Introduction
Hepatocellular carcinoma (HCC) has been ranked the
third most life-threatening cancer, causing approximately 700,000
mortalities each year globally (1). For patients with HCC at Barcelona
Clinic Liver Cancer (BCLC) (2,3)
stage 0 or A, surgical resection is the first-line treatment, which
contributes to 60–80% of patients surviving for 5 years (4,5).
Surgical resection is applicable in patients with HCC as the liver
has a unique capacity to regenerate, following principal tissue
loss (6). Notably, there are
numerous previous studies that have reported that in patients with
HCC beyond BCLC stage A, surgical resection was infrequently the
best choice and improved the rate of survival (2,7,8).
However, in order to remove the lesions thoroughly, extensive liver
resection may be required, leaving an insufficient amount of
residual tissue for the liver to recover to maintain vital function
(9). On this occasion, a transient
fatal form of hepatic failure may develop, leading to the
untreatable small-for-size syndrome (9,10).
Therefore, identifying a strategy to promote liver regeneration and
liver function recovery following partial hepatectomy (PH) is
urgently required.
Nicotinamide (Nam) is a form of vitamin
B3 that is absorbed from our daily diets (11). The therapeutic potential of Nam has
been demonstrated in a diverse range of diseases, including skin
disorders, diabetes and multiple sclerosis (12–14).
Previous studies have revealed that Nam may accelerate the repair
of human skin following injury, suggesting the potency of Nam in
promoting the regeneration of vital tissues (15,16).
It was additionally reported that Nam serves as a precursor of
oxidized nicotinamide adenine dinucleotide (NAD+) during
the metabolism of hepatocytes in liver tissue (17–20).
However, the effect of Nam on liver regeneration remains
unknown.
Sirtuins are class III histone deacetylases that
consume one molecule of NAD+ during each deacetylation
cycle (21). Accumulating evidence
has demonstrated that NAD-dependent protein deacetylase sirtuin-1
(SIRT1), a member of the Sirtuin family, regulates numerous
metabolic processes in the liver, including gluconeogenesis and
lipid synthesis, by directly deacetylating target proteins
(22,23). Furthermore, it has been
demonstrated that downregulation of SIRT1 impairs liver
regeneration (24). According to
our previous study, the expression of SIRT1 in hepatocytes was
significantly upregulated by Nam (25). Collectively, these results suggest
that Nam may promote liver regeneration through the SIRT1-mediated
pathways.
In the present study, it was demonstrated that Nam
induced the process of liver regeneration and improved liver
function through activation of SIRT1, suggesting that Nam has the
potency of becoming a regimen for promoting liver regeneration
following surgical resection.
Materials and methods
Animals and treatment
In the present study, male C57BL/6 mice (6–8 weeks
old; 15–20 g; n=30, 6 per group; Beijing HFK Bioscience Co., Ltd.,
Beijing, China) were raised in specific pathogen-free conditions
(23±1°C; relative humidity, 39–43%; 12 h light/dark cycle; free
access to food and water). Animal care and experimental protocols
were conducted in accordance with guidelines provided by the
Institutional Animal Care and Use Committee of Sichuan University
(Chengdu, China). Ethics approval was obtained from the
Institutional Ethics Committee of Sichuan University. Prior to
subjecting the mice to 70% PH, mice were anesthetized with a
subcutaneous injection of pentobarbital at a dose of 80 mg/kg. For
the study group, Nam (250 mg/kg per day) was administered 6 h prior
to PH via intraperitoneal injection and once daily subsequently for
a total of 3 days. Control animals were treated with the same
frequency and volume of PBS as Nam. All of the study mice were
sacrificed at the indicated time points (24, 36, 48 and 72 h) and
the liver samples were harvested and stored in liquid nitrogen.
Liver recovery rates were calculated as the ratio of the
regenerating liver weight to the mean liver weight prior to PH. The
serum samples (1 ml) were additionally collected from the angular
veins of the mice. Blood glucose (Glu), triglyceride (TG),
aspartate aminotransferase (AST) and alanine aminotransferase (ALT)
were measured using enzymatic methods (ADVIA 165; Siemens
Healthineers, Erlangen, Germany).
Protein isolation and western
blotting
To obtain the total protein, the liver tissues were
frozen in liquid nitrogen, washed twice with PBS, and homogenized
in radioimmunoprecipitation assay lysis buffer (Pierce; Thermo
Fisher Scientific, Inc., Waltham, MA, USA). The samples were
incubated on ice for 30 min prior to centrifugation at 4°C for 15
min at a speed of 12,000 × g. Subsequent to centrifugation, the
supernatant was divided into small fractions and stored at −80°C.
The protein content of each supernatant was quantified using the
Bradford assay (Bio-Rad Laboratories, Inc., Hercules, CA, USA),
according to the manufacturer's protocol. Western blot analysis was
performed as described previously (26). Cyclin D1 (1:1,000; rabbit
monoclonal; cat. no. ab134175), PCNA (1:1,000; mouse monoclonal;
cat. no. ab29), and SIRT1 (1:1,000; mouse monoclonal; cat. no.
ab110304) primary antibodies were obtained from Abcam (Cambridge,
UK); β-actin (1:1,000; mouse monoclonal; cat. no. sc-69879), as
well as the secondary antibodies [horseradish peroxidase
(HRP)-conjugated goat anti-mouse and goat anti-rabbit; both
1:5,000; cat. no. sc-2005 and sc-2004] were obtained from Santa
Cruz Biotechnology, Inc. (Dallas, TX, USA). Densitometric analysis
was performed using ImageJ software (version 1.48; National
Institutes of health, Bethesda, MD, USA).
Histology and
immunohistochemistry
For immunohistochemical analysis and morphological
examination [hematoxylin and eosin (H&E) staining],
formalin-fixed (10% buffered formalin at 4°C overnight), paraffin
embedded liver tissues were sectioned to 5 µm. H&E staining was
performed as described previously (27).
For immunohistochemical analysis, sections were
blocked with 5% bovine serum albumin (Biowest, Nuaillé, France)
diluted in PBS (pH 7.4) for 1 h at 37°C. Proliferation marker
protein Ki-67 (Ki-67) staining was performed with an anti-Ki-67
antibody (1:4,000; rabbit polyclonal; cat. no. ab15580; Abcam) at
4°C overnight. Next, slides were incubated with the HRP-conjugated
goat anti-rabbit antibody at 37°C for 1 h. Liver sections examined
at each time-point were from at least three individual animals in
the treatment and control groups. Images were captured with the
AX10 imager A2 fluorescence microscope (magnification, ×40; Carl
Zeiss AG, Oberkochen, Germany). Percentage Ki-67 expression was
evaluated in five random fields from each immunostained HCC
section. Quantification of Ki-67 staining was performed using
Image-Pro Plus version 6.0 (National Institutes of Health,
Bethesda, MD, USA).
Bioinformatics analysis
The Kyoto Encyclopedia of Genes and Genomes database
(https://www.kegg.jp) was used to explore the
potential genes regulated by SIRT1.
Acetylation assays
To detect the deacetylation of peroxisome
proliferator-activated receptor g coactivator 1-α (PGC-1α) and
oxysterols receptor liver X receptor-α (LXRα) by SIRT1, lysine
acetylation was analyzed by immunoprecipitation and western
blotting, using an acetyl-lysine antibody. Primary antibodies
against PGC-1α, (1:1,000; rabbit polyclonal; cat. no. ab54481) LXRα
(1:1,000; mouse monoclonal; cat. no. ab41902) and acetyl-lysine
(1:1,000; rabbit polyclonal; cat. no. ab21623) were purchased from
Abcam. HRP-conjugated goat anti-mouse and goat anti-rabbit
secondary antibodies were used, as described above, and the western
blotting procedure was the same as aforementioned (26). An immunoprecipitation assay was
performed as previously described (28,29)
using the Pierce™ Classic Immunoprecipitation kit
(Thermo Fisher Scientific, Inc.), according to the manufacturer's
protocol.
Statistical analysis
Statistical analysis was performed using GraphPad
Prism 6 (GraphPad Software, Inc., La Jolla, CA, USA). A Student's
t-test was performed to compare continuous values between the
treatment and control groups. Results are presented as the mean ±
standard deviation of three independent experiments. P<0.05 was
considered to indicate a statistically significant difference.
Results
Nam promotes liver proliferation
following PH
To investigate the effect of Nam on the process of
liver regeneration, 70% PH was conducted on the treatment and
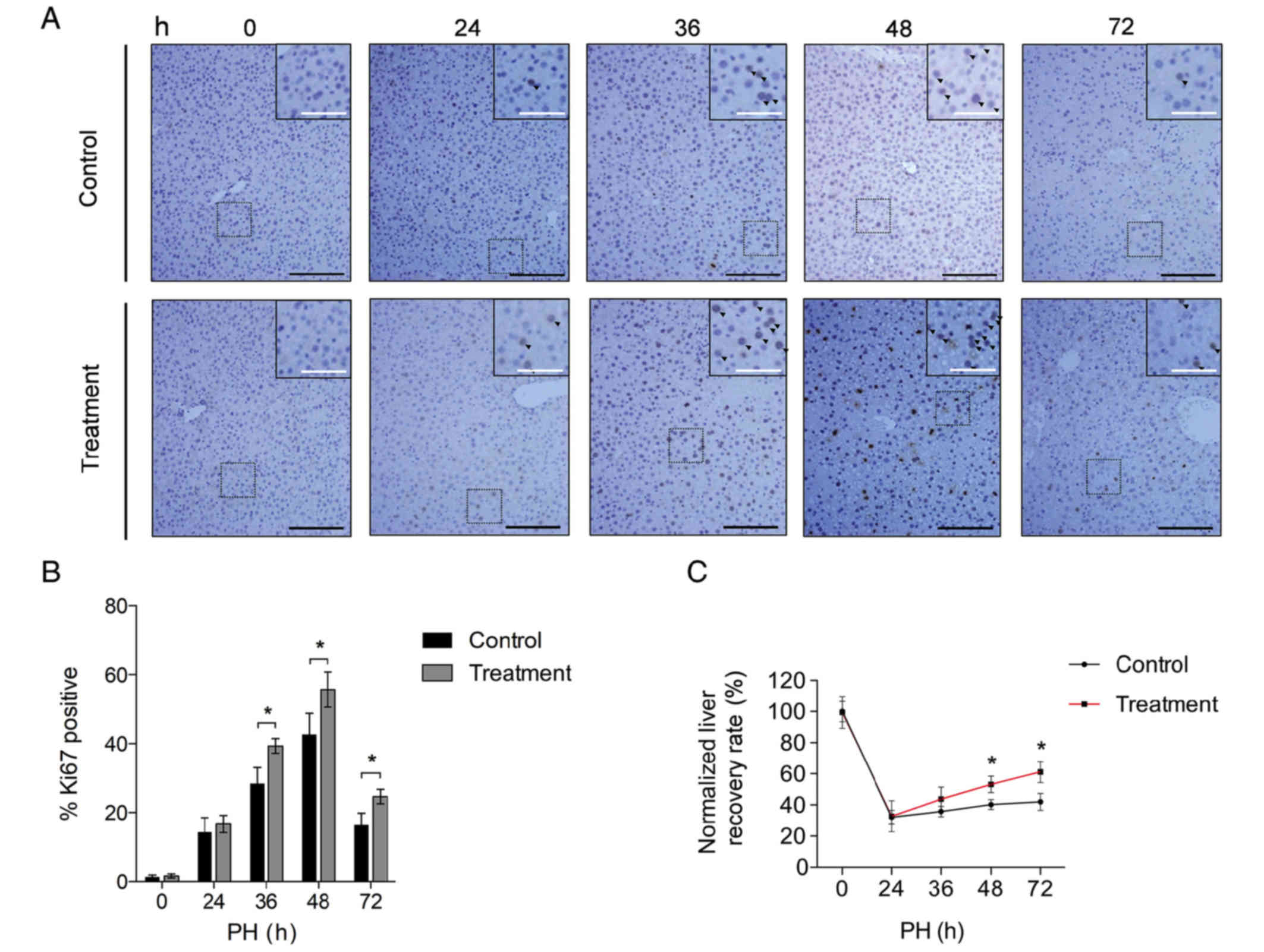
control groups. As demonstrated in Fig. 1A, the Ki-67 immunohistochemistry
staining was performed to validate hepatic cell proliferation
following PH. It was observed that when compared with the control
group, increased active proliferation was observed in the treatment
group at 24–72 h post-operation. Furthermore, quantification of the
Ki-67 staining revealed a significant restoration of hepatocyte
proliferation in the treatment group 36–72 h following surgery
(Fig. 1B; P<0.05). The
treatment group additionally exhibited higher liver recovery rates
compared with the control group (Fig.
1C). These results suggested that the proliferation of primary
hepatocytes was enhanced in the presence of Nam.
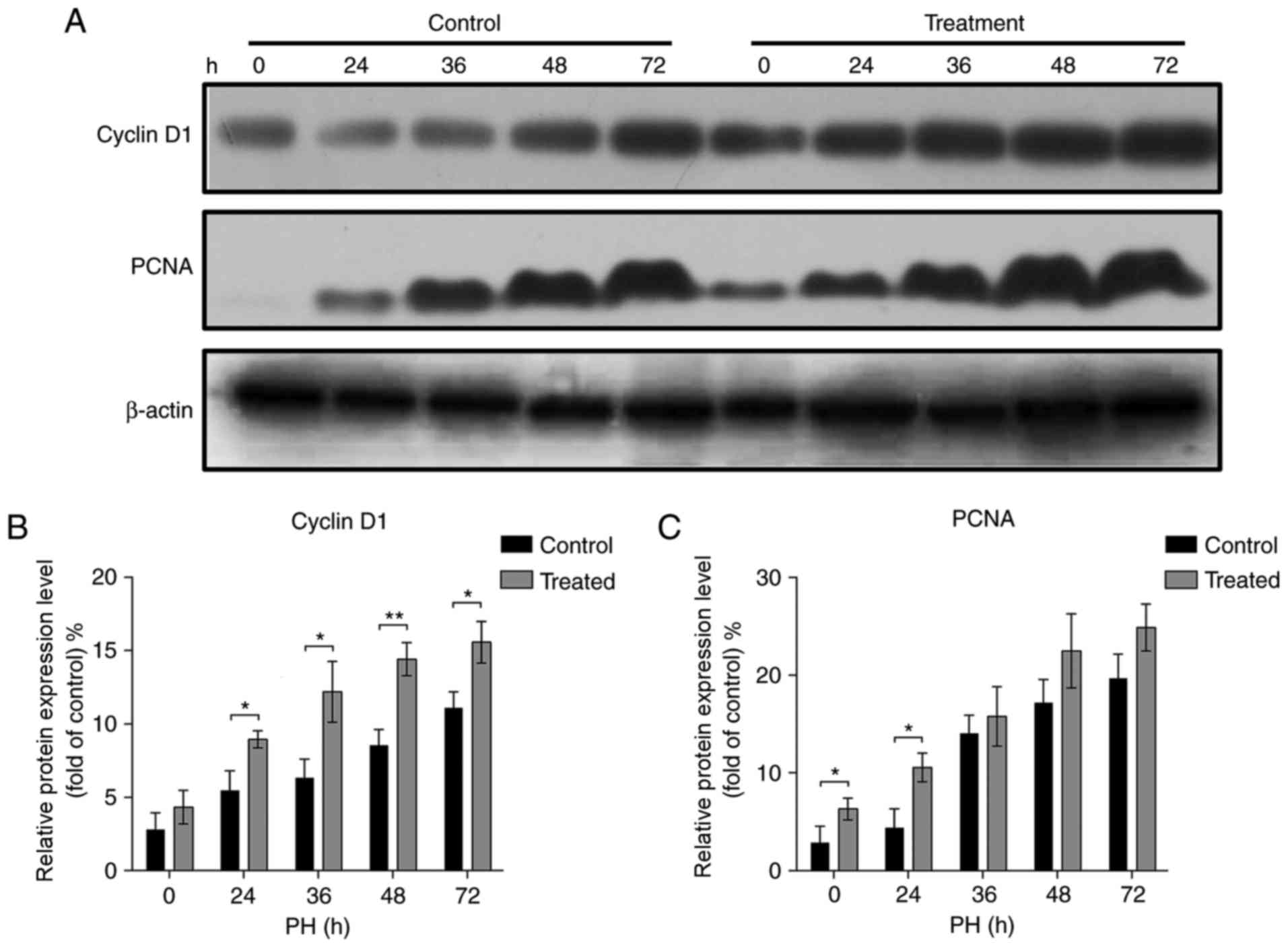
Cell cycle progression serves a vital role during
liver regeneration (30). To
examine the cell cycle of proliferating hepatocytes following PH,
the expression of G1/S-specific cyclin D1 and
proliferating cell nuclear antigen (PCNA), two key regulators of
the cell cycle, was analyzed by western blotting (Fig. 2A). It was identified that the
expression levels of cyclin D1 and PCNA reached a peak at 72 h in
the livers of mice from the treatment and control groups.
Additionally, the livers of mice injected with Nam exhibited
significantly increased expression of cyclin D1 at 24, 36, 48 and
72 h and PCNA at 0 and 24 h when compared with the control
(Fig. 2B and C; P<0.05). These
results suggested that Nam may accelerate liver regeneration
following PH.
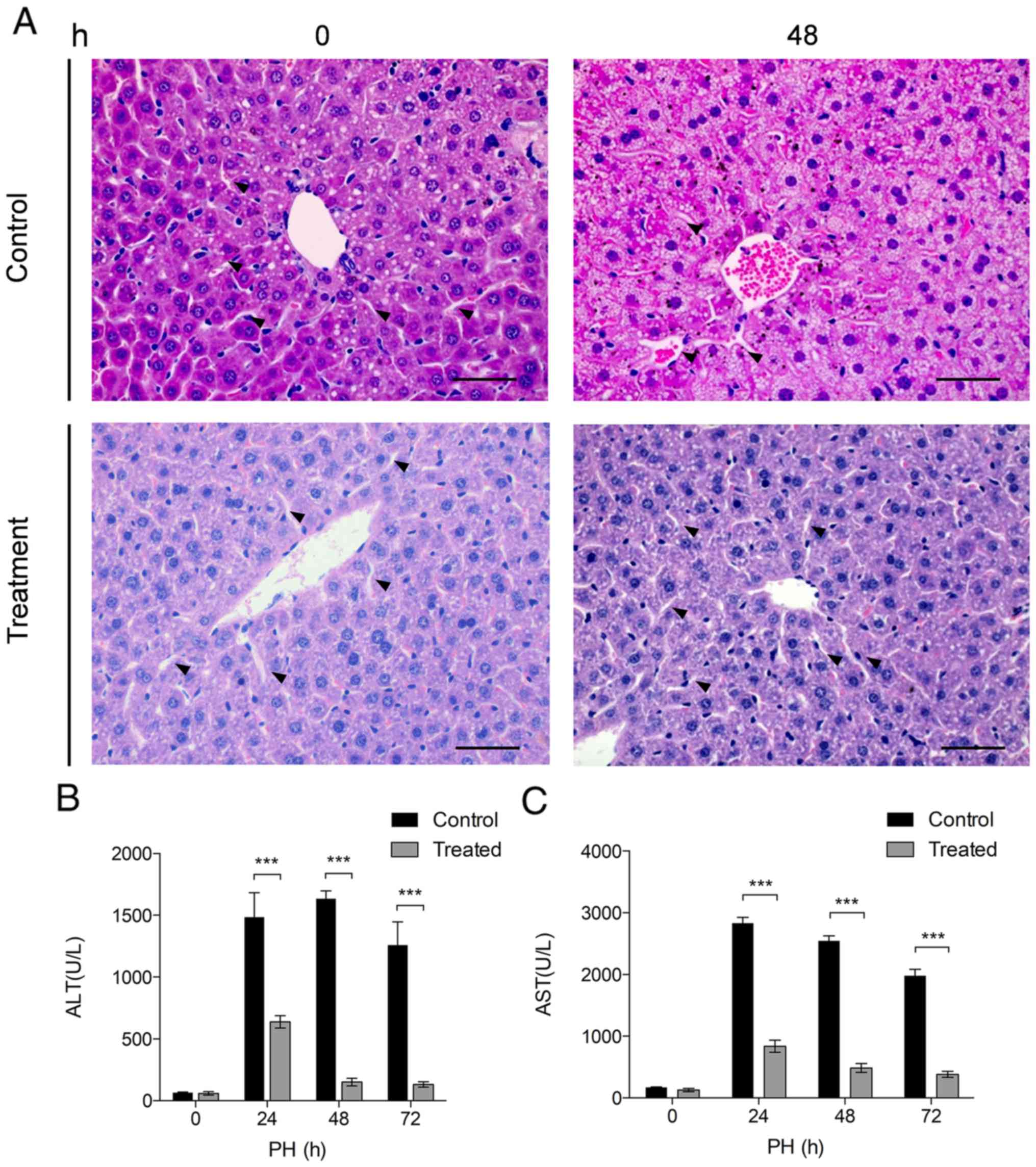
Damage caused by PH is repaired in the
Nam-treated group
A previous study reported that severe liver damage
may occur in short time periods following PH (31). To assess whether Nam may reduce
liver damage following PH, H&E staining was conducted on liver
tissue sections. The results of the H&E staining revealed that
the liver tissues of the control group exhibited evident swelling,
fatty degeneration and nuclear condensation of hepatocytes 48 h
following PH, along with severe sinusoidal narrowing (Fig. 3A). By contrast, sections from the
treatment group demonstrated patent sinusoids, and better
preservation of the cytoplasm and nuclear morphology, suggesting
that treatment with Nam may repair liver damage following PH.
ALT and AST are markers of liver injury following
PH. According to a previous study, these two markers increase
markedly following PH (31). To
further determine whether Nam has the potential to reduce liver
injury following PH, the serum content of ALT and AST was analyzed
(Fig. 3B and C). It was observed
that the serum levels of ALT and AST were significantly decreased
following PH in the treatment group compared with the control group
(Fig. 3B and C; P<0.001),
suggesting that Nam may protect liver function following trauma
caused by PH.
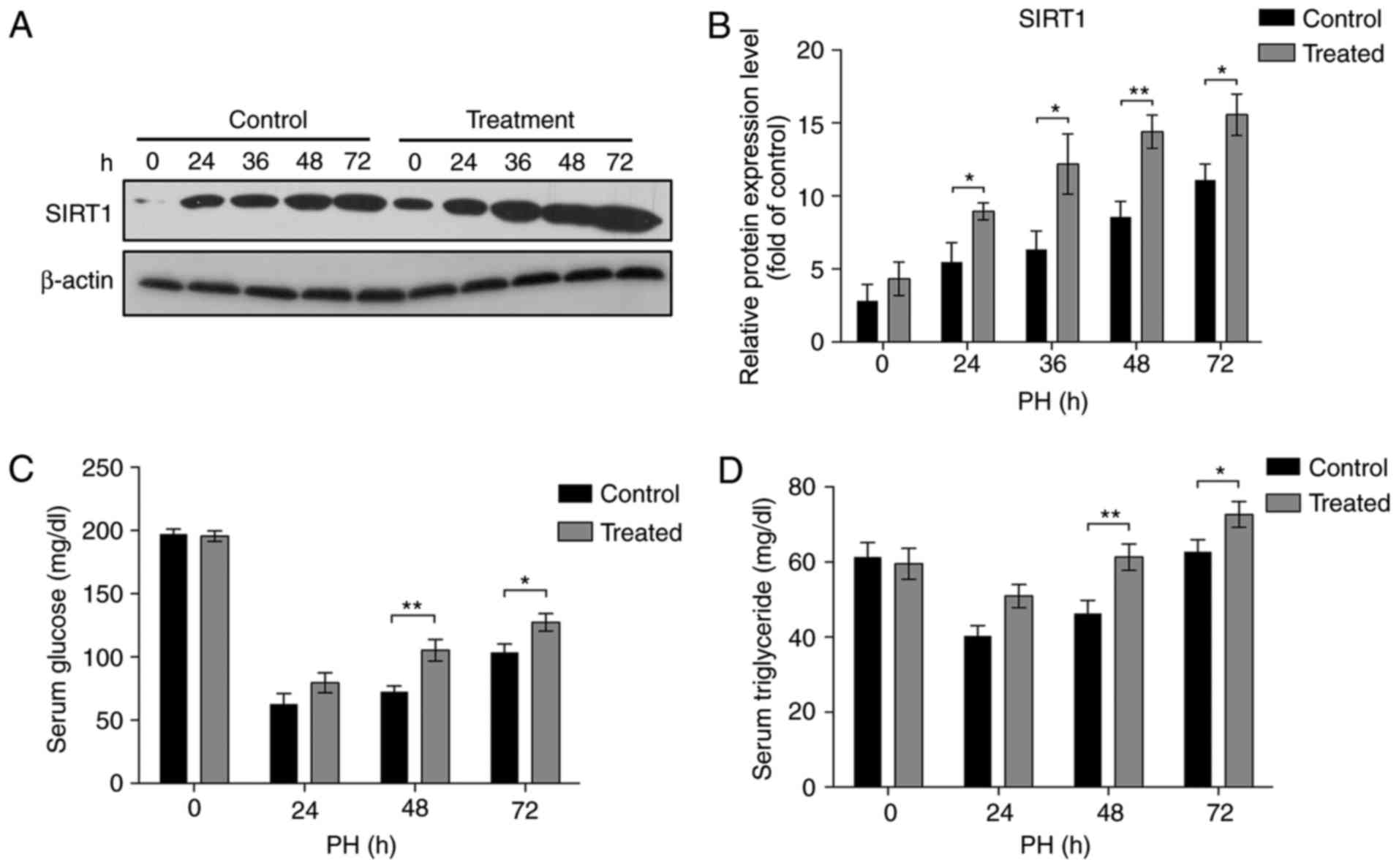
Nam upregulates the expression of
SIRT1
Our previous study demonstrated that the addition of
Nam promoted the expression of SIRT1, a key regulator of liver
regeneration, suggesting that Nam may promote liver regeneration by
upregulating the expression of SIRT1 (25). To verify the mechanisms of Nam in
promoting liver regeneration, western blotting analysis was
conducted to determine the expression of SIRT1 in the treatment and
control groups. As demonstrated in Fig. 4A and B, although the expression of
SIRT1 following PH increased in the two groups in a time-dependent
manner, the addition of Nam further facilitated the significantly
increased expressions of SIRT1 24–72 h following PH (Fig. 4A and B; P<0.05).
Glu and TG metabolisms have been reported to be a
prerequisite for liver regeneration (23,32).
Previous studies have demonstrated that the levels of Glu and TG
are modulated by SIRT1 during the process of liver regeneration
(24,33,34).
To further elucidate the mechanisms of Nam in promoting liver
regeneration, the levels of TG and Glu from serum samples were
analyzed. Compared with the control group, the levels of Glu and TG
were restored faster in the treatment group (Fig. 4C and D), which was consistent with
the result that the expression of SIRT1 was upregulated by Nam.
These results suggest that Nam may elevate the levels of Glu and TG
by promoting the expression of SIRT1, thereby accelerating the
process of liver regeneration.
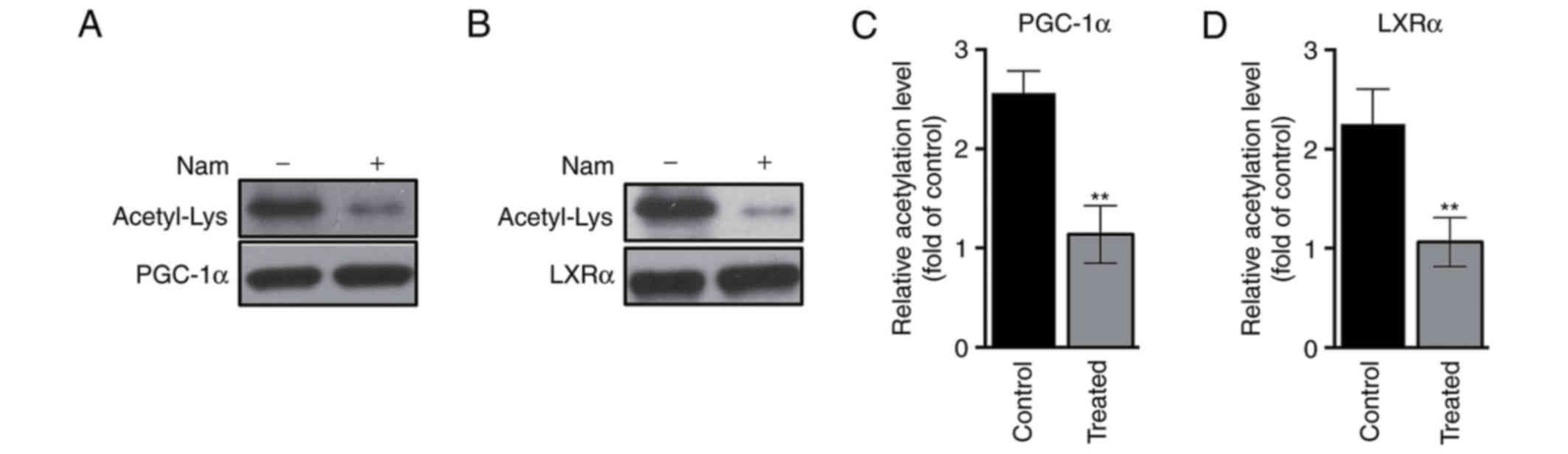
Deacetylating ability of SIRT1 is
enhanced following treatment with Nam
To further investigate the mechanism underlying
SIRT1-mediated metabolic processes following treatment with Nam,
bioinformatics analysis was conducted (data not shown), which
identified two substrates of SIRT1, PGC-1α and LXRα, which may be
deacetylated by SIRT1 and have been previously implicated in the
process of glucogenesis and lipid synthesis, respectively (35,36).
As demonstrated in Fig. 5A and C,
compared with the control group, mice injected with Nam
demonstrated a significant decrease in the lysine acetylation level
of PGC-1α (P<0.01), suggesting that Nam may regulate Glu levels
through the SIRT1-PGC-1α signaling pathways. Likewise, it was
additionally observed that there was a significant decrease in the
lysine acetylation level of LXRα in the treated group (Fig. 5B and D; P<0.01), suggesting that
Nam may regulate lipid levels through the SIRT1-LXRα signaling
pathways.
Discussion
At present, one of the clinical concerns regarding
liver regeneration is identifying effective regimens to accelerate
liver regeneration following surgical resection (37,38).
Conventionally, Nam has been regarded as an anti-inflammatory,
anti-oxidant and anti-apoptotic agent (39). Previous studies have demonstrated
that Nam may promote the expression of SIRT1, an important
regulator in liver regeneration in hepatocytes (25,33,34),
indicating that Nam has the potential to promote liver
regeneration. In the present study, the results demonstrated that
the addition of Nam promoted hepatocyte proliferation and induced
the activation of SIRT1. Furthermore, the present results are
supported as Nam may accelerate skin tissue regeneration following
physicochemical injuries (15,16),
suggesting that Nam has the potential for application in vital
tissue regeneration; however, this requires further
examination.
Hepatic metabolism, including modulations in serum
Glu and lipid, has been revealed to be an essential part of liver
regeneration (26). Within hours
of surgery, mice subjected to PH develop significant hypoglycemia
when compared with the control group (40). Subsequently, adaptation by
modulating the level of blood Glu occurs, including the induction
of hepatic gluconeogenic machinery and the suppression of liver
glycolytic activity (41). The
regulation of lipid is additionally significant for liver
regeneration (42). Lipid has
always been considered to be a structural component of biological
membranes. Furthermore, lipid is involved in the regulation of the
intermediate metabolism of different liver cell types, including
hepatocytes, hepatic stellate cells and Kupffer cells (42). In the present study, it was
demonstrated that Nam promoted the proliferation of hepatocytes, in
addition to Glu and lipid metabolism, which was indicative of an
association between metabolism and liver regeneration.
SIRT1 has been reported to serve an essential role
in various metabolic processes of the liver. During the process of
liver regeneration, SIRT1 has been recognized as an important
regulator of metabolic circadian rhythms, which have been revealed
to be closely associated with cellular proliferation (43–46).
Furthermore, it was observed that SIRT1 serves a central role in
maintaining proper circadian regulation of metabolic processes,
including gluconeogenesis and lipid synthesis, thus affecting the
process of liver regeneration (33,34).
However, the detailed molecular mechanisms underlying
SIRT1-mediated regulation of liver regeneration have not been fully
elucidated. In the present study, it was observed that the
acetylation levels of PGC-1α and LXRα were significantly decreased
upon treatment with Nam, suggesting that Nam-induced SIRT1
activation may promote liver regeneration by inducing the
deacetylation of PGC-1α and LXRα, thereby promoting gluconeogenesis
and lipid synthesis during the process of liver regeneration.
The role of Nam in regulating the activation of
SIRT1 remains controversial. Nam is generally accepted as the
product of SIRT-catalyzed deacetylation reactions and was reported
to inhibit the activity of SIRT1 (35,36).
However, as Nam is the predominant precursor for cellular
NAD+ biosynthesis via the salvage pathway (47,48),
a number of previous studies have reported that Nam supplementation
in cell culture increased intracellular NAD+ levels,
leading to the activation of SIRT1 (49,50).
Our previous study demonstrated that Nam elevated the intracellular
cyclic adenosine 3′,5′-monophosphate (cAMP) expression level by
suppressing phosphodiesterase activity, leading to downstream
cAMP-dependent protein kinase and cAMP-response element binding
activation, thereby inducing the upregulation of SIRT1 (25). By conducting PH in mouse models,
the results of this previous study were validated; Nam may promote
the expression of SIRT1 in vivo. The factors leading to
these conflicted observations may derive from cell type, dosage and
exposure duration. In the present study, the results revealed that
Nam at a dose of 250 mg/kg promoted the expression of SIRT1 and
activated its deacetylating ability in residual liver tissue. Based
on relevant literature, it was hypothesized that SIRT1 may be
upregulated by Nam at a low dosage, and downregulated at a high
dosage. However, since SIRT1-knocked-out mice could not be
obtained, we were not able to clarify the specific mechanism under
which Nam regulates the expression of SIRT1. Further studies are
required to investigate the mechanism of how Nam modulates the
expression of SIRT1 in regulating the metabolic response of the
liver during regeneration.
In conclusion, the present study revealed that Nam
may promote the proliferation of hepatocytes following PH and
stimulate the activation of SIRT1 during liver regeneration. The
present results provided insight into the potency of Nam in
manipulating liver regeneration, which support a preclinical
rationale to examine the clinical use of Nam for surgical resection
recovery.
Acknowledgements
The authors thank the Core Facility of West China
Hospital for their technical support.
Funding
The present study was supported by the Natural
Science Foundation of China (grant nos. 81770615, 81700555,
81672882 and 81502441), the Science and Technology Support Program
of Sichuan Province (grant no. 2017SZ0003), Tianqing Liver Diseases
Research Fund of China Foundation for Hepatitis Prevention and
Control (grant no. TQGB20170067) and Scientific Research Starting
Foundation for Youths of Sichuan University (grant no.
2015SCU11999-9).
Availability of data and materials
The analyzed data sets generated during the study
are available from the corresponding author on reasonable
request.
Authors' contributions
H-FW, H-TL, J-XL and K-FY designed the experiments.
H-FW, H-TL and LX performed the experiments. H-FW, M-HL and LL
analyzed the data. H-FW, H-TL and K-FY wrote the manuscript. H-FW,
K-FY and YZ revised the manuscript. YZ proposed the study concept
and supervised the research. All authors reviewed the
manuscript.
Ethics approval and consent to
participate
Animal care and experimental protocols were in
accordance with guidelines provided by the Institutional Animal
Care and Use Committee of Sichuan University. Ethics approval was
obtained from the Institutional Ethics Committee of Sichuan
University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Llovet JM, Di Bisceglie AM, Bruix J,
Kramer BS, Lencioni R, Zhu AX, Sherman M, Schwartz M, Lotze M,
Talwalkar J, et al: Design and endpoints of clinical trials in
hepatocellular carcinoma. J Natl Cancer Inst. 100:698–711. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Llovet JM, Brú C and Bruix J: Prognosis of
hepatocellular carcinoma: The BCLC staging classification. Semin
Liver Dis. 19:329–338. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
European Association For The Study Of The
Liver; European Organisation For Research And Treatment Of Cancer:
EASL-EORTC clinical practice guidelines: Management of
hepatocellular carcinoma. J Hepatol. 56:908–943. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bruix J and Sherman M: American
Association for the Study of Liver Diseases: Management of
hepatocellular carcinoma: An update. Hepatology. 53:1020–1022.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lehmann K, Tschuor C, Rickenbacher A, Jang
JH, Oberkofler CE, Tschopp O, Schultze SM, Raptis DA, Weber A, Graf
R, et al: Liver failure after extended hepatectomy in mice is
mediated by a p21-dependent barrier to liver regeneration.
Gastroenterology. 143:1609–1619.e4. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Roayaie S, Jibara G, Tabrizian P, Park JW,
Yang J, Yan L, Schwartz M, Han G, Izzo F, Chen M, et al: The role
of hepatic resection in the treatment of hepatocellular cancer.
Hepatology. 62:440–451. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tremosini S, Reig M, de Lope CR, Forner A
and Bruix J: Treatment of early hepatocellular carcinoma: Towards
personalized therapy. Dig Liver Dis. 42 Suppl 3:S242–S248. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Clavien PA, Petrowsky H, DeOliveira ML and
Graf R: Strategies for safer liver surgery and partial liver
transplantation. N Eng J Med. 356:1545–1559. 2007. View Article : Google Scholar
|
|
10
|
Clavien PA, Oberkofler CE, Raptis DA,
Lehmann K, Rickenbacher A and El-Badry AM: What is critical for
liver surgery and partial liver transplantation: Size or quality?
Hepatology. 52:715–729. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen AC and Damian DL: Nicotinamide and
the skin. Australas J Dermatol. 55:169–175. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Oblong JE: The evolving role of the
NAD+/nicotinamide metabolome in skin homeostasis, cellular
bioenergetics, and aging. DNA Repair (Amst). 23:59–63. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Stevens MJ, Li F, Drel VR, Abatan OI, Kim
H, Burnett D, Larkin D and Obrosova IG: Nicotinamide reverses
neurological and neurovascular deficits in streptozotocin diabetic
rats. J Pharmacol Exp Ther. 320:458–464. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kaneko S, Wang J, Kaneko M, Yiu G, Hurrell
JM, Chitnis T, Khoury SJ and He Z: Protecting axonal degeneration
by increasing nicotinamide adenine dinucleotide levels in
experimental autoimmune encephalomyelitis models. J Neurosci.
26:9794–9804. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Thompson BC, Halliday GM and Damian DL:
Nicotinamide enhances repair of arsenic and ultraviolet
radiation-induced DNA damage in HaCaT keratinocytes and ex vivo
human skin. PLoS One. 10:e01174912015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ashkani Esfahani S, Khoshneviszadeh M,
Namazi MR, Noorafshan A, Geramizadeh B, Nadimi E and Razavipour ST:
Topical nicotinamide improves tissue regeneration in excisional
full-thickness skin wounds: A stereological and pathological study.
Trauma Mon. 20:e181932015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gariani K, Ryu D, Menzies KJ, Yi HS, Stein
S, Zhang H, Perino A, Lemos V, Katsyuba E, Jha P, et al: Inhibiting
poly ADP-ribosylation increases fatty acid oxidation and protects
against fatty liver disease. J Hepatol. 66:132–141. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lehwald N, Tao GZ, Jang KY, Papandreou I,
Liu B, Liu B, Pysz MA, Willmann JK, Knoefel WT, Denko NC and
Sylvester KG: β-Catenin regulates hepatic mitochondrial function
and energy balance in mice. Gastroenterology. 143:754–764. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Eriksson S, Prigge JR, Talago EA, Arnér ES
and Schmidt EE: Dietary methionine can sustain cytosolic redox
homeostasis in the mouse liver. Nat Commun. 6:64792015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tummala KS, Gomes AL, Yilmaz M, Graña O,
Bakiri L, Ruppen I, Ximénez-Embún P, Sheshappanavar V,
Rodriguez-Justo M, Pisano DG, et al: Inhibition of de novo NAD(+)
synthesis by oncogenic URI causes liver tumorigenesis through DNA
damage. Cancer Cell. 26:826–839. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Imai S, Armstrong CM, Kaeberlein M and
Guarente L: Transcriptional silencing and longevity protein Sir2 is
an NAD-dependent histone deacetylase. Nature. 403:795–800. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Timchenko NA, Harris TE, Wilde M, Bilyeu
TA, Burgess-Beusse BL, Finegold MJ and Darlington GJ:
CCAAT/enhancer binding protein alpha regulates p21 protein and
hepatocyte proliferation in newborn mice. Mol Cell Biol.
17:7353–7361. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lai HS, Chen WJ and Chen KM: Energy
substrate for liver regeneration after partial hepatectomy in rats:
Effects of glucose vs fat. JPEN J Parenter Enteral Nutr.
16:152–156. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jin J, Iakova P, Jiang Y, Medrano EE and
Timchenko NA: The reduction of SIRT1 in livers of old mice leads to
impaired body homeostasis and to inhibition of liver proliferation.
Hepatology. 54:989–998. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li J, Dou X, Li S, Zhang X, Zeng Y and
Song Z: Nicotinamide ameliorates palmitate-induced ER stress in
hepatocytes via cAMP/PKA/CREB pathway-dependent Sirt1 upregulation.
Biochim Biophys Acta. 1853:2929–2936. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Forbes SJ and Newsome PN: Liver
regeneration-mechanisms and models to clinical application. Nat Rev
Gastroenterol Hepatol. 13:473–485. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cardiff RD, Miller CH and Munn RJ: Manual
hematoxylin and eosin staining of mouse tissue sections. Cold
Spring Harb Protoc. 2014:655–658. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ma S, Zhao Y, Sun J, Mu P and Deng Y:
miR449a/SIRT1/PGC-1α is necessary for mitochondrial biogenesis
induced by T-2 toxin. Front Pharmacol. 8:9542018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang S, Wang C, Turdi S, Richmond KL,
Zhang Y and Ren J: ALDH2 protects against high fat diet-induced
obesity cardiomyopathy and defective autophagy: Role of CaM kinase
II, histone H3K9 methyltransferase SUV39H, Sirt1, and PGC-1α
deacetylation. Int J Obes (Lond). 42:1073–1087. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Fausto N, Campbell JS and Riehle KJ: Liver
regeneration. J Hepatol. 57:692–694. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jin X, Zhang Z, Beer-Stolz D, Zimmers TA
and Koniaris LG: Interleukin-6 inhibits oxidative injury and
necrosis after extreme liver resection. Hepatology. 46:802–812.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tajima T, Goda N, Fujiki N, Hishiki T,
Nishiyama Y, Senoo-Matsuda N, Shimazu M, Soga T, Yoshimura Y,
Johnson RS and Suematsu M: HIF-1alpha is necessary to support
gluconeogenesis during liver regeneration. Biochem Biophys Res
Commun. 387:789–794. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bellet MM, Masri S, Astarita G,
Sassone-Corsi P, Della Fazia MA and Servillo G: Histone deacetylase
SIRT1 controls proliferation, circadian rhythm, and lipid
metabolism during liver regeneration in mice. J Biol Chem.
291:23318–23329. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Jin J, Hong IH, Lewis K, Iakova P, Breaux
M, Jiang Y, Sullivan E, Jawanmardi N, Timchenko L and Timchenko NA:
Cooperation of C/EBP family proteins and chromatin remodeling
proteins is essential for termination of liver regeneration.
Hepatology. 61:315–325. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Rodgers JT, Lerin C, Haas W, Gygi SP,
Spiegelman BM and Puigserver P: Nutrient control of glucose
homeostasis through a complex of PGC-1alpha and SIRT1. Nature.
434:113–118. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Li X, Zhang S, Blander G, Tse JG, Krieger
M and Guarente L: SIRT1 deacetylates and positively regulates the
nuclear receptor LXR. Mol Cell. 28:91–106. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Longo CR, Patel VI, Shrikhande GV, Scali
ST, Csizmadia E, Daniel S, Sun DW, Grey ST, Arvelo MB and Ferran C:
A20 protects mice from lethal radical hepatectomy by promoting
hepatocyte proliferation via a p21waf1-dependent mechanism.
Hepatology. 42:156–164. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Okumura S, Teratani T, Fujimoto Y, Zhao X,
Tsuruyama T, Masano Y, Kasahara N, Iida T, Yagi S, Uemura T, et al:
Oral administration of polyamines ameliorates liver
ischemia/reperfusion injury and promotes liver regeneration in
rats. Liver Transpl. 22:1231–1244. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hwang ES and Song SB: Nicotinamide is an
inhibitor of SIRT1 in vitro, but can be a stimulator in cells. Cell
Mol Life Sci. 74:3347–3362. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Weymann A, Hartman E, Gazit V, Wang C,
Glauber M, Turmelle Y and Rudnick DA: p21 is required for
dextrose-mediated inhibition of mouse liver regeneration.
Hepatology. 50:207–215. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Brinkmann A, Katz N, Sasse D and
Jungermann K: Increase of the gluconeogenic and decrease of the
glycolytic capacity of rat liver with a change of the metabolic
zonation after partial hepatectomy. Hoppe Seylers Z Physiol Chem.
359:1561–1571. 1978. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Delgado-Coello B, Briones-Orta MA,
Macias-Silva M and Mas-Oliva J: Cholesterol: Recapitulation of its
active role during liver regeneration. Liver Int. 31:1271–1284.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Asher G, Gatfield D, Stratmann M, Reinke
H, Dibner C, Kreppel F, Mostoslavsky R, Alt FW and Schibler U:
SIRT1 regulates circadian clock gene expression through PER2
deacetylation. Cell. 134:317–328. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Chang HC and Guarente L: SIRT1 mediates
central circadian control in the SCN by a mechanism that decays
with aging. Cell. 153:1448–1460. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Nakahata Y, Kaluzova M, Grimaldi B, Sahar
S, Hirayama J, Chen D, Guarente LP and Sassone-Corsi P: The
NAD+-dependent deacetylase SIRT1 modulates CLOCK-mediated chromatin
remodeling and circadian control. Cell. 134:329–340. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Sahar S and Sassone-Corsi P: Metabolism
and cancer: The circadian clock connection. Nat Rev Cancer.
9:886–896. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Imai S and Guarente L: Ten years of
NAD-dependent SIR2 family deacetylases: Implications for metabolic
diseases. Trends Pharmacol Sci. 31:212–220. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Haigis MC and Sinclair DA: Mammalian
sirtuins: Biological insights and disease relevance. Annu Rev
Pathol. 5:253–295. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Jang SY, Kang HT and Hwang ES:
Nicotinamide-induced mitophagy: Event mediated by high NAD+/NADH
ratio and SIRT1 protein activation. J Biol Chem. 287:19304–19314.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Liu D, Gharavi R, Pitta M, Gleichmann M
and Mattson MP: Nicotinamide prevents NAD+ depletion and protects
neurons against excitotoxicity and cerebral ischemia: NAD+
consumption by SIRT1 may endanger energetically compromised
neurons. Neuromolecular Med. 11:28–42. 2009. View Article : Google Scholar : PubMed/NCBI
|