Introduction
Colorectal cancer (CRC) is the third most common
cancer and the third leading cause of cancer-related death in the
world (1). There has been remarkable
progress in molecular subtyping of CRC in the past decade and much
of this information has been used for clinical purposes. For
example, K-RAS mutation status can predict treatment response to
cetuximab. The latest National Comprehensive Cancer Network
clinical guideline recommends BRAF and mismatch repair genes to be
examined in routine tumor testing (2). However, cancer is known to be a
heterogeneous disease and thus identification and understanding of
additional prognostic factors would be helpful in guiding
treatments that would ultimately affect survival.
Long non-coding RNAs (lncRNAs) are defined as
transcripts longer than 200 nucleotides (nt) that are 5′ capped and
3′ polyadenylated. lncRNAs function in a wide range of biological
processes and can regulate gene expression in cis or trans by
diverse mechanisms. LncRNA-mediated biology has been implicated in
many cellular processes such as cell proliferation, invasion and so
on. One particularly important lncRNA is lncTCF7 which has been
reported to maintain cancer stem cell self-renewal throughing
Wnt/β-catenin signaling pathway (3).
In the present study we have examined lncTCF7
expression in tissue samples from CRC patients, assessed molecular
mechanisms responsible for its involvement in CRC tumorigenesis,
and assessed its potential value as a biomarker in predicting CRC
prognosis.
Materials and methods
Tissue samples
The CRC surgical samples were obtained from part of
surgical patients in the Department of Surgery at Peking University
First Hospital between August 2011 and December 2012. These were
confirmed to be colorectal adenocarcinoma histologically by
experienced pathologists. Clinicopathologic staging was determined
according to the CRC Guideline of the National Comprehensive Cancer
Network (2). Written informed
consents were obtained from these patients.
Cell culture
The cell lines DLD1 and LoVo were purchased from the
American Type Culture Collection. DLD1 and LoVo were cultured in
DMEM and F-12K (both Gibco; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) culture media, respectively, at 37°C with 5%
CO2. The media were supplemented with 10% fetal bovine
serum (FBS; Gibco), 100 U/ml of penicillin, and 100 µg/ml of
streptomycin (both Keygen Biotech, Nanjing, China).
Cell transfection
The plasmid expressing short hairpin RNA (shRNA)
specifically targeting the lncTCF7 (shRNA-lncTCF7) and its
scrambled RNA (shRNA-nc) control were constructed by GenePharma Co.
(Shanghai, China). The shRNA sequence is
5′-CTTTGGATAAGATCTACGTTT-3′. The plasmid over-expressing lncTCF7
(oe-lncTCF7) and its empty vector (oe-Vec) were obtained from
GenePharma Co. Lipofectamine 3000 (Invitrogen, Carlsbad, CA, USA)
was used for the transfection. Cell clones stably expressing the
shRNA and oe-lncTCF7 were selected with G418.
RNA extraction and qRT-PCR
Total RNA was extracted from the surgical samples
and cultured cells using TRIzol reagent (Takara Bio, Dalian,
China). RQ1 RNase-Free DNase (Promega, Madison, WI, USA) was used
to remove DNA. RNA reverse transcription to cDNA was performed with
RevertAid First Strand cDNA Synthesis kit (Thermo Fisher
Scientific, Inc.) and quantitative real-time PCR (qRT-PCR) analysis
was performed with SYBR-Green PCR Master Mix (Life) according to
the manufacturer's instructions. The results were normalized to the
expression of 18S and β-actin. The primers used for qRT-PCR are
listed below: lncTCF7 Forward, AGGAGTCCTTGGACCTGAGC and Reverse,
AGTGGCTGGCATATAACC-AACA; 18S Forward, GTAACCCGTTGAACCCCATT and
Reverse, CCATCCAATCG-GTAGTAGCG; β-actin Forward,
CATGTACGTTGCTATCCAGGC and Reverse, CTCC-TTAATGTCACGCACGAT. The ΔCq
value was used to represent the expression level of lncRNA in
quantitative qRT-PCR. For each amplicon designed, the ΔCq value was
normalized using the equation: ΔCq=Cq (target)-Cq
(18S/β-actin).
Western blot analysis
Lysate was extracted from surgical samples or
cultured cells by a lysis buffer, and the protein content was
assayed by a protein assay (BCA) kit (Thermo Fisher Scientific,
Inc.). Protein was fractionated in 10% SDS-PAGE and transferred
onto a PVDF membrane (Roche, Mannheim, Germany). Rabbit monoclonal
antibodies against E-cadherin, N-cadherin, Vimentin, Slug, c-Myc,
Cyclin D1 (Cell Signaling Technology, Inc., Danvers, MA, USA), MMP7
(Abcam, Cambridge, UK) and TCF7 (Abgent Inc., San Diego, CA, USA)
were used as the primary antibodies. The antibody against GAPDH
(Cell Signaling Technology, Inc.) was used for loading control.
Secondary antibody was then added and proteins of interest were
visualized by using Pierce ECL Western Blotting Substrate (Thermo
Fisher Scientific, Inc.).
Cell proliferation
The effect of lncTCF7 on proliferation of CRC cell
lines was measured with Cell Counting kit-8 (CCK-8) (Dojindo).
After transfection of shRNA-lncTCF7, shRNA-nc, oe-lncTCF7, or
oe-Vec for 48 h, DLD1 and LoVo cells were re-suspended, seeded at
2×103/100 µl/well in a 96-well plate, and incubated for
24 or 48 h. CCK-8 reagent was added to each well. The plate was
incubated for another 2 h at 37°C, and the absorbance at 450 nm was
measured.
Transwell assays
The migration ability of CRC cells was assayed by a
Transwell plate with a filter of 8 µm pores (BD Biosciences, San
Diego, CA, USA). A total of 5×105 cells in serum-free
medium were added into the upper insert. A total of 600 µl of
medium containing 20% FBS was added into the lower compartment.
After incubation for 48 h, the cells on the upper surface of the
filter were wiped, and the cells adherent on the undersurface of
the filter were fixed in methanol, stained with crystal violet and
counted with a microscope. Three visual fields were selected
randomly for cell counting. An invasion assay was performed in a
similar manner except that the top chamber was pre-coated with 50
µl Matrigel (BD Biosciences).
RNA-FISH
A fragment of 550 bp lncTCF7 cDNA was amplified from
the lncTCF7 plasmid by using a high fidelity DNA polymerase
(Takara) as the DNA template. From this template, a fluorescein
labeled DNA as the lncTCF7 FISH probe was made with
fluorescein-12-dUTP (Roche) and Klenow DNA polymerase following the
manufacturer's protocol. CRC tissue with its adjacent normal tissue
embedded in OCT (Sakura Finetek, Inc., Torrance, CA, USA) was cut
to sections of 4 µm thickness. The slide was soaked in proteinase K
for 5 min and washed in 2× SSC twice. A FISH hybridization solution
(Beijing Dingguo Changsheng Biotechnology Co., Ltd., Beijing,
China) containing 30 ng/µl lncTCF7 FISH probe DNA was applied to
the slide which was subsequently incubated at 37°C for 16 h. The
slide was then washed in 0.4× SSC/0.001% NP-40 at 560C for 5 min
followed by a second wash in 2× SSC/0.0001% NP-40 for another 2
min. The slide was covered with a drop of mounting medium
containing DAPI, and observed under a fluorescence microscope.
Tumor xenograft model
Male BALB/c nude mice of 4–5 weeks age were
purchased from Beijing Vital River Co. (Beijing, China) and
maintained in the Experimental Animal Center of Peking University
First Hospital. DLD1 cells (1×107 cells) stably
expressing shRNA-lncTCF7, or oe-lncTCF7, or respective control
vectors were mixed with 50% Matrigel (BD Biosciences), and were
injected subcutaneously into the right flank of a nude mouse
(n=6/group). Tumor volume was measured every 2 days and calculated
as width2 x length/2. After two weeks, the mice were
sacrificed, the tumors were removed and weight was measured.
Statistical analysis
Two-tailed Student's t-test was used for data
comparison. χ2 test was performed to evaluate the
association between lncTCF7 level and clinicopathological
parameters. Kaplan-Meier method was used to estimate overall
survival of the CRC patients. P<0.05was considered to indicate a
statistically significant difference. Each experiment was repeated
at least 3 times independently.
Results
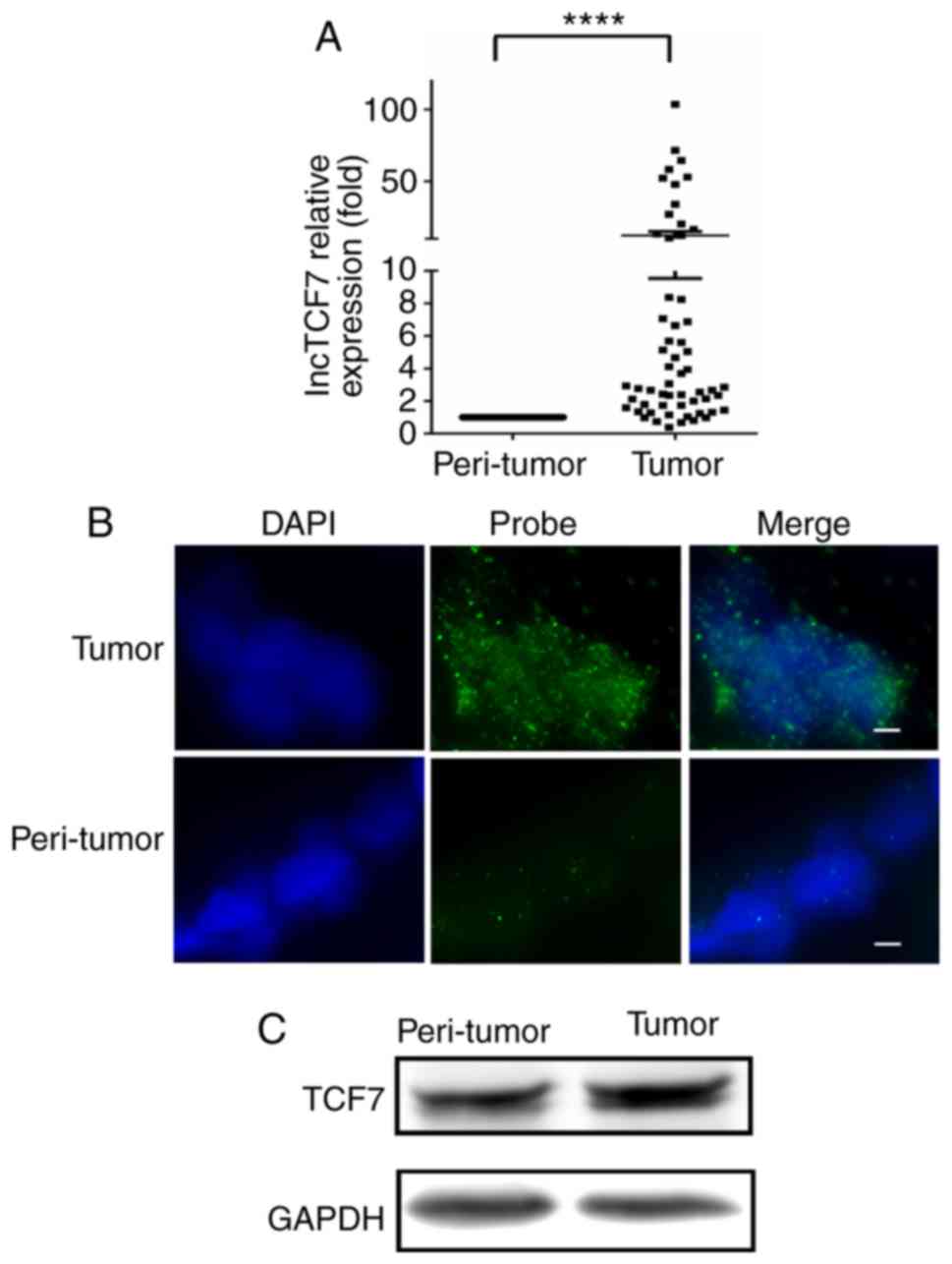
lncTCF7 is upregulated in CRC
tissues
lncRNAs have been reported to play a role in
tumorigenesis and progression and aberrant expression of lncRNAs
has been associated with worse cancer prognosis. We found that
lncTCF7 was significantly increased in cancer tissue in comparison
with peri-tumor control.
In 58 CRC cases, 52 cases showed a higher lncTCF7
expression levels in tumor than in peri-tumor mucosa and the median
ratio increased 3-fold (Fig. 1A).
Using RNA-FISH, we examined the location of lncTCF7 in two pairs of
specimens as shown in Fig. 1B, more
abundant lncTCF7 RNA was seen in cancer tissue compared with
peri-tumor mucosa and it was specifically localized in the nuclei
of CRC specimens. In Fig. 1C,
increased TCF7 protein expression was also noted by western blot in
ten pairs of specimens.
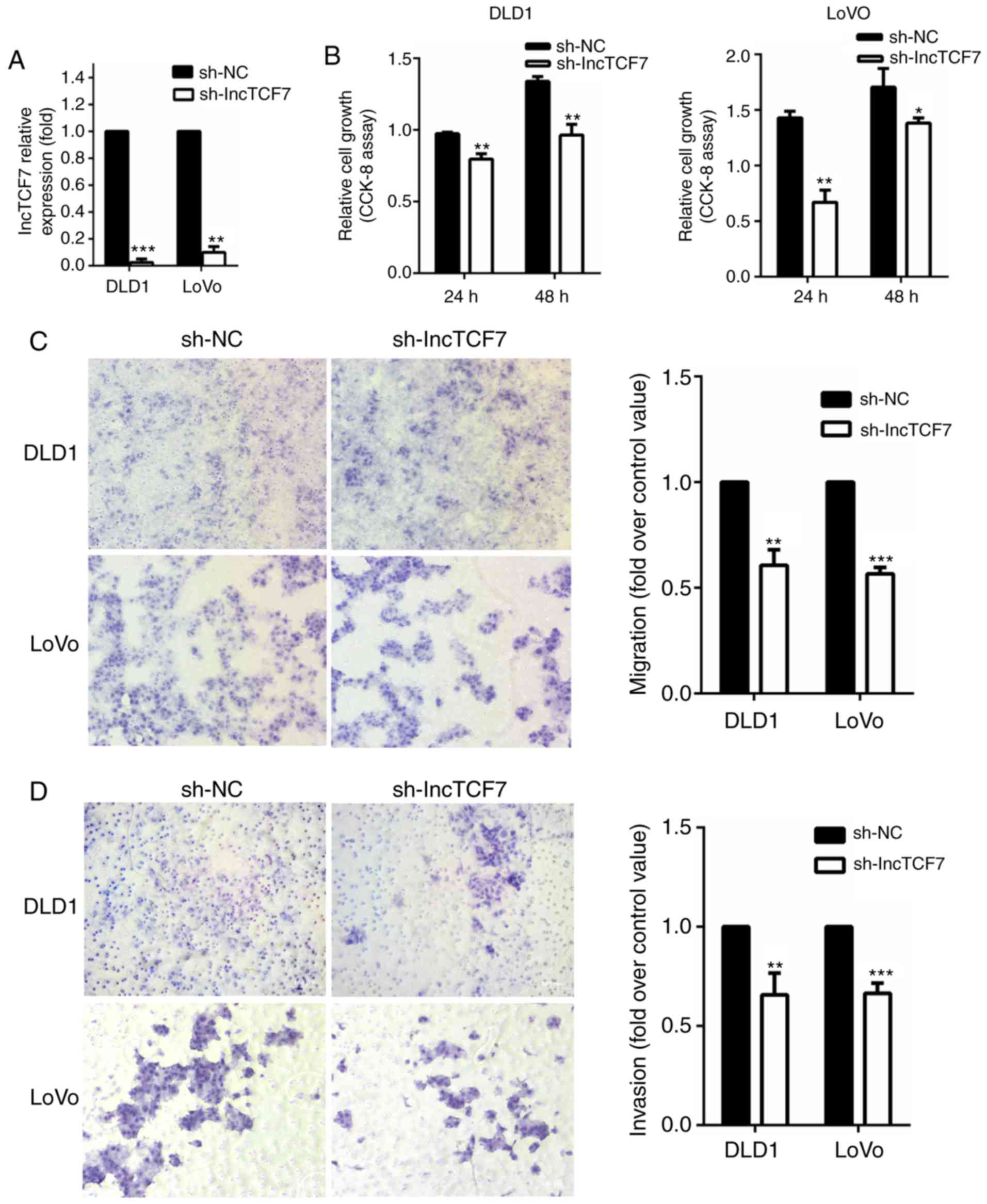
lncTCF7 stimulates CRC cell migration
and invasion in vitro
The DLD1 and LoVo cells transfected with
shRNA-lncTCF7 had reduced expression of lncTCF7 compared to cells
transfected with shRNA-nc as measured by qRT-PCR (Fig. 2A). As shown in Fig. 2B, suppression of lncTCF7 led to a mild
decrease in cell growth. Cell migration was assessed by Transwell
assay (Fig. 2C). In both DLD-1 and
Lovo cell lines, there was a 30–40% decrease in migrated cell
numbers in shRNA-lncTCF7 group compared to that in control. In
addition, reduced expression of lncTCF7 also inhibited invasion by
25~30% (Fig. 2D).
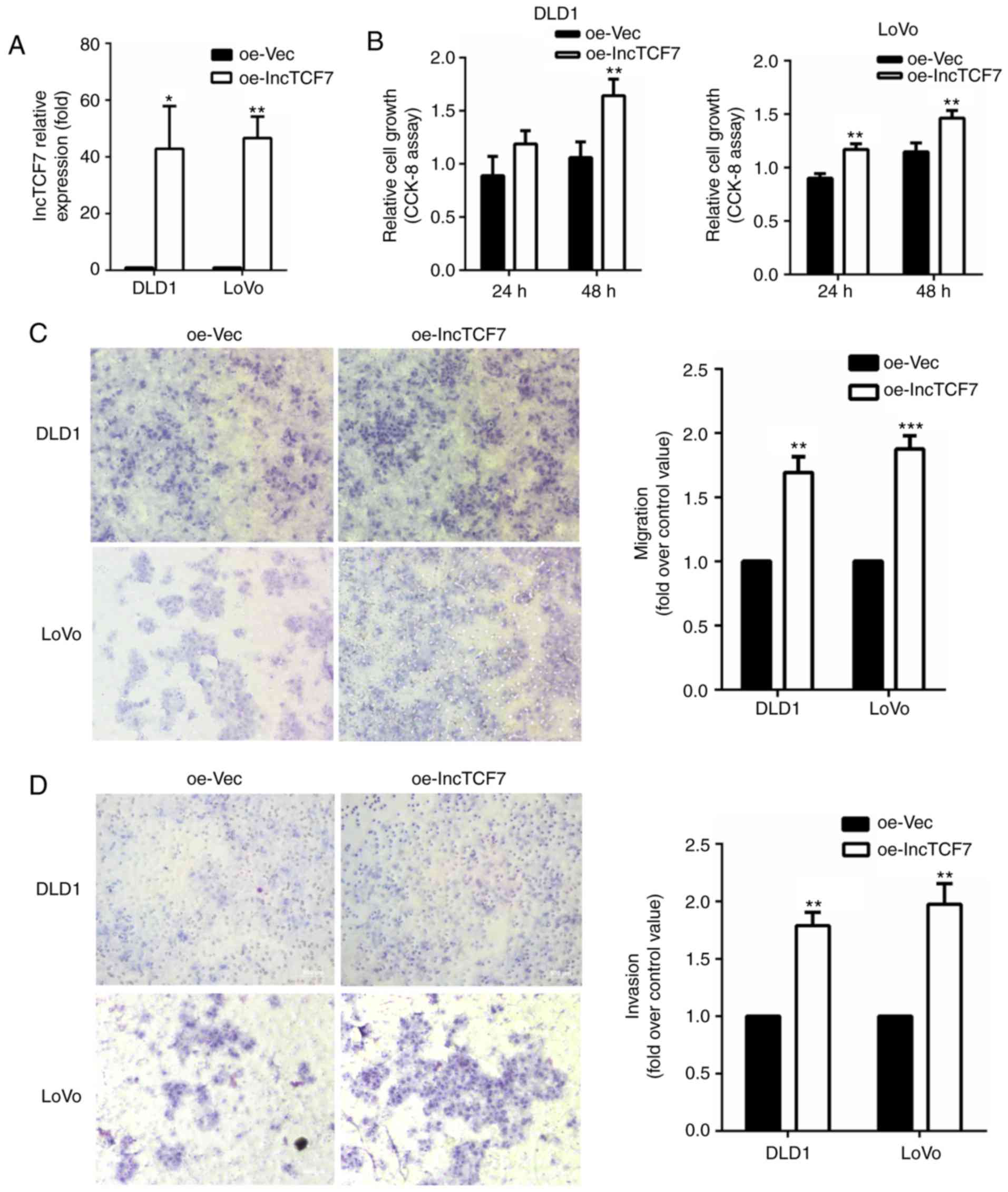
We also measured cell proliferation and migration
after overexpressing lncTCF7 in DLD-1 and Lovo lines (Fig. 3A). As shown in Fig. 3B and C, increased lncTCF7 stimulated
CRC cell proliferation and migration significantly. Fig. 3D showed that overexpression of lncTCF7
increased cell invasion rate by 80~100%.
Taken together, these in vitro experiments
support the hypothesis that lncTCF7 promotes tumor cell migration
and invasion.
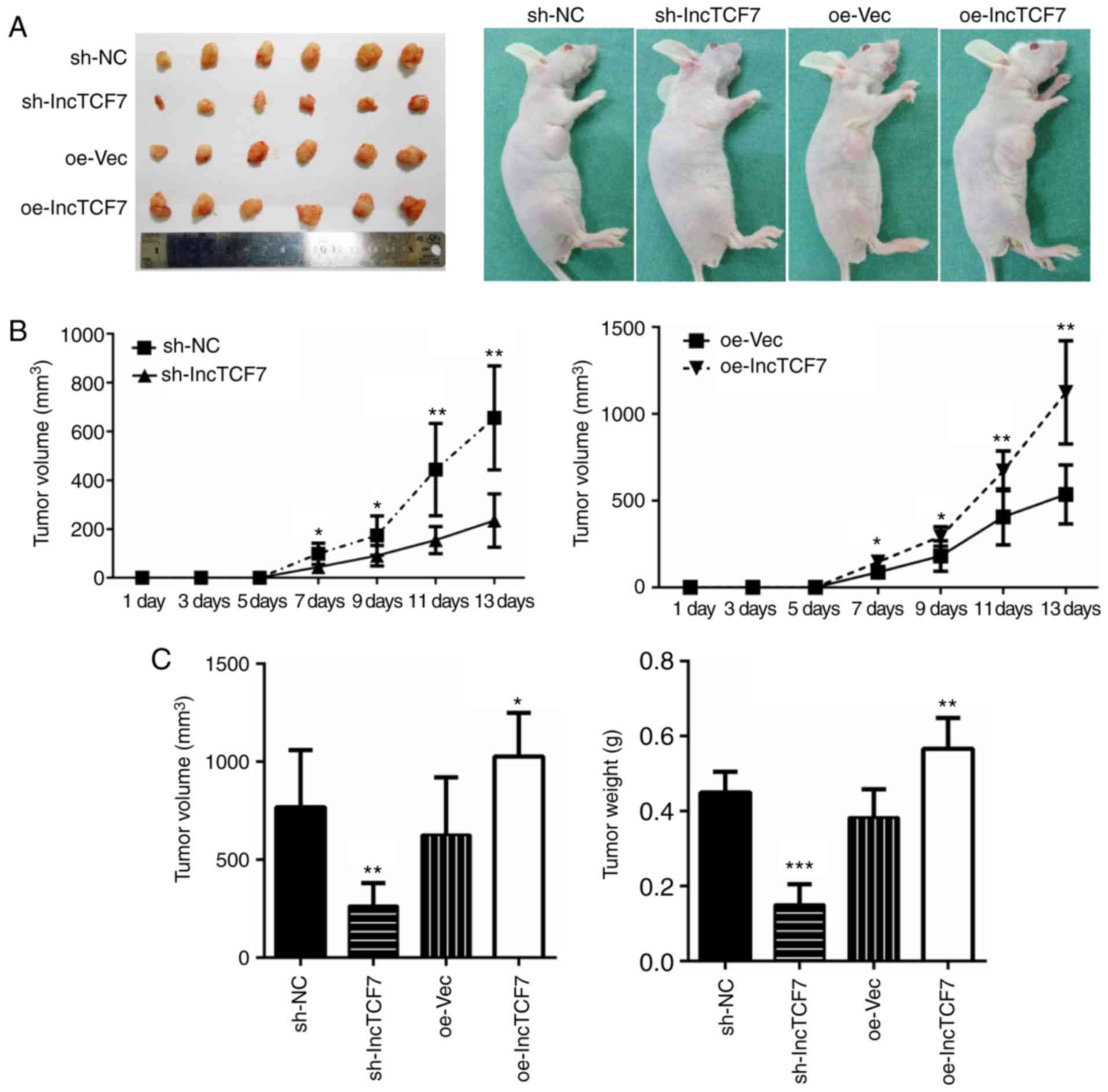
LncTCF7 promotes tumorigenesis in
vivo
Next we detected how lncTCF7 influences
tumorigenesis in vivo. 1×107 cells were
inoculated into nude mice and tumor growth was monitored for 14
days (Fig. 4A). At the sixth day, a
palpable tumor could be recognized. As shown in Fig. 4, reduced expression of lncTCF7 in the
shRNA-lncTCF7 group led to significantly smaller tumor volume and
weight (Fig. 4A, C, left), and a
slower tumor growth (Fig. 4B, left).
On the other hand, overexpression of lncTCF7 with oe-lncTCF7
resulted in faster growth (Fig. 4B,
right), and bigger tumor size (Fig
4C). These data indicate that lncTCF7 promotes CRC
tumorigenesis in vivo.
LncTCF7 regulates
epithelial-mesenchymal transition by activating the Wnt
pathway
Given the finding that lncTCF7 promotes CRC cell
migration and invasion, we attempted to investigate the underlying
molecular mechanisms. Epithelial-mesenchymal transition, EMT, is
pivotal in cancer metastasis and invasion (4) and the Wnt signaling pathway is one of
the key pathways in regulating EMT (5). Thus, we set out to examine if lncTCF7
affects the Wnt signal pathway.
First, the epithelial and mesenchymal markers were
assayed by western blot analysis. Fig.
5A shows that lncTCF7 inhibited the expression of E-cadherin,
an epithelial marker, but enhanced the expression of N-cadherin,
Vimentin and slug, three classic mesenchymal markers, in DLD1 and
LoVo cells.
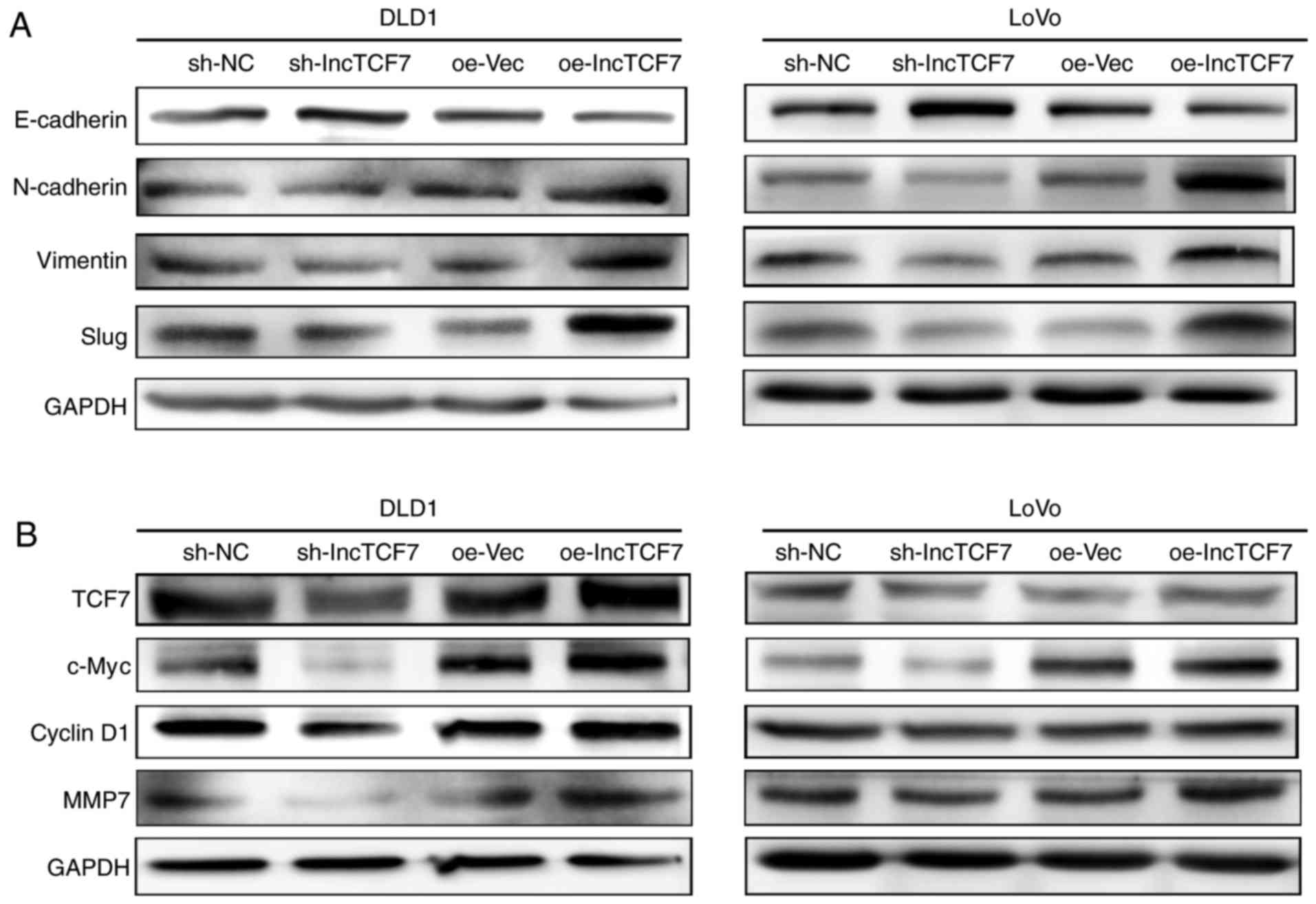 | Figure 5.LncTCF7 initiate EMT by activating
Wnt/β-catenin signaling pathway. DLD1 and LoVo cell lines were
transfected with lncTCF7, shRNA-nc, oe-LncTCF7, or oe-Vec (A) The
expression of E-cadherin, N-cadherin, vimentin and slug in DLD1 and
LoVo cells were detected by western blot. GAPDH was used as an
internal control. (B) The expression of TCF7, c-Myc, CyclinD1, and
MMP7 in DLD1 and LoVo cells were detected by western blot. GAPDH
was used as an internal control. shRNA, short hairpin RNA; lncTCF7,
long non-coding RNA TCF7; oe-lncTCF7, over-expressing lncTCF7. |
Next, we assessed the state of the Wnt pathway upon
lncTCF7 manipulation by measuring several downstream effects. As
shown in Fig. 5B, reduced expression
of lncTCF7 resulted in reduced expression of c-Myc, Cyclin D1 and
MMP7 while overexpressing lncTCF7 demonstrated opposite
effects.
LncTCF7 correlates with poor prognosis
of CRC patients
To test the value of lncTCF7 as a prognostic
biomarker, we studied the association between lncTCF7 expression
and clinical profiles of CRC patients. Demographic and clinical
data were reviewed (Table I). A total
of 58 cases were categorized according to LncRNA expression level.
Group of lncTCF7 was divided according to median expression fold
compared to its peri-tumor. As shown in Table I, there were significant differences
in tumor size, lymph node metastasis, and TNM stage between two
groups.
 | Table I.Clinicopathologic characteristics of
lncTCF7 expression in CRC patients. |
Table I.
Clinicopathologic characteristics of
lncTCF7 expression in CRC patients.
| Clinicopathological
variables | N | High
expressiona | Low
expressiona | χ2 | P-value |
|---|
| Sex |
|
|
|
|
|
| Male | 28 | 14 | 14 | 0 | 1 |
|
Female | 30 | 15 | 15 |
|
|
| Age
(years)a |
|
|
|
|
|
|
<64 | 29 | 12 | 17 | 1.724 | 0.189 |
| ≥64 | 29 | 17 | 12 |
|
|
| Tumor
sizea (cm) |
|
|
|
|
|
|
<4.5 | 25 | 8 | 17 | 5.695 | 0.017 |
| ≥4.5 | 33 | 21 | 12 |
|
|
| Lymph metastasis |
|
|
|
|
|
| Yes | 27 | 18 | 9 | 5.613 | 0.018 |
| No | 31 | 11 | 20 |
|
|
| TNM
classification |
|
|
|
|
|
|
I–II | 27 | 8 | 19 | 8.385 | 0.004 |
|
III–IV | 31 | 21 | 10 |
|
|
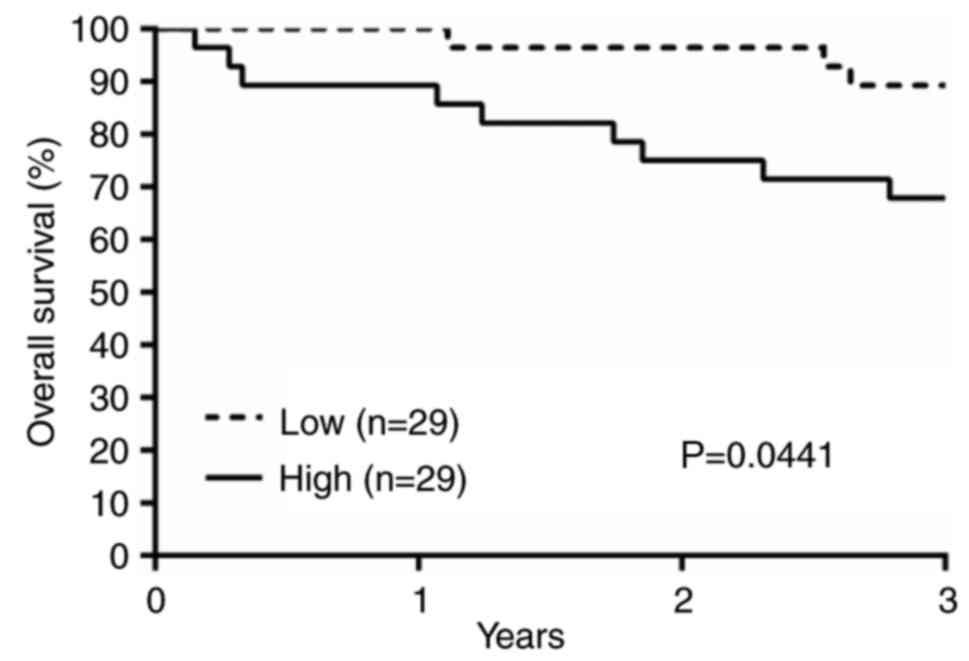
Furthermore, Kaplan-Meier survival analysis revealed
worse prognosis in the high lncTCF7 group. The overall 3-year
survival rate was about 68% in the high group, and 89% in the low
group (Fig 6). The median follow-up
time is 47 months. Log rank test confirmed that the difference is
statistically significant (P=0.0441 with a hazard ratio of 3.513,
95% confidence interval 1.032 to 9.996).
Disscusion
LncRNAs contribute not only to homeostasis, but also
to pathologic processes via regulating expression of many genes.
LncRNAs regulate gene expression by influencing chromatin
remodeling, transcriptional control, post-transcriptional
processing, and protein metabolism (6). It has been thought that lncRNA
dysregulation is associated with tumorigenesis and their aberrant
expression has important pathogenic consequences such as initiating
metastasis. It is known that high expression of HOTAIR, H19,
SAMMSON, AFAP-AS1, UPAT (7–11) correlate with a worse cancer prognosis.
High expression of lncRNAs including MALAT1, HOTAIR, CCAL, 91H
(12–15) were reported to correlate with a poor
prognosis in CRC patients while lower lncRNA Loc285194 expression
was associated with a better survival (16). These findings suggested that lncRNAs
may serve as diagnostic or prognostic markers and could potentially
be useful as treatment targets.
LncTCF7 expression is associated with cancer stem
cell self-renewal (3). However, the
function of lncTCF7 in CRC remains largely unknown. Our study
investigated lncTCF7 expression in surgical samples from CRC
patients and found that lncTCF7 level increased on average about 10
times in CRC tissue. In addition, lncTCF7 promoted cell
proliferation, migration and invasion in vitro and
tumorigenesis in nude mice inoculation experiments.
EMT is a process which closely relates with cancer
invasion and metastasis (17,18). There are several signal pathways
regulating EMT (19,20). One of them is Wnt/β-catenin signaling
cascade (17). Interestingly, lncTCF7
promoted expression of the mesenchymal markers N-cadherin, vimentin
and slug, and suppressed expression of the epithelial marker
E-cadherin. Given the close link between lncTCF7 and Wnt pathway,
we therefore hypothesized that lncTCF7 activated Wnt/β-catenin
signaling and subsequently regulated EMT and resulted in
tumorigenesis.
TCF7 is one of TCF/LEF members which plays an
essential role in Wnt/β-catenin pathway (21–23). It
has been known that lncTCF7 recruits the SWI/SNF complex to the
promoter of TCF7 to facilite its expression (3). We found that lncTCF7 is enriched in the
CRC tissues nucleus. Our research demonstrated that lncTCF7
promotes the expression of TCF7. Therefore, lncTCF7 increased
expression of TCF7 to promote the activation Wnt signal pathway.
The activation of Wnt/β-catenin cascade enhanced expression of
downstream members. In this study, lncTCF7 upregulated c-Myc,
Cyclin D1 and MMP7 levels in CRC cells.
Lastly, lncTCF7 expression is significantly elevated
in CRC samples compared to normal colon epithelium. lncTCF7 level
affects TNM staging and survival and CRC patients with high lncTCF7
levels have worse prognosis. This study thus supports the value of
lncTCF7 as a CRC prognostic marker.
Acknowledgments
The present study is supported by the grant on
potential therapeutic approaches for surgical infection and the
underlying mechanisms from China Health and Medical Development
Foundation. The authors would like to thank Professor Ding-fang Bu
and Qiang Zhu for their technical assistance for RNA-FISH assay,
and Mrs. Yuanyuan Ma for the animal study assistance.
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
National Comprehensive Cancer Network:
NCCN clinical practice guidelines in oncology. https://www.nccn.org/professionals/physician_gls/f_guidelines.asp
|
|
3
|
Wang Y, He L, Du Y, Zhu P, Huang G, Luo J,
Yan X, Ye B, Li C, Xia P, et al: The long noncoding RNA lncTCF7
promotes self-renewal of human liver cancer stem cells through
activation of wnt signaling. Cell Stem Cell. 16:413–425. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Liu CC, Cai DL, Sun F, Wu ZH, Yue B, Zhao
SL, Wu XS, Zhang M, Zhu XW, Peng ZH and Yan DW: FERMT1 mediates
epithelial-mesenchymal transition to promote colon cancer
metastasis via modulation of β-catenin transcriptional activity.
Oncogene. 36:1779–1792. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mondal T and Kanduri C: Noncoding RNA
scaffolds in pluripotency. Circ Res. 110:1162–1165. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gupta RA, Shah N, Wang KC, Kim J, Horlings
HM, Wong DJ, Tsai MC, Hung T, Argani P, Rinn JL, et al: Long
non-coding RNA HOTAIR reprograms chromatin state to promote cancer
metastasis. Nature. 464:1071–1076. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Han D, Gao X, Wang M, Qiao Y, Xu Y, Yang
J, Dong N, He J, Sun Q, Lv G, et al: Long noncoding RNA H19
indicates a poor prognosis of colorectal cancer and promotes tumor
growth by recruiting and binding to eIF4A3. Oncotarget.
7:22159–22173. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Leucci E, Vendramin R, Spinazzi M,
Laurette P, Fiers M, Wouters J, Radaelli E, Eyckerman S, Leonelli
C, Vanderheyden K, et al: Melanoma addiction to the long non-coding
RNA SAMMSON. Nature. 531:518–522. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang F, Ni H, Sun F, Li M and Chen L:
Overexpression of lncRNA AFAP1-AS1 correlates with poor prognosis
and promotes tumorigenesis in colorectal cancer. Biomed
Pharmacother. 81:152–159. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Taniue K, Kurimoto A, Sugimasa H, Nasu E,
Takeda Y, Iwasaki K, Nagashima T, Okada-Hatakeyama M, Oyama M,
Kozuka-Hata H, et al: Long noncoding RNA UPAT promotes colon
tumorigenesis by inhibiting degradation of UHRF1. Proc Natl Acad
Sci USA. 113:pp. 1273–1278. 2016, View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ji Q, Zhang L, Liu X, Zhou L, Wang W, Han
Z, Sui H, Tang Y, Wang Y, Liu N, et al: Long non-coding RNA MALAT1
promotes tumour growth and metastasis in colorectal cancer through
binding to SFPQ and releasing oncogene PTBP2 from SFPQ/PTBP2
complex. Br J Cancer. 111:736–748. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Svoboda M, Slyskova J, Schneiderova M,
Makovicky P, Bielik L, Levy M, Lipska L, Hemmelova B, Kala Z,
Protivankova M, et al: HOTAIR long non-coding RNA is a negative
prognostic factor not only in primary tumors, but also in the blood
of colorectal cancer patients. Carcinogenesis. 35:1510–1515. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ma Y, Yang Y, Wang F, Moyer MP, Wei Q,
Zhang P, Yang Z, Liu W, Zhang H, Chen N, et al: Long non-coding RNA
CCAL regulates colorectal cancer progression by activating
Wnt/β-catenin signalling pathway via suppression of activator
protein 2α. Gut. 65:1494–1504. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Deng Q, He B, Gao T, Pan Y, Sun H, Xu Y,
Li R, Ying H, Wang F, Liu X, et al: Up-regulation of 91H promotes
tumor metastasis and predicts poor prognosis for patients with
colorectal cancer. PLoS One. 9:e1030222014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Qi P, Xu MD, Ni SJ, Huang D, Wei P, Tan C,
Zhou XY and Du X: Low expression of LOC285194 is associated with
poor prognosis in colorectal cancer. J Transl Med. 11:1222013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Liu ZJ, Liu HL, Zhou HC and Wang GC: TIPE2
inhibits hypoxia-induced Wnt/β-catenin pathway activation and EMT
in glioma cells. Oncol Res. 24:255–261. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Jin B, Wang W, Meng XX, Du G, Li J, Zhang
SZ, Zhou BH and Fu ZH: Let-7 inhibits self-renewal of
hepatocellular cancer stem-like cells through regulating the
epithelial-mesenchymal transition and the Wnt signaling pathway.
BMC Cancer. 16:8632016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Nieto MA, Huang RY, Jackson RA and Thiery
JP: EMT: 2016. Cell. 166:21–45. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sun X, Deng Q, Liang Z, Liu Z, Geng H,
Zhao L, Zhou Q, Liu J, Ma J, Wang D, et al: Cigarette smoke extract
induces epithelial-mesenchymal transition of human bladder cancer
T24 cells through activation of ERK1/2 pathway. Biomed
Pharmacother. 86:457–465. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cho YH, Cha PH, Kaduwal S, Park JC, Lee
SK, Yoon JS, Shin W, Kim H, Ro EJ, Koo KH, et al: KY1022, a small
molecule destabilizing Ras via targeting the Wnt/β-catenin pathway,
inhibits development of metastatic colorectal cancer. Oncotarget.
7:81727–81740. 2016.PubMed/NCBI
|
|
22
|
Cui L, Guan Y, Qu Z, Zhang J, Liao B, Ma
B, Qian J, Li D, Li W, Xu GT and Jin Y: WNT signaling determines
tumorigenicity and function of ESC-derived retinal progenitors. J
Clin Invest. 123:1647–1661. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhu Y, Wang W and Wang X: Roles of
transcriptional factor 7 in production of inflammatory factors for
lung diseases. J Transl Med. 13:2732015. View Article : Google Scholar : PubMed/NCBI
|