Introduction
Sarcoidosis is a chronic, inflammatory, systemic
disease affecting primarily the lungs, the mediastinum and the
lymphatic system but also salivary glands, heart, nervous system,
joints and various other organs (1).
Diagnosis depends on the existence of typical clinicoradiological
findings in association with noncaseating epitheloid cell
granulomas in biopsy and the absence of known, alternative or local
causes provoking granulomas (1).
Granulomas are nonspecific inflammatory lesions and can occur
during mycobacterial, fungal or parasitic infections as well as
other diseases like Wegener's granulomatosis (2). Due to the differentiation of granulomas
the pathologists play a pivotal role in finding the correct
diagnosis (2). In sarcoidosis,
granuloma formation is characterized by infiltrating Th1 helper
cells and macrophages. The latter show a transformation process
into epitheloid cells and can fuse into multinucleate giant cells.
Although small amounts of necrosis can be observed, the sarcoid
granuloma is referred to the group of nonnecrotizing or
noncaseating granulomas (2).
Caseating granulomas are typically found in infectious diseases
like syphilis or tularemia or infection with tuberculous and
nontuberculous mycobacterium (2). In
some, especially oncologic patients treated with immunotherapy,
noncaseating granulomas can be found although they do not fulfill
the criteria for systemic sarcoidosis and are thus referred to as
sarcoid-like reactions (3). Due to
the toxicity profile of immunotherapies immune-related adverse
events can provoke those sarcoid-like reactions which may occur in
the organ of tumor origin or in the tumor-draining lymph nodes
(3). Sarcoidosis is associated with
an increased risk for cancer development in several organs like
lung, liver, stomach or for melanoma and lymphoma. Sarcoid-like
reactions can be found in 13.8% of patients with Hodgkin-disease,
7.3% with non Hodgkin lymphoma and 4.4% of cases with carcinomas
(4,5).
Furthermore, simultaneous occurrence of sarcoidosis and cancer is
associated with a diminished survival rate (6). Although no increased risk for malignancy
in the head and neck has been described so far, there are a few
cases that report the simultaneous occurrence of sarcoidosis and
malignoma of the head and neck (7–9). Notably,
in follow-up computed tomography (CT) scans or those done for the
detection of primary tumors or metastatic lesions (e.g.,
18-Fluorodeoxyglucose positron emission tomography-(PET)-CT,
18FDG-PET-CT) sarcoidosis can mimic cancer recurrence or
metastatic progress (10,11). However, it is possible that metastatic
lesions coexist next to lymph nodes with sarcoid-like lesions and
it is unclear whether sarcoidosis has an influence on metastasis of
malignoma. Therefore, a review of the current literature was
performed to analyze the residual risk of metastasis within
radiological suspicious lesions in patients with a history of solid
tumors and sarcoidosis.
Methods
In this review we analyzed reported cases of
patients with solid tumors whose staging or follow-up analysis
revealed an unclear lymphadenopathy owing to metastasis or
sarcoidosis. A systematic literature search was done in Pub Med
data base (from inception to April 2017) without any limitation
using the terms: Sarcoidosis [title] AND metastasis. All cases with
a solid tumor and sarcoidosis were included and the provided
information concerning age, gender, tumor region, tumor entity,
tumor classification, therapy, as well as information about
diagnosis of sarcoidosis were collected. Analyzing the data, the
risk of simultaneous occurrence of metastasis and sarcoidosis in
concordance to positive radiological and histological findings was
elucidated.
Ethical approval
All procedures performed in studies involving human
participants were in accordance with the ethical standards of the
institutional research committee and with the 1964 Helsinki
declaration and its later amendments or comparable ethical
standards. Informed consent: Informed consent was obtained from all
individual participants included in the study. This article does
not contain any studies with animals performed by any of the
authors. For this retrospective study and review of anonymized
clinical records an ethical permission was not required.
Results
Review of the literature
Review of the literature revealed 115 cases in Pub
Med Data Base based on the search criteria mentioned above. Cases
without malignancy were excluded and 59 cases with cancer and
sarcoidosis were identified and are listed in Table I. The median age at cancer diagnosis
was 49.5 +/- 13.4 years and cases of 31 female and 23 male patients
were reported. In 5 cases, the sex of the patient was not
documented. Systemic sarcoidosis treatment consisted of oral
steroids (n=12) or chloroquine (n=1). Local steroids were applied
in one case with uveitis. No therapy was initiated in 15 cases and
30 cases did not provide any information about the sarcoidosis
therapy approach. However, independent from the treatment strategy
the patients' outcome was described to be good with a stable status
or complete remission. The most frequent cancer origin was breast
(n=12) followed by malignoma of the thyroid gland (n=8).
Sarcoidosis occurred in 20 cases after an average of 34.4 months
and in 7 cases 10.25 years before diagnosis of malignoma. In most
reported cases (n=24) sarcoidosis was revealed simultaneously with
diagnosis of malignoma. In 25 cases, surgery was performed to
remove the tumor. 24 patients had a combination of surgery and
radio-, chemo- or radiochemotherapy. Two cases were only treated by
radiochemotherapy and in seven cases, no information about
therapeutic aspects concerning the tumor was provided. Occurrence
of sarcoidosis after surgery was reported in 3/25 cases and in
17/24 cases after surgery and radio-/chemotherapy. In 17% of the
cases, simultaneous detection of sarcoidosis or sarcoid-like
lesions and metastasis was reported (n=10). These reports are now
described in more detail.
 | Table I.Characteristics of patients from
literature review. |
Table I.
Characteristics of patients from
literature review.
|
| Tumor
characteristics | Patient | Sarcoidosis |
|
|---|
|
|
|
|
|
|
|---|
| Author, year | Tumor region | Tumor entity | TNM
classification | Therapy | Age | Sex | Diagnoses tool | Biopsy | Sarcoidosis
manifestations | Time between
Diagnostic (months) | (Refs.) |
|---|
| Altinkaya et
al, 2015 | Breast | Ductal invasive
carcinoma | T1N0M0 | OP | 70 | F | PET | EBUS | LN mediastinal | 0 | (31) |
| Conte et al,
2015 | Breast | Ductal invasive
carcinoma | pT3pN1 | OP | 50 | F | PET | OP+Biopsy | LN pelvic,
supradiaphragmatic | 0 | (12) |
| Zivin et al,
2014 | Breast | Ductal invasive
carcinoma | nk | OP | 32 | F | PET | Biopsy+EBUS | LN hilar +
mediastinal | nk | (32) |
| El Hammoumi et
al, 2015 | Breast | Lobular
carcinoma | T1N2Mx | OP + aRCTx
(Tamoxifen) | 48 | F | CT | Biopsy | LN mediastinal | 36 | (11) |
| Kim et al,
2014 | Breast | Ductal invasive
carcinoma | T1N2M0 | OP + aRCTx | 44 | F | PET |
Mediastinoscopy | LN paraesophageal,
hilar | 24 | (33) |
| Akhtari et
al, 2014 | Breast | Ductal invasive
carcinoma | T2N0Mx | OP + aCTx +
aRT | 47 | F | PET | FNA | LN supraclav.,
mediastinal, periportal | 0 | (34) |
| Braza and Nelson,
2014 | Breast | Ductal invasive
carcinoma | T1N0M0 | nk | 68 | F | MRI | Bone biopsy | Lesions lumbosacral
spine | nk | (35) |
| DeFilippis et
al,2013 | Breast | Lobular
carcinoma | nk | OP + aRTx | 63 | F | MRI | Biopsy | LN axillary | 0 | (36) |
| Bush et al,
2011 | Breast | Ductal invasive
carcinoma | N0 | OP | 42 | F | PET | Biopsy | LN cervical,
abdominal, bone, spleen | 0 | (37) |
| Viswanath et
al, 2009 | Breast | Ductal invasive
carcinoma | T4aN1M0 | OP + aRCTx | 50 | F | CI | Biopsy | Dermal lesions | 24 | (38) |
| Whittington et
al, 1986 | Breast | Carcinoma | N0 | nk | nk | F | nk |
Mediastinoscopy | LN mediastinal | 0 | (39) |
| Whittington et
al, 1986 | Breast | Carcinoma | N0 | nk | nk | F | nk | EBUS | LN hilar | 0 | (39) |
| El Hammoumi et
al, 2015 | Cervix | Epidermoid
Carcinoma | nk | naCTx + OP | 47 | F | PET |
Mediastinoscopy | LN mediastinal | 36 | (11) |
| Tamauchi et
al, 2015 | Endometrium | Adenocarcinoma | T1bN0Mx | OP + aCTx | 67 | F | CT | OP | LN hilar,
paraaortic, pelvine | 0 | (40) |
| Powell et
al, 2005 | Endometrium | Adenocarcinoma | N0 | OP | 67 | F | CT | FNA | LN mediastinal,
liver lesions | 48 | (41) |
| Takanami et
al, 2008 | Esophageal | Squamous cell
carcinoma | N0 | OP | 72 | M | PET | Biopsy | LN mediastinal +
hilar | −168 | (42) |
| Takanami et
al, 2008 | Esophageal | Squamous cell
carcinoma | pT1b | OP | 59 | M | PET | Biopsy | LN mediastinal +
hilar | 0 | (42) |
| Arana et al,
2013 | Ethmoid sinus | Adenocarcinoma | T3N0M0 | OP + aRCTx | 42 | F | PET |
Mediastinoscopy | LN mediastinal +
hilar | 0 | (7) |
| Kachalia et
al, 2014 | Lung | Adenocarcinoma | TxNxM2 | OP | 80 | F | X-ray |
Mediastinoscopy | LN | 0 | (13) |
| Kim et al,
2011 | Lung | Adenocarcinoma | T2N0M0 | OP | 65 | F | PET | Biopsy | LN mediastinal | 0 | (43) |
| Mimura et
al, 2011 | Lung | Squamous cell
carcinoma | pT1N0M0 | OP | 69 | M | CT | Biopsy | LN mediastinal | −120 | (44) |
| Urushiyama et
al, 2015 | Lung | Squamous cell
carcinoma | N0 | OP | 60 | M | CT | Biopsy | LN mediastinal +
hilar | −24 | (45) |
| Bouaziz et
al, 2006 | Lung | Squamous cell
carcinoma | nk | nk | 49 | M | MRI | EBUS | LN mediastinal,
hepatic nodules | 0 | (46) |
| Shields et
al, 2005 | Lung | Papillary
carcinoma | M1 | OP | 57 | F | PET | Radiology | Salivary and
lacrimal glands, LN hilar | 0 | (14) |
| Sato et al,
2003 | Lung | Adenocarcinoma | N1 | nk | nk | nk | nk | Thoracoscopy | LN mediastinal,
interlobar | 0 | (15) |
| Muramatsu et
al, 2000 | Lung | Squamous cell
carcinoma | N0 M0 | OP | 64 | M | CT | Biopsy | LN mediastinal | 0 | (47) |
| Abdel-Galiil et
al, 2008 | Maxilla | Squamous cell
carcinoma | nk | OP | 51 | M | PET |
Mediastinoscopy | LN
mediastinal+hilar + peribronchial | 24 | (8) |
| Yao et al,
2005 | Oropharynx | Squamous cell
carcinoma | T3N2cM0 | RCTx | 49 | M | PET |
Mediastinoscopy | LN mediastinal,
pretracheal, subcarinal | 2 | (9) |
| Yonenaga et
al, 2006 | Ovar | Mucinous
Cystadenocarcinoma | nk | OP + aCTx | 21 | F | PET | nk | Spleen, Liver | 36 | (48) |
| Kim et al,
2013 | Ovar | Papillary
cystadenocarcinoma | nk | OP + aCTx | 52 | F | PET | EBUS | LN paratracheal,
supraclavicular, diaphragmal | 12 | (49) |
| Pollock and
Catalano, 1979 | Parotid gland | Ductal
carcinoma | N2 | OP + aRCTx | 38 | M | CI | Biopsy | LN hilar | −60 | (50) |
| Montini and
Tulchinsky, 2012 | Rectum | Cancer | nk | nk | 45 | M | PET | Biopsy
skeletal | LN
mediastinal, | 0 | (51) |
| Abdi et al,
1987 | Renal | Renal cell
carcinoma | N2 | OP + aRTx
(IFNα) | 57 | F | CT | EBUS | LN mediastinal | 24 | (52) |
| Fukutani et
al, 1987 | Renal | Renal cell
carcinoma | NO | OP | 75 | F | nk | Biopsy | LN pelvic | 0 | (53) |
| Khan and Khan,
1974 | Renal | Hypernephroma | M1 | OP | 52 | M | X-ray | Biopsy | LN hilar | 0 | (19) |
| Gharavi et
al, 2013 | Sacrum | Chordoma | nk | OP | 48 | M | PET | Biopsy | LN iliacal +
femoral | 12 | (54) |
| Wilgenhof et
al, 2012 | Skin | Melanoma | M1c | OP + aCTx
(Dacarbazine, Cisplatin) | 48 | F | PET | EBUS | LN hilar | 84 | (55) |
| Vogel et al,
2012 | Skin | Melanoma | N1 | OP + aCTx
(αCTLA-4) | 49 | M | PET | EBUS | LN mediastinal,
hilar | 168 | (56) |
| Heinzerling et
al, 2010 | Skin | Melanoma | pT4N0M0 | OP + aCTx
(INFα) | 50 | M | nk | Biopsy | LN mediastinal | 7 | (57) |
| Chiagne et
al, 2011 | Skin | Melanoma | nk | OP | 35 | M | PET | Biopsy | LN inguinal | 0 | (18) |
| Heinzerling et
al, 2010 | Skin | Melanoma | pT3bpN1acM0 | OP + aCTx
(INFα) | 47 | M | PET | Biopsy | LN mediastinal+
hilar + peribronchial | nk | (57) |
| Heinzerling et
al, 2010 | Skin | Melanoma | N1 | OP + aCTx
(INFα) | 47 | M | PET |
Mediastinoscopy | LN mediastinal+
hilar + peribronchial | 2 | (57) |
| Suarez-Garcia et
al, 2009 | Skin | Melanoma | N1 | OP + aCTx
(INFα) | 42 | M | CI | Biopsy | Dermal lesions | 3 | (58) |
| Massaguer et
al, 2004 | Skin | Melanoma | nk | CTx (IFNα) | nk | F | CT |
Mediastinoscopy | LN mediastinal | nk | (59) |
| Matsubara et
al, 2015 | Stomach | Adenocarcinoma | N0 | OP | 64 | F | nk | Endoscopy | Gastric
sarcoidosis | −120 | (60) |
| El Hammoumi et
al, 2015 | Stomach | Adenocarcinoma | nk | OP + aCTx | 59 | F | CT |
Mediastinoscopy | LN
paratracheal | 36 | (11) |
| Tissot et
al, 1985 | Stomach | Adenocarcinoma | nk | nk | 63 | F | OP | Biopsy | Combined gastric
lesions | −336 | (61) |
| Claus et al,
2012 | Testis | Seminoma | T2N0M1 | OP + aCTx
(Carboplatin) | 34 | M | CT | EBUS | LN mediastinal | 24 | (62) |
| Claus et al,
2012 | Testis | Seminoma | T1N0M0 | OP + aCTx
(Carboplatin) | 36 | M | CT | Biopsy | LN abdominal | 0 | (62) |
| Teo et al,
2013 | Testis | Seminoma | T1N2M1a | OP + aCTx
(Cisplatin, Etoposid) | 20 | M | CT | EBUS | LN mediastinal | 60 | (63) |
| Tjan-Heijnen et
al, 1998 | Testis | Seminoma | N2 | OP + aRTx | 41 | M | CT |
Mediastinoscopy | LN mediastinal | 24 | (64) |
| Salih et al,
2015 | Thyroid | Papillary thyroid
carcinoma | T2N1Mx | OP | 48 | F | X-ray | Neck
dissection | LN cervical and
hilar | 0 | (10) |
| Lebo et al,
2015 | Thyroid | Papillary thyroid
carcinoma | T1bN1aMx | OP | 41 | F | PET |
Mediastinoscopy | Cervical +
mediastinal | 0 | (16) |
| Myint et al,
2015 | Thyroid | Papillary thyroid
carcinoma | nk | OP + I-131 | 68 | F | PET | Bone biopsy | LN hilar + mediast.
Bone | nk | (65) |
| Ergin and Nasr,
2014 | Thyroid | Papillary thyroid
carcinoma | N0 | OP | nk | nk | nk | OP | Cervical | pre | (17) |
| Ergin and Nasr,
2014 | Thyroid | Papillary thyroid
carcinoma | N0 | OP | nk | nk | nk | FNA | Cervical | post | (17) |
| Ergin and Nasr,
2014 | Thyroid | Papillary thyroid
carcinoma | N1 | OP | nk | nk | PET | OP | Cervical | 0 | (17) |
| Ergin and Nasr,
2014 | Thyroid | Papillary thyroid
carcinoma | N0 | OP | nk | nk | nk | OP | Cervical | 0 | (17) |
| Zimmermann-Belsing
et al, 2000 | Thyroid | Papillary
adenocarcinoma | N2 | OP | 27 | M | Scintigraphy | Biospy | LN hilar | −36 | (66) |
The first case describes a 50-year-old female
patient suffering from ductal invasive breast carcinoma with local
lymph node metastasis (pT3pN1). Chest X-ray and
18FDG-PET-CT were performed for staging of the tumor and
showed a bilateral mediastinal lymphadenopathy and an increased
FDG-uptake in supra-diaphragmatic and pelvic lymph nodes. Biopsy of
an example lesion obtained sarcoidosis (12). Secondly, an 80-year-old female's CT
scan revealed a tumor suspicious mass in the upper lobe of the
right lung, multiple smaller nodules and hilar lymphadenopathy.
Subsequent biopsy of the mass and a mediastinal lymph node showed
just noncaseating granulomas but no malignant cells and led to
insufficient treatment. Six months later, symptoms like cough and
chest pain were exacerbated and a thoracocentesis revealed
adenocarcinoma cells. Further staging examination showed pleural,
pericardial and diaphragmatic metastasis. Due to tumor progress,
palliative care was initiated (13).
The third case describes a 57-year-old female patient who was found
to have a choroidal mass in the left eye. Total body gallium 67
scan showed an increased uptake in salivary and lacrimal glands and
was misinterpreted as typical for sarcoidosis. Progress of symptoms
resulted in enucleation and revealed a choroideal metastasis of a
papillary lung carcinoma (14). Sato
et al (15) report on a
patient with concomitant sarcoidosis and lung adenocarcinoma.
Thoracoscopic biopsy of altered lymph nodes did not detect
metastasis but sarcoidosis. Surgery was performed and a permanent
pathological slide showed that several nodes contained both
sarcoidosis and lung cancer metastasis (15). Three other reports describe patients
suffering from papillary thyroid carcinoma that underwent
thyreoidectomy and modified neck dissection. Pathology revealed
concurrent existence of sarcoidosis and regional lymph node
metastasis (10,16,17). One
patient (27-year-old, male) shows a papillary thyroid carcinoma
upon a previously diagnosed sarcoidosis. Local lymph nodes
contained sarcoidosis mixed with metastasis. A 35-year-old male
with a previous history of melanoma developing metastatic
involvement and sarcoidosis in regional lymph nodes was described
by Chaigne et al (18). Khan
and Khan (19) described a
52-year-old patient with cough and chest pain. Radiologic
examination showed bilateral hilar lymphadenopathy. Furthermore,
within a biopsy of an enlarged lymph node, a metastasis of a left
kidney hypernephroma was detected (19).
Taken together, these ten patients had a median age
of 49 years ranging from 27 to 80 years at the time point of
simultaneous detection of metastasis and sarcoidosis. The gender
ratio was 0.6:1 (male to female) although no information concerning
the sex was provided in two cases. In 8 cases (80%), the metastases
were localized in regional lymph nodes whereas just 2 cases showed
distant metastases. Furthermore, the region and entity of the
associated tumor differed greatly (breast: n=1, lung: n=3, thyroid:
n=4, skin: n=1, kidney: n=1).
Discussion
Sarcoidosis and metastasis
Although there are several published cases
concerning coexistence of malignoma and sarcoidosis, the causal
relationship between these entities is still unclear. On the one
hand, it is possible that patients with sarcoidosis develop
malignancies and, on the other hand, there are oncologic patients
developing sarcoidosis and sarcoid-like reactions, especially after
chemotherapy (3). Several cases
describe a mimicking of metastatic disease by sarcoidosis but just
a few cases actually report a simultaneous occurrence of
sarcoidosis and metastases.
Active sarcoidosis is characterized by an enhanced
local expression of T helper 1 (Th1) and T helper 17 (Th17)
chemokines and cytokines like IFN-γ, TNF-α, IL-17A and IL-22. In
various chronic, autoimmune, inflammatory diseases, such as
sarcoidosis, the percentage of IL-17A+/IFN-γ+
double-producing Th-cells is increased in peripheral blood and is
related to high disease activity. Furthermore, in these
pathological conditions, a dysfunctional response of regulatory
T-cells (Tregs) has been described that is characterized by an
insufficient immunosuppressive function (20). Interestingly, cytotoxic T-lymphocyte
antigen 4 (CTLA-4) expression is decreased while PD-1 (programmed
death-1) expression is increased in Th17-cells in the mediastinal
lymph nodes during sarcoidosis (20).
CTLA-4 is present on Th-cells and mediates an inhibitory effect on
further T cells responses. Hence, a diminished CTLA-4 level
maintains inflammatory reactions. Similarly, PD-1 is expressed on
the surface of T-cells upon activation and is involved in limiting
inflammatory reactions (21). Its
ligand, PD-1L, can be found on tumor cells and provokes
upregulation of PD-1 in T cells. Consequently, activation of tumor
antigen-specific T-cells in pancreatic adenocarcinoma is inhibited
(22). Hence, the bivalent adaptive
immune response in patients with sarcoidosis and metastasis
promotes both pathological conditions by maintaining
sarcoidosis-related inflammation due to the decreased
anti-inflammatory CTLA-4 expression while limiting tumor-specific T
cell activation, marked by an increased PD-1 expression, that
enables tumor escape from the immune system and metastasis.
Increased PD-1 expression on T-cells in sarcoidosis lymph nodes
could thus be a possible predictor of metastasis on the basis of
sarcoidosis. Furthermore, myeloid-derived suppressor cells (MDSC)
might play a pivotal role in the pathogenic association between
sarcoidosis and metastasis. MDSC pursue immunoregulatory and T-cell
suppressive functions (23). Although
it has not been described yet, an influence of MDSC on sarcoidosis
is assumable because of their important role in other inflammatory
diseases (24). Th-17 cells are the
main source of IFN-γ production in sarcoidosis and IFN-γ induces
MDSC differentiation and promotes their immunosuppressive function
(20,25). In cancer models, MDSC accumulation was
promoted by several cytokines and growth factors, such as IL-6,
IL-1ß and S100A8/A9, resulting in an anti-inflammatory tumor
microenvironment (23). MDSC
themselves express cytokines and chemokines like IL-6, TNF, IL-1β,
IL-23 and S100A9 and have the potential to attract both further
myeloid cells and tumor cells (26).
Furthermore, in a melanoma and lung carcinoma mouse model, S100A9
expressing MDSC were identified as important players in enabling
tumor metastasis (27). Consequently,
they are generally recognized as dominant tumor-promoting forces
(28). Patients suffering from
sarcoidosis have increased serum levels of S100A8/A9 and an
enhanced cytoplasmatic S100A8/A9 expression in monocytes and
multinuclear giant cells in granulomas (29). Because of the sarcoidosis-related
inflammation, marked by the expression of IFN-γ, IL-6, IL-23 and
S100A8/A9, we can suspect a similar microenvironment to tumors that
are characterized by an increased accumulation and
immunosuppressive function of MDSC. Accordingly, we can assume that
sarcoidosis has the potential to promote metastasis by inducing
tumor-promoting and immune-regulating cell subsets. Further
analysis is necessary to verify the influence of MDSC on
sarcoidosis as well as the cellular immune response concerning the
association between sarcoidosis and metastasis.
Differentiation between benign and
malignant lesions
PET-CT scan is a very useful diagnostic tool to
identify malignant lesions with a sensitivity between 47 and 100%
and a specificity of 86–100% (30).
Unfortunately, elevated FDG uptake can also be detected in
inflammatory diseases such as sarcoidosis causing a diagnostic
dilemma. The case of a 61-year-old carpenter with a history of
adenocarcinoma of the paranasal sinus and simultaneous occurrence
of multiple cervical metastases and sarcoidosis detected during
follow-up investigation impressively demonstrates the risk to
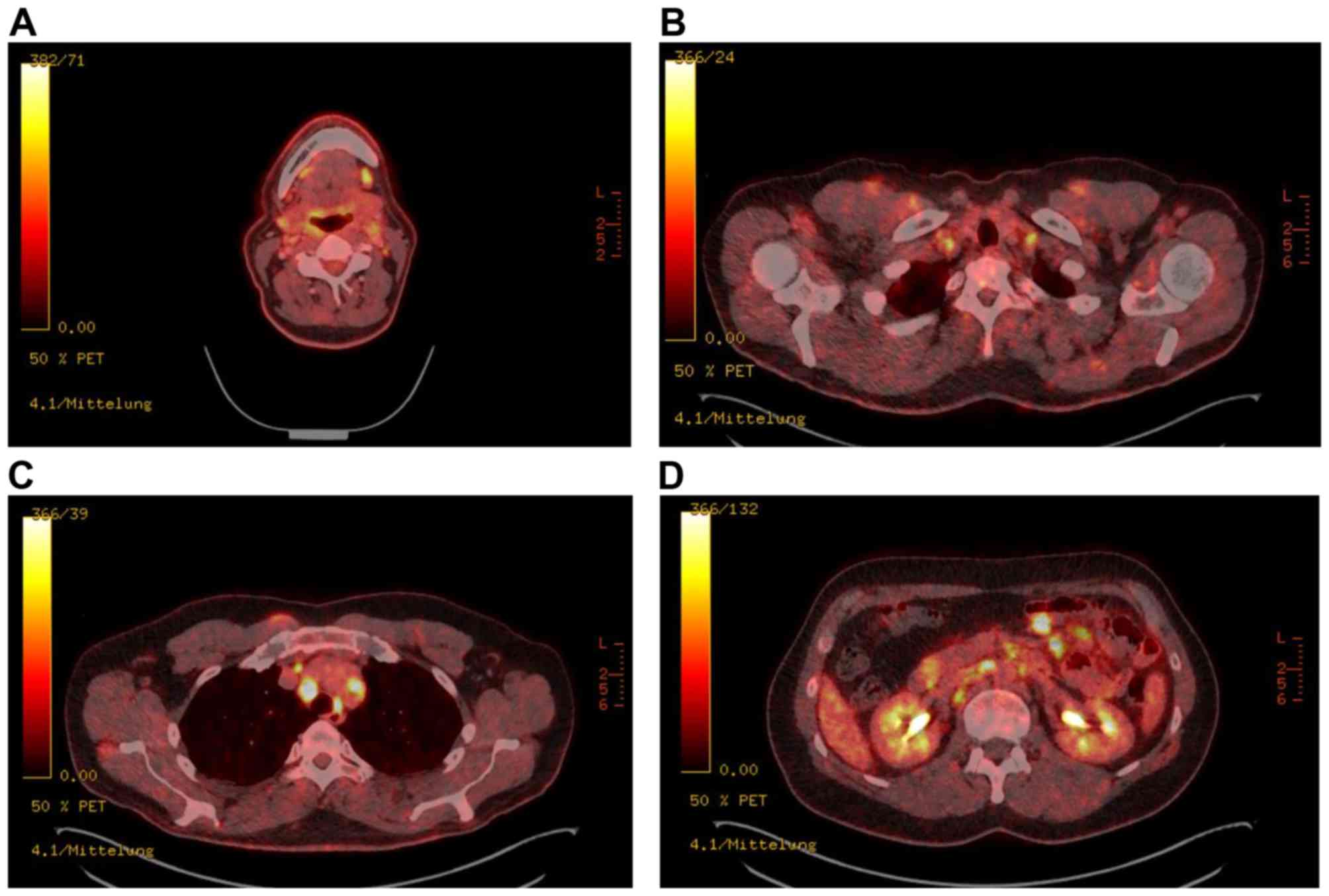
overlook metastatic lesions within sarcoidosis (Fig. 1). Especially PET scans for staging or
restaging of oncologic diseases supply important information about
tumor progression. Decisions on curative or palliative therapy are
based on this information, emphasizing the importance to avoid
misdiagnosis. Inclusion of an additional tracer would be helpful to
differentiate between inflammation and tumor. F18-labeled
3′deoxy-3′fluorothymidine (FLT) is such a promising tracer to
minimize diagnostic and subsequent therapeutic mistakes. By
measuring DNA synthesis instead of metabolic activity that seems to
be more specific to detect tumor diseases, FLT might be a useful
candidate to discriminate between tumor and sarcoidosis lesions
(7).
An additional helpful tool to discriminate
inflammatory bone marrow involvement, like skeletal sarcoidosis,
from metastatic disease might be the diffusion whole-body magnetic
resonance imaging (b-values 50–900s/mm2). In contrast to
malignant lesions (cut-off value of 774 µm2/s),
sarcoidosis or other inflammatory skeletal reactions show high
signal intensity on diffusion-weighted images and a lower apparent
diffuse coefficient (ADC) (12).
Bioptical evaluation of radiologically altered lymph
nodes is necessary for selection of appropriate oncological
treatment strategy. However, examination of each PET-CT positive
lesion is not feasible and the chance to detect metastatic lesions
next to sarcoidosis is rather rare (Fig.
2). Thus, even if pathological findings were suspicious of
sarcoidosis, concomitant metastasis cannot be certainly excluded.
Hence, correlation between the radiological and histological
findings with the probability of distant or local metastasis
corresponding to the tumor entity is important for the careful
assessment of the residual metastasis risk.
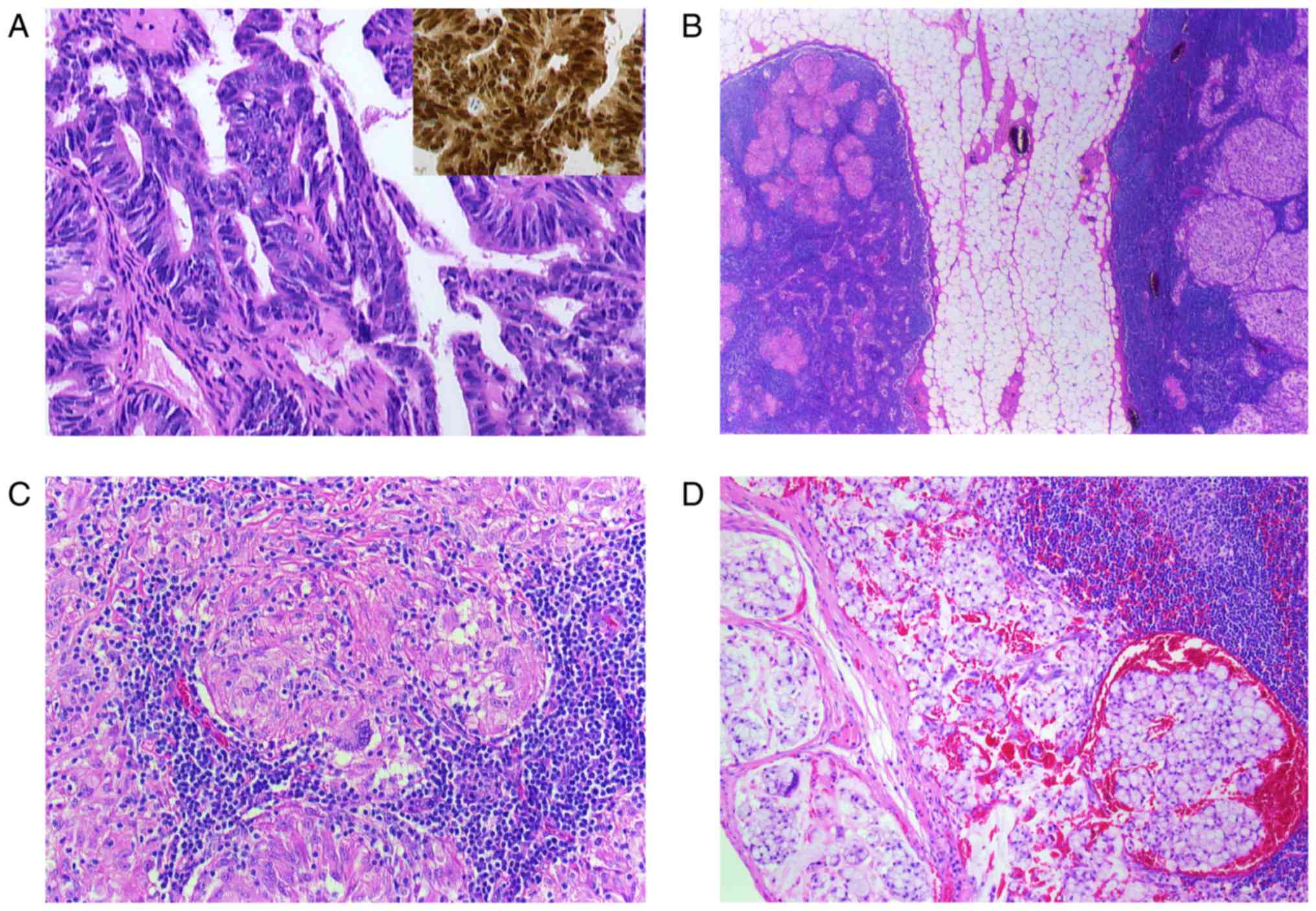 | Figure 2.(A-D) Histological findings of a
resected paranasal sinus adenocarcinoma and lymph node. (A)
Microphotograph shows the adenocarcinoma with atypical tumor cells
(H&E; magnification, ×400) and CDX2 nuclear positive cells,
characteristic for intestinal-type adenocarcinoma (inset, H&E,
magnification, ×400). (B) Two lymph nodes surrounded by perinodal
fat tissue. The right node with noncaseating epitheloid granulomas
and the left one with metastatic lesions (H&E, magnification,
×5). (C) Lymph node with noncaseating epitheloid granuloma in
higher magnification (H&E, magnification, ×200). Multinuclear
giant cells are identifiable. (D) Metastatic carcinoma infiltration
in a lymph node (H&E, magnification, ×200). H&E,
hematoxylin and eosin; CDX2, caudal type homeobox 2. |
Conclusion
Correlation between PET-Scan, histological findings
and knowledge about typical tumor behavior is necessary to avoid
misdiagnosis. Nevertheless, a residual risk of overlooking
metastases in systemic inflammatory diseases still remains
existent. Therefore, it is important for clinical practice to be
aware of the simultaneous occurrence of sarcoidosis and metastatic
malignancy. Further cell subset analysis in these pathologies might
additionally reveal immunological distinct cell populations as
useful markers to distinguish between sarcoidosis, cancer and the
coexistence of these two and help in overcoming the current
diagnostic dilemma.
Acknowledgements
This study was supported by a fellowship from the
medical faculty of the University of Münster, Germany to C.
Spiekermann.
References
|
1
|
Statement on sarcoidosis. Joint Statement
of the American Thoracic Society (ATS), the European Respiratory
Society (ERS) and the World Association of Sarcoidosis and Other
Granulomatous Disorders (WASOG) adopted by the ATS Board of
Directors and by the ERS Executive Committee, February 1999. Am J
Respir Crit Care Med. 160:736–755. 1999.PubMed/NCBI
|
|
2
|
Rosen Y: Pathology of sarcoidosis. Semin
Respir Crit Care Med. 28:36–52. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cohen PR and Kurzrock R: Sarcoidosis and
malignancy. Clin Dermatol. 25:326–333. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Brincker H: Sarcoid reactions in malignant
tumours. Cancer Treat Rev. 13:147–156. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Askling J, Grunewald J, Eklund A,
Hillerdal G and Ekbom A: Increased risk for cancer following
sarcoidosis. Am J Respir Crit Care Med. 160:1668–1672. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Boffetta P, Rabkin CS and Gridley G: A
cohort study of cancer among sarcoidosis patients. Int J Cancer.
124:2697–2700. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yi Arana C, McCue P, Rosen M, Machtay M,
Axelrod R and Morris GJ: Sarcoidosis mimicking metastatic bone
disease in head and neck cancer. Semin Oncol. 40:529–534. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Abdel-Galiil K, Anand R, Sharma S, Brennan
PA, Ramchandani PL and Ilankovan V: Incidence of sarcoidosis in
head and neck cancer. Br J Oral Maxillofac Surg. 46:59–60. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yao M, Funk GF, Goldstein DP, DeYoung BR
and Graham MM: Benign lesions in cancer patients: Case 1.
Sarcoidosis after chemoradiation for head and neck cancer. J Clin
Oncol. 23:640–641. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Salih AM, Fatih SM and Kakamad FH:
Sarcoidosis mimicking metastatic papillary thyroid cancer. Int J
Surg Case Rep. 16:71–72. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
El Hammoumi M, El Marjany M, Moussaoui D,
Doudouh A, Mansouri H and Kabiri el H: Mediastinal sarcoidosis
mimicking lymph malignancy recurrence after anti-neoplastic
therapy. Arch Bronconeumol. 51:e33–e35. 2015.(In English, Spanish).
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Conte G, Zugni F, Colleoni M, Renne G,
Bellomi M and Petralia G: Sarcoidosis with bone involvement
mimicking metastatic disease at (18)F-FDG PET/CT: Problem solving
by diffusion whole-body MRI. Ecancermedicalscience. 9:5372015.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kachalia AG, Ochieng P, Kachalia K and
Rahman H: Rare coexistence of sarcoidosis and lung adenocarcinoma.
Respir Med Case Rep. 12:4–6. 2014.PubMed/NCBI
|
|
14
|
Shields JA, Shields CL and Eagle RC Jr:
Choroidal metastasis from lung cancer masquerading as sarcoidosis.
Retina. 25:367–370. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sato Y, Sasano S, Oyama K, Sakuraba M,
Onuki T and Nitta S: Lung cancer associated with sarcoidosis. Jpn J
Thorac Cardiovasc Surg. 51:21–24. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lebo NL, Raymond F and Odell MJ:
Selectively false-positive radionuclide scan in a patient with
sarcoidosis and papillary thyroid cancer: A case report and review
of the literature. J Otolaryngol Head Neck Surg. 44:182015.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ergin AB and Nasr CE: Thyroid cancer &
sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 31:239–243.
2014.PubMed/NCBI
|
|
18
|
Chaigne B, Perrinaud A, Penaud A, Machet
MC, Venel Y, Marchand-Adam S and Machet L: Melanoma lymph node
metastasis occurring simultaneously with multifocal sarcoidosis
affecting lymph nodes and the lung: A diagnostic pitfall. Eur J
Dermatol. 21:798–799. 2011.PubMed/NCBI
|
|
19
|
Khan A and Khan FA: Hypernephroma: A rare
cause of bilateral adenopathy, and an example of the importance of
tissue diagnosis in suspected cases of sarcoidosis. Chest.
66:722–723. 1974. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Broos CE, Hendriks RW and Kool M: T-cell
immunology in sarcoidosis: Disruption of a delicate balance between
helper and regulatory T-cells. Curr Opin Pulm Med. 22:476–483.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hashemi-Sadraei N, Sikora AG and Brizel
DM: Immunotherapy and checkpoint inhibitors in recurrent and
metastatic head and neck cancer. Am Soc Clin Oncol Educ Book.
35:e277–e282. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Seo YD and Pillarisetty VG: T-cell
programming in pancreatic adenocarcinoma: A review. Cancer Gene
Ther. 24:106–113. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Crook KR and Liu P: Role of
myeloid-derived suppressor cells in autoimmune disease. World J
Immunol. 4:26–33. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Nagaraj S, Collazo M, Corzo CA, Youn JI,
Ortiz M, Quiceno D and Gabrilovich DI: Regulatory myeloid
suppressor cells in health and disease. Cancer Res. 69:7503–7506.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhao Y, Wu T, Shao S, Shi B and Zhao Y:
Phenotype, development, and biological function of myeloid-derived
suppressor cells. Oncoimmunology. 5:e10049832015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu Y, Kosaka A, Ikeura M, Kohanbash G,
Fellows-Mayle W, Snyder LA and Okada H: Premetastatic soil and
prevention of breast cancer brain metastasis. Neuro Oncol.
15:891–903. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yang Q, Li X, Chen H, Cao Y, Xiao Q, He Y,
Wei J and Zhou J: IRF7 regulates the development of granulocytic
myeloid-derived suppressor cells through S100A9 transrepression in
cancer. Oncogene. 36:2969–2980. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zamarron BF and Chen W: Dual roles of
immune cells and their factors in cancer development and
progression. Int J Biol Sci. 7:651–658. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Terasaki F, Fujita M, Shimomura H, Tsukada
B, Otsuka K, Otsuka K, Katashima T, Ikemoto M and Kitaura Y:
Enhanced expression of myeloid-related protein complex (MRP8/14) in
macrophages and multinucleated giant cells in granulomas of
patients with active cardiac sarcoidosis. Circ J. 71:1545–1550.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zanation AM, Sutton DK, Couch ME, Weissler
MC, Shockley WW and Shores CG: Use, accuracy, and implications for
patient management of [18F]-2-fluorodeoxyglucose-positron
emission/computerized tomography for head and neck tumors.
Laryngoscope. 115:1186–1190. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Altinkaya M, Altinkaya N and Hazar B:
Sarcoidosis mimicking metastatic breast cancer in a patient with
early-stage breast cancer. Ulus Cerrahi Derg. 32:71–74.
2015.PubMed/NCBI
|
|
32
|
Zivin S, David O and Lu Y: Sarcoidosis
mimicking metastatic breast cancer on FDG PET/CT. Intern Med.
53:2555–2556. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kim HS, Lee SY, Oh SC, Choi CW, Kim JS and
Seo JH: Case report of pulmonary sarcoidosis suspected to be
pulmonary metastasis in a patient with breast cancer. Cancer Res
Treat. 46:317–321. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Akhtari M, Quesada JR, Schwartz MR, Chiang
SB and Teh BS: Sarcoidosis presenting as metastatic lymphadenopathy
in breast cancer. Clin Breast Cancer. 14:e107–e110. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Braza DW and Nelson PA: Vertebral
sarcoidosis masquerading as breast metastasis. Am J Phys Med
Rehabil. 93:2742014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
DeFilippis EM and Arleo EK: New diagnosis
of sarcoidosis during treatment for breast cancer, with
radiologic-pathologic correlation. Clin Imaging. 37:762–766. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Bush E, Lamonica D and O'Connor T:
Sarcoidosis mimicking metastatic breast cancer. Breast J.
17:533–535. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Viswanath L, Pallade S, Krishnamurthy B,
Naveen T, Preethi BL, Pramod KP, Reddy O and Padma G: Darier-roussy
sarcoidosis mimicking metastatic breast cancer. Case Rep Oncol.
2:251–254. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Whittington R, Lazarus A, Nerenstone S and
Martin A: Sarcoidosis developing during therapy for breast cancer.
Chest. 89:762–763. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Tamauchi S, Shimomura Y and Hayakawa H:
Endometrial cancer with sarcoidosis in regional lymph nodes: A case
report. Case Rep Oncol. 8:409–415. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Powell JL, Cunill ES, Gajewski WH and
Novotny DB: Sarcoidosis mimicking recurrent endometrial cancer.
Gynecol Oncol. 99:770–773. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Takanami K, Kaneta T, Yamada T, Kinomura
S, Yamada S, Fukuda H and Takahashi S: FDG PET for esophageal
cancer complicated by sarcoidosis mimicking mediastinal and hilar
lymph node metastases: Two case reports. Clin Nucl Med. 33:258–261.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kim JJ, Park JK, Wang YP, Choi SH and Jo
KH: Lung cancer associated with sarcoidosis - A case report. Korean
J Thorac Cardiovasc Surg. 44:301–303. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Mimura K, Mochizuki Y, Nakahara Y,
Kawamura T, Sasaki S and Katsuda R: A case of primary lung cancer
with swollen mediastinal lymph nodes due to pre-existing
sarcoidosis. Nihon Kokyuki Gakkai Zasshi. 49:208–213. 2011.(In
Japanese). PubMed/NCBI
|
|
45
|
Urushiyama H, Yamauchi Y, Suzuki S,
Sunohara M, Kouyama T, Ohishi N, Fukami T, Nakajima J, Ushiku T,
Oota S, et al: Case of sarcoidosis with squamous cell carcinoma
which originated from solitary bronchial papilloma. Nihon Kokyuki
Gakkai Zasshi. 48:815–820. 2010.(In Japanese). PubMed/NCBI
|
|
46
|
Bouaziz H, Kaffel N, Charfi N, Fourati M,
Abid H and Abid M: Panhypopituitarism revealing metastasis of
small-cell lung carcinoma associated with sarcoidosis. Ann
Endocrinol (Paris). 67:259–264. 2006.(In French). View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Muramatsu M, Kuriyama M, Takahashi K,
Miyamoto H, Uekusa T, Danbara T and Fukuchi Y: A case of resected
squamous cell carcinoma of the lung complicated with sarcoidosis.
Nihon Kokyuki Gakkai Zasshi. 38:720–725. 2000.(In Japanese).
PubMed/NCBI
|
|
48
|
Yonenaga Y, Kushihata F, Inoue H, Watanabe
J, Tohyama T, Sugita A and Takada Y: Sarcoidosis manifesting as
hepatic and splenic nodules mimicking ovarian cancer metastases: A
case report. Oncol Lett. 10:2166–2170. 2015.PubMed/NCBI
|
|
49
|
Kim MH, Lee K, Kim KU, Park HK, Lee MK and
Suh DS: Sarcoidosis mimicking cancer metastasis following
chemotherapy for ovarian cancer. Cancer Res Treat. 45:354–358.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Pollock JL and Catalano E: Metastatic
ductal carcinoma of the parotid gland in a patient with
sarcoidosis. Arch Dermatol. 115:1098–1099. 1979. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Montini KM and Tulchinsky M:
False-positive bone metastases on PET/CT secondary to sarcoidosis
in a patient with rectal cancer. Clin Nucl Med. 37:307–310. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Abdi EA, Nguyen GK, Ludwig RN and Dickout
WJ: Pulmonary sarcoidosis following interferon therapy for advanced
renal cell carcinoma. Cancer. 59:896–900. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Fukutani K, Kawabe K, Moriyama N, Kitamura
T and Murakami T: Carcinoma of the renal pelvis and bladder
associated with sarcoidosis: A case report. Urol Int. 42:224–226.
1987. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Gharavi MH, Wu HH and Toms SA: High
fluorodeoxyglucose ((18)F)PET-uptake lymph nodes in a patient with
chordoma: Tumor metastasis or sarcoidosis? Am J Case Rep.
14:373–375. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Wilgenhof S, Morlion V, Seghers AC, Four
Du S, Vanderlinden E, Hanon S, Vandenbroucke F, Everaert H and
Neyns B: Sarcoidosis in a patient with metastatic melanoma
sequentially treated with anti-CTLA-4 monoclonal antibody and
selective BRAF inhibitor. Anticancer Res. 32:1355–1359.
2012.PubMed/NCBI
|
|
56
|
Vogel WV, Guislain A, Kvistborg P,
Schumacher TN, Haanen JB and Blank CU: Ipilimumab-induced
sarcoidosis in a patient with metastatic melanoma undergoing
complete remission. J Clin Oncol. 30:e7–e10. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Heinzerling LM, Anliker MD, Muller J,
Schlaeppi M and von Moos R: Sarcoidosis induced by interferon-α in
melanoma patients: Incidence, clinical manifestations, and
management strategies. J Immunother. 33:834–839. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Suarez-Garcia C, Pérez-Gil A,
Pereira-Gallardo S, Codes-Villena M, García-Escudero A and Camacho
Miguel F: Interferon-induced cutaneous sarcoidosis in melanoma.
Melanoma Res. 19:391–394. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Massaguer S, Sánchez M and Castel T:
Mediastinal sarcoidosis induced by high-dose alpha-2-interferon
therapy in a patient with malignant melanoma. Eur Radiol.
14:1716–1717. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Matsubara T, Hirahara N, Hyakudomi R,
Fujii Y, Kaji S, Taniura T and Tajima Y: Early gastric cancer
associated with gastric sarcoidosis. Int Surg. 100:949–953. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Tissot E, Bringeon G, Berger F and Kalb
JC: Cancer and gastric sarcoidosis. J Chir (Paris). 122:479–481.
1985.(In French). PubMed/NCBI
|
|
62
|
Claus F, De Wever L and Moerman P:
Coincidence of seminoma and sarcoidosis in two patients presenting
with peritoneal surface disease. Int J Urol. 19:11262012.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Teo M, McCarthy JE, Brady AP, Curran DR
and Power DG: A case of sarcoidosis in a patient with testicular
cancer post stem cell transplant. Acta Oncol. 52:869–871. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Tjan-Heijnen VC, Vlasveld LT, Pernet FP,
Pauwels P and De Mulder PH: Coincidence of seminoma and
sarcoidosis: A myth or fact? Ann Oncol. 9:321–325. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Myint ZW and Chow RD: Sarcoidosis
mimicking metastatic thyroid cancer following radioactive iodine
therapy. J Community Hosp Intern Med Perspect. 5:263602015.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Zimmermann-Belsing T, Christensen L,
Hansen HS, Kirkegaard J, Blichert-Toft M and Feldt-Rasmussen U: A
case of sarcoidosis and sarcoid granuloma, papillary carcinoma, and
Graves' disease in the thyroid gland. Thyroid. 10:275–278. 2000.
View Article : Google Scholar : PubMed/NCBI
|