Introduction
Oral squamous cell carcinoma (OSCC) is the 11th most
common type of cancer worldwide, and is ranked the most common
malignant tumor in males and fourth most common in females in
India. Despite recent advances in cancer therapy, oral cancer
remains a major health threat due to the lack of improvement in the
5-year overall survival (OS) rate (1). Treatment of OSCC includes surgery,
radiotherapy, chemotherapy or a combination of different systemic
therapies (2). However, an effective
strategy to select suitable patients for these therapies does not
currently exist owing to the complexities associated with radiation
response, and also limitations regarding tolerance of normal
surrounding tissues. The most effective radiotherapy regimens
attain disease-free survival (DFS) rates of between 45 and 55% in
patients with locally advanced head and neck cancer, and between 30
and 40% in oral cancer due to disease recurrence/metastasis
(1). Oral carcinogenesis arises
through the accumulation of genetic and epigenetic changes,
including alterations in the expression of coding and non-coding
RNAs (3,4). Oral cancer types express a specific
miRNA portfolio, which contributes to maintain the epithelial
characteristics of the cells (5,6).
Epithelial-mesenchymal transition (EMT) is a process
by which an epithelial cell adopts a mesenchymal phenotype and
migrates to a different site and proceeds to colonize (7). A previous study demonstrated that cells
with an EMT phenotype become rich sources for cancer stem-like
cells (CSCs) (7). CSCs undergo
self-renewal, initiate tumors development at distal sites and also
serve an important role in chemo/radio-resistance (8–10). The
presence of EMT and CSCs has been implicated in the increased
resistance to radiotherapy through hypoxia, which assists in
maintaining the CSC niche. In total, >90% of cancer-associated
mortalities are due to metastatic events and are reported to be
initiated by the dysregulation of microRNAs (miRNAs), particularly
the microRNA-200 (miR-200) family (11).
miRNAs are a family of small non-coding RNAs that
are between 21 and 25 nucleotides in length, and bind to target
mRNAs through a 6–8-base seed sequence. miRNAs modulate gene
expression at the post-transcriptional level by blocking
translation or degrading the mRNA, depending on the extent of
sequence complementarity with the target mRNA (12). The miR-200 family includes miR-200a,
miR-200b, miR-200c, miR-141 and miR-429, and all share common seed
sequences, which modulate EMT through the regulation of epithelial
(E-)cadherin expression (11). Tumor
invasion and metastasis have previously been demonstrated to be
tightly controlled by the balanced expression of the miR-200 family
members and zinc finger E-box-binding homeobox (ZEB) transcription
factors (9,11). In previous studies, it has been
demonstrated that miR-200 expression is downregulated within cancer
cells, enabling a positive regulatory loop to maintain ZEB1/ZEB2
expression (11,13). Inhibition of miR-200 reduces
E-cadherin expression, increases vimentin expression and induces
the EMT mechanism (14). Long
non-coding RNAs (lncRNAs) are non-protein coding transcripts
>200 nucleotides in length, and have the potential to regulate
gene expression through cis and trans mechanisms.
ZEB2 antisense RNA 1 (ZEB2-AS1) is an lncRNA that overlaps the 5′
splice site of an intron within the 5′ untranslated region (UTR) of
the ZEB2 gene. ZEB2 and ZEB2-AS1 expressed as bidirectional
cis-natural antisense transcripts (NAT) are essential in
downregulating E-cadherin during EMT (15).
In the present study, the expression of miR-200
family and EMT-associated markers in OSCC was analyzed using the
reverse transcription-quantitative polymerase chain reaction
(RT-qPCR) and paired with patients' clinicopathological
characteristic data. Furthermore, head and neck cancer datasets
from The Cancer Genome Atlas (TCGA) database were also analyzed in
order to evaluate the concordance of the present study.
Materials and methods
Clinical specimens and RNA
isolation
The present study was approved by the Institutional
Ethics Committee (IEC), Madras Medical College (Chennai, India; no.
04092010) and was conducted within the ethical guidelines of IEC,
Madras Medical College. OSCC tissue samples (n=40) were obtained
from patients at the Royapettah Government Hospital (Chennai,
India). Informed consent was obtained from each patient in the form
of a standard questionnaire in accordance with IEC guidelines.
Patients' contextual and clinicopathological characteristics are
presented in Table I. Tumor specimens
were obtained under local anesthesia using punch biopsy immersed in
RNAlater® solution (Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) and transported to the laboratory at Department
of Genetics, University of Madras (Chennai, India) in cold storage.
Stored tissues were washed twice with ice-cold PBS to remove
residual RNAlater® solution, and homogenized using a
MicroSmash MS-100 automated homogenizer (Tomy Digital Biology Co.,
Ltd., Tokyo, Japan) using zirconium beads. RNA was isolated using
the RNeasy mini kit (Qiagen GmbH, Hilden, Germany) and miRNAs were
isolated using an miRNeasy mini kit (Qiagen) according to the
manufacturer's protocol.
 | Table I.Demographic and clinicopathological
details of the 40 cancer patients. |
Table I.
Demographic and clinicopathological
details of the 40 cancer patients.
| Clinical
parameters | No. of patients
(%) |
|---|
| Age, years (mean ±
SD, 50.53±10.63) |
|
≤52 | 19 (47.5) |
|
>52 | 21 (52.5) |
| Sex |
|
|
Male | 30 (75) |
|
Female | 10 (25) |
| Anatomical
site |
|
| Buccal
mucosa | 14 (35) |
|
Tongue | 8
(20) |
|
Alveolar ridge | 4
(10) |
|
Lip | 3
(7.5) |
|
Other | 11 (27.5) |
| Clinical stage |
|
|
≤T3 | 15 (37.5) |
|
≥T4 | 25 (62.5) |
| Nodal
metastasis |
|
|
≤N1 | 19 (47.5) |
|
≥N2 | 21 (52.5) |
| Histological
grade |
|
|
Well-differentiated | 13 (32.5) |
|
Moderately differentiated | 19 (47.5) |
| Poorly
differentiated | 8
(20) |
| Risk habit
profile |
|
|
Exclusive smokers | 12 (30) |
|
Exclusive tobacco-chewers | 4
(10) |
| Mixed habits |
|
| Smoking
and alcoholic | 5
(12.5) |
| Smoking
and chewing | 3
(7.5) |
| All
three | 9
(22.5) |
|
None | 7
(17.5) |
Reverse transcription
DNA was reverse-transcribed from total RNA isolated
from the OSCC samples using a custom-designed miRNA seed-specific
stem-loop primer for miRNAs (Table
II), oligo (dT) primer for ZEB2-AS1
[5′-CAGTGCAGGGTCCGAGGTACAGAGCCACCTGGGCAATTTTTTTTTTTVN-3′ with 3′
wobble bases: V-(A, C, G), N-(A, C, G, T)] (16,17), and a
random hexamer primer for coding genes. Prior to RT reactions, all
samples were pre-incubated for RNA secondary structure denaturation
and primer annealing at 65°C for 20 min. All cDNA conversions were
performed using a Reverse Transcription kit (Invitrogen; Thermo
Fisher Scientific, Inc.) using the following reaction conditions:
55°C for 90 min, 72°C for 15 min and final hold at 4°C.
 | Table II.MicroRNA-specific stem-loop primers
used for cDNA synthesis. |
Table II.
MicroRNA-specific stem-loop primers
used for cDNA synthesis.
| miRNA | Stem-loop primers
(5′→3′) |
|---|
| miR-200a |
GTCGTATCCAGTGCGTCGAGTGACACGAGAGCCACCTGGGCAATTTGCACTGGATACGACACATCG |
| miR-200b |
GTCGTATCCAGTGCGTCGAGTGACACGAGAGCCACCTGGGCAATTTGCACTGGATACGACTCATCA |
| miR-200c |
GTCGTATCCAGTGCGTCGAGTGACACGAGAGCCACCTGGGCAATTTGCACTGGATACGACTCCATC |
| miR-429 |
GTCGTATCCAGTGCGTCGAGTGACACGAGAGCCACCTGGGCAATTTGCACTGGATACGACACGGTT |
| miR-141 |
GTCGTATCCAGTGCGTCGAGTGACACGAGAGCCACCTGGGCAATTTGCACTGGATACGACCCATCT |
| RNU44 |
GTCGTATCCAGTGCGTCGAGTGACACGAGAGCCACCTGGGCAATTTGCACTGGATACGACAGTCAG |
Quantitative polymerase chain reaction
(qPCR)
Relative quantification was performed using the
TaqMan® custom designed assays (Thermo Fisher
Scientific, Inc.). qPCRs were performed in triplicate (384-well
optical plates) with 10 µl total volume using cDNAs (diluted
25-fold) with TaqMan® 2X Universal Master mix (No
AmpErase® UNG; Thermo Fisher Scientific, Inc.),
universal reverse primer, either universal reverse lncRNA,
5′-CAGTGCAGGGTCCGAGGT-3′ or universal reverse miRNA,
5′-TCGTATCCAGTGCGTCGAGT-3′, and the following specific forward
primer: miR-200a forward, 5′-AGTAACACTGTCTGGTAACGA-3′; miR-200b
forward, 5′-CGCAGTAATACTGCCTGGT-3′; miR-200c forward,
5′-AGTAATACTGCCGGGTAATGA-3′; miR-429 forward,
5′-CGCAGTAATACTGTCTGGTA-3′; miR-141 forward,
5′-CGCAGTAACACTGTCTGGT-3′; RNU44 forward,
5′-GCAAATGCTGACTGAACATGA-3′; ZEB1-AS1 forward,
5′-TGTGCATGATGAATTTCTGGACTGGA-3′; GAP DH forward,
5′-GAAGAGGGGAGGGGCCTAGG-3′) and universal fluorescein
amidite-labeled minor groove binder probe
(5′-CAGAGCCACCTGGGCAATTTT-3′) for lncRNA (ZEB2-AS1) and miRNAs.
Expression of coding genes was analyzed using SYBR−Green
master mix (Takara Bio, Inc., Otsu, Japan) using gene-specific
primers including: GAPDH forward, 5′-AGGGCTGCTTTTAACTCTGGT-3′ and
reverse, 5′-CCCCACTTGATTTGGAGGGA-3′; ZEB1 forward,
5′-CACTCCCTGCAGCAGAAGCTGA-3′ and reverse,
5′-GCTCTTCTGCACTTGGTTGTGCT-3′; ZEB2 forward,
5′-AACTGGAGGAACGCGATGGTCA-3′ and reverse,
5′-GCAGTTGGGCAAAAGCATCTGGA-3′; E-cadherin forward,
5′-GCTGCTGCAGGTCTCCTCTTG-3′ and reverse,
5′-CCTTTGTCGACCGGTGCAATCT-3′; vimentin forward,
5′-AGCTGCAGGCTCAGATTCAGGA-3′ and reverse,
5′-CGGTTGGCAGCCTCAGAGAGGT-3′. The experiments were carried out
using a 7900HT Real-Time PCR system (Thermo Fisher Scientific,
Inc.) with the following reaction conditions: 50°C for 2 min, 95°C
for 10 min for initial denaturation and enzyme activation followed
by cyclic denaturation at 95°C for 15 sec and primer annealing and
extension at 60°C for 1 min. A negative control without a cDNA
template was included in parallel for all assays. GAPDH and RNU44
were used as endogenous controls for coding/non-coding genes and
miRNAs, respectively. Relative expression levels were quantified
using the 2−ΔΔCq method (18). Each experiment was completed in
triplicate, and the mean was used for analysis.
Clinical evaluation and TCGA data
analysis
All patients with OSCC were treated using
radiotherapy (50–60 Gy) alongside three rounds of chemotherapy
(Cisplatin; Naprod Life Sciences Pvt., Ltd., Mumbai, India) and
5-fluorouracil (Celon Laboratories, Ltd., Telangana, India). Tumor
responses to chemo/radio-therapy were evaluated following 4 weeks
of treatment. Patients who exhibited a partial or complete response
to treatment were categorized as treatment responders, and the
remaining patients were categorized as either poor responders or
resistant to treatment. Patients were monitored for 18 months
following treatment to study the therapeutic outcome. Furthermore,
a comparison study on head and neck cancer were completed using RNA
sequencing datasets obtained from 523 head and neck cancer datasets
from TCGA database using the cBioportal interactive genomics data
portal. Expression levels of the genes obtained from TCGA datasets
were quantified using reads/kb/106 mapped reads
(RPKM).
Statistical analysis
All statistical analyses were performed using
GraphPad Prism (version 6; GraphPad Software, Inc., La Jolla, CA,
USA). Numerical data are presented as the mean ± standard error of
mean. Differences between means were analyzed using Student's
t-test for parametric data and Mann-Whitney for non-parametric
data. A univariate analysis was performed for the association with
clinical features. Survival curves were plotted using the
Kaplan-Meier estimator method, and the fraction survival rate was
tested using the log-rank test. All tests were two-tailed and
P<0.05 was considered to indicate a statistically significant
difference.
Results
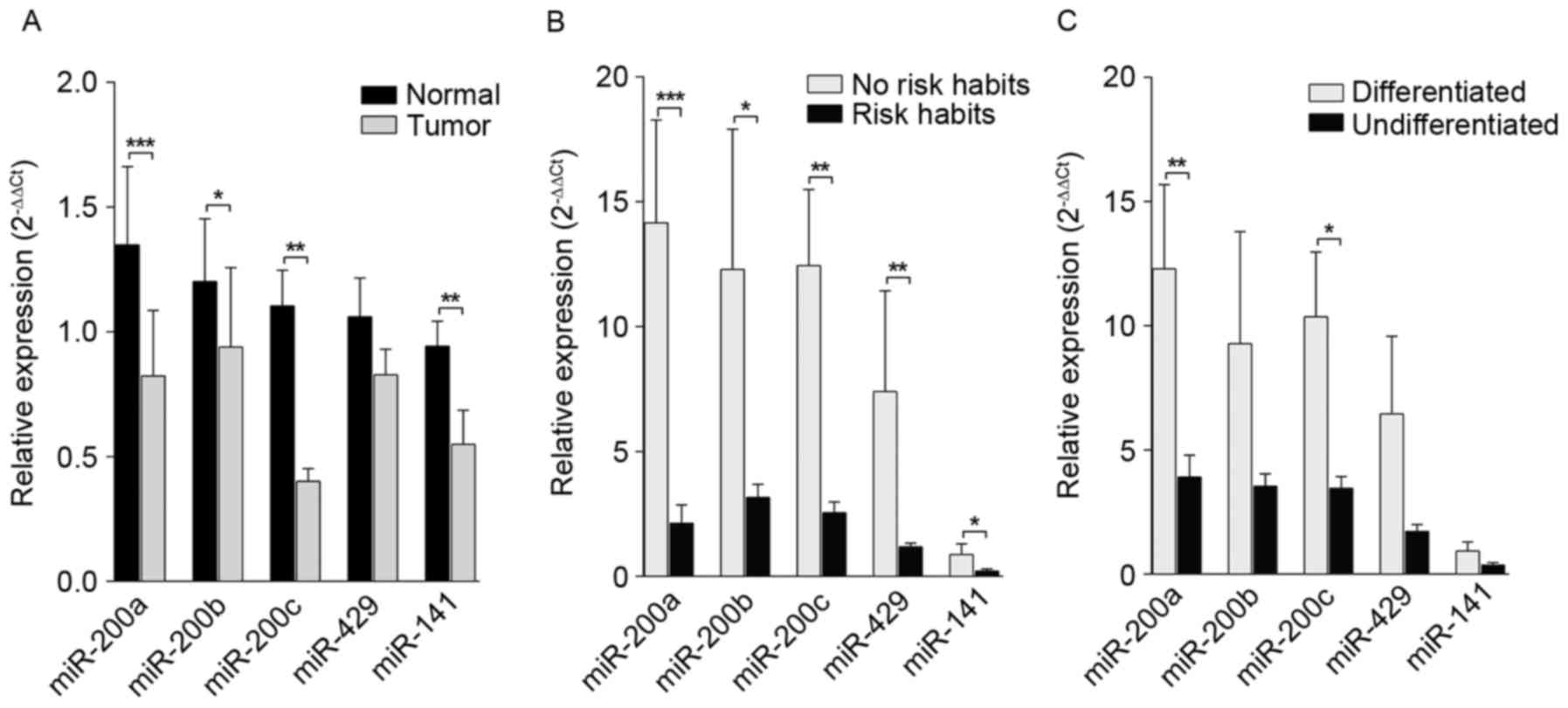
miR-200 family miRNAs are
downregulated in OSCC
Downregulation of the miR-200 family induces EMT and
promotes metastasis in epithelial tumors (19–21).
Expression levels of the miR-200 family in OSCC (n=40) and normal
(n=8) tissues were determined using RT-qPCR. Results demonstrated a
downregulation of the miR-200 family (miR-200a, miR-200b, miR-200c,
miR-429 and miR-141) in OSCC samples compared with normal tissue
samples. Statistical analysis of expression levels demonstrated
that each member of the miR-200 family [miR-200a 33/40 (82.5%),
miR-200b 33/40 (82.5%), miR-200c 25/40 (62.5%) and miR-141 28/40
(70%)] were significantly downregulated (P=0.002, 0.0116, 0.0099
and 0.0035, respectively; Fig.
1A).
Downregulation of the miR-200 family
miRNAs is associated with tobacco chewing/smoking risk habits
Tumors with distinct clinicopathological and
demographic features may be unique due to their anatomical origin.
Expression levels of individual miRNAs were categorized based on
demographic features and analyzed using the univariate analysis.
Results also demonstrated that expression levels of the miR-200
family were markedly influenced by risk factors including tobacco
chewing and smoking. Results presented in Fig. 1B revealed that patients with OSCC and
who are associated with a risk factor/habit (n=33/40) demonstrated
a significant downregulation in miR-200a (82%; n=27; P=0.0006),
miR-200b (79%; n=26; P=0.0467), miR-200c (57.5%; n=19; P=0.0014),
miR-429 (57.6%; n=19; P=0.0087) and miR-141 (60.6%; n=20;
P=0.0230). Furthermore, a significant downregulation in miR-200a
(P=0.0067) and miR-200c (P=0.0248) was detected in undifferentiated
tumors. No significant associations were observed between
expression levels of miR-200 family members, tumor grade and nodal
stage of tumor samples (Fig. 1C).
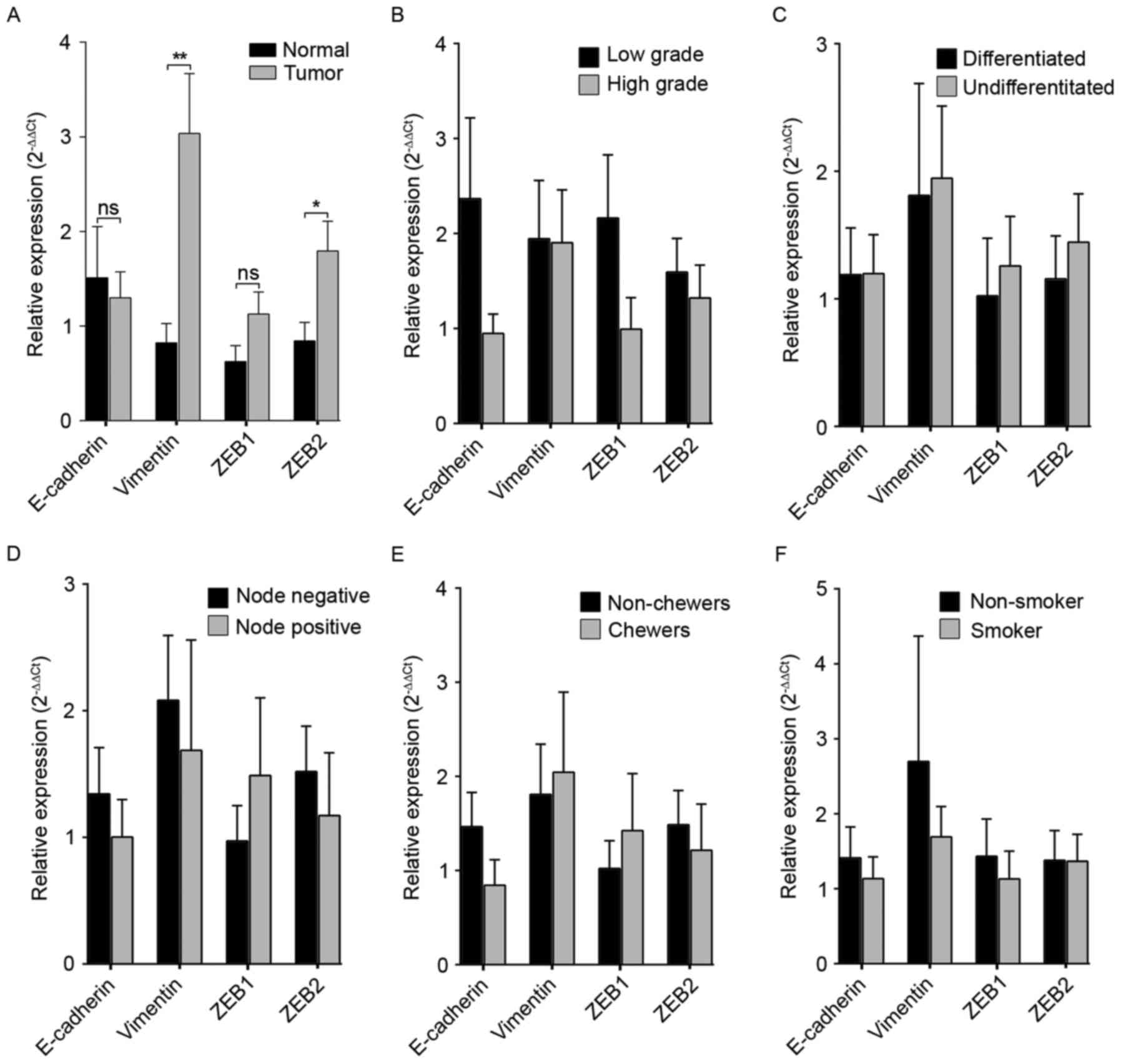
EMT-regulatory genes are overexpressed
in OSCC
Expression of the EMT driver genes ZEB1 and ZEB2,
and the epithelial marker E-cadherin and mesenchymal marker
vimentin were analyzed in OSCC. Results demonstrated an
overexpression of ZEB1 (36.5%; P=0.7733) and ZEB2 (50%; P=0.0451)
in tumor samples compared with normal tissue samples. E-cadherin
was downregulated in 50% of tumors, whereas vimentin was
significantly upregulated in 54.5% of tumors (P=0.0071; Fig. 2A). However, there was no significant
association between EMT-regulatory gene expression with the
clinicopathological features of oral cancer samples (Fig. 2B-F).
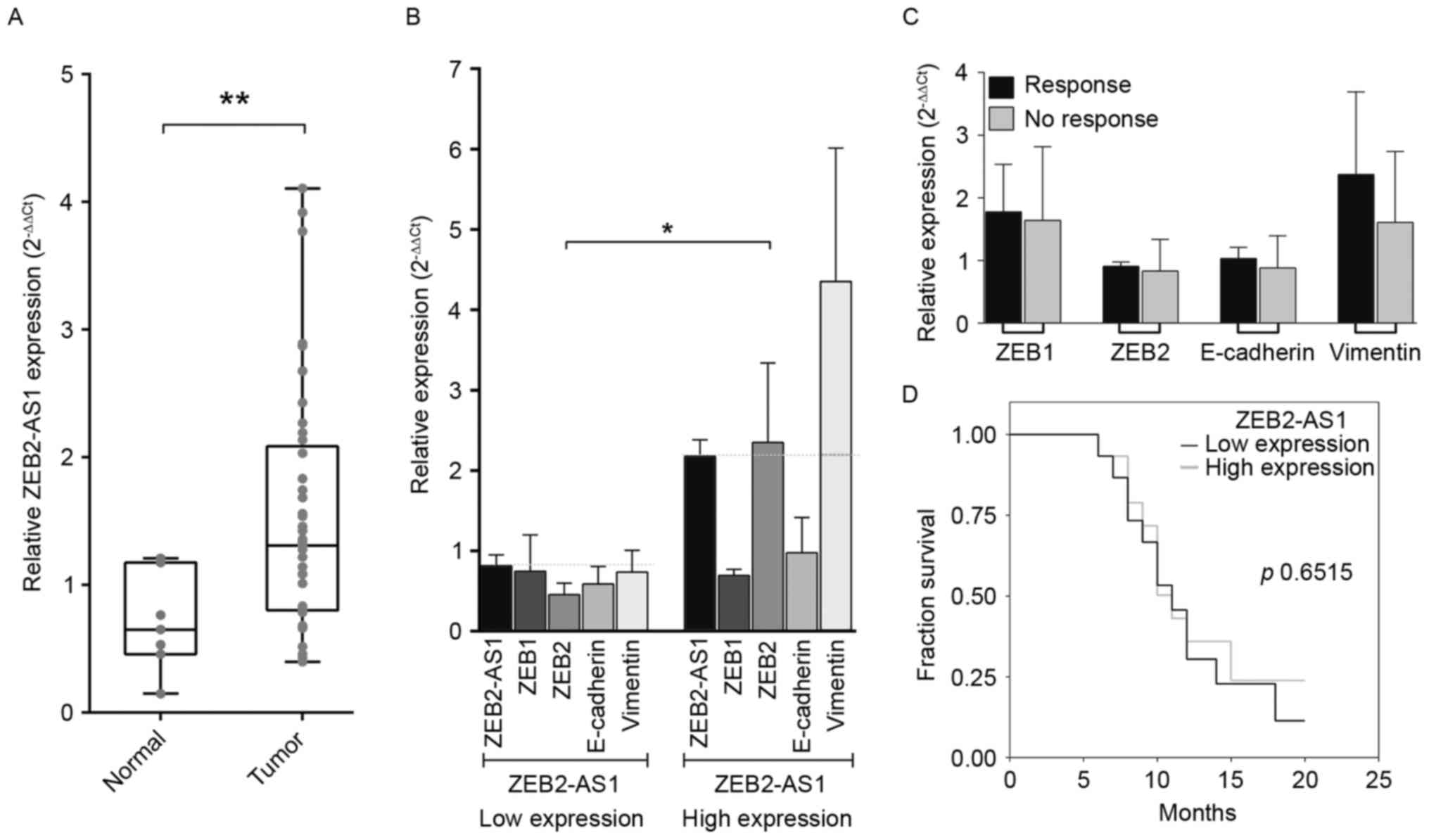
Expression of EMT activators, their
antisense transcripts and the clinical outcomes in OSCC
ZEB1-AS1, a natural antisense transcript has
previously been reported to upregulate the expression of ZEB1 in
tumor cells (22). The present study
investigated the association between the expression of ZEB1 and its
antisense transcript ZEB1-AS1 in head and neck datasets obtained
from TCGA and identified that majority of the tumor samples
co-expressed ZEB1 and ZEB1-AS1 (data not shown). Therefore, further
analysis was performed to assess whether ZEB2-AS1, an antisense
transcript of ZEB2 gene, was similarly able to also upregulate
ZEB2. Notably, the majority of the samples co-expressed ZEB2 and
ZEB2-AS1, and a high-level expression of the ZEB2-AS1 in 16/40 OSCC
tumors (40%; P=0.0075) compared with normal tissues (Fig. 3A). Further comparisons were made
regarding expression levels of ZEB2-AS1 with EMT activators.
Notably, the expression of ZEB2 and vimentin were upregulated 40
and 60%, respectively, in tumor samples, which overexpressed
ZEB2-AS1. Results demonstrated a statistically significant
difference in ZEB2 expression level between the low (54.5%) and
high (45.5%) ZEB2-AS1-expressing tumors (Fig. 3B; Table
III; P=0.0303). Furthermore, the analysis of expression levels
of ZEB1, ZEB2, E-cadherin and vimentin genes with treatment
response demonstrated no significant difference between responder
and non-responder groups (Fig. 2C).
Survival rates of patients in association with the expression level
of ZEB2-AS1 did not demonstrate any statistical significance
(Fig. 3D; P=0.6515).
 | Table III.Expression values of EMT marker genes
in ZEB2-AS1-low and -high expressed oral cancer samples. |
Table III.
Expression values of EMT marker genes
in ZEB2-AS1-low and -high expressed oral cancer samples.
|
| ZEB2-AS1 low
expression | ZEB2-AS1 high
expression |
|
|---|
|
|
|
|
|
|---|
| Gene | Mean | SD | Mean | SD |
P-valuea |
|---|
| ZEB1 | 0.7477 | 1.102 | 0.6926 | 0.1721 | 0.3268 |
| ZEB2 | 0.4551 | 0.3479 | 2.348 | 2.214 | 0.0303a |
| E-cadherin | 0.5882 | 0.5379 | 0.9788 | 0.9756 | 0.7706 |
| Vimentin | 0.7374 | 0.6573 | 4.355 | 3.704 | 0.1775 |
EMT markers are not associated with
clinical outcomes in head and neck squamous cell carcinoma (HNSCC)
in TCGA database
The expression levels of ZEB1 and ZEB1-AS1 were
analyzed in 523 HNSCC samples in TCGA database; results identified
an overexpression of ZEB1 in samples, which also expressed a high
level of ZEB1-AS1. However, the results of the present study could
not confirm the association between ZEB2-AS1 and ZEB2 expression
levels as it is yet to be annotated in TCGA database. Notably,
HNSCC samples obtained from TCGA dataset identified ZEB1 (36%) and
ZEB2 (37.3%) overexpression in tumors that were significantly
hypomethylated (P<0.001 and P<0.001 respectively) at their
promoters (data not shown). Furthermore, the expression levels of
EMT-regulatory genes ZEB1, ZEB2, E-cadherin and vimentin did not
demonstrate any association with tumor stage (Fig. 4A). In addition, the role of other EMT
markers including Twist-related protein 1 (TWIST1), snail family
transcriptional repressor 1 (SNAIL1) and B lymphoma Mo-MLV
insertion region 1 homolog (BMI1) were analyzed and no association
with tumor stage was observed (Fig.
4B). Similarly, the EMT-regulatory genes demonstrated no
significant change in expression levels between disease-free and
disease-recurrence groups (Fig. 4C and
D). Survival analysis of datasets of patients with HNSCC
obtained from TCGA database did not demonstrate any association
with ZEB1, ZEB2, ZEB1-AS1 and ZEB2-AS1 expression (data not shown);
these results are consistent with the present study.
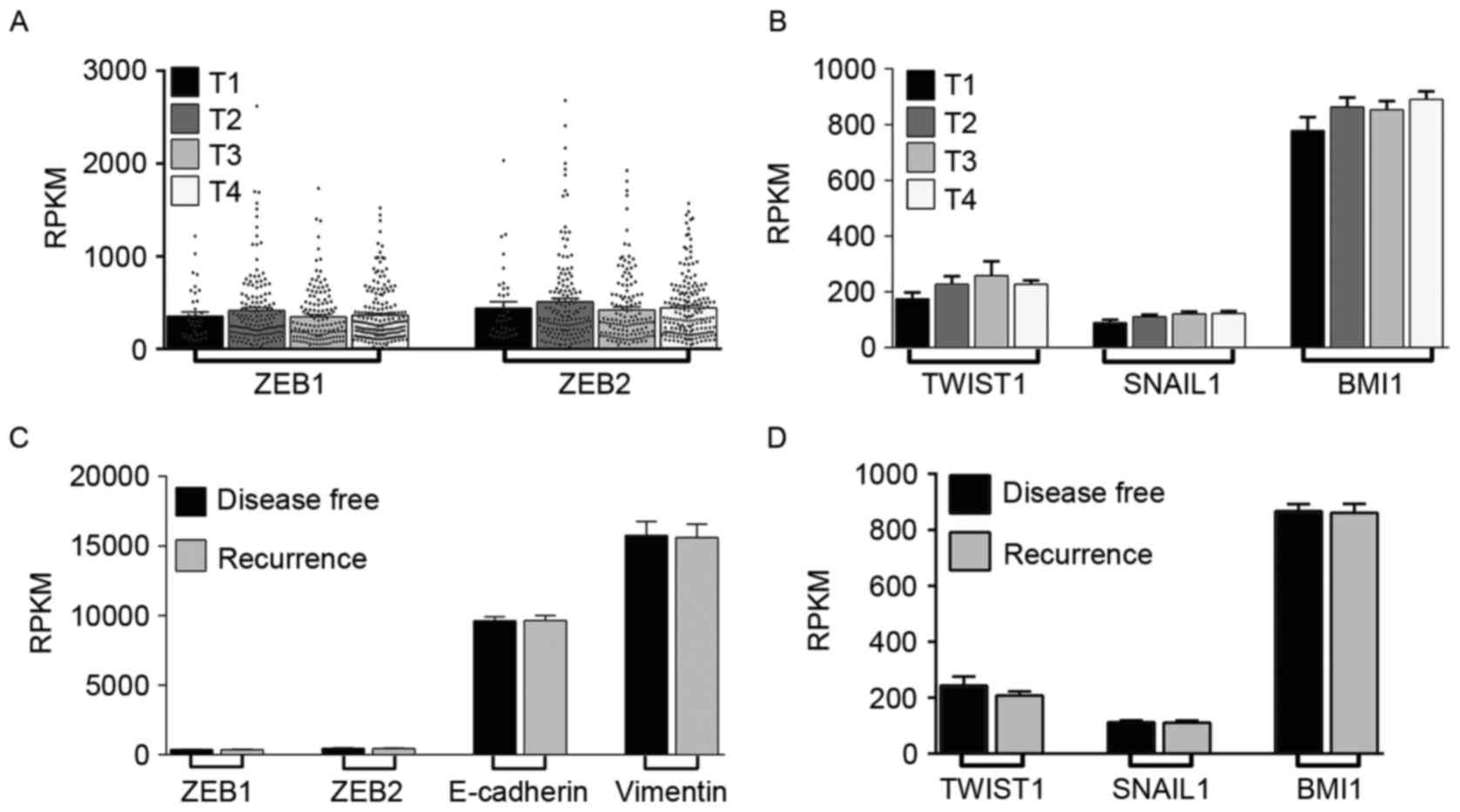 | Figure 4.Expression of EMT markers with tumor
features and overall patient survival from TCGA HNSCC datasets. (A)
Expression of EMT markers ZEB1, ZEB2, E-cadherin and vimentin
between the grade of the tumor (T1, T2, T3 and T4) from TCGA HNSCC
datasets (n=523). There was no significant change in expression
between the groups. (B) Expression of EMT-associated genes TWIST1,
SNAIL1 and BMI1 were unchanged in tumor stages of patients with
HNSCC. (C) Expression levels of ZEB1, ZEB2, E-cadherin and vimentin
were unchanged with disease-free survival and disease recurrence in
patients with HNSCC. (D) Expression of EMT-associated genes TWIST1,
SNAIL1 and BMI1 compared with disease-free and disease recurrence
of patients with HNSCC from TCGA datasets. The expression levels
from TCGA datasets were expressed as RPKM. EMT,
epithelial-mesenchymal transition; ZEB; zinc finger E-box-binding
homeobox; TCGA, The Cancer Genome Atlas; HNSCC, head and neck
squamous cell carcinoma; TWIST1; Twist-related protein 1; SNAIL1,
snail family transcriptional repressor 1; BMI1, B lymphoma Mo-MLV
insertion region 1 homolog; RPKM, reads/kb/106 mapped
reads; E-cadherin, epithelial cadherin. |
Discussion
The miR-200 family is a group of tumor suppressor
miRNAs involved in the regulation of EMT, repression of
self-renewal and differentiation of CSCs, and reversal of
chemoresistance (10). In the present
study, the role of each member within the miR-200 family in OSCC
was analyzed through the observation of expression levels, and
results identified a significant downregulation in miR-200 family
members in OSCC compared with levels detected in normal tissues.
Several previous studies established that miR-200 family members
are critical for the maintenance of the epithelial phenotype, and
has been demonstrated to be deregulated in metastatic cells in
several types of cancer (19,23). Expression levels of miR-200 family
genes have been revealed to inhibit EMT and decrease invasion and
metastasis in tumors (19–21). Environmental risk factors including
areca nut, slacked lime, betel quid, alcohol consumption, and
tobacco chewing and smoking were frequently associated with oral
cancer in India (24). The present
study identified a significant association between the expression
levels of the miR-200 family members and history of tobacco chewing
and smoking. In particular, miR-200a and miR-200c were identified
to be significantly deregulated and associated with tumor
clinicopathological features. In our previous study, miRNAs
deregulation was reported in tobacco-chewing-associated oral cancer
(25). In addition, another study
established the role of tobacco-specific nitrosamine in methylating
gene promoters (26). Tellez et
al (27) reported that tobacco
carcinogens induced the transformation of human lung epithelia
through epigenetic silencing of miR-205 and miR-200, which led to
EMT and stem cell-like properties. miR-200 family miRNAs regulate
EMT through the modulation of E-cadherin expression by targeting
E-cadherin repressors ZEB1 and ZEB2 (21). Inhibition of miR-200 has been
demonstrated to decrease E-cadherin expression, increase the
expression of vimentin and induce EMT (11). The present study therefore
investigated the expression of ZEB1 and ZEB2 in order to determine
the effect(s) of the miR-200 downregulation in OSCC, and
upregulation of EMT inducers, namely ZEB1 and ZEB2, was observed in
the tumor samples compared with the normal tissue, suggesting the
existence of EMT in oral tumors. These results are consistent with
those of several previous studies (11,19,21).
E-cadherin, an epithelial cell marker, was reported to be
downregulated in the metastatic tumors during EMT, and is a proven
target of ZEB1/2 transcription factors (11). Despite detecting a trend, but no
marked change in E-cadherin expression, results identified a
significant overexpression in the mesenchymal marker vimentin,
confirming EMT in OSCC. Therefore, the downregulation of the
miR-200 family and the upregulation of ZEB1 and ZEB2 suggest the
induction of EMT, which may lead to the maintenance and release of
circulating tumor cells in oral tumors, as reported in several
types of cancer with epithelial origin (28,29).
During EMT, ZEB1 and ZEB2 levels may be influenced
by the level of their antisense transcripts ZEB1-AS1 and ZEB2-AS1.
Therefore, analysis of the expression of ZEB2-AS1 in OSCC was
performed and a significant upregulation was identified. In
addition, the expression levels of ZEB2 in the samples that also
expressed increased levels of ZEB2-AS1 were determined, and it was
identified that the expression level of ZEB2 was consistent with
the high-level expression of its natural antisense transcript. In
epithelial cells, ZEB2-AS1 was demonstrated to prevent splicing of
the ZEB2 5′UTR, and promote levels of ZEB2 protein in colon
adenocarcinomas (30). However, an
increased level of ZEB2 in the cytosol was revealed to downregulate
E-cadherin, and enable cells to undergo EMT (31). Similarly, ZEB1-AS1 upregulation has
been reported to increase the promoter activity of the ZEB1 gene
resulting in the repression of E-cadherin, and induction of EMT in
hepatocellular carcinoma (22). The
results of the present study identified a co-expression pattern in
ZEB2 and ZEB2-AS1 in oral tumor samples. Owing to the limitation of
sample size and no annotation of ZEB2-AS1 in TCGA, the present
study could not derive a conclusion that ZEB2 overexpression is due
to the ZEB2-AS1. Therefore, the association between the expression
of ZEB1 and its natural antisense transcript ZEB1-AS1 in 523 TCGA
HNSCC datasets was analyzed and identified that the majority of the
samples co-expressed ZEB1 and ZEB1-AS1. Therefore, our results
suggest that ZEB2 levels may be regulated by its natural antisense
transcript by increasing promoter activity or increasing the levels
of ZEB2 protein through the stabilization of its 5′UTR. In
addition, the methylation signature of the ZEB family gene
promoters from TCGA HNSCC cases were examined, and a high level of
ZEB1 and ZEB2 gene expression was observed in tumors with a
hypomethylated promoter region, suggesting that gene regulation is
achieved by altering promoter methylation events. Chaffer et
al (32) reported that the
induction of transforming growth factor β (TGFβ) expression leads
to the hypomethylation of the ZEB1 promoter region. Therefore, a
similar response may hypomethylate the ZEB2 promoter region and
induce the expression level. Hypomethylation of the ZEB
transcription factor promoter region may enable the overexpression
of ZEB transcription factors (32).
The miR-200 family post-transcriptionally regulates the EMT marker
genes including ZEB1 and ZEB2. The ZEB transcription factors may
downregulate the expression of miR-200 family miRNAs. In addition,
natural anti-sense transcript ZEB1-AS1 was demonstrated to enhance
the transcription of ZEB1 and increase mRNA stability within the
cytosol (30,22). Similarly, ZEB2 stability may also be
enhanced by ZEB2-AS1 expression. Thus, the enhanced expression of
ZEB1 and ZEB2 may downregulate miR-200 family miRNAs more
effectively than normal expression levels, leading to the
activation of EMT.
The present study demonstrated increased expression
levels of EMT-associated genes in tumors compared with normal
tissues, suggesting the existence of EMT in OSCC. Recently, EMT has
been demonstrated to serve a critical role in drug resistance and
distal metastasis, which accounts for tumor recurrence (33). The present study analyzed the
expression of EMT activator genes including TWIST1, SNAIL1 and
BMI1, in addition to ZEB family members and phenotypic marker genes
E-cadherin and vimentin in HNSCC cases obtained from TCGA database.
Notably, there were no significant differences in expression levels
of the EMT markers with reference to the tumor stage, disease
recurrence and treatment response. Survival rate analysis also
demonstrated no association between the expression of
EMT-activating genes and patient survival rate. Differential roles
of EMT have been previously reported in various organ-specific
human cancers (34,35). The expression of TGFβ may also
positively regulate ZEB1/2 levels, thereby promoting metastasis in
tumor cells (11). During embryonic
development, Wnt signaling, fibroblast growth factors, Ras,
phosphoinositide 3-kinase/protein kinase B, bone morphogenetic
proteins, c-myeloblastosis, eomesodermin, mesoderm posterior
proteins and msh homeobox-1-mediated signaling pathways either in
combination or individually are able to induce EMT (36–38). As
metastatic cancer cells behave by way of embryonic stem cells
through the reactivation of embryo-specific genes, EMT in cancer
cells may be initiated by this alternative means of activation.
Despite regular EMT activation, epithelial cells under inflammatory
stress are able to initiate partial EMT to detach from the
epithelium and accumulate at the basal membrane where they switch
to a fibroblast phenotype (36).
Despite EMT activation through the upregulation of
classical EMT-associated transcription factors, deregulation of
miRNAs targeting EMT regulators may also be able to initiate EMT in
cancer. Upregulation of miRNAs including miR-106b-25 cluster,
miR-491-5p, miR-661 and miR-24 leads to decreased cell-cell
adhesion and downregulation of miR-31, miR-124, miR-205 and miR-34,
resulting in the derepression of translation of EMT-activating
transcription factors (38). The
low-level transcripts of EMT-activating factors, through decreased
or lack of post-transcriptional regulation of deregulated miRNAs,
may confer an advantage for a longer half-life in the cytosol, and
may maintain the steady-state level of EMT factors. Furthermore,
non-coding RNAs are also reported to be major components of the
cellular signaling circuit that regulates EMT (39,40).
Regardless of studies reporting the dysregulation of various
factors involved in regulation of EMT, it has been demonstrated
that EMT is not crucial for metastasis to occur in lung cancer;
however, it has been identified to contribute to chemoresistance
(34). Furthermore, knockdown studies
demonstrated that EMT genes (TWIST1 and SNAIL1) were not required
for metastasis; however, they contribute to treatment resistance in
pancreatic cancer (35).
In conclusion, the results of the present study
identified a dysregulation of miR-200 family miRNAs and the
upregulation of EMT-inducer genes in OSCC. Results demonstrated an
association between deregulated expression of miR-200 family
miRNAs, tobacco chewing/smoking and the cellular differentiation
status of oral tumors. To the best of our knowledge, the present
study is the first to identify an association between the
expression of ZEB2-AS1 and ZEB2 in oral cancer, and to demonstrate
the association of ZEB2 levels with its natural antisense
transcript ZEB2-AS1 level. Furthermore, the present study was not
able to confirm any association between the EMT-regulatory genes
and treatment response. A similar result was also observed in HNSCC
cases obtained from TCGA database. The present study was performed
with a limited number of oral cancer samples as the majority of
patients with cancer visited the hospital at an advanced stage of
the disease, and a number of participants discontinued treatment
following one or two rounds of chemotherapy. Further sequencing
genomes and protein estimation will add to the evidence obtained in
the present study. The primary limitation of the present study was
the nature of tissue sample collection as punch biopsies are only
sufficient for histopathological analysis and RNA isolation.
Therefore, genome sequencing and protein-based experiments were not
permitted. Further studies with an increased sample size and
functional dissection are warranted to confirm the differential
role(s) of EMT observed in different types of human cancer.
Acknowledgements
G.A., A.K.D. and M.M. acknowledge University Grant
Commission and Council of Scientific and Industrial Research,
Government of India, respectively for research fellowships. The
authors acknowledge Department of Science and Technology-Fund for
Improvement of S&T Infrastructure, University Grants
Commission-Special Assistance Programme and Department of Health
Research-Multidisciplinary Research Unit for the infrastructural
facility. The authors would like to thank Cancer Institute Adyar,
Chennai for permitting the use of their real-time facility for part
of the study. The present study was supported by a research grant
from Department of Atomic Energy, Board of Research in Nuclear
Sciences (grant no. 35/14/10/2014-BRNS/0210) and Department of
Biotechnology (grant no. BT/PR4820/MED/12/622/2013), Government of
India.
Glossary
Abbreviations
Abbreviations:
|
OSCC
|
oral squamous cell carcinoma
|
|
HNSCC
|
head and neck squamous cell
carcinoma
|
|
TCGA
|
the Cancer Genome Atlas
|
|
EMT
|
epithelial-mesenchymal transition
|
|
DFS
|
disease-free survival
|
|
CSC
|
cancer stem-like cells
|
|
NAT
|
natural antisense transcript
|
|
ZEB2-AS1
|
zinc finger E-box-binding homeobox 2
antisense RNA1
|
References
|
1
|
Bourhis J, Overgaard J, Audry H, Ang KK,
Saunders M, Bernier J, Horiot JC, Le Maître A, Pajak TF, Poulsen
MG, et al: Hyperfractionated or accelerated radiotherapy in head
and neck cancer: A meta-analysis. Lancet. 368:843–854. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Huang SH and O'Sullivan B: Oral cancer:
Current role of radiotherapy and chemotherapy. Med Oral Patol Oral
Cir Bucal. 8:e233–e240. 2013. View Article : Google Scholar
|
|
3
|
Peschansky VJ and Wahlestedt C: Non-coding
RNAs as direct and indirect modulators of epigenetic regulation.
Epigenetics. 9:3–12. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tao H, Yang JJ and Shi KH: Non-coding RNAs
as direct and indirect modulators of epigenetic mechanism
regulation of cardiac fibrosis. Expert Opin Ther Targets.
19:707–716. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lajer CB, Nielsen FC, Friis-Hansen L,
Norrild B, Borup R, Garnæs E, Rossing M, Specht L, Therkildsen MH,
Nauntofte B, et al: Different miRNA signatures of oral and
pharyngeal squamous cell carcinomas: A prospective translational
study. Br J Cancer. 104:830–840. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Manikandan M, Magendhra Rao AK Deva,
Arunkumar G, Manickavasagam M, Rajkumar KS, Rajaraman R and
Munirajan AK: Oral squamous cell carcinoma: microRNA expression
profiling and integrative analyses for elucidation of
tumourigenesis mechanism. Mol Cancer. 15:282016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan
A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M, et al: The
epithelial-mesenchymal transition generates cells with properties
of stem cells. Cell. 133:704–715. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Han J, Fujisawa T, Husain SR and Puri RK:
Identification and characterization of cancer stem cells in human
head and neck squamous cell carcinoma. BMC Cancer. 14:1732014.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kalluri R and Weinberg RA: The basics of
epithelial-mesenchymal transition. J Clin Invest. 119:1420–1428.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Shrivastava S, Steele R, Sowadski M,
Crawford SE, Varvares M and Ray RB: Identification of molecular
signature of head and neck cancer stem-like cells. Sci Rep.
5:78192015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Park SM, Gaur AB, Lengyel E and Peter ME:
The miR-200 family determines the epithelial phenotype of cancer
cells by targeting the E-cadherin repressors ZEB1 and ZEB2. Genes
and Dev. 22:894–907. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tamagawa S, Beder LB, Hotomi M, Gunduz M,
Yata K, Grenman R and Yamanaka N: Role of miR-200c/miR-141 in the
regulation of epithelial-mesenchymal transition and migration in
head and neck squamous cell carcinoma. Int J Mol Med. 33:879–786.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Thompson EW, Torri J, Sabol M, Sommers CL,
Byers S, Valverius EM, Martin GR, Lippman ME, Stampfer MR and
Dickson RB: Oncogene-induced basement membrane invasiveness in
human mammary epithelial cells. Clin Exp Metastasis. 12:181–194.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Balbin OA, Malik R, Dhanasekaran SM,
Prensner JR, Cao X, Wu YM, Robinson D, Wang R, Chen G, Beer DG,
Nesvizhskii AI and Chinnaiyan AM: The landscape of antisense gene
expression in human cancers. Genome Res. 25:1068–1079. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chen C, Ridzon DA, Broomer AJ, Zhou Z, Lee
DH, Nguyen JT, Barbisin M, Xu NL, Mahuvakar VR, Andersen MR, et al:
Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic
Acids Res. 33:e1792005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kang K, Zhang X, Liu H, Wang Z, Zhong J,
Huang Z, Peng X, Zeng Y, Wang Y, Yang Y, et al: A novel real-time
PCR assay of microRNAs using S-Poly(T), a specific oligo(dT)
reverse transcription primer with excellent sensitivity and
specificity. PLoS One. 7:e485362012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gibbons DL, Lin W, Creighton CJ, Rizvi ZH,
Gregory PA, Goodall GJ, Thilaganathan N, Du L, Zhang Y,
Pertsemlidis A and Kurie JM: Contextual extracellular cues promote
tumor cell EMT and metastasis by regulating miR-200 family
expression. Genes Dev. 23:2140–2151. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xu Y, Brenn T, Brown ER, Doherty V and
Melton DW: Differential expression of microRNAs during melanoma
progression: miR-200c, miR-205 and miR-211 are downregulated in
melanoma and act as tumour suppressors. Br J Cancer. 106:553–561.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Koutsaki M, Spandidos DA and Zaravinos A:
Epithelial-mesenchymal transition-associated miRNAs in ovarian
carcinoma, with highlight on the miR-200 family: Prognostic value
and prospective role in ovarian cancer therapeutics. Cancer Lett.
351:173–181. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li T, Xie J, Shen C, Cheng D, Shi Y, Wu Z,
Deng X, Chen H, Shen B, Peng C, et al: Upregulation of long
noncoding RNA ZEB1-AS1 promotes tumor metastasis and predicts poor
prognosis in hepatocellular carcinoma. Oncogene. 35:1575–1584.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Olson P, Lu J, Zhang H, Shai A, Chun MG,
Wang Y, Libutti SK, Nakakura EK, Golub TR and Hanahan D: MicroRNA
dynamics in the stages of tumorigenesis correlate with hallmark
capabilities of cancer. Genes Dev. 23:2152–2165. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Manjari M, Popli R, Paul S, Gupta VP and
Kaholon SK: Prevalence of oral cavity, pharynx, larynx and nasal
cavity malignancies in Amritsar, Punjab. Indian J Otolaryngol Head
Neck Surg. 48:191–195. 1999.
|
|
25
|
Manikandan M, Magendhra Rao AK Deva,
Rajkumar KS, Rajaraman R and Munirajan AK: Altered levels of
miR-21, miR-125b-2*, miR-138, miR-155, miR-184, and miR-205 in oral
squamous cell carcinoma and association with clinicopathological
characteristics. J Oral Pathol Med. 44:792–800. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
La DK, Upton PB and Swenberg JA:
Carcinogenic alkylating agentsComprehensive Toxicology (revised
ed). 12. Pergamon Press; Oxford: pp. 111–140. 2010
|
|
27
|
Tellez CS, Juri DE, Do K, Bernauer AM,
Thomas CL, Damiani LA, Tessema M, Leng S and Belinsky SA: EMT and
stem cell-like properties associated with miR-205 and miR-200
epigenetic silencing are early manifestations during
carcinogen-induced transformation of human lung epithelial cells.
Cancer Res. 71:3087–3097. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cristofanilli M, Budd GT, Ellis MJ,
Stopeck A, Matera J, Miller MC, Reuben JM, Doyle GV, Allard WJ,
Terstappen LW and Hayes DF: Circulating tumor cells, disease
progression and survival in metastatic breast cancer. N Engl J Med.
351:781–791. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cohen SJ, Punt CJ, Iannotti N, Saidman BH,
Sabbath KD, Gabrail NY, Picus J, Morse M, Mitchell E, Miller MC, et
al: Relationship of circulating tumor cells to tumor response,
progression-free survival and overall survival in patients with
metastatic colorectal cancer. J Clin Oncol. 26:3213–3221. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Beltran M, Puig I, Peña C, García JM,
Álvarez AB, Peña R, Bonilla F and de Herreros AG: A natural
antisense transcript regulates Zeb2/Sip1 gene expression during
Snail1-induced epithelial-mesenchymal transition. Genes Dev.
22:756–769. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wellner U, Schubert J, Burk UC,
Schmalhofer O, Zhu F, Sonntag A, Waldvogel B, Vannier C, Darling D,
zur Hausen A, et al: The EMT-activator ZEB1 promotes tumorigenicity
by repressing stemness-inhibiting microRNAs. Nat Cell Biol.
11:1487–1495. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chaffer CL, Marjanovic ND, Lee T, Bell G,
Kleer CG, Reinhardt F, D'Alessio AC, Young RA and Weinberg RA:
Poised chromatin at the ZEB1 promoter enables breast cancer cell
plasticity and enhances tumorigenicity. Cell. 154:61–74. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liem KF Jr, Jessell TM and Briscoe J:
Regulation of the neural patterning activity of sonic hedgehog by
secreted BMP inhibitors expressed by notochord and somites.
Development. 127:4855–4866. 2000.PubMed/NCBI
|
|
34
|
Fischer KR, Durrans A, Lee S, Sheng J, Li
F, Wong ST, Choi H, El Rayes T, Ryu S, Troeger J, et al:
Epithelial-to-mesenchymal transition is not required for lung
metastasis but contributes to chemoresistance. Nature. 527:472–476.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zheng X, Carstens JL, Kim J, Scheible M,
Kaye J, Sugimoto H, Wu CC, LeBleu VS and Kalluri R:
Epithelial-to-mesenchymal transition is dispensable for metastasis
but induces chemoresistance in pancreatic cancer. Nature.
527:525–530. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Garg M: Epithelial-mesenchymal
transition-activating transcription factors-multifunctional
regulators in cancer. World J Stem Cells. 5:188–195. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Yuan JH, Yang F, Wang F, Ma JZ, Guo YJ,
Tao QF, Liu F, Pan W, Wang TT, Zhou CC, et al: A long noncoding RNA
activated by TGF-β promotes the invasion-metastasis cascade in
hepatocellular carcinoma. Cancer Cell. 25:666–681. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Li W and Kang Y: A new Lnc in metastasis:
Long noncoding RNA mediates the prometastatic functions of TGF-β.
Cancer Cell. 25:557–559. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Shang Y, Cai X and Fan D: Roles of
epithelial-mesenchymal transition in cancer drug resistance. Curr
Cancer Drug Targets. 13:915–929. 2013. View Article : Google Scholar : PubMed/NCBI
|