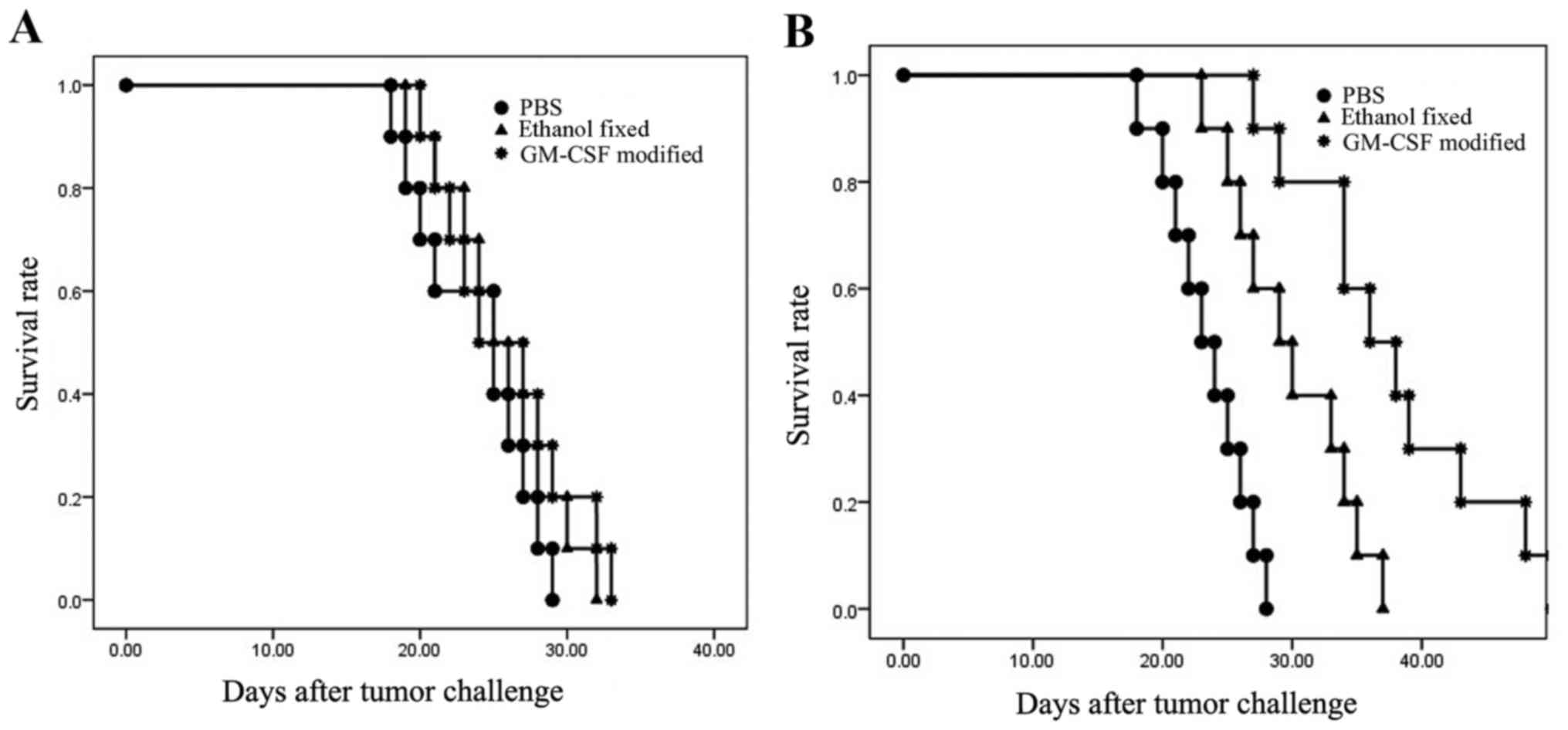
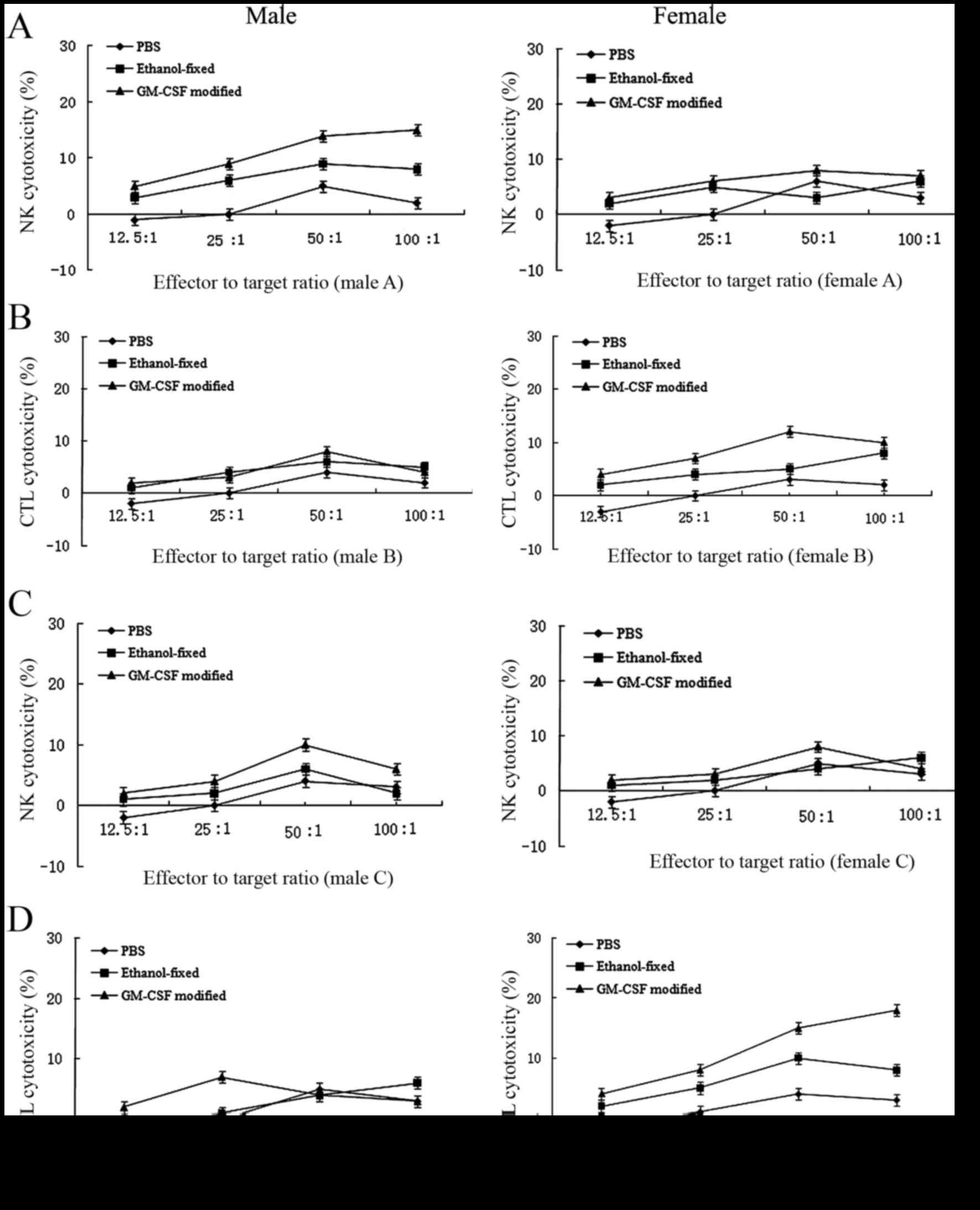
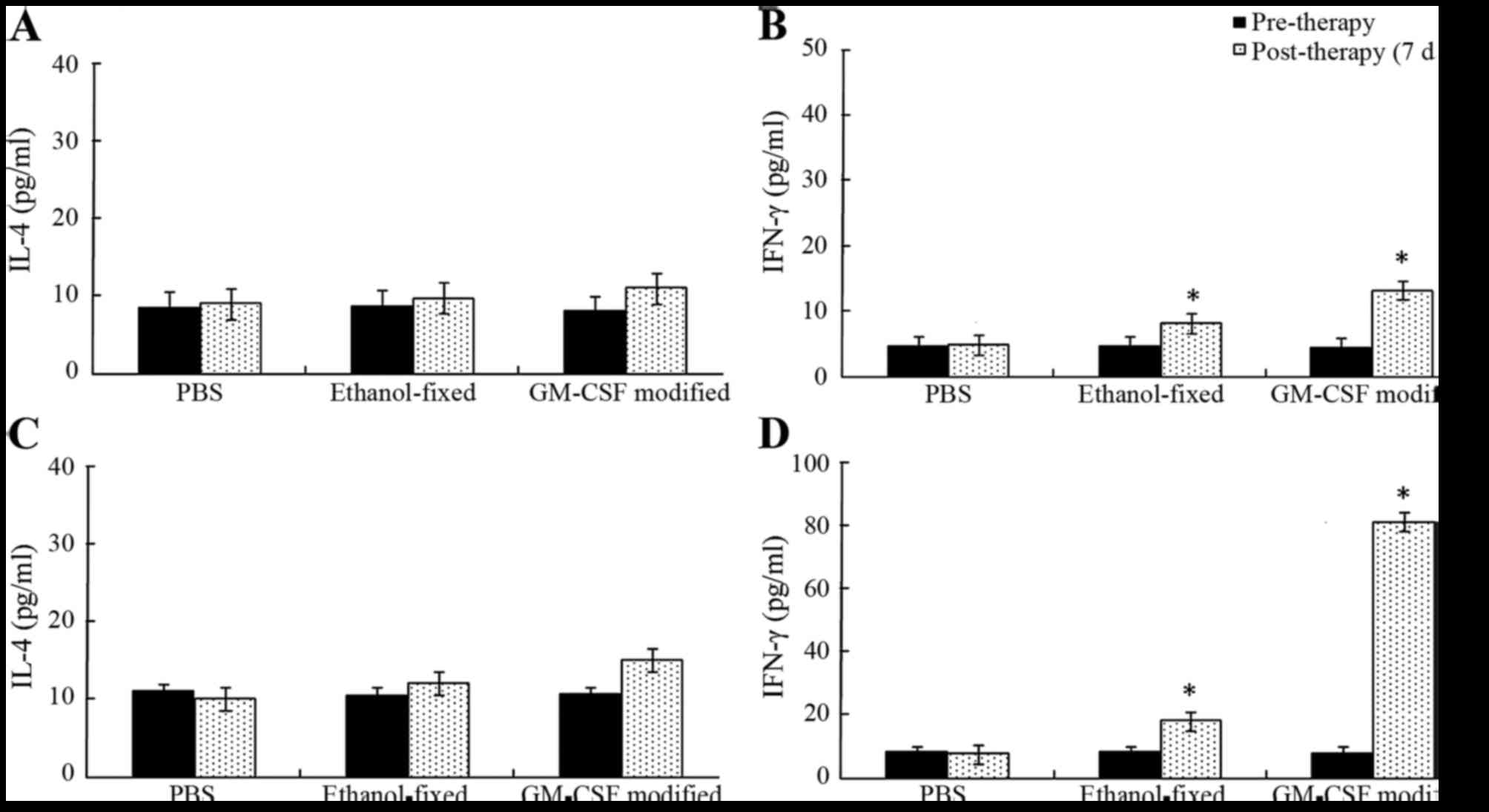
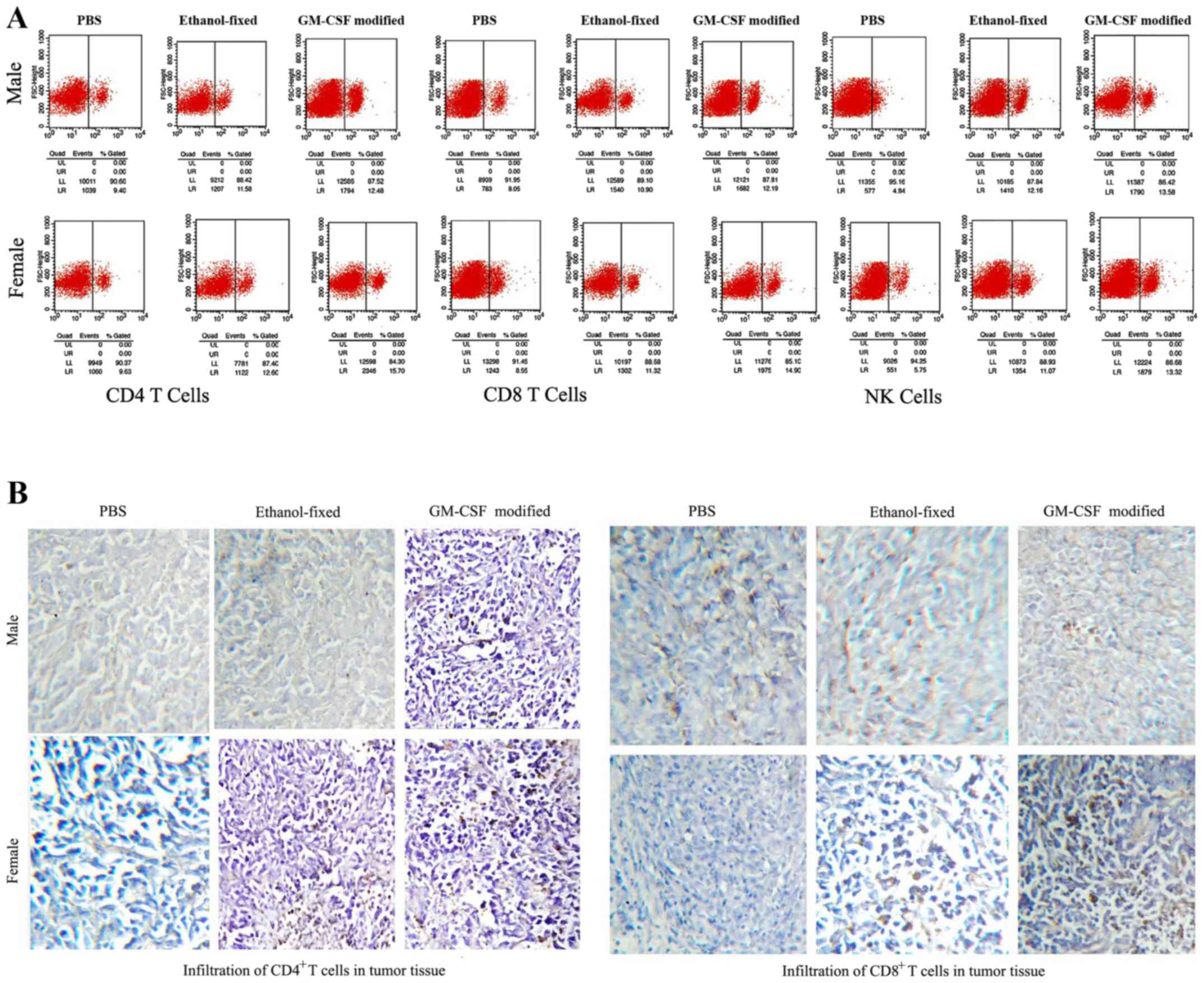
|
1
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Crawford ED, Blumenstein BA, Goodman PJ,
Davis MA, Eisenberger MA, McLeod DG, Spaulding JT, Benson R and
Dorr FA: Leuprolide with and without flutamide in advanced prostate
cancer. Cancer. 66(5 Suppl): S1039–S1044. 1990. View Article : Google Scholar
|
|
3
|
Quinn DI, Tangen CM, Hussain M, Lara PN
Jr, Goldkorn A, Moinpour CM, Garzotto MG, Mack PC, Carducci MA,
Monk JP, et al: Docetaxel and atrasentan versus docetaxel and
placebo for men with advanced castration-resistant prostate cancer
(SWOG S0421): A randomised phase 3 trial. Lancet Oncol. 14:893–900.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Armstrong AJ and Carducci MA: Chemotherapy
for advanced prostate cancer: Results of new clinical trials and
future studies. Curr Oncol Rep. 7:220–227. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Haigh PI, Difronzo LA, Gammon G and Morton
DL: Vaccine therapy for patients with melanoma. Oncology (Williston
Park). 13:1561–1574. 1999.PubMed/NCBI
|
|
6
|
Li H, Jiang HJ, Ma MQ, Wei F, An XM and
Ren XB: Vaccination with allogeneic GM-CSF gene-modified lung
cancer cells: Antitumor activity comparing with that induced by
autologous vaccine. Cancer Biother Radiopharm. 22:790–798. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Obata C, Zhang M, Moroi Y, Hisaeda H,
Tanaka K, Murata S, Furue M and Himeno K: Formalin-fixed tumor
cells effectively induce antitumor immunity both in prophylactic
and therapeutic conditions. J Dermatol Sci. 34:209–219. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yin W, He Q, Hu Z, Chen Z, Qifeng M,
Zhichun S, Zhihui Q, Xiaoxia N, Li J and Gao J: A novel therapeutic
vaccine of GM-CSF/TNFalpha surface-modified RM-1 cells against the
orthotopic prostatic cancer. Vaccine. 28:4937–4944. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Suckow MA, Wolter WR and Pollard M:
Prevention of de novo prostate cancer by immunization with
tumor-derived vaccines. Cancer Immunol Immunother. 54:571–576.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kotera Y, Shimizu K and Mulé JJ:
Comparative analysis of necrotic and apoptotic tumor cells as a
source of antigen(s) in dendritic cell-based immunization. Cancer
Res. 61:8105–8109. 2001.PubMed/NCBI
|
|
11
|
Ahram M, Flaig MJ, Gillespie JW, Duray PH,
Linehan WM, Ornstein DK, Niu S, Zhao Y, Petricoin EF III and
Emmert-Buck MR: Evaluation of ethanol-fixed, paraffin-embedded
tissues for proteomic applications. Proteomics. 3:413–421. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Baley PA, Yoshida K, Qian W, Sehgal I and
Thompson TC: Progression to androgen insensitivity in a novel in
vitro mouse model for prostate cancer. J Steroid Biochem Mol Biol.
52:403–413. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Griffith TS, Kawakita M, Tian J, Ritchey
J, Tartaglia J, Sehgal I, Thompson TC, Zhao W and Ratliff TL:
Inhibition of murine prostate tumor growth and activation of
immunoregulatory cells with recombinant canarypox viruses. J Natl
Cancer Inst. 93:998–1007. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Labarthe MC, Theocharous P, Russell N,
Todryk S, Bangma C, Thraves P, Dalgleish AG and Whelan MA: A novel
murine model of allogeneic vaccination against prostate cancer.
Cancer Immunol Immunother. 57:453–465. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
He Q, Li J, Yin W, Song Z, Zhang Z, Yi T,
Tang J, Wu D, Lu Y, Wang Z, et al: Low-dose paclitaxel enhances the
anti-tumor efficacy of GM-CSF surface-modified whole-tumor-cell
vaccine in mouse model of prostate cancer. Cancer Immunol
Immunother. 60:715–730. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cooper MD and Alder MN: The evolution of
adaptive immune systems. Cell. 124:815–822. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li M, Davey GM, Sutherland RM, Kurts C,
Lew AM, Hirst C, Carbone FR and Heath WR: Cell-associated ovalbumin
is cross-presented much more efficiently than soluble ovalbumin in
vivo. J Immunol. 166:6099–6103. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gallucci S, Lolkema M and Matzinger P:
Natural adjuvants: Endogenous activators of dendritic cells. Nat
Med. 5:1249–1255. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sauter B, Albert ML, Francisco L, Larsson
M, Somersan S and Bhardwaj N: Consequences of cell death: Exposure
to necrotic tumor cells, but not primary tissue cells or apoptotic
cells, induces the maturation of immunostimulatory dendritic cells.
J Exp Med. 191:423–434. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Grant JF, Iwasawa T, Sinn HW, Siemens DR,
Griffith TS, Takacs EB and Ratliff TL: Induction of protective
immunity to RM-1 prostate cancer cells with
ALVAC-IL-2/IL-12/TNF-alpha combination therapy. Int J Cancer.
119:2632–2641. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Carbone FR, Kurts C, Bennett SR, Miller JF
and Heath WR: Cross-presentation: A general mechanism for CTL
immunity and tolerance. Immunol Today. 19:368–373. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chhikara M, Huang H, Vlachaki MT, Zhu X,
Teh B, Chiu KJ, Woo S, Berner B, Smith EO, Oberg KC, et al:
Enhanced therapeutic effect of HSV-tk+GCV gene therapy and ionizing
radiation for prostate cancer. Mol Ther. 3:536–542. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Schroten-Loef C, de Ridder CM, Reneman S,
Crezee M, Dalgleish A, Todryk SM, Bangma CH and Kraaij R: A
prostate cancer vaccine comprising whole cells secreting IL-7,
effective against subcutaneous challenge, requires local GM-CSF for
intra-prostatic efficacy. Cancer Immunol Immunother. 58:373–381.
2009. View Article : Google Scholar : PubMed/NCBI
|