|
1
|
Zweegman S, Engelhardt M and Larocca A:
EHA SWG on ‘Aging and Hematology’: Elderly patients with multiple
myeloma: Towards a frailty approach? Curr Opin Oncol. 29:315–321.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chesi M and Bergsagel PL: Molecular
pathogenesis of multiple myeloma: Basic and clinical updates. Int J
Hematol. 97:313–323. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ramsenthaler C, Kane P, Gao W, Siegert RJ,
Edmonds PM, Schey SA and Higginson IJ: Prevalence of symptoms in
patients with multiple myeloma: A systematic review and
meta-analysis. Eur J Haematol. 97:416–429. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dimopoulos MA and Terpos E: Renal
insufficiency and failure. Hematology Am Soc Hematol Educ Program.
2010:431–436. 2010.PubMed/NCBI
|
|
5
|
Knudsen LM, Hippe E, Hjorth M, Holmberg E
and Westin J: Renal function in newly diagnosed multiple myeloma-a
demographic study of 1353 patients. The Nordic Myeloma Study Group.
Eur J Haematol. 53:207–212. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rekhtina IG and Mendeleeva LP: Current
approaches to treating of patients with multiple myeloma with renal
failure: Questions and proofs. Ter Arkh. 89:112–117. 2017.(In
Russian; Abstract available in Russian from the publisher).
View Article : Google Scholar : PubMed/NCBI
|
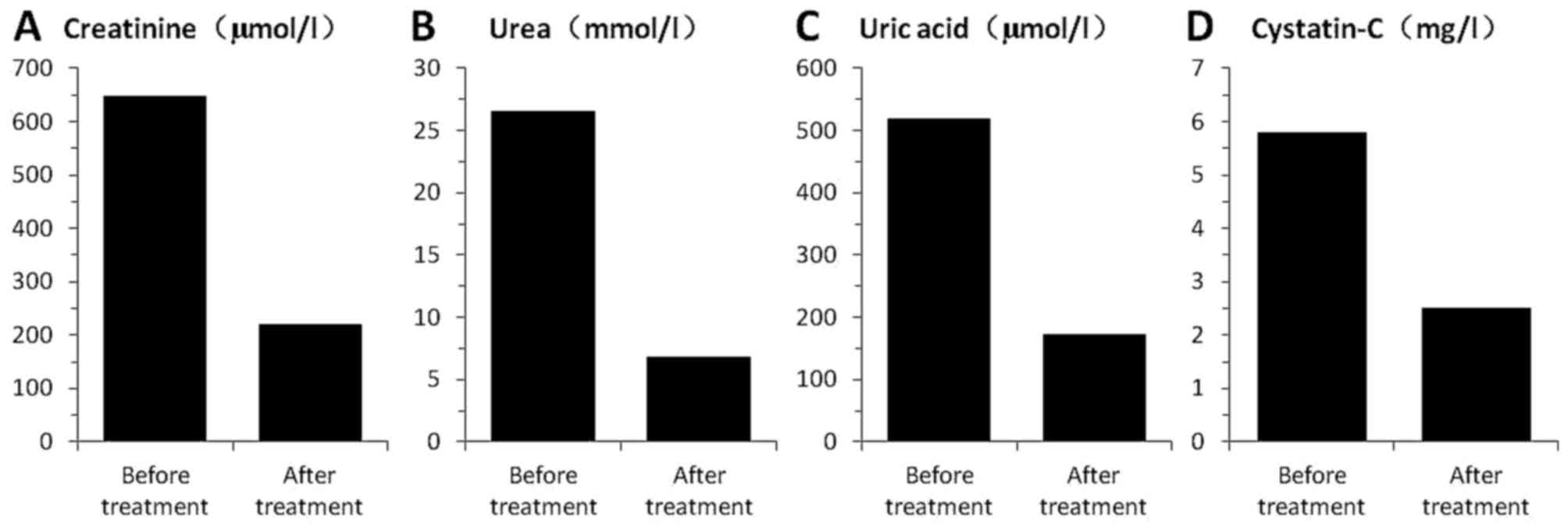
|
7
|
Joseph NS, Gentili S, Kaufman JL, Lonial S
and Nooka AK: High-risk mutiple myiloma: Definition and management.
Clin Lymphoma Myeloma Leuk. 17S:S80–S87. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Caillon H, Irimia A, Simon JS, Axel A,
Sasser K, Scullion MJ, Ligneel T, Nouadje G, Moreau P and Dejoie T:
Overcoming the interference of daratumumab with immunofixation
electrophoresis (IFE) using an industry-developed dira test:
Hydrashift 2/4 daratumumab. Blood. 128:2063. 2016.
|
|
9
|
Puig N, Sarasquete ME, Balanzategui A,
Martínez J, Paiva B, García H, Fumero S, Jiménez C, Alcoceba M,
Chillón MC, et al: Critical evaluation of ASO RQ-PCR for minimal
residual disease evaluation in multiple myeloma. A comparative
analysis with flow cytometry. Leukemia. 28:391–397. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ross FM, Avet-Loiseau H, Ameye G,
Gutiérrez NC, Liebisch P, O'Connor S, Dalva K, Fabris S, Testi AM,
Jarosova M, et al: Report from the european myeloma network on
interphase FISH in multiple myeloma and related disorders.
Haematologica. 97:1272–1277. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Abramova TV, Obukhova TN, Mendeleeva LP,
Pokrovskaya OS, Gribanova EO, Ryzhko VV, Grebenyuk LA, Nareyko MV,
Solovyev MV, Votyakova OM, et al: Prognostic value of 1q21
amplification in multiple myeloma. Ter Arkh. 89:32–38. 2017.(In
Russian). View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Modesto AP, Usvyat L, Calice-Silva V,
Spigolon DN, Figueiredo AE, de Moraes TP, Olandoski M, Shimakura
SE, Barretti P, Kotanko P and Pecoits-Filho R: Impact of the
karnofsky performance status on survival and its dynamics during
the terminal year of peritoneal dialysis patients. Perit Dial Int:
pii. pdi.2015.00241. 2017. View Article : Google Scholar
|
|
13
|
Qian J, Jin J, Luo H, Jin C, Wang L, Qian
W and Meng H: Analysis of clinical characteristics and prognostic
factors of multiple myeloma: A retrospective single-center study of
787 cases. Hematology. 22:472–476. 2017.PubMed/NCBI
|
|
14
|
Alexanian R, Barlogie B and Dixon D: Renal
failure in multiple myeloma. Pathogenesis and prognostic
implications. Arch Intern Med. 150:1693–1695. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Blade´ J, Fernández-Llama P, Bosch F,
Montolíu J, Lens XM, Montoto S, Cases A, Darnell A, Rozman C and
Montserrat E: Renal failure in multiple myeloma: Presenting
features and predictors of outcome in 94 patients from a single
institution. Arch Intern Med. 158:1889–1893. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kyle RA, Gertz MA, Witzig TE, Lust JA,
Lacy MQ, Dispenzieri A, Fonseca R, Rajkumar SV, Offord JR, Larson
DR, et al: Review of 1027 patients with newlydiagnosed multiple
myeloma. Mayo Clin Proc. 78:21–33. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Eleutherakis-Papaiakovou V, Bamias A, Gika
D, Simeonidis A, Pouli A, Anagnostopoulos A, Michali E,
Economopoulos T, Zervas K and Dimopoulos MA: Greek Myeloma Study
Group: Renal failure in multiple myeloma: Incidence, correlations,
and prognostic significance. Leuk Lymphoma. 48:337–341. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Torra R, Bladé J, Cases A, López-Pedret J,
Montserrat E, Rozman C and Revert L: Patients with multiple myeloma
requiring long-term dialysis: Presenting features, response to
therapy, and outcome in a series of 20 cases. Br J Haematol.
91:854–859. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Eleftherakis-Papapiakovou E, Kastritis E,
Roussou M, Gkotzamanidou M, Grapsa I, Psimenou E, Nikitas N, Terpos
E and Dimopoulos MA: Renal impairment is not an independent adverse
prognostic factor in patients with multiple myeloma treated upfront
with novel agent-based regimens. Leuk Lymphoma. 52:2299–2303. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Dimopoulos MA, Roussou M, Gkotzamanidou M,
Nikitas N, Psimenou E, Mparmparoussi D, Matsouka C,
Spyropoulou-Vlachou M, Terpos E and Kastritis E: The role of novel
agents on the reversibility of renal impairment in newly diagnosed
symptomatic patients with multiple myeloma. Leukemia. 27:423–429.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chanan-Khan AA, San Miguel JF, Jagannath
S, Ludwig H and Dimopoulos MA: Novel therapeutic agents for the
management of patients with multiple myeloma and renal impairment.
Clin Cancer Res. 18:2145–2163. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gaballa MR, Laubach JP, Schlossman RL,
Redman K, Noonan K, Mitsiades CS, Ghobrial IM, Munshi N, Anderson
KC and Richardson PG: Management of myeloma-associated renal
dysfunction in the era of novel therapies. Expert Rev Hematol.
5:51–66; quiz 67–68. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Costa LJ, Abbas J, Ortiz-Cruz KL, Kang Y
and Stuart RK: Outcomes of patients with multiple myeloma and renal
impairment treated with bortezomib, cyclophosphamide, and
dexamethasone without plasma exchange. Eur J Haematol. 89:432–434.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mulkerin D, Remick S and Takimoto C:
Safety, tolerability and pharmacology of bortezomib in cancer
patients with renal failure requiring dialysis: Results from a
prospective phase 1 study. ASH Annu Meet Abstr. 110:34772007.
|
|
25
|
Jagannath S, Barlogie B, Berenson JR,
Singhal S, Alexanian R, Srkalovic G, Orlowski RZ, Richardson PG,
Anderson J, Nix D, et al: Bortezomib in recurrent and/or refractory
multiple myeloma. Initial clinical experience in patients with
impaired renal function. Cancer. 103:1195–1200. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
San-Miguel JF, Richardson PG, Sonneveld P,
Schuster MW, Irwin D, Stadtmauer EA, Facon T, Harousseau JL,
Ben-Yehuda D, Lonial S, et al: Efficacy and safety of bortezomib in
patients with renal impairment: Results from the APEX phase 3
study. Leukemia. 22:842–849. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chanan-Khan AA, Kaufman JL, Mehta J,
Richardson PG, Miller KC, Lonial S, Munshi NC, Schlossman R,
Tariman J and Singhal S: Activity and safety of bortezomib in
multiple myeloma patients with advanced renal failure: A
multicenter retrospective study. Blood. 109:2604–2606. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ying WZ, Wang PX, Aaron KJ, Basnayake K
and Sanders PW: Immunoglobulin light chains activate nuclear
factor-κB in renal epithelial cells through a Src-dependent
mechanism. Blood. 117:1301–1307. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sarközi R, Perco P, Hochegger K, Enrich J,
Wiesinger M, Pirklbauer M, Eder S, Rudnicki M, Rosenkranz AR, Mayer
B, et al: Bortezomib-induced survival signals and genes in human
proximal tubular cells. J Pharmacol Exp Ther. 327:645–656. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Uttamsingh V, Lu C, Miwa G and Gan LS:
Relative contributions of the five major human cytochromes P450,
1A2, 2C9, 2C19, 2D6, and 3A4, to the hepatic metabolism of the
proteasome inhibitor bortezomib. Drug Metab Dispos. 33:1723–1728.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Pekol T, Daniels JS, Labutti J, Parsons I,
Nix D, Baronas E, Hsieh F, Gan LS and Miwa G: Human metabolism of
the proteasome inhibitor bortezomib: Identification of circulating
metabolites. Drug Metab Dispos. 33:771–777. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Labutti J, Parsons I, Huang R, Miwa G, Gan
LS and Daniels JS: Oxidative deboronation of the peptide boronic
acid proteasome inhibitor bortezomib: Contributions from reactive
oxygen species in this novel cytochrome P450 reaction. Chem Res
Toxicol. 19:539–546. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dimopoulos MA, Richardson PG, Schlag R,
Khuageva NK, Shpilberg O, Kastritis E, Kropff M, Petrucci MT,
Delforge M, Alexeeva J, et al: VMP (Bortezomib, Melphalan, and
Prednisone) is active and well tolerated in newly diagnosed
patients with multiple myeloma with moderately impaired renal
function, and results in reversal of renal impairment: Cohort
analysis of the phase III VISTA study. J Clin Oncol. 27:6086–6093.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Morabito F, Gentile M, Mazzone C, Rossi D,
Di Raimondo F, Bringhen S, Ria R, Offidani M, Patriarca F, Nozzoli
C, et al: Safety and efficacy of
bortezomib-melphalan-prednisone-thalidomide followed by
bortezomib-thalidomide maintenance (VMPT-VT) versus
bortezomib-melphalan-prednisone (VMP) in untreated multiple myeloma
patients with renal impairment. Blood. 118:5759–5766. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ludwig H, Adam Z, Hajek R, Greil R,
Tóthová E, Keil F, Autzinger EM, Thaler J, Gisslinger H, Lang A, et
al: Light chain-induced acute renal failure can be reversed by
bortezomib-doxorubicin-dexamethasone in multiple myeloma: Results
of a phase II study. J Clin Oncol. 28:4635–4641. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Dimopoulos MA, Roussou M, Gavriatopoulou
M, Zagouri F, Migkou M, Matsouka C, Barbarousi D, Christoulas D,
Primenou E, Grapsa I, et al: Reversibility of renal impairment in
patients with multiple myeloma treated with bortezomib-based
regimens: Identification of predictive factors. Clin Lymphoma
Myeloma. 9:302–306. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Batuman V, Dreisbach AW and Cyran J:
Light-chain binding sites on renal brush-border membranes. Am J
Physiol. 258:F1259–F1265. 1990.PubMed/NCBI
|
|
38
|
Batuman V and Guan S: Receptor-mediated
endocytosis of immunoglobulin light chains by renal proximal tubule
cells. Am J Physiol. 272:F521–F530. 1997.PubMed/NCBI
|
|
39
|
Batuman V, Verroust PJ, Navar GL, Kaysen
JH, Goda FO, Campbell WC, Simon E, Pontillon F, Lyles M, Bruno J
and Hammond TG: Myeloma light chains are ligands for cubilin
(gp280). Am J Physiol. 275:F246–F254. 1998.PubMed/NCBI
|
|
40
|
Santostefano M, Zanchelli F, Zaccaria A,
Poletti G and Fusaroli M: The ultrastructural basis of renal
pathology in monoclonal gammopathies. J Nephrol. 18:659–675.
2005.PubMed/NCBI
|
|
41
|
Guan S, el-Dahr S, Dipp S and Batuman V:
Inhibition of Na-K-ATPase activity and gene expression by a myeloma
light chain in proximal tubule cells. J Investig Med. 47:496–501.
1999.PubMed/NCBI
|
|
42
|
Leung N, Gertz M, Kyle RA, Fervenza FC,
Irazabal MV, Eirin A, Kumar S, Cha SS, Rajkumar SV, Lacy MQ, et al:
Urinary albumin excretion patterns of patients with cast
nephropathy and other monoclonal gammopathy-related kidney
diseases. Clin J Am Soc Nephrol. 7:1964–1968. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Mayo MM and Johns GS: Serum free light
chains in the diagnosis and monitoring of patients with plasma cell
dyscrasias. Contrib Nephrol. 153:44–65. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Hutchison CA, Cockwell P, Harding S, Mead
GP, Bradwell AR and Barnett AH: Quantitative assessment of serum
and urinary polyclonal free light chains in patients with type II
diabetes: An early marker of diabetic kidney disease? Expert Opin
Ther Targets. 12:667–676. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Hutchison CA, Plant T, Drayson M, Cockwell
P, Kountouri M, Basnayake K, Harding S, Bradwell AR and Mead G:
Serum free light chain measurement aids the diagnosis of myeloma in
patients with severe renal failure. BMC Nephrol. 9:112008.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Durie BG, Harousseau JL, Miguel JS, Bladé
J, Barlogie B, Anderson K, Gertz M, Dimopoulos M, Westin J,
Sonneveld P, et al: International uniform response criteria for
multiple myeloma. Leukemia. 20:1467–1473. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Terpos E, Katodritou E, Tsiftsakis E, et
al: Cystatin-C is a sensitive marker of renal impairment with an
independent predictive value for survival in multiple myeloma;
reduction post bortezomib monotherapy. ASH Annu Meet Abstr.
110:14842007.
|
|
48
|
Dimopoulos MA, Christoulas D, Kastritis E,
et al: Tubular damage is ubiquitous in newly-diagnosed patients
with multiple myeloma: Comparison of three urinary and two serum
markers of kidney injury. ASH Annu Meet Abstr. 120:29192012.
|
|
49
|
Gupta N, Goh YT, Min CK, Lee JH, Kim K,
Wong RS, Chim CS, Hanley MJ, Yang H, Venkatakrishnan K, et al:
Pharmacokinetics and safety of ixazomib plus
lenalidomide-dexamethasone in Asian patients with
relapsed/refractory myeloma: A phase 1 study. J Hematol Oncol.
8:1032015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Rychak E, Mendy D, Shi T, Ning Y, Leisten
J, Lu L, Miller K, Narla RK, Orlowski RZ, Raymon HK, et al:
Pomalidomide in combination with dexamethasone results in
synergistic anti-tumour responses in pre-clinical models of
lenalidomide-resistant multiple myeloma. Br J Haematol.
172:889–901. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Weiss BM, Abadie J, Verma P, Howard RS and
Kuehl WM: A monoclonal precedes multiple myeloma in most patients.
Blood. 113:5418–5422. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Agarwal A and Ghobrial IM: Monoclonal
gammopathy of undetermined significance and smoldering multiple
myeloma: A review of the current understanding of epidemiology,
biology, risk stratification, and management of myeloma precursor
disease. Clin Cancer Res. 19:985–994. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Bahlis NJ: Darwinian evolution and tiding
clones in multiple myeloma. Blood. 120:927–928. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Magrangeas F, Avet-Loiseau H, Gouraud W,
Lodé L, Decaux O, Godmer P, Garderet L, Voillat L, Facon T, Stoppa
AM, et al: Minor clone provides a reservoir for relapse in multiple
myeloma. Leukemia. 27:473–481. 2013. View Article : Google Scholar : PubMed/NCBI
|