–5880.
1991.PubMed/NCBI
|
|
36
|
Sinkovics JG and Horvath JC: Natural and
genetically engineered viral agents for oncolysis and gene therapy
of human cancers. Arch Immunol Ther Exp (Warsz). 56 Suppl 1:3S–59S.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Yoshida Y, Sadata A, Zhang W, Saito K,
Shinoura N and Hamada H: Generation of fiber-mutant recombinant
adenoviruses for gene therapy of malignant glioma. Hum Gene Ther.
9:2503–2515. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kusnierczyk H, Pajtasz-Piasecka E, Koten
JW, Bijleveld C, Krawczyk K and Den Otter W: Further development of
local IL-2 therapy of cancer: Multiple versus single IL-2 treatment
of transplanted murine colon carcinoma. Cancer Immunol Immunother.
53:445–452. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Pantuck AJ and Belldegrun AS: Phase I
clinical trial of interleukin 2 (IL-2) gene therapy for prostate
cancer. Curr Urol Rep. 2:332001. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Baek S, Kim YM, Kim SB, Kim CS, Kwon SW,
Kim Y, Kim H and Lee H: Therapeutic DC vaccination with IL-2 as a
consolidation therapy for ovarian cancer patients: A phase I/II
trial. Cell Mol Immunol. 12:87–95. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Tan Y, Xu M, Wang W, Zhang F, Li D, Xu X,
Gu J and Hoffman RM: IL-2 gene therapy of advanced lung cancer
patients. Anticancer Res. 16:1993–1998. 1996.PubMed/NCBI
|
|
42
|
Gilly FN, Beaujard A, Bienvenu J, Trillet
Lenoir V, Glehen O, Thouvenot D, Malcus C, Favrot M, Dumontet C,
Lombard-Bohas C, et al: Gene therapy with Adv-IL-2 in unresectable
digestive cancer: Phase I–II study, intermediate report.
Hepatogastroenterology. 46 Suppl 1:S1268–S1273. 1999.
|