Introduction
Glioma, a major malignant tumor of the central
nervous system, is characterized by high incidence, recurrence and
mortality rates, and low cure rate (1). In-depth analysis of the mechanism of
tumorigenesis and the identification of specific molecular markers
and intervention targets has become a hot topic in tumor research.
Several studies have confirmed that, Wnt signaling pathway, which
is closely related to central nervous system diseases, is involved
in the proliferation, migration and differentiation processes of
neural stem cells (2). The classic
Wnt/β-catenin pathway has been extensively studied (2). Autophagy, also known as type II
programmed cell death, has been proved to be closely related to the
maintenance of cell homeostasis and occurrence of disease.
Autophagy can participate in the cell cycle, cell proliferation,
cell apoptosis and angiogenesis of tumor cells, thus affecting
occurrence and development of tumors (3,4). A variety
of chemotherapy drugs can induce autophagic activity of glioma
cells, which in turn show a curative effect, and reduce drug
resistance (5). Inflammation is also
an important factor involved in the development, progression and
treatment processes of various tumor cells (6). Based on the above, the aim of this study
was to analyze the mechanism of Wnt signaling pathway in the
regulation of autophagy and inflammatory response in glioma
cells.
Materials and methods
Experimental materials
Human glioma cell line U118 was purchased from
Research Science (Shanghai, China); High glucose DMEM medium and
fetal bovine serum were purchased from Sigma (St. Louis, MO, USA);
Wnt inhibitor IWR-1 was from Beyotime (Jiangsu, China); RIPA lysate
and phenylmethylsulfonyl fluoride (PMSF) were purchased from
Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd.; BCA kits
were from Europe B.V. (Venlo, The Netherlands); mouse anti-human
Wnt5a, β-catenin, marker
microtubule-associated-proteins-1A/1B-light-chain-3C (LC-3) II and
Beclin I, IL-6 and TNF-α monoclonal antibodies were from Santa Cruz
Biotechnology, Inc. (Santa Cruz, CA, USA) (cat. nos. sc-365370,
sc-65480, sc-398822, sc-48341, sc-130326 and sc-130349); β-actin
antibody and rabbit anti-mouse polyclonal antibody were purchased
from Invitrogen (Carlsbad, CA, USA) (cat. no. MA5-15739, A-11059);
TBST and ECL solutions were purchased from General Electric Co.
(Fairfield, CT, USA); Cell culture plates and CO2
incubators were from Bio-Rad (Hercules, CA, USA), electrophoresis
apparatus trophoresis was from Applied Biosystems (City Foster, CA,
USA); Polyvinylidene fluoride film (PDVF) was from R&D Systems
(Minneapolis, MN, USA).
Experimental grouping
After resuscitation, U118 cells were cultured with
high glucose DMEM medium containing 10% fetal bovine serum in an
incubator (37°C, 5% CO2). Subcultures were prepared and
medium was replaced every other day. Cells were collected during
the logarithmic growth phase and resuspended in PBS to adjust the
density to 2×106/ml. Cells were divided into 3 groups:
the Wnt signal pathway inhibitor IWR-1 group (5 µmol/l, observation
group), the PBS negative control group (PBS group) and the blank
control group. All groups were incubated for another 24 h.
Observation indicators and examination
methods
Western blot was used to evaluate Wnt5a/β-catenin
protein, autophagy marker
microtubule-associated-proteins-1A1B-light-chain-3C (LC-3) II and
Beclin I, and inflammatory factors IL-6 and TNF-α protein
expression levels. Western blotting: medium containing cells of
each group was placed on ice, and 150 µl of RIPA lysate and 1.5 µl
of PMSF were added and incubated for 30 min, followed by
centrifugation (2,000 × g) at 4°C for 20 min and the supernatant
was collected. Protein concentration was measured using BCA method.
Protein (15 µg) from each sample was subjected to 8% polyacrylamide
gel (SDS-PAGE) electrophoresis, followed by transfer to PDVF
membrane. Membrane was blocked with 5% skimmed milk at room
temperature for 2 h. Mouse anti-human Wnt5a, β-catenin, LC-3 II,
Beclin I, IL-6 and TNF-α monoclonal antibodies (1:2,000) were then
added and incubated overnight at 4°C. After washing with TBST,
rabbit anti-mouse polyclonal antibody (1:500) was added and
incubated at room temperature for 4 h. After washing with TBST,
color development with ECL was performed, and results were scanned
and recorded. Lab Works 4.5 gel imaging software (Invitrogen) was
used for semi-quantitative analysis with results expressed as
integrated optical density (IOD). The relative expression levels of
targeted proteins were represented by the ratio of IOD to that of
β-actin (endogenous control).
Statistical analysis
Statistical analysis was performed using SPSS 20.0
software (SPSS Inc., Chicago, IL, USA). Measurement data were
expressed as mean ± standard deviation. Single factor ANOVA
analysis was used for comparisons among multiple groups and LSD-t
method was used for the comparisons between 2 groups. P<0.05 was
considered to indicate a statistically significant difference.
Results
Expression levels of Wnt5a/β-catenin
protein
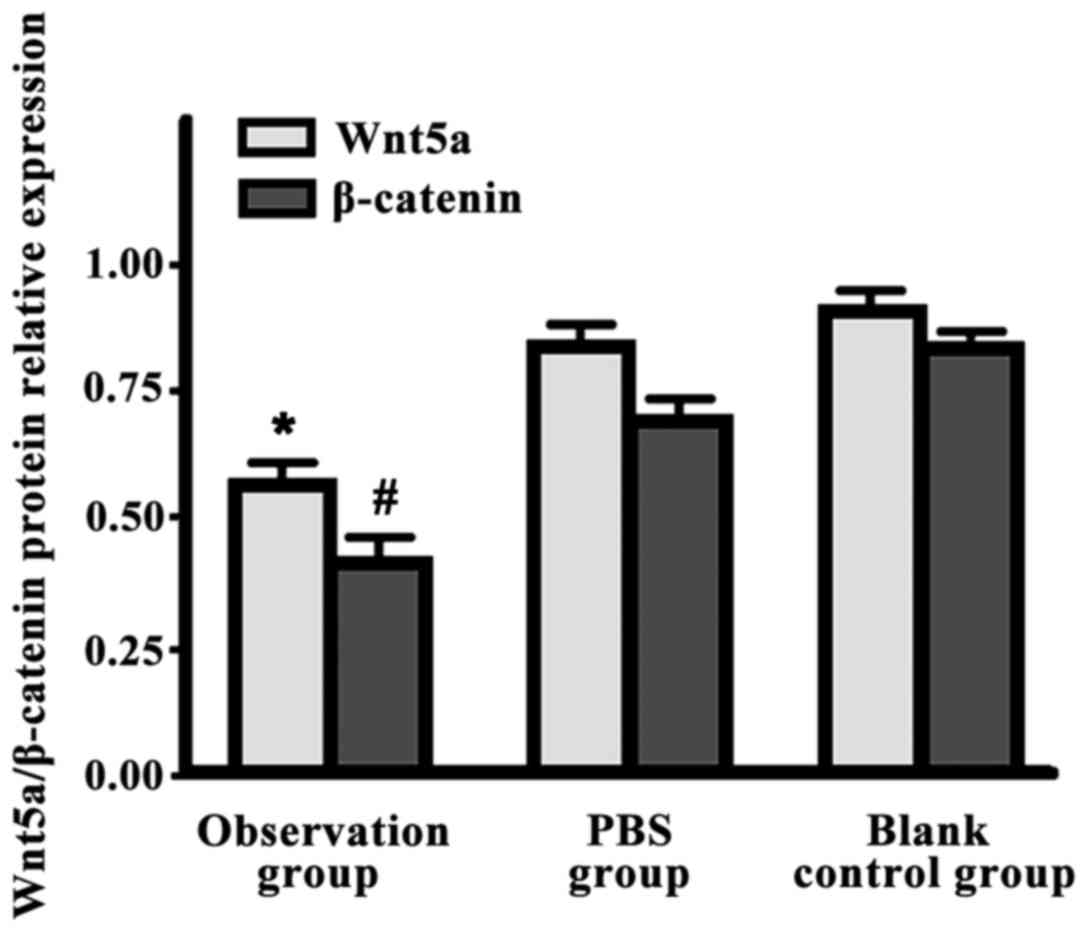
Wnt5a/β-catenin protein expression levels in the
observation group were significantly lower than those in the other
two groups (P<0.05) (Fig. 1).
Expression levels of LC-3 II and
Beclin I
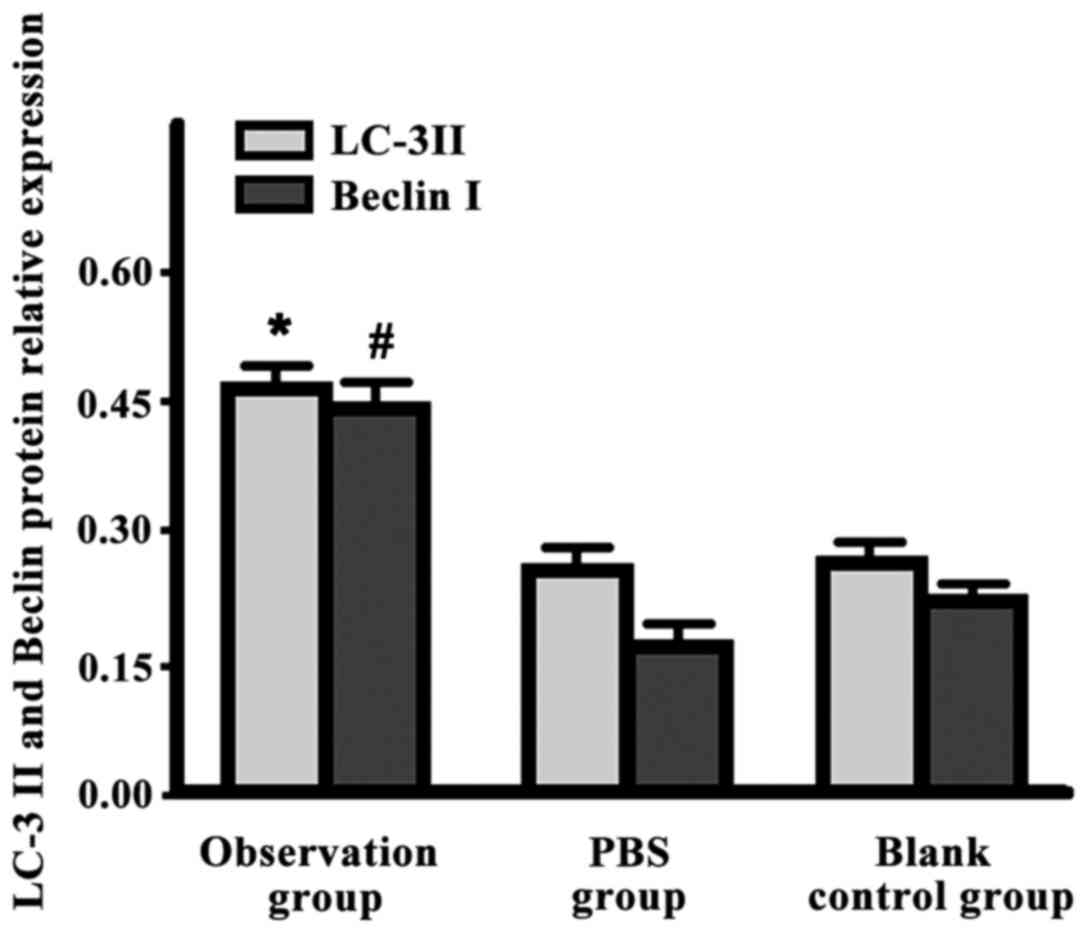
LC-3 II and Beclin I expression levels in the
observation group were significantly higher than those in the other
two groups (P<0.05) (Fig. 2).
Expression levels of IL-6 and
TNF-α
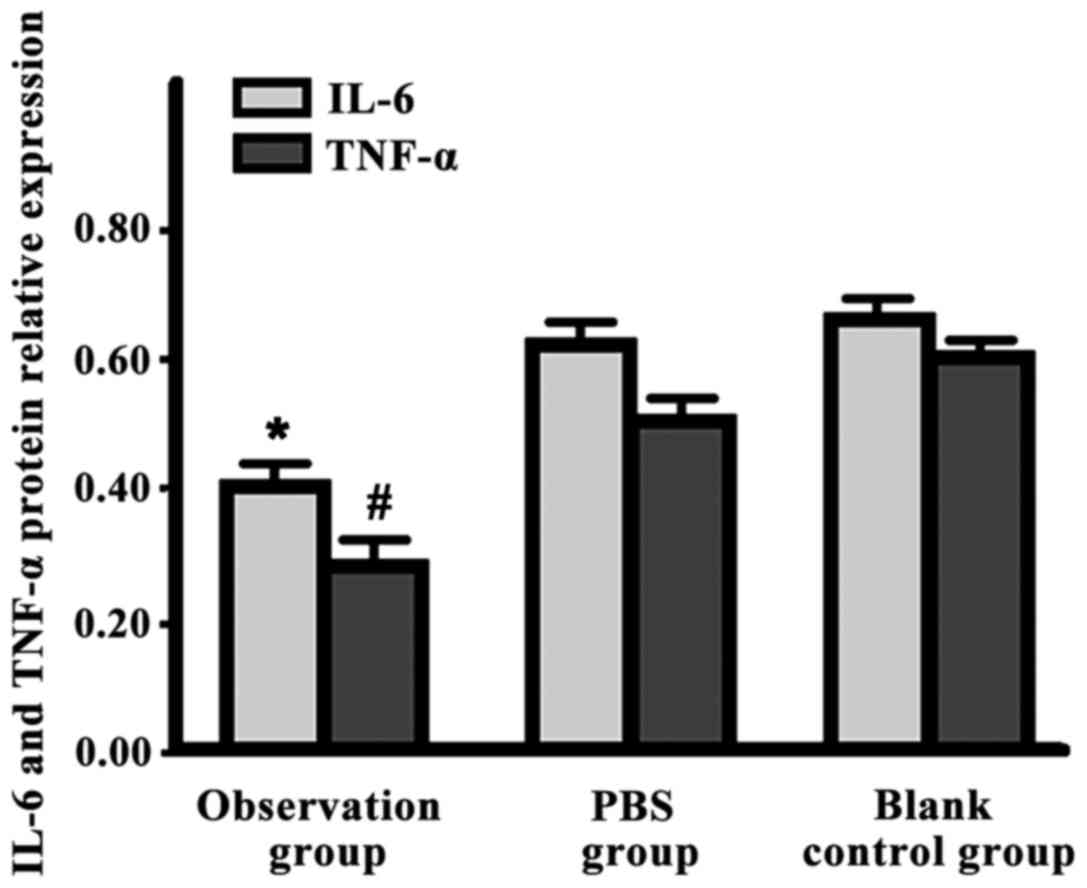
IL-6 and TNF-α expression levels in the observation
group were significantly lower than those in the other two groups
(P<0.05) (Fig. 3).
Discussion
Wnt pathway includes classical Wnt/β-catenin,
Wnt/PCP (7) and Wnt/Ca2+
pathway (8), within which the
activation of Wnt/β-catenin can lead to the binding of Wnt protein
to specific cell surface frizzled receptor proteins and low density
lipoprotein receptor-related protein 5/6 (9). This binding can activate the
intracellular PDZ domain containing dishevelled protein and Axin
protein to inhibit the phosphorylation of glycogen synthase kinase
3 (GSK 3), and consequently lead to the accumulation of β-catenin
in cytoplasm (9). β-catenin can enter
nucleus and bind to the transcription factor T
cytokine/lymphotropic enhancer family protein and initiate the
transcription, thereby regulating the expression of downstream
related target genes and promoting proliferation, invasion and
metastasis of tumor cells (10). Our
results showed that Wnt5a/β-catenin, IL-6 and TNF-α protein
expression levels were significantly lower and LC-3II and Beclin I
protein expression levels were significantly higher in observation
group. Therefore, we believe that Wnt5a/β-catenin signaling pathway
regulates the autophagy and inflammatory response of glioma
cells.
Prior studies confirmed (11) that Wnt is a key factor in the
self-renewal of neural stem cells, and Wnt also participates in
neural formation in adult brain tissue. Mutations of Wnt gene and
knockout of Wnt1 gene can lead to the loss of midbrain in mice
(12). Wnt can act on mouse neural
precursor cells to increase the number of new neurons (13). Wnt pathway can also affect the
synaptic transmission and plasticity of hippocampal neurons
(14). Wnt/β-catenin pathway in adult
brain is closely related to the occurrence of Alzheimer's disease
(15). Wnt5a protein is a member of
the Wnt protein family, which can mediate nonclassical pathways to
inhibit classical pathways, and the inhibition of classic pathwaty
can in turn trigger Wnt5a pathway to participate in regulating the
development of the central nervous system (16). IWR-1 is a small molecule inhibitor of
the Wnt pathway that plays a role in stabilizing Axin degradation
complex (17). LC3 is a marker
protein used for detecting autophagy. LC3-I has the ability to bind
to phosphatidylethanolamine after autophagy and form LC3-II. LC3-II
is localized in the phagocytic endometrium and adventitia and can
be kept stably in autophagic membrane until the fusion with
lysosomes (18). LC3-II level can
reflect the number of autophagosomes and the degree of autophagy
(18). Beclin I is the first
identified mammalian autophagy protein, and it has been shown that
Beclin I gene deletion is directly related to the development of
human breast cancer (19), colorectal
cancer (20) and prostate cancer
(21). The recovery of Beclin I
expression can induce autophagy and inhibit the occurrence and
development of tumors. Mechanism of autophagy in treatment response
of cancer is complex. Autophagy can both suppress and protect tumor
cells (22) and there are researchers
who believe that the regulation of autophagy may be a new target
for the treatment of a variety of tumors.
In addition, inflammatory response with IL-6 and
TNF-α as the central network factors, is also an important
mechanism involved in tumorigenesis. It has been confirmed that,
the occurrence, development, treatment and prognosis of lung cancer
(23), liver cancer (24), colorectal cancer (25) among other cancers are closely related
to inflammation. Proinflammatory stimulation factors such as IL-6
and TNF-α can mediate the activation of a variety of inflammatory
cells such as neutrophils and fibroblasts and inflammatory
mediators such as IL-1, IL-8, INF-γ and NF-κB, which in turn cause
inflammatory disorders. High levels of IL-6 and TNF-α have been
reported in glioma patients, and these levels have been shown to be
associated with the chemotherapy outcomes and the prognosis
(26,27).
We concluded that the occurrence of glioma is
related to Wnt5a/β-catenin signaling pathway activation, cell
autophagy and inflammatory response. Our study provided a new
direction for bio-targeted therapy.
References
|
1
|
Ostrom QT, Gittleman H, Fulop J, Liu M,
Blanda R, Kromer C, Wolinsky Y, Kruchko C and Barnholtz-Sloan JS:
CBTRUS Statistical Report: Primary brain and central nervous system
tumors diagnosed in the United States in 2008–2012. Neuro Oncol. 17
Suppl 4:iv1–iv62. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Duchartre Y, Kim YM and Kahn M: The Wnt
signaling pathway in cancer. Crit Rev Oncol Hematol. 99:141–149.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen N and Karantza V: Autophagy as a
therapeutic target in cancer. Cancer Biol Ther. 11:157–168. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kaza N, Kohli L and Roth KA: Autophagy in
brain tumors: A new target for therapeutic intervention. Brain
Pathol. 22:89–98. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Xue H, Yuan G, Guo X, Liu Q, Zhang J, Gao
X, Guo X, Xu S, Li T, Shao Q, et al: A novel tumor-promoting
mechanism of IL6 and the therapeutic efficacy of tocilizumab:
Hypoxia-induced IL6 is a potent autophagy initiator in glioblastoma
via the p-STAT3-MIR155-3p-CREBRF pathway. Autophagy. 12:1129–1152.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chen W, Xia T, Wang D, Huang B, Zhao P,
Wang J, Qu X and Li X: Human astrocytes secrete IL-6 to promote
glioma migration and invasion through upregulation of cytomembrane
MMP14. Oncotarget. 7:62425–62438. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kikuchi A, Yamamoto H and Kishida S:
Multiplicity of the interactions of Wnt proteins and their
receptors. Cell Signal. 19:659–671. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lerner UH and Ohlsson C: The WNT system:
Background and its role in bone. J Intern Med. 277:630–649. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Prakash S and Swaminathan U: β catenin in
health: A review. J Oral Maxillofac Pathol. 19:230–238. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bielen H and Houart C: The Wnt cries many:
Wnt regulation of neurogenesis through tissue patterning,
proliferation, and asymmetric cell division. Dev Neurobiol.
74:772–780. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Nusse R: Wnt signaling and stem cell
control. Cell Res. 18:523–527. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Adachi K, Mirzadeh Z, Sakaguchi M,
Yamashita T, Nikolcheva T, Gotoh Y, Peltz G, Gong L, Kawase T,
Alvarez-Buylla A, et al: Beta-catenin signaling promotes
proliferation of progenitor cells in the adult mouse subventricular
zone. Stem Cells. 25:2827–2836. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Van Camp JK, Beckers S, Zegers D and Van
Hul W: Wnt signaling and the control of human stem cell fate. Stem
Cell Rev. 10:207–229. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cerpa W, Godoy JA, Alfaro I, Farías GG,
Metcalfe MJ, Fuentealba R, Bonansco C and Inestrosa NC: Wnt-7a
modulates the synaptic vesicle cycle and synaptic transmission in
hippocampal neurons. J Biol Chem. 283:5918–5927. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Maguschak KA and Ressler KJ: A role for
WNT/β-catenin signaling in the neural mechanisms of behavior. J
Neuroimmune Pharmacol. 7:763–773. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Boitard M, Bocchi R, Egervari K, Petrenko
V, Viale B, Gremaud S, Zgraggen E, Salmon P and Kiss JZ: Wnt
signaling regulates multipolar-to-bipolar transition of migrating
neurons in the cerebral cortex. Cell Rep. 10:1349–1361. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lee SC, Kim OH, Lee SK and Kim SJ: IWR-1
inhibits epithelial-mesenchymal transition of colorectal cancer
cells through suppressing Wnt/β-catenin signaling as well as
survivin expression. Oncotarget. 6:27146–27159. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu WT, Huang CY, Lu IC and Gean PW:
Inhibition of glioma growth by minocycline is mediated through
endoplasmic reticulum stress-induced apoptosis and autophagic cell
death. Neuro Oncol. 15:1127–1141. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Al Dhaheri Y, Attoub S, Ramadan G, Arafat
K, Bajbouj K, Karuvantevida N, AbuQamar S, Eid A and Iratni R:
Carnosol induces ROS-mediated beclin1-independent autophagy and
apoptosis in triple negative breast cancer. PLoS One.
9:e1096302014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wu S, Sun C, Tian D, Li Y, Gao X, He S and
Li T: Expression and clinical significances of Beclin1, LC3 and
mTOR in colorectal cancer. Int J Clin Exp Pathol. 8:3882–3891.
2015.PubMed/NCBI
|
|
21
|
Suh Y, Afaq F, Khan N, Johnson JJ, Khusro
FH and Mukhtar H: Fisetin induces autophagic cell death through
suppression of mTOR signaling pathway in prostate cancer cells.
Carcinogenesis. 31:1424–1433. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hamed HA, Yacoub A, Park MA, Eulitt P,
Sarkar D, Dimitrie IP, Chen CS, Grant S, Curiel DT, Fisher PB, et
al: OSU-03012 enhances Ad.7-induced GBM cell killing via ER stress
and autophagy and by decreasing expression of mitochondrial
protective proteins. Cancer Biol Ther. 9:526–536. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tan X, Carretero J, Chen Z, Zhang J, Wang
Y, Chen J, Li X, Ye H, Tang C, Cheng X, et al: Loss of p53
attenuates the contribution of IL-6 deletion on suppressed tumor
progression and extended survival in Kras-driven murine lung
cancer. PLoS One. 8:e808852013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liu F, Zhang W, Yang F, Feng T, Zhou M, Yu
Y, Yu X, Zhao W, Yi F, Tang W, et al: Interleukin-6-stimulated
progranulin expression contributes to the malignancy of
hepatocellular carcinoma cells by activating mTOR signaling. Sci
Rep. 6:212602016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Banday MZ, Balkhi HM, Hamid Z, Sameer AS,
Chowdri NA and Haq E: Tumor necrosis factor-α (TNF-α)-308G/A
promoter polymorphism in colorectal cancer in ethnic Kashmiri
population - a case control study in a detailed perspective. Meta
Gene. 9:128–136. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shan Y, He X, Song W, Han D, Niu J and
Wang J: Role of IL-6 in the invasiveness and prognosis of glioma.
Int J Clin Exp Med. 8:9114–9120. 2015.PubMed/NCBI
|
|
27
|
Zhao X, Laver T, Hong SW, Twitty GB Jr,
Devos A, Devos M, Benveniste EN and Nozell SE: An NF-κB p65-cIAP2
link is necessary for mediating apoptotic resistance to TNF-α in
gliomas. J Neurooncol. 102:367–381. 2011. View Article : Google Scholar : PubMed/NCBI
|