Introduction
Nasopharyngeal carcinoma (NPC) is associated with
Epstein-Barr virus infection (1).
In 2018, there were an estimated 129,079 new cases of NPC and the
an estimated 72,987 deaths from NPC worldwide (2). Epidemiological trends have shown that
the incidence of NPC has declined progressively and related
mortality has reduced substantially in the past decade (3). Given the complex proximity of the
nasopharynx, surgical excision is rarely used to treat primary NPC
(4). For a long time, radiotherapy
has been recognized as the mainstay for the treatment of NPC
(5), but ~50% of patients
experienced recurrent tumor (6).
Therefore, there is an urgent need to identify more effective
agents.
Ophiopogonin B (OP-B) is one of the main active
components of Radix Ophiopogon japonicus (7). Studies have revealed that OP-B
suppresses tumorigenesis and induces apoptosis in gastric (8), colorectal (9) and lung cancers (10). OP-B has been shown to regulate
multiple cancer-related signaling mechanisms, including JNK/c-Jun
(9), EPH Receptor A2/AKT (11), PI3K/AKT (12) and ERK signaling pathways (12). However, the anti-cancer function of
OP-B and the underlying specific mechanisms remain to be
elucidated.
The Hippo signaling pathway consists of a kinase
cascade and transcription coactivators (13). Aberrant regulation of the Hippo
pathway has been reported in several types of cancer, including NPC
(14–17). However, no studies have reported on
the anticancer effect of OP-B by regulating the Hippo pathway in
NPC. The objective of the present study was to evaluate the effect
of OP-B against NPC and investigate the underlying mechanisms.
Materials and methods
Cell culture
The present study chose human EBV positive NPC cell
(C666-1) and EBV negative cells (HK1) (18). C666-1 and HK1 cells were purchased
from the National Infrastructure of Cell Line Resource, Peking
Union Medical College (Beijing, China). C666-1 and HK1 cells were
cultured in Dulbecco's modified Eagle medium (HyClone; Cytiva)
containing 10% fetal bovine serum (HyClone; Cytiva). All cells were
then incubated at 37°C in a humidified atmosphere of 5%
CO2.
Ophiopogonin B (OP-B; purity of ≥97%; Shanghai Tauto
Biotech Co., Ltd.) was dissolved in dimethyl sulfoxide (DMSO;
Sigma-Aldrich; Merck KGaA). For treatment of cells, OP-B was
diluted in culture medium to a final concentration of 5, 10 and 20
µM (0.01% DMSO) at 25°C for 30 min.
MTT assay
Cells were seeded (5×104 cells/well) in
96-well plates and then OP-B (5, 10 and 20 µM) was added for 12,
24, 48 or 72 h. MTT (20 µl; 5 mg/ml; Sigma-Aldrich; Merck KGaA) was
added and incubated for another 4 h at 37°C. Afterwards, 150 µl
dimethyl sulfoxide was added at room temperature for 10 min.
Absorbance was assessed at 490 nm using a microplate reader (BioTek
Instruments, Inc.).
EdU assay
Cell proliferation was detected using the BeyoClick™
EdU-594 detection kit (cat. no. C0078S; Beyotime Institute of
Biotechnology). Briefly, following treatment with OP-B, cells were
incubated with 50 mM EdU for 2 h at 37°C and incubated with
4′,6-diamidino-2-phenylindole for 30 min at 37°C. After staining,
images were photographed under a fluorescence microscope
(Olympus).
Apoptosis analysis
The Annexin V-FITC/propidium iodide (PI) apoptosis
detection kit was used to detect cell apoptosis. Briefly, following
treatment with OP-B, cells were collected. 5 µl Annexin V-FITC and
5 µl PI were then added, mixed and incubated for 15 min at room
temperature. Apoptosis was evaluated using a flow cytometer (BD
Accuri C6 Plus; BD Biosciences) and FlowJo software (v10.6.2;
FlowJo, LLC). The apoptotic rate was calculated as the percentage
of early + late apoptotic cells.
Measurement of mitochondrial membrane
potential (MMP)
MMP was examined using the fluorescent probe
5,5′,6,6-tetrachloro-1,
1′,3,3′-tetraethyl-benzimidazolylcarbocyanine iodide (JC-1,
Beyotime Institute of Biotechnology). Briefly, following treatment
with OP-B, cells were incubated with JC-1 staining solution at 37°C
for 20 min. MMP was detected using flow cytometry as aforementioned
(BD Biosciences).
Measurement of intracellular reactive
oxygen species (ROS)
Intracellular ROS were detected using an ROS assay
kit (Nanjing Jiancheng Bioengineering Institute). In brief,
following treatment with OP-B, 10 µM 2,7-dichlorodihydrofluorescein
diacetate (DCFH-DA) was added to the cells at 37°C for 20 min. The
median fluorescence intensity of ROS was measured using a flow
cytometer as aforementioned (BD Biosciences).
Malondialdehyde (MDA) assay and
superoxide dismutase (SOD) assay
MDA and SOD activity were assessed using MDA assay
kit and SOD assay kit (Nanjing Jiancheng Bioengineering
Institute).
Small interfering RNA (siRNA)
transfection
The yes-associated protein (YAP) siRNA (sense:
5′-ACUUUUCGCUGCAAGUUGCUA-3′; antisense:
3′-GCAACUUGCAGCGAAAAGUUU-5′), control siRNA (sense:
5′-UUCUCCGAACGUGUCACGUTT-3′; antisense:
3′-ACGUGACACGUUCGGAGAATT-5′) were synthesized by Shanghai
GenePharma Co., Ltd. C666-1 and HK1 cells (106
cells/well) were seeded in six-well plates. The cells were
transfected with 100 nM of YAP siRNA or control siRNA using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) at 37°C, and the medium was changed for fresh
after 6 h. After 48 h, the C666-1 and HK1 cells were harvested and
exposed to OP-B (5 µM) for 24 h at 37°C.
Western blotting
C666-1 and HK1 cells (1×106) were
harvested and lysed them with radioimmunoprecipitation assay buffer
(Beyotime Institute of Biotechnology). Total protein was quantified
by using an Enhanced BCA Protein Assay kit (Beyotime Institute of
Biotechnology). Total protein (50 µg) was loaded on 12% sodium
dodecyl sulphate polyacrylamide gel electrophoresis. Subsequently,
the gel was transferred to a polyvinylidene fluoride membrane
(Millipore). The membrane was blocked by using 5% non-fat milk for
1 h at 25°C. After blocking, the membrane was incubated with
anti-YAP (1:1,000; cat. no. 4912), anti-phosphorylated (p-)YAP
(S127; 1:1,000; cat. no. 4911), anti-mammalian sterile 20-like
kinase 1 (MST1; 1:1,000; cat. no. 3682), anti-large tumor
suppressor 1 (LATS1; 1:1,000; cat. no. 3477), anti-transcriptional
enhanced associate domain 1 (TEAD1; 1:1,000; cat. no. 12292),
anti-Bcl-2 (1:1,000; cat. no. 3498), anti-Bax (1:1,000; cat. no.
2774), anti-caspase-3 (1:1,000; cat. no. 9662), anti-cleaved
caspase-3 (1:1,000; cat. no. 9661), anti-poly(ADP-ribose)
polymerase (PARP; 1:1,000; cat. no. 9542), anti-cleaved-PARP
(1:1,000; cat. no. 9545), anti-forkhead box transcription factor O1
(FOXO1; 1:1,000; cat. no. 2880), anti-p-FOXO1 (1:1,000; cat. no.
2486; all from Cell Signaling Technology, Inc.) and
anti-glyceraldehyde-3-phosphate dehydrogenase (1:5,000; cat. no.
P30008; Abmart Pharmaceutical Technology Co., Ltd.) overnight at
4°C. Membranes were incubated with secondary antibody (1:5,000;
cat. no. M21002, Abmart Pharmaceutical Technology Co., Ltd.) at
room temperature for 2 h. Protein bands were detected using a
chemiluminescence kit and a gel imaging system (Tanon 2500; Tanon
Science and Technology Co., Ltd.).
Statistical analysis
Statistical analyses were performed using GraphPad
Prism 7 (GraphPad Software, Inc.). The data represented mean ±
standard deviation from three independent experiments. One-way
ANOVA and Tukey's post hoc test were conducted to evaluate changes
among groups. P<0.05 was considered to indicate a statistically
significant difference.
Results
OP-B inhibits the proliferation of NPC
cells
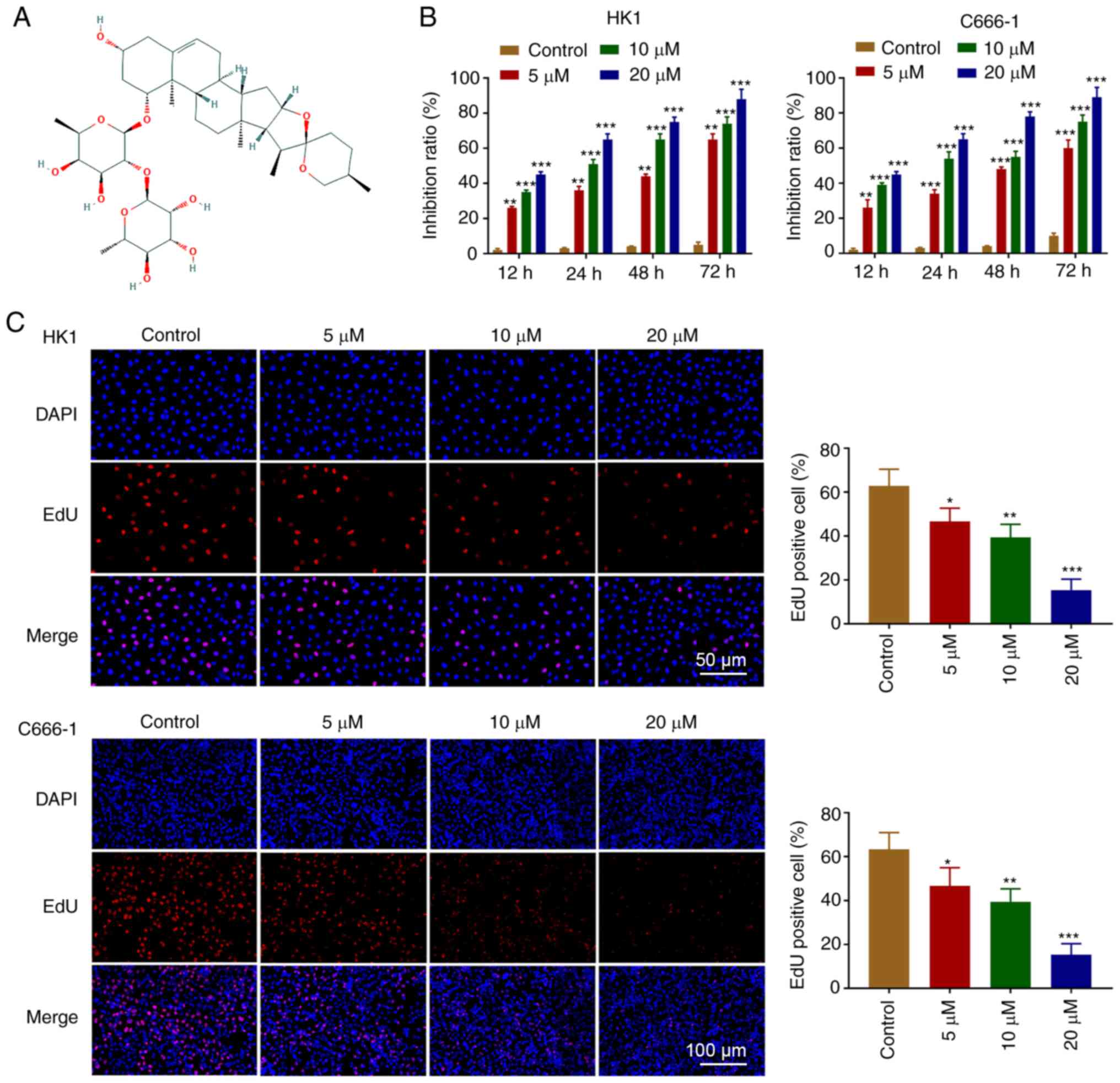
The chemical structure of OP-B is shown in Fig. 1A. The effect of OP-B on cell
proliferation was investigated in C666-1 and HK1 cell lines. The
MTT assay demonstrated that OP-B inhibited C666-1 and HK1 cells
proliferation in a dose and time-dependent manner (Fig. 1B). As shown in Fig. 1C, OP-B effectively inhibited the
proliferation of C666-1 and HK1 cells.
OP-B induces apoptosis in NPC
cells
Next, whether OP-B induced apoptosis was
investigated. Flow cytometry assays were used to confirm that OP-B
activated apoptosis in C666-1 and HK1 cells (Fig. 2A). Furthermore, OP-B increased the
expression of Bax, cleaved-PARP and cleaved-caspase-3, whereas the
expression of Bcl-2, PARP and caspase-3 was decreased by OP-B in
C666-1 and HK1 cells (Fig. 2B).
OP-B induced a concentration-dependent decrease in red/green
fluorescence ratios in C666-1 and HK1 cells (Fig. 2C).
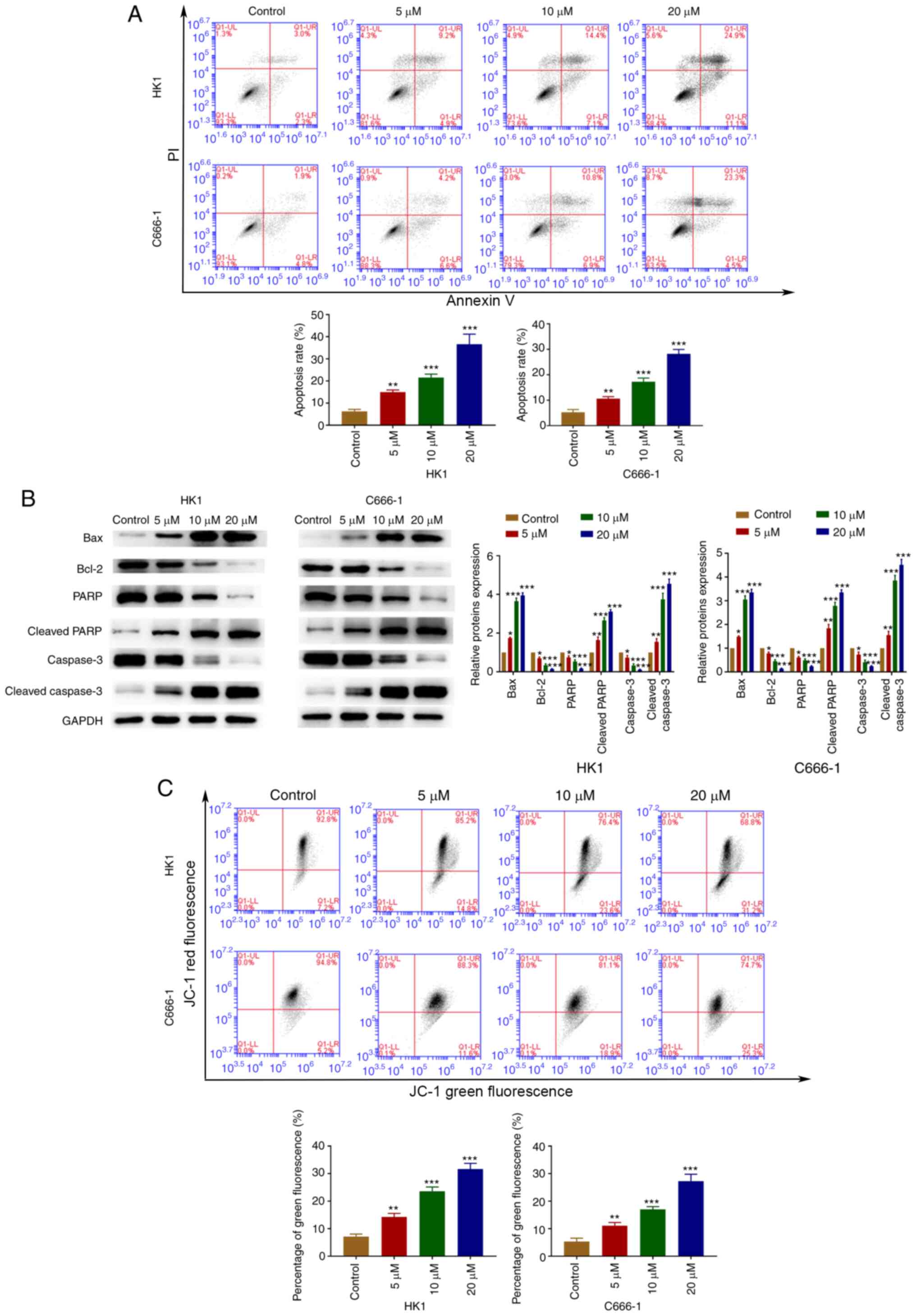 | Figure 2.OP-B induced apoptosis of
nasopharyngeal carcinoma cells. (A) Annexin V/PI analysis was used
to detected OP-B induced apoptosis rates. (B) The expression of
Bax, Bcl-2, cleaved caspase-3, caspase-3, cleaved PARP1 and PARP1
was detected by western blotting. (C) Mitochondrial membrane
potential was evaluated using JC-1 staining. *P<0.05;
**P<0.01; and ***P<0.001 vs. control group. OP-B,
Ophiopogonin B; JC-1, 5,5′,6,6-tetrachloro-1,
1′,3,3′-tetraethyl-benzimidazolylcarbocyanine iodide. |
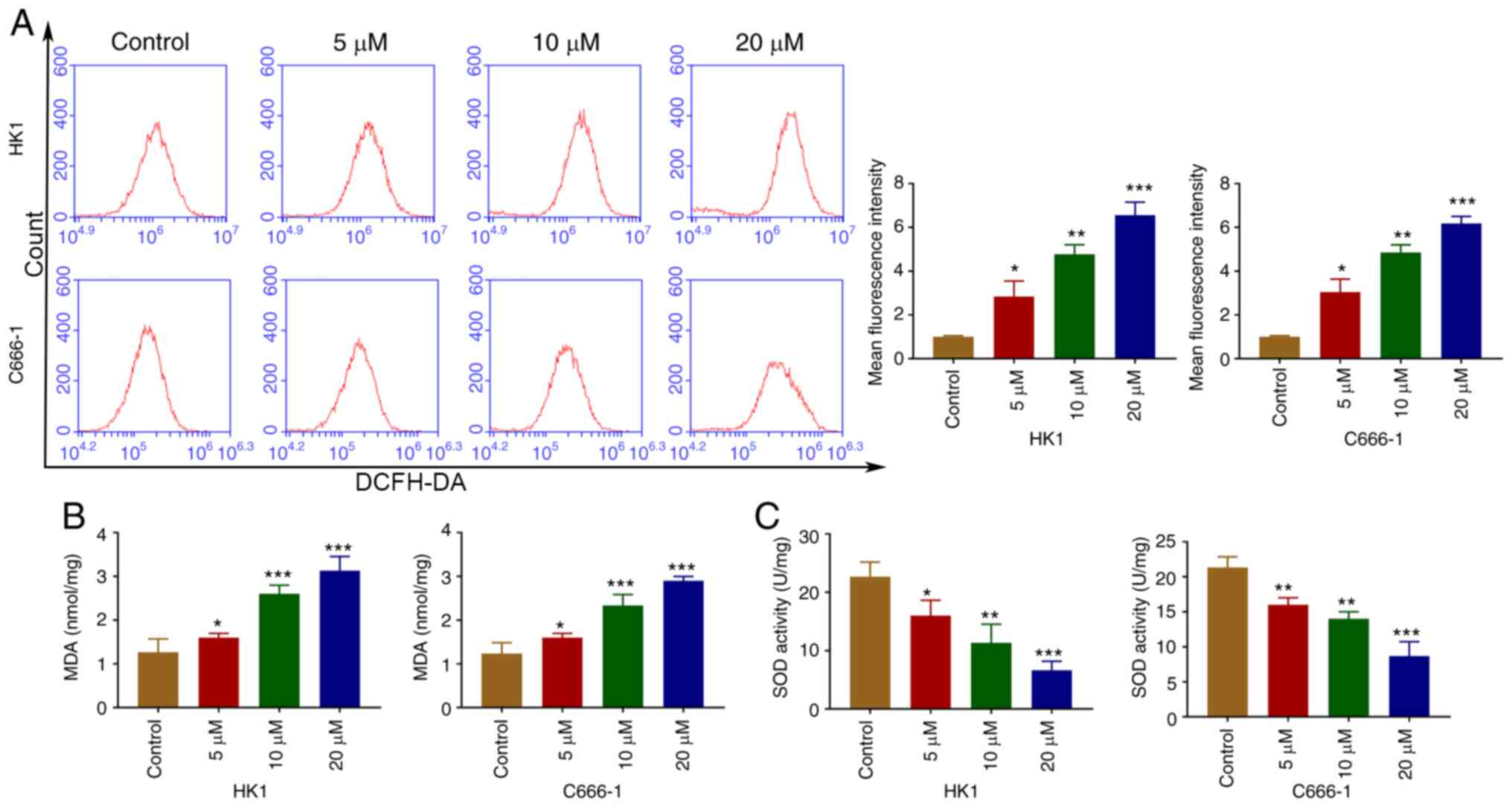
OP-B induces ROS in NPC cells
ROS from mitochondria are related to cell apoptosis
(19). Following treatment with
OP-B, intracellular ROS levels were increased in C666-1 and HK1
cells (Fig. 3A). As shown in
Fig. 3B and C, OP-B increased the
MDA content and decreased SOD activity.
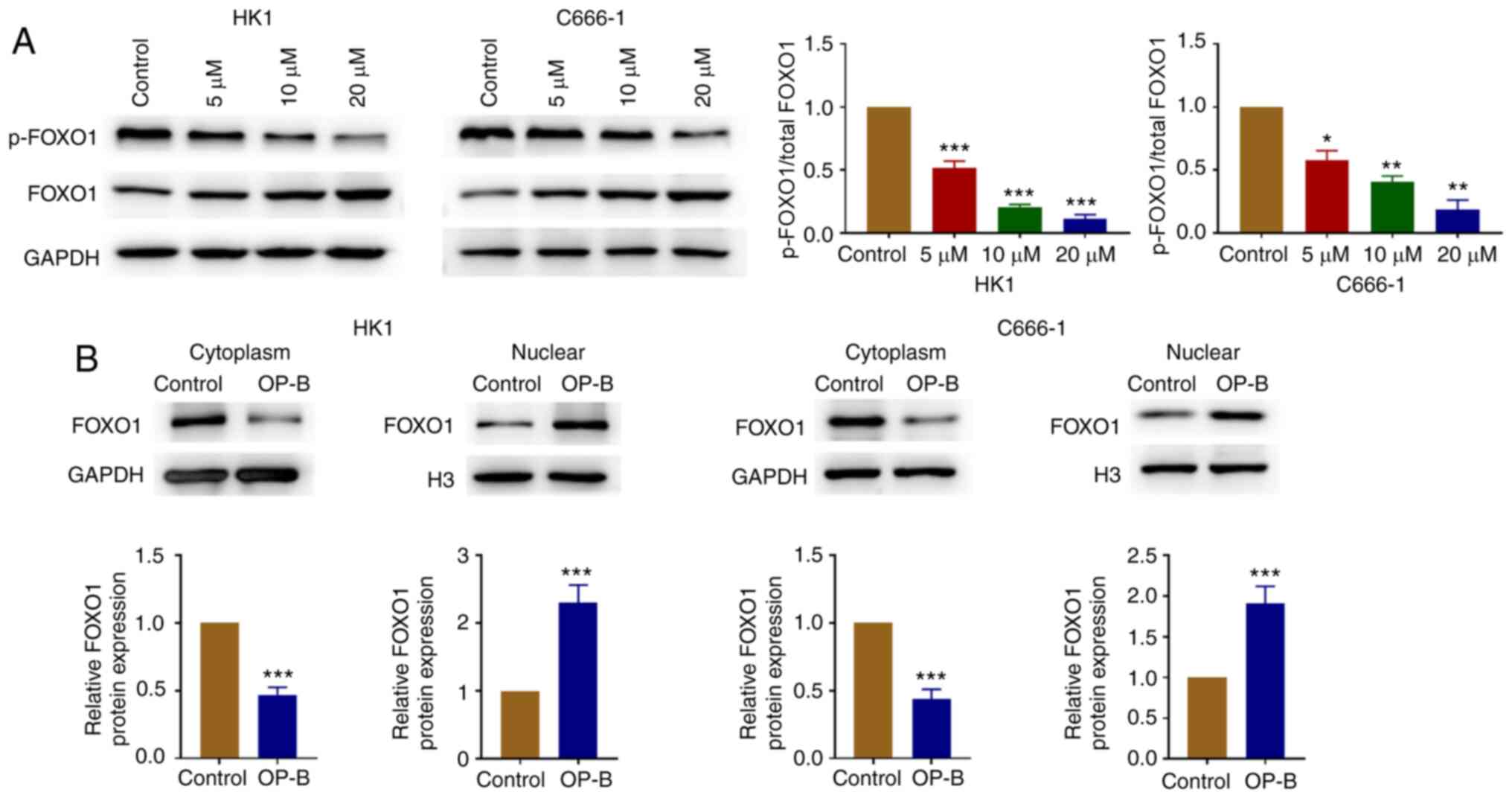
OP-B increases the expression of FOXO1
in the nuclear fraction
OP-B treatment decreased the ratio of p-FOXO1 vs.
total FOXO1 in C666-1 and HK1 cells (Fig. 4A). In addition, FOXO1 protein level
decreased in cytosolic fraction and increased in the nuclear
fraction following treatment with OP-B (Fig. 4B).
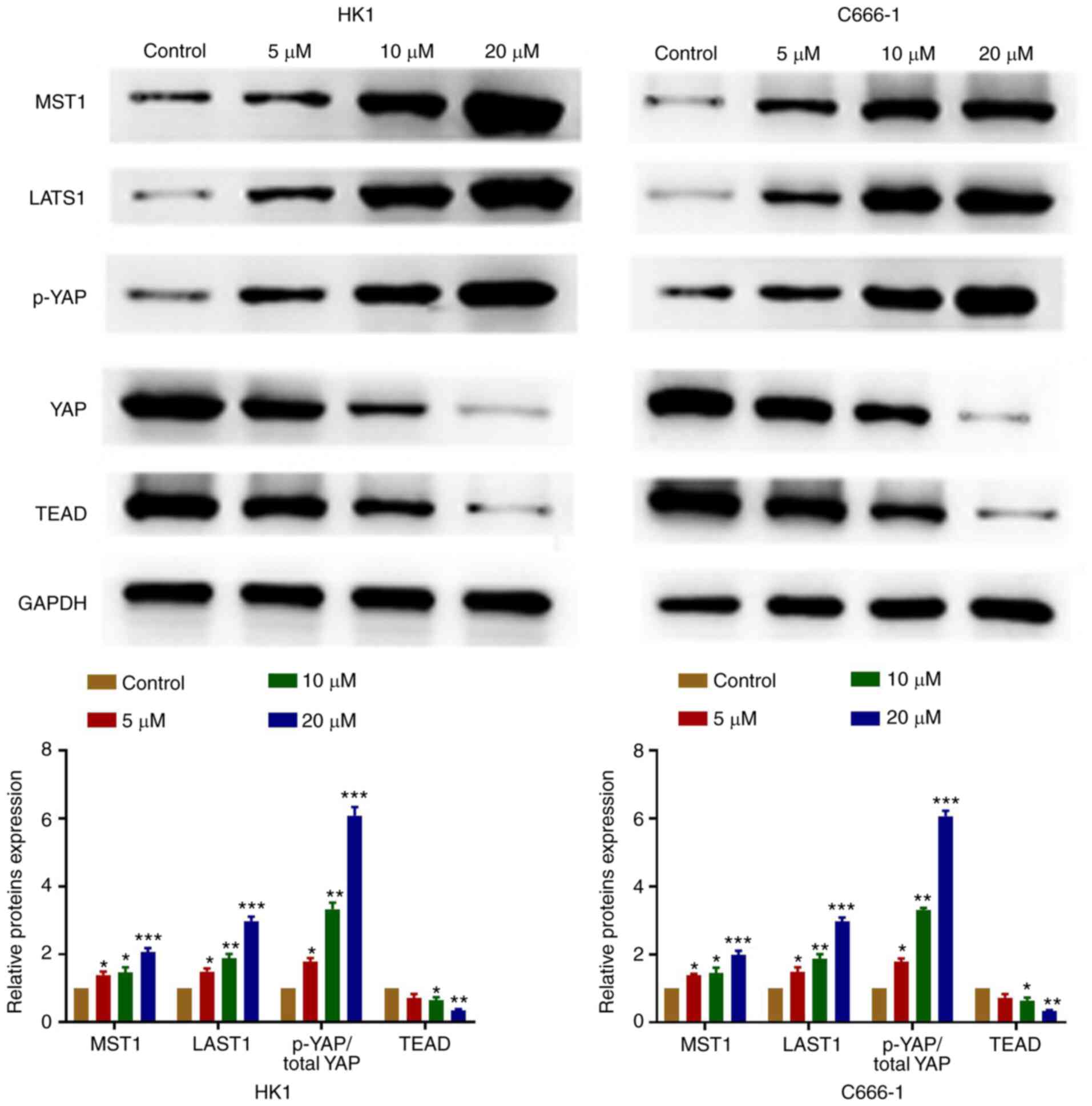
OP-B regulates the Hippo-YAP signaling
pathway in NPC cells
OP-B significantly increased the expression of MST1
and LATS1, increased the ratio of p-YAP vs. total YAP and decreased
TEAD protein levels (Fig. 5).
Therefore, the results suggested that OP-B inhibited NPC cells
tumorigenesis through the regulation of the Hippo signaling
pathway.
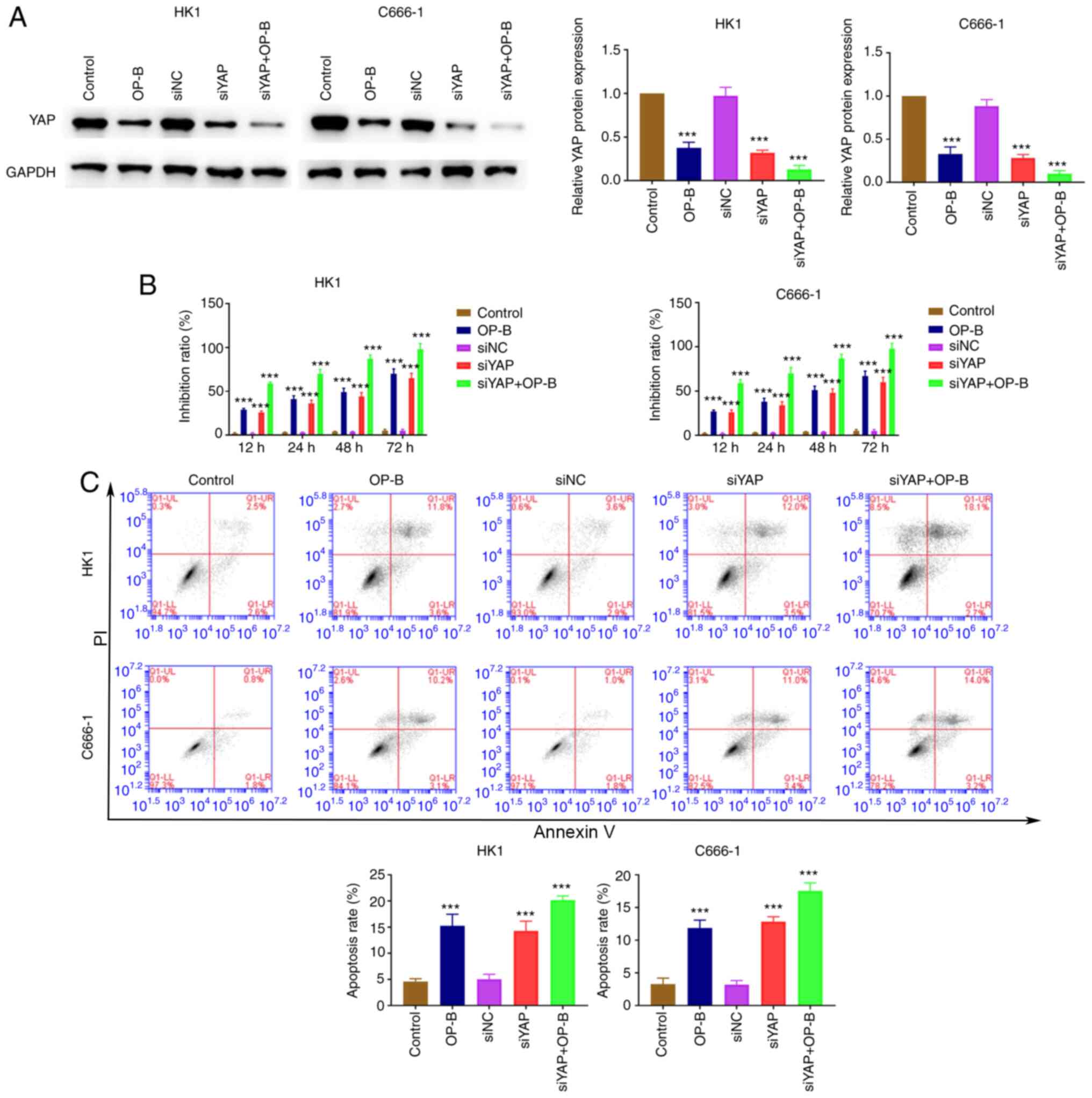
Dysfunction of hippo-YAP signaling
increases the antitumor function of OP-B
siYAP was transfected into C666-1 and HK1 cells and
the cell proliferation and apoptosis ability of these two NPC cell
lines detected. The expression of YAP decreased rapidly due to OP-B
treatment, YAP knockdown significantly reduced YAP expression
(Fig. 6A). YAP knockdown promoted
the inhibitory effect of OP-B on the proliferation of C666-1 and
HK1 cells (Fig. 6B). In addition,
YAP knockdown induced the enhancing effect of OP-B on apoptosis in
C666-1 and HK1 cells (Fig. 6C).
Discussion
The current study revealed that OP-B exerted its
anti-cancer effects by inhibiting cell proliferation, inducing cell
apoptosis and regulating the Hippo signaling pathway.
The mitochondrial pathway is a crucial mechanism of
ophiopogonin-mediated cell death in gastric cancer and prostate
cancer (8,20). The balance of proapoptotic protein
(Bax) and antiapoptotic protein (Bcl-2) maintains a healthy
survival/death balance in cells (21). Caspase-3 is a critical molecule for
stimulating cancer apoptosis (22),
which is activated by pro-apoptotic factors (23). The present study found that OP-B
increased the Bax/Bcl-2 ratio and activated caspase-3 level. PARP
could be cleaved by caspase-3 during apoptosis (24) and was also clearly detected
following OP-B treatment in NPC cells. Meanwhile, suppression of
MMP was found in the process of OP-B-induced NPC cell apoptosis,
which was consistent with results of a previous study (8). This finding indicated that the
mitochondria-mediated apoptotic pathway contributes to the process
of OP-B-induced apoptosis in NPC cells.
The mitochondria of cancer cells overproduce ROS
(25). ROS induce DNA-damage and
cell death (26). The generation of
ROS and reduction in MMP has been reported to be important in
triggering apoptosis (27). Natural
products have attracted attention as good candidate
chemotherapeutic drugs for cancer therapies, due to their ability
to maintain oxidative metabolism with minimal toxicity (28). For example, plumbagin is known to
induce apoptosis in lung cancer through ROS production (29). OP-B participates in ROS generation
in the gastric cancer cells (8).
SOD is an antioxidant that removes superoxide radicals (30). It has been reported that targeting
SOD is a promising approach to selectively kill cancer cells
(31). The present study found that
OP-B accelerated mitochondrial ROS production, decreased SOD levels
and promoted apoptotic cell death in NPC cells.
FOXO1 serves an important role in cell proliferation
and chemosensitivity in NPC cells (32). The results of the present study are
in accordance with previous findings that trifluoperazine increases
the expression of FOXO1 in the nucleus and enhances the expression
of Bax, but decreases the expression of Bcl-2 (33).
The Hippo pathway was originally found in
Drosophila melanogaster (34). It is generally accepted that the
highly-conserved Hippo pathway serves a vital role in maintaining
tissue and organ size, stem cell and tumorigenesis (35–37).
YAP and PDZ-binding motif (TAZ) actively promote cell proliferation
through a transcriptional program mediated by TEAD family
transcription factors (38).
Mechanistically, YAP/TAZ depletion diminishes glycolysis-dependent
proliferation and increases mitochondrial respiration and ROS
production, resulting in oxidative stress-induced cell death
(39). In NPC, the Hippo pathway
has been found to be dysregulated. Li et al (40) reported that the Hippo pathway
attenuates the sensitivity of NPC cells to cisplatin by inducing
epithelial-mesenchymal transition. Elevated expression of YAP, a
Hippo pathway effector, is observed in NPC (41). Overexpression of YAP rescues the
effect of testis-associated oncogenic lncRNA knockdown on NPC cell
stemness and sensitivity of NPC cells to cisplatin (42). In the present study, OP-B markedly
increased the activation of Hippo pathway components, such as MST1,
LATS1 and phosphorylated YAP. Additionally, the protein expression
of YAP and TEAD was decreased in NPC cells following treatment with
OP-B. Previous studies have shown the LATS-mediated phosphorylation
of YAP at Ser127 and its nuclear localization (43–45).
Moreover, YAP knockdown promoted the inhibitory effect of OP-B on
proliferation and induced the effect of OP-B on apoptosis in C666-1
and HK1 cells. This result is partly consistent with a recent study
that indicates that artemisinin inhibits hepatocellular carcinoma
cell proliferation, migration and invasion by suppressing Hippo
signaling (46). The results of the
present study suggested that OP-B regulated Hippo signaling pathway
in NPC cells.
The present study indicated that OP-B inhibited NPC
cells survival by activating mitochondria-mediated apoptosis via
the Hippo pathway. OP-B appears to be a potential therapeutic agent
for NPC patients.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
WD and HD designed the study and performed the
research; QD and HD analyzed the data and wrote the manuscript. WD
and HD confirm the authenticity of all the raw data. All authors
read and approved the final manuscript and agree to be accountable
for all aspects of the work in ensuring that the accuracy or
integrity of any part of the work are appropriately investigated
and resolved.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Glastonbury CM: Head and neck squamous
cell cancer: Approach to staging and surveillance. Diseases of the
Brain, Head and Neck, Spine 2020–2023: Diagnostic Imaging
[Internet]. Hodler J, Kubik-Huch RJ and von Schulthess GK: Cham
(CH): Springer. Chapter 17. 2020, View Article : Google Scholar
|
|
2
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen YP, Chan ATC, Le QT, Blanchard P, Sun
Y and Ma J: Nasopharyngeal carcinoma. Lancet. 394:64–80. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lee HM, Okuda KS, González FE and Patel V:
Current perspectives on nasopharyngeal carcinoma. Adv Exp Med Biol.
1164:11–34. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sun XS, Li XY, Chen QY, Tang LQ and Mai
HQ: Future of radiotherapy in nasopharyngeal carcinoma. Br J
Radiol. 92:92019. View Article : Google Scholar
|
|
6
|
Caponigro F, Longo F, Ionna F and Perri F:
Treatment approaches to nasopharyngeal carcinoma: A review.
Anticancer Drugs. 21:471–477. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen MH, Chen XJ, Wang M, Lin LG and Wang
YT: Ophiopogon japonicus-A phytochemical, ethnomedicinal and
pharmacological review. J Ethnopharmacol. 181:193–213. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang W, Zhang Q, Jiang Y, Li F and Xin H:
Effects of ophiopogonin B on the proliferation and apoptosis of
SGC-7901 human gastric cancer cells. Mol Med Rep. 13:4981–4986.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gao GY, Ma J, Lu P, Jiang X and Chang C:
Ophiopogonin B induces the autophagy and apoptosis of colon cancer
cells by activating JNK/c-Jun signaling pathway. Biomed
Pharmacother. 108:1208–1215. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hu C, Jiang R, Cheng Z, Lu Y, Gu L, Li H,
Li L, Gao Q, Chen M and Zhang X: Ophiopogonin-B Suppresses
Epithelial-mesenchymal transition in human lung adenocarcinoma
cells via the Linc00668/miR-432-5p/EMT axis. J Cancer.
10:2849–2856. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen M, Hu C, Guo Y, Jiang R, Jiang H,
Zhou Y, Fu H, Wu M and Zhang X: Ophiopogonin B suppresses the
metastasis and angiogenesis of A549 cells in vitro and in
vivo by inhibiting the EphA2/Akt signaling pathway. Oncol Rep.
40:1339–1347. 2018.PubMed/NCBI
|
|
12
|
Chen M, Du Y, Qui M, Wang M, Chen K, Huang
Z, Jiang M, Xiong F, Chen J, Zhou J, et al: Ophiopogonin B-induced
autophagy in non-small cell lung cancer cells via inhibition of the
PI3K/Akt signaling pathway. Oncol Rep. 29:430–436. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pan D: The hippo signaling pathway in
development and cancer. Dev Cell. 19:491–505. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang Y, Wang M, Xu X, Liu Y and Xiao C:
Matrine promotes apoptosis in SW480 colorectal cancer cells via
elevating MIEF1-related mitochondrial division in a manner
dependent on LATS2-Hippo pathway. J Cell Physiol. 234:22731–22741.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kim SH, Jin H, Meng RY, Kim DY, Liu YC,
Chai OH, Park BH and Kim SM: Activating hippo pathway via Rassf1 by
ursolic acid suppresses the tumorigenesis of gastric cancer. Int J
Mol Sci. 20:47092019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang L, Wang J, Cao Y, Li W, Wang Y, Xu J
and Xu G: Molecular evidence for better efficacy of hypocrellin A
and oleanolic acid combination in suppression of HCC growth. Eur J
Pharmacol. 842:281–290. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhu Y, He D, Bo H, Liu Z, Xiao M, Xiang L,
Zhou J, Liu Y, Liu X, Gong L, et al: The MRVI1-AS1/ATF3 signaling
loop sensitizes nasopharyngeal cancer cells to paclitaxel by
regulating the Hippo-TAZ pathway. Oncogene. 38:6065–6081. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wu RW, Chu ES, Huang Z, Xu CS, Ip CW and
Yow CM: FosPeg® PDT alters the EBV miRNAs and LMP1
protein expression in EBV positive nasopharyngeal carcinoma cells.
J Photochem Photobiol B. 127:114–122. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chong SJ, Low IC and Pervaiz S:
Mitochondrial ROS and involvement of Bcl-2 as a mitochondrial ROS
regulator. Mitochondrion. 19:39–48. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lu Z, Wang H, Zhu M, Song W, Wang J, Wu C,
Kong Y, Guo J, Li N, Liu J, et al: Ophiopogonin D', a natural
product from radix ophiopogonis, induces in vitro and in vivo
RIPK1-dependent and caspase-independent apoptotic death in
androgen-independent human prostate cancer cells. Front Pharmacol.
9:4322018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hassan M, Watari H, AbuAlmaaty A, Ohba Y
and Sakuragi N: Apoptosis and molecular targeting therapy in
cancer. Biomed Res Int. 2014:1508452014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Crowley LC and Waterhouse NJ: Detecting
cleaved caspase-3 in apoptotic cells by flow cytometry. Cold Spring
Harb Protoc. Nov 1–2016.(Epub ahead of print). doi:
10.1101/pdb.prot087312. View Article : Google Scholar
|
|
23
|
Li J, Wu Y, Wang D, Zou L, Fu C, Zhang J
and Leung GP: Oridonin synergistically enhances the anti-tumor
efficacy of doxorubicin against aggressive breast cancer via
pro-apoptotic and anti-angiogenic effects. Pharmacol Res.
146:1043132019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Boulares AH, Yakovlev AG, Ivanova V,
Stoica BA, Wang G, Iyer S and Smulson M: Role of poly(ADP-ribose)
polymerase (PARP) cleavage in apoptosis. Caspase 3-resistant PARP
mutant increases rates of apoptosis in transfected cells. J Biol
Chem. 274:22932–22940. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yang Y, Karakhanova S, Hartwig W, D'Haese
JG, Philippov PP, Werner J and Bazhin AV: Mitochondria and
mitochondrial ROS in cancer: Novel targets for anticancer therapy.
J Cell Physiol. 231:2570–2581. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kocyigit A and Guler EM: Curcumin induce
DNA damage and apoptosis through generation of reactive oxygen
species and reducing mitochondrial membrane potential in melanoma
cancer cells. Cell Mol Biol. 63:97–105. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
He J, Wei W, Yang Q and Wang Y:
Phillygenin exerts in vitro and in vivo antitumor effects in
drug-resistant human esophageal cancer cells by inducing
mitochondrial-mediated apoptosis, ROS generation and inhibition of
the nuclear factor kappa B NF-κB signalling pathway. Med Sci Monit.
25:739–745. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
NavaneethaKrishnan S, Rosales JL and Lee
KY: ROS-mediated cancer cell killing through dietary
phytochemicals. Oxid Med Cell Longev. 2019:90515422019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tripathi SK, Rengasamy KRR and Biswal BK:
Plumbagin engenders apoptosis in lung cancer cells via caspase-9
activation and targeting mitochondrial-mediated ROS induction. Arch
Pharm Res. 43:242–256. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
He L, He T, Farrar S, Ji L, Liu T and Ma
X: Antioxidants maintain cellular redox homeostasis by elimination
of reactive oxygen species. Cell Physiol Biochem. 44:532–553. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Huang P, Feng L, Oldham EA, Keating MJ and
Plunkett W: Superoxide dismutase as a target for the selective
killing of cancer cells. Nature. 407:390–395. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhao M, Luo R, Liu Y, Gao L, Fu Z, Fu Q,
Luo X, Chen Y, Deng X, Liang Z, et al: miR-3188 regulates
nasopharyngeal carcinoma proliferation and chemosensitivity through
a FOXO1-modulated positive feedback loop with
mTOR-p-PI3K/AKT-c-JUN. Nat Commun. 7:113092016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Jiang J, Huang Z, Chen X, Luo R, Cai H,
Wang H, Zhang H, Sun T and Zhang Y: Trifluoperazine activates
FOXO1-related signals to inhibit tumor growth in hepatocellular
carcinoma. DNA Cell Biol. 36:813–821. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Meng Z, Moroishi T and Guan KL: Mechanisms
of Hippo pathway regulation. Genes Dev. 30:1–17. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Dong L, Lin F, Wu W, Liu Y and Huang W:
Verteporfin inhibits YAP-induced bladder cancer cell growth and
invasion via Hippo signaling pathway. Int J Med Sci. 15:645–652.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hsu PC, Jablons DM, Yang CT and You L:
Epidermal Growth Factor Receptor (EGFR) Pathway, Yes-Associated
Protein (YAP) and the Regulation of Programmed Death-Ligand 1
(PD-L1) in Non-Small Cell Lung Cancer (NSCLC). Int J Mol Sci.
20:38212019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Choi W, Kim J, Park J, Lee DH, Hwang D,
Kim JH, Ashktorab H, Smoot D, Kim SY, Choi C, et al: YAP/TAZ
initiates gastric tumorigenesis via upregulation of MYC. Cancer
Res. 78:3306–3320. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Koo JH and Guan KL: Interplay between
YAP/TAZ and Metabolism. Cell Metab. 28:196–206. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
White SM, Avantaggiati ML, Nemazanyy I, Di
Poto C, Yang Y, Pende M, Gibney GT, Ressom HW, Field J, Atkins MB
and Yi C: YAP/TAZ inhibition induces metabolic and signaling
rewiring resulting in targetable vulnerabilities in NF2-deficient
tumor cells. Dev Cell. 49:425–443.e9. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Li S, Zhang X, Zhang R, Liang Z, Liao W,
Du Z, Gao C, Liu F, Fan Y and Hong H: Hippo pathway contributes to
cisplatin resistant-induced EMT in nasopharyngeal carcinoma cells.
Cell Cycle. 16:1601–1610. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Song L, Tang H, Liao W, Luo X, Li Y, Chen
T and Zhang X: FOXC2 positively regulates YAP signaling and
promotes the glycolysis of nasopharyngeal carcinoma. Exp Cell Res.
357:17–24. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Gao L, Cheng XL and Cao H: LncRNA THOR
attenuates cisplatin sensitivity of nasopharyngeal carcinoma cells
via enhancing cells stemness. Biochimie. 152:63–72. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Suemura S, Kodama T, Myojin Y, Yamada R,
Shigekawa M, Hikita H, Sakamori R, Tatsumi T and Takehara T: CRISPR
Loss-of-Function screen identifies the hippo signaling pathway as
the mediator of regorafenib efficacy in hepatocellular carcinoma.
Cancers. 11:13622019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ye XY, Luo QQ, Xu YH, Tang NW, Niu XM, Li
ZM, Shen SP, Lu S and Chen ZW: 17-AAG suppresses growth and
invasion of lung adenocarcinoma cells via regulation of the
LATS1/YAP pathway. J Cell Mol Med. 19:651–663. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Han Y, Tang Z, Zhao Y, Li Q and Wang E:
TNFAIP8 regulates Hippo pathway through interacting with LATS1 to
promote cell proliferation and invasion in lung cancer. Mol
Carcinog. 57:159–166. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Li Y, Lu J, Chen Q, Han S, Shao H, Chen P,
Jin Q, Yang M, Shangguan F, Fei M, et al: Artemisinin suppresses
hepatocellular carcinoma cell growth, migration and invasion by
targeting cellular bioenergetics and Hippo-YAP signaling. Arch
Toxicol. 93:3367–3383. 2019. View Article : Google Scholar : PubMed/NCBI
|