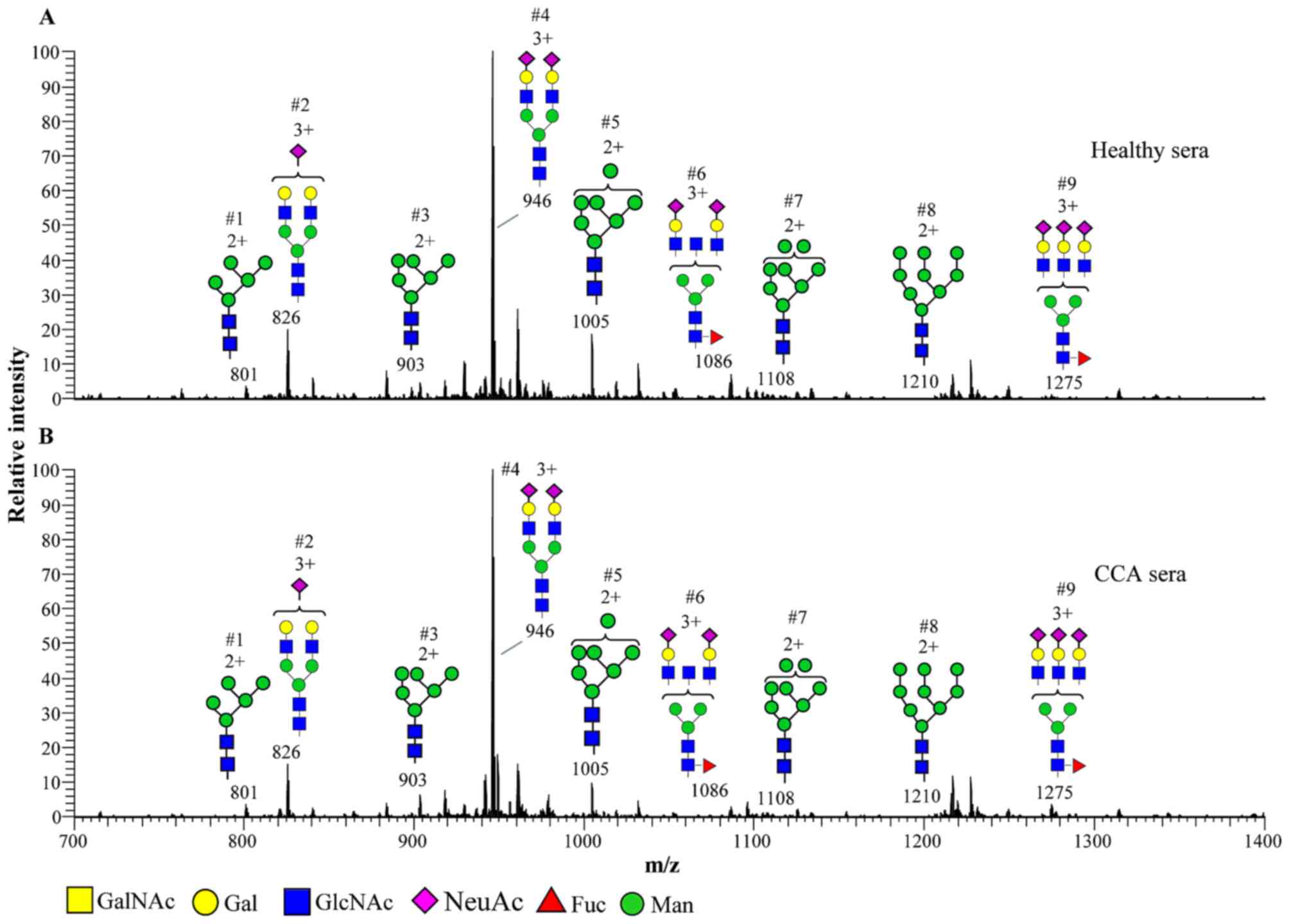
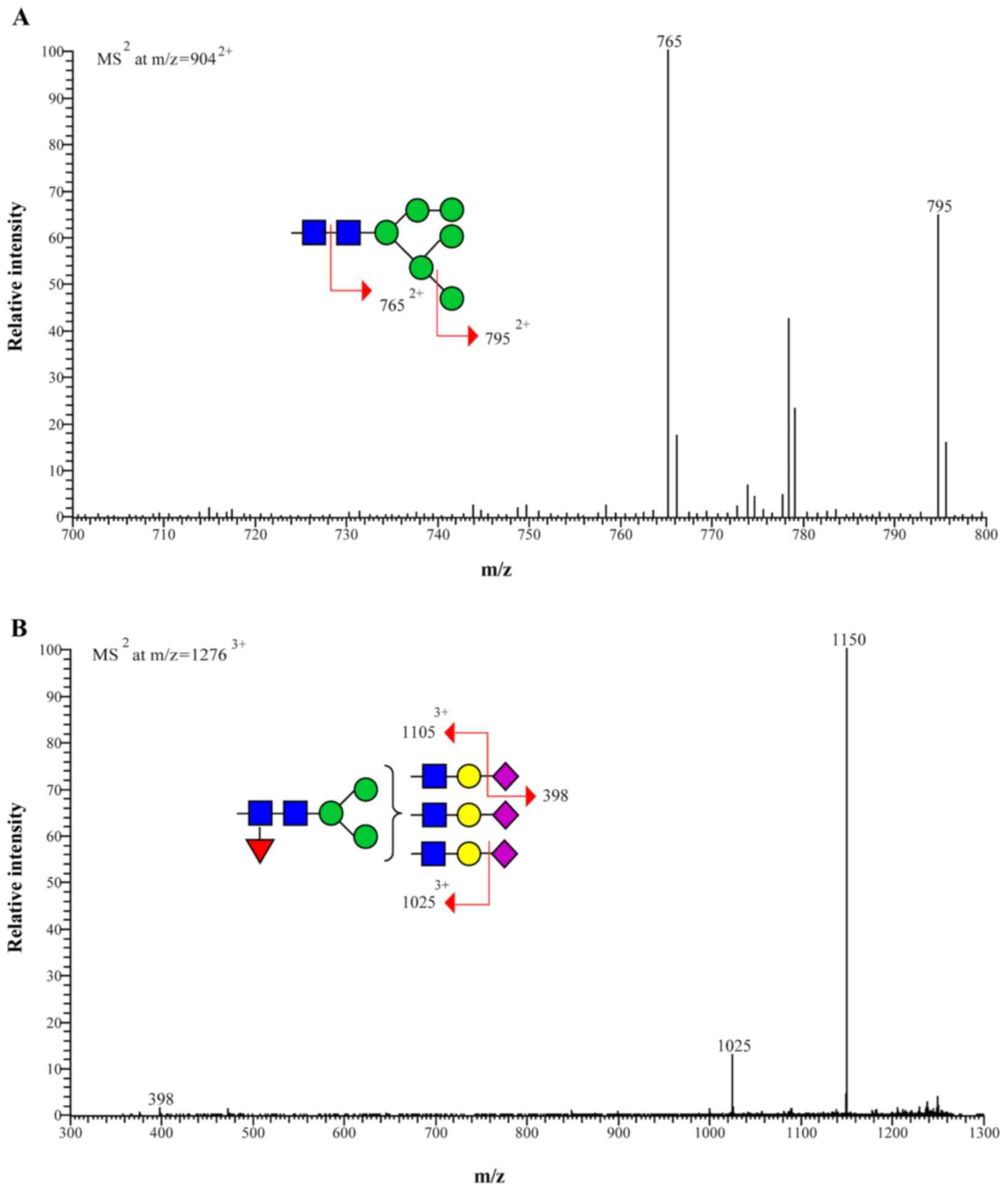
|
1
|
Sripa B: Pathobiology of opisthorchiasis:
An update. Acta Trop. 88:209–220. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Patel T: Increasing incidence and
mortality of primary intrahepatic cholangiocarcinoma in the United
States. Hepatology. 33:1353–1357. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shaib YH, Davila JA, McGlynn K and
El-Serag HB: Rising incidence of intrahepatic cholangiocarcinoma in
the United States: A true increase? J Hepatol. 40:472–477. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Apweiler R, Hermjakob H and Sharon N: On
the frequency of protein glycosylation, as deduced from analysis of
the SWISS-PROT database. Biochim Biophys Acta. 1473:4–8. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wong CH: Protein glycosylation: New
challenges and opportunities. J Org Chem. 70:4219–4225. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Varki A and Gagneux P: Biological
Functions of GlycansEssentials of Glycobiology. 3rd. Varki A,
Cummings RD, Esko JD, Stanley P, Hart GW, Aebi M, Darvill AG,
Kinoshita T, Packer NH, Prestegard JH, Schnaar RL and Seeberger PH:
Cold Spring Harbor (NY): 2015, View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kim EH and Misek DE: Glycoproteomics-based
identification of cancer biomarkers. Int J Proteomics.
2011:6019372011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Juntavee A, Sripa B, Pugkhem A, Khuntikeo
N and Wongkham S: Expression of sialyl Lewis(a) relates to poor
prognosis in cholangiocarcinoma. World J Gastroenterol. 11:249–254.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Silsirivanit A, Araki N, Wongkham C,
Pairojkul C, Narimatsu Y, Kuwahara K, Narimatsu H, Wongkham S and
Sakaguchi N: A novel serum carbohydrate marker on mucin 5AC: Values
for diagnostic and prognostic indicators for cholangiocarcinoma.
Cancer. 117:3393–3403. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sawanyawisuth K, Silsirivanit A, Kunlabut
K, Tantapotinan N, Vaeteewoottacharn K and Wongkham S: A novel
carbohydrate antigen expression during development of opisthorchis
viverrini-associated cholangiocarcinoma in golden hamster: A
potential marker for early diagnosis. Parasitol Int. 61:151–154.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Indramanee S, Silsirivanit A, Pairojkul C,
Wongkham C and Wongkham S: Aberrant glycosylation in
cholangiocarcinoma demonstrated by lectin-histochemistry. Asian Pac
J Cancer Prev. 13 Suppl:S119–S124. 2012.
|
|
12
|
Phoomak C, Silsirivanit A, Wongkham C,
Sripa B, Puapairoj A and Wongkham S: Overexpression of
O-GlcNAc-transferase associates with aggressiveness of mass-forming
cholangiocarcinoma. Asian Pac J Cancer Prev. 13 Suppl:S101–S105.
2012.
|
|
13
|
Silsirivanit A, Araki N, Wongkham C,
Vaeteewoottacharn K, Pairojkul C, Kuwahara K, Narimatsu Y, Sawaki
H, Narimatsu H, Okada S, et al: CA-S27: A novel Lewis a associated
carbohydrate epitope is diagnostic and prognostic for
cholangiocarcinoma. Cancer Sci. 104:1278–1284. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Matsuda A, Kuno A, Nakagawa T, Ikehara Y,
Irimura T, Yamamoto M, Nakanuma Y, Miyoshi E, Nakamori S, Nakanishi
H, et al: Lectin microarray-based sero-biomarker verification
targeting aberrant O-Linked glycosylation on mucin 1. Anal Chem.
87:7274–7281. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Talabnin K, Talabnin C, Ishihara M, Azadi
P, Wongkham S and Sripa B: Differential expression of
O-glycoprotein glycans in cholangiocarcinoma cell lines. Asian Pac
J Cancer Prev. 17:691–695. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Talabnin K, Aoki K, Saichua P, Wongkham S,
Kaewkes S, Boons GJ, Sripa B and Tiemeyer M: Stage-specific
expression and antigenicity of glycoprotein glycans isolated from
the human liver fluke, Opisthorchis viverrini. Int J Parasitol.
43:37–50. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Aoki K, Perlman M, Lim JM, Cantu R, Wells
L and Tiemeyer M: Dynamic developmental elaboration of N-linked
glycan complexity in the Drosophila melanogaster embryo. J Biol
Chem. 282:9127–9142. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Anumula KR and Taylor PB: A comprehensive
procedure for preparation of partially methylated alditol acetates
from glycoprotein carbohydrates. Anal Biochem. 203:101–108. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Domon B and Costello CE: Structure
elucidation of glycosphingolipids and gangliosides using
high-performance tandem mass spectrometry. Biochemistry.
27:1534–1543. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liu X, Nie H, Zhang Y, Yao Y, Maitikabili
A, Qu Y, Shi S, Chen C and Li Y: Cell surface-specific N-glycan
profiling in breast cancer. PLoS One. 8:e727042013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
de Leoz ML, Young LJ, An HJ, Kronewitter
SR, Kim J, Miyamoto S, Borowsky AD, Chew HK and Lebrilla CB:
High-mannose glycans are elevated during breast cancer progression.
Mol Cell Proteomics. 10:M110.0027172011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sethi MK, Thaysen-Andersen M, Smith JT,
Baker MS, Packer NH, Hancock WS and Fanayan S: Comparative N-glycan
profiling of colorectal cancer cell lines reveals unique bisecting
GlcNAc and alpha-2,3-linked sialic acid determinants are associated
with membrane proteins of the more metastatic/aggressive cell
lines. J Proteome Res. 13:277–288. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Nakagawa T, Miyoshi E, Yakushijin T,
Hiramatsu N, Igura T, Hayashi N, Taniguchi N and Kondo A: Glycomic
analysis of alpha-fetoprotein L3 in hepatoma cell lines and
hepatocellular carcinoma patients. J Proteome Res. 7:2222–2233.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Schwedler C, Kaup M, Weiz S, Hoppe M,
Braicu EI, Sehouli J, Hoppe B, Tauber R, Berger M and Blanchard V:
Identification of 34 N-glycan isomers in human serum by capillary
electrophoresis coupled with laser-induced fluorescence allows
improving glycan biomarker discovery. Anal Bioanal Chem.
406:7185–7193. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Dennis JW, Laferté S, Waghorne C, Breitman
ML and Kerbel RS: Beta 1–6 branching of Asn-linked oligosaccharides
is directly associated with metastasis. Science. 236:582–585. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Taniguchi N and Kizuka Y: Glycans and
cancer: Role of N-glycans in cancer biomarker, progression and
metastasis and therapeutics. Adv Cancer Res. 126:11–51. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kronewitter SR, De Leoz ML, Strum JS, An
HJ, Dimapasoc LM, Guerrero A, Miyamoto S, Lebrilla CB and
Leiserowitz GS: The glycolyzer: Automated glycan annotation
software for high performance mass spectrometry and its application
to ovarian cancer glycan biomarker discovery. Proteomics.
12:2523–2538. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ozcan S, Barkauskas DA, Renee Ruhaak L,
Torres J, Cooke CL, An HJ, Hua S, Williams CC, Dimapasoc LM, Han
Kim J, et al: Serum glycan signatures of gastric cancer. Cancer
Prev Res (Phila). 7:226–235. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Dall'Olio F, Malagolini N, Trinchera M and
Chiricolo M: Sialosignaling: Sialyltransferases as engines of
self-fueling loops in cancer progression. Biochim Biophys Acta.
1840:2752–2764. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Vanhooren V, Laroy W, Libert C and Chen C:
N-glycan profiling in the study of human aging. Biogerontology.
9:351–356. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ding N, Nie H, Sun X, Sun W, Qu Y, Liu X,
Yao Y, Liang X, Chen CC and Li Y: Human serum N-glycan profiles are
age and sex dependent. Age Ageing. 40:568–575. 2011. View Article : Google Scholar : PubMed/NCBI
|