Introduction
Osteosarcoma (OS) is the most common form of bone
malignancy in children and adults, and is characterized by high
mortality and a poor curative rate (1–3).
Neoadjuvant chemotherapy, surgical resection and limb preservation
surgery are the leading therapeutic strategies employed for the
treatment of OS (4). For
high-grade OS, the 5-year survival rate of OS patients without
cancer metastasis has been ≤60-70% due to effective treatments
(5,6). The survival rate of OS patients with
low-grade lesions is notably higher (7,8).
Unfortunately, the majority of patients with OS are diagnosed at
late stages and the prognosis of these patients remains poor
(7,8). Molecular targeted therapy has been
considered as a potential strategy for the treatment of OS
(9). Therefore, it is important to
identify the novel targets and the underlying molecular mechanisms
that drive the pathogenesis of OS.
MicroRNAs (miRNAs/miRs) are short noncoding RNAs
that regulate gene expression by binding with the 3′-untranslation
region (3′-UTR) of the targeted mRNAs, consequently inhibiting the
translation or inducing the degradation of mRNAs (10). Increasing evidence has demonstrated
the regulatory function of miRNAs in the initiation and progression
of OS (11,12). For instance, miR-449a is
downregulated in OS, which promotes the apoptosis of OS cells via
targeting B-cell lymphoma 2 (13).
In addition, miR-20a is overexpressed in OS, which induces the
proliferation of OS cells (14).
Previously, miR-185 was reported to suppress the growth of human
none small cell lung cancer by inducing cell cycle arrest (15). Additionally, miR-185 inhibits the
growth of hepatocellular carcinoma cells through the DNA
(cytosine-5)-methyltransferase 1 (DNMT1)/phosphatase and tensin
homolog/protein kinase B signaling pathway (16). It has been demonstrated that
miR-185 is a potential prognostic biomarker for early stage
hepatocellular carcinoma (17).
Interestingly, previous studies have revealed that miR-185
regulated the invasion, metastasis and radioresistance of
nasopharyngeal carcinoma by targeting Wnt family member 2B in
vitro (18,19). These studies indicate the important
roles of miR-185 in numerous types of cancer; however, the function
of miR-185 in OS remains unclear.
Aerobic glycolysis is considered as the hallmark of
cancer cells, which regulates the metabolism of glucose into
lactate as opposed to the mitochondrial oxidative phosphorylation
pathway (20). The glucose
transporters are responsible for the translocation of glucose
across the plasma membrane. The metabolism of glucose in cancer
cells is catalyzed by a variety of enzymes (21,22).
During glycolysis, hexokinase 2 (HK2) is the first rate-limiting
enzyme, which regulates the conversion of glucose into
glucose-6-phosphate (23–28). These findings indicate that the
expression of HK2 is associated with the glucose metabolism of
cancer cells. Previous studies have demonstrated upregulation of
HK2 in various cancers, which promotes the glucose consumption of
cancer cells (29–32). Interestingly, HK2 has been reported
to be the target of miRNAs in cancers (33–36).
For instance, miR-181b downregulates HK2, consequently suppressing
the glycolysis and proliferation of gastric cancer cells (36). miR-143 was reported to be a tumor
suppressor in prostate cancer via targeting HK2 (35). In OS, miR-125b inhibits the aerobic
glycolysis of OS cells by downregulating the expression of HK2
(37). These findings indicate
that inhibition of HK2 may be a promising strategy for the
treatment of cancer.
In the present study, the expression of miR-185 in
OS tissues and cells was evaluated. Further molecular study
revealed that miR-185 inhibited the expression of HK2 in OS cells.
These findings suggested the potential regulatory function of
miR-185 in OS.
Materials and methods
Cell culture
The normal human osteoplastic cell line (NHOst) and
OS cell lines HOS, U2OS, Saos-2 and MG-63 were purchased from the
American Type Culture Collection. Cells were cultured in Dulbecco's
Modified Eagle's medium (DMEM; Gibco; Thermo Fisher Scientific,
Inc.) supplemented with 10% fetal bovine serum, 100 U/ml penicillin
(Invitrogen; Thermo Fisher Scientific, Inc.) and 100 µg/ml of
streptomycin (Invitrogen; Thermo Fisher Scientific, Inc.) at 37°C
in a humidified incubator containing 5% CO2.
Clinical samples
A total of 30 paired OS tissues and adjacent normal
tissues (0.5–1 cm in diameter) were collected from the metaphyseal
regions of long bones of the OS patients (age, 8–45 years old;
female:male=1.35:1) at The Central Hospital of Chao Zhou between
April 2015 and October 2016. The adjacent normal tissues were ≥5 cm
away from the edge of the OS tissues. The exclusion criteria of the
patients included: i) Received chemo- or radiotherapy prior to
surgery; ii) patients were unsuitable for surgery; iii) Informed
consent was not obtained; and iv) serious infection. The basic
clinical characteristics of these patients were provided in
Table I. The samples were
snap-frozen in liquid nitrogen and stored at −80°C. Written
informed consent was obtained from all the patients. Tissues were
staged according to the Vanderbilt system (38). The procedures for tissue collection
and the following experiments were approved by the Ethics Committee
of The Third Affiliated Hospital of Southern Medical
University.
 | Table I.Clinical parameters of the
osteosarcoma patients involved in this study. |
Table I.
Clinical parameters of the
osteosarcoma patients involved in this study.
| Clinical
characteristics | Number |
|---|
| Age (years) |
|
|
≤50 | 10 |
|
>50 | 20 |
| Sex |
|
|
Male | 17 |
|
Female | 13 |
| Tumor size
(cm) |
|
| ≤5 | 11 |
|
>5 | 19 |
|
Differentiation |
|
|
Moderate | 15 |
|
Poor | 15 |
| TNM stage |
|
|
I–II | 18 |
|
III–IV | 12 |
| Lymph node
metastasis |
|
| N0 | 16 |
|
N1-3 | 14 |
miRNA transfection
miR-185 mimics (5′-UGGAGAGAAAGGCAGUUCCUGA), control
miRNA (5′-GGUUCGUACGUACACUGUUCA-3′), miR-185 antagomir
(5′-UCAGGAACUGCCUUUCUCUCCA-3′) and negative control miRNA
(5′-CGGUACGAUCGCGGCGGGAUAUC-3′) were synthesized by Sangon Biotech
Co., Ltd. For miRNA transfection, both U2OS and Saos-2 cells
(10,000 cells per well) were seeded on 6-well plates with DMEM and
20 nm miRNAs were transfected into the cells using
Lipofectamine® 3000 (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocols. At 36
h post-transfection, the expression levels of miR-185 were
determined by reverse transcription-quantitative polymerase chain
reaction (RT-qPCR). The HA-HK2 transfection was also performed
using Lipofectamine® 3000 (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocols.
RNA extraction and RT-qPCR
Total RNA was extracted from the tissues or cells
using an RNeasy Mini kit (cat. no. 74104; Qiagen GmbH) according to
the manufacturer's protocols. The concentration of RNA was
determined using a NanoDrop 2000 spectrophotometer (NanoDrop
Technologies; Thermo Fisher Scientific, Inc). Extracted RNA (1 µg)
was reverse transcribed using the Prime-Script RT kit (Takara
Biotechnology Co., Ltd., Dalian, China) according to the
manufacturer's protocols. qPCR was performed using the miScript PCR
system (Qiagen GmbH) according to the manufacturer's instructions.
U6 RNA and GAPDH were used to normalize the expression of miR-185
and HK2, respectively. The PCR conditions were as follows: 95°C for
10 min, followed by 40 cycles of 95°C for 10 sec and 58°C for 60
sec. The primer sequences used in this study were synthesized as:
MiR-185 forward, 5′-CAATGGAGAGAAAGGCAGTTCC-3′ and reverse,
5′-AATCCATGAGAGATCCCTACCG-3′; U6 forward, 5′-CTCGCTTCGGCAGCACA-3′
and reverse, 5′-AACGCTTCACGAATTTGCGT-3′; HK2 forward,
5′-ATGATCGCCTGCTTATTCACG-3′ and reverse,
5′-CGCCTAGAAATCTCCAGAAGGG-3′; actin forward,
5′-GTGACGTTGACATCCGTAAAGA-3′ and reverse,
5′-GCCGGACTCATCGTACTCC-3′. The experiments were performed with
three independent repeats.
Cell proliferation assay
The proliferation of OS cells transfected with
miR-185 mimics or control miRNA was determined using Cell Titer
96® Aqueous Cell Proliferation Assay kit (Promega
Corporation) according to the manufacturer's protocols. Briefly,
transfected OS cells (1,000 cells per well) were seeded in 96-well
plates with DMEM. Cell proliferation was evaluated by adding MTT
reagent to the medium. Following the incubation at 37°C for 3 h,
the absorbance at 480 nm at day 1, 2, 3, 4 and 5 was determined
using a microplate reader.
Luciferase reporter assay
Wild-type (WT) or mutant 3′-UTR of HK2 containing
the putative miR-185 binding sites were amplified and inserted into
pMIR-Luciferase-Reporter vectors (Promega Corporation) according to
the manufacturer's protocols. The plasmids were transfected into OS
cells in the presence of miR-185 mimics or control miRNA using
Lipofectamine 3000. At 48 h post-transfection, luciferase activity
was determined using Dual-GLO Luciferase Assay System (Promega
Corporation) according to the manufacturer's protocols. The
activity of Renilla was detected as the normalization. The
experiments were performed in triplicate.
Western blotting
Following transfection for 48 h, total proteins of
cells were extracted using radioimmunoprecipitation lysis buffer
(Beyotime Institute of Biotechnology) and the protein concentration
was determined using a BCA Assay kit (Pierce; Thermo Fisher
Scientific, Inc.). Equal amounts of protein (20 µg) were separated
using 15% SDS-PAGE and then transferred onto polyvinylidene
difluoride membranes (EMD Millipore). The membrane was blocked with
5% non-fat milk for 1 h at room temperature and incubated with
antibodies against HK2 (1:2,000; cat. no. GTX124375; GeneTex) at
room temperature for 2 h. The goat anti rabbit secondary antibody
conjugated with horseradish peroxidase (1:5,000; cat. no. 305005;
Bio-Rad Laboratories, Inc.) was then applied for 1 h at room
temperature. The bands were visualized using an enhanced
chemiluminescence protein detection kit (EMD Millipore) and exposed
using X-ray film (Kodak, Inc.). β-actin was used as the loading
control (1:3,000; cat. no. AC004; ABclonal Biotech Co., Ltd.).
Determination of glucose consumption
and lactate production
To evaluate glucose uptake and lactate production,
OS cells (10,000 cells) were seeded in the 6-well plates with DMEM
and transfected with miRNAs. At 48 h post-transfection, the culture
medium was collected. The glucose consumption and lactate
production were determined using an Amplex® Red
Glucose/Glucose oxidase Assay kit (Invitrogen; Thermo Fisher
Scientific, Inc.) and lactate assay kit (Sigma-Aldrich; Merck KGaA)
according to the manufacturer's protocols.
Colony formation
OS cells were transfected with miR-185 mimics or
control miRNA and seed in the 6-well plates with the density of
1,000 cells per well. Cells were maintained in DMEM containing 10%
FBS. Following culture for 10 days, colonies were washed with PBS
and stained with 0.1% crystal violet (Beyotime Institute of
Biotechnology) for 15 min at room temperature. The number of
colonies was counted with the light microscopy (magnification,
×100; Olympus Corporation).
Wound-healing assay
OS cells transfected with miR-185 mimics or control
miRNA were seeded in a 6-well plate (~10,000 cells per well).
Following culture for 24 h, cells were wounded with a 200 µl
pipette tip and the cell debris was removed by washing with PBS.
Cells were cultured with fresh DMEM for 24 h at 37°C. Subsequently,
the migration of the cells was observed using a light microscope
(Olympus Corporation; magnification, ×40).
Statistical analysis
The data were presented as the mean + standard
deviation from three independent repeats and analyzed with the
GraphPad Prism software (Version 5.0; GraphPad Software, Inc.).
Statistical comparisons between two groups were performed using a
Student's t-test. Comparisons between multiple groups were
performed using a one-way analysis of variance followed by a
Newman-Keuls post-hoc comparison. The correlation between the
expression of miR-185 and HK2 was analyzed with the Spearman's
correlation analysis P<0.05 was considered to indicate a
statistically significant difference.
Results
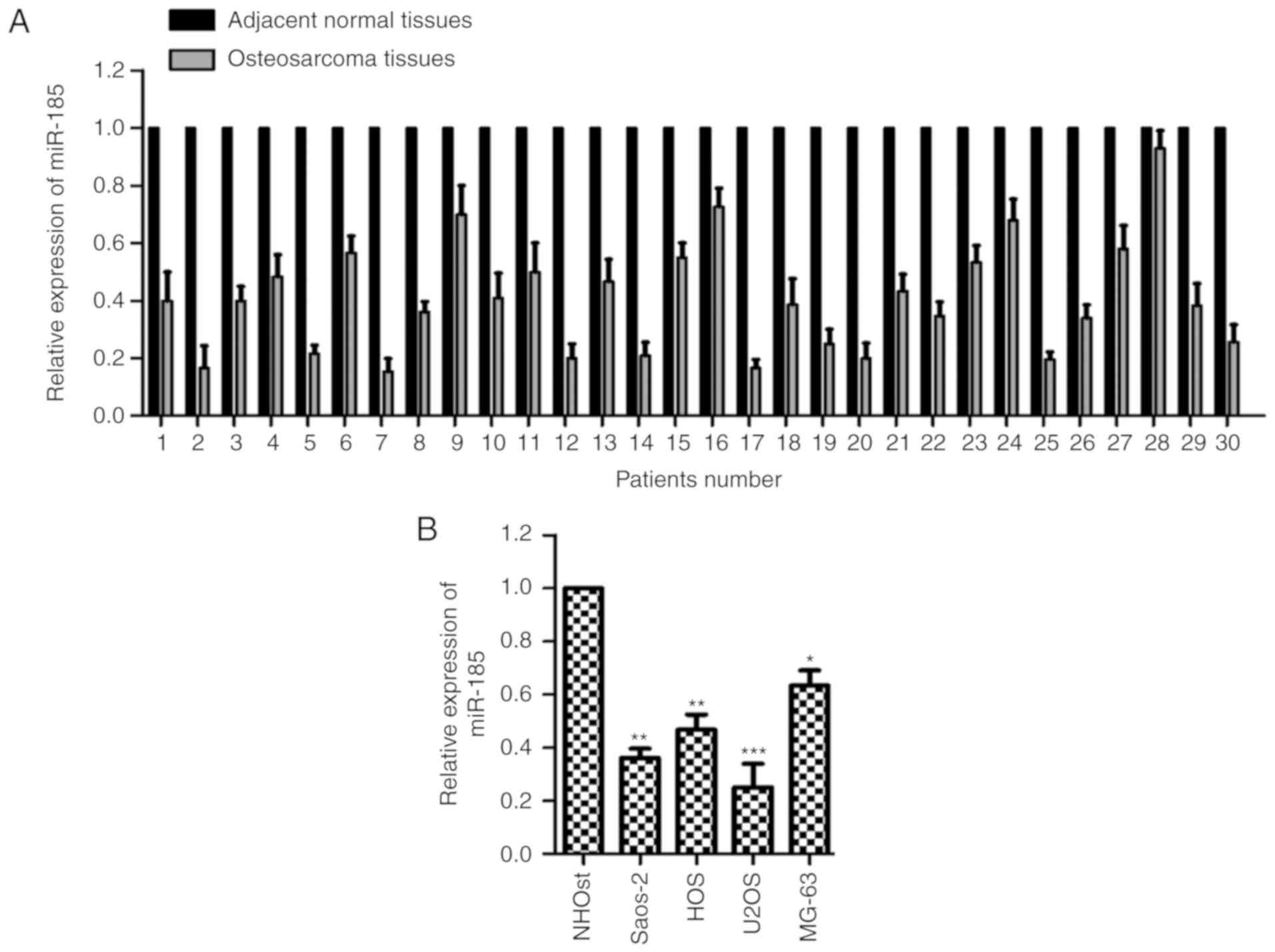
miR-185 is downregulated in OS tissues
and cells
To characterize the potential function of miR-185 in
OS, the expression of miR-185 was determined in paired OS and
adjacent normal tissues. The expression levels of miR-185 were
notably reduced in OS tissues compared with the adjacent
noncancerous tissues (Fig. 1A).
Additionally, the expression of miR-185 was significantly
downregulated in OS cells compared with normal cells (P<0.05;
Fig. 1B). These data revealed the
downregulated expression of miR-185 in OS. The relative expression
levels of miR-185 in U2OS and Saos-2 cells were the lowest; thus,
these two cell lines were selected for further cytological study to
overexpress miR-185.
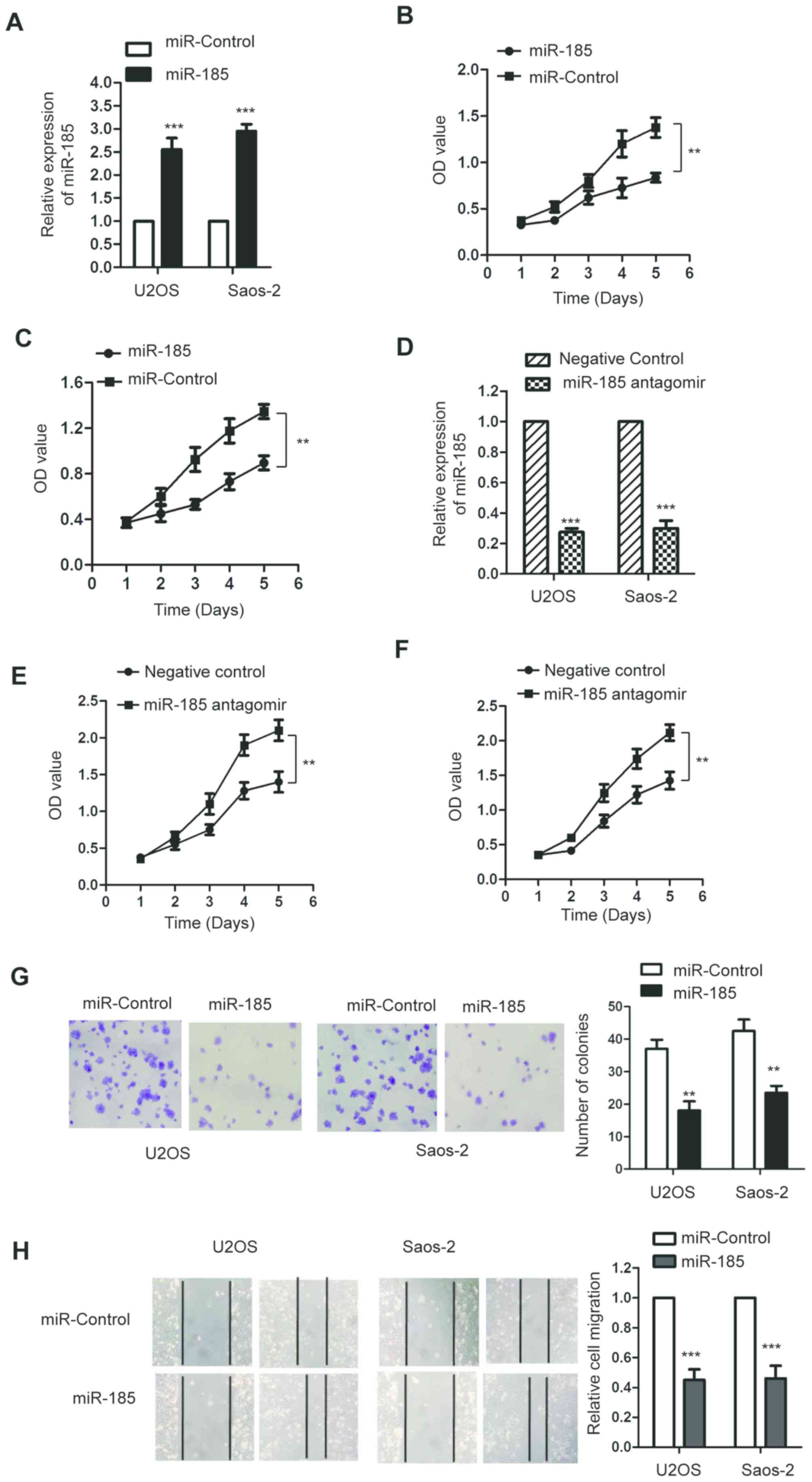
miR-185 suppresses the proliferation
of OS cells
To investigate the function of miR-185 in the
progression of OS, both U2OS and Saos-2 cells were transfected with
miR-185 mimics or the control miRNA. The expression levels of
miR-185 were significantly upregulated following the transfection
of miR-185 in U2OS and Saos-2 cells compared with the control
(P<0.05; Fig. 2A). To determine
the effect of miR-185 on the proliferation of OS cells, an MTT
assay was performed and the results revealed that miR-185
overexpression significantly inhibited the growth of OS cells
compared with the control (P<0.05; Fig. 2B and C). To further investigate the
regulatory function of miR-185 on the growth of OS cells, U2OS and
Saos-2 cells were transfected with miR-185 antagomir or negative
control. Transfection of miR-185 antagomir significantly
downregulated the expression of miR-185 (P<0.05; Fig. 2D). Furthermore, the results of MTT
assay revealed that inhibition of miR-185 significantly promoted
the proliferation of U2OS and Saos-2 cells compared with the
control (Fig. 2E and F).
Consistent with these results, the results of colony formation
assay also indicated that miR-185 upregulation inhibited the colony
formation ability of U2OS and Saos-2 cells compared with the
control (Fig. 2G). To further
investigate the effect of miR-185 on the migration of OS cells, a
wound-healing assay was performed using U2OS and Saos-2 cells
transfected with miR-185 mimics or control miRNA. Overexpression of
miR-185 in U2OS and Saos-2 cells significantly inhibited the
migration ability compared with the control group (P<0.05;
Fig. 2H). These findings indicated
that miR-185 could inhibit the growth of OS cells.
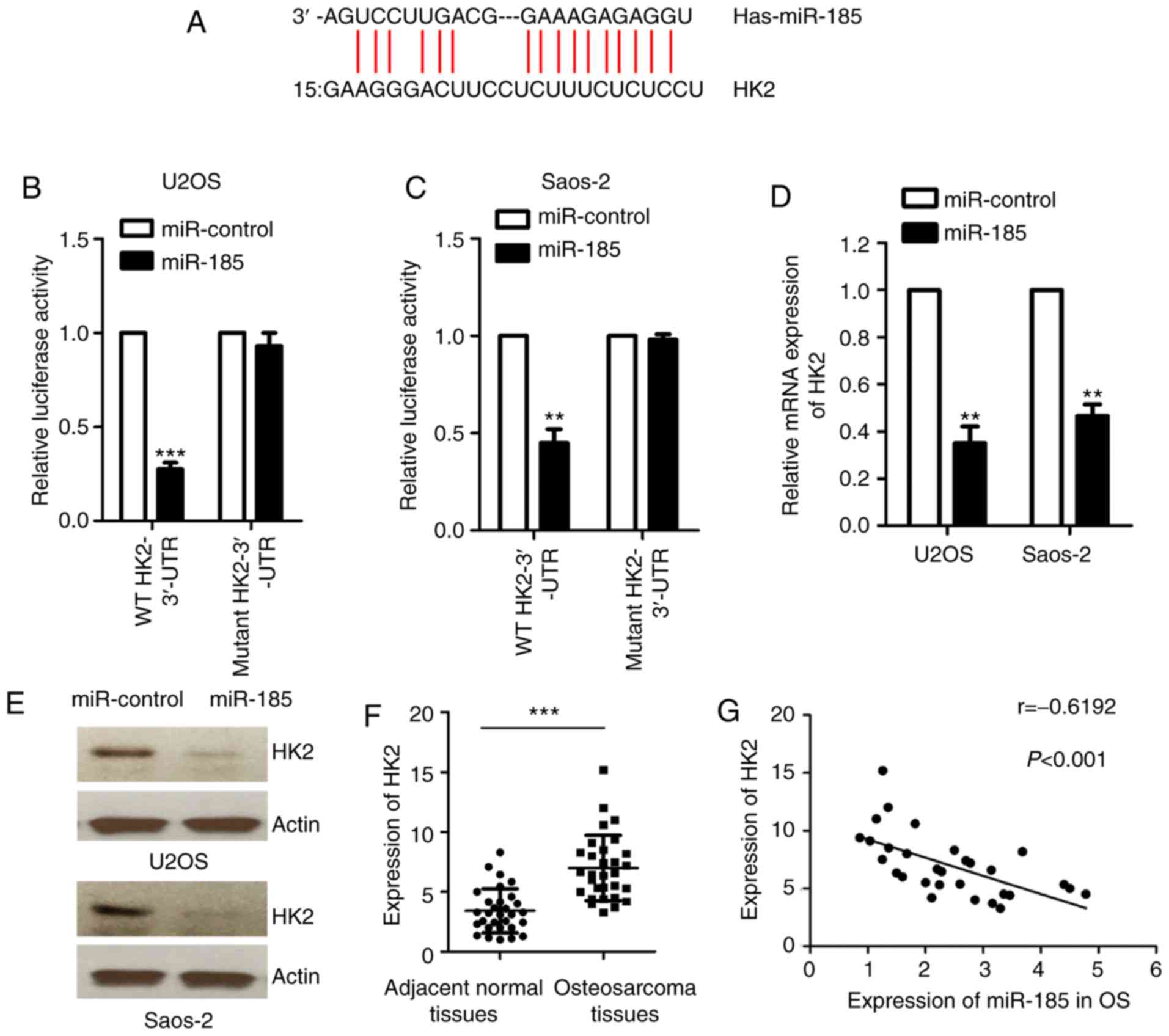
miR-185 downregulates the expression
of HK2 by binding to the 3′-UTR of HK2
To further investigate the regulatory function of
miR-185 in OS, the targets of miR-185 were identified using
bioinformatic analysis (http://34.236.212.39/microrna/getMirnaForm.do). HK2
was predicted to be a potential target of miR-185. The putative
binding sequence of miR-185 at the 3′-UTR of HK2 was indicated in
Fig. 3A. To confirm this potential
binding interaction, a luciferase reporter assay was performed
using a luciferase vector containing the WT or mutant 3′-UTR of HK2
in the presence of miR-185 mimics or control miRNA. The results
revealed that miR-185 significantly inhibited the luciferase
activity of WT but not the mutant 3′-UTR of HK2 (P<0.05;
Fig. 3B and C). Furthermore, the
mRNA and protein levels of HK2 were also determined in U2OS and
Saos-2 cells transfected with miR-185 mimics or control miRNA.
Ectopic expression of miR-185 significantly downregulated the mRNA
levels of HK2 in U2OS and Saos-2 cells compared with the control
(P<0.05; Fig. 3D). Consistent
with these results, the protein levels of HK2 were also reduced in
OS cells overexpressing miR-185 (Fig.
3E). These results demonstrated that miR-185 inhibited the
expression of HK2 in OS cells. To confirm the association between
the expression of miR-185 and HK2, the mRNA levels of HK2 in OS
tissues were determined using RT-qPCR compared with adjacent normal
tissues. The data revealed that the expression of HK2 was
significantly upregulated in OS tissues compared with the control
(P<0.05; Fig. 3F). The results
of Spearman's correlation analysis indicated that the expression
levels of miR-185 and HK2 in OS tissues were inversely correlated
(Fig. 3G). These results
demonstrated that HK2 could be a potential downstream target of
miR-185 in OS cells.
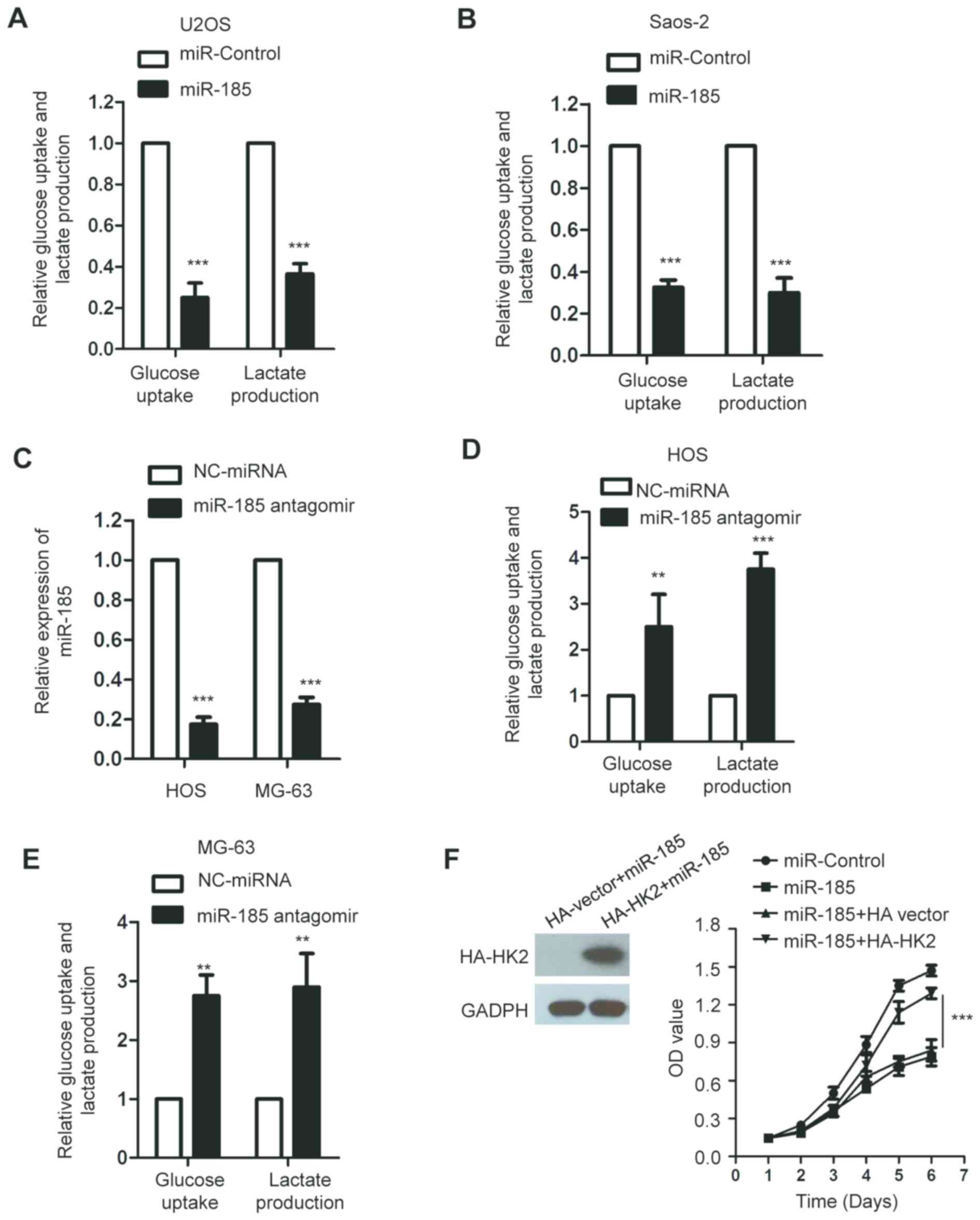
miR-185 inhibits the glucose
metabolism of OS cells
HK2 serves important roles in the glycolysis of
cancer cells (39). As miR-185
downregulated the expression of HK2, we hypothesized that miR-185
could regulate the glycolysis of OS cells. To confirm this, the
glucose consumption and lactate production of U2OS and Saos-2 cells
transfected with miR-185 or control miRNA were determined. The data
revealed that overexpression of miR-185 significantly suppressed
glucose uptake and lactate production of OS cells compared with the
control (P<0.05; Fig. 4A and
B). To further investigate the inhibitory effect of miR-185 on
the glycolysis of OS cells, the endogenous expression of miR-185
was abolished by transfecting miR-185 antagomir into HOS and MG-63
cells, which express miR-185 at a relatively higher level among the
OS cell lines we used. The results revealed that miR-185 was
significantly downregulated in OS cells transfected with miR-185
antagomir (P<0.05; Fig. 4C). In
addition, inhibition of miR-185 significantly promoted the
glycolysis of HOS and MG-63 cells compared with the control
(Fig. 4D and E). To determine
whether miR-185 inhibited the growth of OS cells through
downregulating HK2, the expression of HK2 was restored by
transfecting U2OS cells with HA-HK2. The ectopic expression of HK2
suppressed the inhibitory effect of miR-185 on the growth of OS
cells (Fig. 4F).
Discussion
The regulatory functions of miRNAs in the
progression of cancers have been highlighted in previous studies
(40–42). miR-185 was reported to function as
a tumor suppressor in numerous types of cancer, including
hepatocellular carcinoma and non-small cell lung cancer (15,16).
In the present study, the expression levels of miR-185 in OS were
evaluated, and the downstream targets of miR-185 were identified.
The results indicated that miR-185 was significantly downregulated
in OS tissues and cells. Ectopic overexpression of miR-185
suppressed the growth and migration of OS cells. These findings
suggested the potential tumor suppressive role of miR-185 in
OS.
To identify the underlying molecular mechanisms of
miR-185-mediated inhibition in the proliferation of OS cells, the
potential targets of miR-185 were predicted. A previous study
identified AKT1 as one of the targets of miR-185 in non-small cell
lung cancer (43). The potential
tumor suppressive function of miR-185 through the E2F1 and DNMT1
pathways was also revealed in triple-negative breast cancer
(44). Furthermore, recent studies
reported that miR-185 targeted WNT2B to regulate the invasion and
metastasis of nasopharyngeal carcinoma (18,19).
Additionally, miR-185 suppressed the proliferation of pancreatic
cells by targeting a transcriptional co-activator with a
PDZ-binding motif (45). miR-185
was also reported to inhibit the Wnt/β-catenin pathway in
colorectal cancer (46).
In the present study, HK2 was revealed to be a
direct target of miR-185 in OS cells. Overexpressed miR-185
suppressed the mRNA and protein levels of HK2. In addition, an
inverse correlation between the expression of miR-185 and HK2 was
also observed in OS tissues. HK2 controls the first step of
glycolysis, consequently inducing the ATP-dependent phosphorylation
of glucose into glucose-6-phasphatase (47). Upregulated HK2 has been detected in
cancer cells, and was associated with poor prognosis of patients
(48,49). HK2 was determined to be involved in
the initiation and development of Kras-driven lung cancer and
ErbB2-driven breast cancer in HK2 conditional knockout mice
(50). In addition, previous
studies suggested that HK2 was a target of numerous miRNAs in
cancers, which could be associated with cancer progression
(35,36). Furthermore, recent findings
indicated that miR-155 inhibited HK2-mediated glycolysis and
sensitized lung cancer cells to irradiation (51). HK2 was also revealed as a target of
miR-143 in prostate cancer (35).
These results indicated that HK2 was regulated by miRNAs in
numerous types of cancer and consequently regulated the progression
of tumor. In the present study, miR-185 overexpression suppressed
the glucose consumption and lactate production of OS cells, while
restoration of HK2 rescued the growth of OS cells. As HK2 is
involved in aerobic glycolysis, these results suggested that
miR-185 could regulate the proliferation of OS cells in a
HK2-dependent manner.
Due to the tumor suppressive role of miR-185 in OS,
it might be interesting to identify the upstream regulators of
miR-185. Accumulating evidence suggested that some small molecules
or natural compounds, such as docosahexaenoic acid, resveratrol,
curcumin, regulated the expression of miRNAs (52). Further studies could be conducted
to identify the molecules that regulate the expression of miR-185
in OS. Additionally, to further characterize the potential tumor
suppressive function of miR-185 in OS, in vivo experiments
may also be performed. Additionally, as numerous downstream targets
of miR-185 have been identified in a variety of cancers (43,44,53–55),
the regulatory function of miR-185 on its potential targets in OS
could be explored.
In conclusion, the results of the present study
demonstrated that miR-185 inhibited the proliferation and
glycolysis of OS cells through targeting HK2. The suppressive
function of miR-185 on the growth of OS cells indicated that
miR-185 may be a novel therapeutic candidate for the treatment of
OS in the future.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
CL and HL designed the study. CL performed the
experiments. LC performed the luciferase assay. HL wrote the
manuscript.
Ethics approval and consent to
participate
Written informed consent was provided by all the
patients enrolled in this study. The procedures for tissue
collection and the following experiments were approved by the
Ethics Committee of The Third Affiliated Hospital of Southern
Medical University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Friebele JC, Peck J, Pan X, Abdel-Rasoul M
and Mayerson JL: Osteosarcoma: A Meta-analysis and review of the
literature. Am J Orthop (Belle Mead NJ). 44:547–553.
2015.PubMed/NCBI
|
|
2
|
Abarrategi A, Tornin J, Martinez-Cruzado
L, Hamilton A, Martinez-Campos E, Rodrigo JP, González MV, Baldini
N, Garcia-Castro J and Rodriguez R: Osteosarcoma: Cells-of-Origin,
cancer stem cells, and targeted therapies. Stem Cells Int.
2016:36317642016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Brown HK, Tellez-Gabriel M and Heymann D:
Cancer stem cells in osteosarcoma. Cancer Lett. 386:189–195. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Moore DD and Luu HH: Osteosarcoma. Cancer
Treat Res. 162:65–92. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ottaviani G and Jaffe N: The epidemiology
of osteosarcoma. Cancer Treat Res. 152:3–13. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bernthal NM, Federman N, Eilber FR, Nelson
SD, Eckardt JJ, Eilber FC and Tap WD: Long-term results (>25
years) of a randomized, prospective clinical trial evaluating
chemotherapy in patients with high-grade, operable osteosarcoma.
Cancer. 118:5888–5893. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Luetke A, Meyers PA, Lewis I and Juergens
H: Osteosarcoma treatment-where do we stand? A state of the art
review. Cancer Treat Rev. 40:523–532. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Geller DS and Gorlick R: Osteosarcoma: A
review of diagnosis, management, and treatment strategies. Clin Adv
Hematol Oncol. 8:705–718. 2010.PubMed/NCBI
|
|
9
|
Zhou W, Hao M, Du X, Chen K, Wang G and
Yang J: Advances in targeted therapy for osteosarcoma. Discov Med.
17:301–307. 2014.PubMed/NCBI
|
|
10
|
Cai Y, Yu X, Hu S and Yu J: A brief review
on the mechanisms of miRNA regulation. Genomics Proteomics
Bioinformatics. 7:147–154. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chang L, Shrestha S, LaChaud G, Scott MA
and James AW: Review of microRNA in osteosarcoma and
chondrosarcoma. Med Oncol. 32:6132015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sampson VB, Yoo S, Kumar A, Vetter NS and
Kolb EA: MicroRNAs and Potential Targets in Osteosarcoma: Review.
Front Pediatr. 3:692015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chen J, Zhou J, Chen X, Yang B, Wang D,
Yang P, He X and Li H: miRNA-449a is downregulated in osteosarcoma
and promotes cell apoptosis by targeting BCL2. Tumour Biol.
36:8221–8229. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yuan G, Zhao Y, Wu D, Gao C and Jiao Z:
miRNA-20a upregulates TAK1 and increases proliferation in
osteosarcoma cells. Future Oncol. 14:461–469. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Takahashi Y, Forrest AR, Maeno E,
Hashimoto T, Daub CO and Yasuda J: MiR-107 and MiR-185 can induce
cell cycle arrest in human non small cell lung cancer cell lines.
PLoS One. 4:e66772009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Qadir XV, Han C, Lu D, Zhang J and Wu T:
miR-185 inhibits hepatocellular carcinoma growth by targeting the
DNMT1/PTEN/Akt pathway. Am J Pathol. 184:2355–2364. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhi Q, Zhu J, Guo X, He S, Xue X, Zhou J,
Hu B, Li H, Chen S, Zhao H and Kuang Y: Metastasis-related miR-185
is a potential prognostic biomarker for hepatocellular carcinoma in
early stage. Biomed Pharmacother. 67:393–398. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu C, Li G, Ren S, Su Z, Wang Y, Tian Y,
Liu Y and Qiu Y: miR-185-3p regulates the invasion and metastasis
of nasopharyngeal carcinoma by targeting WNT2B in vitro.
Oncol Lett. 13:2631–2636. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li G, Wang Y, Liu Y, Su Z, Liu C, Ren S,
Deng T, Huang D, Tian Y and Qiu Y: miR-185-3p regulates
nasopharyngeal carcinoma radioresistance by targeting WNT2B in
vitro. Cancer Sci. 105:1560–1568. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Akram M: Mini-review on glycolysis and
cancer. J Cancer Educ. 28:454–457. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li XB, Gu JD and Zhou QH: Review of
aerobic glycolysis and its key enzymes-new targets for lung cancer
therapy. Thorac Cancer. 6:17–24. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zheng J: Energy metabolism of cancer:
Glycolysis versus oxidative phosphorylation (Review). Oncol Lett.
4:1151–1157. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Coelho RG, Calaca IC, Celestrini DM,
Correia-Carneiro AH, Costa MM, Zancan P and Sola-Penna M:
Hexokinase and phosphofructokinase activity and intracellular
distribution correlate with aggressiveness and invasiveness of
human breast carcinoma. Oncotarget. 6:29375–29387. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Dai W, Wang F, Lu J, Xia Y, He L, Chen K,
Li J, Li S, Liu T, Zheng Y, et al: By reducing hexokinase 2,
resveratrol induces apoptosis in HCC cells addicted to aerobic
glycolysis and inhibits tumor growth in mice. Oncotarget.
6:13703–13717. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Deng Y and Lu J: Targeting hexokinase 2 in
castration-resistant prostate cancer. Mol Cell Oncol.
2:e9744652015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lu CL, Qin L, Liu HC, Candas D, Fan M and
Li JJ: Tumor cells switch to mitochondrial oxidative
phosphorylation under radiation via mTOR-mediated hexokinase II
inhibition-a Warburg-reversing effect. PLoS One. 10:e01210462015.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Viticchie G, Agostini M, Lena AM, Mancini
M, Zhou H, Zolla L, Dinsdale D, Saintigny G, Melino G and Candi E:
p63 supports aerobic respiration through hexokinase II. Proc Natl
Acad Sci USA. 112:11577–11582. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhou Y, Lu N, Qiao C, Ni T, Li Z, Yu B,
Guo Q and Wei L: FV-429 induces apoptosis and inhibits glycolysis
by inhibiting Akt-mediated phosphorylation of hexokinase II in
MDA-MB-231 cells. Mol Carcinog. 55:1317–1328. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Katabi MM, Chan HL, Karp SE and Batist G:
Hexokinase type II: A novel tumor-specific promoter for
gene-targeted therapy differentially expressed and regulated in
human cancer cells. Hum Gene Ther. 10:155–164. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Brown RS, Goodman TM, Zasadny KR, Greenson
JK and Wahl RL: Expression of hexokinase II and Glut-1 in untreated
human breast cancer. Nucl Med Biol. 29:443–453. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Goel A, Mathupala SP and Pedersen PL:
Glucose metabolism in cancer. Evidence that demethylation events
play a role in activating type II hexokinase gene expression. J
Biol Chem. 278:15333–15340. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jin Z, Gu J, Xin X, Li Y and Wang H:
Expression of hexokinase 2 in epithelial ovarian tumors and its
clinical significance in serous ovarian cancer. Eur J Gynaecol
Onco. 35:519–524. 2014.
|
|
33
|
Peschiaroli A, Giacobbe A, Formosa A,
Markert EK, Bongiorno-Borbone L, Levine AJ, Candi E, D'Alessandro
A, Zolla L, Finazzi Agrò A and Melino G: miR-143 regulates
hexokinase 2 expression in cancer cells. Oncogene. 32:797–802.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Guo W, Qiu Z, Wang Z, Wang Q, Tan N, Chen
T, Chen Z, Huang S, Gu J, Li J, et al: MiR-199a-5p is negatively
associated with malignancies and regulates glycolysis and lactate
production by targeting hexokinase 2 in liver cancer. Hepatology.
62:1132–1144. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhou P, Chen WG and Li XW: MicroRNA-143
acts as a tumor suppressor by targeting hexokinase 2 in human
prostate cancer. Am J Cancer Res. 5:2056–2063. 2015.PubMed/NCBI
|
|
36
|
Li LQ, Yang Y, Chen H, Zhang L, Pan D and
Xie WJ: MicroRNA-181b inhibits glycolysis in gastric cancer cells
via targeting hexokinase 2 gene. Cancer Biomark. 17:75–81. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wu Y, He H, Wu B, Wen J, Guo Z, Luo Y and
Cao G: miR-125b suppresses the aerobic glycolysis of osteosarcoma
HOS cells by downregulating the expression of hexokinase-2. Xi Bao
Yu Fen Zi Mian Yi Xue Za Zhi. 33:1365–1370. 2017.(In Chinese).
PubMed/NCBI
|
|
38
|
Cates JMM: Simple staging system for
osteosarcoma performs equivalently to the AJCC and MSTS systems. J
Orthop Res. 36:2802–2808. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Garcia SN, Guedes RC and Marques MM:
Unlocking the potential of HK2 in cancer metabolism and
therapeutics. Curr Med Chem. Dec 12–2018.doi:
10.2174/0929867326666181213092652 (Epub ahead of print). View Article : Google Scholar
|
|
40
|
Gentilin E, Degli Uberti E and Zatelli MC:
Strategies to use microRNAs as therapeutic targets. Best Pract Res
Clin Endocrinol Metab. 30:629–639. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Farazi TA, Spitzer JI, Morozov P and
Tuschl T: miRNAs in human cancer. J Pathol. 223:102–115. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Kwak PB, Iwasaki S and Tomari Y: The
microRNA pathway and cancer. Cancer Sci. 101:2309–2315. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Li S, Ma Y, Hou X, Liu Y, Li K, Xu S and
Wang J: MiR-185 acts as a tumor suppressor by targeting AKT1 in
non-small cell lung cancer cells. Int J Clin Exp Pathol.
8:11854–11862. 2015.PubMed/NCBI
|
|
44
|
Tang H, Liu P, Yang L and Xie X, Ye F, Wu
M, Liu X, Chen B, Zhang L and Xie X: miR-185 suppresses tumor
proliferation by directly targeting E2F6 and DNMT1 and indirectly
upregulating BRCA1 in triple-negative breast cancer. Mol Cancer
Ther. 13:3185–3197. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Xia D, Li X, Niu Q, Liu X, Xu W, Ma C, Gu
H, Liu Z, Shi L, Tian X, et al: MicroRNA-185 suppresses pancreatic
cell proliferation by targeting transcriptional coactivator with
PDZ-binding motif in pancreatic cancer. Exp Ther Med. 15:657–666.
2018.PubMed/NCBI
|
|
46
|
Dong-Xu W, Jia L and Su-Juan Z:
MicroRNA-185 is a novel tumor suppressor by negatively modulating
the Wnt/β-catenin pathway in human colorectal cancer. Indian J
Cancer. 52 (Suppl 3):E182–E185. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wilson JE: Isozymes of mammalian
hexokinase: Structure, subcellular localization and metabolic
function. J Exp Biol. 206:2049–2057. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Anderson M, Marayati R, Moffitt R and Yeh
JJ: Hexokinase 2 promotes tumor growth and metastasis by regulating
lactate production in pancreatic cancer. Oncotarget. 8:56081–56094.
2016.PubMed/NCBI
|
|
49
|
Katagiri M, Karasawa H, Takagi K, Nakayama
S, Yabuuchi S, Fujishima F, Naitoh T, Watanabe M, Suzuki T, Unno M
and Sasano H: Hexokinase 2 in colorectal cancer: A potent
prognostic factor associated with glycolysis, proliferation and
migration. Histol Histopathol. 32:351–360. 2017.PubMed/NCBI
|
|
50
|
Patra KC, Wang Q, Bhaskar PT, Miller L,
Wang Z, Wheaton W, Chandel N, Laakso M, Muller WJ, Allen EL, et al:
Hexokinase 2 is required for tumor initiation and maintenance and
its systemic deletion is therapeutic in mouse models of cancer.
Cancer Cell. 24:213–228. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Lv X, Yao L, Zhang J, Han P and Li C:
Inhibition of microRNA-155 sensitizes lung cancer cells to
irradiation via suppression of HK2-modulated glucose metabolism.
Mol Med Rep. 14:1332–1338. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Lin Q, Ma L, Liu Z, Yang Z, Wang J, Liu J
and Jiang G: Targeting microRNAs: A new action mechanism of natural
compounds. Oncotarget. 8:15961–15970. 2017.PubMed/NCBI
|
|
53
|
Zhao L, Zhang Y, Liu J, Yin W, Jin D, Wang
D and Zhang W: MiR-185 inhibits cell proliferation and invasion of
non-small cell lung cancer by targeting KLF7. Oncol Res. May
1–2018.10.3727/096504018X15247341491655 (Epub ahead of print).
View Article : Google Scholar
|
|
54
|
Afshar S, Najafi R, Sedighi Pashaki A,
Sharifi M, Nikzad S, Gholami MH, Khoshghadam A, Amini R, Karimi J
and Saidijam M: MiR-185 enhances radiosensitivity of colorectal
cancer cells by targeting IGF1R and IGF2. Biomed Pharmacother.
106:763–769. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Jiang CY, Ruan Y, Wang XH, Zhao W, Jiang
Q, Jing YF, Han BM, Xia SJ and Zhao FJ: MiR-185 attenuates androgen
receptor function in prostate cancer indirectly by targeting
bromodomain containing 8 isoform 2, an androgen receptor
co-activator. Mol Cell Endocrinol. 427:13–20. 2016. View Article : Google Scholar : PubMed/NCBI
|