Introduction
Breast cancer is the most common malignant tumor in
females (1). The prognosis of breast
cancer depends on the age of the patient, tumor grade and treatment
received. Individualizing treatment plans based on
histopathological types of tumor is recommended by tumor experts
for the treatment of breast cancer (2). Based on the phenotypes of breast cancer,
at least four molecular subtypes of breast cancer have been
identified, including luminal A, luminal B, triple
negative/basal-like and human epidermal growth factor receptor 2
(HER2) (3). The multiple molecular
subtypes of breast cancer exhibit distinct sensitivities to
chemotherapy, and different prognoses. Patients with luminal breast
cancer typically have good prognoses and benefit less from
chemotherapy compared with patients with other subtypes of breast
cancer (4). Currently, no effective
chemotherapy drugs are available for triple negative/basal-like
breast cancer. Multi-drug resistance is common in patients with
advanced breast cancer. Furthermore, since the majority of
chemotherapy drugs possess side effects, selecting effective and
safe chemotherapy drugs for patients with advanced breast cancer
poses a substantial challenge. The development of chemotherapy
drugs that are independent of HER2, estrogen and progesterone
receptors is crucial for the future successful treatment of breast
cancer, particularly advanced breast cancer.
Multiple Chinese herbs have been hypothesized to
facilitate the treatment of tumors. However, the molecular
mechanisms underlying the anti-tumor effects associated with
certain Chinese herbs remain to be fully understood. Pteris
semipinnata is a traditional Chinese herb of the Pteridaceae
family. P. semipinnata has been used to treat snake bites,
hepatitis and enteritis due to its detoxifying, swelling-reducing
and pain-relieving effects (5).
Ent-11α-hydroxy-15-oxo-aur-16-en-19-olic acid (5F), a compound
isolated from the leaves of P. semipinnata, crystallizes in
the monoclinic system. The structure consists of three six-membered
rings. Additionally, 5F possesses seven chiral atoms, the
configuration of which, as obtained by anomalous dispersion, is
R-C4, S-C5, R-C8, S-C9, R-C12 and R-C15 (6). Previous studies have reported that 5F
induces apoptosis in and inhibits the proliferation of lung, liver,
anaplastic thyroid, stomach, colon and throat cancer cells
(7–11).
However, whether 5F exerts anti-tumor effects during
the progression of breast cancer remains unclear. The present study
examined the functions of 5F in the growth and apoptosis of three
breast cancer cell lines, including MDA-MB-231, MCF-7 and
SK-BR-3.
Materials and methods
Materials
The human breast cancer cell lines MDA-MB-231,
MCF-7, and SK-BR-3 were obtained from the Central Laboratory of the
Third Affiliated Hospital of Sun Yat-sen University (Guangzhou,
China). Dulbecco's modified Eagle's medium (DMEM), fetal bovine
serum, trypsin, and dimethyl sulfoxide were purchased from Gibco;
Thermo Fisher Scientific, Inc. (Waltham, MA, USA). Cell Counting
Kit-8 (CCK-8) was purchased from Shanghai Dongren Chemical
Technology Co., Ltd. (Shanghai, China). The Annexin V-fluorescein
isothiocyanate (FITC)/propidium iodide (PI) apoptosis kit and
ethanol were purchased from Nanjing KeyGen Biotech Co., Ltd.
(Nanjing, China). PI and ribonuclease (RNase) were purchased from
Sigma-Aldrich; Merck KGaA (Darmstadt, Germany). The BCA Protein
Assay Kit for protein quantification was purchased from Applygen
Technologies, Inc. (Beijing, China). The Cell Total Protein
Extraction Kit used for protein extraction was purchased from
Beyotime Institute of Biotechnology (Haimen, China). B-cell
lymphoma 2 (Bcl-2; cat. no. sc-509), Bcl-2-associated X, apoptosis
regulator (Bax; cat. no. sc-6236), Bcl-2 antagonist/killer (Bak;
cat. no. sc-832), β-actin (cat. no. sc-130300) and caspase-3 (cat.
no. sc-271759) antibodies were purchased from Santa Cruz
Biotechnology, Inc. (Dallas, TX, USA). The reactive oxygen species
detection kit was purchased from Shanghai GenePharma Co., Ltd.
(Shanghai, China). The Key Laboratory of Natural Drug Research and
Development (Guangdong Medical College, Zhanjiang, China) provided
the 5F. 5F was extracted and diluted from P. semipinnata as
described in a previous study (10).
The 5F was dissolved in propylene glycol, volume fraction 0.15, and
diluted with distilled water to prepare 5F stock solutions (1,000
mg/l). The 5F solutions were sterilized using filtration and stored
in a −20°C freezer.
Analysis of cell proliferation
MDA-MB-231, MCF-7 and SK-BR-3 cells were digested
using trypsin, concentration 0.25%, during the logarithmic growth
phase. Using DMEM, breast cancer cell suspensions ranging from
2.0–10.0×104/ml in concentration were prepared. Cancer
cells were inoculated onto a 96-well culture plate
(5.0×103 cells/100 µl/well). Following culture for 24 h,
DMEM was removed and 100 µl 5F solution (0, 5, 10, 20 or 40 µg/ml)
was added to the culture. Eight parallel wells were used for a
combination of one drug and one type of breast cancer cell.
Following incubation for 24, 48 and 72 h at 37°C, the medium
containing 5F at different concentrations was removed, and 100 µl
DMEM and 10 µl CCK-8 were added and incubated for 2 h at 37°C. The
absorbance value (A) at 450 nm was determined using a microplate
reader (BioTek Instruments, Inc., Winooski, VT, USA). The
proliferation-inhibiting effect of 5F on cancer cells was evaluated
according to the following cancer cell proliferation inhibition
rate equation, as obtained from triplicate experiments: Cancer cell
proliferation rate (%) = [1 - (A value of the positive group - A
value of the blank group)/A value of the negative group - A value
of the blank group)] × 100%.
Morphological changes of breast cancer
cells
The concentration of MDA-MB-231, MCF-7 and SK-BR-3
cells at the logarithmic growth phase was set to
2.0×105/ml. Cancer cells (2 ml) were added to a 6-well
plate with coverslips and incubated for 24 h at 37°C until cancer
cells were 100% confluent. Following this, 100 µl 5F (0 or 40
µg/ml) was added and the cells were cultured in DMEM for 48 h at
37°C. Cells were subsequently rinsed in cold PBS twice and fixed in
4% paraformaldehyde at 4°C for 30 min followed by paraffin
embedding at room temperature for 30 min in accordance with the
standard methodology (12). Slices
were cut automatically and manually at thickness of 5 µm by an
automatic microtome. Morphological changes of cells were identified
under transmission electron microscopy (TEM) (Hitachi Model H-7000;
Hitachi, Ltd., Tokyo, Japan), magnification, ×200. Subsequent to
treatment with 0 or 40 µg/ml 5F for 24 h, cells were rinsed with
PBS twice and stained in 10 mg/ml Hoechst 33342 at 37°C for 15 min.
Cells were subsequently rinsed with PBS twice and observed using
fluorescence microscopy.
Flow cytometry
The concentration of MDA-MB-231, MCF-7 and SK-BR-3
cells at the logarithmic growth phase was set to
2.0×105/ml. Cells (2 ml) were inoculated onto a 6-well
plate. Subsequent to incubation for 24 h, when the cells were 100%
confluent, 5F was added to the culture medium at concentrations of
0 and 40 µg/ml. Following incubation in DMEM for 48 h at 37°C, the
morphology of the cells was observed using an inverted microscope.
Subsequently, cells were rinsed with cold PBS twice and digested
with trypsin, concentration 0.25%. Cells were harvested via
centrifugation at 1,000 × g for 5 min at 4°C, rinsed with PBS and
suspended in Annexin V-FITC (5 µl) for 30 min for flow cytometry
analysis on a FACS Calibur flow cytometer (BD Biosciences, Franklin
Lakes, NJ, USA). Data were collected and analyzed using FlowJo
software version 7.6.1 (Tree Star, Inc., Ashland, OR, USA).
Western blotting
The expression of multiple apoptosis-associated
genes, including Bcl-2, Bax and caspase-3, was evaluated using
western blotting. The concentration of MDA-MB-231, MCF-7 and
SK-BR-3 cells at the logarithmic growth phase were set to
2.0×105/ml. Cells (2 ml) were inoculated onto a 6-well
plate. Following 24 h of incubation, during which cells attached to
the wells, 5F was added to the culture to reach concentrations of
0, 20, and 40 µg/ml. Following incubation in DMEM for 24 h at 37°C,
cells were harvested via centrifugation at 1,000 × g for 5 min at
4°C and rinsed with cold PBS twice at 4°C. The harvested cells were
subsequently digested with 200 µl lysate buffer (Gibco; Thermo
Fisher Scientific, Inc.) for 30 min at 4°C. Supernatant was
collected via centrifugation at 1,000 × g for 5 min at 4°C and
proteins were quantified using the Bradford protein quantification
method. Proteins (50 µg) were separated using 12.5% SDS-PAGE and
transferred onto a nitrocellulose membrane. Following blocking in
5% nonfat milk at room temperature for 1 h, the nitrocellulose
membrane was rinsed with Tris buffered saline with Tween [TBST, 20
mM Tris-Hcl (pH=7.6), 137 mM Nacl and 0.01% Tween-20] and incubated
with primary antibodies against Bak (1:200), Bcl-2 (1:200), or
caspase-3 (1:300) at room temperature overnight. β-actin (1:500)
was used as an internal control. Following three rinses with TBST
for 15 min, the nitrocellulose membrane was incubated with
horseradish peroxidase (HRP)-labeled goat-anti rabbit
immunoglobulin G (IgG; 1:5,000; cat. no. sc-2004; Santa Cruz
Biotechnology, Inc. TX, USA) at room temperature for 1 h.
Subsequently, the nitrocellulose membrane was rinsed with TBST for
15 min three times. Following rinsing with PBS for 1 min, the
nitrocellulose membrane was stained with 10 ng/ml enhanced
chemiluminescence reagents (Tiangen, Beijing, China) at 4°C for 1 h
and exposed three times. The western blot analysis was repeated
three times. Then, the blots were analyzed by Image J software
version 1.41 (National Institutes of Health, Bethesda, MD,
USA).
Statistical analyses
Experimental data were presented as the mean ±
standard deviation. Statistical analyses were performed using SPSS
16.0 statistical software (SPSS, Inc., Chicago, IL, USA).
Comparison between two groups was performed by means of independent
samples t test. Comparison among multiple groups was performed by
one-way analysis of variance followed by Tukey's and Tamhane's T2
post-hoc tests. P<0.05 was considered to indicate a
statistically significant difference.
Results
5F inhibited the proliferation of
human breast cancer cells
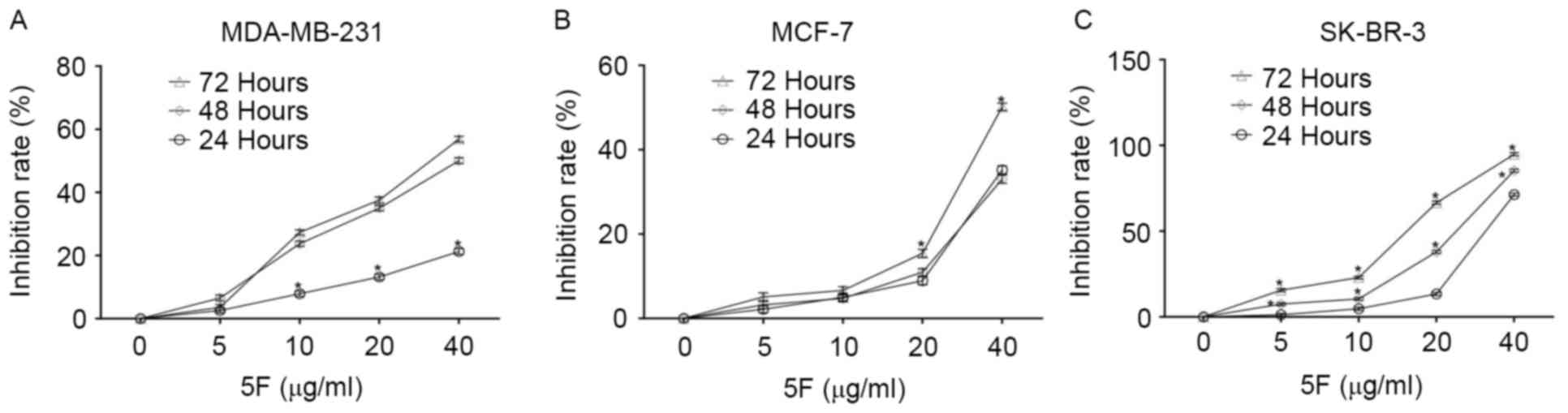
The CCK-8 staining experiment demonstrated that 5F
inhibited the proliferation of the three types of human breast
cancer cell assessed in the present study, MDA-MB-231, MCF-7 and
SK-BR-3 cells, in a time- and concentration-dependent manner
(Fig. 1). SK-BR-3 cells were the most
sensitive to 5F of the three human breast cancer cells; <6% of
SK-BR-3 cells survived following culturing in DMEM containing 5F at
a concentration of 40 µg/ml for 72 h. Following culture in DMEM
containing 5F at a concentration of 40 µg/ml for 72 h, ~50.00 and
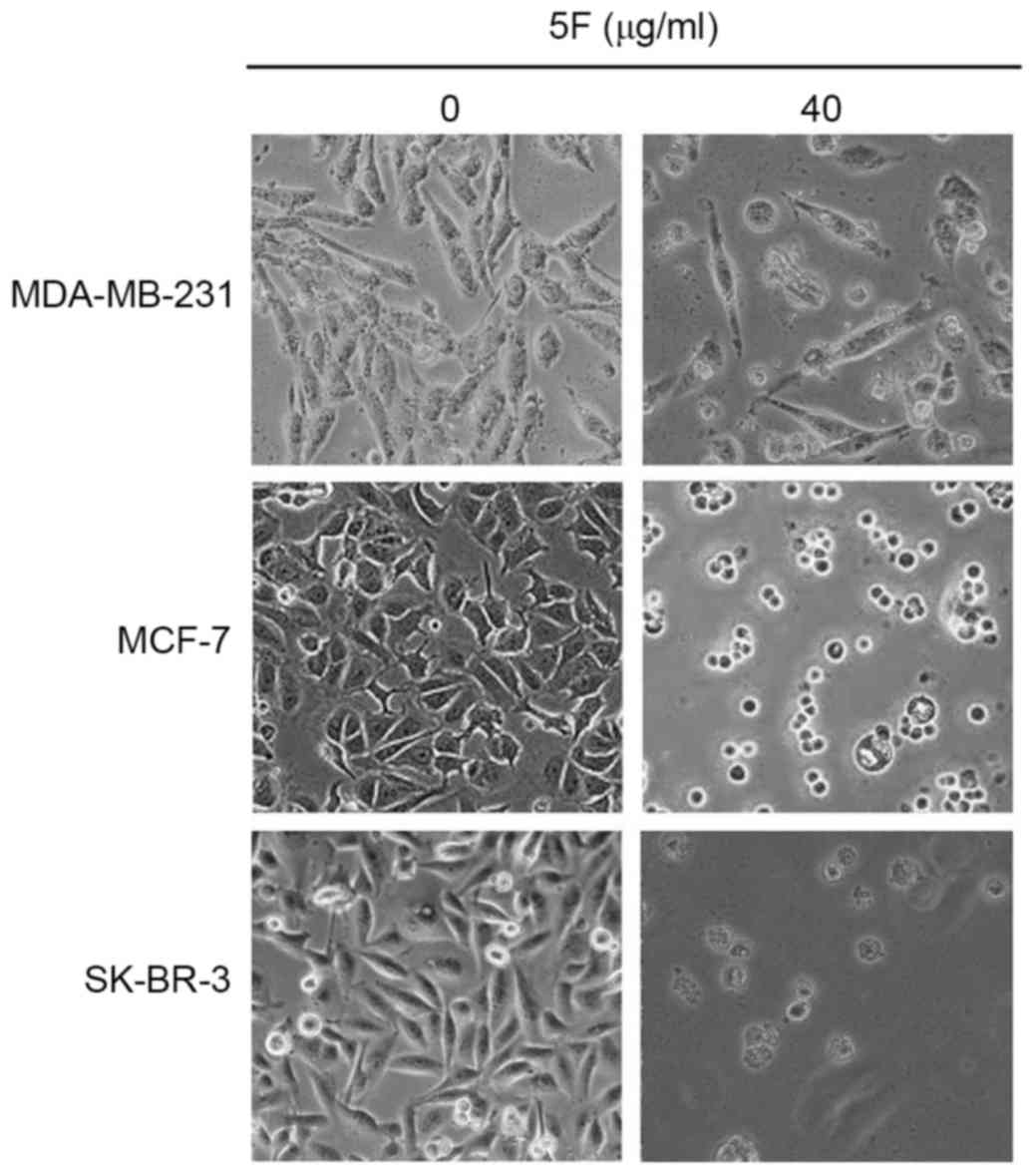
44.13% of MCF-7 and MDA-MB-231 cells survived. Morphological
changes, including membrane rupture and atrophy, disconnection from
wells, and granular bodies in the cytoplasm were observed in all
cancer cells following incubation with DMEM containing 5F at a
concentration of 40 µg/ml for 24 h (Fig.
2).
5F induced the apoptosis of human
breast cancer cells
To assess whether 5F had any effect on the apoptosis
of MDA-MB-231, MCF-7 and SK-BR-3 cells the morphological changes
and the expression of multiple apoptosis-associated proteins in
these human breast cancer cells were evaluated. The cells were
incubated in 5F for 48 h and the morphological changes of the cells
were subsequently observed using TEM. Since few MDA-MB-231 (50%),
MCF-7 (44.13%) and SK-BR-3 (6%) cells survived the 48 h incubation
(Fig. 2), fluorescence microscopy was
used to evaluate the morphological changes of cancer cells
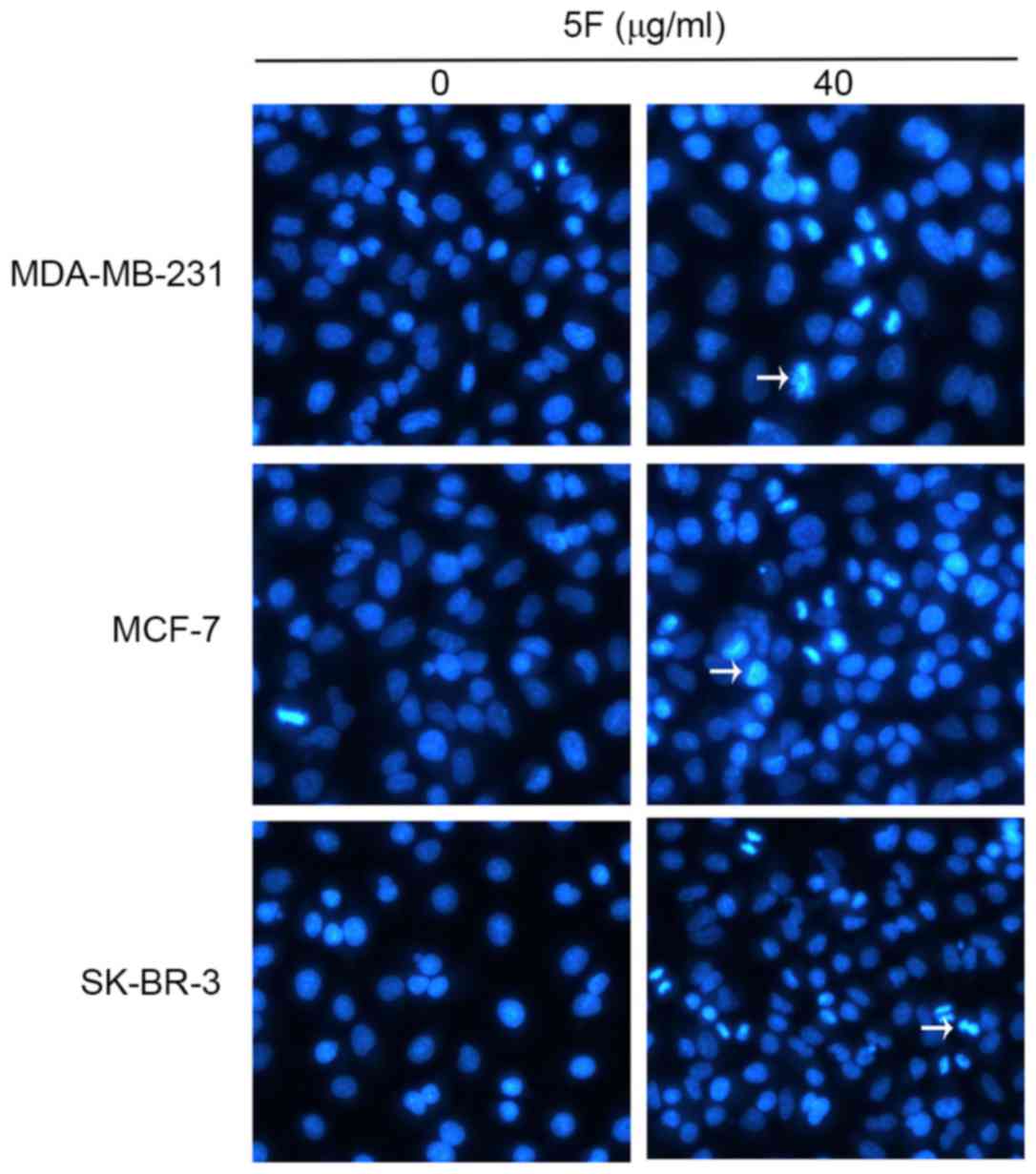
incubated in DMEM containing 5F (40 µg/ml) for 24 h. Cells were
stained with Annexin V/FITC. Condensation and degradation of cancer
cell nuclei were induced by 5F under fluorescence microscopy
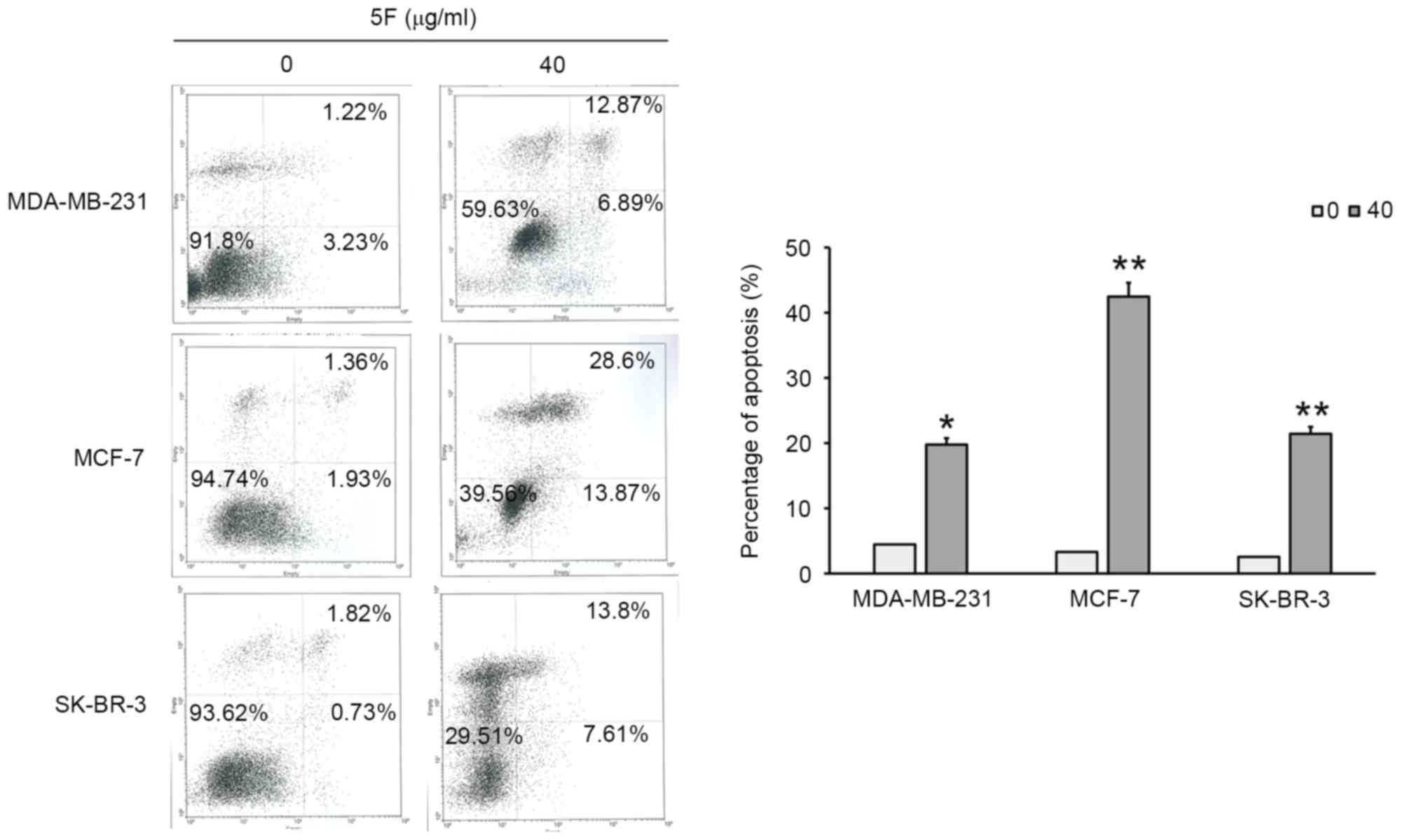
(Fig. 3). The results of flow
cytometry demonstrated that 6.89, 13.87 and 7.61% of MDA-MB-231,
MCF-7, and SK-BR-3 cells, respectively, exhibited in early
apoptosis. The results of flow cytometry also demonstrated that
12.87, 28.6 and 13.8% of MDA-MB-231, MCF-7, and SK-BR-3 cells,
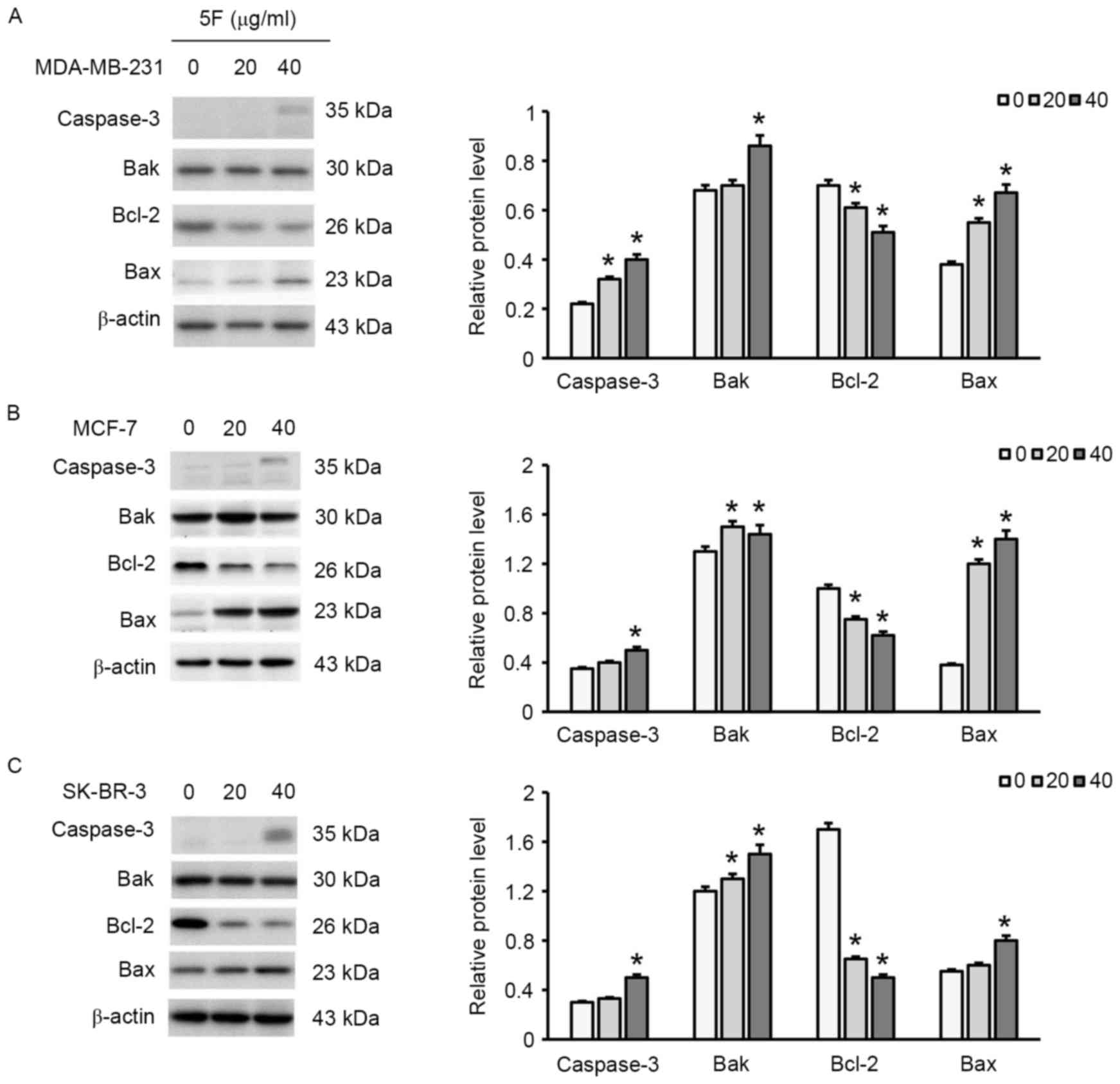
respectively, exhibited late apoptosis (Fig. 4). Furthermore, a decrease in the
expression of Bcl-2 and an increase in the expression of Bax, Bak
and caspase-3 were observed (Fig. 5),
suggesting that 5F induced breast cancer cell apoptosis by
regulating the expression of these apoptosis-associated proteins.
In addition, 40 µg/ml 5F exhibited greater effects on the
expression of Bcl-2, Bax, Bak and caspase-3 than 20 µg/ml 5F,
suggesting that 5F affected the expression of these
apoptosis-associated proteins in a concentration-dependent
manner.
Discussion
5F exerts anti-tumor effects against liver (8), lung (7),
stomach (13), and anaplastic thyroid
(5) cancers, but whether 5F exhibits
anti-tumor effects against breast cancer remains unclear. The
present study assessed the potential function of 5F in the growth
of breast cancer in three different types of breast cancer cell
line: MCF-7, MDA-MB-231 and SK-BR-3. Each line possesses different
hormone receptors: Thyroid hormone receptors, steroid hormone
receptors and estrogen receptor in MCF-7, MDA-MB-231 and SK-BR-3
cell lines, respectively (14,15), and
different HER2 and p53 expression levels. The results of the
present study demonstrated that 5F inhibited the proliferation of
each of these three types of breast cancer cell in a concentration-
and time-dependent manner, suggesting that the anti-tumor effects
of 5F are independent of the hormone receptors, HER2 expression,
and p53 expression in breast cancer cells.
The results of the present study also suggested that
5F promoted the apoptosis of the three breast cancer cell lines in
a concentration- and time-dependent manner. However, the rate of
apoptosis induced by 5F differed among the three breast cancer cell
lines, suggesting that the three lines differed in 5F sensitivity.
This result may be explained by the different characteristics of
the three breast cancer cell lines. However, certain key molecules
may serve crucial functions in the proliferation and apoptosis of
breast cancer cells, though the underlying molecular mechanisms
remain to be fully understood. Apoptosis is an important step in
cell development. The dysregulation of apoptosis leads to numerous
diseases, including the development of cancer (16). Intrinsic and extrinsic pathways are
involved in apoptosis. Extrinsic apoptosis signals are transduced
into cells from cell surface death receptors, a pathway known as
the death receptor pathway (17).
Cell surface death receptors include the tumor necrosis factor and
nerve growth factor superfamilies. Extracellular death signals are
transduced into cells through the binding of death receptors and
ligands. Intrinsic apoptosis pathways include the mitochondrial
pathway and the endoplasmic reticulum pathway (18). In the mitochondrial pathway, the Bax
subfamily protein inserts into the mitochondrial membrane from the
outer mitochondrial membrane or cytoplasm, causing changes in the
permeability of the mitochondrial membrane. Subsequently, the
mitochondrial membrane potential decreases and cytochrome C,
multiple other proteins and apoptosis-inducing factors are released
from the mitochondrion into the cytoplasm (19). The release of cytochrome C is key in
the mitochondrial apoptosis pathway; activated cytochrome C
activates caspase-9 and downstream caspase-3, and induces apoptosis
(20).
The activation, expression and regulation of
numerous genes is crucial in apoptosis (21). Of these genes, the Bcl-2 family genes
serve key functions in the regulation of apoptosis. Bcl-2 family
proteins are divided into two groups: Anti-apoptotic and
pro-apoptotic. Anti-apoptotic members include Bcl-2, Bcl-W and
Bcl-2 family apoptosis regulator; pro-apoptotic members include
Bax, Bak, Bcl-extra small, Bcl-2 associated agonist of cell death,
harakiri, and Bcl-2 homology region 3 interacting domain death
agonist (22). Apoptosis occurs when
the activities of anti-apoptotic proteins are lower than those of
pro-apoptotic proteins (23). The
western blotting results of the present study demonstrated that 5F
increased the expression of Bax and decreased the expression of
Bcl-2 in breast cancer cells. Furthermore, increasing the
concentration of 5F increased the expression of caspase-3. A
previous study reported that 5F was involved in the regulation of
apoptosis and of the cell cycle by affecting the expression of
extracellular regulated kinase 1/2, c-jun N-terminal kinase, and
p38 (24). Therefore, the present
study speculated that 5F arrests the cell cycle and promotes
apoptosis by downregulating Bcl-2 and Bcl-extra large, upregulates
Bax and Bak and inhibits the expression of proliferation-associated
proteins (10). The results of the
present study suggested that 5F induced the apoptosis of breast
cancer cells by affecting the balance of activity of
apoptosis-promoting and -inhibiting proteins of the Bcl-2
family.
To conclude, 5F inhibited the proliferation of
breast cancer cells in a time- and concentration-dependent manner.
Furthermore, 5F induced the apoptosis of breast cancer cells.
Acknowledgements
The present study was supported by the Guangdong
Medical Scientific Research Fund (grant no. A2016004) and the
Medical Scientific Research Foundation of Guangdong Province [grant
no. (2016) 568].
References
|
1
|
Benson JR and Jatoi I: The global breast
cancer burden. Future Oncol. 8:697–702. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Nandy A, Gangopadhyay S and Mukhopadhyay
A: Individualizing breast cancer treatment-The dawn of personalized
medicine. Exp Cell Res. 320:1–11. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Slamon DJ, Leyland-Jones B, Shak S, Fuchs
H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M,
et al: Use of chemotherapy plus a monoclonal antibody against HER2
for metastatic breast cancer that overexpresses HER2. N Engl J Med.
344:783–792. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lim E and Winer EP: Adjuvant chemotherapy
in luminal breast cancers. Breast. 20 Suppl 3:S128–131. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu ZM, Chen GG, Vlantis AC, Liang NC,
Deng YF and van Hasselt CA: Cell death induced by
ent-11alpha-hydroxy-15-oxo-kaur-16-en-19-oic-acid in anaplastic
thyroid carcinoma cells is via a mitochondrial-mediated pathway.
Apoptosis. 10:1345–1356. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Brunocolmenarez J, Peña A, Alarcón L,
Usubillaga A and Delgadoméndez P: Structure of
ent-15a-hydroxy-kaur-16-en-19-oic acid. Avances En Química.
6:16–20. 2011.
|
|
7
|
Li L, Chen GG, Lu YN, Liu Y, Wu KF, Gong
XL, Gou ZP, Li MY and Liang NC:
Ent-11α-Hydroxy-15-oxo-kaur-16-en-19-oic-acid inhibits growth of
human lung cancer A549 cells by arresting cell cycle and triggering
apoptosis. Chin J Cancer Res. 24:109–115. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen GG, Leung J, Liang NC, Li L, Wu K,
Chan UP, Leung BC, Li M, Du J, Deng YF, et al:
Ent-11α-hydroxy-15-oxo-kaur-16-en-19-oic-acid inhibits
hepatocellular carcinoma in vitro and in vivo via stabilizing IkBα.
Invest New Drugs. 30:2210–2218. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu Z, Ng EK, Liang NC, Deng YF, Leung BC
and Chen GG: Cell death induced by Pteris semipinnata L. Is
associated with p53 and oxidant stress in gastric cancer cells.
FEBS Lett. 579:1477–1487. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen GG, Liang NC, Lee JF, Chan UP, Wang
SH, Leung BC and Leung KL: Over-expression of Bcl-2 against Pteris
semipinnata L-induced apoptosis of human colon cancer cells via a
NF-kappa B-related pathway. Apoptosis. 9:619–627. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vlantis AC, Lo CS, Chen GG, Liang Ci N,
Lui VW, Wu K, Deng YF, Gong X, Lu Y, Tong MC and van Hasselt CA:
Induction of laryngeal cancer cell death by
Ent-11-hydroxy-15-oxo-kaur-16-en-19-oic acid. Head Neck.
32:1506–1518. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mgbonyebi OP, Russo J and Russo IH:
Roscovitine induces cell death and morphological changes indicative
of apoptosis in MDA-MB-231 breast cancer cells. Cancer Res.
59:1903–1910. 1999.PubMed/NCBI
|
|
13
|
Chen JF, Chen YX, Li P, Fu M, Lv YN and Li
L: Effect of Ent-11α-hydroxy-15-oxo-kaur-16-en-19-oic-acid on human
gastric cancer cells and its mechanism. Nan Fang Yi Ke Da Xue Xue
Bao. 31:1345–1348. 2011.(In Chinese). PubMed/NCBI
|
|
14
|
Crépin M, Salle V, Raux H, Berger R,
Hamelin R, Brouty-Boyé D and Israel L: Steroid hormone receptors
and tumorigenicity of sublines from breast tumor metastatic MDA-MB
231 cell line. Anticancer Res. 10:1661–1666. 1990.PubMed/NCBI
|
|
15
|
Duncan RE and Archer MC: Farnesol induces
thyroid hormone receptor (THR) beta1 but inhibits THR-mediated
signaling in MCF-7 human breast cancer cells. Biochem Biophys Res
Commun. 343:239–243. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Reed JC: Dysregulation of apoptosis in
cancer. J Clin Oncol. 17:2941–2953. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gupta S: Molecular steps of death receptor
and mitochondrial pathways of apoptosis. Life Sci. 69:2957–2964.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pinton P, Ferrari D, Rapizzi E, Di
Virgilio F, Pozzan T and Rizzuto R: The Ca2+ concentration of the
endoplasmic reticulum is a key determinant of ceramide-induced
apoptosis: Significance for the molecular mechanism of Bcl-2
action. EMBO J. 20:2690–2701. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gross A, McDonnell JM and Korsmeyer SJ:
BCL-2 family members and the mitochondria in apoptosis. Genes Dev.
13:1899–1911. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Eldering E, Mackus WJ, Derks IA, Evers LM,
Beuling E, Teeling P, Lens SM, van Oers MH and van Lier RA:
Apoptosis via the B cell antigen receptor requires Bax
translocation and involves mitochondrial depolarization, cytochrome
C release, and caspase-9 activation. Eur J Immunol. 34:1950–1960.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen F, Jiang X, Chen X, Liu G and Ding J:
Effects of downregulation of inhibin alpha gene expression on
apoptosis and proliferation of goose granulosa cells. J Genet
Genomics. 34:1106–1113. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Pilchova I, Klacanova K, Chomova M,
Tatarkova Z, Dobrota D and Racay P: Possible contribution of
proteins of Bcl-2 family in neuronal death following transient
global brain ischemia. Cell Mol Neurobiol. 35:23–31. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Guo B, Zhai D, Cabezas E, Welsh K,
Nouraini S, Satterthwait AC and Reed JC: Humanin peptide suppresses
apoptosis by interfering with Bax activation. Nature. 423:456–461.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li MY, Liang NC and Chen GG:
Ent-11α-hydroxy-15-oxo-kaur-16-en-19-oic-acid induces apoptosis of
human malignant cancer cells. Curr Drug Targets. 13:1730–1737.
2012. View Article : Google Scholar : PubMed/NCBI
|