Introduction
Neurofibromatosis is characterized by café-au-lait
spots, iris Lisch nodules and cutaneous or subcutaneous skin tumors
termedneurofibromas (1). This
disease may cause tissue along the nerves to grow uncontrollably,
inducing serious damage by compressing the nerves. Two types of
neurofibromatosis, type 1 (NF1) and type 2 (NF2), have been
distinguished and genetically mapped to different loci, namely
chromosomes 17q11.2 and 22q112 (2). NF1 predominates in neurofibromatosis
patients (~98%), while NF2 comprises 2% of this population and is
reported as a more rare disease (1:35,000) (3).
NF1 occurs in ~1 out of 3,500 births (4) and is defined as an autosomal dominant
genetic disorder correlated with point mutations or gross deletions
of the NF1 gene (5). Neoplasms in
the ends of the limbs are one symptom of NF1. Although uncommon,
NF1 patients may exhibit macrodactylia of the fingers or toes
accompanied by the proliferation of the vessels, phalanges and
subcutaneous fat (6). The exact
cellular genes that participate in nerve growth in NF1 patients
remain unclear.
In the present study, abnormal bulky terminal nerves
were obtained from three diagnosed NF1 patients, and the expression
profiles of nerves in the NF1 patients and normal controls were
analyzed by microarray analysis. The genes likely to be involved in
promoting nerve growth were also identified.
Materials and methods
Patients and tissues
Three confirmed NF1 patients (male; age, 20–30
years), with macrodactylia of the fingers or toes, and five healthy
controls were recruited from the Department of General Surgery at
The Third Affiliated Hospital of Southern Medical University
(Guangzhou, China) between 2009 and 2011. The healthy controls all
underwent amputations due to car accidents or fires. None of the
patients and healthy controls were suffering from systemic
disorders or viral infections. The nerve tissues of the two groups
were separated during surgery. Subsequent to splitting them into
small sections, the tissues were powdered in liquid nitrogen and
the samples were kept refrigerated at −80°C. All study participants
provided written informed consent and the study was approved by the
Ethics Committee of Jilin University (Changchun, China).
Microarray analysis
RNA was extracted using the RNeasy mini kit (Qiagen,
Hilden, Germany). Aliquots of the RNA were analyzed by a
Bioanalyser 2100 (Agilent Technologies, Santa Clara, CA, USA) and
quantified by a Nano Drop ND-1000 (Labtech International, Uckfield,
UK). The Whole Human Genome Microarray (Agilent Technologies) was
selected to screen for the expression profiles of the different
genes in the NF1 patients and healthy controls. Following
hybridization, the microarrays were scanned in the Agilent
Microarray Scanner, analyzed with the Feature Extraction software
and the raw data were normalized by the Quantile Algorithm. To
control the false discovery rate, the Q-value of all genes used in
this analysis was assessed using Q<0.05.
Cell culture
PC12 rat pheochromocytoma cells were initially
obtained from the American Type Culture Collection (Manassas, VA,
USA) and cultured in RPMI-1640 supplemented with 10% (v/v) fetal
calf serum, cultivated at 37°C in a 5% CO2 incubator. At
70–90% confluency, the cells (2.5×105) were seeded onto
12-well plates (Iwaki, Tokyo, Japan). Following a 24-h incubation,
differentiation was induced by adding 50 ng/ml nerve growth factor
(NGF; Chemicon, Temecula, CA, USA) (7). The medium was changed every two days.
Following a 96-h incubation, the PC12 cells were transformed into
neuronal-like cells.
RNA extraction and RT-PCR
The PC12 cells were harvested following
differentiation and the total RNA was isolated using TRIzol
(Invitrogen Life Technologies, Carlsbad, CA, USA) according to the
manufacturer’s instructions. The RNA pellets were stored in sterile
ribonuclease-free water. Reverse transcription was conducted using
1 μg total RNA, 0.5 μg oligo(dT) and Superscript II enzyme (Gibco,
Carlsbad, CA, USA). To check the relative mRNA levels, the primers
shown in Table I were used.
 | Table IPrimers for checking relative mRNA
levels. |
Table I
Primers for checking relative mRNA
levels.
| Primer | Sequence | Length, bp |
|---|
| ACSBG1 | Forward:
ACGCCATCATTGACACCC
Reverse: CGTAGCCCGCATACAAGC | 630 |
| ADAMTS4 | Forward:
AGGTCCCATGTGCAACG
Reverse: TGGAGCCTCTGGTTTGTCT | 724 |
| DGKB | Forward:
GGACCATATTTTACCACC
Reverse: AACTTCCACCTGTCCAAC | 600 |
| FPR1 | Forward:
TCTCCCCACGAACATCTC
Reverse: TTGTGGATCTTGGTGGC | 672 |
| HIF3α | Forward:
CAGTAGCAGCCACTCCCAG
Reverse: CAGGGGCTCATTCAGGTT | 635 |
| HOXC8 | Forward:
CTGTTCTCCAAATACAAAGCCG
Reverse: CTTCTTCCTCCACCTTCTCCTC | 652 |
| IL1β | Forward:
GCTTATTACAGTGGCAATGAGG
Reverse: GGATCTACACTCTCCAGCTGTAGAG | 572 |
| MMP9 | Forward:
CTCCAGAAGCAACTGTCCC
Reverse: CCATCAGCATTGCCGTC | 635 |
| RCAN1 | Forward:
GTGGCCGGTCCCCAG
Reverse: TTGGCTTAGGTCTCCTCATTCT | 697 |
| SLC11A1 | Forward:
CCACGACTACGCCAAGAT
Reverse: AAGGAGCCCATACAGGAAG | 640 |
Western blotting
The cells were harvested by trypsinization, lysed in
RIPA buffer (Aidlab, Beijing, China) on ice for 30 mins and then
centrifuged (Eppendorf, Hamburg, Germany) at 4°C and 12,000 × g for
10 mins. The whole cell lysate was subjected to sodium dodecyl
sulfate-polyacrylamide gel electrophoresis and transferred onto
polyvinylidene difluoride membranes (Amersham Life Sciences,
Arlington Heights, IL, USA). The membranes were incubated with 5%
skimmed dry milk and then with polyclonal goat anti-human primary
antibodies [GAPDH, acyl-CoA synthetase bubblegum family member 1
(ACSBG1), hypoxia inducible factor 3α (HIF3α) and HOXC8; Santa Cruz
Biotechnology, Inc., Santa Cruz, CA, USA]. Following washing with
phosphate-buffered saline with Tween 20 (PBST), the membranes were
incubated with horseradish peroxidase-conjugated mouse anti-goat
IgG (Santa Cruz Biotechnology Inc.). The immunoreactivity was
visualized with the enhanced chemiluminescence system (ECL;
Amersham Life Sciences).
Statistical analysis
Statistical analyses were performed using the Stat
View software (SAS Institute, Inc., Cary, NC, USA). P<0.05 was
considered to indicate a statistically significant difference.
Microarray analyses were analyzed with Feature Extraction software
10.7 and the raw data were normalized by the Quantile Algorithm,
Gene Spring Software 11.0 (Agilent Technologies). Student’s t-tests
were used to identify the differentially-expressed genes.
Results
Differential gene expression profiles in
three NF1 patients
To investigate the etiology of nerve growth in the
NF1 patients, the genes that shared the same change trends in the
NF1 patients were analyzed to try to explain their similar
phenotypes, particularly nerve hyperplasia, vessel overgrowth and
subcutaneous fat generation. Various genes were compared between
each NF1 patient and the healthy controls. From nearly 30,000
genes, 132, 144 and 158 genes were identified to have significant
changes (P<0.01) in the three NF1 patients, respectively. A
common set of 61 genes exhibited the same trend in the three
patients, and among these, 28 genes changed markedly >5-fold.
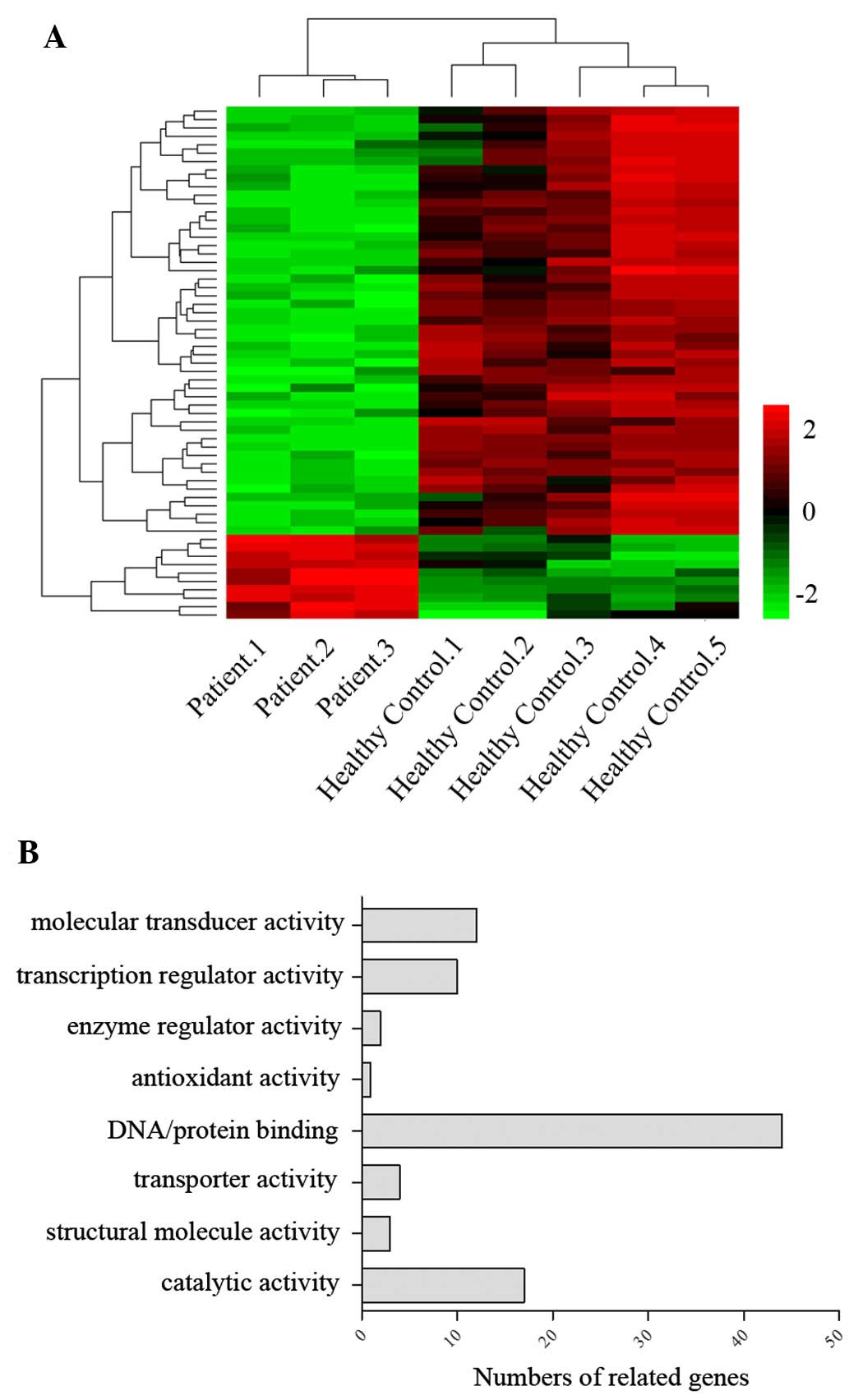
Gene ontology (GO) analysis revealed that genes associated with
DNA/protein binding represented the largest proportion (48%) of the
total number of different genes (Fig.
1), while others involved in regulating molecular transduction
(23%) and catalysis (18%) also represented a high percentage.
Confirmation of possible genes in
regulating nerve overgrowth of NF1
Transcriptional level
Of the 28 evidently changed genes, the
metastasis-associated genes, lymphocyte function-associated antigen
1 (LFA1), C-X-C chemokine receptor type 4 (CXCR4) and intracellular
adhesion molecule 1 (ICAM1), and the inflammatory cytokines,
interleukin (IL)1β, IL6 and IL8, were generally expressed at a low
level, and the genes involved in the cell cycle, cyclin-dependent
kinase 4 inhibitor, cyclin dependent kinase 4 and
Serine/threonine-protein kinase 1, and apoptosis, LMNB1, BIRC3 and
FOSL1, were also significantly downregulated. It is noteworthy that
>75% of these genes were downregulated, and that the majority of
them were confirmed to be significant in promoting tumor
proliferation and metastasis. Thus, the mechanism of the abnormal
overgrowth of the nerves, including the vessels, phalanges and
subcutaneous fat around the vessels, in NF1 patients varied from
that of cancer tissues. To identify the main factors that affect
nerve growth, genes that were up- or downregulated >10-fold
change were investigated. The screened genes are listed in Table II. The levels of mRNA expression
of these genes are shown in Fig.
2.
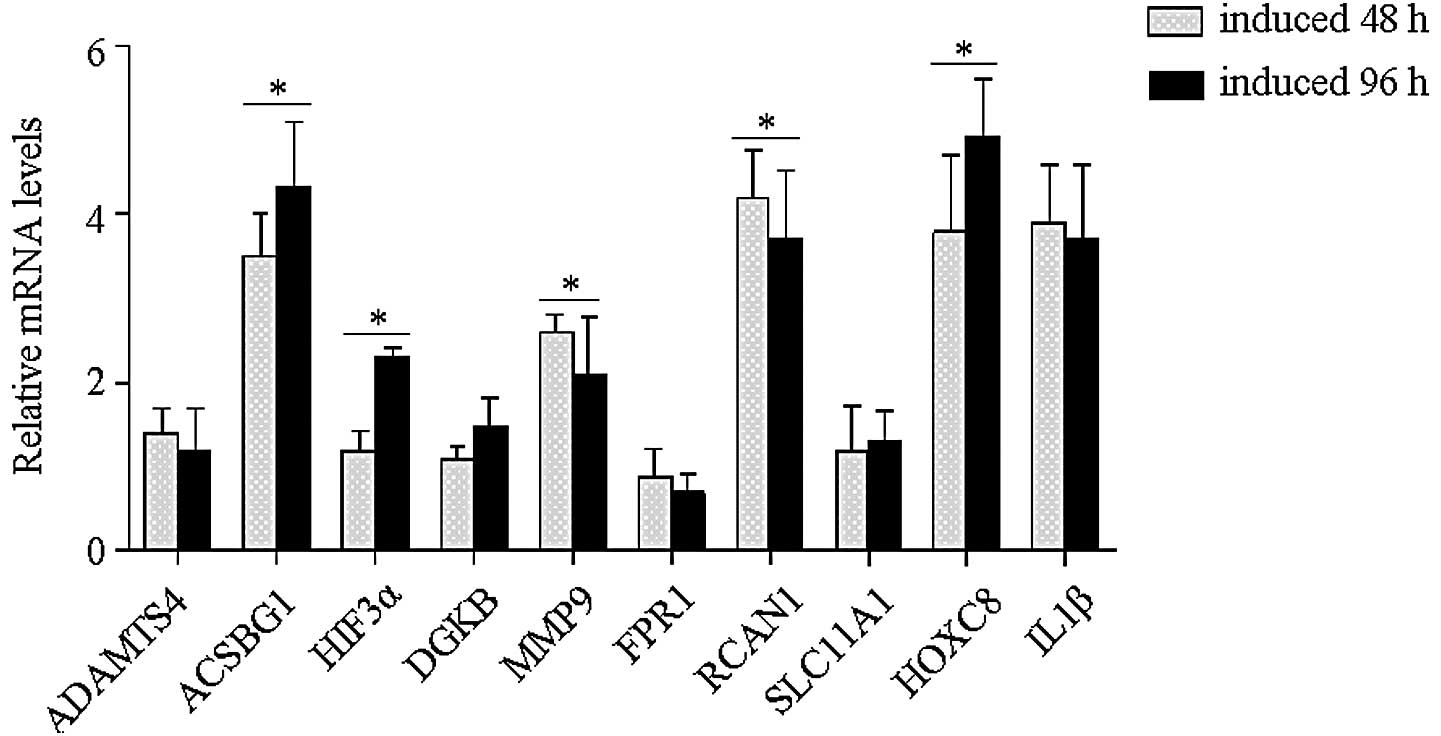 | Figure 2mRNA expression levels of possible
genes related with NF1 development. PC12 cells were induced to
differentiate using NGF. Following 96 h of induction, the cells
were harvested and the total RNA isolated. The 10 genes that
changed >10-fold were analyzed, including the calcium ions
channel-related factors, FPR1, RCAN1 and DGKB, the
inflammatory-related cytokines, IL1β and SLC11A1, the
matrix-degrading genes, ADAMTS4 and MMP9, the transcription
modulators, HIF3α and HOXC8, and the metabolism-related factor,
ACSBG1. The expression levels were determined with GAPDH as a
reference. ACSBG1, HIF3α and HOXC8 genes showed evident
upregulation within the differentiation of the PC12 cells
(*P<0.05). NF1; neurofibromatosis type 1, NGF; nerve
growth factor; ADAMTS4, a disinterin and metalloproteinase with
thrombospondin motifs 4; ACSBG1, acyl-CoA synthetase bubblegum
family member 1; HIF3α, hypoxia inducible factor 3α; DGKB,
diacylglycerol kinase; MMP9, matrix metallopeptidase 9; FPR1,
formyl peptide receptor 1; RCAN1, regulator of calcineurin 1;
SLC11A1, solute family carrier 11; IL1β, interleukin 1β. |
 | Table IIGenes upregulated >10-fold and
their corresponding functions. |
Table II
Genes upregulated >10-fold and
their corresponding functions.
| Probe ID | Gene | Chromosome No. | Fold change in
NF1 | Gene function |
|---|
| 1660497 | ADAMTS4 | 1 | 0.052 | Metalloproteases;
inflammation-related |
| 1030270 | FPR1 | 19 | 0.0674 | Inflammation-related;
interacting with calcium ions |
| 620239 | RCAN1 | 21 | 0.0685 | Inhibit
calcineurin-dependent pathways |
| 840685 | IL1β | 2 | 0.074 | Inflamatory
cytokine |
| 5390220 | MMP9 | 16 | 0.0825 | Degradation of the
extracellular matrix |
| 780465 | SLC11A1 | 2 | 0.0828 | Divalent transition
metal transporter; immunological response-related |
| 4640059 | HOXC8 | 12 | 10.531 | Modulates the
frequency of transcription |
| 4540192 | DGKB | 7 | 10.4188 | Interacting with
calcium ions; associated with glucose metabolism |
| 1110520 | HIF3α | 19 | 12.637 | Modulate
transcription; responsor of lowered oxygen tension |
| 2480730 | ACSBG1 | 15 | 14.536 | Related to metabolic
processes of fatty acids |
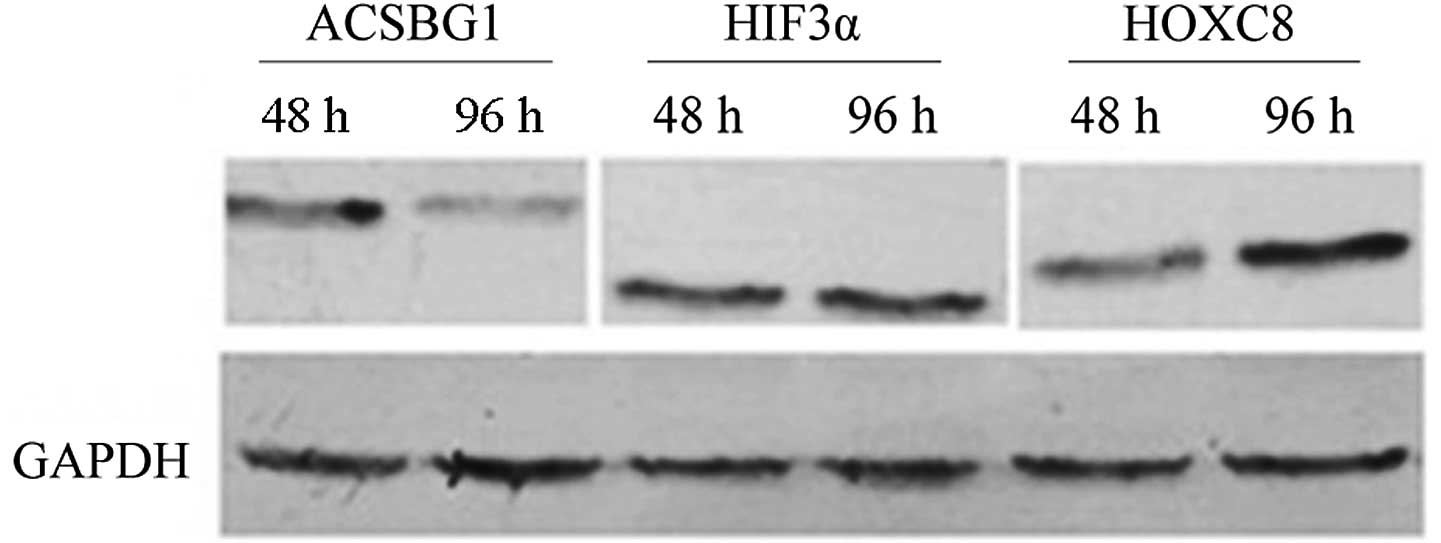
Protein level
To determine whether the screened genes that changed
at the transcriptional level were also altered at the protein
level, the expression of ACSBG1, hHIF3α and HOXC8, which possessed
statistical significant changes in mRNA expression was analyzed. By
inducing PC12 cells to differentiate into neuron-like cells that
were characterized by axon growth, HOXC8 was identified to be
upregulated during the process of PC12 differentiation (Fig. 3). This implies that HOXC8 may be
involved in promoting nerve growth in NF1.
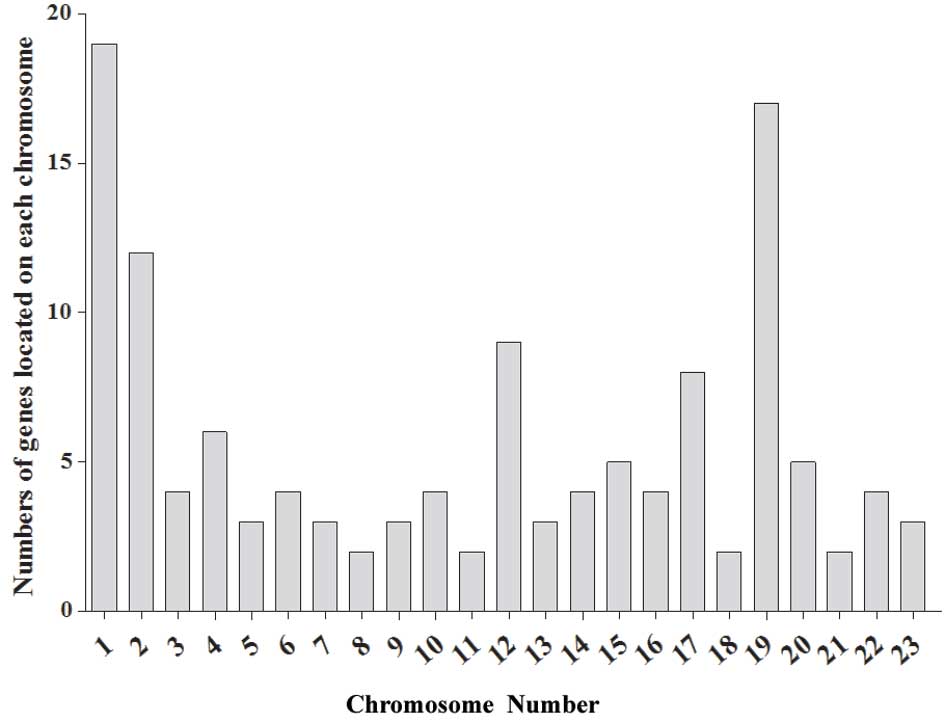
Location analysis of altered genes
The common locations of genes that shared the same
change trend in the three patients were clarified in this study.
The results revealed that genes located on chromosomes 1, 2, 12, 17
and 19 possessed the most marked changes in the NF1 patients. The
genes on chromosomes 1 and 19 in particular indicated potential
gene clusters associated with the nerve growth abnormalities. The
chromosomal localization was displayed as shown in Fig. 4.
Discussion
Although NF1 has attracted attention for a long
period of time, the pathogenetic mechanism and intracellular
factors involved in nerve growth during NF1 development remain
unclear. In the present study, comparisons of gene expression
profiles between NF1 and healthy controls were investigated by
microarray analysis. A total of 132, 144 and 158 genes from the
nerve tissues of the three NF1 patients showed statistically
significant changes, respectively, and among these genes,
downregulated genes accounted for >70% in each patient. A total
of 28 genes that changed >5-fold showed the same trend in all
the NF1 patients, including the intracellular protein binding
mediators, CXCR4, ICAM1, integrin αX, LFA1 and major facilitator
superfamily domain containing 6, involved in matrix degradation and
metastasis (8), and the
inflammatory related factors, IL1β, IL6 and IL8, involved in
regulating the extracellular microenvironment, angiogenesis and
tumorigenesis (9). The general
downregulation of the tumor-stimulating factors indicated negative
regulation of the immune system and of tumor spreading, which may
explain why NF1 has always been considered to be a benign tumor
that is less likely to become malignant.
To confirm the most likely genes contributing to the
effects on nerve growth in NF1 patients, genes that changed up to
10-fold, including the calcium related factors, formyl peptide
receptor 1 (FPR1), regulator of calcineurin 1 (RCAN1) and
diacylglycerol kinase, the inflammatory cytokines, IL1β and solute
family carrier 11, the matrix-degrading genes, a disinterin and
metalloproteinase with thrombospondin motifs 4 and matrix
metallopeptidase, the transcription modulators, HIF3α and HOXC8,
and the metabolism-related factor, ACSBG1, were investigated. With
the exception of the inflammatory cytokines and the
matrix-degrading genes, which have been reported to be involved in
tumor proliferation and metastasis, FPR1 and RCAN1 are expressed in
malignant cancers (10–12). The marked downregulation of these
genes once again provided evidence that inflammation and basement
membrane degradation are suppressed in NF1 patients, which may
reduce the risk of tumor metastasis. HIF3α acts as a negative
regulator of hypoxia, so it may induce the tolerance of the
extracellular environment (13).
Whether HIF3α is involved in promoting nerve growth remains to be
verified. HOXC8 is an essential factor in oncogenesis and tumor
metastasis. HOXC8 is downregulated in esophageal and prostate
cancers (14), but significantly
elevated in metastatic breast cancer cells (15). The complex function of HOXC8 may
indicate a temporal role for HOXC8 in tumor progression. To confirm
the screened genes that regulate nerve overgrowth, the ACSBG1,
HIF3α and HOXC8 genes that changed markedly at the transcriptional
level were checked. The results indicated that HOXC8 exhibited a
positive correlation with the differentiation of the PC12 cells,
thus it may be a potential stimulating factor involved in nerve
axon growth.
The chromosomal localization of genes exhibiting a
>5-fold change was also analyzed. This confirmed that genes
located on chromosome 1, which affect nerve system development
(16), and on chromosome 19, which
usually associate with genetic diseases (17), were mainly correlated with the
development of NF1. The key cluster of genes that stimulate nerve
growth on chromosomes 1 and 19 require identification in future
studies.
In conclusion, the etiology of NF1 is complicated
and results from changes in multiple genes. Therefore, the
identification of such genes is essential in order to explain NF1
formation, metastasis and recurrence. Through large-scale
microarray analysis of NF1 patients, possible genes that contribute
to the promotion of nerve growth during NF1 development were
investigated, which may deepen our understanding of NF1 and provide
a novel direction for the research into nerve repair and
regeneration.
Acknowledgements
The authors are grateful to the staff of the
Department of General Surgery of The Third Affiliated Hospital of
Southern Medical University. This study was supported by a grant
from the Key Supporting Project of the Jilin Provincial Science and
Technology Department (grant no. 20110456).
References
|
1
|
Lammert M, Friedman JM, Kluwe L and
Mautner VF: Prevalence of neurofibromatosis 1 in German children at
elementary school enrollment. Arch Dermatol. 141:71–74. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bausch B, Borozdin W, Mautner VF, et al;
European-American Phaeochromocytoma Registry Study Group. Germline
NF1 mutational spectra and loss-of-heterozygosity analyses in
patients with pheochromocytoma and neurofibromatosis type 1. J Clin
Endocrinol Metab. 92:2784–2792. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Evans DG, Huson SM, Donnai D, et al: A
genetic study of type 2 neurofibromatosis in the United Kingdom. I
Prevalence, mutation rate, fitness, and confirmation of maternal
transmission effect on severity. J Med Genet. 29:841–846. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rasmussen SA and Friedman JM: NF1 gene and
neurofibromatosis 1. Am J Epidemiol. 151:33–40. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bottillo I, Ahlquist T, Brekke H, et al:
Germline and somatic NF1 mutations in sporadic and NF1-associated
malignant peripheral nerve sheath tumours. J Pathol. 217:693–701.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jouhilahti EM, Peltonen S, Heape AM and
Peltonen J: The pathoetiology of neurofibromatosis 1. Am J Pathol.
178:1932–1939. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nishimura T, Ishima T, Iyo M and Hashimoto
K: Potentiation of nerve growth factor-induced neurite outgrowth by
fluvoxamine: role of sigma-1 receptors, IP3 receptors and cellular
signaling pathways. PLoS One. 3:e25582008. View Article : Google Scholar
|
|
8
|
Albini A and Sporn MB: The tumour
microenvironment as a target for chemoprevention. Nat Rev Cancer.
7:139–147. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
de Visser KE, Eichten A and Coussens LM:
Paradoxical roles of the immune system during cancer development.
Nat Rev Cancer. 6:24–37. 2006.
|
|
10
|
Rescher U, Danielczyk A, Markoff A and
Gerke V: Functional activation of the formyl peptide receptor by a
new endogenous ligand in human lung A549 cells. J Immunol.
169:1500–1504. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kim SS and Seo SR: The regulator of
calcineurin 1 (RCAN1/DSCR1) activates the cAMP response
element-binding protein (CREB) pathway. J Biol Chem.
286:37841–37848. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Blanco-Mezquita T, Martinez-Garcia C,
Proença R, et al: Nerve growth factor promotes corneal epithelial
migration by enhancing expression of matrix metalloprotease-9.
Invest Ophthalmol Vis Sci. 54:3880–3890. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Augstein A, Poitz DM, Braun-Dullaeus RC,
Strasser RH and Schmeisser A: Cell-specific and hypoxia-dependent
regulation of human HIF-3α: inhibition of the expression of HIF
target genes in vascular cells. Cell Mol Life Sci. 68:2627–2642.
2011.PubMed/NCBI
|
|
14
|
Miller GJ, Miller HL, van Bokhoven A,
Lambert JR, Werahera PN, Schirripa O, et al: Aberrant HOXC
expression accompanies the malignant phenotype in human prostate.
Cancer Res. 63:5879–5888. 2003.PubMed/NCBI
|
|
15
|
Li Y, Zhang M, Chen H, Dong Z, Ganapathy
V, Thangaraju M and Huang S: Ratio of miR-196s to HOXC8 messenger
RNA correlates with breast cancer cell migration and metastasis.
Cancer Res. 70:7894–904. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Garlow SJ, Boone E, Kinkead B and Nemeroff
CB: Genetic analysis of the hypothalamic neurotensin system.
Neuropsychopharmacology. 31:535–543. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Grimwood J, Gordon LA, Olsen A, et al: The
DNA sequence and biology of human chromosome 19. Nature.
428:529–535. 2004. View Article : Google Scholar : PubMed/NCBI
|