Introduction
Pilomatrix carcinoma (PC) is a rare and malignant
adnexal tumor of hair matrix origin (1). The tumor is a dermo-hypodermic in
nature, with a low metastatic potential, but a high risk of
recurrence following excision. PC is characterized by a
locally-aggressive and low-potential malignant lesion, which is
likely to be misdiagnosed. However, distant metastasis and
mortality have also been reported (2). To highlight a further case of this
extremely rare occurrence, the present study reports a case of PC
of the parotid region that was misdiagnosed numerous times, and
reviews the pertinent literature.
Case report
A 43-year-old female first visited the Department of
Head and Neck Surgery of Jiangyou City People's Hospital (Jiangyou,
Sichuan, China) in 2009 due to a tenacious subcutaneous nodule that
was 1.0 cm in diameter on the left side of the parotid region. The
small, subcutaneous nodule was asymptomatic, and had been slowly
increasing in size for the past 5 years. The nodule was surgically
excised under local anesthesia with a false clinical diagnosis of
an adenolymphoma of the parotid gland. However, 1 year later, a
tenacious mass with an unclear boundary reappeared at the previous
site. A secondary excision was performed at the same hospital, and
the histological evaluation revealed a squamous epithelial cell
mass and cells with hyperchromatic nuclei among fibrous tissue,
reactive hyperplasia of the lymph node and partial epithelial
proliferation among the tissues. A gray-black nodule with swelling
and effusion appeared 3 months later at the site of the primary
lesion. The patient was admitted to the same hospital once more. A
biopsy of the nodes revealed a well-differentiated squamous cancer.
The patient was then transferred to our hospital for further
diagnosis and therapy.
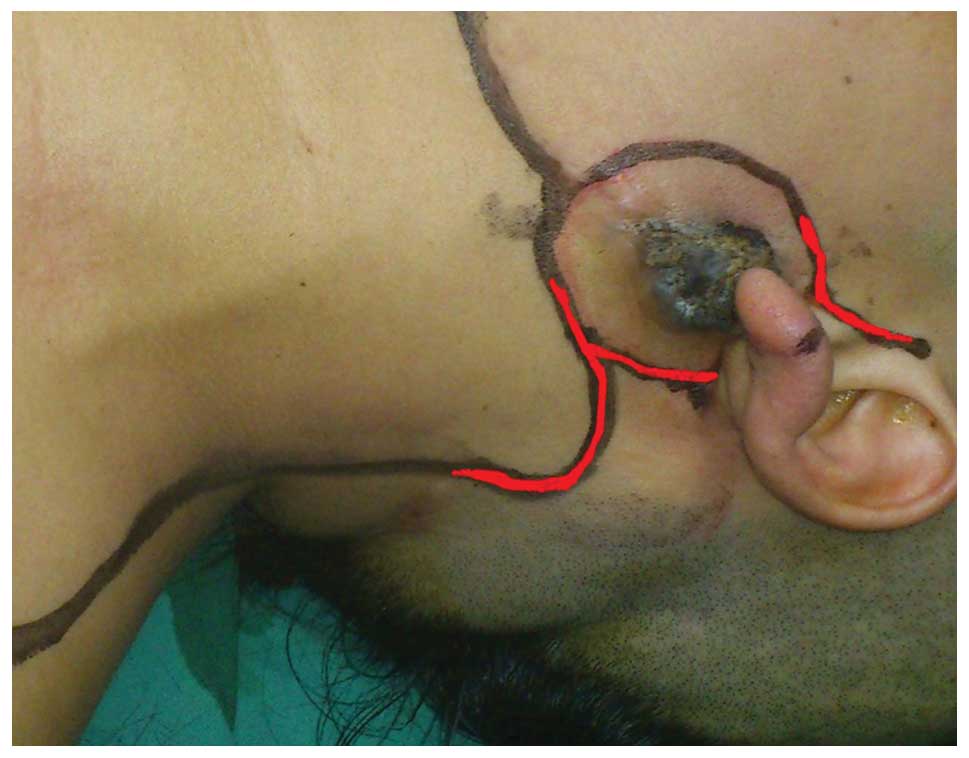
Upon physical examination, a 2.0×2.0-cm, tender,
firm, non-fluctuant, gray-black lesion was noted on the skin
surface of the left side of the parotid region (Fig. 1). There was no palpable
lymphadenopathy of the left side of the neck. Sensation around the
area and salivation were normal. There was no facial nerve
dysfunction or detectable adenopathy. The remainder of the physical
examination was otherwise unremarkable, as well as the results of
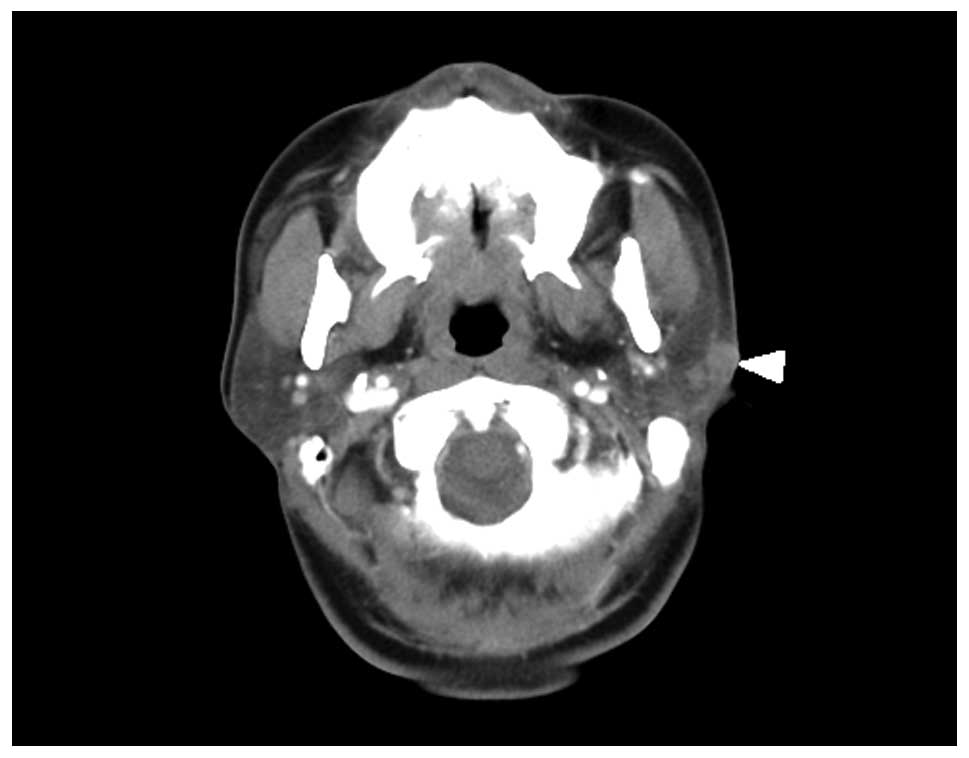
routine laboratory tests and chest X-rays. Computed tomography
revealed a solid mass measuring 2.0×2.1×1.5 cm, which infiltrated
the left parotid gland (Fig. 2). As
the exact nature of the lesion was not certain, frozen sections
were analyzed: Microscopic examination revealed that the tumor was
composed of sheets and nests of basaloid cells with prominent
central necroses and nuclear pleomorphism, and a diagnosis of
cancer, likely to be a pigmented basal cell carcinoma, was
determined. The selected treatment strategy consisted of resection
of the whole tumor together with the left parotid gland,
submaxillary salivary gland and cervical lymph nodes of the left
side of the neck. The tumor was located subcutaneously, reaching
the parotid fascia, buccal branches and ramus marginalis mandibulae
nervi facialis. After confirming a complete resection
histologically, a bilobed flap reconstruction was immediately
performed to cover the resection area (Fig. 1). The resected tumor was submitted for
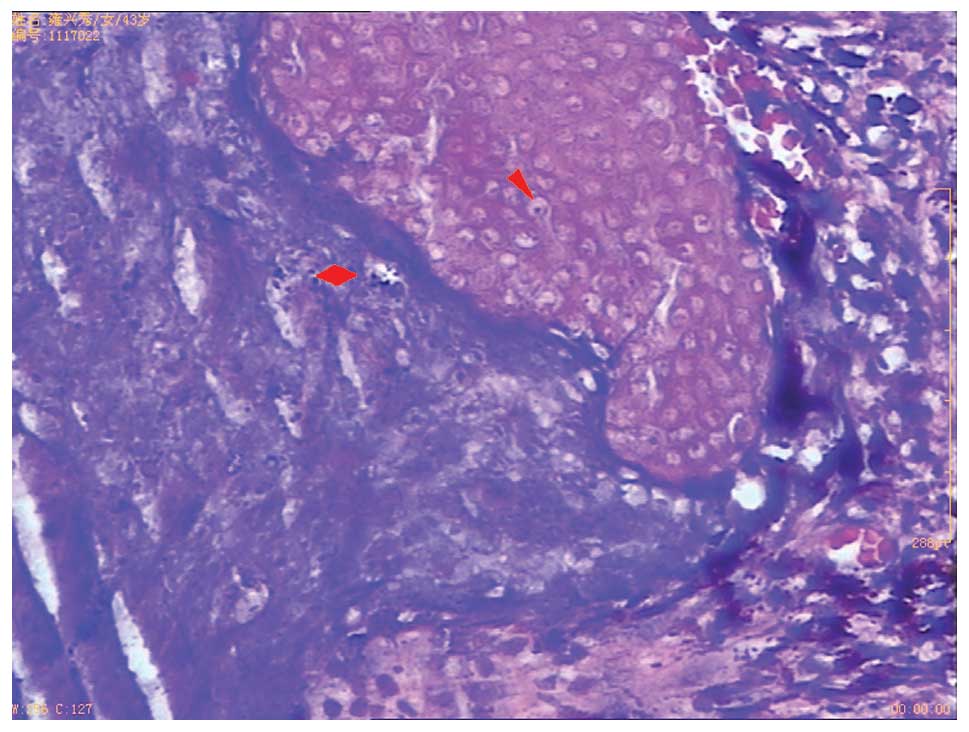
pathological examination. Microscopic examination revealed that the
tumor was composed of sheets and nests of basaloid cells with
prominent central necrosis and nuclear pleomorphism, and numerous
shadow cells in the necrotic area (Fig.
3). A diagnosis of PC was therefore formed. The patient
experienced an uneventful post-operative course and excellent wound
healing, with nearly symmetrical facial nerve function. Due to the
previous recurrences following multiple surgeries, the patient
received fractionated external beam radiation therapy (RT) to the
left parotid region and neck at a total dose of 50 Gy in 25
fractions. Short-term follow-up, including clinical and ultrasound
examinations, was performed for the early detection of possible
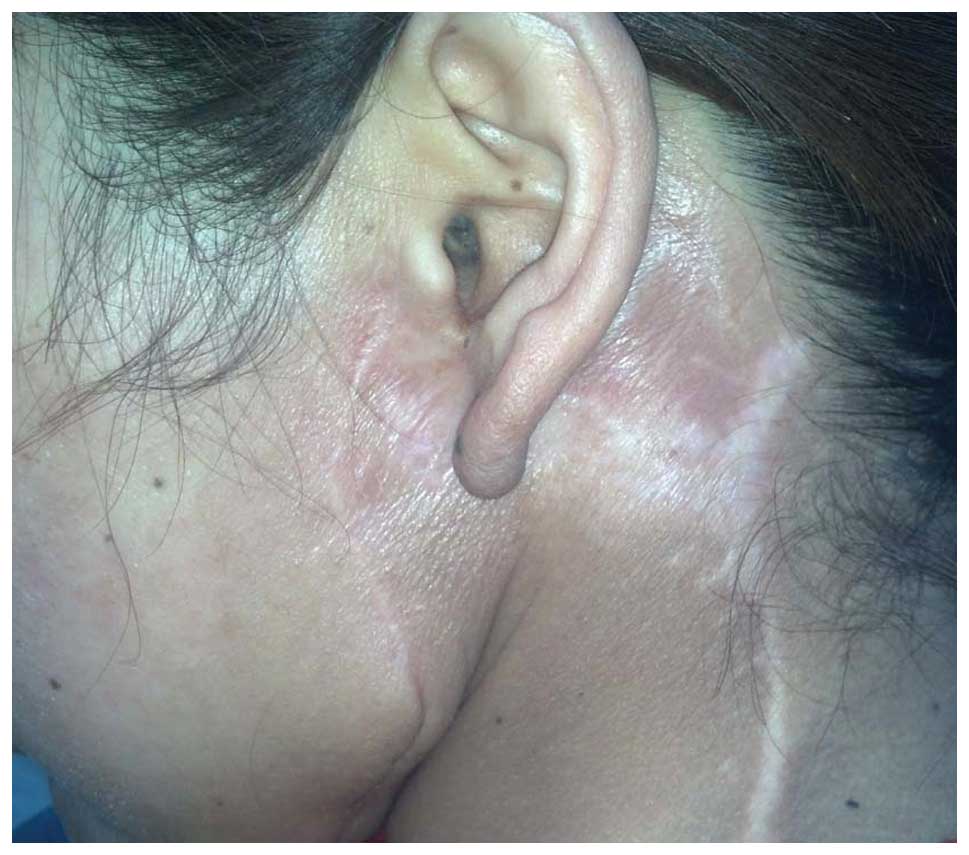
tumor recurrence at 12-week intervals. At the 2-year post-RT
follow-up, a routine pathological and ultrasound examination
demonstrated that the patient remained free of local recurrence and
metastasis (Fig. 4).
The protocol for the present retrospective study was
approved by the Ethics Committee of Sichuan Cancer Hospital, and
written informed consent was obtained from the study patient.
Discussion
Since PC was initially described by Lopansri and
Mihm in 1980, only 136 cases of PC (including 15 cases published in
Chinese with English abstracts) have been reported in the English
literature worldwide (3–5). Another 28 such cases have been reported
in the Chinese literature. To the best of our knowledge, only a few
cases have been reported in the parotid region (6–8). Although
PC occurs more commonly in males than in females (ratio, 3:1) in
the published English literature excluding Chinese cases, the rate
is approximately 1:1 (22:23) in the Chinese cases (including the
present case). The patients in these previously reported cases
range in age from 14 months to 93 years, with a mean of 46.3 years
(9). The most common site of PC is
the head and neck, occurring in 60% of patients, followed by the
upper extremities, trunk and lower extremities (10).
PC manifests clinically as a multicolored,
asymptomatic cystic or solid mass. On presentation, PC varies in
size from 0.5–20.0 cm (average, 3.6 cm; median, 2.5 cm) and the
lesion can be present from several months to years prior to
treatment. Due to a lack of clear histological criteria to
distinguish PC from other matrical tumors, the diagnosis of this
neoplasm can be challenging. PC tumors consist of pleomorphic
basaloid cells with prominent nucleoli and frequent mitoses (a
number of which are atypical) accompanied by central areas with
keratotic material, shadow cells and foci of necrosis (11). The transition to squamous cells, clear
cells, areas of necrosis and frequent mitoses is associated with
metrical cornification in PC (3).
Additional characteristics include the appearance of an
infiltrative pattern that is asymmetric with frequent invasion of
fat and other underlying structures, such as vascular, lymphatic or
perineural invasion (3,12). In the present case, sheets and nests
of basaloid cells with prominent central necroses and nuclear
pleomorphism, typical shadow cells, atypical mitoses and
infiltration of the parotid gland and nervi facialis were
observed.
The differential histological diagnosis is necessary
to exclude other tumors derived from hair follicles, including
proliferating pilar cysts, basal cell carcinoma with metrical
differentiation, pilomatrixoma, trichoepithelioma, squamous cell
carcinoma, lymphoepithelioma-like carcinoma of the skin and mixed
tumors of the skin (13,14). A proliferating trichilemmal cyst
comprises lobules or sheets of squamous nests with trichilemmal
keratinization, however, ghost cells or basaloid cells are absent
in these lesions (14). Proliferating
pilomatrixoma and basal cell carcinoma with matrical
differentiation may resemble PC clinically and histologically.
However, PC can be distinguished from these tumors by the
appearance of sheets and islands of basaloid matrical cells with
considerable nuclear pleomorphism and an asymmetric infiltrative
pattern, with frequent invasion of fat and other underlying
structures (12). Trichoepitheliomas
consist of islands of basaloid cells, which may contain shadow
cells and keratin horn cysts. However, characteristics of
malignancy, including infiltrative growth, necrosis, prominent
nucleoli and pleomorphism, are absent in these tumors (14). Squamous cell carcinoma may contain
cytological features similar to the basaloid cells of PC, but lack
ghost cells (15).
Lymphoepithelioma-like carcinoma contains epithelial cell islands
that may resemble PC. However, the tumors lack shadow cells or
squamous differentiation, while PC lacks a prominent
lymphoplasmacytic infiltrate (14).
Shadow cells in large horn cysts may be present in mixed tumors of
the skin, however, characteristics of malignancy, including
infiltrative growth, necrosis, prominent nucleoli and pleomorphism,
are absent (15). Due to the rarity
of PC, the difficulty of diagnosis and the lack of experience in
such cases, the present case was misdiagnosed numerous times. As
this tumor is inclined to arise in the head and neck region,
particularly the parotid region, the rare occurrence and atypical
presentation of such tumors pose a diagnostic dilemma, often
resulting in a misdiagnosis of primary parotid neoplasm. In the
present case, the PC was misdiagnosed as adenolymphoma. Diagnostic
imaging may be of use in the clinical setting in order to clearly
demonstrate the association between the mass and the parotid
parenchyma (16), prior to parotid
infiltration.
The ability of PC for infiltration both laterally
and in depth is significant. Local relapses are frequent, as
observed in the present case. PC can disseminate via the lymphatic
or the blood system, and forms metastases. As PC is a rare entity,
the standards of surgical management are not well-defined.
Therefore, the preferred treatment method involves excision of the
tumor with wide safety margins. Previous studies have recorded
margins ranging from 0.5–3.0 cm where feasible (14,15,17). In
the present case, a complete tumor resection was performed using a
wide local resection with clear margins (1.0–2.0 cm), and the
larger defect was reconstructed by a bilobed flap. With regard to
RT, only a limited number of cases employing the treatment have
been described and its role in the therapy for PC remains unclear,
mainly due to the lack of experience and diverse results reported
following its use (18). Tselis et
al summarized 6 cases that had been treated initially with RT
or with RT following surgery, and observed no local recurrence
after 2 years of follow-up (18). The
present patient was administered a total RT dose of 50 Gy as a
post-operative supplementary treatment, and has remained free of
local recurrence and metastasis for 2 years. We suggest that RT may
therefore be an effective method for the treatment of PC. As a
result of prior therapy, the tumor bed is generally more hypoxic
with recurrent lesions. The higher radiation doses that are applied
with interstitial high dose rate brachytherapy may be of advantage
in treating not only recurrent PC, but also residual or newly
diagnosed PC (18). Certain authors
consider that chemotherapy has not been proven effective (1,3). However,
to the best of our knowledge, systemic chemotherapy has been used
in 2 cases of metastatic PC, and these cases have exhibited a
response (1,19).
In conclusion, PC is a rare malignant tumor that is
easily misdiagnosed. The histological diagnosis, based on several
factors (shadow cells, necrosis, nuclear atypia, infiltration and
abnormal mitosis) is difficult to prove. An optimal treatment
regimen has not been established. A surgical procedure with wide
margins is recommended to avoid recurrence when the staging shows
no metastasis. RT has provided mixed results and has been most
commonly used as an adjuvant therapy for PC. Chemotherapy is
generally considered to be ineffective, although 2 cases of
metastatic PC have shown a response.
Acknowledgements
The study was supported by grants from the Sichuan
Provincial Science and Technology Department (no. 2012JY0125) and
the Sichuan Provincial Bureau of Health (nos. 110259, 090538 and
130230).
References
|
1
|
Eluecque H, Gisquet H, Kitsiou C, et al:
Pilomatrix carcinoma: A case report. J Clin Exp Dermatol Res.
3:1–3. 2012.
|
|
2
|
Sassmannshausen J and Chaffins M:
Pilomatrix carcinoma: A report of a case arising from a previously
excised pilomatrixoma and a review of the literature. J Am Acad
Dermatol. 44((2 Suppl): 358–361. 2001.PubMed/NCBI
|
|
3
|
Melancon JM, Tom WL, Lee RA, et al:
Management of pilomatrix carcinoma: A case report of successful
treatment with Mohs micrographic surgery and review of the
literature. Dermatol Surg. 37:1798–1805. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wang X, Ma JM and Wang N: Malignant
pilomatricoma in the upper eyelid. BMJ Case Rep.
2009.bcr07.2008.0410. 2009.
|
|
5
|
Li X, Jiang H and Li A:
Clinicopathological study on 15 cases of pilomatrix carcinoma.
Zhonghua Bing Li Xue Za Zhi. 26:100–102. 1997.(In Chinese).
PubMed/NCBI
|
|
6
|
Karaaslan O, MelihCan M, Ozlem Karatas
Silistreli A, Kaan Bedir Y and Caliskan G: Malignant pilomatrixoma
arising on the previously irradiated face: Case report and
literature review. J Cutan Med Surg. 16:341–343. 2012.PubMed/NCBI
|
|
7
|
Joshi A, Sah SP, Agrawal CS, Jacob M and
Agarwalla A: Pilomatrix carcinoma in a child. Acta Derm Venereol.
79:476–477. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Carbonaro V, Pietribiasi F and Penno A:
Malignant pilomatrixoma of the face. Acta Otorhinolaryngol Ital.
17:444–447. 1997.(In Italian). PubMed/NCBI
|
|
9
|
Hardisson D, Linares MD, Cuevas-Santos J
and Contreras F: Pilomatrix carcinoma: A clinicopathologic study of
six cases and review of the literature. Am J Dermatopathol.
23:394–401. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Aherne NJ, Fitzpatrick DA, Gibbons D,
Collins CD and Armstrong JG: Recurrent malignant pilomatrixoma
invading the cranial cavity: Improved local control with adjuvant
radiation. J Med Imaging Radiat Oncol. 53:139–141. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Jani P, Chetty R and Ghazarian DM: An
unusual composite pilomatrix carcinoma with intralesional
melanocytes: Differential diagnosis, immunohistochemical
evaluation, and review of the literature. Am J Dermatopathol.
30:174–177. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kaddu S, Soyer HP, Wolf IH and Kerl H:
Proliferating pilomatricoma. A histopathologic simulator of
matrical carcinoma. J Cutan Pathol. 24:228–234. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
de Gálvez-Aranda MV, Herrera-Ceballos E,
Sánchez-Sánchez P, Bosch-García RJ and Matilla-Vicente A:
Pilomatrix carcinoma with lymph node and pulmonary metastasis:
Report of a case arising on the knee. Am J Dermatopathol.
24:139–143. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cornejo KM and Deng A: Pilomatrix
carcinoma: A case report and review of the literature. Am J
Dermatopathol. 35:389–394. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Caubet-Biayna J, Ramos-Asensio R, Ortabe I
and Mas M: Pilomatrix carcinoma of the face. J Oral Maxillofac
Surg. 57:609–611. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cozzi DA, d'Ambrosio G, Cirigliano E, et
al: Giant pilomatricoma mimicking a malignant parotid mass. J
Pediatr Surg. 46:1855–1858. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sable D and Snow SN: Pilomatrix carcinoma
of the back treated by mohs micrographic surgery. Dermatol Surg.
30:1174–1176. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tselis N, Heyd R, Vogt HG and Zamboglou N:
Pilomatrix carcinoma with lymph node and pulmonary metastases.
Strahlenther Onko. 182:727–732. 2006. View Article : Google Scholar
|
|
19
|
Kim KU, Lee MK, Kim YS, et al: Pilomatrix
carcinoma with lung and lymph node metastases. J Lung Cancer.
7:90–92. 2008. View Article : Google Scholar
|