Introduction
Lower back pain caused by degenerative spinal
conditions is a serious public health problem. It is estimated that
between 2.8 and 5% of visits to health-care professionals in the
United States are due to this problem, and considering direct and
indirect costs, the overall cost is >$100 billion per year
(1). It has been suggested that
intervertebral disc degeneration (IDD) is a major causative factor
in the development of spinal degenerative diseases (2); however, the specific pathogenesis of
IDD remains unclear. Extracellular matrix synthesis and catabolic
imbalance are believed to be direct causes of IDD (3). It is well known that inflammatory
cytokines, including interleukin IL-1, tumor necrosis factor-α and
IL-6, serve an important role in cellular apoptosis and
extracellular matrix metabolism in the process of IDD (4,5).
Leukemia inhibitory factor (LIF) was first reported
in a study regarding myeloid leukemia M1 cells (6). It is a 180-amino acid glycoprotein
that belongs to the IL-6 sub-family, which is comprised of seven
members, including IL-6, IL-11, IL-27, oncostatin M (OSM), ciliary
neurotrophic factor, cardiotrophin-1 and cardiotrophin-like
cytokine/cytokine-like factor (7).
The aforementioned cytokines share the same receptors, including
LIF receptor β (LIFRβ) and glycoprotein 130 (gp130) (8). LIF is a multifunctional cytokine that
exerts various activities on numerous systems, including bone
remodeling, and the nervous, muscular, endocrine and reproductive
systems (9). Previous studies have
reported that LIF is involved in several osteoarticular
pathological conditions (10–12).
Lotz et al demonstrated that LIF promotes cartilage
catabolism and contributes to the pathogenesis of arthritis
(13). Although chondrocytes and
nucleus pulposus cells have been reported to share many cellular
phenotypes (14), whether LIF is
positively expressed in the nucleus pulposus of intervertebral
discs, and its roles in IDD, have yet to be evaluated.
The present study aimed to evaluate the expression
of LIF in the degenerative nucleus pulposus of an animal model and
in patients with lumbar disc herniation. Furthermore, the effects
of LIF on extracellular matrix synthesis, proliferation and
apoptosis were detected in degenerative nucleus pulposus cells
(DNPCs).
Materials and methods
Materials
A total of 50 adult male New Zealand rabbits
(weight, 2.5±0.2 kg), were purchased from the Experimental Animal
Center of Chongqing Medical University (Chongqing, China). Animals
were maintained in a 26°C room with 40–70% humidity and a 12-h
light/dark cycle, and were allowed free access to food and water.
In addition, degenerative disc samples were obtained from six
patients undergoing lumbar disc herniation surgery (age, 27–68
years; mean age, 48.6 years; three men and three women; Pfirrmann
degeneration grading, III–IV). Nucleus pulposus tissues were also
obtained from a control group (age, 17–49 years; mean age, 37.1
years, three men and two women), which consisted of young trauma
patients (three cases) and adolescent patients with scoliosis (two
cases). All animal and patient procedures were approved by the
Ethics Committee of the First Affiliated Hospital of Chongqing
Medical University (Chongqing, China). Written informed consent was
obtained from adult patients and the parents of adolescent patients
permitting the use of their samples and the publication of any
associated data.
Reagents
Pentobarbital sodium was purchased from Merck KGaA
(Darmstadt, Germany). Safranin O-Fast Green staining reagent was
obtained from Beijing Leagene Biotech Co., Ltd. (Beijing, China).
Dulbecco's modified Eagle's medium (DMEM)/F12 and fetal bovine
serum were obtained from HyClone; GE Healthcare Life Sciences
(Logan, UT, USA). Recombinant Human LIF Protein was purchased from
Novus Biologicals, LLC (Littleton, CO, USA). Collagenase type II
was obtained from Sigma-Aldrich; Merck KGaA. Collagen type II α1
(COL2α1) and LIF primary antibodies were from BIOSS (Beijing,
China; bs-1068R and bs-1058R), and aggrecan primary antibody was
from Abcam (Cambridge, MA, USA; ab3778). Cell Counting Kit-8
(CCK-8) was purchased from Beyotime Institute of Biotechnology
(Haimen, China) and the Annexin V-phycoerythrin (PE)/propidium
iodide (PI) apoptosis detection kit was obtained from BD
Biosciences (Franklin Lakes, NJ, USA).
Animal grouping and model
establishment
A total of 50 New Zealand white rabbits were
randomly divided into the following 5 groups, with 10 rabbits in
each group: The control group, and the 0, 2, 4 and 8 week groups
according to modeling time. According to the method introduced by
Masuda et al (15), 3%
pentobarbital sodium (30 mg/kg) was injected into the marginal
veins of each rabbit, intervertebral discs between L4-5, L5-6 and
L6-7 were exposed via the posterior lateral approach and the
annulus fibrosus was punctured using a 18G needle (5 mm depth for 5
sec). The incision was then closed in layers.
Imaging evaluation
After modeling at 0, 2, 4 and 8 weeks, sagittal
T2-weighted magnetic resonance imaging (MRI) scans of the lumbar
spine were performed under the following parameter settings: Repeat
time, 2,500 msec; echo time, 85 msec; scanning layer, 11; layer
thickness, 2 mm; matrix, 196×196; bandwidth, 31.25; field of view,
18×18. The intervertebral discs were evaluated according to the
method introduced by Masudaet al (15). Images and scoring of the discs were
assessed by two experienced, independent radiologists.
Histological assessment
Rabbits were sacrificed on the day that MRI
examinations were conducted, after which, discs were removed and
fixed in 4% paraformaldehyde for 24 h at room temperature. They
were then decalcified in EDTA solution for 4–5 weeks (decalcifying
solution was replaced every 3–4 days). Disc tissues were embedded
in paraffin, sliced into 5 µm sections on the median sagittal,
dewaxed in water and then stained with Safranin O-Fast Green,
according to the manufacturer's protocol. Pathological assessment
of degeneration was performed according to the Masuda criteria
under a confocal microscope (Olympus Corporation, Tokyo, Japan)
(15).
Measurement of LIF expression in
intervertebral disc nucleus pulposus
Total proteins were extracted from nucleus pulposus
samples harvested from rabbit and human discs by grinding in liquid
nitrogen (PROTTOT-1KT, Sigma-Aldrich; Merck KGaA). Protein
concentrations were then measured using the bicinchoninic acid
(BCA) method and the protein expression levels of LIF were detected
in each group by western blot analysis. Briefly, 50 µg of extracted
proteins were separated by 10% SDS–PAGE, then were electro-blotted
onto PVDF membranes (EMD Millipore, Billerica, MA, USA). The
membranes were blocked using 5% blocking buffer (P0252; Beyotime
Institute of Biotechnology) at room temperature for 1 h. After
incubation with rabbit anti-LIF antibody (1:1,000; SAB2102317;
Sigma-Aldrich; Merck KGaA), membranes were washed 3 times with TBST
(P0023C; Beyotime Institute of Biotechnology) for 10 min and
followed by incubation with a secondary antibody (1:1,000;
Sigma-Aldrich; Merck KGaA) for 2 h at room temperature, and were
then washed 3 times again with TBST. Relative levels of
immunoreactivity were quantified using the Kodak In-vivo Imaging
System (Kodak, Rochester, NY, USA). Rabbit anti-GADPH (1:2,000;
SAB2108668; Sigma-Aldrich; Merck KGaA) was used as an internal
control for the concentration of proteins loaded. Images were
analyzed using Image J version 2× (National Institutes of Health,
Bethesda, MD, USA).
Culture of human DNPCs
Human nucleus pulposus cells were isolated from the
obtained degenerative discs of patients as described previously
(16). Blood in nucleus pulposus
samples was washed away using PBS supplemented with penicillin
(800,000 U/l) and streptomycin (1,000,000 U/l). The samples were
cut into pieces using ophthalmic scissors, and were then digested
using 0.25% trypsin and 0.2% type II collagenase. After sieving
through a 200-mesh cell sieve, the cells were centrifuged (300 × g,
4°C, 5 min) and suspended in a culture flask and were cultured in
DMEM/F12 medium containing 20% fetal bovine serum at 37°C in an
incubator containing 5% CO2.
Detection of aggrecan and COL2α1
protein
Passage (P)2-P3 cells were seeded into 10-cm culture
dishes and treated with recombinant human LIF protein (rhLIF) at 0,
10, 20, 50 or 100 ng/ml for 24 h at room temperature, or were
treated with 100 ng/ml rhLIF for 0, 24, 48 or 72 h, as described
previously (17). Subsequently,
cells from each group were collected to extract total protein.
After protein concentration was measured using the BCA method, the
protein expression levels of aggrecan and COL2α1 were detected by
western blot analysis.
P2 cells were also seeded onto glass slides and
treated with 0 or 100 ng/ml rhLIF for 48 h. The slides were
collected, rinsed with PBS, fixed with 40 g/l paraformaldehyde for
30 min at room temperature, and rinsed with PBS three times (5
min/wash). Subsequently, the cells were stained with 1 g/l
toluidine blue stain for 10 min, washed with double distilled water
for 1 min, and then placed under an inverted phase contrast
microscope to detect proteoglycan staining (18).
Cell proliferation assay
P2 cells were treated with various concentrations of
rhLIF and then incubated with serum-free medium for 24 h at room
temperature. Cell proliferation was measured using the CCK-8 kit,
according to the manufacturer's protocol. Cells were incubated with
CCK-8 solution for 4 h and cell proliferation rate was assessed by
measuring absorbance at 450 nm.
Apoptosis analysis
The apoptotic rate was detected by flow cytometry
using the Annexin V-PE/PI apoptosis detection kit. P2 cells were
treated with various concentrations of rhLIF and then incubated
with serum-free medium for 24 h at room temperature. Cells were
then collected and suspended in binding buffer. Subsequently, cells
were counted and incubated with 5 µl Annexin V-PE and 5 µl PI for
30 min at room temperature in the dark. Finally, flow cytometry was
performed within 1 h. Apoptotic cells were defined as Annexin V
(+)/PI (−) and Annexin V (+)/PI (+).
Statistical analysis
All analyses were performed using SPSS version 19.0
(IBM Corp., Armonk, NY, USA). All data were presented as the means
± standard deviation. For each independent in vitro
experiment, at least three technical replicates were analyzed.
Differences between groups were analyzed by one-way analysis of
variance followed by Student-Newman-Keuls post hoc test to
determine multiple comparisons. Correlation analysis of the MRI
score, histological score and modeling time was conducted using the
Spearman Rank Correlation test. P<0.05 was considered to
indicate a statistically significant difference.
Results
Successful generation of an IDD
model
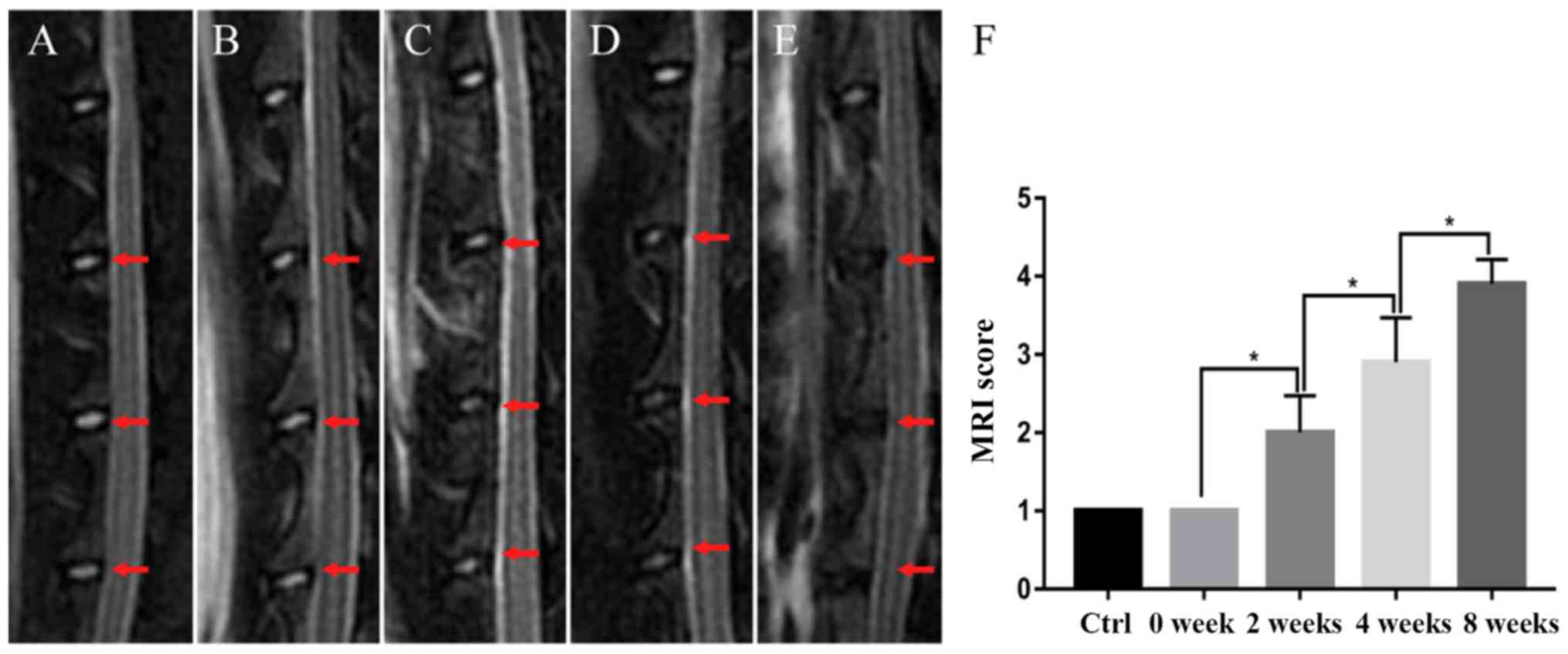
Nuclei from the control group and the 0 week
modeling group exhibited a uniform, high signal in MRI. However,
the nuclei signals from the 2 week modeling group were slightly
decreased, and the signals were further reduced in the 4 week
modeling group. In the 8 week modeling group, no signal was
detected in the nuclei. According to the modified Thompson scoring
system, scores of the control group and the 0 week modeling group
were consistent with each other, whereas scores of the 2, 4 and 8
week modeling groups increased with time. The differences among the
groups were statistically significant (1.000±0.000, 1.000±0.200,
2.000±0.471, 2.900±0.568 and 3.900±0.316 in the control, 0, 2, 4
and 8 week groups, respectively; P<0.05; Fig. 1).
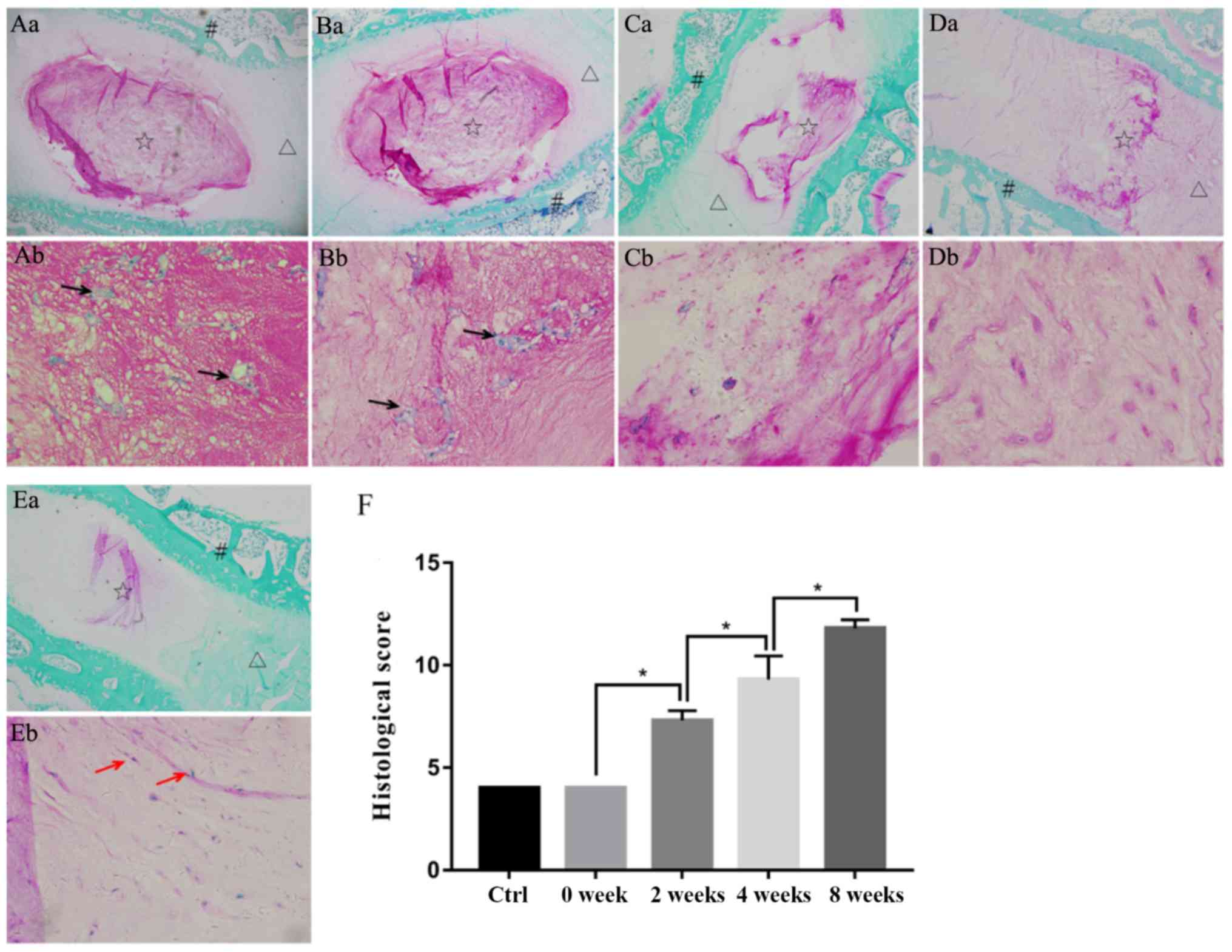
At low magnification, the nucleus pulposus of the
control group and the 0 week group (magnification, ×40) was round
or oval shaped, and the annulus fibrosis was in a regular circular
arrangement. At high magnification (magnification, ×400), the foamy
gel-like matrix was abundant and nucleated cells were clustered or
had aggregated into a ring. In the 2 week modeling group, the
nucleus pulposus was smaller than in the 0 week modeling group and
the junction with the annulus fibrosis was unclear or irregular. In
addition, mild extracellular matrix shrinkage and scattered nucleus
pulposus cells were observed. In the 4 week modeling group, the
nucleus pulposus exhibited obvious extracellular matrix shrinkage
and fibrosis, and the number of normal nucleus pulposus cells was
markedly reduced. In the 8 week modeling group, fibrosis in the
nucleus pulposus was further aggravated and it was continuous with
the annulus fibrosus; the junction between the two regions was
obviously fractured and delaminated. According to Masuda's
histological scoring, there was no significant difference between
the control group and the 0 week modeling group. However, the
degeneration scores increased with the prolongation of modeling
time, and differences among the modeling groups were statistically
significant (4.000±0.000, 4.000±0.0400, 7.300±0.483, 9.300±1.160
and 11.800±0.422 in the control, 0, 2, 4 and 8 week groups,
respectively; P<0.05; Fig.
2).
Degeneration scores of intervertebral
discs are significantly correlated with modeling time
There was a significant positive correlation between
modeling time and MRI score (r=0.939; P<0.05). In addition, the
correlation between modeling time and histological score was
significantly positively (r=0.958; P<0.05). MRI score was also
significantly correlated with histological score (r=0.967;
P<0.05) -. The results of this correlation analysis indicated
that the annulus puncture degeneration model was successfully
established, and with the extension of modeling time, degeneration
of intervertebral discs was aggravated.
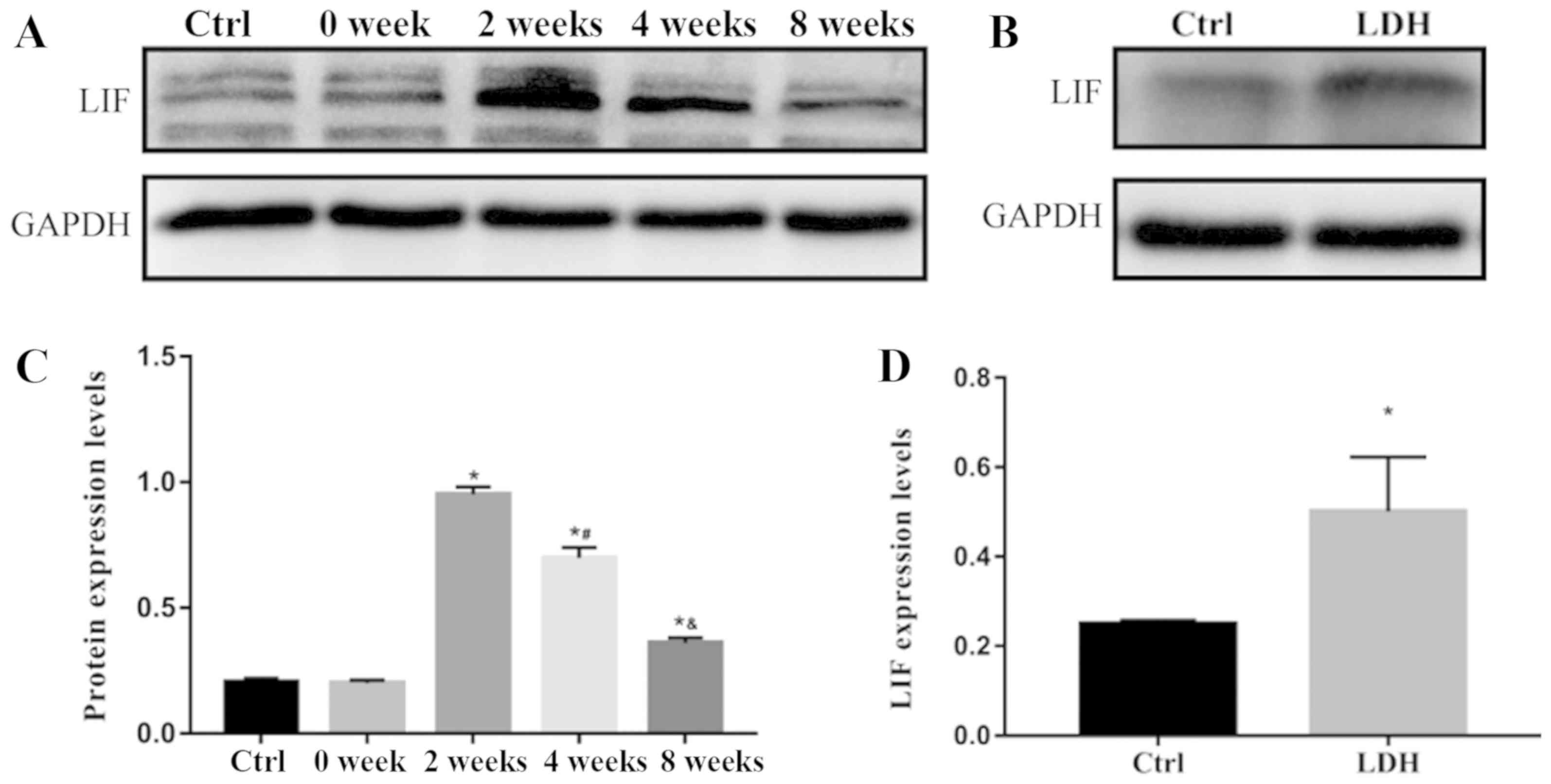
LIF is highly expressed in the
degenerated nucleus pulposus
Western blotting was used to detect LIF protein
expression (Fig. 3). The protein
expression levels of LIF in nucleus pulposus samples from the
control group and the 0 week modeling group were low, and the
difference between them was not statistically significant
(0.205±0.147 vs. 0.200±0.114; P=0.459). Conversely, LIF protein
expression was significantly higher in the 2 week modeling group;
however, as the duration of modeling time and the progression of
disc degeneration were prolonged, LIF protein expression was
gradually decreased in the 4 and 8 week modeling groups. The
differences between the 0, 2, 4 and 8 week modeling groups were
statistically significant (0.200±0.114, 0.950±0.301, 0.698±0.410,
0.360±0.210 in the control, 0, 2, 4 and 8 week groups,
respectively; P<0.05; Fig. 3A and
3C). The protein expression levels of LIF were also
significantly increased in the nucleus pulposus samples of patients
with lumbar disc herniation compared with in the control group
(0.250±0.007 vs. 0.501±0.012; P<0.05) (Fig. 3B and 3D).
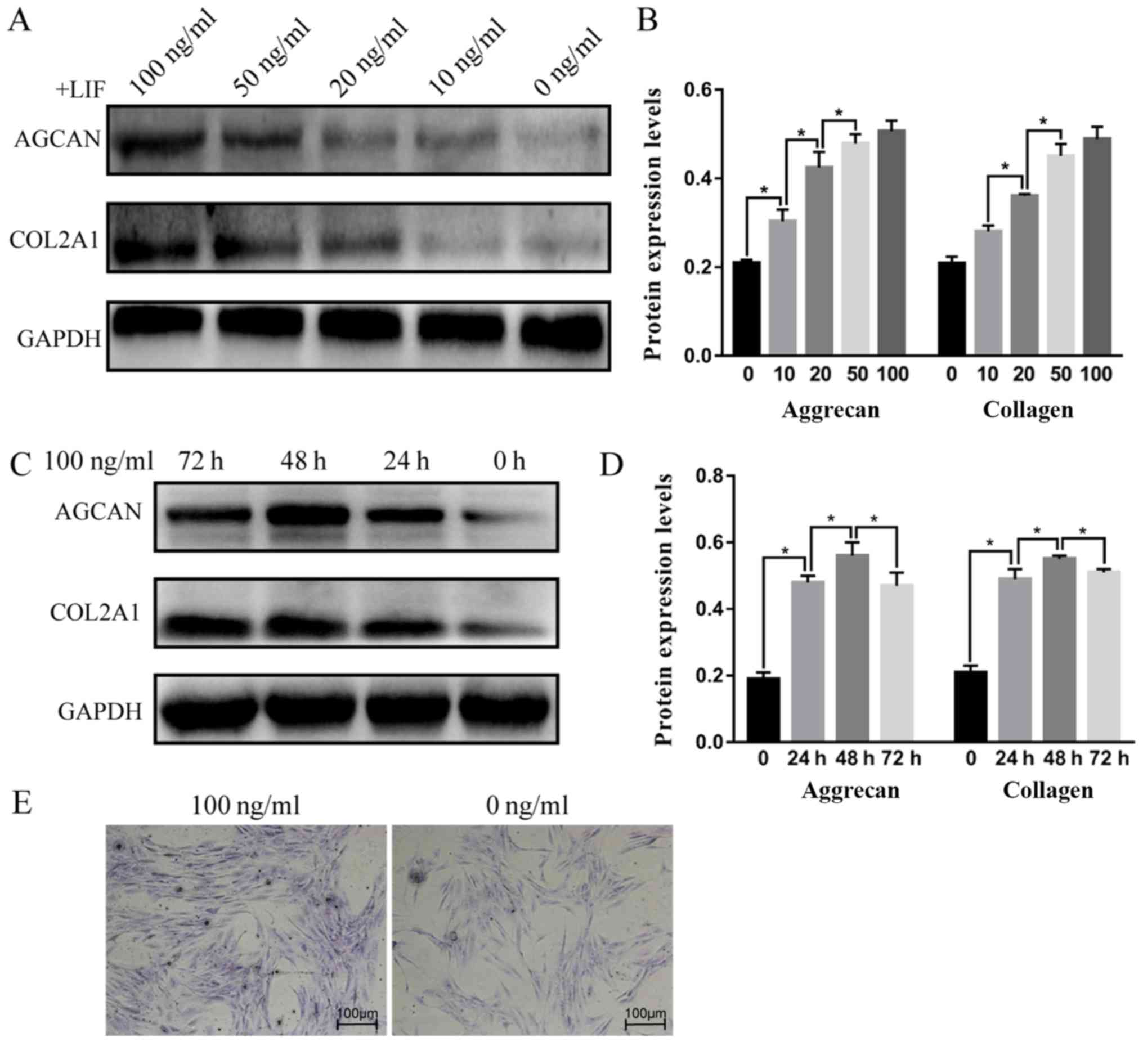
LIF promotes aggrecan and COL2α1
protein expression in human DNPCs
Western blot analysis revealed that the protein
expression levels of aggrecan and COL2α1 were increased in response
to increasing rhLIF concentrations (Fig. 4A-D). The protein expression levels
of aggrecan and COL2α1 in the rhLIF-treated groups were
significantly higher compared with in the 0 ng/ml group [aggrecan:
0.210±0.007, 0.304±0.023, 0.425±0.035, 0.507±0.024 and 0.479±0.020
in the 0, 10, 20, 50 and 100 ng/ml groups, respectively
(P<0.05); COL2α1, 0.209±0.015, 0.281±0.013, 0.361±0.004,
0.451±0.027 and 0.489±0.028 in the 0, 10, 20, 50 and 100 ng/ml
groups, respectively (P<0.05)]. At 100 ng/ml, rhLIF exhibited
the strongest enhancing effect on aggrecan and COL2α expression,
and this effect was maintained for ≥72 h. The results of toluidine
blue staining were consistent with those of the western blot
analysis (Fig. 4E). Aggrecan was
uniformly expressed in the cytoplasm in both groups; however,
compared with the control group, staining intensity was markedly
increased in the 100 ng/ml treatment group.
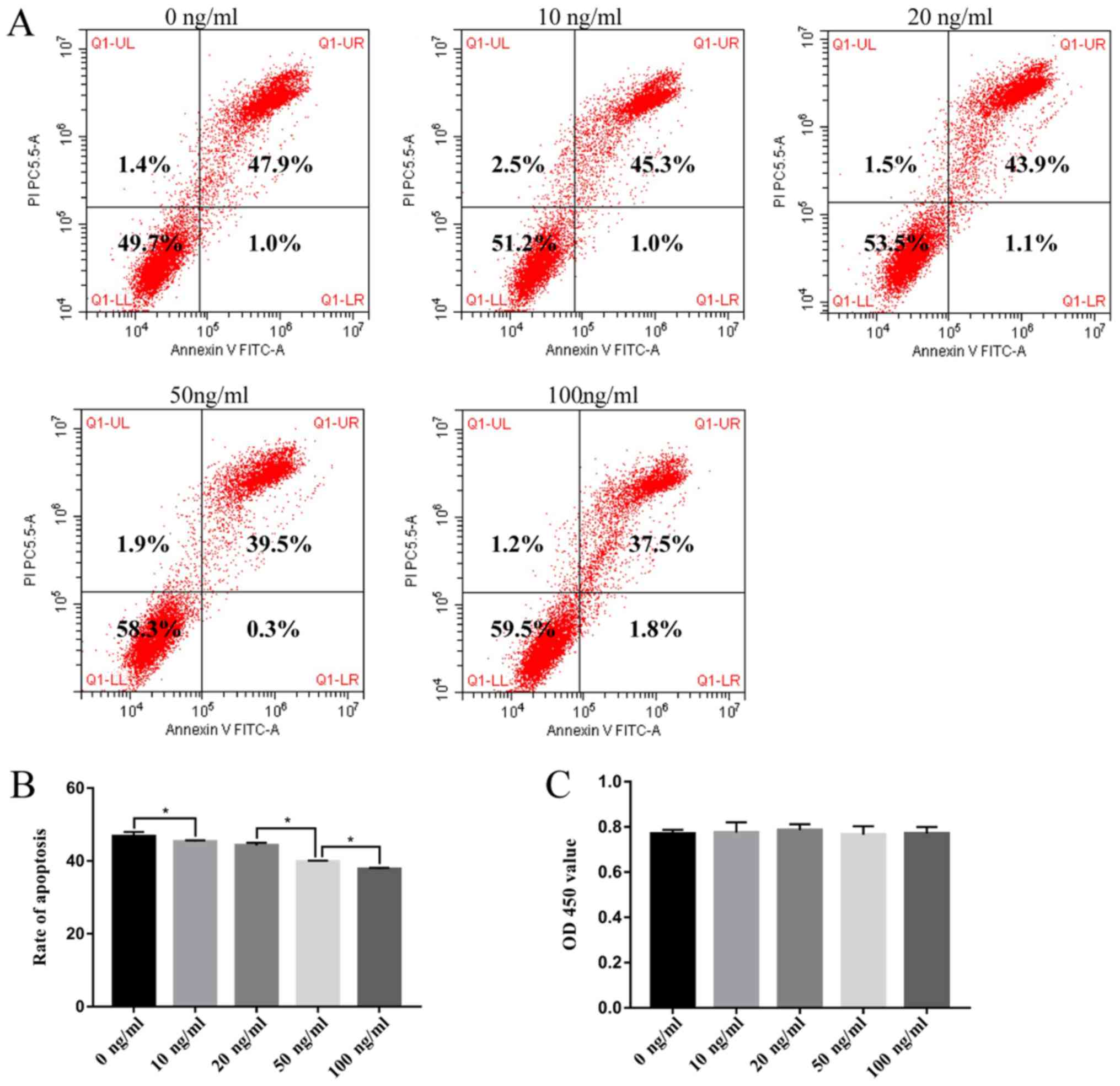
LIF inhibits apoptosis of human
DNPCs
rhLIF inhibited the apoptosis of human DNPCs in a
dose-dependent manner (Fig. 5A and
B). As shown in Fig. 5A and B,
the apoptotic rate of DNPCs was gradually decreased in response to
increasing concentrations of rhLIF, and the rates were
significantly reduced compared with the rate in the control group
(47.89±1.185, 45.310±0.345, 43.883±0.738, 39.520±0.243 and
37.493±0.340 in the 0, 10, 20, 50 and 100 ng/ml groups,
respectively; P<0.05). Conversely, rhLIF had no effect on the
proliferation of human DNPCs (0.770±0.017, 0.774±0.046,
0.785±0.027, 0.765±0.037 and 0.771±0.029 in the 0, 10, 20, 50 and
100 ng/ml groups, respectively; P=0.900) (Fig. 5C).
Discussion
Using the method introduced by Masuda et al
(15), the present study
successfully constructed an animal model of degenerative disc
disease (19). A similar method of
modeling was reported in a previously published study (20). Although some researchers recommend
a longer modeling time of 12 weeks (21), it has been reported that the MRI
signal of the discs generally reaches grade 4 degeneration at 8
weeks and typically stabilizes at later time points (22).
In vivo experiments demonstrated that in
rabbits and humans, the protein expression levels of LIF were
increased in degenerative nucleus pulposus compared with in the
control group. Previous studies have reported that, in articular
cartilage, LIF tends to act as a negative catabolic factor
(12,13). In a mouse model of K/BΧN serum
transfer arthritis, LIF knockout (LIF−/−) mice display a
>50% reduction in clinical arthritis severity. In addition,
significantly lower histological cartilage scores are observed in
LIF−/− mice compared with in wild-type controls
(23). Our previous study also
suggested that LIF expression is highest in advanced articular
cartilage tissue (24). In order
to further clarify the role of LIF in the degeneration of
intervertebral disc tissue, in vitro experiments were
conducted.
Reduced synthesis of aggrecan and COL2α1 proteins,
and decreased numbers of nucleus pulposus cells are important
features of disc degeneration (25,26).
Therefore, with reference to the concentration gradient set by
Upadhyay et al (17),
cultured primary human DNPCs were treated with various
concentrations of rhLIF for different time periods and the protein
expression levels of aggrecan and COL2α1 were detected. The results
revealed that rhLIF promoted the expression levels of both of these
proteins. Toluidine blue staining, which preferentially stains
proteoglycans such as aggrecan (27), confirmed the positive regulatory
effect of rhLIF. Further results suggested that rhLIF had no effect
on the proliferation of DNPCs, but reduced the apoptotic rate of
DNPCs.
In vivo, an increase in LIF was associated
with reduced breakdown of the extracellular matrix and loss of
cells in the intervertebral disc tissue at a relatively early stage
of degeneration (2 week modeling group). As the degeneration
process progressed, the number of nucleus pulposus cells was
decreased, alongside reduced LIF secretion (4 and 8 week modeling
groups). Although the present study indicated that LIF may exert
protective effects on the nucleus pulposus in intervertebral discs,
the amount of LIF naturally secreted during the degenerative
process is not sufficient to resist the progression of IDD.
Therefore, administration of exogenous LIF may be able to slow
degeneration to a certain extent.
LIF, IL-6 and OSM are members of the IL-6 family.
Their amino acid sequences and secondary structures are notably
similar, and they have the same receptor conversion mechanism
(8). Previous studies have
reported that IL-6 can enhance the expression of TIMP
metallopeptidase inhibitor 1 (TIMP-1) in the synovium and
chondrocytes, thus blocking IL-1-mediated collagenolysis. IL-6 can
also induce the production of an IL-1 receptor antagonist and thus
block the proinflammatory effects of IL-1 (28,29).
OSM significantly promotes TIMP-1 expression in chondrocytes, thus
acting as a chondroprotective factor (30). Notably, Upadhyay et al
reported that LIF increases the expression of TIMP-1 in
chondrocytes by ~4-fold (17). The
present results also indicated that LIF may exist as a protective
factor in DNPCs.
The present study had several limitations. Although
it was demonstrated that LIF promoted the expression of aggrecan
and COL2α1, and inhibited apoptosis of DNPCs in vitro,
whether those effects were sustained in vivo was not
investigated. Furthermore, the molecular mechanisms underlying how
LIF was induced in degenerative nucleus pulposus, and how it acted
as a protective factor during the process of IDD was not
investigated.
LIF is a pleiotropic cytokine with a wide range of
activities (31), but there is no
intrinsic tyrosine kinase activity within the LIF receptor;
instead, its two signaling chains, gp130 and LIFRβ, are
constitutively associated with members of the Janus kinase (JAK)
family of tyrosine kinases (32).
Therefore, future mechanistic studies may focus on JAK signaling,
including the JAK/signal transducer and activator of transcription,
mitogen-activated protein kinase and phosphatidylinositol 3-kinase
pathways (9).
In conclusion, the present study explored the
expression of LIF in degenerative intervertebral discs and
discussed the protective effects of LIF on nucleus pulposus cells.
LIF was upregulated during the process of IDD, and promoted the
expression of extracellular matrix components. In addition, it was
suggested that LIF may act as a potential protective factor by
inhibiting apoptosis of DNPCs without affecting cell proliferation.
However, to clarify the specific mechanism underlying the ability
of LIF to alleviate IDD, further investigations are required.
Acknowledgements
Not applicable.
Funding
This study was supported, in part, by the National
Natural Science Foundation of Jiangxi Province (grant no.
20151BAB205092) and the Key Researching and Developing Project of
Jiangxi Province (grant no. 20161BBG70125).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
QX and JHZ participated in the entire process of the
study. QX and HZ carried out the animal experiments. QHQ, BK and LD
helped to perform the in vitro experiments, JHZ performed
the statistics. ZMH and JR helped conduct the histomorphological
and immunohistochemical staining, and revised the manuscript. MD
designed and coordinated the study. All authors read and approved
the final manuscript.
Ethics approval and consent to
participate
All animal and patient procedures were approved by
the Ethics Committee of the First Affiliated Hospital of Chongqing
Medical University. Written informed consent was obtained from
adult patients and from the parents of adolescent patients
permitting the use of their samples.
Patient consent for publication
Written informed consent was obtained from adult
patients and from the parents of adolescent patients permitting the
publication of any associated data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Kepler CK, Ponnappan RK, Tannoury CA,
Risbud MV and Anderson DG: The molecular basis of intervertebral
disc degeneration. Spine J. 13:318–330. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Global Burden of Disease Study 2013
Collaborators: Global, regional, and national incidence,
prevalence, and years lived with disability for 301 acute and
chronic diseases and injuries in 188 countries, 1990–2013: A
systematic analysis for the Global Burden of Disease Study 2013.
Lancet. 386:743–800. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Vo N, Niedernhofer LJ, Nasto LA, Jacobs L,
Robbins PD, Kang J and Evans CH: An overview of underlying causes
and animal models for the study of age-related degenerative
disorders of the spine and synovial joints. J Orthop Res.
31:831–837. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Risbud MV and Shapiro IM: Shapiro, role of
cytokines in intervertebral disc degeneration: Pain and disc
content. Nat Rev Rheumatol. 10:44–56. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Freemont AJ: The cellular pathobiology of
the degenerate intervertebral disc and discogenic back pain.
Rheumatology (Oxford). 48:5–10. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ichikawa Y: Differentiation of a cell line
of myeloid leukemia. J Cell Physiol. 74:223–234. 1969. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Trouillas M, Saucourt C, Guillotin B,
Gauthereau X, Taupin JL, Moreau JF and Boeuf H: The LIF cytokine:
Towards adulthood. Eur Cytokine Netw. 20:51–62. 2009.PubMed/NCBI
|
|
8
|
Gearing DP, Comeau MR, Friend DJ, Gimpel
SD, Thut CJ, McGourty J, Brasher KK, King JA, Gillis S, Mosley B,
et al: The IL-6 signal transducer, gp130: An oncostatin M receptor
and affinity converter for the LIF receptor. Science.
255:1434–1437. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nicola NA and Babon JJ: Leukemia
inhibitory factor (LIF). Cytokine Growth Factor Rev. 26:533–544.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Carroll GJ and Bell MC: Leukaemia
inhibitory factor stimulates proteoglycan resorption in porcine
articular cartilage. Rheumatol Int. 13:5–8. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Naka T and Kishimoto T: Joint disease
caused by defective gp130-mediated STAT signaling. Arthritis Res.
4:154–156. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bell MC and Carroll GJ: Rheumatoid
synovial fluid contains bioactive leukemia inhibitory factor with
cartilage degrading activity-another target for chondroprotective
intervention. J Rheumatol. 27:332–338. 2000.PubMed/NCBI
|
|
13
|
Lotz M, Moats T and Villiger PM: Leukemia
inhibitory factor is expressed in cartilage and synovium and can
contribute to the pathogenesis of arthritis. J Clin Invest.
90:888–896. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lee CR, Sakai D, Nakai T, Toyama K,
Mochida J, Alini M and Grad S: A phenotypic comparison of
intervertebral disc and articular cartilage cells in the rat. Eur
Spine J. 16:2174–2185. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Masuda K, Aota Y, Muehleman C, Imai Y,
Okuma M, Thonar EJ, Andersson GB and An HS: A novel rabbit model of
mild, reproducible disc degeneration by an anulus needle puncture:
correlation between the degree of disc injury and radiological and
histological appearances of disc degeneration. Spine (Phila Pa
1976). 30:5–14. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang X, Hu Z, Hao J and Shen J: Low
intensity pulsed ultrasound promotes the extracellular matrix
synthesis of degenerative human nucleus pulposus cells through
FAK/PI3K/Akt pathway. Spine (Phila Pa 1976). 41:E248–E254. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Upadhyay A, Sharma G, Kivivuori S, Raye
WS, Zabihi E, Carroll GJ and Jazayeri JA: Role of a LIF antagonist
in LIF and OSM induced MMP-1, MMP-3, and TIMP-1 expression by
primary articular chondrocytes. Cytokine. 46:332–338. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ahadiat O, Higgins S, Ly A and Wysong A:
Toluidine blue stain of dermatofibrosarcoma protuberans:
Highlighting its use in mohs. Dermatol Surg. 43:1496–1498. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Han B, Zhu K, Li FC, Xiao YX, Feng J, Shi
ZL, Lin M, Wang J and Chen QX: A simple disc degeneration model
induced by percutaneous needle puncture in the rat tail. Spine
(Phila Pa 1976). 33:1925–1934. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Vaudreuil N, Kadow T, Yurube T, Hartman R,
Ngo K, Dong Q, Pohl P, Coelho JP, Kang J, Vo N and Sowa G: NSAID
use in intervertebral disc degeneration: What are the effects on
matrix homeostasis in vivo? Spine J. 17:1163–1170. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Daly C, Ghosh P, Jenkin G, Oehme D and
Goldschlager T: A review of animal models of intervertebral disc
degeneration: Pathophysiology, regeneration, and translation to the
clinic. Biomed Res Int 2016. 59521652016.
|
|
22
|
Liu H-T, Wang W-M, Lin Z-J, Chen G-X, Lin
G-Y and Li P-S: Alterations in imaging and histopathology after
aspiration of nucleus pulposus of rabbit lumbar intervertebral
disc. J Clin Rehabil Tissue Eng Res. 18:1313–1318. 2014.
|
|
23
|
Upadhyay A, Senyschyn D, Santos L, Gu R,
Carroll GJ and Jazayeri JA: K/BxN serum transfer arthritis is
delayed and less severe in leukaemia inhibitory factor
(LIF)-deficient mice. Clin Exp Immunol. 169:71–78. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jiang Y, Xiao Q, Hu Z, Pu B, Shu J, Yang
Q, Lao H and Hao J: Tissue levels of leukemia inhibitory factor
vary by osteoarthritis grade. Orthopedics. 37:e460–e464. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Adams MA and Roughley PJ: What is
intervertebral disc degeneration, and what causes it? Spine (Phila
Pa 1976). 31:2151–2161. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Videman T, Gibbons LE and Battié MC:
Age-and pathology-specific measures of disc degeneration. Spine
(Phila Pa 1976). 33:2781–2788. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Terry DE, Chopra RK, Ovenden J and
Anastassiades TP: Differential use of Alcian blue and toluidine
blue dyes for the quantification and isolation of anionic
glycoconjugates from cell cultures: Application to proteoglycans
and a high-molecular-weight glycoprotein synthesized by articular
chondrocytes. Anal Biochem. 285:211–219. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Silacci P, Dayer JM, Desgeorges A, Peter
R, Manueddu C and Guerne PA: Interleukin (IL)-6 and its soluble
receptor induce TIMP-1 expression in synoviocytes and chondrocytes,
and block IL-1-induced collagenolytic activity. J Biol Chem.
273:13625–13629. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tilg H, Dinarello CA and Mier JW: IL-6 and
APPs: Anti-inflammatory and immunosuppressive mediators. Immunol
Today. 18:428–432. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Nemoto O, Yamada H, Mukaida M and Shimmei
M: Stimulation of TIMP-1 production by oncostatin M in human
articular cartilage. Arthritis Rheum. 39:560–566. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Metcalf D: Leukemia inhibitory factor-a
puzzling polyfunctional regulator. Growth factors. 7:169–173. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wilks AF: Two putative protein-tyrosine
kinases identified by application of the polymerase chain reaction.
Proc Natl Acad Sci USA. 86:1603–1607. 1989. View Article : Google Scholar : PubMed/NCBI
|