Introduction
Epithelial ovarian cancer is the leading cause of
mortality among gynecological types of cancer. Surgical debulking
combined with chemotherapy is the standard therapeutic strategy.
However, the relapse rate is high, primarily due to the development
of chemotherapy resistance (1),
therefore, novel modalities must be explored.
Dopamine receptor (DR) expression may be associated
with the development of various types of cancer. Patients with
schizophrenia who receive DR antagonists have a reduced incidence
of cancer of the rectum, colon, prostate and uterine cervix
(2,3).
Patients with Parkinson's disease, which functionally similar to
disease-induced DR antagonism, also have a lower incidence of
cancer. It was hypothesized that DR may be a biomarker for cancer
(4). Knockdown/blocking of DR2
inhibited the proliferation of cancer cells, including cancer stem
cells (5). This suggested that DR2
may be a treatment target for types of cancer that expresses
dopamine receptor 2.
Thioridazine is a DR2 antagonist and has been
clinically approved to treat schizophrenia and other psychotic
disorders (6). Of note, thioridazine
exhibits anticancer action in breast cancer, leukemia, hepatoma and
cervical carcinoma (5,7–9). Ovarian
cancer cells express a number of DRs, with the exception of DR3
(10), suggesting that thioridazine
may be used to treat ovarian cancer. In the present study, the
effects of thioridazine on ovarian cancer were explored in
vitro and in vivo. The findings suggested that
thioridazine may be a promising candidate drug for ovarian cancer
therapy.
Materials and methods
Reagents
Thioridazine was obtained from Sigma-Aldrich (Merck
KGaA, Darmstadt, Germany) and kept as 50 mM stock solutions in
water. Autophagy inhibitor 3-methyl adenine (3-MA) was purchased
from Sigma-Aldrich (Merck KGaA) and kept as 50 mM stock solutions
in dimethyl sulfoxide.
Cell culture
A2780 and SKOV3 human ovarian cancer cell lines
(China Center for Type Culture Collection, Wuhan, China) were
cultured in RPMI-1640 medium (Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) supplemented with 10% fetal bovine serum
(Zhejiang Hisun Chemical Co., Ltd., Taizhou, China) and 1%
penicillin-streptomycin solution at 37°C and 5% CO2.
Cell survival
SKOV3 and A2780 cells were seeded into a 96-well
plate at a density of 1×104 cells/well, followed by
thioridazine treatment with increasing doses (5, 10, 15, 20 and 25
µm) for 24 h at 37°C. The controls were not exposed to the
thioridazine. The cell viability was determined by performing a
tetrazolium assay (Cell Counting kit-8; Dojindo Molecular
Technologies, Inc., Kumamoto, Japan) according to the protocol of
the manufacturer. A concentration of 15 µM thioridazine induced
cell inhibition, which was then utilized in subsequent experiments.
Cells (5×105) were seeded in a 6-well plate, then 15 µM
thioridazine was added and maintained at 37°C for various time
periods (6, 12 and 24 h) prior to assay analysis. Following
exposure to 15 µM thioridazine for various time periods (6, 12 and
24 h), the morphological changes and the protein expression related
to the DR2, apoptosis and autophagy were detected. Experiments were
performed in triplicate.
Detection of reactive oxygen species
(ROS)
Cells were incubated with 10 µM
2′,7′-dichlorofluorescein diacetate (Sigma-Aldrich; Merck KGaA) for
30 min at 37°C, rinsed with PBS, trypsinized, centrifuged (800 × g
for 5 min at room temperature), resuspended in 1 ml PBS and then
analyzed by flow cytometry, and 4 fields of view were observed
under a fluorescence microscope at magnification, ×200.
Detection of light chain 3 (LC3) using
immunofluorescence
Cells were rinsed with PBS three times, fixed in 4%
paraformaldehyde for 15 min, blocked with 5% bovine serum albumin
(Beyotime Institute of Biotechnnology, Haimen, China) for 30 min
and permeabilized with 1% Triton X-100 for 10 min, all at room
temperature. Subsequently, cells were incubated with a rabbit
anti-LC3 antibody (1:100) (Sigma-Aldrich; Merck KgaA; cat. no.
L8918) at 4°C overnight. Following incubation with a secondary
antibody (goat anti-rat IgG (H+L), FITC conjugate, 1:1,000 (cat.
no. SA00003-11); ProteinTech Group, Inc., Chicago, IL, USA) for 1 h
at room temperature and nucleus staining using DAPI (Beyotime
Institute of Biotechnnology), the cells in 4 fields of view were
visualized using a fluorescence microscope at magnification,
×200.
Detection of apoptosis using Annexin V. SKOV3 and
A2780 cells were pre-treated with 5 mM 3-MA (3-Methyladenine)
(Sigma-Aldrich; Merck KGaA; cat. no. M9281) or not, followed by a
24-h thioridazine treatment. Cells were stained using fluorescein
isothiocyanate-labelled Annexin V (Nanjing KeyGen Biotech Co.,
Ltd., Nanjing, China) according to the protocol of the
manufacturer, and analyzed using flow cytometry (FACSCanto II)
according to the protocol of the manufacturer.
Western blotting
Nuclear factor erythroid 2-related 2 (Nrf2),
phosphorylated Nuclear factor erythroid 2-related 2 (p-Nrf2),
NAD(P)H quinone dehydrogenase 1 (NQO1), heme oxygenase 1 (HO-1),
c-Jun N-terminal kinase (JNK), p-JNK, p38, p-p38, extracellular
signal-related kinase (ERK), p-ERK, protein kinase B (AKT), p-AKT,
hypoxia inducible factor-1 (HIF-1), vascular endothelial growth
factor (VEGF), P62, LC3 and cleaved caspase-3 were detected using
western blotting. Total proteins were harvested by lysing cells in
a buffer supplemented with protease inhibitor for 10 min in an ice
bath. Equal amounts (25 µg/µl) of protein were separated using
8–12% gradient SDS-PAGE (Bio-Rad Laboratories, Inc., Hercules, CA,
USA) and then transferred to polyvinylidene difluoride membranes.
Following blocking 5% skimmed milk powder for 2 h at room
temperature, the membranes were incubated with various primary
antibodies (1:1,000) overnight. Antibodies against Nrf2 (cat. no.
12721s), NQO1 (cat. no. 62262S), HO-1 (cat. no. 1:5853S), JNK (cat.
no. 4672S), p-JNK (cat. no. 4668S), p38 (cat. no. 8690S), p-p38
(cat. no. 9216S), ERK (cat. no. 9102S), p-ERK (cat. no. 4377T), AKT
(cat. no. 4685S), p-AKT (cat. no. 13038S), HIF-1 (cat. no. 14179S),
VEGF (cat. no. 2463S), P62 (cat. no. 8025S) and cleaved caspase-3
(cat. no. 9664S) were purchased from Cell Signaling Technology,
Inc. (Danvers, MA, USA), the antibody against LC3 was obtained from
Sigma-Aldrich (cat. no. L8918; Merck KGaA), and the antibody
against P-Nrf2 (cat. no. ab76026) was purchased from Abcam (USA).
Subsequently, the blots were rinsed 3 times with PBS, incubated
with a goat anti-rat IgG (H+L) horseradish peroxidase-conjugated
secondary antibody (cat. no. SA00001-15; dilution, 1:1,000;
ProteinTech Group, Inc., Chicago, IL, USA) at 4°C overnight and
observed using an ECL reagents kit (KeyGEN BioTECH, Nanjing, China)
according to the protocol of the manufacturer. GAPDH served as the
reference. Results were analyzed using Image Lab software from
Bio-Rad Laboratories, Inc. (Hercules, CA, USA; v.4.1.0.0).
Experiments were performed in triplicate.
Detection of DNA damage using an
alkaline comet assay
DNA damage was detected using the alkaline Comet
Assay (11), using normal-(cat. no.
A9414) and low-melting point agarose (A9414; Sigma-Aldrich; Merck
KGaA). In brief, a slide was pre-coated with 150 µl 1%
normal-melting-point agarose and mounted on a coverslip for 20 min
at 4°C. Subsequently, 70 µl (containing 1×104 cells) 1%
low-melting-point agarose was spread on the agarose pre-coated
slide and then mounted using a coverslip for 20 min at 4°C. The
slide was immersed in 40 ml 176.55 g/l freshly prepared alkaline
lysis solution (2.5 M NaCl, 100 mM EDTA, 10 mM Tris, 34 mM
N-lauroylsarcosine sodium at pH 10.0–10.5 with freshly added 1%
Triton X-100) at 4°C overnight. Following rinsing with PBS, the
slide was placed in an electrophoresis chamber filled with freshly
prepared alkaline electrophoresis buffer (300 mM NaOH, 1 mM EDTA,
pH 13) for 20 min at 4°C in the dark and electrophoresed at 300 mA
and 25 V for 20 min. Subsequently, the slide was washed with 0.4 M
Tris buffer (pH 7.5) for 10 min and dried at room temperature for 1
h. The slide was stained with 5 µg/ml DAPI (Beyotime Institute of
Biotechnnology) for 15 min at room temperature, rinsed with cold
distilled water, and 4 fields of view were observed under a
fluorescence microscope at microscope, ×200. The comet formation
rate, or the comet quantity/total number of cells was used to
evaluate the degree of DNA damage.
In vivo therapeutic efficacy
Female BALB/c nude mice aged 5–6-weeks (16–17 g)
were obtained from the Peking University Health Science Center
(Laboratory Animals Science Department, Beijing, China) and kept in
an atmosphere of 21–24°C, with a relative humidity of 40–67%. Air
exchange was performed 14.4–17.2 times/h. Food and water were
provided ad libitum. The mice were injected subcutaneously
with 1×107 SKOV3 cells. Tumors reached 100
mm3 following ~14 days. Nude mice were administered an
intraperitoneal injection of 25 mg/kg thioridazine dissolved in
physiological saline into the abdominal cavity, and length (L, mm)
and width (W, mm) of the tumors were determined every once every
three days for 3 weeks, so that each mouse received 7 injections.
Tumor volume (V, mm3) was subsequently evaluated using
the following equation: V=(LxW2)/2. A total of 3 days
following the final injection, the nude mice were sacrificed via
cervical dislocation and the tumors were excised. All procedures
for animal experiments were approved by the Committee on the Use
and Care of Animals (Chongqing Medical University, Chongqing,
China) and performed in accordance with the institution's
guidelines.
Statistical analysis
Data are presented as the mean ± standard deviation.
Statistical analysis was performed using SPSS version 17.0 (SPSS
Inc., Chicago, IL, USA) and comparison of groups was analyzed by
the Student's t-test. P<0.05 was considered to indicate a
statistically significant difference.
Results
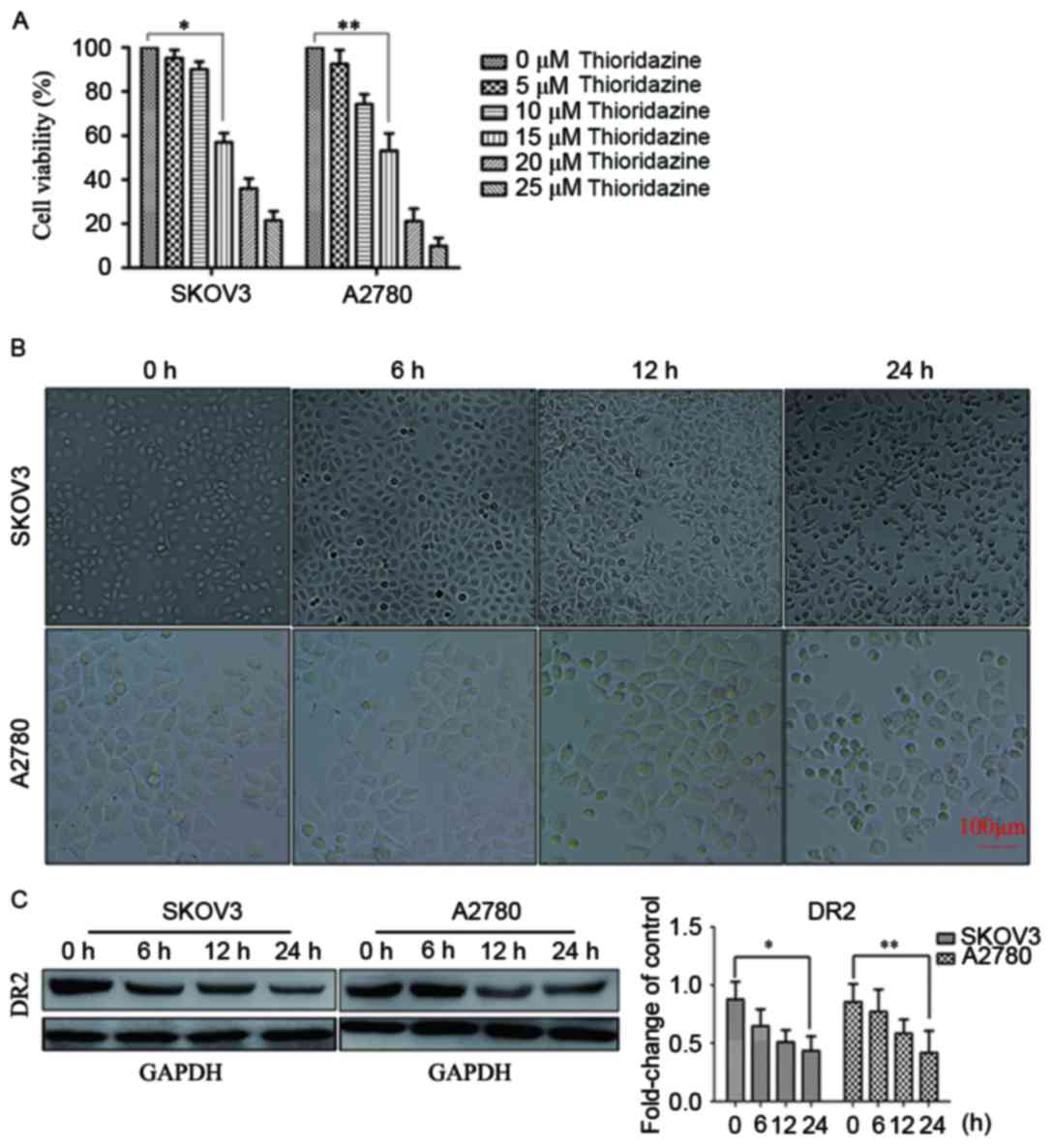
Thioridazine blocks DR2 and suppresses
cell proliferation
SKOV3 and A2780 cells were exposed to thioridazine
for 24 h. The concentration that produced significant cell
inhibition was determined to be 15 µM (P<0.05; Fig. 1A). The cell viability values for each
cell type at each concentration of thioridazine were as follows:
SKOV3: 5 µm, 95.24±3.64%; 10 µm, 90.7±3.48%; 15 µm, 57.00±4.13%; 20
µm, 36.04±4.52%; 25 µm, 21.45±4.23%; and A2780: 5 µm, 92.51±6.36%;
10 µm, 74.21±4.54%; 15 µm, 53.22±7.81%; 20 µm, 21.17±5.74%; 25 µm,
9.97±3.62%. The morphological changes observed included cellular
rounding, vacuolation and detachment (Fig. 1B). Western blotting indicated that the
expression of DR2 in SKOV3 and A2780 was downregulated following
treatment with 15 µM thioridazine (Fig.
1C). These results suggested that the inhibition of cell
proliferation by thioridazine may be associated with the
suppression of DR2.
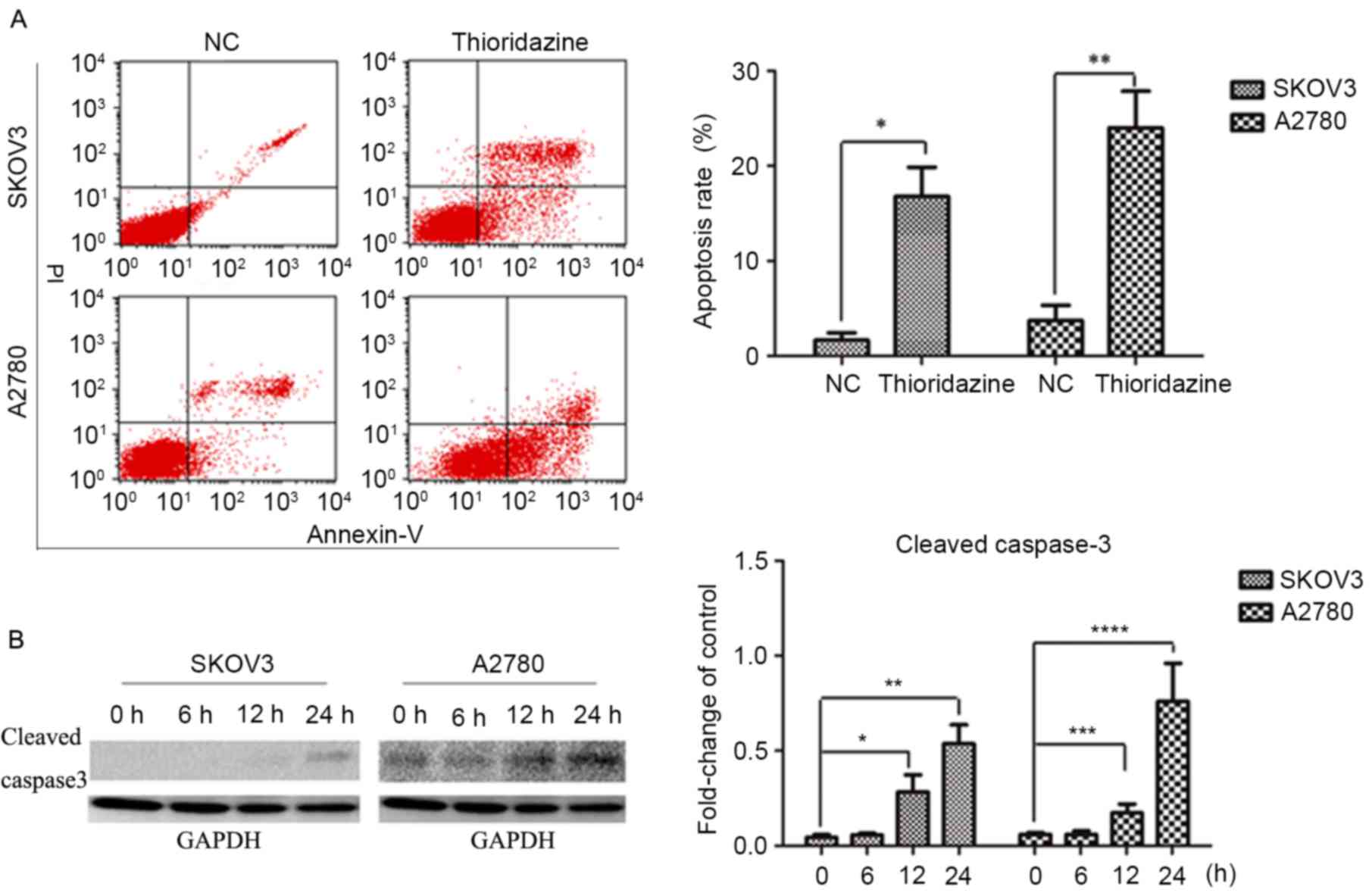
Thioridazine induced apoptosis
In order to investigate the mechanisms underlying
the cytocidal effects of thioridazine, SKOV3 and A2780 cells were
treated with 15 µM thioridazine for various durations (0, 6, 12 or
24 h). Flow cytometry analysis revealed a higher percentage of
apoptotic cells following a 24-h thioridazine treatment (Fig. 2A). Apoptosis was confirmed with the
activation of caspase 3 and the subsequent production of cleaved
caspase-3, which was detected by western blot analysis. The
expression levels of cleaved caspase-3 (a marker of apoptosis) were
increased after a 24-h treatment (Fig.
2B).
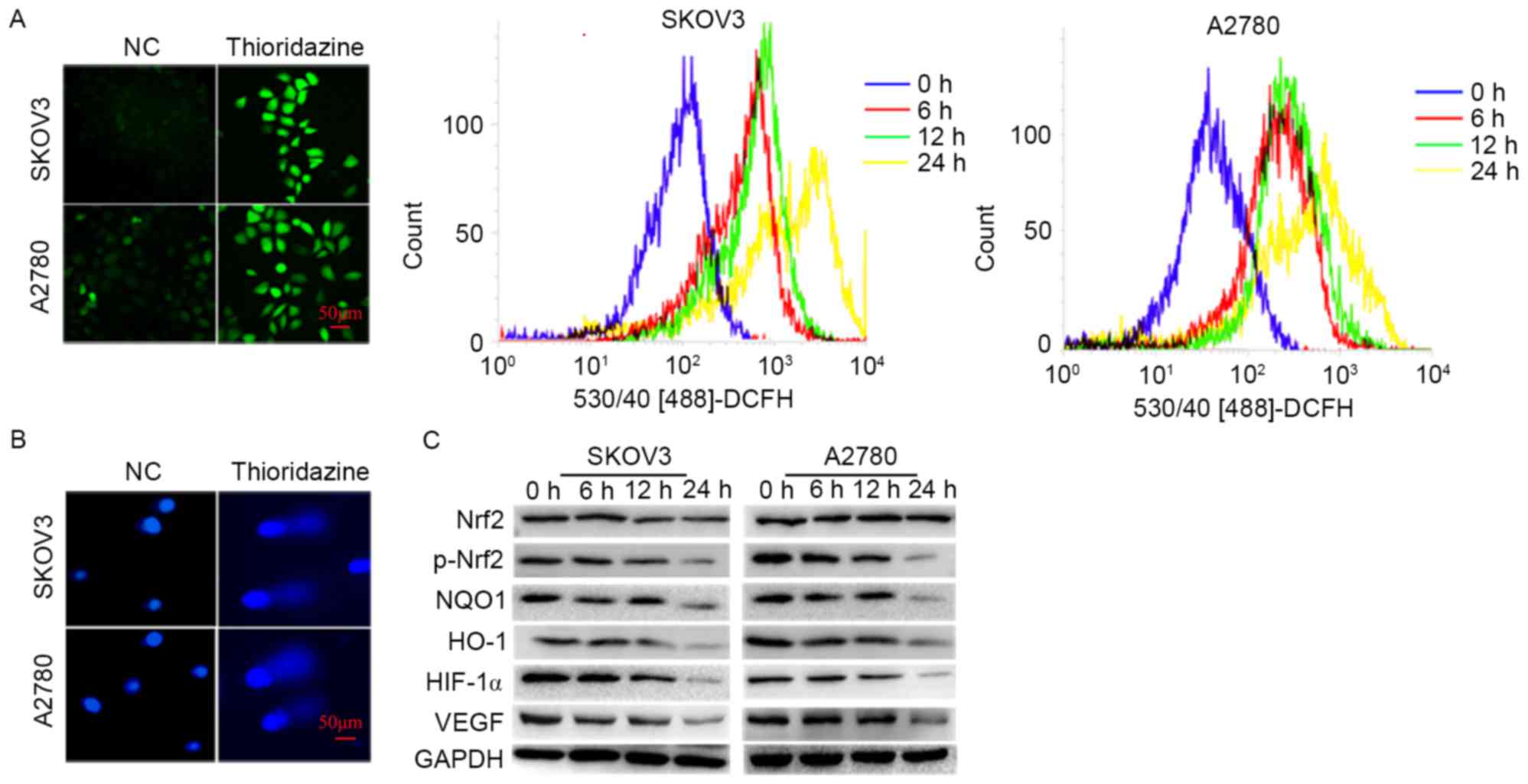
Thioridazine induces ROS production
and DNA damage
In order to investigate the underlying mechanisms of
thioridazine induced apoptosis, the cellular ROS, DNA damage and
protein expression associated with oxidative stress were evaluated.
The ROS levels and comet formation rates (SKOV3: 8.33±1.53 vs.
30.33±3.21%; P=0.002; A2780: 5.67±2.08%; P=0.002) were increased
following 15 µm thioridazine treatment for 24 h (Fig. 3A and B). The expression levels of
p-Nrf2, a pivotal transcriptional factor involved in the cellular
responses to oxidative stress, and its downstream targets, HO-1,
NQO1 and HIF-1α, were significantly decreased (P<0.05). The
expression level of VEGF, a pivotal enabling factor for tumor
angiogenesis, was reduced. VEGF is regulated by HIF-1α (Fig. 3C) (10).
Thioridazine induces autophagy
To investigate whether thioridazine induces
autophagy, the immunofluorescence assay was used to detect the
localization of LC3. LC3-green punctate were observed in 15 µm
thioridazine-treated cells (Fig. 4A).
The expression levels of LC3 (a marker of autophagy) and P62 (a
substrate of autophagy) were evaluated. LC3-II was significantly
upregulated in a dose- and time-dependent manner in the two cell
lines following treatment with 0, 5, 10 and 15 µm thioridazine for
0, 6, 12 and 24 h (Fig. 4B and C;
P<0.05). Notably, autophagy occurred with a relatively lower
drug concentration (5 µM) and earlier drug processing time (6 h).
To investigate the pro-survival or pro-cell death mechanisms
underlying thioridazine-induced autophagy, SKOV3 and A2780 cells
were pre-treated with 5 mM 3-MA for 2 h to inhibit the formation of
autophagosomes, followed by a 24-h treatment with thioridazine
(Fig. 4D). The apoptosis rate (SKOV3:
14.43±1.76%; A2780; 29.71±1.98%) was increased in the 3-MA
pretreated group (SKOV3: 36.24±3.93%, *P=0.004; A2780: 44.35±3.78%,
**P=0.021). These results indicated that thioridazine induces
autophagy, which may be a pro-survival mechanism associated with
thioridazine-induced cytotoxicity in ovarian cancer cells.
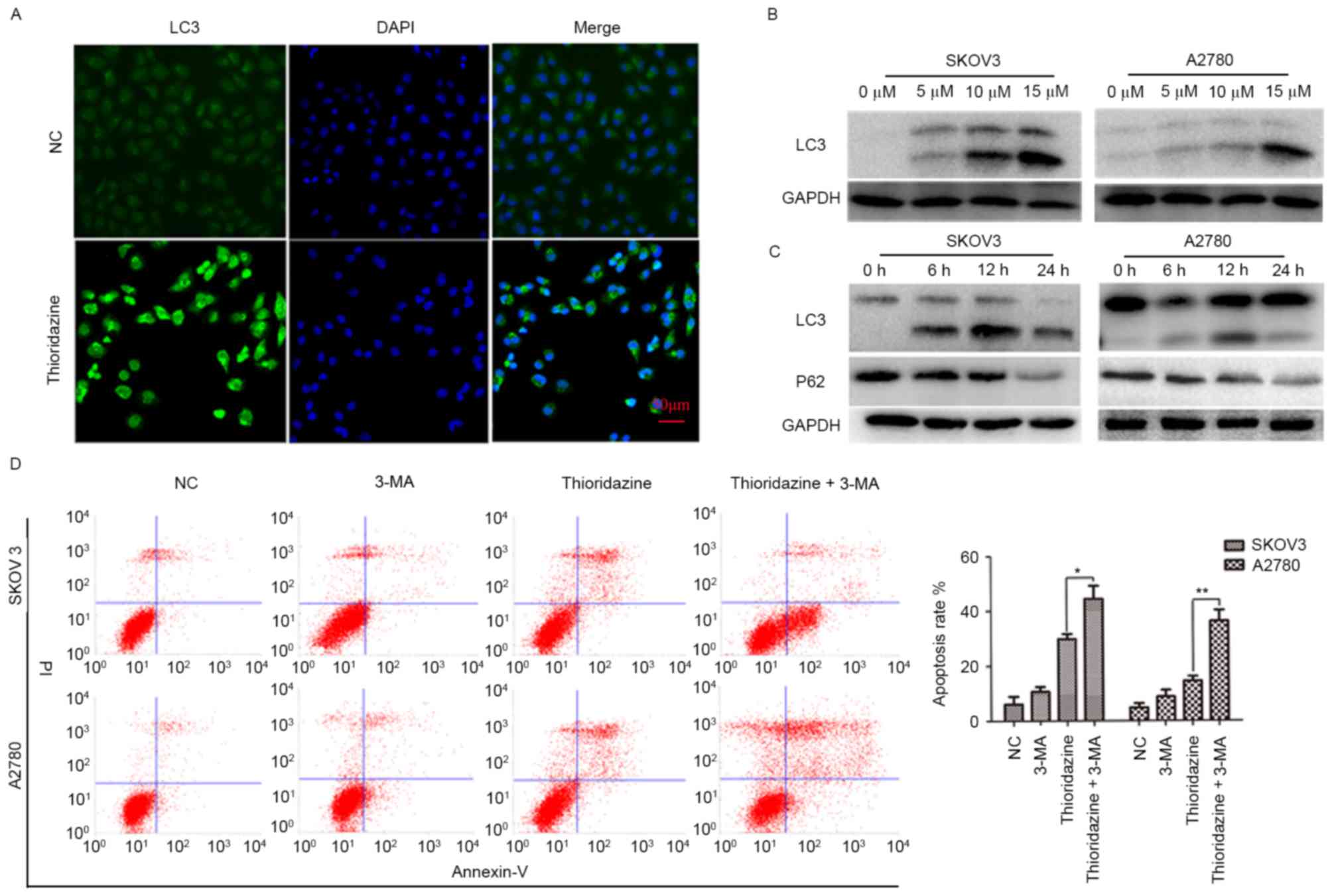 | Figure 4.Thioridazine induced autophagy in
ovarian cancer cells. (A) LC3 localization following treatment with
thioridazine for 12 h (scale bar, 50 µm). (B) Representative
western blot analysis demonstrating the expression levels of LC3
following treatment with thioridazine (0, 5, 10 and 15 µM) for 24
h. GAPDH was used as an internal control. (C) Representative
western blot analysis demonstrating the expression levels of LC3
and P62 following treatment with 15 µM thioridazine for various
lengths of time (0, 6, 12 and 24 h). GAPDH was used as an internal
control. (D) Ovarian cancer cells were pre-treated with 5 mM 3-MA
for 2 h, and then treated with 15 µM thioridazine for 24 h. Annexin
V/PI double staining was used to analyze the effect of thioridazine
on ovarian cancer cells. Data are representative of the results of
two independent experiments. *P=0.004, **P=0.021 compared with the
control group. 3-MA, 3-methyl adenine; NC, negative control; PI,
protease inhibitor; LC3, light chain 3. |
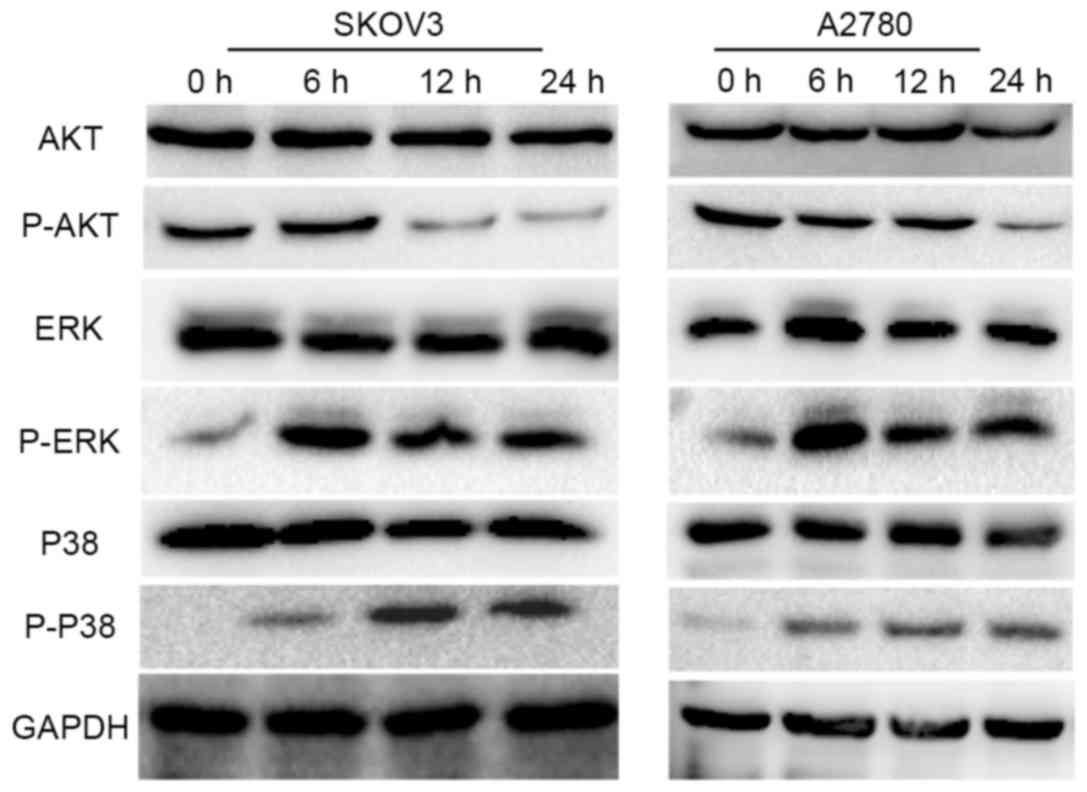
Thioridazine induces autophagy via the
AKT/ERK signaling pathway
It has previously been demonstrated that DR2
expression is associated with the phosphorylation/activation of AKT
and ERK [members of the mitogen activated protein kinase (MAPK)
family], which serve an essential role in the proliferation,
apoptosis and autophagy of cancer cells (12–14). The
activity of ERK and AKT was evaluated by western blot analysis,
which revealed that p-AKT was downregulated (Fig. 4A) and that the expression of p-ERK was
upregulated following treatment with 15 µM thioridazine.
Furthermore, the activity of P38 and JNK, which are also members of
the MAPK family and share a 40–50% sequence identity to ERK, was
analyzed. Consistent with the expression pattern of p-ERK, the
expression level of p-P38 was also increased following treatment
with 15 µM thioridazine (Fig. 5).
However, the expression level of p-JNK was not detected (data not
presented).
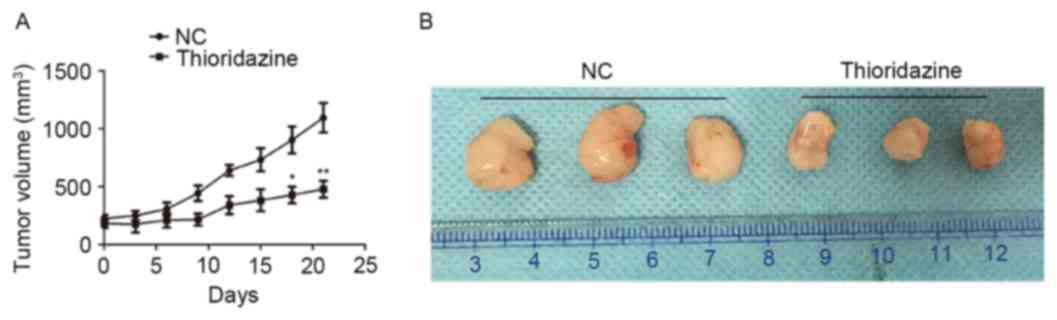
Thioridazine inhibits the growth of
SKOV3 xenografts in nude mice
The effect of thioridazine on the growth of ovarian
cancer cells in vivo was investigated using a xenograft
mouse model. The tumor size was smaller in the thioridazine
treatment group (mice treated with 25 mg/kg for 3 weeks) on the day
21 (tumor volume: 1094.74±127.31 mm3) compared with the
control group (tumor volume: 478.07±72.63 mm3; P=0.004;
Fig. 6A). The morphology of tumors
with or without treatment of 25 mg/kg thioridazine on day 21 is
demonstrated in Fig. 6B.
Discussion
Treatment with the DR2 blocker, thioridazine,
inhibited the proliferation of ovarian cancer cells in vivo
and in vitro. The concentration of 15 µM is within the range
of tolerability in human patients (15,16). This
suggests that thioridazine may be a candidate drug for ovarian
cancer treatment. In the present study, thioridazine was observed
to induce apoptosis and autophagy. Apoptosis is the primary
mechanism underlying the action of anticancer drugs (17). Autophagy, the intracellular
degradation of cytoplasmic components, serves an intricate and
paradoxical role in tumor chemotherapy (18,19). The
results of the present study indicated that autophagy may act as a
pro-survival mechanism in the anticancer action of thioridazine.
Conversely, a previous study reported that it was autophagy, not
apoptosis, induced by thioridazine in glioblastoma cells that
appeared to be the principal pro-cell death mechanism (20). In certain cell systems with apoptotic
dysfunction, autophagy has acted as a cell death mechanism
(21). The role of autophagy in
apoptotic competent cells may depend on the type of stimulus
(22). In other cases, the role of
autophagy in initiating cell death has been identified: In response
to hypoxia, ER stress, chemotherapeutic, virus infection and toxin
(23–26). The diverse role of autophagy in
different cells appears complex, and further research is
required.
Thioridazine induced a higher level of cellular ROS
and DNA damage in comparison with the control group.
Simultaneously, antioxidative stress-associated proteins were
downregulated, including p-Nrf2 and its downstream targets, HO-1,
NQO1 and HIF1α. This inhibition of the expression of certain
cellular antioxidant stress-associated proteins, and consequent
increase in intracellular levels of ROS, may explain the mechanism
by which thioridazine induces apoptosis and autophagy. HIF1α binds
to the promoter of VEGF (27,28), which activated the proliferation and
migration of endothelial cells during microvessel formation
(29). Consistent with the expression
of HIF1α, VEGF was downregulated. Inhibition of angiogenesis may
result in anticancer efficacy (30).
Overexpression or hyper-activation of Nrf2 may participate in
tumorigenesis. Knockdown of Nrf2 may reverse cisplatin resistance
in ovarian cancer (31); the present
study demonstrated that thioridazine inhibited the expression of
p-Nrf2, and suggested that it may be an potential adjuvant for
cisplatin therapy. A previous study demonstrated that the
co-delivery of thioridazine and doxorubicin (DOX) using polymeric
micelles eradicated cancer cells and DOX-resistant cancer stem
cells (32).
DR may be associated with cancer chemotherapy
(33,34) Thioridazine and its analogs have
exhibited antitumor effects in melanoma (35). The development of certain small
molecules with a high specificity for DR2 or knockdown of DR2 may
confer an improved therapeutic prognosis (34,36).
However, the DR2 agonist bromocriptin has inhibited proliferation
in MCF-7 breast cancer cells (37).
Thus, a strategy specifically targeting DR2 must be developed
dependent on the type of cancer.
In conclusion, DR2 may be a therapeutic target for
ovarian cancer. The DR2 blocker thioridazine exhibited anticancer
effects in vivo and in vitro by inducing oxidative
stress and apoptosis, inhibiting tumor angiogenesis and interacting
with the AKT and ERK signaling pathways. Thus, thioridazine may be
a promising candidate drug for ovarian cancer.
Acknowledgements
The present study was supported by the Natural
Science Foundation of Chongqing (grant no. CSTC 2012JJB10030) and
The Natural Science Foundation of China (grant no. 81172492).
References
|
1
|
Shih KK and Chi DS: Maximal cytoreductive
effort in epithelial ovarian cancer surgery. J Gynecol Oncol.
21:75–80. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Dalton SO, Mellemkjaer L, Thomassen L,
Mortensen PB and Johansen C: Risk for cancer in a cohort of
patients hospitalized for schizophrenia in Denmark, 1969–1993.
Schizophr Res. 75:315–324. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mortensen PB: The incidence of cancer in
schizophrenic patients. J Epidemiol Community Health. 43:43–47.
1989. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Driver JA, Logroscino G, Buring JE,
Gaziano JM and Kurth T: A prospective cohort study of cancer
incidence following the diagnosis of Parkinson's disease. Cancer
Epidemiol Biomarkers Prev. 16:1260–1265. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sachlos E, Risueño RM, Laronde S,
Shapovalova Z, Lee JH, Russell J, Malig M, McNicol JD, Fiebig-Comyn
A, Graham M, et al: Identification of drugs including a dopamine
receptor antagonist that selectively target cancer stem cells.
Cell. 149:1284–1297. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kilts CD, Knight DL, Mailman RB, Widerlöv
E and Breese GR: Effects of thioridazine and its metabolites on
dopaminergic function: Drug metabolism as a determinant of the
antidopaminergic actions of thioridazine. J Pharmacol Exp Ther.
231:334–342. 1984.PubMed/NCBI
|
|
7
|
Yin T, He S, Shen G, Ye T, Guo F and Wang
Y: Dopamine receptor antagonist thioridazine inhibits tumor growth
in a murine breast cancer model. Mol Med Rep. 12:4103–4108. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lu M, Li J, Luo Z, Zhang S, Xue S, Wang K,
Shi Y, Zhang C, Chen H and Li Z: Roles of dopamine receptors and
their antagonist thioridazine in hepatoma metastasis. OncoTargets
Ther. 8:1543–1552. 2015.
|
|
9
|
Mao M, Yu T, Hu J and Hu L: Dopamine D2
receptor blocker thioridazine induces cell death in human uterine
cervical carcinoma cell line SiHa. J Obstet Gynaecol Res.
41:1240–1245. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Moreno-Smith M, Lee SJ, Lu C, Nagaraja AS,
He G, Rupaimoole R, Han HD, Jennings NB, Roh JW, Nishimura M, et
al: Biologic effects of dopamine on tumor vasculature in ovarian
carcinoma. Neoplasia. 15:502–510. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Olive PL and Banáth JP: The comet assay: A
method to measure DNA damage in individual cells. Nat Protoc.
1:23–29. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pozzi L, Håkansson K, Usiello A, Borgkvist
A, Lindskog M, Greengard P and Fisone G: Opposite regulation by
typical and atypical anti-psychotics of ERK1/2, CREB and Elk-1
phosphorylation in mouse dorsal striatum. J Neurochem. 86:451–459.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rodrigues-Ferreira S and Nahmias C:
G-protein coupled receptors of the renin-angiotensin system: New
targets against breast cancer? Front Pharmacol. 6:242015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Berhow MT, Hiroi N and Nestler EJ:
Regulation of ERK (extracellular signal regulated kinase), part of
the neurotrophin signal transduction cascade, in the rat mesolimbic
dopamine system by chronic exposure to morphine or cocaine. J
Neurosci. 16:4707–4715. 1996.PubMed/NCBI
|
|
15
|
Nahata MC, Ford C and Ruymann FB:
Pharmacokinetics and safety of prochlorperazine in paediatric
patients receiving cancer chemotherapy. J Clin Pharm Ther.
17:121–123. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Morgan RJ Jr, Synold T, Carr BI, Doroshow
JH, Womack EP, Shibata S, Somlo G, Raschko J, Leong L, McNamara M,
et al: Continuous infusion prochlorperazine: Pharmacokinetics,
antiemetic efficacy, and feasibility of high-dose therapy. Cancer
Chemother Pharmacol. 47:327–332. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Adams JM: Ways of dying: Multiple pathways
to apoptosis. Genes Dev. 17:2481–2495. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wu HM, Jiang ZF, Ding PS, Shao LJ and Liu
RY: Hypoxia-induced autophagy mediates cisplatin resistance in lung
cancer cells. Sci Rep. 5:122912015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
García-Cano J, Ambroise G, Pascual-Serra
R, Carrión MC, Serrano-Oviedo L, Ortega-Muelas M, Cimas FJ, Sabater
S, Ruiz-Hidalgo MJ, Sanchez Perez I, et al: Exploiting the
potential of autophagy in cisplatin therapy: A new strategy to
overcome resistance. Oncotarget. 6:15551–15565. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cheng HW, Liang YH, Kuo YL, Chuu CP, Lin
CY, Lee MH, Wu AT, Yeh CT, Chen EI, Whang-Peng J, et al:
Identification of thioridazine, an antipsychotic drug, as an
antiglioblastoma and anticancer stem cell agent using public gene
expression data. Cell Death Dis. 6:e17532015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jin S and White E: Role of autophagy in
cancer: Management of metabolic stress. Autophagy. 3:28–31. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Boya P, González-Polo RA, Casares N,
Perfettini JL, Dessen P, Larochette N, Métivier D, Meley D,
Souquere S, Yoshimori T, et al: Inhibition of macroautophagy
triggers apoptosis. Mol Cell Biol. 25:1025–1040. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Daido S, Kanzawa T, Yamamoto A, Takeuchi
H, Kondo Y and Kondo S: Pivotal role of the cell death factor BNIP3
in ceramide-induced autophagic cell death in malignant glioma
cells. Cancer Res. 64:4286–4293. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kanzawa T, Kondo Y, Ito H, Kondo S and
Germano I: Induction of autophagic cell death in malignant glioma
cells by arsenic trioxide. Cancer Res. 63:2103–2108.
2003.PubMed/NCBI
|
|
25
|
Paglin S, Hollister T, Delohery T, Hackett
N, McMahill M, Sphicas E, Domingo D and Yahalom J: A novel response
of cancer cells to radiation involves autophagy and formation of
acidic vesicles. Cancer Res. 61:439–444. 2001.PubMed/NCBI
|
|
26
|
Yu L, Alva A, Su H, Dutt P, Freundt E,
Welsh S, Baehrecke EH and Lenardo MJ: Regulation of an ATG7-beclin
1 program of autophagic cell death by caspase-8. Science.
304:1500–1502. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yang Y, Cong H, Han C, Yue L, Dong H and
Liu J: 12-Deoxyphorbol 13-palmitate inhibits the expression of VEGF
and HIF-1α in MCF-7 cells by blocking the PI3K/Akt/mTOR signaling
pathway. Oncol Rep. 34:1755–1760. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ioannou M, Paraskeva E, Baxevanidou K,
Simos G, Papamichali R, Papacharalambous C, Samara M and Koukoulis
G: HIF-1alpha in colorectal carcinoma: Review of the literature. J
BUON. 20:680–689. 2015.PubMed/NCBI
|
|
29
|
Risau W: Mechanisms of angiogenesis.
Nature. 386:671–674. 1997. View
Article : Google Scholar : PubMed/NCBI
|
|
30
|
Park MS, Dong SM, Kim BR, Seo SH, Kang S,
Lee EJ, Lee SH and Rho SB: Thioridazine inhibits angiogenesis and
tumor growth by targeting the VEGFR-2/PI3K/mTOR pathway in ovarian
cancer xenografts. Oncotarget. 5:4929–4934. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bao LJ, Jaramillo MC, Zhang ZB, Zheng YX,
Yao M, Zhang DD and Yi XF: Nrf2 induces cisplatin resistance
through activation of autophagy in ovarian carcinoma. Int J Clin
Exp Pathol. 7:1502–1513. 2014.PubMed/NCBI
|
|
32
|
Ke XY, Lin Ng VW, Gao SJ, Tong YW, Hedrick
JL and Yang YY: Co-delivery of thioridazine and doxorubicin using
polymeric micelles for targeting both cancer cells and cancer stem
cells. Biomaterials. 35:1096–1108. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gabalec F, Beranek M, Netuka D, Masopust
V, Nahlovsky J, Cesak T, Marek J and Cap J: Dopamine 2 receptor
expression in various pathological types of clinically
non-functioning pituitary adenomas. Pituitary. 15:222–226. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Gatto F and Hofland LJ: The role of
somatostatin and dopamine D2 receptors in endocrine tumors. Endocr
Relat Cancer. 18:R233–R251. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gil-Ad I, Shtaif B, Levkovitz Y,
Nordenberg J, Taler M, Korov I and Weizman A: Phenothiazines induce
apoptosis in a B16 mouse melanoma cell line and attenuate in vivo
melanoma tumor growth. Oncol Rep. 15:107–112. 2006.PubMed/NCBI
|
|
36
|
Xu HN, Huang WD, Cai Y, Ding M, Gu JF, Wei
N, Sun LY, Cao X, Li HG, Zhang KJ, et al: HCCS1-armed,
quadruple-regulated oncolytic adenovirus specific for liver cancer
as a cancer targeting gene-viro-therapy strategy. Mol Cancer.
10:1332011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Sheikhpour M, Ahangari G, Sadeghizadeh M
and Deezagi A: A novel report of apoptosis in human lung carcinoma
cells using selective agonist of D2-like dopamine receptors: A new
approach for the treatment of human non-small cell lung cancer. Int
J Immunopathol Pharmacol. 26:393–402. 2013. View Article : Google Scholar : PubMed/NCBI
|