Introduction
Stage IV or metastatic breast cancer is the most
prevalent type of cancer worldwide, and is currently an incurable
condition with 20% of patients surviving 5 years or less after
diagnosis (1–3). Numerous studies have identified a role
for the tumor-expressed chemokine receptor CXCR4 in the regulation
of breast cancer metastasis to the axillary lymph node, bone, liver
and lung, the four most common metastatic destinations (4). These destinations, in turn, express
high levels of the CXCR4 ligand CXCL12 (reviewed in ref. 5). This study showed that a number of
primary breast cancer cells and cell lines express CXCR7, a second
receptor for CXCL12 (6). CXCR7 also
binds CXCL11; however, the binding of either chemokine ligand does
not trigger cell migration or intracellular calcium mobilization
(7). Recently, a small molecule
that blocks CXCL12 binding to CXCR7 and inhibits CXCR4-mediated
tumor cell transendothelial migration to CXCL12 was identified
(8). Small molecules that
selectively target CXCR7 may therefore prevent the metastasis of
CXCR4+CXCR7+ breast cancer cells that have
gained access to the circulatory system.
A key step in cancer cell metastasis is the
degradation of the interstitial matrix and basement membrane by
matrix metalloproteinases (MMPs), such as MMP-3. MMP-3 substrates
include collagens, gelatin, fibronectin, laminin and aggrecan;
MMP-3 is itself proteolytically cleaved and activated by MMP-2, -3,
-13 and plasmin (reviewed in ref. 9). Overexpression of an auto-activating
MMP-3 variant in mouse mammary epithelium was found to result in
neoplastic transformation of the mammary tissue (10,11).
Furthermore, high levels of MMP-3 are reported in tumor epithelial
cells and the surrounding stromal cells in human breast cancer
biopsy samples (12), as well as in
the tumor and stroma in xenografts of human breast cancer cell
lines implanted in nude mice (13).
The present study found that MMP-3 secretion was
increased in human mammary tumor cells that overexpress CXCR7. In a
xenograft model, SCID mice implanted with MDA-MB-435s human breast
cancer cells transfected with CXCR7 (435-CXCR7) tumor cells showed
elevated levels of circulating hMMP-3 relative to mice bearing
mock-transfected 435 tumors. Furthermore, in 4T1 mouse mammary
tumor cells that express endogenous CXCR7, mMMP-3 secretion was
significantly reduced following CXCR7-RNAi knockdown. Together,
these data suggest a correlation between CXCR7 expression and MMP-3
secretion in breast cancer that may contribute to metastatic
disease.
Materials and methods
Cell lines
Mouse 4T1 (4T1-WT) and human MDA-MB-435s (435-WT)
breast cancer cell lines were purchased from American Type Culture
Collection and cultured in RPMI-1640 or DMEM with 10% fetal bovine
serum (FBS), respectively. 435-CXCR7, 435-empty vector control
transfectants, and 4T1- CXCR7-RNAi-985 cell lines were generated
and previously characterized (6).
Animal experiments
Animal procedures were approved by the ChemoCentryx
Institutional Animal Care Committee. A total of 1 million 435-WT or
435-CXCR7 cells were injected subcutaneously into 6- to 8-week-old
female CB17 SCID mice (Charles River Laboratories). Animals were
sacrificed when the tumor volumes reached 1,000 mm3 or
the animals lost >20% of their initial body weight.
Protein quantification
The 435-WT or 435-CXCR7 cells were seeded at 200,000
cells/well in 6-well plates and cultured for 2 days in DMEM with 10
or 1% FBS. The conditioned media (CM) were collected and sent to
Rules-Based Medicine, Inc. (Austin, TX, USA) for multi-protein
analysis via a quantitative and multiplexed immunoassay. Human or
murine MMP-3 was measured with ELISA reagents purchased from
R&D Systems, Inc. (Minneapolis, MN, USA). In the xenograft
model, mouse serum samples were obtained after mice were sacrificed
on day 56 or 92, and serum was collected following cardiac
puncture. Human and mouse MMP-3 levels were measured in the serum
samples. 4T1-WT or 4T1-CXCR7-RNAi-985 cells were seeded at
200,000/well in 6-well plates and cultured for 2-3 days in RPMI
with 10% FBS. The CM were collected, and mMMP3 was measured using
ELISA.
Results
Secretion of MMP-3 is increased in
435-CXCR7 human breast cancer cells
In mouse tumor models, CXCR7+ breast
cancer cells are significantly more aggressive than their
CXCR7– counterparts and rapidly form large tumors when
implanted in vivo (6). To
examine whether CXCR7 overexpression results in the secretion of
factors that may enhance tumor growth, we evaluated, using a
quantitative and multiplexed immunoassay, the CM of WT MDA-MB-435s
human breast cancer cells (435-WT) and cells transfected with CXCR7
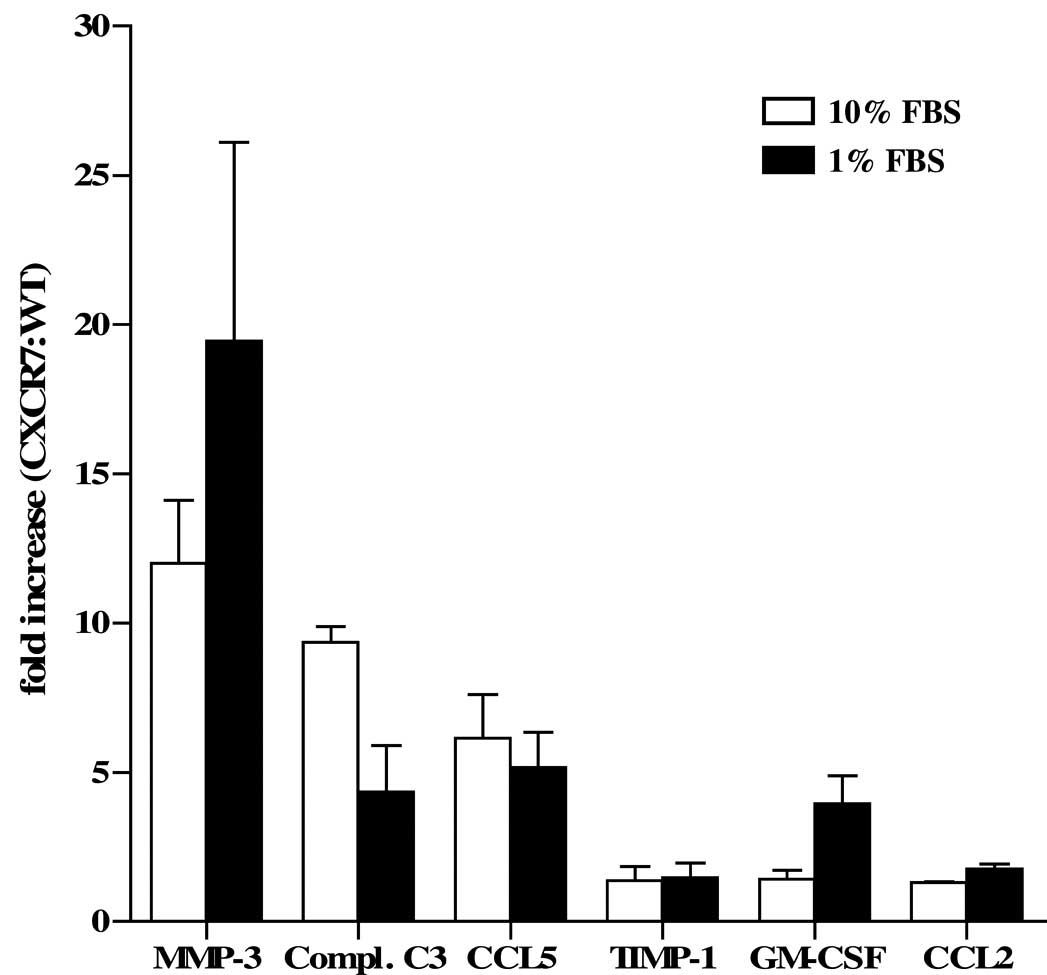
(435-CXCR7). Of the 70 analytes examined, we found the largest
differences in the levels of MMP-3, which were markedly elevated in
435-CXCR7 CM, when compared with the 435-WT CM (Table I and Fig. 1). This observation was consistent
whether the cells were grown in optimal culture conditions (10%
FBS) or exposed to environmental stress, such as serum starvation
(1% FBS) which the cells may encounter in the tumor
microenvironment (Table I and
Fig. 1). Complement C3
anaphylatoxin (Compl. C3), CCL5 (RANTES), tissue inhibitor of
metalloprotease 1 (TIMP-1), GM-CSF and CCL2 (MCP-1) were also
elevated to varying degrees in the 435-CXCR7 CM when compared to
the 435-WT CM (Table I and Fig. 1). The manner in which these
mediators contribute to tumorigenesis or cell growth has yet to be
determined. However, the complement system has recently been found
to play a role in promoting the growth of malignant cervical
carcinoma in mice through the recruitment of myeloid-derived
suppressor cells (reviewed in ref. 14).
 | Table IEvaluation of secreted
analytes.a |
Table I
Evaluation of secreted
analytes.a
| Analyte | 435- | 10% FBS (mean ±
range) | 1% FBS (mean ±
range) |
|---|
| MMP-3 (ng/ml) | X7 | 34.4±13.3 | 12.8±4.8 |
| WT | 2.8±0.6 | 0.6±0.0 |
| Compl. C3
(ng/ml) | X7 | 6.7±1.0 | 3.0±0.0 |
| WT | 0.7±0.1 | 0.7±0.3 |
| CCL5 (pg/ml) | X7 | 323.5±13.5 | 206.0±24.0 |
| WT | 56.5±15.7 | 43.3±14.6 |
| TIMP-1 (ng/ml) | X7 | 205.6±194.5 | 125.7±111.4 |
| WT | 114.7±102.4 | 67.8±53.3 |
| GM-CSF (pg/ml) | X7 | 16.2±2.7 | 28.7±7.6 |
| WT | 11.6±0.6 | 7.2±0.2 |
| CCL2 (pg/ml) | X7 | 3145±375 | 1500±40 |
| WT | 2410±230 | 851±54 |
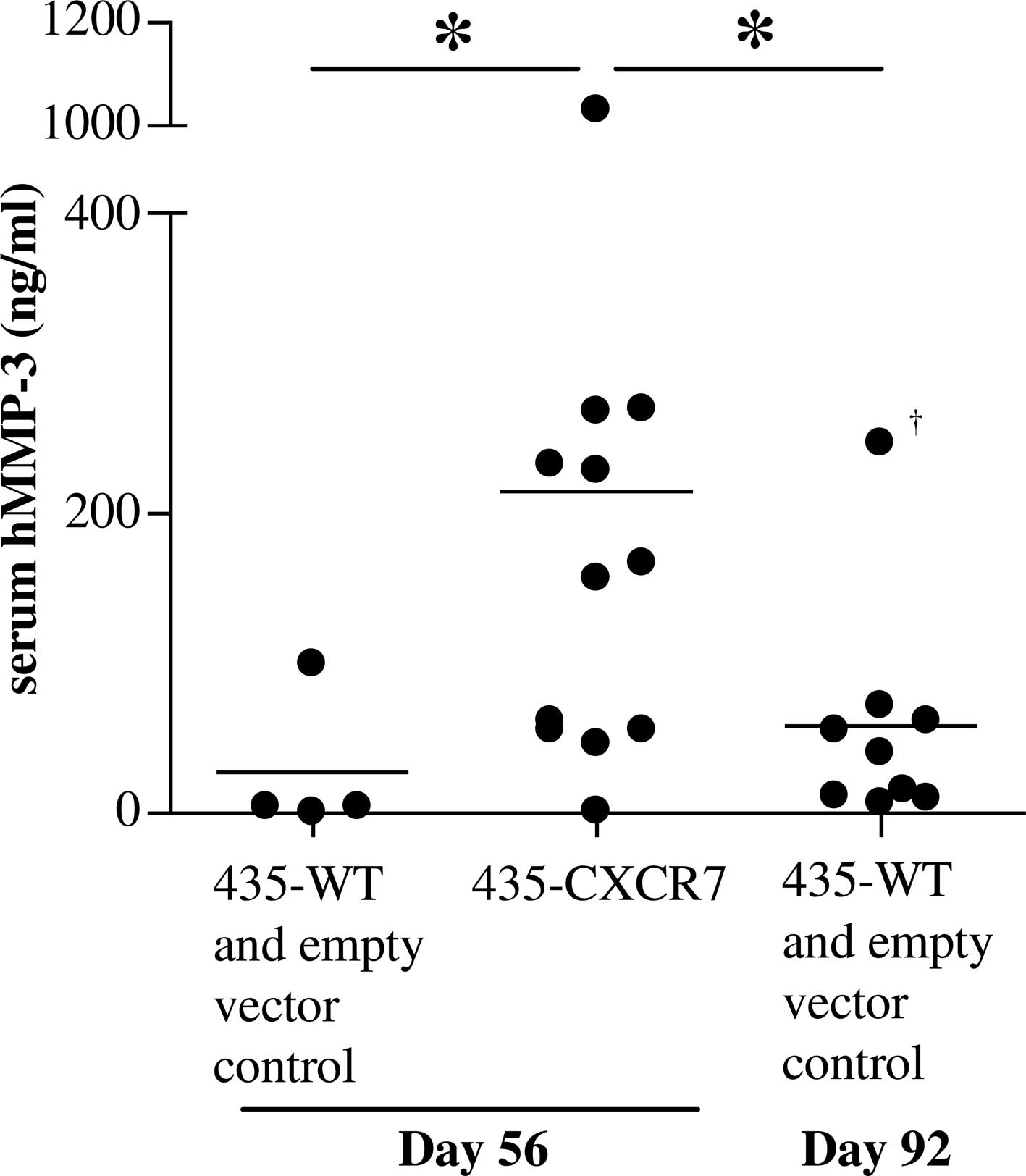
To examine whether the enhanced MMP-3 secretion is
maintained in vivo, a xenograft model was used. SCID mice
were implanted with 435-CXCR7, 435-WT or 435-empty vector tumor
cells. As previously reported, the 435-CXCR7 tumors grew
significantly more rapidly than the control tumors (6). Serum from the 435-CXCR7 mice collected
at the termination of the study had significantly higher levels of
human MMP-3 than serum from the mice implanted with control tumors
(Fig. 2). Mouse MMP-3 levels among
the groups were below the limits of detection (data not shown).
Since 435 cells are the only source of human proteins, the
overexpression of CXCR7 correlates with enhanced MMP-3 secretion in
breast cancer cells in vitro and in vivo. Since the
control tumors were smaller than the 435-CXCR7 tumors on day 56,
the control tumors were allowed to grow until day 92, when they
reached the average size (~900 mm3) comparable to that
of the 435-CXCR7 tumors on day 56 [(6) and data not shown]. These control mice
contained lower serum human MMP-3 levels than the 435-CXCR7
tumor-bearing mice (Fig. 2).
Reduced MMP-3 secretion in
CXCR7-RNAi-targeted 4T1 mouse breast cancer cells
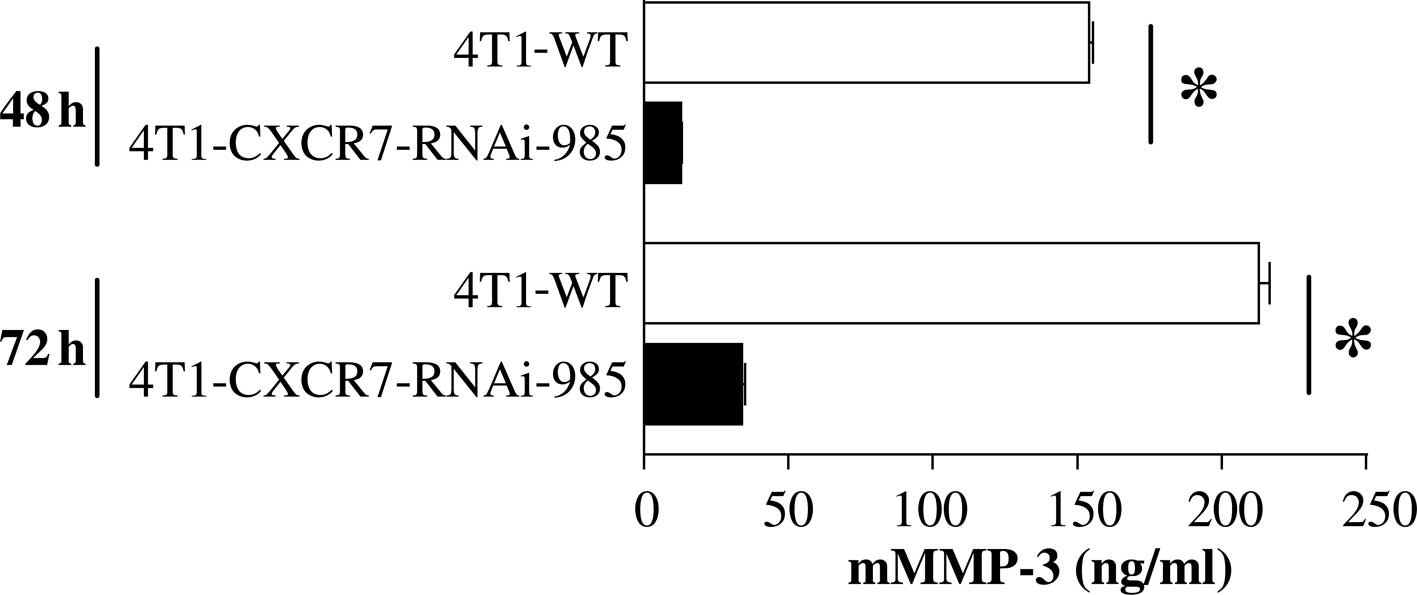
We aimed to ascertain whether inhibiting CXCR7
expression in breast cancer cells results in a reduction in MMP-3
secretion. Since 4T1 mouse mammary tumor cells endogenously express
CXCR7 protein (6), we used this
cell line to examine the effects of CXCR7-targeted RNAi knockdown
on MMP-3 release. Results showed that mMMP-3 secretion was
significantly reduced following CXCR7-RNAi knockdown (Fig. 3). Thus, using two complementary
approaches, we showed that CXCR7 expression correlates with
enhanced in vivo tumor growth (6) and MMP-3 secretion in human and mouse
breast cancer cell lines.
Discussion
Metastatic breast cancer is a leading cause of death
in women and is currently incurable (1). MMP-3 is expressed by primary breast
cancer cells and/or the underlying stroma (12,13).
Additionally, MMP-3 has been implicated as a pro-metastatic factor,
both in transforming tumor cells and in degrading the basement
membrane for subsequent tumor cell dissemination (10). CXCR7 protein is expressed in breast
cancer cells and may contribute to CXCR4-mediated tumor cell
transendothelial migration and metastasis (6,8). This
study showed that the CXCR7 protein expression correlates with
enhanced in vivo tumor growth (6) and the elevated secretion of MMP-3 by
breast cancer cells. Therapeutic agents that target CXCR7 may
therefore act on multiple pathways to prevent metastatic
disease.
Acknowledgements
This work was funded by ChemoCentryx, Inc.
References
|
1
|
Morris PG, McArthur HL and Hudis CA:
Therapeutic options for metastatic breast cancer. Expert Opin
Pharmacother. 10:967–981. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Brunello A, Roma A, Falci C and Basso U:
Chemotherapy and targeted agents for elderly women with advanced
breast cancer. Recent Pat Anticancer Drug Discov. 3:187–201. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gonzalez-Angulo AM, Morales-Vasquez F and
Hortobagyi GN: Overview of resistance to systemic therapy in
patients with breast cancer. Adv Exp Med Biol. 608:1–22. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hess KR, Varadhachary GR, Taylor SH, Wei
W, Raber MN, Lenzi R and Abbruzzese JL: Metastatic patterns in
adenocarcinoma. Cancer. 106:1624–1633. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zlotnik A: Involvement of chemokine
receptors in organ-specific metastasis. Contrib Microbiol.
13:191–199. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Miao Z, Luker KE, Summers BC, Berahovich
R, Bhojani MS, Rehemtulla A, Kleer CG, Essner JJ, Nasevicius A,
Luker GD, Howard MC and Schall TJ: CXCR7 (RDC1) promotes breast and
lung tumor growth in vivo and is expressed on tumor-associated
vasculature. Proc Natl Acad Sci USA. 104:15735–15740. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Burns JM, Summers BC, Wang Y, Melikian A,
Berahovich R, Miao Z, Penfold ME, Sunshine MJ, Littman DR, Kuo CJ,
Wei K, McMaster BE, Wright K, Howard MC and Schall TJ: A novel
chemokine receptor for SDF-1 and I-TAC involved in cell survival,
cell adhesion, and tumor development. J Exp Med. 203:2201–2213.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zabel BA, Wang Y, Lewen S, Berahovich RD,
Penfold ME, Zhang P, Powers J, Summers BC, Miao Z, Zhao B, Jalili
A, Janowska-Wieczorek A, Jaen JC and Schall TJ: Elucidation of
CXCR7-mediated signaling events and inhibition of CXCR4-mediated
tumor cell transendothelial migration by CXCR7 ligands. J Immunol.
183:3204–3211. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chakraborti S, Mandal M, Das S, Mandal A
and Chakraborti T: Regulation of matrix metalloproteinases: an
overview. Mol Cell Biochem. 253:269–285. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sternlicht MD, Lochter A, Sympson CJ, Huey
B, Rougier JP, Gray JW, Pinkel D, Bissell MJ and Werb Z: The
stromal proteinase MMP3/stromelysin-1 promotes mammary
carcinogenesis. Cell. 98:137–146. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sternlicht MD, Bissell MJ and Werb Z: The
matrix metalloproteinase stromelysin-1 acts as a natural mammary
tumor promoter. Oncogene. 19:1102–1113. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Haupt LM, Irving RE, Weinstein SR, Irving
MG and Griffiths LR: Matrix metalloproteinase localisation by in
situ-RT-PCR in archival human breast biopsy material. Mol Cell
Probes. 22:83–89. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Haupt LM, Thompson EW, Trezise AE, Irving
RE, Irving MG and Griffiths LR: In vitro and in vivo MMP gene
expression localisation by in situ-RT-PCR in cell culture and
paraffin embedded human breast cancer cell line xenografts. BMC
Cancer. 6:182006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Markiewski MM and Lambris JD: Unwelcome
complement. Cancer Res. 69:6367–6370. 2009. View Article : Google Scholar : PubMed/NCBI
|