Introduction
Hepatocellular carcinoma (HCC) is an aggressive
neoplasm with an increasing incidence, representing the fifth most
common cancer globally (1). It is
also the third leading cause of cancer-associated mortality
(2). Surgical resection is the
cornerstone of therapeutic strategy in the early stages of HCC.
However, numerous patients with HCC with advanced or distant
metastatic disease are not candidates for these therapies (3). HCC is well characterized as
demonstrating an inherently chemotherapy-refractory nature, and
systemic cytotoxic chemotherapy agents exhibit minimal
effectiveness on improving patient survival (4,5).
Therefore, exploring novel anticancer agents that are more
effective for HCC is important. A number of traditional Chinese
medicines, with wide use in clinical practice in China, have been
the source of novel anticancer drug developments (6–10).
Delisheng, a Chinese medicinal compound that consists of ginseng,
milk vetch root, bufonis secretions and cantharidium (11), has previously attracted attention
(11), but the potential molecular
mechanisms underlying its anticancer effect are unknown.
During carcinogenesis, angiogenesis exhibits a key
role in regulating physiological functions (12). This process is characterized by
decreasing levels of antiangiogenic proteins and increasing levels
of factors that stimulate vasculogenesis (13). Despite previous studies demonstrating
that vascular endothelial growth factor(VEGF), osteopontin (OPN)
and endostatin (ES) are involved in tumor angiogenesis (14,15), their
association with HCC is poorly understood.
In the present study, human hepatocarcinoma Hep3B
cells and normal liver HL-7702 cells were utilized to further study
the underlying mechanisms of the anticancer effects of Delisheng.
It was demonstrated that Delisheng exhibits anti-proliferative and
pro-apoptotic effects on Hep3B cells, and that the angiogenic
proteins VEGF, OPN and ES are involved in this process.
Materials and methods
Cell culture
The human hepatocarcinoma Hep3B and normal liver
HL-7702 cell lines were purchased from Chinese Academy of Sciences
Institute of Life Sciences Cell Resource Center (Shanghai, China).
They were grown in RPMI-1640 medium and Dulbecco's modified Eagle's
medium (DMEM; GE Healthcare Life Sciences; Hyclone, Logan, UT,
USA), respectively. The cell cultures contain 10% fetal bovine
serum (GE Healthcare Life Sciences; Hyclone) and 100 U/ml
penicillin and streptomycin (Gibco; Thermo Fisher Scientific,
Waltham, MA, USA) at 37°C with 5% CO2. Then, cells were
harvested in the logarithmic growth phase for subsequent
experiments.
MTT assays
Hep3B cells (2×105 cells) and the normal
liver HL-7702 cell (2×105 cells) suspended in 150 µl
DMEM and RPMI-1640 medium with 10% FBS, respectively. They were
seeded into each well of 96-well microtiter plates overnight, and
then treated with 25, 50 and 100 µl/ml Delisheng. Cells were
cultured at 37°C and 5% CO2 for 24, 48 and 72 h at each
concentration of Delisheng. Subsequently, 20 µl MTT (5 mg/ml) was
added to each well for 4 h at 37°C and formazan crystals were
dissolved in 200 µl dimethyl sulfoxide for each well. The optical
density (OD) value was recorded at a wavelength of 492 nm. The rate
of growth inhibition (%) = (1 - OD of treated group)/(OD of
control) × 100. This experiment was repeated in triplicate.
Cell-cycle analysis
The Hep3Bcells treated with Delisheng
(2×105 cells/group) were trypsinized, washed twice with
cold PBS and harvested by centrifugation at 4,000 × g and 37°C for
5 min, and then incubated in 100 µg/ml propidium iodide (PI) with
RNaseA (5 µg/ml) following fixation with 70% ice-cold ethanol at
room temperature (RT) for 30 min. Cellular DNA content was
subsequently analyzed by flow cytometry analysis on a BD
FACSCalibur system (BD Biosciences, Franklin Lakes, NJ, USA), as
previously described (16).
Cell apoptosis assay
Hep3B and HL-7702 cells treated with 25, 50 and 100
µl/ml Delisheng were collected and washed twice with cold PBS, and
centrifuged at 5,000 × g for 5 min, followed by staining with 5 µl
Annexin V-fluorescein isothiocyanate and 5 µl PI for 15 min at 37°C
in darkness. The apoptotic cells were subsequently analyzed by
fluorescence-activated cell sorting (FACSCalibur; BD Biosciences)
using Cell Quest Research 3.3 Software (BD Biosciences).
Immunocytochemistry staining
This procedure has been described previously
(15). Briefly, following treatment
with 25 µl/ml Delisheng, the Hep3B cells were washed with PBS and
then stained with the primary antibodies (Beijing Zhongshan Golden
Bridge Biotechnology Co., Ltd. Beijing, China) against VEGF
(cat.no., ZA-0509), OPN (cat. no., ZM-0174) and ES (cat.no.,
TA507124) (dilutions, 1:100) overnight at 4°C. The next day, the
cells were washed with PBS 3 times, followed by incubation with a
secondary antibody (Biotinylated goat anti-rabbit IgG; cat. no.,
bs-0294P; Xiamen Bosen Biotechnology Co. Ltd., Xiamen, China)
diluted to 1:500 for 20 min at 37°C, and then immunostained using
an avidin-biotin complex (ABC) protocol (ABC Staining Kits; Xiamen
Bosen Biotechnology Co., Ltd. Xiamen, China) for 30 min, according
to the protocol of the manufacturer. The color reaction was
visualized through a light microscope (Olympus BH-12, Tokyo, Japan)
at magnification, ×400 using 3,3′-diaminobenzidinefor 5 min. The
nuclei were counterstained with hematoxylin (10 mg/ml) solution
(Beyotime Institute of Biotechnology, Shanghai, China) at 37°C for
10 sec.
Evaluation of immunocytochemistry
staining
The immunoreactivity was evaluated according to the
cell staining degree and percentage rate of the positive cell.
Staining intensity was graded as follows: 0, no staining; 1, weak;
2, moderate; and 3, strong. The percentage of positively stained
cells was scored as follows: 0, ≤5%; 1, 5–25%; 2, 26–50%; and 3,
>50%. The score of staining intensity and percentage score of
positive cells were multiplied. In these groups, tumors with 0–2
scores were noted as negative, while 3–9 scores were designated as
positive. The immunocytochemical results were quantitatively
analyzed using LeicaQ550cw imaging analysis system (Leica
Microsystems GmbH, Wetzlar, Germany).
Western blotting
Following treatment with 25 µl/ml Delisheng, the
Hep3B cells were harvested in ice-cold PBS and lysed with
radioimmunoprecipitation cell lysis buffer containing protease and
phosphatase inhibitors (Xi'an Wolsen Biotechnology Co. Ltd., Xi'an,
China). Protein concentrations in the cell extracts were determined
using the BCA Protein Assay reagents (Pierce; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol. Each
protein sample (30 µg) was subject to 15% SDS-PAGE and transferred
onto polyvinylidene difluoride membranes (Merck KGaA, Darmstadt,
Germany), blocked with 8% non-fat milk for 2 h at RT. The membranes
were labeled with primary antibodies overnight at 4°C.
Subsequently, blots were probed with horseradish
peroxidase-conjugated secondary anti-rabbit IgG antibodies
(1:5,000; cat. no., sc-2004; Santa Cruz Biotechnology, Inc.,
Dallas, TX, USA) at RT for 1 h. The primary antibodies VEGF (cat.
no., ab46154), OPN (cat. no., ab8448), ES (cat. no., ab64569) at
dilutions of 1:1,000 and β-actin (cat. no., ab8227; 1:5,000) were
purchased from Abcam (Cambridge, UK). For the quantification of
protein levels, films were scanned and analyzed using Labworks 4.0
software (World BioHazTec Corporation, NM, USA).
Statistical analysis
All results were presented as the mean ± standard
deviation. Statistical analysis was performed by one-way analysis
of variance followed by Tukey's post hoc test. Student's t-test and
Spearman's rank test using SPSS v.19.0 (IBM Corp., Armonk, NY,
USA). P<0.05 was considered to indicate a statistically
significant difference.
Results
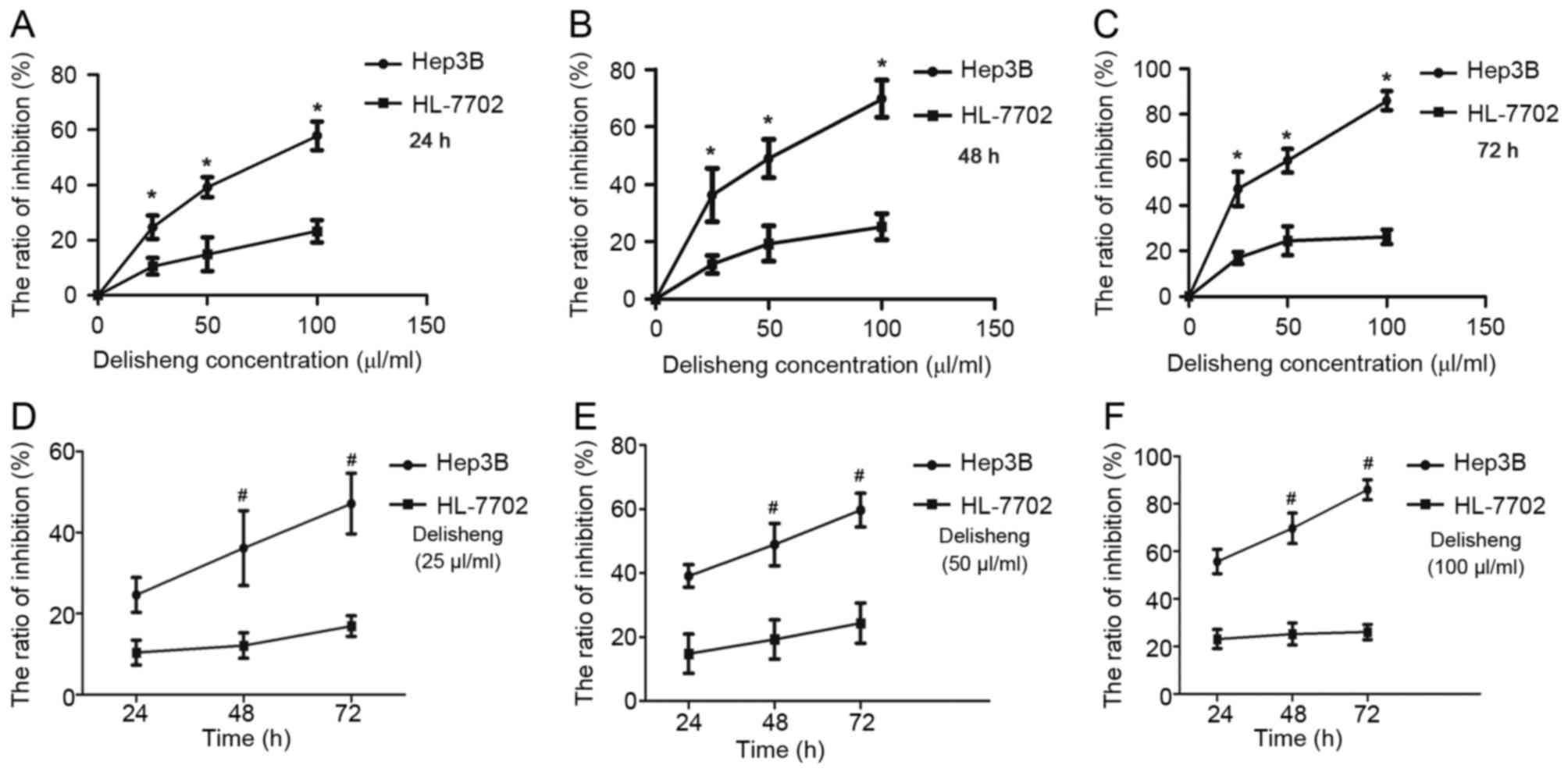
Antiproliferative activity of
Delisheng on Hep3B and HL-7702 cells
The cytotoxic effects of Delisheng were measured by
MTT assay. Based on Table I and
Fig. 1, exposure of these cells to
Delisheng at the indicated concentrations for 24–72 h periods
exhibited growth inhibition effects of Hep3B in a time- and
dose-dependent manner. Compared to Hep3B cells, HL-7702 cells were
less sensitive to the antiproliferative effects of Delisheng
(Table I).
 | Table I.Delisheng inhibits the proliferation
of Hep3B and HL-7702 cell lines. |
Table I.
Delisheng inhibits the proliferation
of Hep3B and HL-7702 cell lines.
|
|
| Inhibition rate,
% |
|
|---|
|
|
|
|
|
|---|
| Cells | Concentration,
µl/ml | 24 h | 48 h | 72 h | P-value |
|---|
| Hep3B | 25 |
24.62±4.31a |
36.17±9.24a |
47.14±7.48a | 0.037 |
|
| 50 |
39.13±3.52b |
48.94±6.65b |
59.69±5.26b | 0.021 |
|
| 100 |
57.73±5.13c |
69.72±6.42c |
85.92±4.19c | 0.003 |
| HL-7702 | 25 |
10.41±3.06d |
12.15±3.15d |
16.94±2.53d | 0.216 |
|
| 50 |
14.85±6.12e |
19.32±6.14e |
24.39±6.31e | 0.125 |
|
| 100 |
23.14±4.02f |
25.24±4.63f |
26.09±3.18f | 0.766 |
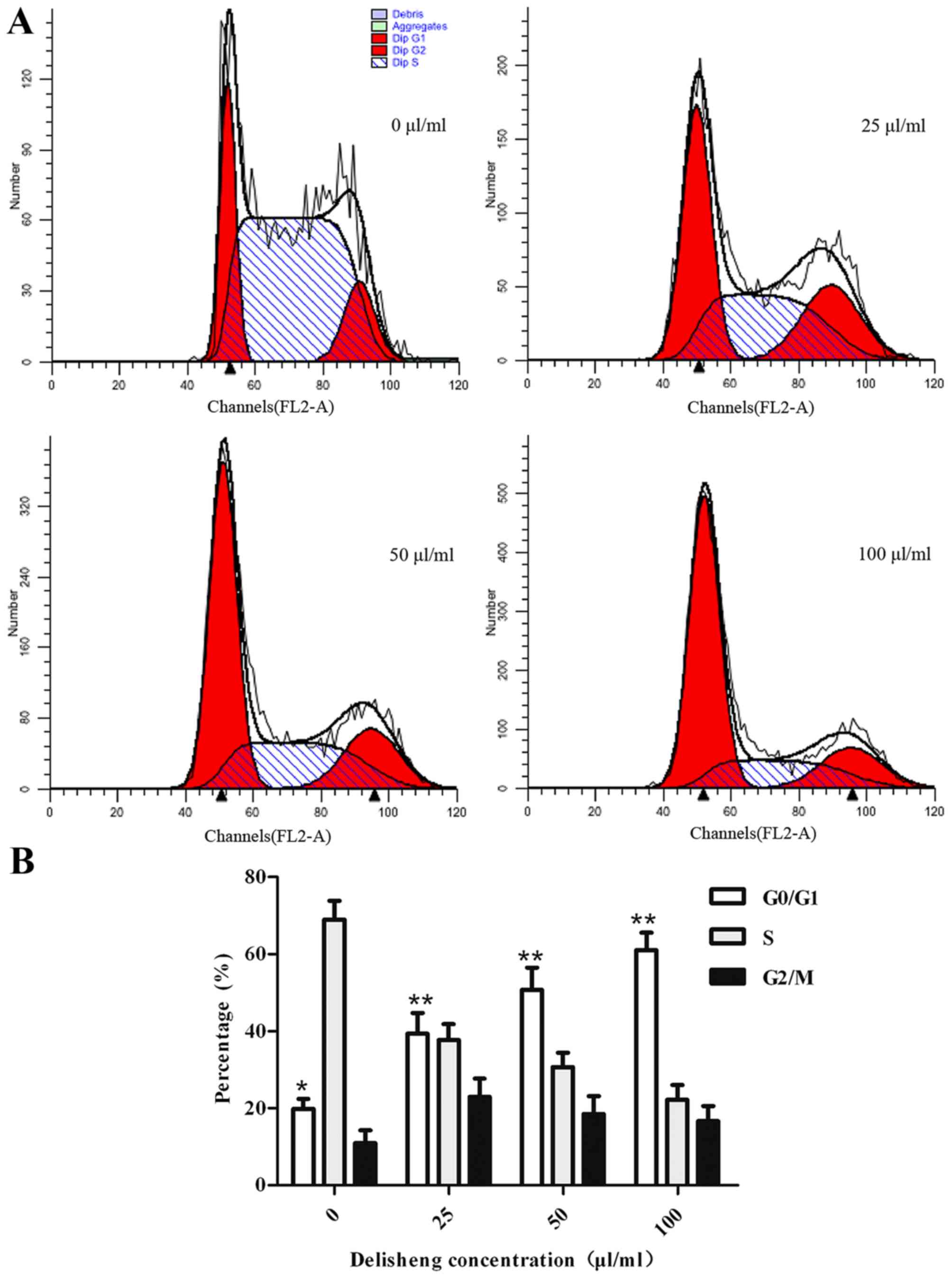
Effects of Delisheng on the cell
cycles of Hep3B cells
To determine the mechanism underlying the
antiproliferative activity of Delisheng on Hep3B cells, cell cycles
were tested by flow cytometric analysis. As demonstrated in
Table II and Fig. 2, in Hep3B cells treated with 25, 50
and 100 µl/ml Delisheng, G0/G1 cell cycle
arrest was observed. In addition, the results indicated the cells
exhibited G0/G1 phase arrest in a
concentration-dependent manner.
 | Table II.Change in cell cycle distribution of
Hep3B cells treated with Delisheng. |
Table II.
Change in cell cycle distribution of
Hep3B cells treated with Delisheng.
|
| Cell cycles |
|---|
|
|
|
|---|
| Concentration,
µl/ml |
G0/G1 cycle | S cycle |
G2/Mcycle |
|---|
| 0 | 19.90±2.57 | 69.05±4.83 | 11.06±3.22 |
| 25 |
39.36±5.41a | 37.70±4.16 | 22.94±4.79 |
| 50 |
50.75±5.82a | 30.72±3.73 | 18.53±4.63 |
| 100 |
61.06±4.58a | 22.26±3.84 | 16.68±3.98 |
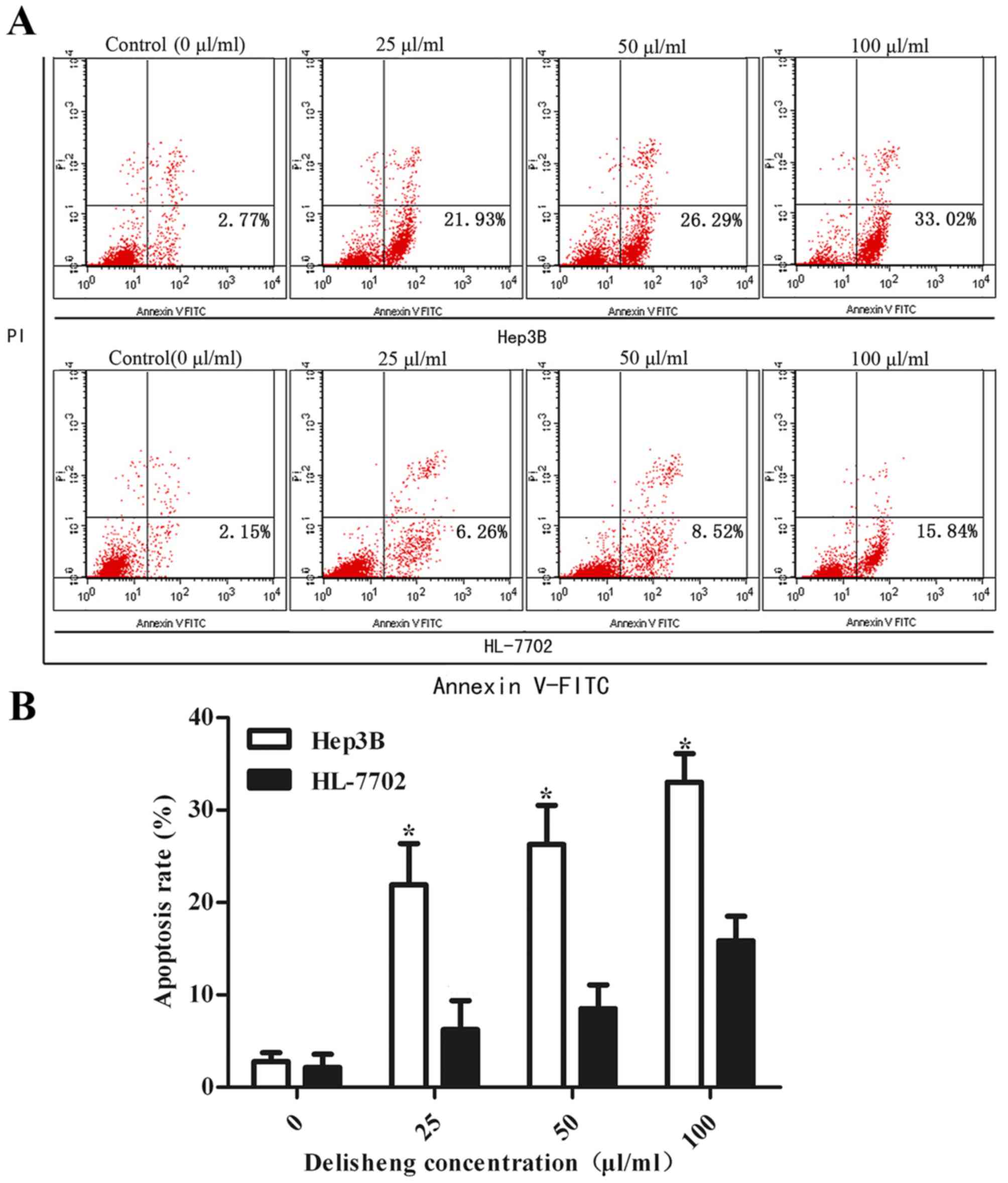
Delisheng induces apoptosis
To additionally determine the potential mechanism of
the anticancer effects of Delisheng, cell apoptosis was measured by
flow cytometry. As indicated by Fig.
3 and Table III, following
treatment with 25, 50 and 100 µl/ml Delisheng at 24 h, the
apoptotic rates of Hep3B cells were 21.93±0.98, 26.29±4.45 and
33.02±3.09%, respectively (P<0.05 vs. untreated cells). The
HL-7702 cells, which exhibited apoptotic rates of 6.26±3.12,
8.52±2.55 and 15.84±2.67%, at 25, 50 and 100 µl/ml Delisheng at 24
h, respectively, and Delisheng-untreated Hep3B cells did not
demonstrate evidence of apoptosis.
 | Table III.Delisheng-induced apoptosis in Hep3B
and HL-7702 cell lines. |
Table III.
Delisheng-induced apoptosis in Hep3B
and HL-7702 cell lines.
|
| Early apoptosis
rate, %a |
|---|
|
|
|
|---|
| Cells | Delisheng, 0
µl/ml | Delisheng, 25
µl/ml | Delisheng, 50
µl/ml | Delisheng, 100
µl/ml |
|---|
| Hep3B |
2.77±0.98b |
21.93±4.45b |
26.29±4.24b |
33.02±3.09b |
| HL-7702 |
2.15±1.44c |
6.26±3.12c |
8.52±2.55c |
15.84±2.67c |
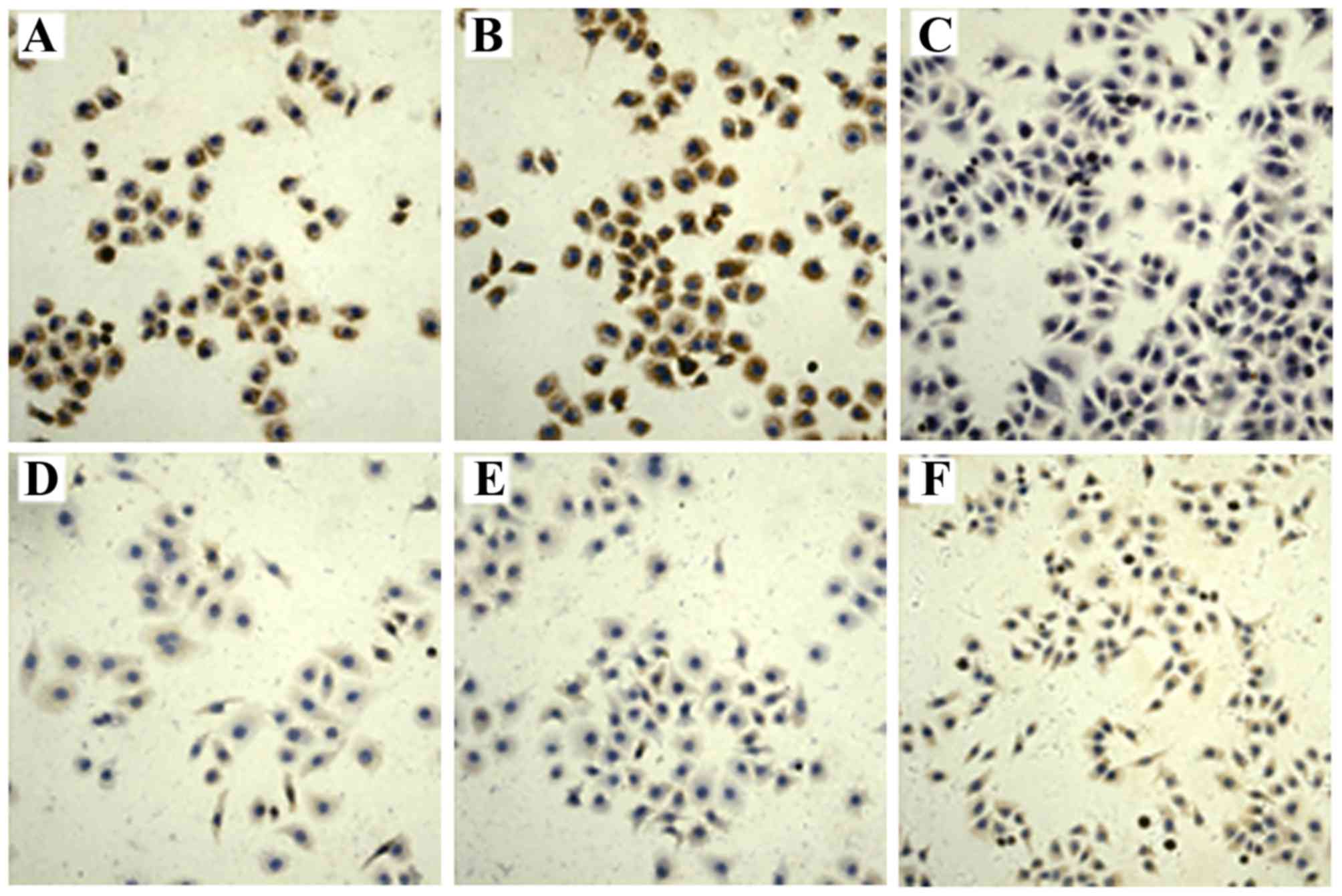
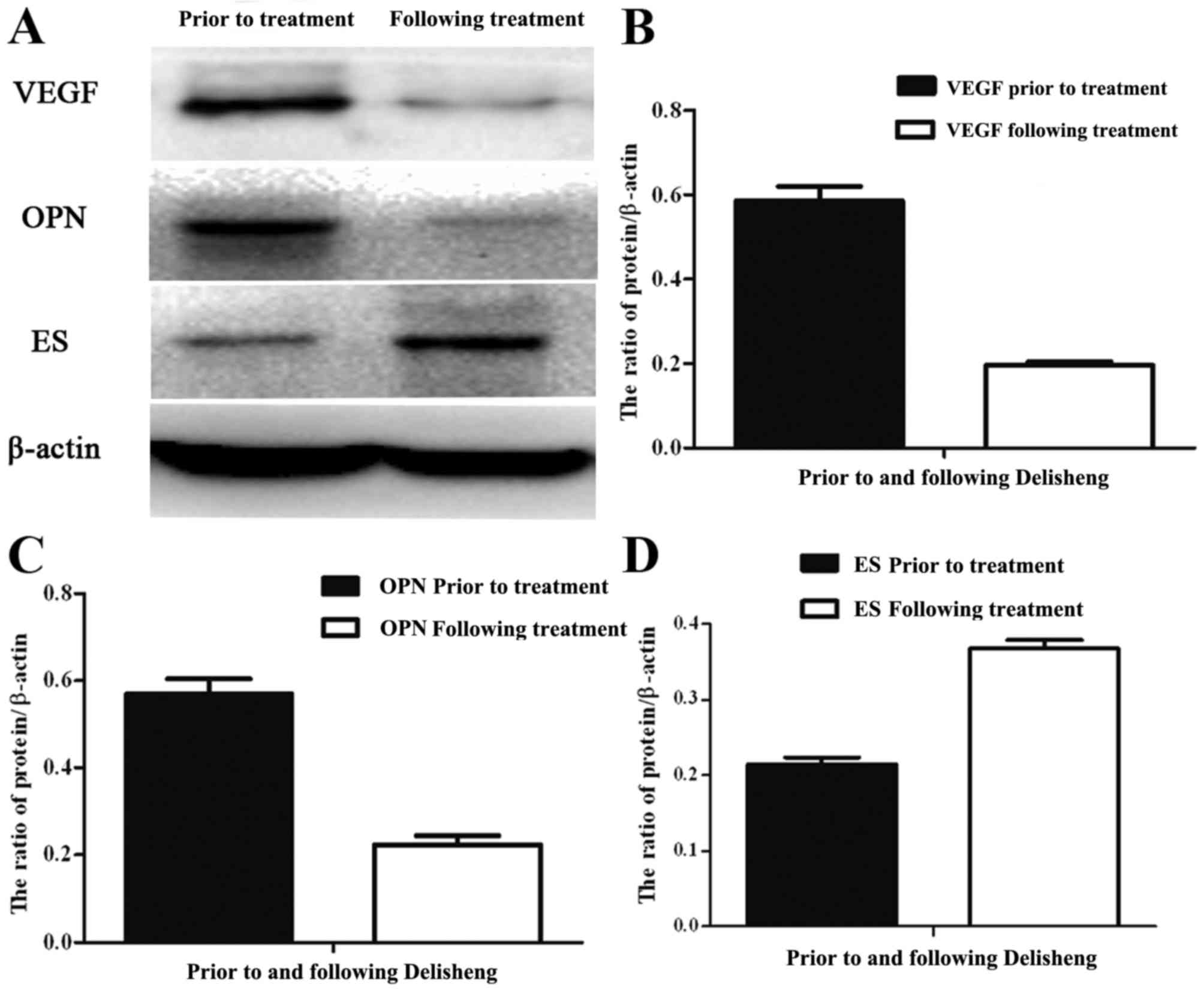
VEGF, OPN and ES are involved in Hep3B
cells treated with Delisheng
Immunocytochemistry analysis demonstrated that the
levels of VEGF, OPN and ES expression were observed in the
cytoplasm of Hep3B cells. Fig. 4
indicates that Delisheng treatment increased VEGF and OPN and
decreased ES protein expression levels (P<0.05). As observed in
Fig. 5 and Table IV, western blotting analysis
additionally confirmed the results. Furthermore, the Spearman's
rank correlation analysis indicated that OPN was positively
correlated with VEGF expression (r=0.58, P=0.011). However, ES was
negatively correlated with VEGF (r=0.57, P=0.009) and OPN
expression (r=0.69, P=0.025).
 | Table IV.Expression of VEGF, OPN and ES in
Hep3B cell lines treated with Delisheng. |
Table IV.
Expression of VEGF, OPN and ES in
Hep3B cell lines treated with Delisheng.
|
| Mean grey
value |
|---|
|
|
|
|---|
| Protein | Delisheng, 0
µl/ml | Delisheng, 25
µl/ml | Delisheng, 50
µl/ml | Delisheng, 100
µl/ml |
|---|
| VEGF |
180.26±5.43a |
192.36±5.78a |
201.98±6.54a |
218.51±8.27a |
| OPN |
186.34±6.11b |
197.96±7.45b |
206.64±4.83b |
229.37±7.62b |
| ES |
145.56±7.52c |
132.31±4.16c |
120.15±4.25c |
107.69±4.31c |
Discussion
Delisheng has demonstrated antitumor activities for
specific types of cancer (17–19).
Ginsenoside Rg3 (Rg3), one of the bioactive extracts from
Delisheng, exerts an antiproliferation effect on prostate cancer
cells (20). For ~80% of patients
with prostate cancer, Delisheng treatment alone may alleviate
symptom of weakness, improve quality of life and decrease prostate
specific antigen (PSA) levels (21).
Our previous studies suggested that Delisheng exhibited cytotoxic
activities and triggered time- and concentration-dependent
apoptosis in HepG2 cells by activating the mitochondria-mediated
and death receptor-mediated apoptotic pathways (11,17).
Cell cycle progression serves a pivotal role in
maintaining homeostasis in healthy tissues. Numerous anticancer
agents arrest the cell cycle and then induce cell death (22–25). In
the present study, the results demonstrated that the
antiproliferative and proapoptotic activities of Delisheng may be
attributed to G0/G1 cell cycle arrest.
Delisheng induced antiproliferation and apoptosis in Hep3B cells,
but exhibited no significant effect on HL-7702 cells. These data
suggested that Delisheng may exhibit less toxicity in normal liver
cells compared with in HCC cells.
VEGF and OPN may induce tumor angiogenesis, which is
crucial for the malignant process of cancer (26–29). VEGF
exerts multiple functions including endothelial cell mitogenesis
and migration, remodeling of the extracellular matrix, increased
vascular permeability and maintenance of the survival of novel
blood vessel formation (30). A
number of tumors highly express VEGF, which is correlated with
tumor invasiveness, vascular density, metastasis/recurrence and
unfavorable prognosis (31–33). OPN is a phosphorylated acidic
glycoprotein which functions in cell attachment, and is also a
cytokine that signals through αvβ3-integrin and cluster of
differentiation 44. Binding of OPN to these cell surface receptors
stimulates cell adhesion, proliferation and migration (34). Previous studies indicated that the
expression of VEGF was associated with OPN expression, and that
they may serve as prognostic factors for non-small-cell lung cancer
(27). The data of the present study
demonstrated that Delisheng downregulated VEGF and OPN expression
in Hep3B cell ina concentration-dependent manner. ES is a
proteolytic cleavage product of type XVIII collagen, and interacts
with multiple cell-surface molecules (35). It inhibits endothelial cells
proliferation, migration/invasion and increases tumor cells
apoptosis (36). Furthermore, ES
suppressed the expression of VEGF, matrix metalloproteinases,
fibroblast growth factor and cell adhesion molecule (37,38).
Certain previous studies revealed that tumor growth was inhibited
in transgenic mice overproducing ES (39,40). Based
on the results of the present study, the upregulation of ES and
downregulation of VEGF and OPN may be responsible for the
anticancer effect of Delisheng on Hep3B cells.
Taken together, the results of the present study
provide novel evidence that Delisheng induces antiproliferation and
apoptosis in Hep3B cells, at least in part through downregulating
VEGF and OPN and upregulating ES, which may assist in the
validation of the clinical application of Delisheng. Additional
studies investigating the underlying anticancer mechanisms and for
its potential as a novel anticancer therapeutic in HCC are
required.
Acknowledgements
The present study was supported by grants from the
National Natural Science Foundation of China (grant no.
81372581).
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2015. CA Cancer J Clin. 65:5–29. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Buell J: Hepatoma Research: The beginning
of a new forum. Hepatoma Res. 1:1–5. 2015. View Article : Google Scholar
|
|
3
|
El-Serag HB, Marrero JA, Rudolph L and
Reddy KR: Diagnosis and treatment of hepatocellular carcinoma.
Gastroenterology. 134:1752–1763. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Choi KJ, Baik IH, Ye SK and Lee YH:
Molecular targeted therapy for hepatocellular carcinoma: Present
status and future directions. Biol Pharm Bull. 38:986–991. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wu T, Jiao M, Jing L, Wang MC, Sun HF, Li
Q, Bai YY, Wei YC, Nan KJ and Guo H: Prognostic value of Notch-1
expression in hepatocellular carcinoma: A meta-analysis. Onco
Targets Ther. 8:3105–3114. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang X, Wang N, Cheung F, Lao L, Li C and
Feng Y: Chinese medicines for prevention and treatment of human
hepatocellular carcinoma: Current progress on pharmacological
actions and mechanisms. J Integr Med. 13:142–164. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhai XF, Qiao CX, Liu Q, Chen Z and Ling
CQ: Quality assessment of clinical research on liver cancer treated
by intra-arterial infusion of Chinese medicine. Chin J Integr Med.
20:870–875. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wu P, Dugoua JJ, Eyawo O and Mills EJ:
Traditional Chinese Medicines in the treatment of hepatocellular
cancers: A systematic review and meta-analysis. J Exp Clin Cancer
Res. 28:1122009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Qi F, Li A, Inagaki Y, Gao J, Li J, Kokudo
N, Li XK and Tang W: Chinese herbal medicines as adjuvant treatment
during chemo- or radio-therapy for cancer. Biosci Trends.
4:297–307. 2010.PubMed/NCBI
|
|
10
|
Qi F, Li A, Zhao L, Xu H, Inagaki Y, Wang
D, Cui X, Gao B, Kokudo N, Nakata M and Tang W: Cinobufacini, an
aqueous extract from Bufo bufo gargarizans Cantor, induces
apoptosis through a mitochondria-mediated pathway in human
hepatocellular carcinoma cells. J Ethnopharmacol. 128:654–661.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang SH, Wang YC, Nie YL, Hai YN, Sun HF,
Yuan ZL and Nan KJ: Antiproliferative activity of the chinese
medicinal compound, delisheng, compared with Rg3 and gemcitabine in
HepG2 Cells. Indian J Pharm Sci. 75:578–584. 2013.PubMed/NCBI
|
|
12
|
Sredni B: Immunomodulating tellurium
compounds as anti-cancer agents. Semin Cancer Biol. 22:60–69. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hanahan D and Folkman J: Patterns and
emerging mechanisms of the angiogenic switch during tumorigenesis.
Cell. 86:353–364. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Raja R, Kale S, Thorat D, Soundararajan G,
Lohite K, Mane A, Karnik S and Kundu GC: Hypoxia-driven osteopontin
contributes to breast tumor growth through modulation of
HIF1α-mediated VEGF-dependent angiogenesis. Oncogene. 33:2053–2064.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bao Y, Feng WM, Tang CW, Zheng YY, Gong HB
and Hou EG: Endostatin inhibits angiogenesis in hepatocellular
carcinoma after transarterial chemoembolization.
Hepato-gastroenterology. 59:1566–1568. 2012.PubMed/NCBI
|
|
16
|
Gai P, Sun H, Wang G, Xu Q, Qi X, Zhang Z
and Jiang L: miR-22 promotes apoptosis of osteosarcoma cells via
inducing cell cycle arrest. Oncol Lett. 13:2354–2358.
2017.PubMed/NCBI
|
|
17
|
Lu CX, Nan KJ, Nie YL, Hai YN and Jiao M:
Delisheng, a Chinese medicinal compound, exerts anti-proliferative
and pro-apoptotic effects on HepG2 cells through extrinsic and
intrinsic pathways. Mol Biol Rep. 37:3407–3412. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dong XL, Gong Y, Chen ZZ and Wang YJ:
Delisheng injection (), a Chinese medicinal compound, enhanced the
effect of cis-platinum on lung carcinoma cell line PGCL3. Chin J
Integr Med. 20:286–291. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhao D, Liu XJ, Xie Q, Huang ZP, Zou BX
and Ge YB: Intensity-modulated radiation combined with Delisheng
injection for naspharyngeal carcinoma. Nan Fang Yi Ke Da Xue Xue
Bao. 26:874–875. 2006.(In Chinese). PubMed/NCBI
|
|
20
|
Pan XY, Guo H, Han J, Hao F, An Y, Xu Y,
Xiaokaiti Y, Pan Y and Li XJ: Ginsenoside Rg3 attenuates cell
migration via inhibition of aquaporin 1 expression in PC-3M
prostate cancer cells. Eur J Pharmacol. 683:27–34. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang SW, Zhou SY, Shao JC and Qu XW:
Primary research on Chinese medicine treatment of
androgen-independent prostate cancer. Chin J Integr Med.
15:168–169. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lee HH, Lee S, Lee K, Shin YS, Kang H and
Cho H: Anti-cancer effect of Cordyceps militaris in human
colorectal carcinoma RKO cells via cell cycle arrest and
mitochondrial apoptosis. Daru. 23:352015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tsui KC, Chiang TH, Wang JS, Lin LJ, Chao
WC, Chen BH and Lu JF: Flavonoids from Gynostemma pentaphyllum
exhibit differential induction of cell cycle arrest in H460 and
A549 cancer cells. Molecules. 19:17663–17681. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lee YS, Choi KM, Kim W, Jeon YS, Lee YM,
Hong JT, Yun YP and Yoo HS: Hinokitiol inhibits cell growth through
induction of S-phase arrest and apoptosis in human colon cancer
cells and suppresses tumor growth in a mouse xenograft experiment.
J Nat Prod. 76:2195–2202. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Feng R and Dong L: Knockdown of
microRNA-127 reverses adriamycin resistance via cell cycle arrest
and apoptosis sensitization in adriamycin-resistant human glioma
cells. Int J Clin Exp Pathol. 8:6107–6116. 2015.PubMed/NCBI
|
|
26
|
Zeng Z, Huang WD, Gao Q, Su ML, Yang YF,
Liu ZC and Zhu BH: Arnebin-1 promotes angiogenesis by inducing
eNOS, VEGF and HIF-1α expression through the PI3K-dependent
pathway. Int J Mol Med. 36:685–697. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lin Q, Guo L, Lin G, Chen Z, Chen T, Lin
J, Zhang B and Gu X: Clinical and prognostic significance of OPN
and VEGF expression in patients with non-small-cell lung cancer.
Cancer Epidemiol. 39:539–544. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ramchandani D and Weber GF: Interactions
between osteopontin and vascular endothelial growth factor:
Implications for cancer. Biochim Biophys Acta. 1855:202–222.
2015.PubMed/NCBI
|
|
29
|
Gong X, Tong Q, Chen Z, Zhang Y, Xu C and
Jin Z: Microvascular density and vascular endothelial growth factor
and osteopontin expression during the implantation window in a
controlled ovarian hyperstimulation rat model. Exp Ther Med.
9:773–779. 2015.PubMed/NCBI
|
|
30
|
Ferrara N: Vascular endothelial growth
factor: Basic science and clinical progress. Endocr Rev.
25:581–611. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chen P, Zhu J, Liu DY, Li HY, Xu N and Hou
M: Over-expression of survivin and VEGF in small-cell lung cancer
may predict the poorer prognosis. Med Oncol. 31:7752014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li C, Li R, Song H, Wang D, Feng T, Yu X,
Zhao Y, Liu J, Yu X, Wang Y and Geng J: Significance of AEG-1
expression in correlation with VEGF, microvessel density and
clinicopathological characteristics in triple-negative breast
cancer. J Surg Oncol. 103:184–192. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li J, Li L, Li Z, Gong G, Chen P, Liu H,
Wang J, Liu Y and Wu X: The role of miR-205 in the VEGF-mediated
promotion of human ovarian cancer cell invasion. Gynecol Oncol.
137:125–133. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chen J, Zhu C, He Z, Geng M, Li G, Tao X
and Zhang F: Association of OPN overexpression with tumor stage,
differentiation, metastasis and tumor progression in human
laryngeal squamous cell carcinoma. Int J Clin Exp Med. 8:7116–7124.
2015.PubMed/NCBI
|
|
35
|
Gao P, Gao YJ and Liang HL: Effect of
NF-κB inhibitor PDTC on VEGF and endostatin expression of mice with
Lewis lung cancer. Asian Pac J Trop Med. 8:220–224. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hu Y, Hu MM, Shi GL, Han Y and Li BL:
Imbalance between vascular endothelial growth factor and endostatin
correlates with the prognosis of operable non-small cell lung
cancer. Eur J Surg Oncol. 40:1136–1142. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lim J, Duong T, Lee G, Seong BL, El-Rifai
W, Ruley HE and Jo D: The effect of intracellular protein delivery
on the anti-tumor activity of recombinant human endostatin.
Biomaterials. 34:6261–6271. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Xu W, Ye P, Li Z, Shi J, Wang W and Yao K:
Endostar, a recently introduced recombinant human endostatin,
inhibits proliferation and migration through regulating growth
factors, adhesion factors and inflammatory mediators in
choroid-retinal endothelial cells. Mol Biol (Mosk). 44:664–670.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sund M, Hamano Y, Sugimoto H, Sudhakar A,
Soubasakos M, Yerramalla U, Benjamin LE, Lawler J, Kieran M, Shah A
and Kalluri R: Function of endogenous inhibitors of angiogenesis as
endothelium-specific tumor suppressors. Proc Natl Acad Sci USA.
102:pp. 2934–2939. 2005, View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lee SH, Jeung IC, Park TW, Lee K, Lee DG,
Cho YL, Lee TS, Na HJ, Park YJ, Lee HG, et al: Extension of the in
vivo half-life of endostatin and its improved anti-tumor activities
upon fusion to a humanized antibody against tumor-associated
glycoprotein 72 in a mouse model of human colorectal carcinoma.
Oncotarget. 6:7182–7194. 2015. View Article : Google Scholar : PubMed/NCBI
|