Introduction
Celastrus orbiculatus belongs to the family
Celastraceae and the genus Celastrus (1). The stem, roots and leaves of this plant
are used as a folk medicine to treat rheumatoid arthritis (2). Celastrus and various of its
defined constituents possess anti-cancer, anti-inflammatory and
anti-oxidant properties (1–8). Our previous studies demonstrated that
the ethyl acetate extract of Celastrus orbiculatus (COE) has
significant anti-tumor effects in vitro and in vivo
(1,3,4,9–12). COE
induced cytotoxicity and promoted the apoptosis of human
hepatocellular carcinoma cells by inhibiting the Akt signaling
pathway (9), and inhibited tumor
angiogenesis by modulating the vascular endothelial growth factor
signaling pathway (10). COE reduced
the invasion and migration of gastric adenocarcinoma MGC-803 cells
by reducing the expression and enzymatic activity of matrix
metallopeptidase-9, which was regulated by inactivation of the
phosphatidylinositol-4,5-bisphosphate 3-kinase (PI3K)/Akt signaling
pathway and nuclear factor-κB activity (11). COE also displayed anti-metastatic
activity on human gastric adenocarcinoma by inhibiting the
epithelial-mesenchymal transition (12). In previous experiments, the present
authors also observed that COE induced autophagy and apoptosis in
HT-29 cells; however, the effectiveness of COE induced-autophagy
and its cellular mechanism in colorectal cells is still
unknown.
Cell death can occur by apoptosis [also known as
programmed cell death (PCD) I] or by necrosis and autophagy (which
is known as PCD II) (13,14). Drugs that induce apoptosis remain the
most common chemotherapeutic agents used in medical oncology
(13). Cancer cells hijack cell
processes to survive, allowing them to escape from apoptosis and to
acquire drug resistance (13). Drugs
having both apoptotic and autophagic activities offer an added
advantage to overcome these escape mechanisms. Autophagy is
normally a degradative mechanism for the removal and turnover of
bulk cytoplasmic constituents through the endosomal/lysosomal
system (14–16); however, it is also an adaptive
response to environmental changes, including nutrient deprivation,
hypoxia and cell injury (17–19). Previous studies revealed that
autophagy was involved in the cell death induced by anti-cancer
drugs such as 5-fluorouracil and rapamycin (20,21), and
that autophagy may improve the efficacy of chemotherapy by
enhancing the apoptosis of cells (22–24). By
contrast, it has also been shown that autophagy is important in
promoting cell survival against apoptosis (25–28).
Despite this dual role of autophagy, the tissue/cancer-specific
role of autophagy should be further explored to determine its
potential as a therapeutic strategy for cancer and other human
diseases. Thus, the aim of the present study was to investigate the
effect of COE on autophagy and apoptotic cell death mechanisms, as
well as their possible association in HT-29 cells treated with
COE.
Materials and methods
Plant material
The stems of C. orbiculatus plants
(production batch no. 070510) were purchased from Zhixin
Pharmaceutical Co., Ltd. (Guangzhou, China). The COE was prepared
and characterized at the Department of Chinese Materia Medica
Analysis, China Pharmaceutical University (Nanjing, China). The
chemical constituents and preparation procedure of COE were
described previously (29,30). Prior to being used to treat cells, COE
was dissolved in dimethyl sulfoxide (DMSO) and diluted to different
concentrations. The final concentration of DMSO in the cell medium
did not exceed 0.1%.
Chemical reagents and antibodies
Antibodies against Beclin 1 (cat. no. 3495), light
chain (LC) 3 (cat. no. 12741), B-cell lymphoma (Bcl)-2 (cat. no.
2872) and Bcl-2-associated X (Bax; cat. no. 2772), Akt (cat. no.
4691), Akt-Ser308 (cat. no. 9275), mechanistic target of rapamycin
(mTOR; cat. no. 2983), mTOR-Ser2448 (cat. no. 5536), p70 ribosomal
protein S6 kinase (p70S6K; cat. no. 2708) and p70S6K-Thr389 (cat.
no. 9234) were purchased from Cell Signaling Technology, Inc.
(Danvers, MA, USA). The enhanced chemiluminescence (ECL) kit was
purchased from GE Healthcare Life Sciences (Chalfont, UK). DMSO and
MTT were purchased from Sigma-Aldrich (Merck Millipore, Darmstadt,
Germany). Other reagents were purchased from Beyotime Institute of
Biotechnology (Jiangsu, China).
Cell culture
The human colorectal cancer cell line HT-29 was
acquired from the Cell Bank of Chinese Academy of Sciences,
Shanghai Institute of Cell Biology (Shanghai, China). HT-29 cells
were cultured in RPMI-1640 medium (Gibco; Thermo Fisher Scientific,
Inc., Waltham, MA, USA) containing 10% fetal bovine serum (Gibco;
Thermo Fisher Scientific, Inc.) and maintained at 37°C in a
humidified incubator in an atmosphere of 5% CO2.
Cell viability assay
Cell viability was measured by MTT assay. HT-29
cells were grown until logarithmic phase, and then seeded in a
96-well plate at a density of 1×104 cells/well overnight
prior to treatment. The cells were then incubated with different
concentrations of COE (0, 20, 40, 80, 160 and 320 mg/l) in
triplicate. After 24, 48 and 72 h of incubation, the cells were
incubated with medium containing MTT for 4 h, and the formazan
crystals were dissolved with 150 µl DMSO. The plates were incubated
on an agitator for 15 min at room temperature, and the absorbance
was measured at 490 nm with a microplate reader. The drug
concentration at which the cell viability was reduced to 50%
[defined as the half maximal inhibitory concentration
(IC50)] by 24 h of treatment was then determined. The
effects of COE on cell viability were also determined in the
absence or presence of the autophagy inhibitor 3-methyladenine
(3-MA) (5 mM; Sigma-Aldrich; Merck Millipore) 3 h prior to COE
treatment or the autophagy inducer rapamycin (100 nM;
Sigma-Aldrich; Merck Millipore) 1 h prior to COE treatment to
investigate the effects of COE-induced autophagy on the viability
of HT-29 cells.
Annexin V-fluorescein isothiocyanate
(FITC)/propidium iodide (PI) dual staining assay
Apoptosis was detected with the Annexin V-FITC/PI
Apoptosis Detection kit (Nanjing KeyGen Biotech Co., Ltd., Nanjing,
China). In brief, after 24 h of treatment with different
concentrations of COE, cells were washed twice with ice-cold PBS
and re-suspended in 500 µl binding buffer. A total of 5 µl annexin
V-FITC and 5 µl PI were added to the cell suspension and then
incubated for 15 min at room temperature in the dark. FITC and PI
fluorescence was analyzed in a FACSort flow cytometer (BD
Biosciences, Franklin Lakes, NJ, USA) with CellQuest Pro software
(BD Biosciences). The results were generated from three independent
experiments.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Following treatment with COE, total RNA was
extracted from the cells using the TRIzol reagent (Invitrogen;
Thermo Fisher Scientific, Inc.) under RNase-free conditions, and RT
was performed in a 20-µl reaction with 200 ng total RNA using the
Two-Step RT-PCR kit (Takara Biotechnology Co., Ltd., Dalian,
China). RT-qPCR was performed on a 7500 Real-Time PCR System
(Applied Biosystems; Thermo Fisher Scientific, Inc.) using the SYBR
Premix Ex Taq kit (Takara Biotechnology Co., Ltd.) in
Axygen® 96-well reaction plates (Thermo Fisher
Scientific, Inc.). PCR was performed under the following
conditions: Initiation step of 95°C for 10 min, denaturation step
of 95°C for 15 sec, and the annealing and extension step at 60°C
for 60 sec for 40 cycles.
Primers were obtained from Shanghai Shenggong
Biology Engineering Technology Service, Ltd. (Shanghai, China), and
their sequences were: Beclin 1 (NM_003093), forward primer
5′-GGCTGAGAGACTGGATCAGG-3′ and reverse primer
5′-CTGCGTCTGGGCATAACG-3′; LC3-II (NM_003094), forward primer
5′-GAGAAGCAGCTTCCTGTTCTGG-3′ and reverse primer
5′-GTGTCCGTTCACCAACAGGAAG-3′; and GAPDH (NM_002046), forward primer
5′-TGGCACCCAGCACAATGAA-3′ and reverse primer
5′-CTAAGTCATAGTCCGCCTAGA-3′. GAPDH was used as an internal control,
and the data were analyzed using the 2−ΔΔCq method
(31).
Ultrastructures observed by
transmission electron microscopy (TEM)
Following treatment with COE, the cells were washed
with PBS, collected by centrifugation at 1,500 × g for 5 min
at 4°C, and fixed in 2.5% electron microscopy-grade glutaraldehyde
(SenBeiJia Biological Technology Co. Ltd., Nanjing, China). The
specimens were subsequently rinsed with 0.1 M PBS, fixed in 1%
osmium tetroxide (Nanjing Zhongjingkeyi Technology Co., Ltd.,
Nanjing, China), dehydrated through a graded series of ethanol and
processed for Epon™ embedding (Sigma-Aldrich; Merck Millipore).
Ultra-thin sections (60 nm) stained with uranyl acetate and lead
citrate (Nanjing Zhongjingkeyi Technology Co., Ltd.) were observed
with a JEM-1230 electron microscope (JEOL, Ltd., Tokyo, Japan).
Detection of acidic vascular
organelles (AVOs) with acridine orange
To detect the AVOs in COE-treated cells in the
presence of 3-MA and rapamycin, cells were stained with acridine
orange (Beyotime Institute of Biotechnology). In acridine
orange-stained cells, the cytoplasm fluoresces bright green,
whereas the acidic compartments fluoresce bright red (20). The intensity of the red fluorescence
is proportional to the degree of acidity and/or the volume of the
cellular acidic compartment (20).
Therefore, changes in the degree of acidity and/or the fractional
volume of the cellular acidic compartments can be determined.
Following treatment with COE for 24 h, cells were fixed with
chilled 70% ethanol and stained with 5 mM acridine orange.
Fluorescence was then observed using a fluorescence microscope
(BX51; Olympus Corporation, Tokyo, Japan).
Western blot analysis
The expression levels of Beclin 1, LC3, Bax, Bcl-2
and mTOR-p70S6K signaling proteins (Akt, Akt-308, mTOR,
mTOR-Ser2448, p70S6K and p70S6K-Thr389) in HT-29 cells were
determined by western blot analysis. Briefly, a cell lysis solution
was prepared using Cytoplasmic Extraction Reagent II (Fermentas;
Thermo Fisher Scientific, Inc., Pittsburgh, PA, USA). A 50-µg
sample of supernatant was subjected to 10% SDS-PAGE and then
transferred onto nitrocellulose membranes (EMD Millipore,
Billerica, MA, USA). The blot was blocked with 5% not-fat dry milk
for 2 h at room temperature, and then incubated with primary
antibodies (1:1,000) and agitated overnight at 4°C. Subsequently,
the membranes were washed three times with washing buffer for 10
min and incubated with secondary antibodies (1:1,000; G130321;
Hangzhou Huaan Biotechnology Co., Ltd., Hangzhou, China) for 2 h at
room temperature. β-actin (a housekeeping gene) was used as a
loading control. ECL was used to detect the signals, using the
SuperSignal West Pico Chemiluminescent Substrate (Thermo Fisher
Scientific, Inc.) on a Molecular Imager ChemiDoc XRS System
(Bio-Rad Laboratories, Inc., Hercules, CA, USA). Densitometry was
determined using Quantity One 1-D analysis software (Bio-Rad
Laboratories, Inc.).
Statistical analysis
Results are expressed as the mean ± standard
deviation. Each experiment was repeated ≥3 times. Statistical
comparisons of ≥2 groups were conducted using analysis of variance
followed by a Bonferroni post hoc test. All statistical analyses
were performed using the SPSS 18.0 statistical software (SPSS,
Inc., Chicago, IL, USA), and P<0.05 was considered to indicate a
statistically significant difference.
Results
COE inhibits the proliferation of
HT-29 cells
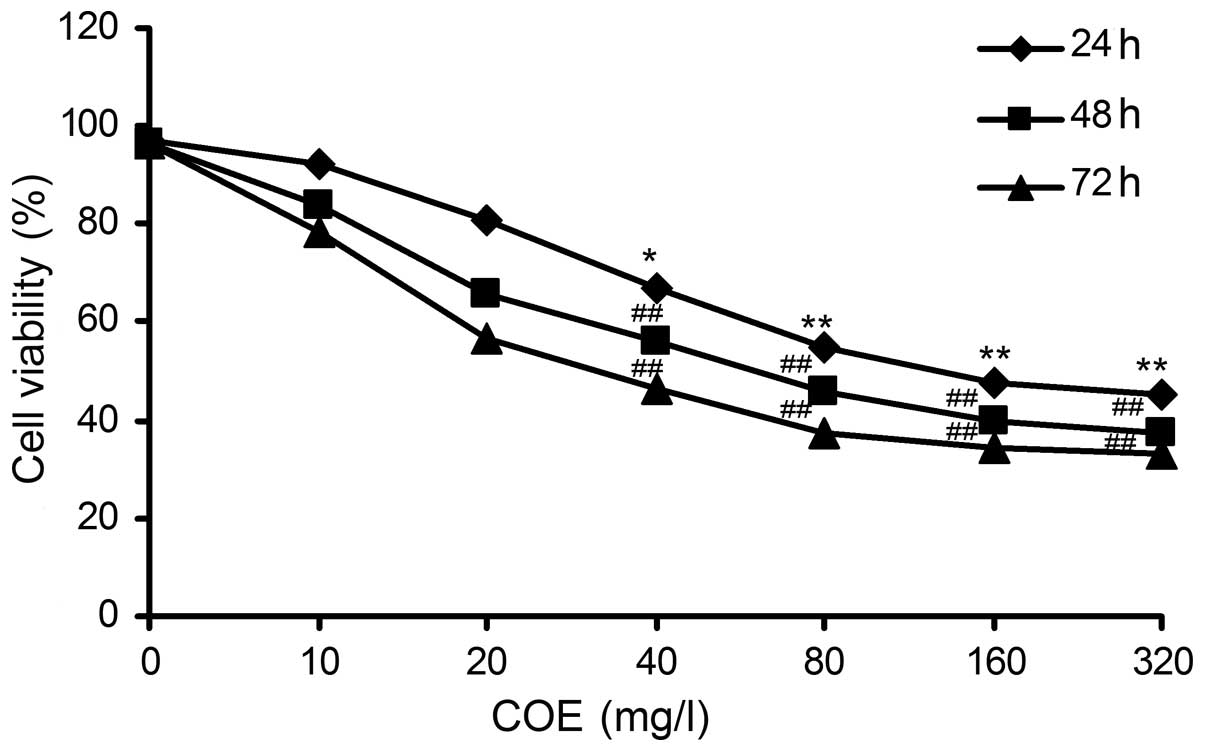
The results of the MTT assay revealed that HT-29
cell viability decreased in a dose-dependent manner upon
administration of COE. A strong dose-dependent inhibition of cell
proliferation was also observed in the HT-29 cell line. Upon
treatment with 149.65±0.49 mg/l of COE for 24 h, the cell viability
was reduced to 50% (Fig. 1). These
results indicate that COE exerts a potent anti-proliferative
activity against colorectal cancer cells.
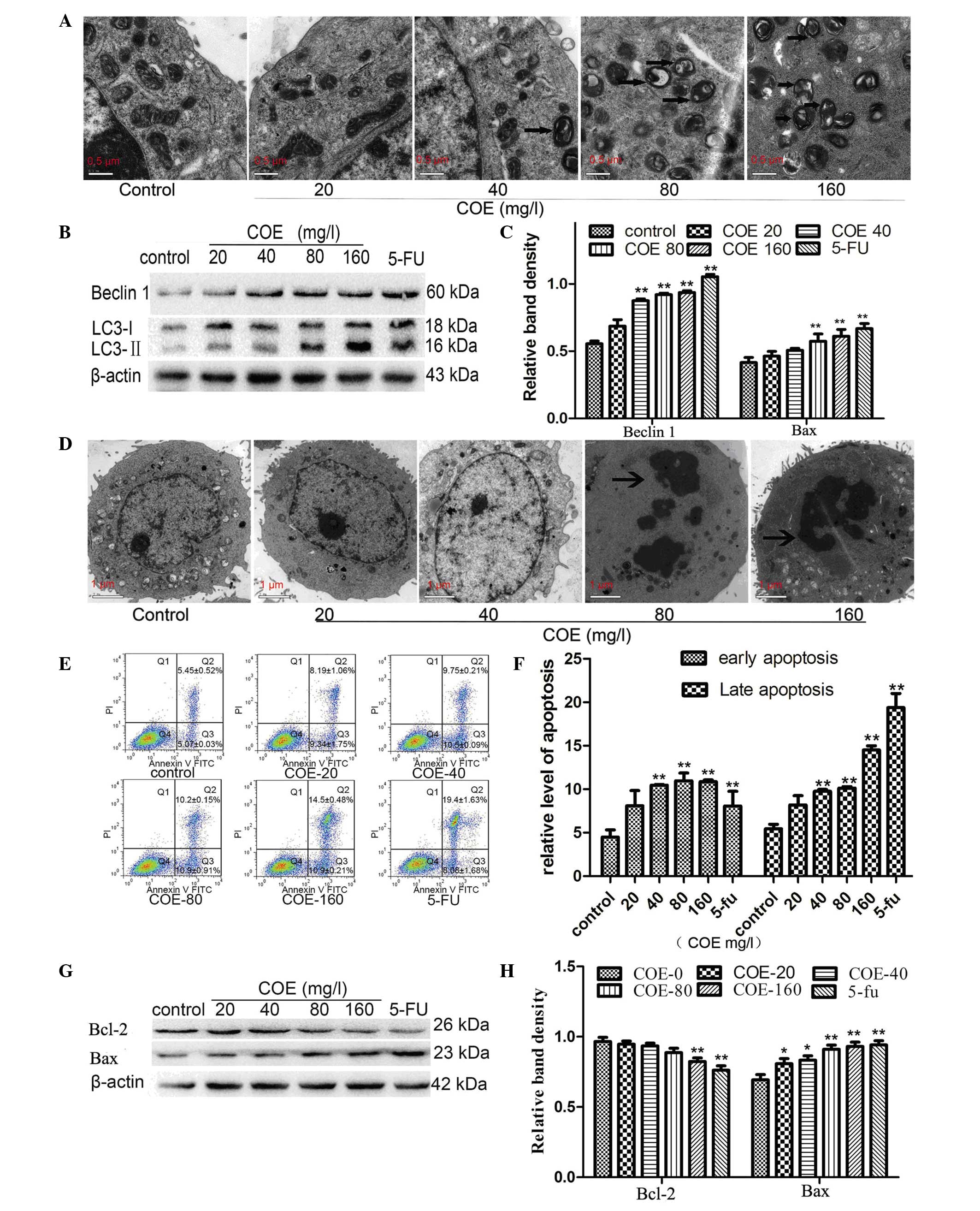
COE induces autophagy
The induction of autophagy is characterized by the
formation of autophagosomes enclosed in a double membrane that fuse
with lysosomes to form autolysosomes (17,18).
Electron microscopy observation of these cellular structures is the
gold standard for detecting autophagy (22). In the present study, cells in the
control group presented intact organelles. By contrast, large
autophagosomes and autolysosomes were present in HT-29 cells
treated with 80 or 160 mg/l COE for 24 h (Fig. 2A).
Under normal conditions, the protein LC3 is
dispersed throughout the cytoplasm in a dissociated form (LC3-I)
(21). During autophagy induced by
factors such as nutrient depletion, LC3-I is converted to its
phosphatidylethanolamine-conjugated form (LC3-II), which localizes
to both sides of the autophagosome (14). Beclin 1 is also indispensable for
autophagy induction (13). The
western blotting results revealed that the levels of LC3-II and
Beclin 1 increased significantly in HT-29 cells treated with 80 or
160 mg/l COE for 24 h compared with control cells (Fig. 2B and C). No significant change was
observed for LC3-II or Beclin 1 messenger RNA transcript
levels.
COE induces apoptosis in colorectal
cancer cells
As shown in Fig. 2D,
ultrastructures in HT-29 cells were observed by TEM after 24 h of
COE treatment at different concentrations. Cells in the control
group exhibited intact organelles and round shape with nucleus and
nucleolus chromatin. However, upon treatment with 80 or 160 mg/l
COE, cell shrinkage, chromatin condensation and nuclear collapse
were observed.
The percentage of total apoptotic cells was
calculated by adding the percentages of early apoptotic-gated cells
[annexin V+, quadrant (Q) 3 area] and late
apoptotic-gated cells (annexin V+/PI+, Q2
area) (Fig. 2E). The results revealed
a progressive increment in the cell population at the right-side
coordinates upon COE administration in a dose-dependent manner
compared with the control group (Fig. 2E
and F).
Members of the Bcl-2 family serve a vital role in
the regulation of apoptosis (32,33). Thus,
western blot analysis was used to determine the levels of Bax and
Bcl-2. Compared with the control group, Bcl-2 expression decreased
with increasing concentrations of COE, while Bax expression
increased in a dose-dependent manner (Fig. 2G and H).
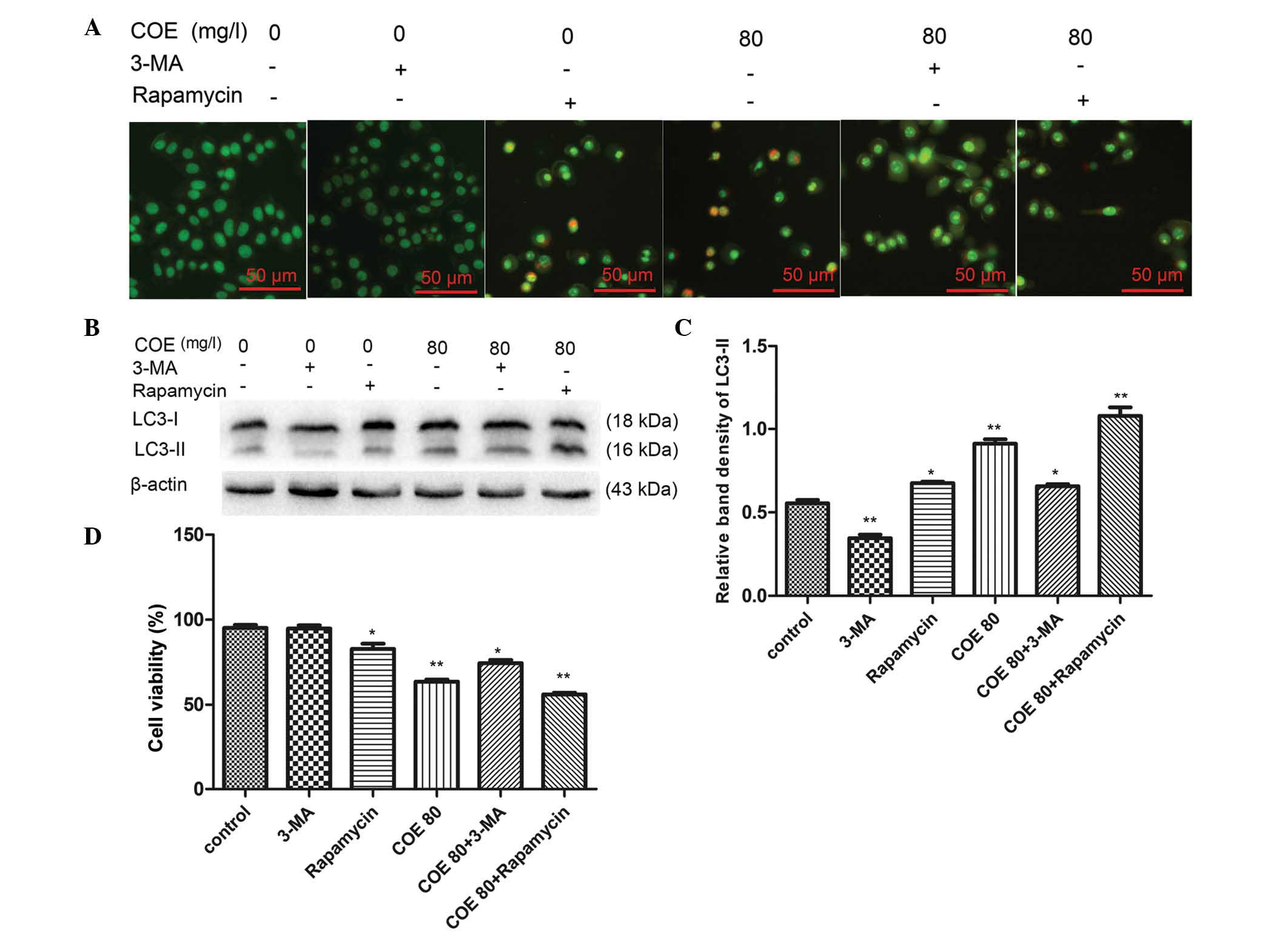
COE-induced cell death involves
autophagy in HT-29 cells
Our data revealed that <20 mg/l COE had no
significant impact on cell viability, while 80 mg/l COE
significantly inhibited HT-29 cell viability (P<0.01; Fig. 1). Based on the IC50 value,
80 mg/l COE was selected explore the effects of COE-induced
autophagy on HT-29 cell viability. To confirm whether COE-induced
autophagy was inhibited or enhanced by 3-MA or rapamycin,
respectively, AVOs were observed by fluorescence microscopy. The
results revealed that AVO formation induced by 80 mg/l COE was
reduced in the presence of 3-MA and increased in the presence of
rapamycin (Fig. 3A). Additionally,
western blot analysis demonstrated that the level of LC3-II was
increased in the presence of rapamycin and decreased in the
presence of 3-MA upon treatment with 80 mg/l COE (Fig. 3B and C). In addition, to confirm
whether COE-induced autophagy increased cell survival or cell
death, a cell viability assay was performed following treatment of
HT-29 cells with 80 mg/l COE in the presence of 3-MA (an autophagy
inhibitor) or rapamycin (an autophagy inducer) (13,22). The
ability of COE to inhibit proliferation was reduced in the presence
of 3-MA and enhanced in the presence of rapamycin (Fig. 3D). These results indicate that
COE-induced autophagy enhanced cell death rather than cell
survival.
The PI3K-Akt-mTOR signaling pathway
mediates COE cytotoxicity
To determine whether the PI3K-Akt-mTOR signaling
pathway serves a role in mediating COE-induced apoptosis and
autophagy, the levels of Akt, mTOR and p70S6K were determined by
western blotting. Following COE treatment, there was a robust and
sustained decrease in Akt-Thr308, mTOR-Ser2448 and p70S6K-Thr389
levels in HT-29 cells (Fig. 4A and
B). This indicated that COE may inhibit the proliferation of
HT-29 cells by mediating the phosphorylation of Akt and reducing
the kinase activity of mTOR. The reduction in mTOR activity
promoted autophagy in HT-29 cells.
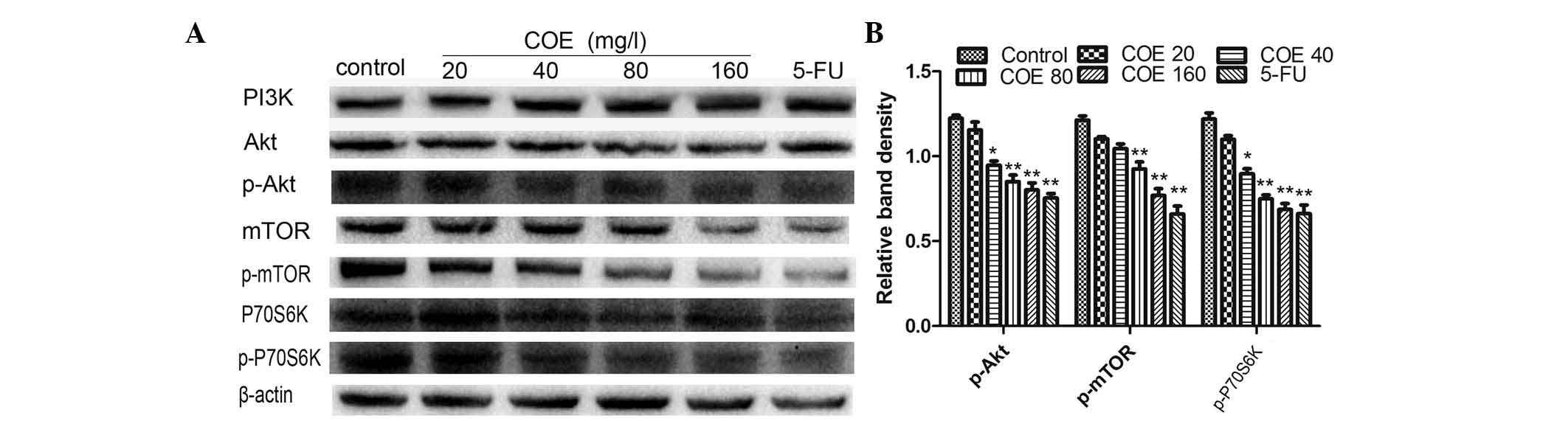 | Figure 4.Protein expression of the
PI3K/Akt/mTOR signaling pathway components in HT-29 cells treated
with different concentrations of COE for 24 h. (A) The expression
levels of PI3K, Akt, Akt-Ser308, mTOR, mTOR-Ser2448, p70S6K and
p70S6K-Thr389 were analyzed by western blotting. (B) The band
intensities of Akt-Ser308, mTOR-Ser2448 and p70S6K-Thr389 relative
to the untreated control cells were determined upon normalizing to
β-actin expression, and are expressed as the mean ± standard
deviation of three independent experiments. *P<0.05 and
**P<0.01 compared with the untreated control. p, phosphorylated;
FU, fluorouracil; PI3K, phosphatidylinositol-4,5-bisphosphate
3-kinase; mTOR, mechanistic target of rapamycin; p70S6K, p70
ribosomal protein S6 kinase; COE, Celastrus orbiculatus
extract. |
Discussion
As surgery and adjuvant chemotherapy are associated
with low survival rates in colorectal carcinoma, there is an urgent
requirement to identify novel therapeutic agents for this type of
cancer (34). An ideal anti-cancer
drug should initiate PCD in cancer cells, and consistent with this
expectation, COE was observed to initiate this type of cell death
(25,34). In the current study, the
anti-proliferative effects of COE were first examined. After
incubation with different concentrations of COE for 24, 48 and 72
h, the proliferation of HT-29 cells was significantly inhibited in
a time- and dose-dependent manner. When HT-29 cells were incubated
with 80 or 160 mg/l COE for 24 h, autophagy and apoptosis were
significantly induced (Fig. 2A and
B).
Autophagy is normally considered to be a cell
survival mechanism induced by stress, starvation and other
environmental cues, but recent reports have provided evidence of
cell death mediated by autophagy (13,15).
Numerous tumor suppressors, including death-associated protein
kinase, phosphatase and tensin homolog and p53, can effectively
stimulate autophagy in cancer cells (26). A previous study demonstrated that
knocking down autophagy-related (Atg) 7, an important gene involved
in autophagy, leads to uncontrolled proliferation of liver cancer
cells beyond a normal lifespan in mice (35). Autophagy is now considered to be one
of the most important types of PCD (type II PCD) (32). It begins with sequestering cytoplasmic
organelles in a double-membrane vacuole called autophagosome
(13,14). Autophagosomes then fuse with lysosomes
to form autolysosomes, where the internal materials are degraded
and recycled (17). Detection of the
autophagosome and autolysosomes inside cells is the best way to
confirm induction of autophagy (17,19). In
the present study, observation of HT-29 cells by electron
microscopy revealed that more autophagosomes and autolysosomes were
present in cells treated with 80 or 160 mg/l COE for 24 h COE than
in cells treated with 0, 20, 40 mg/l COE for 24 h (Fig. 2A). LC3, a mammalian homolog of yeast
Atg8, is another reliable marker of autophagosomes. During
autophagy, the cytoplasmic form of LC3 (LC3-I, 18 kDa) is processed
and recruited to autophagosomes, where the LC3-II form (16 kDa) is
generated by site-specific proteolysis and lipidation near the
C-terminus of LC3-I (19,20). In the present study, immunoblotting of
LC3 revealed a significant conversion of LC3-I to the
autophagosomal membrane-bound LC3-II upon treatment with 80 or 160
mg/l COE for 24 h (Fig. 2E and G).
Beclin 1 is part of the class III PI3K lipid kinase complex, and
plays a central role in the induction of autophagy (33). Immunoblot analysis demonstrated that
the expression of Beclin 1 increased in COE-treated cells (Fig. 2E and G), suggesting that COE-induced
autophagy was associated with augmented Beclin 1 expression. Taken
together, these findings confirmed the induction of autophagy in
HT-29 cells treated with COE.
It is well established that autophagy, mainly a
self-digestion process, degrades intracellular structures in
response to stress, leading to either cell survival or cell death
(15,16). To determine whether COE-induced
autophagy increased cell survival or cell death, HT-29 cells were
treated with 80 mg/l COE for 24 h in the presence of 3-MA, a
pharmacologic autophagy inhibitor that inhibits class III PI3K,
which is known to inhibit autophagic sequestration (13,22). The
results revealed that treatment with 3-MA reduced the effect of COE
on cell viability (Fig. 3D) and AVO
formation (Fig. 3A). Furthermore,
immunoblot analysis demonstrated that the expression of LC3-II was
downregulated in cells treated with 80 mg/l COE for 24 h in the
presence of 3-MA compared with that in cells treated with COE alone
(Fig. 3C). By contrast, AVO formation
increased upon treatment with 80 mg/l COE in the presence of
rapamycin in HT-29 cells (Fig. 3A),
and the expression of LC3-II was upregulated (Fig. 3C). Of particular significance is the
marked reduction in cell viability observed when autophagy was
induced by COE administration in the presence of rapamycin
(Fig. 3D). This finding confirmed
that COE-induced autophagy did not enhance survival pathways but
instead increased cell death in human colorectal cancer cells.
Our experimental results also demonstrated that, in
addition to activating autophagy, COE induced pro-apoptotic
signaling pathways leading to cell death. Electron microscopy
observation of the COE-treated HT-29 cells revealed chromatin
condensation, nuclear collapse and apoptotic body formation in
cells treated with 80 or 160 mg/l COE (Fig. 2B). Apoptotic cell death was also
quantified by determining the percentage of early apoptotic-gated
cells (annexin V+) and late apoptotic-gated cells
(annexin V+/PI+). The results revealed a
progressive increase in the percentage of apoptotic cells in a COE
dose-dependent manner (Fig. 2C and
D). Bcl-2 and Bax are the key proteins for controlling the
release of cytochrome c and other pro-apoptotic factors from
mitochondria, which leads to caspase activation and apoptotic cell
death (36). The present study
demonstrated that COE significantly decreased the expression of
Bcl-2 but increased the expression of Bax (Fig. 2F and H). This finding indicated that
COE induced apoptosis by regulating the expression of the Bcl-2
family of proteins.
To further understand the mechanism of apoptosis and
autophagy induced by COE, the PI3K/Akt/mTOR pathway was explored,
which is frequently deregulated in cancer and is important for
tumorigenesis, since this pathway can modulate apoptosis and
autophagy (13,15). mTOR exists in two conserved protein
complexes, mTORC complex (mTORC) 1 and mTORC2 (21,36,37).
mTORC1 acts as a nutrient sensor and has been described as the
master regulator of autophagy (37).
When Akt is hyper-activated, mTORC1 can phosphorylate and activate
mTOR, with the consequent inhibition of autophagy and apoptosis
(38,39). In the present study, immunoblot
analysis revealed a robust and sustained decrease in Akt-Thr308,
mTOR-Ser2448 and p70S6K-Thr389 levels upon treatment with
increasing doses of COE (Fig. 4),
indicating that COE may inhibit the proliferation of HT-29 cells by
mediating the phosphorylation of Akt and reducing the kinase
activity of mTOR. The reduction in mTOR activity promotes autophagy
in HT-29 cells.
In conclusion, COE-induced autophagy and apoptosis
synergize to inhibit colorectal cancer growth. The present study
provides novel evidence that COE may be a promising agent against
human colorectal cancer, particularly in light of the fact that
cancer cells can escape induction of apoptosis, resulting in drug
resistance. The present study may aid in the development of new
strategies for the formulation of effective therapeutic drugs that
would selectively target cancer cells and induce autophagic cell
death independently of the normal apoptotic pathway.
Acknowledgements
The present study was financially supported by
grants from the National Natural Science Foundation of China
(Beijing, China; grant nos. 81403232, 81274141 and 81450051) and
the Natural Science Foundation of Jiangsu Province of China
(Nanjing, China; grant nos. BK 2012686 and SBK 2014021480).
References
|
1
|
Guo YQ, Li X, Xu J, Li N, Meng DL and Wang
JH: Sesquiterpene esters from the fruits of Celastrus orbiculatus.
Chem Pharm Bull (Tokyo). 52:1134–1136. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jin HZ, Hwang BY, Kim HS, Lee JH, Kim YH
and Lee JJ: Antiinflammatory constituents of Celastrus orbiculatus
inhibit the NF-kappa B activation and NO production. J Nat Prod.
65:89–91. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li GQ, Liu D, Zhang Y, Qian Y, Zhang H,
Guo S, Sunagawa M, Hisamitsu T and Liu Y: Celastrol inhibits
lipopolysaccharide-stimulated rheumatoid fibroblast-like
synoviocyte invasion through suppression of TLR4/NF-κB-mediated
matrix metalloproteinase-9 expression. PloS One. 8:e689052013.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Li GQ, Liu D, Zhang Y, Qian YY, Zhu YD,
Guo SY, Sunagawa M, Hisamitsu T and Liu YQ: Anti-invasive effects
of celastrol in hypoxia-induced fibroblast-like synoviocyte through
suppressing of HIF-1α/CXCR4 signaling pathway. Int Immunopharmacol.
17:1028–1036. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Spivey AC, Weston M and Woodhead S:
Celastraceae sesquiterpenoids: Biological activity and synthesis.
Chem Soc Rev. 31:43–59. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kim SE, Kim YH, Lee JJ and Kim YC: A new
sesquiterpene ester from Celastrus orbiculatus reversing multidrug
resistance in cancer cells. J Nat Prod. 61:108–111. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Westerheide SD, Bosman JD, Mbadugha BN,
Kawahara TL, Matsumoto G, Kim S, Gu W, Devlin JP, Silverman RB and
Morimoto RI: Celastrols as inducers of the heat shock response and
cytoprotection. J Biol Chem. 279:56053–56060. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nam NH: Naturally occurring NF-kappaB
inhibitors. Mini Rev Med Chem. 6:945–951. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang H, Qian Y, Liu Y, Li G, Cui P, Zhu
Y, Ma H, Ji X, Guo S and Tadashi H: Celastrus orbiculatus extract
induces mitochondrial-mediated apoptosis in human hepatocellular
carcinoma cells. J Tradit Chin Med. 32:621–626. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Qian YY, Zhang H, Hou Y, Yuan L, Li GQ,
Guo SY, Hisamits T and Liu YQ: Celastrus orbiculatus extract
inhibits tumor angiogenesis by targeting vascular endothelial
growth factor signaling pathway and shows potent antitumor activity
in hepatocarcinomas in vitro and in vivo. Chin J Integr Med.
18:752–760. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhu YD, Liu YQ, Qian YY, Zhang H, Li GQ
and Yang L: Extracts of Celastrus orbiculatus exhibit
anti-proliferative and anti-invasive effects on human gastric
adenocarcinoma cells. Chin J Integr Med. 2014.(Epub ahead of
print). View Article : Google Scholar
|
|
12
|
Zhu Y, Liu Y, Qian Y, Dai X, Yang L, Chen
J, Guo S and Hisamitsu T: Antimetastatic effects of Celastrus
orbiculatus on human gastric adenocarcinoma by inhibiting
epithelial-mesenchymal transition and NF-κB/snail signaling
pathway. Integr Cancer Ther. 14:271–281. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lefranc F, Facchini V and Kiss R:
Proautophagic drugs: A novel means to combat apoptosis-resistant
cancers, with a special emphasis on glioblastomas. Oncologist.
12:1395–1403. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Levine B and Klionsky DJ: Development by
self-digestion: Molecular mechanisms and biological functions of
autophagy. Dev Cell. 6:463–477. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Levine B and Kroemer G: Autophagy in the
pathogenesis of disease. Cell. 132:27–42. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mizushima N, Levine B, Cuervo AM and
Klionsky DJ: Autophagy fights disease through cellular
self-digestion. Nature. 451:1069–1075. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li J, Hou N, Faried A, Tsutsumi S and
Kuwano H: Inhibition of autophagy augments 5-fluorouracil
chemotherapy in human colon cancer in vitro and in vivo model. Eur
J Cancer. 46:1900–1909. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sharma N, Thomas S, Golden EB, Hofman FM,
Chen TC, Petasis NA, Schönthal AH and Louie SG: Inhibition of
autophagy and induction of breast cancer cell death by mefloquine,
an antimalarial agent. Cancer Lett. 326:143–154. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Xi G, Hu X, Wu B, Jiang H, Young CY, Pang
Y and Yuan H: Autophagy inhibition promotes paclitaxel-induced
apoptosis in cancer cells. Cancer Lett. 307:141–148. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pan X, Zhang X, Sun H, Zhang J, Yan M and
Zhang H: Autophagy inhibition promotes 5-fluorouraci-induced
apoptosis by stimulating ROS formation in human non-small cell lung
cancer A549 cells. PloS One. 8:e566792013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ravikumar B, Vacher C, Berger Z, Davies
JE, Luo S, Oroz LG, Scaravilli F, Easton DF, Duden R, O'Kane CJ and
Rubinsztein DC: Inhibition of mTOR induces autophagy and reduces
toxicity of polyglutamine expansions in fly and mouse models of
Huntington disease. Nat Genet. 36:585–595. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cui Q, Tashiro S, Onodera S, Minami M and
Ikejima T: Autophagy preceded apoptosis in oridonin-treated human
breast cancer MCF-7 cells. Biol Pharm Bull. 30:859–864. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Iwamaru A, Kondo Y, Iwado E, Aoki H,
Fujiwara K, Yokoyama T, Mills GB and Kondo S: Silencing mammalian
target of rapamycin signaling by small interfering RNA enhances
rapamycin-induced autophagy in malignant glioma cells. Oncogene.
26:1840–1851. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Maiuri MC, Le Toumelin G, Criollo A, Rain
JC, Gautier F, Juin P, Tasdemir E, Pierron G, Troulinaki K,
Tavernarakis N, et al: Functional and physical interaction between
Bcl-X(L) and a BH3-like domain in Beclin-1. EMBO J. 26:2527–2539.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shintani T and Klionsky DJ: Autophagy in
health and disease: A double-edged sword. Science. 306:990–995.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Levine B and Yuan J: Autophagy in cell
death: An innocent convict? J Clin Invest. 115:2679–2688. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kenific CM, Thorburn A and Debnath J:
Autophagy and metastasis: Another double-edged sword. Curr Opin
Cell Biol. 22:241–245. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lock R and Debnath J: Extracellular matrix
regulation of autophagy. Curr Opin Cell Biol. 20:583–588. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li JJ, Yang J, Lu F, et al: Chemical
constituents from the stems of Celastrus Orbiculatus. Chin J Nat
Med. 10:279–283. 2012. View Article : Google Scholar
|
|
30
|
Zan K, Chen X-Q, Wang Q, et al: Chemical
constituents in stem of Celastrus orbiculatus. Chin Trad Herbal
Drugs. 38:14552007.
|
|
31
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tait SW, Ichim G and Green DR: Die another
way-non-apoptotic mechanisms of cell death. J Cell Sci.
127:2135–2144. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Levine B, Sinha S and Kroemer G: Bcl-2
family members: Dual regulators of apoptosis and autophagy.
Autophagy. 4:600–606. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ferlay J, Autier P, Boniol M, Heanue M,
Colombet M and Boyle P: Estimates of the cancer incidence and
mortality in Europe in 2006. Ann Oncol. 18:581–592. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ramakrishnan S, Nguyen TM, Subramanian IV
and Kelekar A: Autophagy and angiogenesis inhibition. Autophagy.
3:512–515. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Thomas S, Quinn BA, Das SK, Dash R, Emdad
L, Dasgupta S, Wang XY, Dent P, Reed JC, Pellecchia M, et al:
Targeting the Bcl-2 family for cancer therapy. Expert Opin Ther
Targets. 17:61–75. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Jung CH, Ro SH, Cao J, Otto NM and Kim DH:
mTOR regulation of autophagy. FEBS Lett. 584:1287–1295. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Maiso P, Liu Y, Morgan B, Azab AK, Ren P,
Martin MB, Zhang Y, Liu Y, Sacco A, Ngo H, et al: Defining the role
of TORC1/2 in multiple myeloma. Blood. 118:6860–6870. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Corradetti MN and Guan KL: Upstream of the
mammalian target of rapamycin: Do all roads pass through mTOR?
Oncogene. 25:6347–6360. 2006. View Article : Google Scholar : PubMed/NCBI
|