Introduction
Locally advanced cervical cancer (LACC), which is
classified by International Federation of Gynecology and Obstetrics
(FIGO, 2009) as stages IB2-IVA, is a group of high risk cervical
cancers with poor prognosis (1). For
young patients with LACC, neoadjuvant chemotherapy is now becoming
a preferable therapy option prior to radical surgery (2,3).
Nevertheless, the systemic adverse reactions due to
chemotherapeutics, the low complete remission rate from single
chemotherapeutics as well as tumor cell resistance all hamper the
efficacy of chemotherapy. Therefore, more effective treatment
strategies are still required. Wild-type p53 (wt-p53) gene is an
essential cancer suppressor gene. Evidence generated over the past
decade has shown that the p53 tumor suppressor is the most
frequently altered gene in >50% of all types of human cancers.
In addition, gene therapy with wt-p53 has emerged as a promising
antitumor strategy for some malignant tumors (4,5). Wt-p53 is
a ‘housekeeping gene’ and functions as an inhibitor of
carcinogenesis by maintaining genomic integrity; the wt-p53 protein
is a primary mediator of cell cycle arrest, DNA repair and
apoptosis. The inactivated p53 protein and the aberrant p53 tumor
suppressor pathway plays important roles in the pathogenesis of
cervical cancer. It would be interesting to reconstruct the p53
pathway by gene therapy. In our previous study, we confirmed that
recombinant human adenovirus-p53 (rhAd-p53) could inhibit the
proliferation of HeLa cells in vitro and the p53 agent
combined with chemotherapeutics significantly inhibited cancer cell
proliferation and effectively induced apoptosis (6). The p53 agent potentiated the sensitivity
of cancer cells to chemotherapy. However, there has been a paucity
of reports on the clinical applications of p53 agent to cervical
cancer. In this study, rhAd-p53 was intratumorally injected into
LACC in patients of FIGO stages IB2-IIIA, coupled with PVB
chemotherapy, which may be able to provide a novel clinical regimen
for LACC.
Materials and methods
Patients and tissues
A total of 40 patients with cervical cancer of FIGO
stages IB2-IIIA hospitalized in the Maternity and Child Health Care
Hospital Affiliated to Xuzhou Medical College (Xuzhou, China) were
recruited. Experimental protocol was approved by the hospital
Ethics Committee, and written informed consent was obtained from
each patient. Inclusion criteria: Premenopausal patients aged ≤45
years, cervical squamous cell carcinoma diagnosed by cervix biopsy,
expected survival of at least six months, lesions clinically
measurable and feasible for drug injection, no prior chemotherapy
and/or radiotherapy, normal functions of heart, lungs, liver and
kidneys, void of contraindications to chemotherapy.
A total of 40 patients were randomized into 2 groups
(n=20 each): PVB group (cisplatin + vincristine + bleomycin,
intravenously) and combined group (rhAd-p53 gene therapy + PVB
chemotherapy). Both groups underwent chemotherapy, the only
exception was the administration of the rhAd-p53 solution
1×1012 VP intratumorally at an interval of 3 days thrice
in the combined group.
According to the 2009 amendments to the clinical
staging criteria by FIGO, the 40 patients were classified IB2 (6
cases), stage IIA (13 cases), stage IIB (18 cases) and stage IIIA
(3 cases). The PVB group was aged from 28 to 45 years (mean age, 40
years); their FIGO clinical staging was IB2 (4 cases), IIA (10
cases), IIB (6 cases) and IIIA (0 case); their pre-treatment tumor
size was averaged at 19.91±5.40 cm2. The combined group
was aged from 32 to 45 years (mean age, 42 years); their FIGO
clinical staging was IB2 (2 cases), IIA 3 cases, IIB (12 cases),
IIIA (3 cases); their pre-treatment tumor size was averaged at
22.76±6.02 cm2. There were no significant differences in
age and FIGO clinical staging (P>0.05) or in pre-treatment tumor
size (t=1.573, P=0.124) between the two groups.
Tissue samples were obtained from patients in both
the PVB and combined group (n=20 each) that were undergoing tumor
resection. A total of 18 samples were obtained from control
patients with cervical cancer who did not undergo chemotherapy,
radiotherapy or gene therapy.
Regimens and efficacy assessments
rhAd-p53 injections (1×1012 VP/ampule)
(Shenzhen SiBiono GeneTech Co., Ltd., Shenzhen, China), approved by
the China Food and Drug Administration were stored at −20°C prior
to the experiment. Both groups were administered a chemotherapy
regimen that comprised of cisplatin, vincristine and bleomycin for
3 consecutive days, whereas the combined group proceeded to receive
multiple intratumoral injection of rhAd-p53 solution
1×1012 VP at an interval of three days thrice after end
of chemotherapy. For tumors <4 cm in diameter, rhAd-p53 solution
was diluted to 2 ml by addition of normal saline, and for tumors
>4 cm in diameter, rhAd-p53 was diluted to 4 ml. Efficacy was
assessed 3 weeks thereafter. Tumor sizes were compared prior and
subsequent treatment consistently by ultrasonography. Tumor size is
defined as mass product of the two greatest perpendicular
diameters. In conformity with the WHO evaluation of efficacy of
solid tumors of complete response (CR), partial remission (PR),
stable disease (SD), progressive disease (PD), our curative
efficiencies CR and PR were considered effective, whereas SD and PD
were null. Moreover, adverse events were observed in all patients
throughout the treatment.
Immunohistochemistry
The expression of vascular endothelial growth factor
(VEGF), mutant p53 protein and micro-vessel density (MVD) in tumors
was determined before and after treatment by immunohistochemistry
using VEGF and CD34 reagents (Boster Biological Technology Co.
Ltd., Wuhan, China) and the p53 protein detection reagent (Beijing
Zhongshan Golden Bridge Biotechnology Co., Ltd., Beijing, China).
Positive staining of VEGF was demonstrated by the presence of
brown-yellow granules in the cytoplasm, and positive staining of
p53 denoted tan-colored nuclei.
Semi-quantitative immunohistochemical scoring: i)
Positive cell scoring: 0, for proportion of positive cells below
1%; 1, for 1–10%; 2, for 11–50%; 3, for 51–80%; and 4, for >80%;
ii) staining intensity scoring: 1, for weak staining; 2, for
moderate staining; and 3, for strong staining. The sum of two items
represented the eventual result: 1, (−); 2–3, (+); 4–5, (++); and
6–7, (+++). CD34 stain is claybank located on vascular endothelial
cell membrane of the tumor and stroma. Microvessel count criteria:
any single endothelial cell or endothelial cell clusters that are
CD34 dyed brown are regarded as a microvessel as long as they are
separated from the adjacent microvessels, tumor cells or other
connective tissue. Similarly, it is a vessel if the structure is
not connected to its branching structure.
Routine clinical examination and
management of adverse events
Routine tests of blood, urine and stools, as well as
blood biochemistry, ECG and chest X-ray were conducted at intervals
and coupled with surveillance of gastrointestinal tract, hepatic
functionalities, temperature variations and signs of bone marrow
suppression. A few hours after the rhAd-p53 injection, patients may
experience a self-limited temporary fever, for which antipyretic
(indomethacin suppository) could be administered if needed.
Statistical analysis
Statistical analysis was performed using the
statistical software SPSS 12.0 (SPSS, Inc., Chicago, IL, USA). MVD
counts and tumor size before and after treatment are shown as mean
± standard deviation. Post-therapeutic focal shrinkage was compared
using t-test and comparison of the efficacies between the groups
was performed with the rank sum test and Spearman correlation test.
Adverse events and expression of VEGF and p53 between groups was
compared and analyzed by χ2 test. The MVD counts between
groups were analyzed with one-way analysis of variance. P<0.05
was considered to indicate a statistically significant
difference.
Results
Therapeutic efficacy
Post-treatment cancer foci were reduced to
11.42±2.78 and 15.25±4.00 cm2 in the PVB group and the
combined group, respectively (t=3.504, P=0.001). The total efficacy
rate in the PVB group which consisted of 0 CR, 15 PR, 5 SD and 0 PD
was 75%, whereas that in combined group comprised of 2 CR, 17 PR, 1
SD and 0 PD was 95%; the results were statistically significant
(Z=2.137, P=0.033).
Detection of VEGF, mutant p53 protein
and MVD in tumor tissues
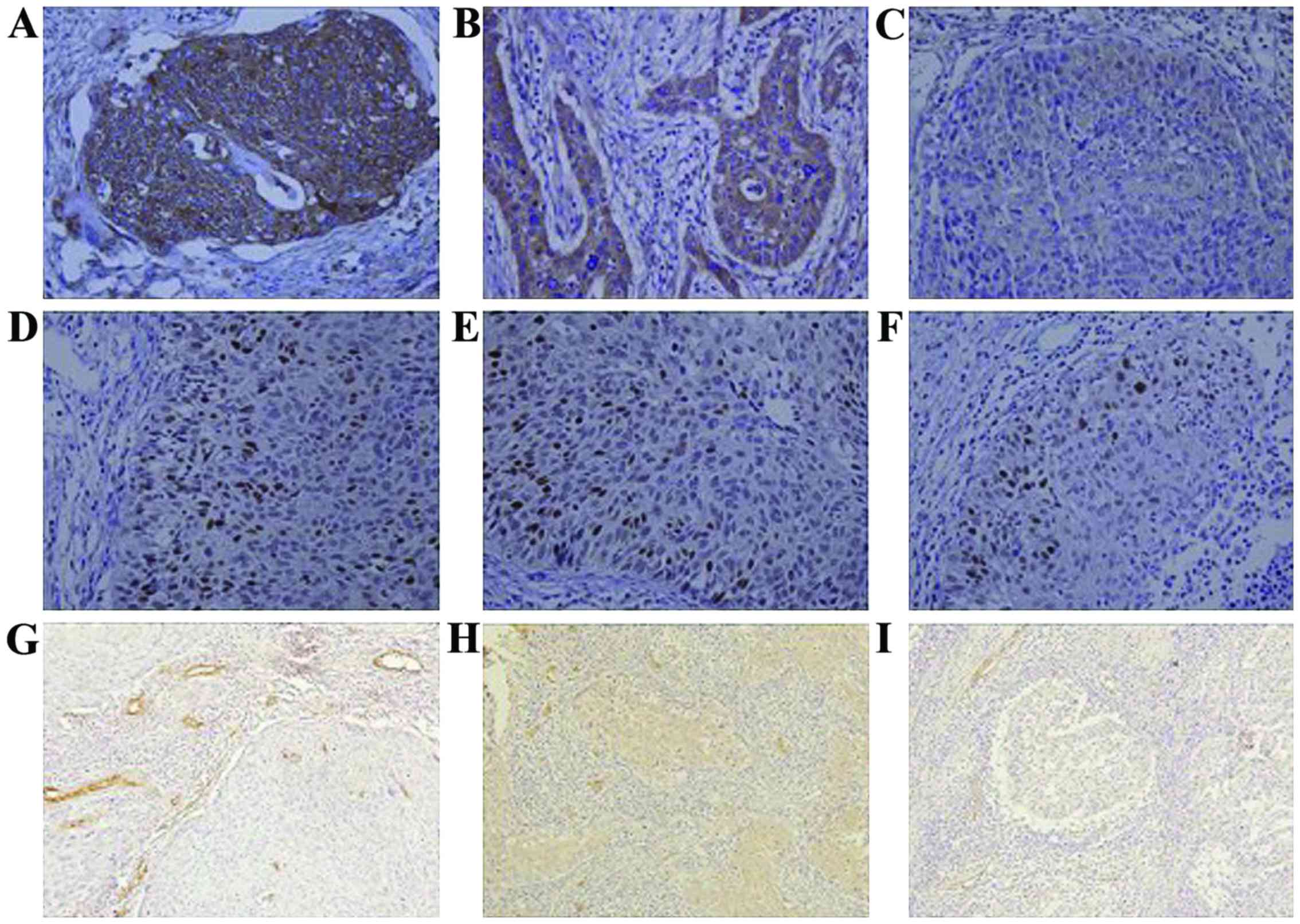
The expression of VEGF in control, PVB group and
rhAd-p53 + PVB group was 88.9 (16/18), 80.0 (16/20) and 50.0%
(10/20), respectively; results were statistically significant
(P<0.05) (Fig. 1A-C). The
expression of the mutant p53 protein in each of the three groups
was 83 (15/18), 60 (12/20) and 25% (5/20), respectively; results
were statistically significant (P<0.05) (Fig. 1D-F). MVD in the three groups counted
for 64.7±2.3, 42.5±2.2 and 32.5±1.3, respectively; results were
statistically significant (P<0.05; Fig. 1).
Adverse reactions
Both groups suffered from adverse reactions, mainly
gastrointestinal reactions, bone marrow suppression, liver damage
and fever. In the PVB group, 15 cases of gastrointestinal
discomfort such as nausea and vomiting were observed, 3 patients
had second-degree bone marrow suppression and 1 patient had
third-degree bone marrow suppression. All were given recombinant
human granulocyte colony-stimulating factors to support symptoms
improvement after treatment. No patient showed signs of infection.
Three patients had elevated aminotransferase levels, which improved
after being treated for liver disease. Ten patients presented with
fever, which improved after symptomatic treatment. In rhAd-p53 +
PVB group, 13 cases of patients had gastrointestinal discomfort,
which was tolerated. One patient had bone marrow suppression and
was given recombinant human granulocyte colony-stimulating factor
to support symptoms improved after treatment. Two patients who had
elevated aminotransferase levels improved after being given liver
treatment. Fifteen patients that were given the rhAd-p53 injection
had fever, among which 10 had a temperature of 38°C, which returned
to normal 12–24 h after drinking a lot of water; the other 5 had a
temperature of 39°C, which returned to normal 12–24 h after
indomethacin anal plug symptomatic treatment. None of the patients
showed signs of upper respiratory tract infection. There was no
statistically significant difference in fever, gastrointestinal
reactions, bone marrow suppression and liver damage rate
(P>0.05).
Discussion
rhAd-p53 is a replication-defective living
adenovirus carrying the p53 gene. The advantage of the adenovirus
delivery system is that it does not result in integration of the
vector DNA into the host cells (7).
Once cells have been infected, virus particles cannot breed, while
it can import the p53 gene, which exerted biological functions. DNA
damage within cancer cells is the important internal factor which
triggers the p53 gene and (or) p53 protein responses. Basic
mechanism of rhAd-p53 reagents are: i) Inhibit tumor growth by cell
cycle arrest and induce ‘programmed cell death’; ii) enhance
chemotherapy-induced cell cycle arrest and apoptosis; iii)
stimulate the body to produce anti-tumor immune response in a way
that the local injection site of tumors gather a large number of
immune cells; iv) inhibit tumor VEGF to suppress angiogenesis and
tumor growth through the ‘bystander effect’ (8), so the injection site of the local tumor
tissue will block blood supply and induce tumor necrosis.
The basic theory for gene therapy is completely
different from that of the traditional treatment such as
chemotherapy and radiotherapy; in fact, gene therapy and
chemotherapy could complement each other (9–11). Our
previous study found that rhAd-p53 can inhibit the proliferation of
cervical cancer (HeLa) cells in vitro; furthermore, a p53
agent combined with chemotherapeutics leads to a greater inhibition
in cell proliferation and a higher apoptosis rate (8). Some studies have also shown that gene
therapy in combination with chemotherapy leads to a beneficial
effect for head and neck squamous cell carcinoma, lung cancer and
gastric cancer (12–14).
In this study, we combine gene therapy and
chemotherapy for the treatment of LACC and found that the PVB group
and rhAd-p53 + PVB group can shrink the tumor volume. Moreover, the
shrunken volume is 1.34 times higher in rhAd-p53 + PVB group than
the PVB group alone, and the efficiency of the rhAd-p53 + PVB group
is 1.27 times that of PVB group. Therefore, rhAd-p53 has a
significant synergistic effect with cervical cancer
chemotherapy.
VEGF is the most critical growth factor to stimulate
tumor angiogenesis (15), which
specifically targets vascular endothelium to promote endothelial
cell proliferation and migration. It favors the formation of new
blood vessels and increases the permeability of existing blood
vessels (16). Consequently, it
contributes to the growth, invasion and metastasis of tumor cells
(17). The wt-p53 expression was
inversely associated with VEGF expression, suggesting that wt-p53
is involved in the suppression of the VEGF gene (18). Some researchers (19) reported that a downregulation of VEGF
was observed in tumor biopsy tissue treated with an intratumoral
injection of rhAd-p53. Significant necrosis was found in tumor
biopsy tissues of patients who received intratumoral injections of
rhAd-p53, which may be correlated with rhAd-p53-induced
downregulation of VEGF. In our study, the expression of VEGF in the
PVB group and the rhAd-p53 + PVB group was significantly decreased
compared to the control. In addition, the rhAd-p53 + PVB group had
a more significant decrease in VEGF than the PVB group. Hence, the
expression of VEGF is effectively decreased when gene therapy is
combined with chemotherapy, which inhibits the formation of
angiogenesis in cervical cancer.
The expression of the p53 gene in cervical cancer
was significantly decreased (20),
and the inactivation of p53 gene led to an increase in the invasion
of cervical cancer (21). The mutant
p53 protein gathers in the cell nucleus and the half-life period is
significantly extended, which helps us detect abnormal expression
of the p53 protein. In our study, the expression of the mutant p53
protein in PVB and rhAd-p53 + PVB group was significantly decreased
when compared to the control, and rhAd-p53 + PVB was decreased much
more than the PVB group. The above results suggest that the mutant
p53 protein enhances the transcription and expression of VEGF
(22). rhAd-p53 reagents can
effectively inhibit the expression of mutant p53 protein. Also,
they inhibit tumor angiogenesis by suppressing the transcription
and expression of VEGF.
The formation of blood vessels accelerated the
growth and metastasis of tumor cells, and MVD in the tumor tissue
reflected the malignancy and invasiveness of the tumors. The CD34
antigen, a highly glycosylated type I transmembrane protein with a
relative molecular mass of 90×103-120×103, is selectively expressed
on the surface of human vascular endothelial cells. CD34 is an
antigen that is related to new small blood vessels, which is a more
specific marker of mature vascular endothelial cells. We used CD34
antigens as markers to count microvessels, which can reflect tumor
microvessel quantity accurately and objectively (23). In our study, the MVD count in PVB and
rhAd-p53 + PVB group is significantly decreased, with the latter
demonstrating a more considerable decrease. This result is
consistent with a decrease in mutated p53 protein and VEGF, which
suggests that gene therapy, in combination with chemotherapy, can
be more effective in inhibiting tumor angiogenesis and limiting the
growth of tumors.
In this study, the rhAd-p53 reagent caused a
self-limiting fever in patients. Febrile reactions mainly occured
several hours after the injection and few on the following day.
Symptoms eased without obvious discomfort after drinking more water
or the use of antipyretic drugs, which does not affect subsequent
treatment. There was no statistical significance in fever,
gastrointestinal reactions, bone marrow suppression and liver
damage between the PVB group and rhAd-p53 + PVB group, which tells
us that gene dosage does not have serious side effects.
Through this study, we realized that rhAd-p53 can
effectively inhibit the expression of the mutant p53 protein, and
inhibit the transcription and expression of VEGF once it gets into
tumor cells. This reduces tumor angiogenesis and is more effective
in shrinking tumor volume after interaction with chemotherapeutic
drugs. Furthermore, it provides conditions for surgical treatment
by reducing tumor staging for patients with advanced cervical
cancer. There were no other adverse reactions except, for a brief
period of time, self-limiting heating during the administration of
rhAd-p53 reagent, which suggests that, in addition to its
anti-cancer effect, rhAd-p53 reagent can increase sensitivity to
chemotherapy for cervical cancer. Therefore, the rhAd-p53 reagent,
when combined with chemotherapy drugs, is effective for cervical
cancer patients.
Acknowledgements
This study was supported by Clinical Cancer Research
Fund of Health Bureau of Jiangsu Province (no. P200943). We are
grateful to Ms. Yao Qier, our professional nurse; her dedication is
outstanding and invaluable.
Glossary
Abbreviations
Abbreviations:
|
LACC
|
locally advanced cervical cancer
|
|
FIGO
|
International Federation of Gynecology
and Obstetrics
|
|
rhAd-p53
|
recombinant human adenovirus-p53
|
|
VEGF
|
vascular endothelial growth factor
|
|
MVD
|
micro-vessel density
|
|
CR
|
complete response
|
|
PR
|
partial remission
|
|
SD
|
stable disease
|
|
PD
|
progressive disease
|
References
|
1
|
Pecorelli S: Revised FIGO staging for
carcinoma of the vulva, cervix, and endometrium. Int J Gynaecol
Obstet. 105:103–104. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rydzewska L, Tierney J, Vale CL and
Symonds PR: Neoadjuvant chemotherapy plus surgery versus surgery
for cervical cancer. Cochrane Database Syst Rev.
12:CD0074062012.PubMed/NCBI
|
|
3
|
Gadducci A, Sartori E, Maggino T, Zola P,
Cosio S, Zizioli V, Lapresa M, Piovano E and Landoni F:
Pathological response on surgical samples is an independent
prognostic variable for patients with Stage Ib2-IIb cervical cancer
treated with neoadjuvant chemotherapy and radical hysterectomy: An
Italian multicenter retrospective study (CTF Study). Gynecol Oncol.
131:640–644. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nemunaitis J, Clayman G, Agarwala SS,
Hrushesky W, Wells JR, Moore C, Hamm J, Yoo G, Baselga J, Murphy
BA, et al: Biomarkers predict p53 gene therapy efficacy in
recurrent squamous cell carcinoma of the head and neck. Clin Cancer
Res. 15:7719–7725. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lehmann BD and Pietenpol JA: Targeting
mutant p53 in human tumors. J Clin Oncol. 30:3648–3650. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zheng XL, Wang TT, Fu M, Zhou J and Liu
FM: Inhibitory effect of recombinant human adenovirus-P53 combined
with paclitaxel on human cervical cancer HeLa cells and its
mechanism. Chin J Cancer Biother. 20:192–196. 2013.(In
Chinese).
|
|
7
|
Lane DP, Cheok CF and Lain S: p53-based
cancer therapy. Cold Spring Harb Perspect Biol. 2:a0012222010.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Burdak-Rothkamm S, Rothkamm K, McClelland
K, Al Rashid ST and Prise KM: BRCA1, FANCD2 and Chk1 are potential
molecular targets for the modulation of a radiation-induced DNA
damage response in bystander cells. Cancer Lett. 356(Pt B 2):
454–461. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Jiang G, Xin Y, Zheng JN and Liu YQ:
Combining conditionally replicating adenovirus-mediated gene
therapy with chemotherapy: A novel antitumor approach. Int J
Cancer. 129:263–274. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Prados J, Alvarez PJ, Melguizo C,
Rodriguez-Serrano F, Carrillo E, Boulaiz H, Vélez C, Marchal JA,
Caba O, Ortiz R, et al: How is gene transfection able to improve
current chemotherapy? The role of combined therapy in cancer
treatment. Curr Med Chem. 19:1870–1888. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wirth T, Parker N and Ylä-Herttuala S:
History of gene therapy. Gene. 525:162–169. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tassone P, Old M, Teknos TN and Pan Q:
p53-based therapeutics for head and neck squamous cell carcinoma.
Oral Oncol. 49:733–737. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li D, Zhang Y, Xie Y, Xiang J, Zhu Y and
Yang J: Enhanced tumor suppression by adenoviral PTEN gene therapy
combined with cisplatin chemotherapy in small-cell lung cancer.
Cancer Gene Ther. 20:251–259. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chen GX, Zheng LH, Liu SY and He XH:
rAd-p53 enhances the sensitivity of human gastric cancer cells to
chemotherapy. World J Gastroenterol. 17:4289–4297. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cao Y, Guangqi E, Wang E, Pal K, Dutta SK,
Bar-Sagi D and Mukhopadhyay D: VEGF exerts an
angiogenesis-independent function in cancer cells to promote their
malignant progression. Cancer Res. 72:3912–3918. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tomao F, Papa A, Rossi L, Zaccarelli E,
Caruso D, Zoratto F, Panici P Benedetti and Tomao S: Angiogenesis
and antiangiogenic agents in cervical cancer. Onco Targets Ther.
7:2237–2248. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Perrot-Applanat M and Di Benedetto M:
Autocrine functions of VEGF in breast tumor cells: Adhesion,
survival, migration and invasion. Cell Adhes Migr. 6:547–553. 2012.
View Article : Google Scholar
|
|
18
|
Holmgren L, Jackson G and Arbiser J: p53
induces angiogenesis-restricted dormancy in a mouse fibrosarcoma.
Oncogene. 17:819–824. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li X, Xiao S, Li Y and Zhang S: Clinical
antiangiogenic effect of recombinant adenovirus-p53 combined with
hyperthermia for advanced cancer. Chin J Cancer Res. 25:749–755.
2013.PubMed/NCBI
|
|
20
|
Crinelli R, Bianchi M, Menotta M, Carloni
E, Giacomini E, Pennati M and Magnani M: Ubiquitin over-expression
promotes E6AP autodegradation and reactivation of the p53/MDM2
pathway in HeLa cells. Mol Cell Biochem. 318:129–145. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yang X and Lu L: Expression of HPV-16 E6
protein and p53 inactivation increases the uterine cervical cancer
invasion. Drug Res (Stuttg). 65:70–73. 2015.PubMed/NCBI
|
|
22
|
Yu YF, Zhang Y, Shen N, Zhang RY and Lu
XQ: Effect of VEGF, P53 and telomerase on angiogenesis of gastric
carcinoma tissue. Asian Pac J Trop Med. 7:293–296. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ding S, Li C, Lin S, Yang Y, Liu D, Han Y,
Zhang Y, Li L, Zhou L and Kumar S: Comparative evaluation of
microvessel density determined by CD34 or CD105 in benign and
malignant gastric lesions. Hum Pathol. 37:861–866. 2006. View Article : Google Scholar : PubMed/NCBI
|