Introduction
Gestational hypertension is a complication that
specifically occurs in late pregnancy, and includes gestational
hypertension, preeclampsia, eclampsia and chronic hypertension
(1,2). A clinical investigation suggested
that hypertensive disorder is a complication during pregnancy, is a
common pregnancy-associated disease and is becoming increasingly
present worldwide (3). However,
the etiology of gestational hypertension is yet to be clearly
investigated. Currently, insulin resistance,
renin-angiotensin-aldosterone system (RAS) dysfunction,
inflammation, obesity-induced interaction of neurohumoral and renal
mechanisms are considered to be contributing factors of gestational
hypertension (4,5). In addition, genetic factors that
contribute to gestational hypertension have been well documented in
a previous study (6). Furthermore,
gestational hypertension seriously affects the life expectancy of
the mother and fetus in the clinical setting (7,8).
Gestational hypertension is one of the leading causes of maternal,
fetal and neonatal morbidity and mortality (4,9).
Reports have suggested that the incidence rate of gestational
hypertension is approxinately 7–12% in China, presenting a higher
than the average incidence throughout the world (10).
Evidence has revealed that clinical assisted
reproductive technology treatment, including artificial
insemination and ovulation induction, may increase the incidence of
gestational hypertension and preeclampsia (11). In addition, there are a large
number of factors that may induce gestational hypertension, and
have been found to serve a role in the etiology of these
hypertensive disorders, including work stress, depression and
anxiety (12,13). Reports also have indicated that
angiotensin II type 1 receptor (Ang-IITR), vasodilation converting
enzyme (VCE) and α-1A adrenergic receptor (α-ADR) serve crucial
roles in the process of gestational hypertension and may be
potential targets for the treatment of patients with gestational
hypertension in clinical (3,12,13).
Therefore, a large number of drugs targeting Ang-IITR were
introduced and their therapeutic effects for the treatment of
gestational hypertension were evaluated.
Ang-IITR has been demonstrated to be a potential
target in the progression of gestational hypertension and higher
expression levels of this receptor exist in vascular endothelial
cells in patients with gestational hypertension, compared with
healthy pregnant women (14,15).
Antihypertensive agents targeting Ang-IITR are the most common used
drugs for remission patients with gestational hypertension
(16,17). Candesartan
(C24H20N6O3), is an
antihypertensive drug, and is a selective Ang-IITR antagonist that
acts by reducing peripheral vascular resistance and
vasoconstriction (18,19). Candesartan is used to treat
hypertension and prevents the constriction (narrowing) of blood
vessels, including veins and arteries. In addition, fibrinolysis
and insulin sensitivity was improved by treatment with Imidapril
and Candesartan in patients with hypertension (20). The effect of Candesartan treatment
on lipid metabolism in patients with hypertension has also been
investigated in a retrospective longitudinal survey (21). However, the efficacy of Candesartan
for gestational hypertension has not been previously studied.
In the present study, the therapeutic effects of
Candesartan on gestational hypertension in a mouse model was
investigated, in addition to the underlying molecular mechanisms by
which it contributes to attenuating gestational hypertension
induced by homocysteine. The aim of the present study was to
characterize alterations in Ang-IITR, soluble fms-like tyrosine
kinase 1 (sFlt-1), insulin resistance homeostasis model assessment
score and associated cardiovascular risk factors in mice with
gestational hypertension after treatment with Candesartan. NF-κB
activation and nuclear factor NF-κB p65 subunit (p65), inhibitor of
NF-κB kinase subunit β (IKK-β) and NF-κB inhibitor α (IκBα)
expression levels were also investigated in vascular endothelial
cells in mice treated by Candesartan.
Materials and methods
Ethics statement
The present study was carried out in strict
accordance with the recommendations in the Guide for Shandong
Provincial Hospital affiliated to Shandong University. All surgery
and euthanasia were performed to minimize suffering. This study was
approved by the ethics committee of Shangdong University and Taian
City Central Hospital (Jinan, China).
Cell culture and reagents
Vascular endothelial cells were isolated from
experimental mice and cultured in Minimum Essential media (Merck
KGaA, Darmstadt, Germany) supplemented with 10% fetal bovine serum
(Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA).
Vascular endothelial cells were cultured at 37°C in a humidified
atmosphere of 5% CO2. Cells were treated with
Candesartan and/or NF-κB activity inhibitor JSH23 (Sigma-Aldrich;
Merck KGaA) for 12 h at 37°C.
Western blot analysis
Vascular endothelial cells were homogenized in
radioimmunoprecipitation assay lysis buffer (Sigma-Aldrich; Merck
KGaA) containing protease-inhibitor and centrifuged at 6,000 × g at
4°C for 10 min. The resulting supernatant was used for analysis of
proteins. Protein concentration was determined with a bicinchoninic
acid protein assay kit. Protein (10 µg/lane) was separated by 12.5%
SDS-PAGE and subsequently transferred onto polyvinylidene fluoride
membranes as previously described (22). For western blotting, membranes were
incubated with the following primary antibodies for 12 h at 4°C:
Superoxide dismutase (SOD; 1:1,000; cat. no. A91960Hu01; Shanghai
Wuqi Biotechnology Co., Ltd., Shanghai, China) sFlt-1 (1:1,000;
cat. no. ab230516), tumor necrosis factor α (TNF-α; 1:1,000; cat.
no. ab6671), interleukin-2 (1:1,000; cat. no. ab80780), reactive
oxygen species (ROS; 1:1,000; cat. no. ab191522), transcription
factor p65 (1:1,000; cat. no. ab16502), inhibitor of nuclear factor
κ-B kinase subunit β (Ikk-β; 1:1,000; cat. no. ab17870), NF-κB
inhibitor α (IκBα; 1:1,000; cat. no. ab16502) and β-actin (1:1,000;
cat. no. ab8227; all Abcam, Cambridge, MA, USA) were added
following blocking in 5% skimmed milk for 60 min at 37°C. After
washing with PBS three times, horseradish peroxidase-conjugated
goat anti-rabbit IgG monoclonal secondary antibodies (1:2,000; cat.
no. PV-6001; OriGene Technologies, Inc., Beijing, China) were added
for 2 h at 37°C. The results were visualized with chemiluminescence
substrate (Roche Diagnostics, Basel, Switzerland). Band density was
analyzed by Quantity One software (version 4.62; Bio-Rad
Laboratories, Inc., Hercules, CA, USA).
Animal study
Female C57BL/6 mice (aged 8 weeks; 25–30 g) were
purchased from Jackson Laboratory (Ben Harbor, ME, USA) and housed
in a 12 h light-dark cycle at 23±1°C with a relative humidity of
50±5%. All mice had free to access food and water. The mouse model
of gestational hypertension was established according to previous
studies (23,24). Mice with gestational hypertension
were divided into two groups and received treatment with
Candesartan (0.2 mg/kg) or with PBS as a control once daily for a
total of 7 days.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from vascular endothelial
cells by using an RNAeasy Mini kit (Qiagen GmbH, Hilden, Germany).
RNA was reversed to cDNA using a one-step reverse transcription kit
(Thermo Fisher Scientific, Inc.). Vascular endothelial growth
factor (VEGF), transforming growth factor β (TGF-β), angiopoietin-1
(Ang-1), placental growth factor (PLGF), angiotensin, Ang-IITR,
cytokine-induced neutrophil chemoattractant 1 (CINC-1),
lipopolysaccharide induced CXC chemokine (LIX), monokine, α-ADR
expression levels in vascular endothelial cells were determined by
RT-qPCR with β-actin as an endogenous control (25) (Invitrogen; Thermo Fisher
Scientific, Inc.). All forward and reverse primers were synthesized
by Invitrogen; Thermo Fisher Scientific, Inc. (Table I). The thermocycling conditions
were as follows: Initial denaturation at 94°C for 2 min, followed
by 35 cycles of 95°C for 30 sec, 54°C for 30 sec and 72°C for 10
min. The reaction volume (50 µl) contained 50 ng genomic cDNA, 200
µM dNTPs, 200 µM primers, 2.5 U Taq DNA polymerase (Thermo Fisher
Scientific, Inc.) and 2.5 U SYBR-Green (Thermo Fisher Scientific,
Inc.). Relative mRNA expression levels were calculated by the
2−ΔΔCq method (26).
The results are expressed as the n-fold way compared to
control.
 | Table I.Primer sequences used for reverse
transcription-quantitative polymerase chain reaction. |
Table I.
Primer sequences used for reverse
transcription-quantitative polymerase chain reaction.
| Gene name | Reverse primer | Forward primer |
|---|
| Angiotensin |
5′-GCAAGCGCAAGACCACTAAC-3′ |
5′-GCCGCTGTAATCCATCATGC-3′ |
| Ang-IITR |
5′-AAGAGAGCTTCCGTAAGGCG-3′ |
5′-GCATCCTCTTCAGTTACGTCC-3′ |
| VEGF |
5′-TTGCTGCTCTACCTCCAC-3′ |
5′-AATGCTTTCTCCGCTCTG-3′ |
| TGF-β |
5′-CCCCTGGAAAGGGCTCAACAC-3′ |
5′-TCCAACCCAGGTCCTTCCTAAAGTC-3′ |
| Ang-1 |
5′-GAAGGAAACCGAGCCTATTCAC-3′ |
5′-CCACAAGCATCAAACCACCA-3′ |
| PLGF |
5′-GTTCAGCCCATCCTGTGTCT-3′ |
5′-CTTCATCTTCTCCCGCAGAG-3′ |
| CINC-1 |
5′-GAAGATAGATTGCACCGATG-3′ |
5′-CATAGCCTCTCACACATTTC-3′ |
| LIX |
5′-GCATCTCATCTGTTACAGC-3′ |
5′-GCAAGCGCAAGACCACTAAC-3′ |
| Monokine |
5′-AGGCCTCCTGGGCTTCAT-3′ |
5′-GGAGTAGAAGTCCCGCAGGAT-3′ |
| α-ADR |
5′-AAACCTGTCCAACTACCTC-3′ | 5′-
TAATCCTCGTCTCCTTCC-3′ |
| β-actin |
5′-CGGAGTCAACGGATTTGGTC-3′ |
5′-AGCCTTCTCCATGGTCGTGA-3′ |
Immunohistochemical staining
Immunohistochemical staining was performed using the
avidin-biotin-peroxidase technique. Paraffin-embedded tumor tissue
sections (5 µm) were fixed with 10% paraformaldehyde for 2 h at
37°C were prepared and subsequently deparaffinized in xylene and
washed in PBS with Tween-20 (1%) three times at room temperature,
followed by epitope retrieval using Tris-EDTA buffer solution (pH
9.0; Sigma-Aldrich; Merck KGaA; cat. no. SRE0063) for 60 min at
65°C. The paraffin sections were subjected to hydrogen peroxide
(3%) for 10–15 min, and were subsequently blocked with 5% bovine
serum albumin (Sigma-Aldrich; Merck KGaA) for 10–15 min at 37°C.
Finally, the sections were incubated with rabbit anti-mouse p65
(1:1,000; cat. no. ab16502), Ikk-β (1:1,000; cat. no. ab17870),
IκBα (1:1,000; cat. no. ab16502; all Abcam) t 4°C for 12 h. All
sections were washed three times and incubated with Alexa
Fluor® 488-conjugated goat anti-rabbit IgG secondary
antibody (1:1,000; cat. no. ab150077; Abcam) for 1 h at 37°C and
were observed in six random fields of view under a fluorescence
microscope at ×40 magnification.
Enzyme-linked immunosorbent assay
(ELISA)
Plasma concentration levels of angiotensin, VCE and
α-ADR from mice with gestational hypertension were detected by
ELISA after receiving treatment with Candesartan, using
commercialized ELISA kits for ACE (cat. no. DL-ACE2-Mu), α-ADR
(cat. no. KL-EL001391SH; both Shanghai Kanglang Biotechnology Co.,
Ltd., Shanghai, China), according to the manufacturer's protocol.
The absorbance was measured at a wavelength of 450 nm on an ELISA
plate reader.
Analysis of Th1/Th2
Plasma concentration levels Th1/Th2 were measured
using Th1/Th2 kit (LHC0015M, Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol.
Activity of NF-κB
Activity of NF-κB in vascular endothelial cells from
healthy mice and mice with gestational hypertension was analyzed
after receiving treatment with Candesartan. The activity of NF-κB
was determined with the NF-κB p65 Transcription Factor Assay Kit
(cat. no. 10007889; Shenzen Xinbo Sheng Biological Technology Co.,
Ltd., Shenzen, China) according to the manufacturer's
instructions.
Statistical analysis
All data are presented as mean ± standard error of
three replicates within the same experiment and performed using
GraphPad Prism software 5.0 (GraphPad Software, Inc., La Jolla, CA,
USA). Statistical differences between experimental groups were
analyzed by Student's t-test or a one-way analysis of variance
followed by Tukey's post-hoc test. P<0.05 was considered to
indicate a statistically significant difference.
Results
Effects of Candesartan treatment on
expression of Ang-IITR in vascular endothelial cells
To assess the impact of Candesartan on angiotensin
release and Ang-IITR expression in a gestational hypertension model
system, isolated endothelial cells were incubated with Candesartan.
Angiotensin expression levels were upregulated in control vascular
endothelial cells, whereas Candesartan significantly inhibited
angiotensin release compared with control mice; there was no
significant compared with healthy mice (Fig. 1A). In vitro assays indicated
that homocysteine also induced angiotensin release compared with
the control group, whereas Candesartan treatment inhibited
angiotensin release compared with the control group (Fig. 1B). In addition, Ang-IITR expression
levels (Fig. 1C) and angiotensin
plasma concentration levels (Fig.
1D) were upregulated in control group compared with the healthy
group and were downregulated after Candesartan treatment in
vivo compared with the control group. Furthermore, we analyzed
expression levels of hypertension-related proteins. As demonstrated
in Fig. 1E-H, VEGF, TGF-β, Ang-1
and PLGF expression levels were upregulated by Candesartan compared
with the control group. Taken together, these results suggested
that Candesartan treatment may be beneficial for the downregulation
of angiotensin release and Ang-IITR expression in vascular
endothelial cells and mice with gestational hypertension induced by
homocysteine.
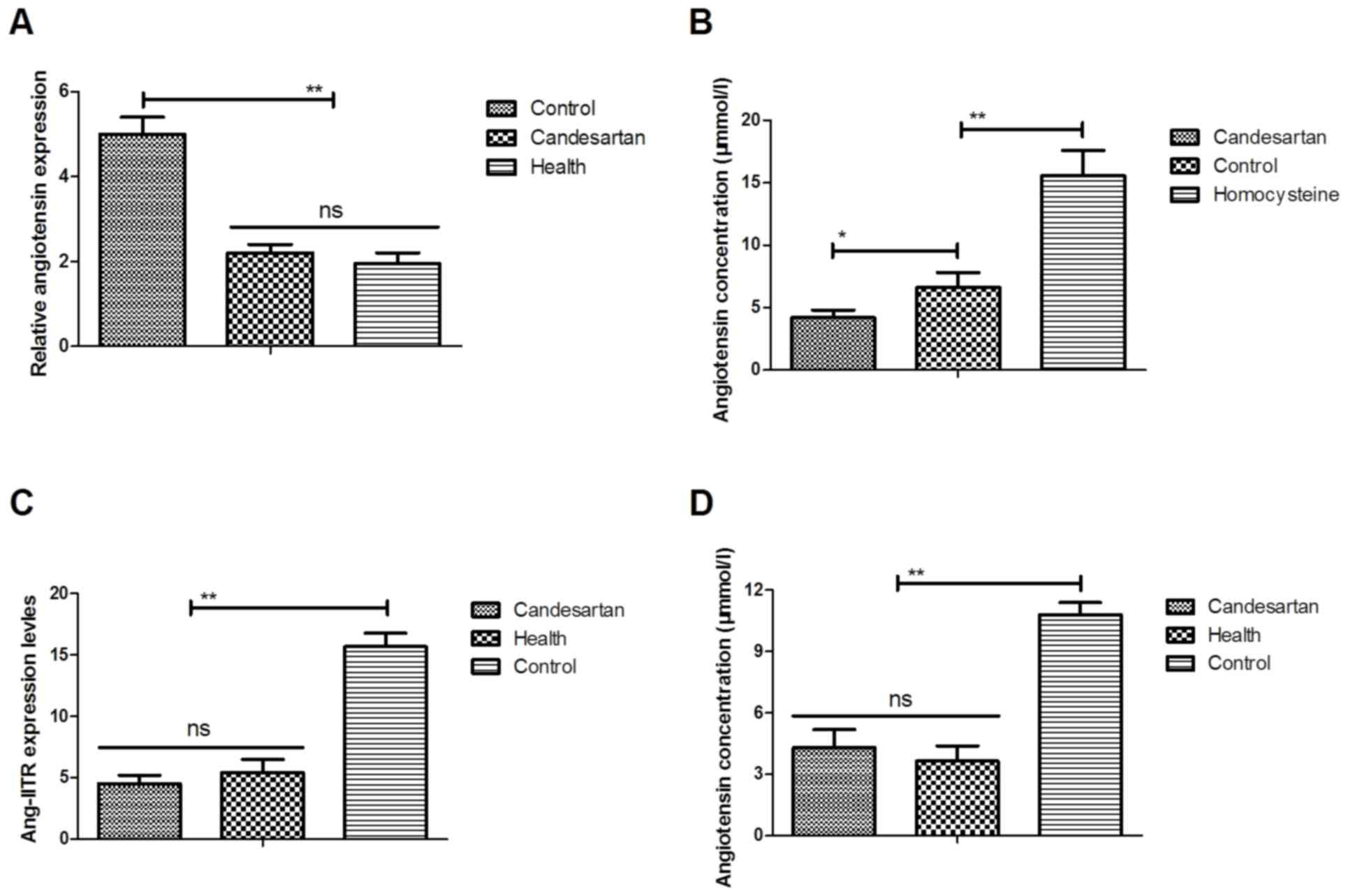 | Figure 1.Effects of Candesartan on Ang-IITR
and expression levels of hypertension-related proteins in vascular
endothelial cells. (A) Angiotensin expression levels in control,
healthy or Candesartan-treated vascular endothelial cells. (B)
Angiotensin release in vascular endothelial cells after treatment
with homocysteine or Candesartan. (C) Ang-IITR expression levels
and (D) angiotensin plasma concentration levels in control or
Candesartan-treated mice with gestational hypertension. Analysis of
(E) VEGF, (F) TGF-β, (G) Ang-1 and (H) PLGF expression levels in
control, healthy or Candesartan-treated vascular endothelial cells.
The data are presented as the mean ± standard error. *P<0.05 and
**P<0.01 vs. control. ns, no significant difference; VEGF,
vascular endothelial growth factor; TGF-β, transforming growth
factor-β; Ang-1, angiopoietin-1; PLGF, placental growth factor;
Ang-IITR, angiotensin II type 1 receptor. |
Candesartan treatment markedly
improves sFlt-1, insulin resistance, and cardiovascular risk
factors
Gestational hypertension is characterized by
widespread endothelial cell dysfunction, the progressive elevation
of insulin resistance and higher expression of cardiovascular risk
factors (27). Western blot
analyses of sFlt-1 revealed that Candesartan treatment
significantly decreased sFlt-1 expression levels compared with
control vascular endothelial cells (Fig. 2A). Insulin resistance of mice was
significantly improved in mice with gestational hypertension after
treatment with Candesartan compared with the control group
(Fig. 2B). In addition, renal
prorenin receptor expression levels were also downregulated by
Candesartan compared with the control group (Fig. 2C). Furthermore, cardiovascular risk
factors including inflammation and oxidative stress were studied in
the vascular endothelial cells after treatment with Candesartan. As
demonstrated in Fig. 2D-F, tumor
necrosis factor-α and interleukin-2 expression and the balance of
Th1/Th2 was downregulated by Candesartan compared with the control
group, in the serum of mice with gestational hypertension. Western
blot analysis of oxidative stress demonstrated that expression
levels of SOD (Fig. 1G) and ROS
(Fig. 1H) were downregulated by
Candesartan compared with control vascular endothelial cells. Taken
together, these results suggested that Candesartan treatment
markedly improved sFlt-1, insulin resistance and cardiovascular
risk factors.
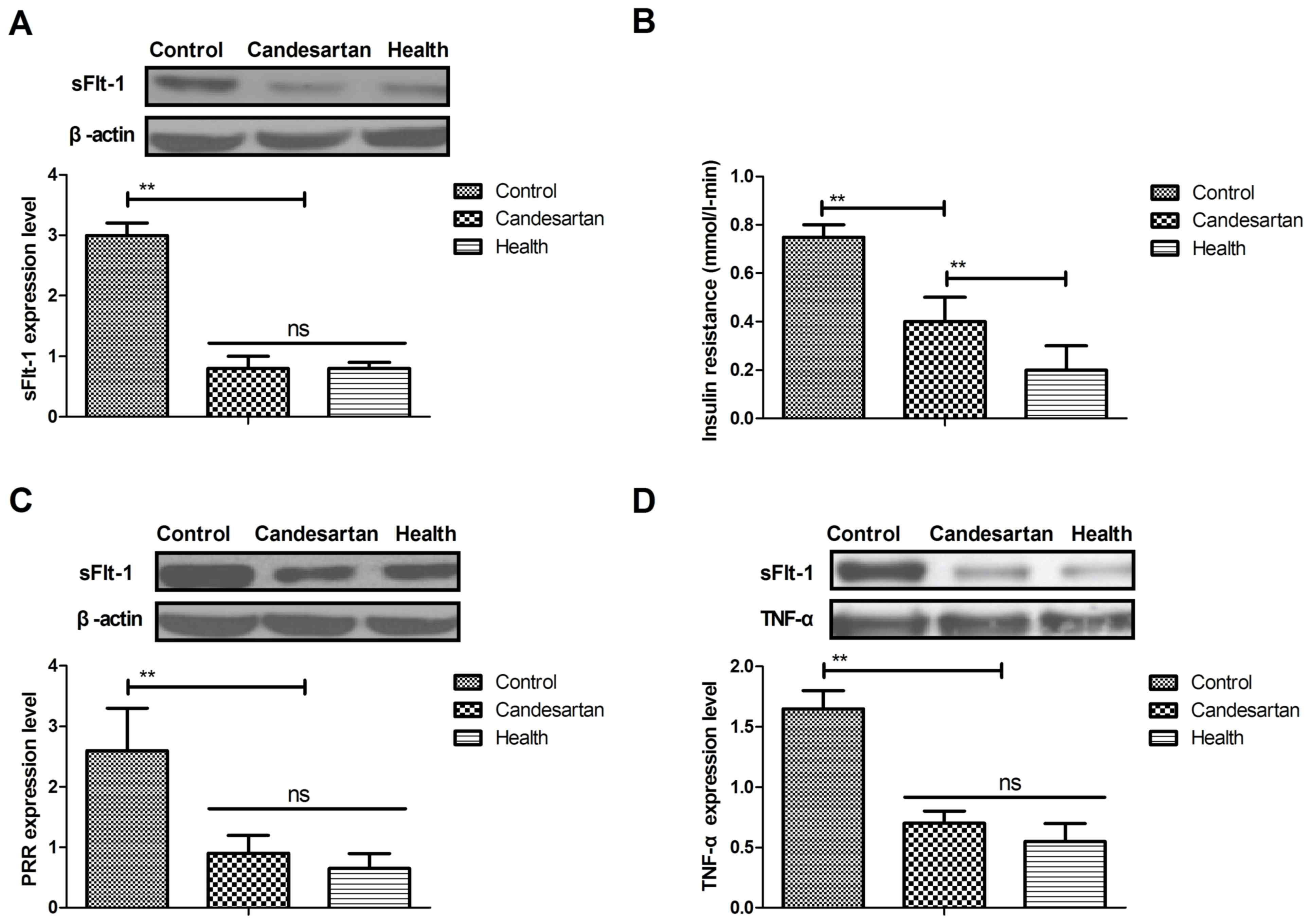 | Figure 2.Analysis of sFlt-1, insulin
resistance and cardiovascular risk factors in mice with gestational
hypertension after treatment with Candesartan. (A) Expression
levels of sFlt-1 in control, healthy or Candesartan-treated
vascular endothelial cells. (B) Insulin resistance of control,
healthy or Candesartan-treated mice with gestational hypertension.
(C) Expression levels of PRR in control, healthy or
Candesartan-treated vascular endothelial cells. Expression levels
of (D) TNF-α and (E) IL-2, and (F) balance of Th1/Th2 in the serum
of control, healthy or Candesartan-treated mice with gestational
hypertension. Expression levels of (G) SOD and (H) ROS in control,
healthy or Candesartan-treated vascular endothelial cells. The data
are presented as the mean ± standard error. **P<0.01 vs.
control. ns, no significant difference. sFlt-1, soluble fms-like
tyrosine kinase 1; PRR, prorenin receptor; TNF-α, tumor necrosis
factor-α; IL-2, interleukin-2; Th1, T-helper cell class 1; Th2,
T-helper cell class 2; SOD, superoxide dismutase; ROS, reactive
oxygen species. |
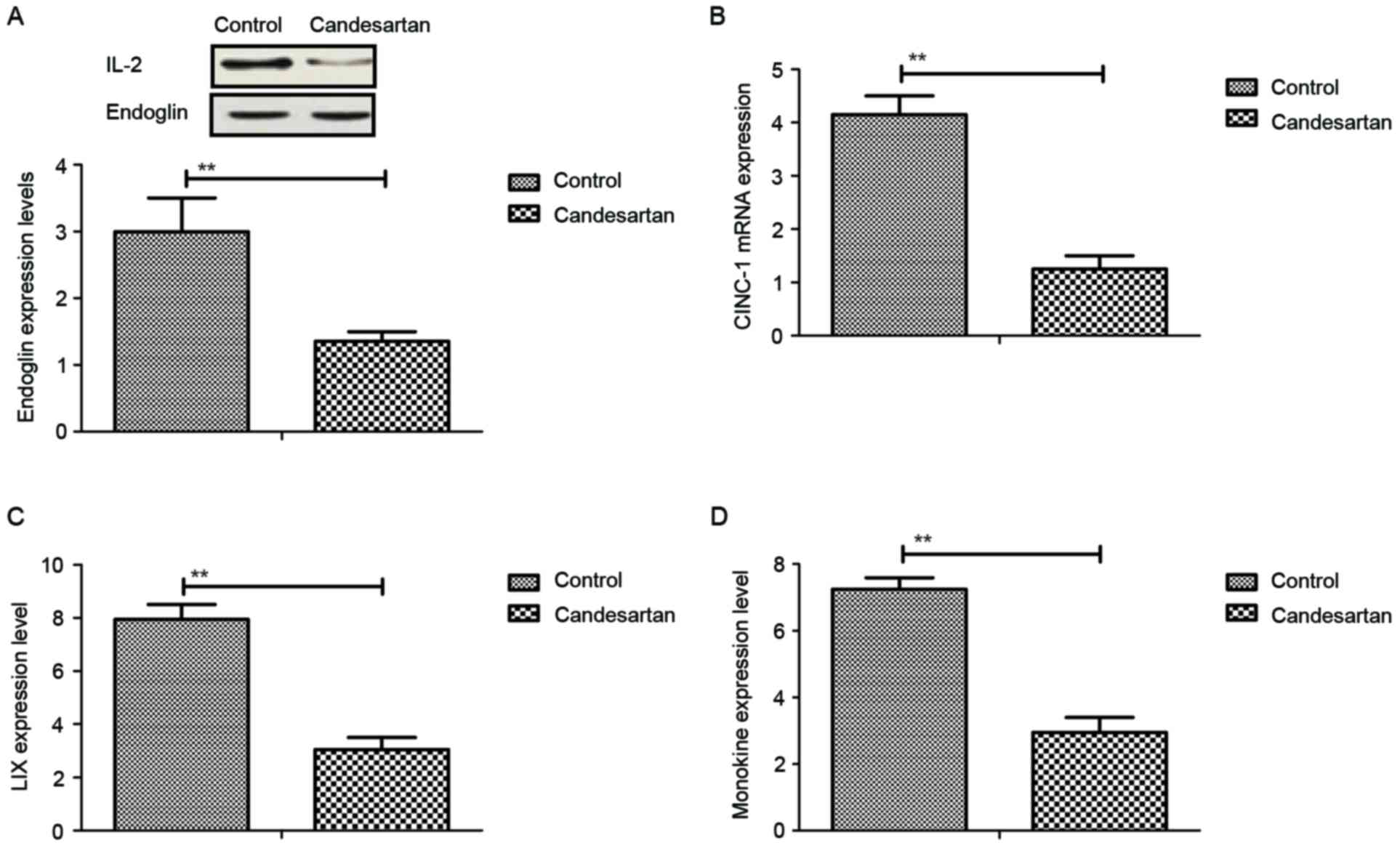
Candesartan regulates soluble endoglin
via the NF-κB signaling pathway
To analyze the mechanism of Candesartan-mediated
benefits for gestational hypertension, the NF-κB signaling pathway
was investigated. Soluble endoglin expression was analyzed in
vascular endothelial cells. As demonstrated in Fig. 3A, endoglin expression was
downregulated by Candesartan compared with the control. Cytokine
mRNA expressions levels of CINC-1 (Fig. 3B), LIX (Fig. 3C) and monokine (Fig. 3D) were decreased after treatment
with Candesartan compared with the control. In addition, it was
observed that p65, IKK-β and IκBα expression levels were
upregulated by Candesartan in vascular endothelial cells, compared
with the control (Fig. 3E). NF-κB
activity was upregulated by Candesartan in vascular endothelial
cells compared with the control (Fig.
3F). Inhibition of NF-κB activity by JSH23 reversed
Candesartan-mediated reduction of sFlt-1 and endoglin expression
levels (Fig. 3G, H). Taken
together, the data suggested that Candesartan may mediate
gestational hypertension via the endoglin-mediated NF-κB signaling
pathway.
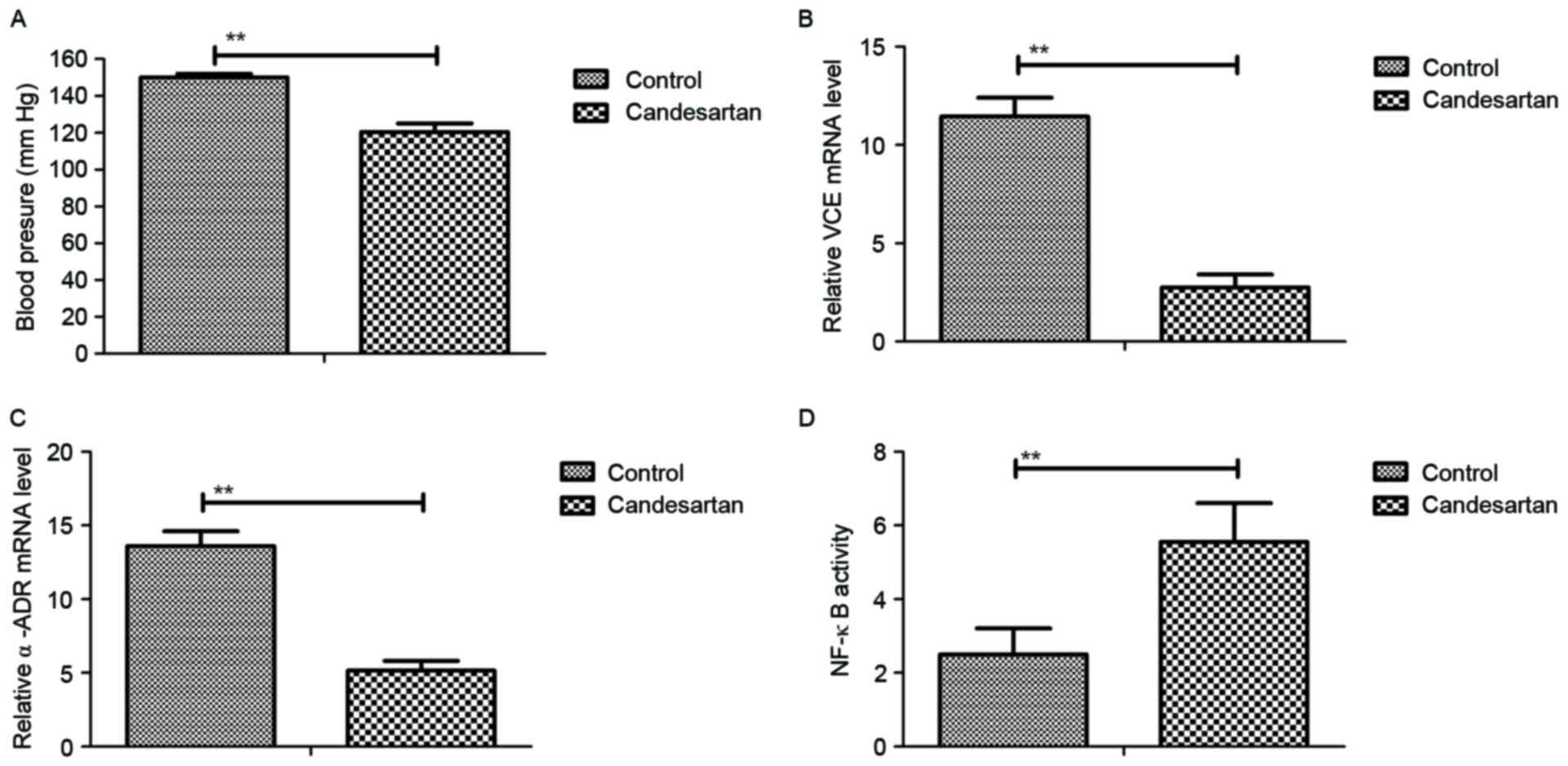
Analysis of the efficacy of
Candesartan treatment on mice with gestational hypertension induced
by homocysteine
To determine the therapeutic effects of Candesartan
for gestational hypertension, a mouse model of gestational
hypertension was established using homocysteine. As demonstrated in
Fig. 4A, hypertension was
decreased by Candesartan treatment compared with the control.
Plasma concentrations of VCE and α-ADR were also downregulated by
Candesartan treatment compared with the control, as determined by
RT-qPCR (Fig. 4B and C).
Candesartan treatment also upregulated NF-κB activation and nuclear
translocation in vivo (Fig. 4D
and E). Immunostaining assays revealed that p65, IKK-β and IκBα
expression levels in vascular endothelial cells were increased
after 14-day treatment with Candesartan (Fig. 4F). In addition, ELISA assays
demonstrated that Candesartan treatment significantly inhibited
aldosterone (Fig. 4G) and renin
(Fig. 4H) release in serum in mice
with gestational hypertension. Taken together, these results
suggested that Candesartan treatment may be beneficial for the
treatment of gestational hypertension induced by homocysteine.
Discussion
Reports have indicated angiotensin release and
Ang-IITR expression serve a crucial role in the initiation and
development of gestational hypertension, which respectively target
angiotensin and α-ADR (28,29).
Research also suggests that VCE and α-ADR are potential targets for
the treatment of gestational hypertension (30). The underlying mechanism of
formation of gestational arterial hypertension for women during
pregnancy have been investigated and results have indicated that
Ang-IITR contributes to the progression of gestational hypertension
(31). Therefore, Ang-IITR may be
a potential target and studies have revealed the efficacy of an
Ang-IITR antagonist for hypertension in the clinic (32–34).
In the present study, the therapeutic effects of Candesartan (an
Ang-IITR antagonist) was investigated in pregnant mice with
hypertension, which was induced by homocysteine. Although various
previous studies have investigated the efficacy of Candesartan for
the treatment of hypertension, the therapeutic effects of
Candesartan for gestational hypertension is infrequently reported
(35,36).
Candesartan is in a class of drugs called
angiotensin II receptor antagonists. Candesartan prevents the
constriction (narrowing) of veins and arteries and has been used in
the treatment of essential hypertension (37). Currently, the classical RAS has
been studied extensively for decades and has yielded numerous
effective therapies for gestational hypertension and gestational
hypertension-induced complications (19,38).
Research has indicated that Ang-IITR blockers are antihypertensive
drugs that may be an efficient way to improve the renin-angiotensin
system by binding to Ang-IITR, leading to improvement of RAS,
gestational hypertension and gestational hypertension-associated
target organ damage in hypertensive animal models (39–41).
Therefore, the present study determined whether Candesartan may
have therapeutic effects on gestational hypertension. Candesartan
treatment markedly improved gestational hypertension in a mouse
model of gestational hypertension induced by homocysteine. The
findings of the present study have suggested that Candesartan
improves gestational hypertension via the NF-κB signaling
pathway.
Previous studies revealed that the NF-κB signaling
pathway may be involved in the biochemical metabolism of vascular
endothelial cells and progression of gestational hypertension
(42,43). Henke et al (44) demonstrated that vascular
endothelial cell-specific NF-κB inhibition not only attenuates
hypertension, but may also improve hypertension-induced renal
damage. Alonso et al (45)
revealed that an angiotensin II- and NF-κB-dependent mechanism may
increase connexin 43 in murine arteries by targeting of
renin-dependent hypertension. In addition, the NF-κB signaling
pathway may be involved in obesity-induced hypertension (46). The NF-κB pathway is also involved
in C-reactive protein-induced effects on pulmonary arterial
endothelial cells in chronic thromboembolic pulmonary hypertension.
Furthermore, the effect of antihypertensive drugs on the expression
of NF-κB in monocrotaline-induced pulmonary arterial hypertension
of rats have been investigated by Li et al (47). These reports suggested that the
NF-κB signaling pathway may be associated with the initiation and
progression of hypertension.
The results of the present study suggested that the
NF-κB signaling pathway influences Candesartan-mediated soluble
endoglin and cytokine (CINC-1, LIX and monokine) expressions levels
in vascular endothelial cells. Blocking of the NF-κB signaling
pathway suppressed Candesartan-mediated improvement of sFlt-1 and
endoglin expression levels. sFlt-1 and endoglin are two important
predictive biomarkers of gestational hypertension (48,49).
Alterations in sFlt-1 and endoglin expression levels may predict
the progression of gestational hypertension (50,51).
The results of the present study suggested that sFlt-1 and endoglin
expression levels are altered after Candesartan treatment. Notably,
gestational hypertension is markedly decreased after Candesartan
treatment once daily for a total of 14 days.
To conclude, the present study investigated the
clinical efficacy of Candesartan in mice with gestational
hypertension induced by homocysteine. Although previous studies
have reported the direct effects of Candesartan on hypertension,
the present study investigated the effects of Candesartan in
gestational hypertension and gestational hypertension-associated
factors (52,53). This study suggested that
Candesartan has a novel role in gestational hypertension management
and an increasing number of preclinical reports show promising
results (54–56). Of note, our results suggested that
the NF-κB signaling pathway is involved in Candesartan-mediated
initiation and progression of gestational hypertension. Overall,
our findings suggested that mice with gestational hypertension
treated by Candesartan exhibited beneficial effects for
inflammation, oxidative stress and hypertension. However, future
work should be performed in a larger cohort to support the data of
the present study.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The analyzed data sets generated during the study
are available from the corresponding author on reasonable
request.
Author's contributions
XZ performed the experiments. XW designed the
experiments and analyzed the data.
Ethics approval and consent to
participate
The present study was carried out in strict
accordance with the recommendations in the Guide for Shandong
Provincial Hospital affiliated to Shandong University. All surgery
and euthanasia were performed to minimize suffering. This study was
approved by the ethics committee of Shangdong University and Taian
City Central Hospital (Jinan, China).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Kennedy DA, Woodland C and Koren G: Lead
exposure, gestational hypertension and pre-eclampsia: A systematic
review of cause and effect. J Obstet Gynaecol. 32:512–517. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Huang YH, Chen YP, Liang CC, Chang YL and
Hsieh CC: Impetigo herpetiformis with gestational hypertension: A
case report and literature review. Dermatology. 222:221–224. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Razi Mohamad ZR and Schindler AE: Review
on role of progestogen (dydrogesterone) in the prevention of
gestational hypertension. Horm Mol Biol Clin Investig. 27:73–76.
2016.PubMed/NCBI
|
|
4
|
Pengo MF, Rossi GP and Steier J:
Obstructive sleep apnea, gestational hypertension and preeclampsia:
A review of the literature. Curr Opin Pulm Med. 20:588–594. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhu YC, Sun Y and Yang HX: Chronic
hypertension superimposed on preeclampsia at 13 gestational weeks:
A case report with review. Chin Med J(Engl). 125:2067–2069.
2012.PubMed/NCBI
|
|
6
|
Grandone E, Margaglione M, Colaizzo D,
Cappucci G, Sciannamé N, Montanaro S, Paladini D, Martinelli P and
Di Minno G: Prothrombotic genetic risk factors and the occurrence
of gestational hypertension with or without proteinuria. Thromb
Haemost. 81:349–352. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Vasapollo B, Novelli GP, Gagliardi G,
Tiralongo GM, Pisani I, Manfellotto D, Giannini L and Valensise H:
Medical treatment of early-onset mild gestational hypertension
reduces total peripheral vascular resistance and influences
maternal and fetal complications. Ultrasound Obstet Gynecol.
40:325–331. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Banhidy F, Acs N, Puho EH and Czeizel AE:
The efficacy of antihypertensive treatment in pregnant women with
chronic and gestational hypertension: A population-based study.
Hypertens Res. 33:460–466. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Schoenaker DA, Soedamah-Muthu SS and
Mishra GD: The association between dietary factors and gestational
hypertension and pre-eclampsia: A systematic review and
meta-analysis of observational studies. BMC Med. 12:1572014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rashid Zainul MR, Lim JF, Nawawi NH,
Luqman M, Zolkeplai MF, Rangkuty HS, Nor Mohamad NA, Tamil A, Shah
SA, Tham SW and Schindler AE: A pilot study to determine whether
progestogen supplementation using dydrogesterone during the first
trimester will reduce the incidence of gestational hypertension in
primigravidae. Gynecol Endocrinol. 30:217–220. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang YA, Chughtai AA, Farquhar CM, Pollock
W, Lui K and Sullivan EA: Increased incidence of gestational
hypertension and preeclampsia after assisted reproductive
technology treatment. Fertil Steril. 105:920–926, e922. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hua X, Zhang J, Guo Y, Shen M, Gaudet L,
Janoudi G, Walker M and Wen SW: Effect of folic acid
supplementation during pregnancy on gestational
hypertension/preeclampsia: A systematic review and meta-analysis.
Hypertens Pregnancy. 35:447–460. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Basaran A, Basaran M, Topatan B and Martin
JN Jr: Effect of chorionic villus sampling on the occurrence of
preeclampsia and gestational hypertension: An updated systematic
review and meta-analysis. J Turk Ger Gynecol Assoc. 17:65–72. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chinnathambi V, More AS, Hankins GD,
Yallampalli C and Sathishkumar K: Gestational exposure to elevated
testosterone levels induces hypertension via heightened vascular
angiotensin II type 1 receptor signaling in rats. Biol Reprod.
91:62014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Seremak-Mrozikiewicz A, Drews K, Chmara E,
Mrozikiewicz PM and Slomko Z: Gestational hypertension (GH) and
a1166c polymorphism of angiotensin II type 1 receptor. Ginekol Pol.
71:783–788. 2000.(In Polish). PubMed/NCBI
|
|
16
|
Chen PM, Lai TS, Chen PY, Lai CF, Wu V,
Chiang WC, Chen YM, Wu KD and Tsai TJ: Renoprotective effect of
combining pentoxifylline with angiotensin-converting enzyme
inhibitor or angiotensin II receptor blocker in advanced chronic
kidney disease. J Formos Med Assoc. 113:219–226. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mavrakanas TA, Gariani K and Martin PY:
Mineralocorticoid receptor blockade in addition to angiotensin
converting enzyme inhibitor or angiotensin II receptor blocker
treatment: An emerging paradigm in diabetic nephropathy: A
systematic review. Eur J Intern Med. 25:173–176. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tsutamoto T, Nishiyama K, Yamaji M,
Kawahara C, Fujii M, Yamamoto T and Horie M: Comparison of the
long-term effects of candesartan and olmesartan on plasma
angiotensin II and left ventricular mass index in patients with
hypertension. Hypertens Res. 33:118–122. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kjeldsen SE, Stålhammar J, Hasvold P,
Bodegard J, Olsson U and Russell D: Effects of losartan vs
candesartan in reducing cardiovascular events in the primary
treatment of hypertension. J Hum Hypertens. 24:263–273. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fogari R, Zoppi A, Salvadeo SA, Mugellini
A, Lazzari P, Santoro T and Derosa G: Fibrinolysis and insulin
sensitivity in imidapril and candesartan (FISIC study) recipients
with hypertension. Hypertens Res. 34:509–515. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nishida Y, Takahashi Y, Nakayama T, Soma
M, Kitamura N and Asai S: Effect of candesartan monotherapy on
lipid metabolism in patients with hypertension: A retrospective
longitudinal survey using data from electronic medical records.
Cardiovasc Diabetol. 9:382010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wai-Hoe L, Wing-Seng L, Ismail Z and
Lay-Harn G: SDS-PAGE-Based quantitative assay for screening of
kidney stone disease. Biol Proced Online. 11:145–160. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Armstrong DW, Tse MY, O'Tierney-Ginn PF,
Wong PG, Ventura NM, Janzen-Pang JJ, Matangi MF, Johri AM, Croy BA,
Adams MA and Pang SC: Gestational hypertension in atrial
natriuretic peptide knockout mice and the developmental origins of
salt-sensitivity and cardiac hypertrophy. Regul Pept. 186:108–115.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Partyka R, Chmiel B, Sikora J, Grabowska
T, Wróbel B, Praisner A, Pióro A, Jałowieicki P and Kokocinska D:
Lipofuscin, homocysteine and tissue polypeptide specific antigen in
gestational hypertension. Neuro Endocrinol Lett. 28:311–314.
2007.PubMed/NCBI
|
|
25
|
Xiao S, Wang J and Xiao N: MicroRNAs as
noninvasive biomarkers in bladder cancer detection: A diagnostic
meta-analysis based on qRT-PCR data. Int J Biol Markers.
31:e276–e285. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yang L, Zhang W, Zhang L, Zhang S, Yang Y,
Wang Q, Shao J, Chen G and Wang Y: Gestational hypertension risk
evaluation based on epidemiological, biochemical, and hemodynamic
factors. Clin Exp Obstet Gynecol. 40:61–65. 2013.PubMed/NCBI
|
|
28
|
Van Guilder GP, Pretorius M, Luther JM,
Byrd JB, Hill K, Gainer JV and Brown NJ: Bradykinin type 2 receptor
BE1 genotype influences bradykinin-dependent vasodilation during
angiotensin-converting enzyme inhibition. Hypertension. 51:454–459.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Iwatsubo K and Umemura S: Alpha adrenergic
receptor blockers for patients with hypertension. Nihon Rinsho. 64
Suppl 6:S294–S299. 2006.(In Japanese).
|
|
30
|
Mancia G: Introduction to a compendium on
hypertension. Circ Res. 116:923–924. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Davidovich IM, Bloshchinskaia IA and
Petrichko TA: Gestational arterial hypertension. Mechanisms of
formation. Treatment with normodipin. Ter Arkh. 75:50–54. 2003.(In
Russian). PubMed/NCBI
|
|
32
|
Kong J and Li YC: Effect of ANG II type I
receptor antagonist and ACE inhibitor on vitamin D receptor-null
mice. Am J Physiol Regul Integr Comp Physiol. 285:R255–R261. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hagiwara S, Iwasaka H, Hidaka S, Hasegawa
A, Koga H and Noguchi T: Antagonist of the type-1 ANG II receptor
prevents against LPS-induced septic shock in rats. Intensive Care
Med. 35:1471–1478. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Rakusan K, Chvojkova Z, Oliviero P,
Ostadalova I, Kolar F, Chassagne C, Samuel JL and Ostadal B: ANG II
type 1 receptor antagonist irbesartan inhibits coronary
angiogenesis stimulated by chronic intermittent hypoxia in neonatal
rats. Am J Physiol Heart Circ Physiol. 292:H1237–H1244. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yasuno S, Fujimoto A, Nakagawa Y, Kuwahara
K and Ueshima K: Fixed-dose combination therapy of candesartan
cilexetil and amlodipine besilate for the treatment of hypertension
in Japan. Expert Rev Cardiovasc Ther. 10:577–583. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Barrios V and Escobar C: Candesartan in
the treatment of hypertension: What have we learnt in the last
decade? Expert Opin Drug Saf. 10:957–968. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kwon BJ, Jang SW, Choi KY, Kim DB, Cho EJ,
Ihm SH, Youn HJ and Kim JH: Comparison of the efficacy between
hydrochlorothiazide and chlorthalidone on central aortic pressure
when added on to candesartan in treatment-naive patients of
hypertension. Hypertens Res. 36:79–84. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Henriksson M, Russell D, Bodegard J,
Kjeldsen S, Hasvold P, Stålhammar J and Levin LÅ: Health-care costs
of losartan and candesartan in the primary treatment of
hypertension. J Hum Hypertens. 25:130–136. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Moltzer E, Raso Mattace FU, Karamermer Y,
Boersma E, Webb GD, Simoons ML, Danser AH, van den Meiracker AH and
Roos-Hesselink JW: Comparison of candesartan versus metoprolol for
treatment of systemic hypertension after repaired aortic
coarctation. Am J Cardiol. 105:217–222. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Meredith PA, Murray LS and McInnes GT:
Comparison of the efficacy of candesartan and losartan: A
meta-analysis of trials in the treatment of hypertension. J Hum
Hypertens. 24:525–531. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ogihara T, Nakao K, Fukui T, Fukiyama K,
Fujimoto A, Ueshima K, Oba K, Shimamoto K, Matsuoka H and Saruta T:
CASE-J Trial Group: The optimal target blood pressure for
antihypertensive treatment in Japanese elderly patients with
high-risk hypertension: A subanalysis of the candesartan
antihypertensive survival evaluation in Japan (CASE-J) trial.
Hypertens Res. 31:1595–1601. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Gu JW, Tian N, Shparago M, Tan W, Bailey
AP and Manning RD Jr: Renal NF-kappaB activation and TNF-alpha
upregulation correlate with salt-sensitive hypertension in Dahl
salt-sensitive rats. Am J Physiol Regul Integr Comp Physiol.
291:R1817–R1824. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Ortiz LA, Champion HC, Lasky JA, Gambelli
F, Gozal E, Hoyle GW, Beasley MB, Hyman AL, Friedman M and Kadowitz
PJ: Enalapril protects mice from pulmonary hypertension by
inhibiting TNF-mediated activation of NF-kappaB and AP-1. Am J
Physiol Lung Cell Mol Physiol. 282:L1209–L1221. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Henke N, Schmidt-Ullrich R, Dechend R,
Park JK, Qadri F, Wellner M, Obst M, Gross V, Dietz R, Luft FC, et
al: Vascular endothelial cell-specific NF-kappaB suppression
attenuates hypertension-induced renal damage. Circ Res.
101:268–276. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Alonso F, Krattinger N, Mazzolai L, Simon
A, Waeber G, Meda P and Haefliger JA: An angiotensin II- and
NF-kappaB-dependent mechanism increases connexin 43 in murine
arteries targeted by renin-dependent hypertension. Cardiovasc Res.
87:166–176. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Purkayastha S, Zhang G and Cai D:
Uncoupling the mechanisms of obesity and hypertension by targeting
hypothalamic IKK-β and NFκB. Nat Med. 17:883–887. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Li XW, Guo B, Shen YY and Yang JR: Effect
of chrysin on expression of NOX4 and NF-kappaB in right ventricle
of monocrotaline-induced pulmonary arterial hypertension of rats.
Yao Xue Xue Bao. 50:1128–1134. 2015.(In Chinese). PubMed/NCBI
|
|
48
|
Carney EF: Hypertension: sFlt-1 removal
seems to be beneficial in women with pre-eclampsia. Nat Rev
Nephrol. 11:6902015. View Article : Google Scholar
|
|
49
|
Valbuena-Diez AC, Blanco FJ, Oujo B, Langa
C, Gonzalez-Nuñez M, Llano E, Pendas AM, Díaz M, Castrillo A,
Lopez-Novoa JM and Bernabeu C: Oxysterol-induced soluble endoglin
release and its involvement in hypertension. Circulation.
126:2612–2624. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Daly AL, Robertson A, Johnson P, Middleton
S, Bobek G, Sullivan C and Hennessy A: PP162. sFlt-1 controlled by
CPAP in a pregnant patient with chronic hypertension. Pregnancy
Hypertens. 2:3272012. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Gluchowska M, Kowalska-Koprek U and
Karowicz-Bilińska A: Evaluation of the usefulness of endoglin level
as a predictor of preeclampsia in pregnant women with hypertension.
Ginekol Pol. 84:835–840. 2013.(In Polish). View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Muda P, Kampus P, Teesalu R, Zilmer K,
Ristimäe T, Fischer K and Zilmer M: Effects of amlodipine and
candesartan on oxidized LDL level in patients with mild to moderate
essential hypertension. Blood Press. 15:313–318. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Ikeda H, Inoue T, Uemura S, Kaibara R,
Tanaka H and Node K: Effects of candesartan for middle-aged and
elderly women with hypertension and menopausal-like symptoms.
Hypertens Res. 29:1007–1012. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Nzelu D, Dumitrascu-Biris D, Hunt KF,
Cordina M and Kametas NA: Pregnancy outcomes in women with previous
gestational hypertension: A cohort study to guide counselling and
management. Pregnancy Hypertens. 2017. View Article : Google Scholar
|
|
55
|
Pauli JM, Lauring JR, Stetter CM, Repke
JT, Botti JJ, Ural SH and Ambrose A: Management of gestational
hypertension-the impact of HYPITATa. J Perinat Med. 41:415–420.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Sibai BM: Management of late preterm and
early-term pregnancies complicated by mild gestational
hypertension/pre-eclampsia. Semin Perinatol. 35:292–296. 2011.
View Article : Google Scholar : PubMed/NCBI
|