Introduction
Breast cancer is the most common type of cancer in
females and one of the most devastating cancers worldwide (1,2). Although
the underlying mechanism that causes breast cancer remains
uncertain, substantial evidence suggests that the Notch signaling
pathway may serve an important role in breast cancer pathogenesis
and, thus, may be a novel therapeutic target (3,4).
The Notch pathway serves an important role in normal
breast cell development, cell fate determination and stem cell
self-renewal (3). It is also
implicated in breast cancer development and progression as the
aberrant activation of this pathway is associated with breast
cancer (3–5). Inhibition of Notch signaling by gamma
secretase inhibitors, anti-Notch1 or anti-delta-like 4 (DLL4)
monoclonal antibodies have been revealed to result in antitumor
activity in a variety of tumors, including T-cell acute
lymphoblastic leukemia (T-ALL) and solid tumors, through multiple
mechanisms inclusive of the induction of cell cycle arrest or
apoptosis, and also the disruption of angiogenesis (4–10). Notch
signaling serves important roles in various physiological and
pathological processes including cell differentiation,
proliferation, invasion, angiogenesis, tumor metastasis and
apoptosis, which contribute to the development of various types of
human cancer (5,11). Furthermore, an activated Notch
signaling pathway and its target genes are commonly observed in
breast cancer, and the upregulation of Notch1 expression has
been revealed to protect breast cancer cells from apoptosis
(12). Currently known gene targets
for Notch signaling include the hairy enhancer of split
(Hes) genes, p21, cyclinD1, c-Myc, nuclear factor κB,
B-cell lymphoma-2 (Bcl-2) and Bcl-extra large
(Bcl-xl) (13–18).
Previous studies suggest that xanthohumol (XN), a
prenylated chalcone derived from hops (Humulus lupulus), may
inhibit cell growth and induce apoptosis in numerous types of human
cancer, including breast, prostate, leukemia and colon cancer cells
(19–23); however, the underlying mechanism of
this inhibition remains unknown. In the present study, the
therapeutic potential of XN in breast cancer cell lines was
investigated, focusing on its ability to inhibit breast cancer cell
proliferation, cell cycle arrest, apoptosis induction in
vitro and slowing tumor growth in vivo. Additionally,
possible XN-mediated inhibition in human breast cancer growth via
the regulation of the Notch1 signaling pathway was
investigated. To the best of our knowledge, the present study was
the first to observe that XN suppressed breast carcinoma growth by
inhibiting the Notch signaling pathway in vitro and in
vivo. Therefore, blocking of the Notch signaling pathway may be
a novel therapeutic approach for the treatment of breast cancer, by
inhibiting tumor development, progression and metastasis.
Materials and methods
Cell lines and cell culture
Human MCF-7, MDA-MB-231 and HEK-293T cells,
h-TERT-BJ, MCF-10A and murine 4T1 cells were obtained from the
American Type Culture Collection (Manassas, VA, USA). They were
cultured in Dulbecco's modified Eagle's medium (DMEM; Gibco; Thermo
Fisher Scientific, Inc., Waltham, MA, USA) supplemented with 10%
fetal bovine serum (FBS; Thermo Fisher Scientific, Inc.) and
maintained at 37°C in a humidified atmosphere of 5% CO2
in air.
Animals
All studies evaluating in vivo toxicity and
therapeutic effectiveness were performed using 26 female BALB/c
mice (17–18 g; 8 weeks of age) and obtained from Lanzhou Veterinary
Research Institute, Chinese Academic of Agricultural Sciences
(Lanzhou, China). The Institutional Animal Care and Use Committee
of Lanzhou University approved use of BALB/c mice for the present
study, and procedures involving animals and their care complied
with the Guide for the Care and Use of Laboratory Animals. Mice
were maintained in a temperature-controlled (23±4°C) environment
with a strict 12 h light/dark cycle. Food was purchased from
Wanqianjiaxing Biotech Corp (Wuhan, China) and water was
autoclaved. Food and water were freely available to the mice.
Reagents and antibodies
XN (purity 98.6%) was provided by Yumen Tuopu
Science Development and Technology Co., Ltd. (Yumen, China).
Duration of Dual Antiplatelet Therapy (DAPT) was purchased from
Sigma-Aldrich (Merck Millipore, Darmstadt, Germany). Primary
antibodies against Notch 1 (120 kDa; cat. no., 3268; dilution,
1:1,000), p21 (cat. no., 2947; dilution, 1:1,000), cyclin-dependent
kinase 4 (CDK4) (cat. no., 12790; dilution, 1:1,000), c-Myc (cat.
no., 5605; dilution, 1:1,000), survivin (cat. no., 2808; dilution,
1:1,000), Bcl-2 (cat. no., 15071; dilution, 1:500), Bcl-xL (cat.
no., 2764; dilution, 1:1,000), cyclin D1 (cat. no., 2978; dilution,
1:1,000), caspase-3 (cat. no., 9665; dilution, 1:1,000) and poly
(ADP-ribose) polymerase (PARP; cat. no., 9532; dilution, 1:1,000)
were purchased from Cell Signaling Technology, Inc. (Danvers, MA,
USA). Hes1 (cat. no., ABIN2779597; dilution, 1:1,000) was acquired
from Abnova Biotechnology (Taipei, Taiwan ROC). GAPDH (cat. no.,
sc-47724; dilution, 1:5,000) was supplied by Santa Cruz
Biotechnology, Inc. (Dallas, TX, USA). The secondary antibodies,
peroxidase-conjugated AffiniPure goat anti-rabbit (cat. no.,
ZB-2301; dilution, 1:500) and anti-mouse (cat. no., ZB-2305;
dilution, 1:500) IgG (H+L), were purchased from ZSGB-Bio (Beijing,
China).
MTT assay
The h-TERT-BJ, MCF-10A, MCF-7 and MDA-MB-231 cells
(5×103) were seeded in a 96-well culture plate;
following a 24-h incubation the cells were treated with 5, 10, 15
and 25 µmol/l XN for 24 and 48 h in incubator. Control cells were
treated with 0.1% dimethyl sulfoxide (DMSO) in culture medium.
Following treatment, the cells were incubated with MTT reagent (0.5
mg/ml) at 37°C for 4 h. The resulting formazan crystals were
solubilized by the addition of 200 µl DMSO to each well. The
absorbance was read at 490 nm in a Vector3 Multilevel Plate Counter
(PerkinElmer, Inc., Waltham, MA, USA) and all MTT experiments were
performed in triplicate and repeated ≥3 times.
Transient transfection and luciferase
reporter assay
Transient transfections were performed with
Lipofectamine® (Invitrogen; Thermo Fisher, Inc.),
according to the manufacturer's protocol. Briefly, HEK-293T cells
were plated in 24-well plates at a density of 1×105
cells/well. Following a 24-h incubation at 37°C, cells were
transfected with plasmids of 0.8 µg promoter-linked luciferase
vector (23A, 4xCBF1 binding element plasmid) and 0.2 µg pGL4.20
vector for 4 h in DMEM (Sigma-Aldrich; Merck Millipore). Following
DLL-4 (100 ng/ml) stimulation, cells were treated with XN for 12 h
at 37°C. The cell lysates were evaluated in a luciferase assay
using a dual luciferase reporter assay kit (E1910; Promega,
Madison, WI, USA), and the emitted light was determined with a
luminometer (Wallac 1420 VICTOR, Inc., Waltham, MA, USA) as
previously described (24).
Luciferase activity was normalized to β-galactosidase and plotted
as relative light units.
Flow cytometry analysis of cell
cycle
The cell cycle was analyzed by flow cytometry. The
MCF-7 and MDA-MB-231 cells (1×106) were collected and
washed in PBS, prior to being fixed in 75% alcohol at 20°C
overnight. Following washing in cold PBS three times, cells were
resuspended in 1 ml PBS solution with 50 µg propidium iodide (PI;
Sigma-Aldrich; Merck Millipore) and 100 µg RNase A (Sigma-Aldrich;
Merck Millipore), for 30 min at 37°C. Samples were then analyzed
for their DNA content by fluorescence-activated cell sorting (FACS;
BD Biosciences, San Jose, CA, USA). Each experiment was repeated ≥3
times.
Flow cytometric analysis of
apoptosis
Cell apoptosis was assessed by flow cytometry using
an Annexin-V-fluorescein isothiocyanate/PI apoptosis detection kit
(BD Biosciences, San Jose, CA, USA), according to the
manufacturer's instructions. The pretreated MCF-7 and MDA-MB-231
cells were harvested and washed twice with PBS (4°C). Following
this, they were resuspended in 1X binding buffer at a concentration
of 1×106 cells/ml, stained with Annexin V/PI and kept on
ice for 30 min in the dark. The cells were then analyzed on a
FACSCalibur™ flow cytometer (BD Biosciences). Assays were performed
three times in triplicate.
Semi-quantitative reverse
transcription polymerase chain reaction (RT-PCR)
Total RNA was isolated from cell lines using an
RNeasy kit as described by the manufacturer (Qiagen Inc., Valencia,
CA, USA). The reverse transcription reaction was performed using
the SuperScript® First-Strand Synthesis System
(Invitrogen; Thermo Fisher Scientific, Inc.) in a final volume of
20 µl, containing 5 µg total RNA, 200 ng random hexamers, 1X
reverse transcription buffer, 2.5 mM MgCl2, 1 mM
deoxynucleotide triphosphate mixture, 10 mM DTT, RNaseOUT
recombinant ribonuclease inhibitor (Invitrogen; Thermo Fisher
Scientific, Inc.), 50 U superscript reverse transcriptase and
diethylpyrocarbonate-treated water. Following incubation at 42°C
for 50 min, the reverse transcription reaction was terminated by
heating to 85°C for 5 min. The newly synthesized cDNA was amplified
by PCR. The reaction mixture contained 2 µl cDNA template, 1.5 mM
MgCl2, 2.5 U Taq polymerase and 0.5 µM primers (primers are
presented in Table I). The reactions
were performed under the following conditions: 5 min at 94°C, 30
sec at 94°C, 30 sec at 55°C, 30 sec at 72°C, 25 cycles of 30 sec at
94°C, 5 min at 72°C and 30 min at 4°C. The mRNA levels of Hes1 and
Hes related family BHLH transcription factor with YRPW motif 1
(Hey1) were normalized to GAPDH.
 | Table I.Primer sequences for reverse
transcription-polymerase chain reaction. |
Table I.
Primer sequences for reverse
transcription-polymerase chain reaction.
| Gene | Upstream
primer | Downstream
primer |
|---|
| Hes1 |
5′-TTGGAGGCTTCCAGGTGGTA-3′ |
5′-GGCCCCGTTGGGAATG-3′ |
| Hey1 |
5′-CGAGGTGGAGAAGGAGAGTG-3′ |
5′-CTGGGTACCAGCCTTCTCAG-3′ |
| GAPDH |
5′-TCTCATCACCATCTTCCA-3′ |
5′-CATCACGCCACAGTTTCC-3′ |
Boyden chamber assay
For Boyden chamber assays, 1×105
MDA-MB-231 cells were seeded per well in a Boyden chamber with a
pore size of 8 µM (Corning Incorporated, Corning, NY, USA), without
fetal bovine serum. For the control and treated samples (following
XN and DAPT treatment), the lower compartment was filled with
medium containing 10% FBS. Following 6 or 12 h, cells were fixed
with 4% paraformaldehyde solution and stained with crystal violet
(Beyotime Institute of Biotechnology, Haimen, China; C0121). Cells
on top of the filter were removed with a cotton bud and the bottom
sides were photographed using a bright field light microscope (4×
objective). For quantification, the membranes sectioned, and 0.1 ml
33% acetic acid elution was used to dissolve the cells, then 40 µl
was added into 96 plates and the values were determined at 570
nm.
Western blot analysis
Western blotting was performed as described in a
previous study (24). Briefly, total
proteins from the two human breast cancer cell lines were lysed in
lysis buffer (cat. no., P00138; Beyotime Institute of
Biotechnology) and incubated for 15 min at 4°C. The concentrations
of total proteins were determined using a bicinchoninic acid assay
protein assay kit (Thermo Fisher Scientific, Inc.). Total proteins
were fractionated using 10% SDS-PAGE and the gels were then
transferred onto a nitrocellulose membrane. The membranes were
blocked with 5% non-fat milk in Tris-buffered saline containing
0.1% Tween-20 prior to incubation with the appropriate primary
antibodies overnight at 4°C. Horseradish peroxidase-conjugated
anti-goat IgG was used as the secondary antibody, and the protein
bands were detected using the electrochemiluminescence (ECL) method
(cat. no., RPN2134; Western Blotting Detection Reagent, GE
Healthcare Life Sciences, Chalfont, UK). Western blot analysis was
quantified by laser densitometry, and the results are presented as
the mean of three independent experiments with error bars
representing the standard deviation.
In vivo tumorigenicity assays and
immunohistochemistry
Firstly, 2×105 4T1 tumor cells from
cultures, suspended in 0.2 ml DMEM without FBS, were injected into
the right flank of 8-week-old BALB/c mice. Tumor-bearing mice were
randomly assigned in three groups [n=10 control mice; n=16 XN mice
(8 received 100 mg/kg XN and 8 received 200 mg/kg XN)]. Following
24-h, the mice were gavaged with 200 µl vehicle or 200 µl XN at the
appropriate concentrations (200 and 100 mg/kg). Intragastric
administration was performed once a day for two weeks and the mice
were weighed daily. Following two weeks, all of the mice were
sacrificed by breaking of the neck and the stripped subcutaneous
sarcorna tumors were subsequently weighed to evaluated the
antitumor effect. Tumors treated with 200 mg/kg XN were stored in
formaldehyde for immunohistochemistry as described in a previous
study (25).
Statistical analysis
The statistical analysis was performed using SPSS,
Inc., version 12.0 software (Chicago, IL, USA). All data were
expressed as the mean ± SD. Student's t-test was performed for
comparisons between the two treatment groups. The three treatment
groups were compared by one-way analysis of variance, followed by
the significant difference method for multiple comparisons.
P<0.05 was considered to indicate a statistically significant
difference and P<0.01 was considered to indicate a highly
statistically significant difference.
Results
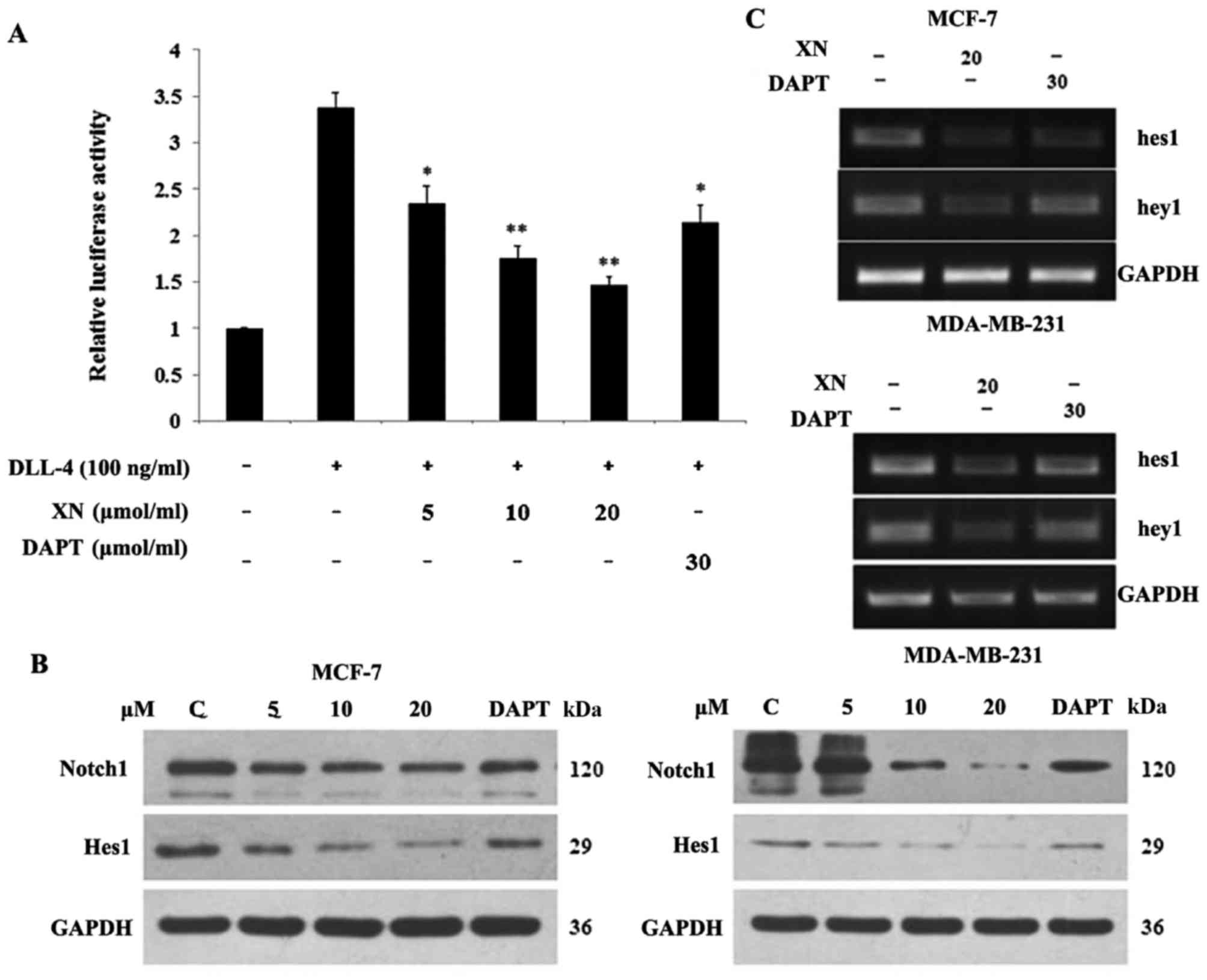
XN regulates Notch1 signal pathway
activity and downstream targets
In order to determine if the Notch pathway was
targeted by XN, a Notch1 functional assay and DAPT (gamma secretase
inhibitor) was used as a positive control. Upon binding of Notch1
to the CBF1 transgene, luciferase activity may be determined in the
documented reporter assay. The aim of the present study was to
determine if XN reduced the binding activity of Notch1 to CBF1. The
cells were stimulated with/without DLL-4 (100 ng/ml). The
stimulated groups were treated with XN (5, 10, 20 µM) and DAPT (30
µM). Relative to the control, a 62% and a 34% decrease in the
luciferase signal were identified following treatment with XN (20
µM) and DAPT (30 µM), respectively, for 24 h in 293T cells
(Fig. 1A). Subsequently, the level of
XN, which was associated with a reduction in Notch 1 protein
expression, was investigated. Cells were harvested following 24 h
of treatment and analyzed by western blotting with antibodies
specific for Notch 1 and Hes1 (Fig.
1B). Notch 1 and Hes1 expression levels decreased in a
dose-dependent manner in both cell lines. This reduction in Notch 1
activity led to an evaluation into whether XN was functioning
similar to a γ-secretase inhibitor, or if it is operating at the
transcriptional level. RNA was isolated following one day of XN or
DAPT treatment, and RT-PCR was performed. Lastly, the downstream
target genes of Notch were investigated. It was revealed that
treatment with XN resulted in decreased Hes1 and Hey1 transcription
in MCF-7 and MDA-MB-231 cells. Concomitantly, a decrease in Hes1
and Hey1 transcription in MCF-7 and MDA-MB-231 cells was observed
following treatment with DAPT (Fig.
1C).
XN inhibits cell growth and migration
in breast cancer cell lines
The treatment of MCF-7 and MDA-MB-231 breast cancer
cells for 24–48 h with 5, 10, 15, 20 and 25 µM XN resulted in cell
growth inhibition in a dose- and time-dependent manner, and the
inhibition was revealed to be more pronounced with 25 µM XN
treatment by an MTT assay (Fig. 2A,
left). Comparison of the effects on cell viability among MCF-7,
MDA-MB-231, hTERT-BJ and MCF-10A cells following 24 and 48 h drug
treatments (Fig. 2A, right),
demonstrated that XN induced a higher level of apoptosis for DU145
and MDA-MB-231 cells compared with hTERT-BJ and MCF-10A cells,
subsequent to treatment (Fig. 2A).
The inhibition of cell growth was also identified by viewing with
an inverted microscope (×10 magnification; Fig. 2B).
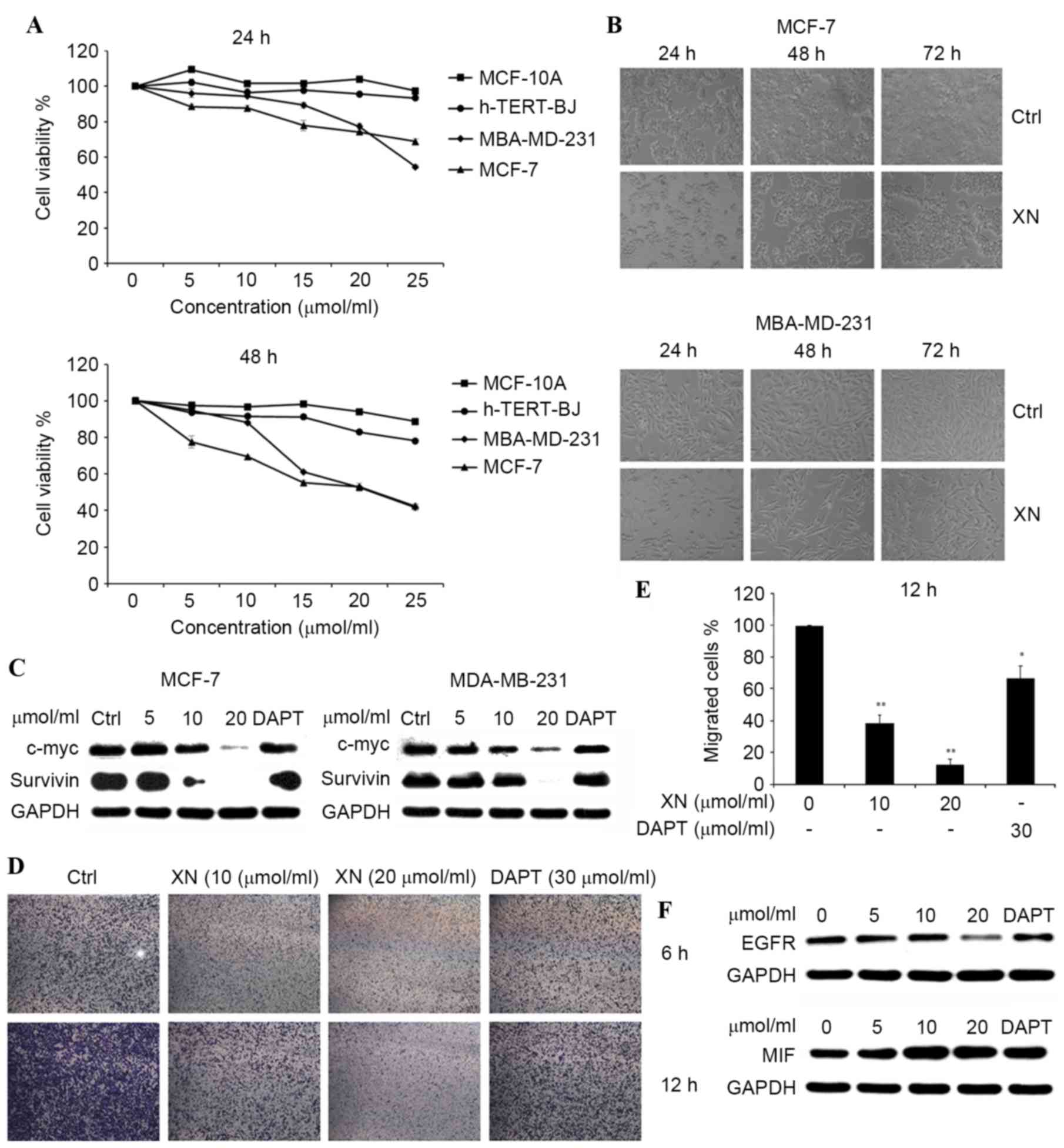 | Figure 2.XN inhibited breast cancer cell
proliferation in a dose- and time-dependent manner. (A) h-TERT-BJ,
MCF-10A, MCF-7 and MDA-MB-231 were plated equally (six replicates)
and treated with XN. MTT assays were performed daily for two days.
DMSO was a vehicle control. (B) MDA-MB-231 and MCF-7 cells were
treated with 20 µM XN for 24–72 h and visualized by light
microscopy. (C) The c-Myc and survivin protein expression levels
were detected by western blotting. (D) The Boyden chamber transwell
assay demonstrated MDA-MB-231 cell migration. MCF-7 and MDA-MB-231
cells were treated with XN (data are presented as the mean ± SEM of
three independent experiments). (E) Quantification of migrated
MDA-MB-231 cells. Data are presented as the mean ± SEM of three
independent experiments. *P<0.05, **P<0.01 vs. control. (F)
EGFR and MIF expression levels were determined by western blot
analysis. SEM; standard error of the mean; DMSO, dimethyl
sulfoxide; EGFR, epidermal growth factor receptor; MIF, tumor
metastasis-associated protein; CDK4, cyclin-dependent kinase 4;
DAPT, Duration of Dual Antiplatelet Therapy; XN, xanthohumol; Ctrl,
control. |
Western blot analysis of c-Myc and
survivin was also performed. It has been observed that
expression of c-Myc and survivin were gradually
decreasing compared with the control in both cell lines, following
treatment with XN for 24 h (Fig. 2C).
These results indicated that XN may inhibit breast cancer cell
growth and migration by blocking the Notch 1 signaling pathway.
The effect of XN on the transmigration of MDA-MB-231
cells was determined with a Boyden chamber assay. Cell migration
was inhibited significantly by XN treatment at 10 or 20 µM, down to
40 or 30%, respectively. Similar effects (83%) were observed with
DAPT treatment, which was used as positive control (Fig. 2D and E). The effect of XN on epidermal
growth factor receptor (EGFR) and tumor metastasis-associated
protein (MIF) was determined by using a western blot assay. There
was a notable decrease in EGFR expression levels and an increase in
MIF expression levels, as the concentration of XN increased in the
control and treatment groups in MDA-MB-231 cells (Fig. 2F).
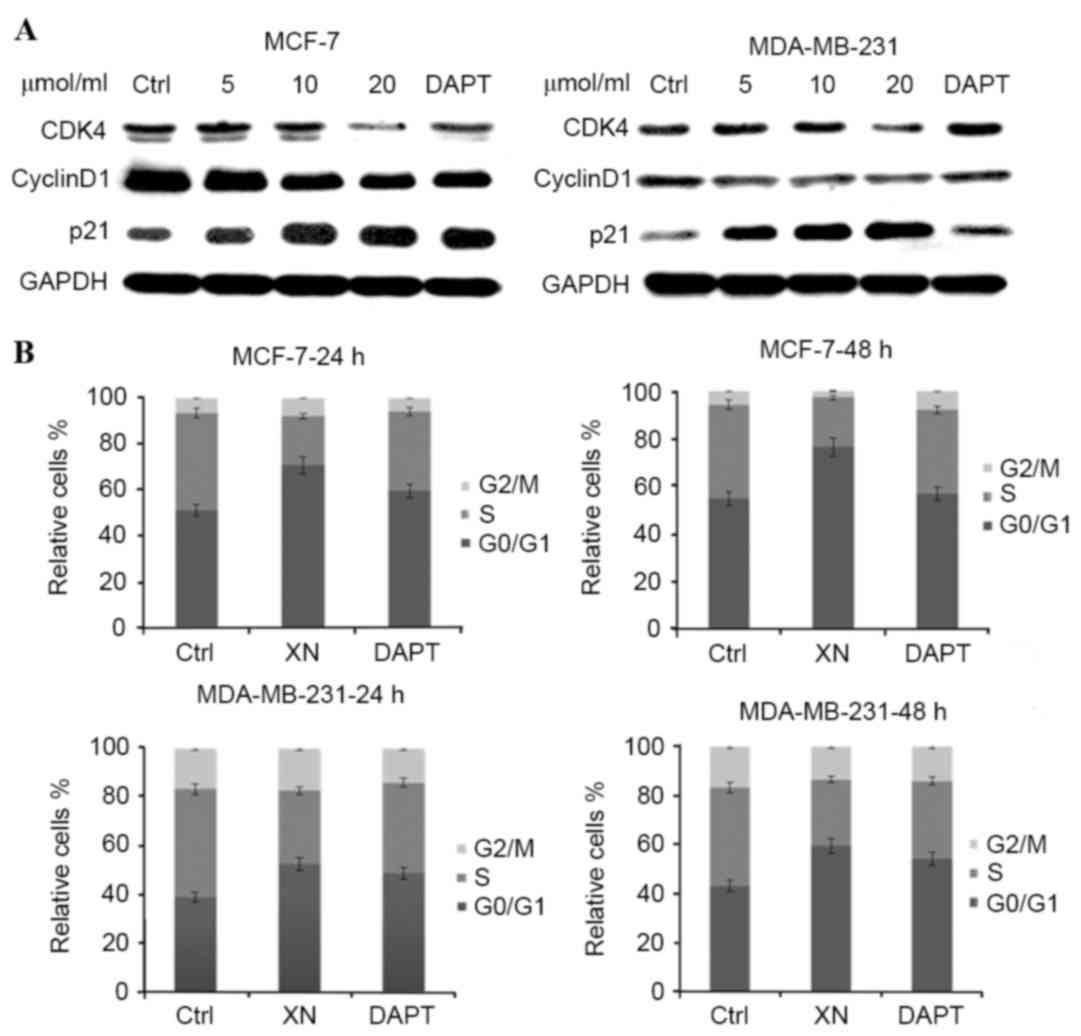
XN treatment is associated with cell
cycle arrest and apoptosis
To determine whether cell cycle arrest was an
underlying mechanism by which XN may be effecting reduced cell
growth in breast cancer, the present study performed western blot
analysis of certain cell cycle associated proteins. There was a
notable increase in p21WAF1/CIP1 expression levels in MCF-7 cells
as the concentration of XN treatment increased, in addition to an
increase in p21WAF1/CIP1 expression levels between the control and
treatment groups in MDA-MB-231 cells (Fig. 3A). As anticipated, from an increase in
p21WAF1/CIP1 expression levels, flow cytometry experiments revealed
that cells responded in time-dependent and dose-dependent manners
to XN, through arresting cells in the G0/G1
phase of the cell cycle (Fig. 3B).
Cells were treated with XN for 24 or 48 h, and the percentage of
control cells in G0/G1 phase was 51.1 and
10%; however, following treatment with 20 µM XN, 70.6 and 24.8% of
the cells were in the G0/G1 phase. Cell cycle
arrest was confirmed by a decrease in CDK4 and cyclin D1 expression
levels in the MCF-7 and MDA-MB-231 cells (Fig. 3A). As a consequence of
G0/G1 phase cell cycle arrest, the present
study investigated whether XN inhibits breast cancer cells by
inducing apoptosis; this was determined by an Annexin V/PI assay
and cleavage of PARP-1, caspase-3 and Bcl-xL and Bcl-2 expression
levels. As presented in Fig. 4 (a
representative experiment), following exposure to various media
(DMEM, XN or DAPT for 48 h, the percentage of apoptotic cells in
the XN treated groups increased significantly in comparison with
the control groups. DAPT was also revealed to induce the apoptosis
of breast cancer cells; however, the XN treatment groups contained
a higher number of apoptotic cells compared with the DAPT treated
groups. Apoptosis assessed by western blot assay demonstrated the
same trends as the Annexin-V/PI assay (Fig. 4A and B). An increase in cleaved
caspase-3 and cleaved PARP was revealed with increasing doses of XN
and DAPT, suggesting that the cells went through the caspase
cascade-mediated apoptotic pathway (Fig.
4C). These results indicated that XN exerts an inhibitory
effect on cell proliferation and also promotes cell apoptosis.
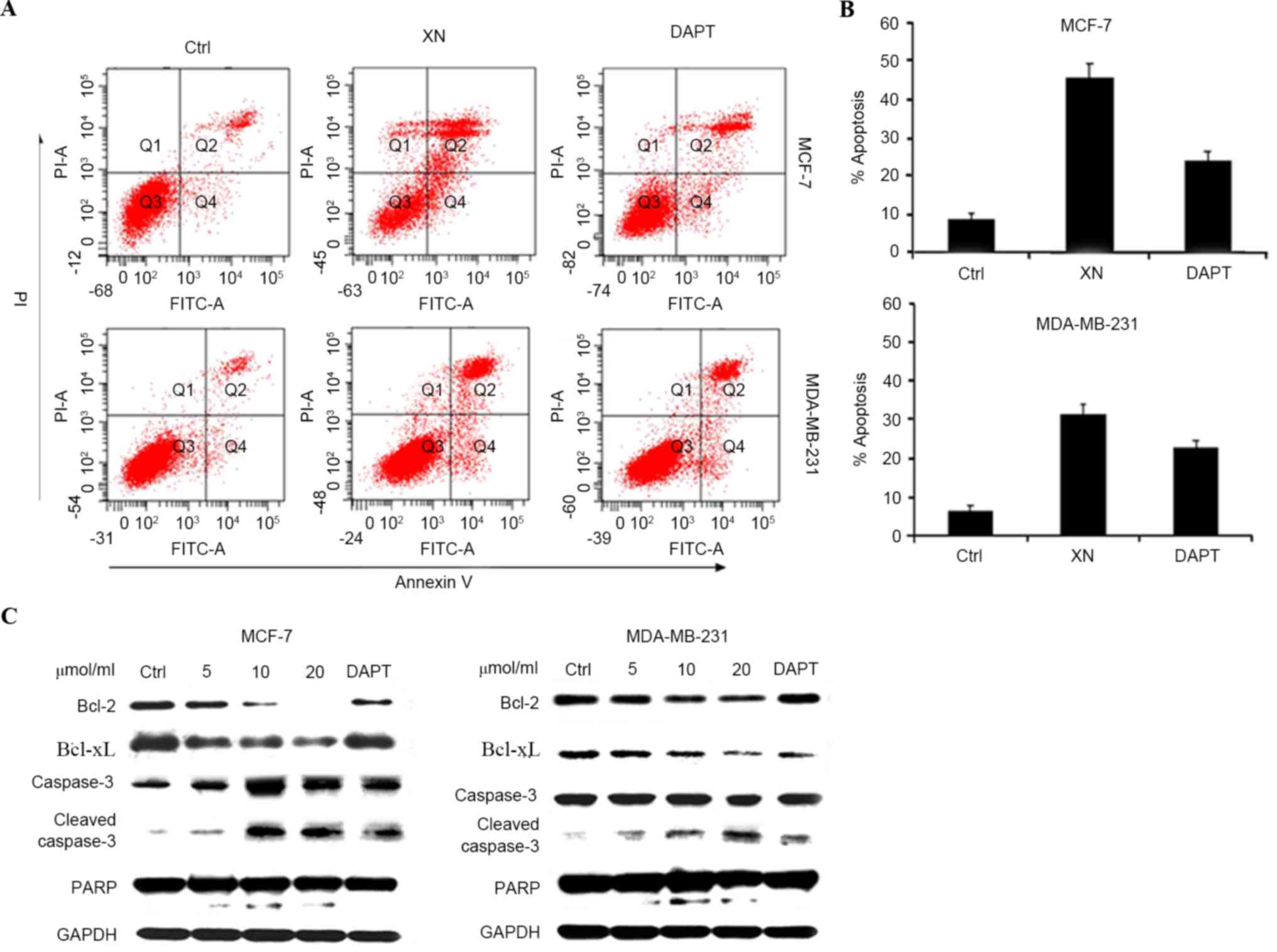 | Figure 4.XN promote apoptosis of breast cells.
(A) MCF-7 and MDA-MB-231 cells were harvested for apoptotic
analysis using Annexin V-FITC staining. (B) Quantification of
apoptosis. Data are presented as the mean ± SD error of the mean of
three independent experiments. (C) Apoptosis associated proteins
Bcl-2, Bcl-xL and caspase-3 and cleaved PARP were analyzed by
western blotting. GAPDH was used as the control. FITC, fluorescein
isothiocyanate; FITC-A, FITC-annexin; Ctrl, control; XN,
xanthohumol; DAPT, Duration of Dual Antiplatelet Therapy; PI-A,
propidium iodide annexin; Bcl-2, B cell lymphoma-2; Bcl-xL, B cell
lymphoma extra 1; PARP, poly (ADP-ribose) polymerase. |
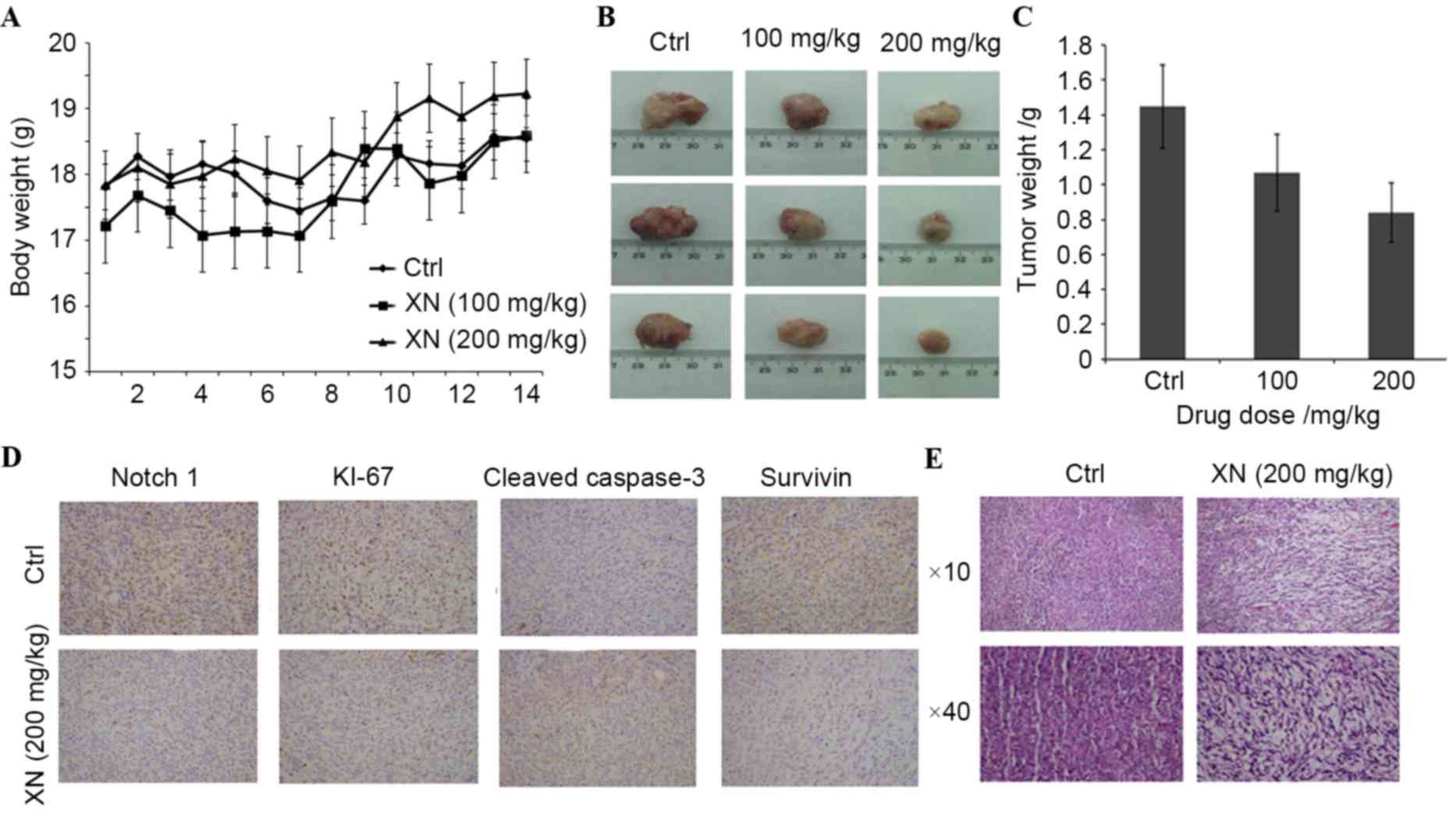
XN effects mouse tumor growth and
Notch 1 protein expression levels in vivo
In order to evaluate whether XN downregulation
affects tumor growth in vivo, BALB/c mice were used as an
in vivo model of mammary carcinoma. A mouse tumor model was
generated by endermic injection of 4T1 murine mammary cells. Mice
treated by oral gavage with XN three times a week did not exhibit
any symptoms of toxicity, and no effects were observed in the body
weight profiles when XN treated mice were compared with vehicle-fed
controls (data not presented). At the time of necropsy, all animals
were examined for gross pathology, and no evidence of edema or
abnormal organ size in target and non-target organs was
observed.
There was a significant difference in mouse tumor
model weight between the control and the 200 mg/kg XN-fed group
(Fig. 5A). Additionally, a marked
decrease in tumor size in a dose dependent manner was revealed
(Fig. 5B): Tumor weight decreased
27.22 and 46.79% following various XN treatments (100 and 200
mg/kg), respectively (Fig. 5C). These
results demonstrated that XN effectively inhibited breast tumor
growth in vivo.
In the present study, the possibility of XN serving
a role in Notch1-associated protein expression in
vivo was investigated. The expression levels of Notch 1, Ki-67,
survivin and caspase-3 by immunohistochemistry was determined.
Fig. 5D demonstrates that XN inhibits
the expression of Notch1 and Ki-67. Survivin was
downregulated and cleaved caspase-3 was upregulated in XN treated
tissues (Fig. 5D). Hematoxylin and
eosin staining revealed that XN produced obvious cell injury in the
tumor tissues of mice (Fig. 5E).
These results demonstrated that XN efficiently inhibited the growth
of tumors and promoted tumor cell apoptosis, which further
indicates that XN has an antitumor effect.
Discussion
The Notch signaling pathway decides cell fate, an
important carcinogenic factor of breast cancer (7,26).
Notch1 expression levels are connected to breast cancer cell
proliferation and survival (27).
Farnie and Clarke (28) reported that
aberrant activation of the Notch signaling pathway is an early
event in breast cancer, and high expression levels of the Notch 1
intracellular domain was indicative of a reduced time to five-year
post-surgical recurrence. Therefore, the Notch signaling pathway
may represent a breast cancer novel therapeutic target. Notch
signaling may be downregulated via inhibition of Notch ligands,
inhibiting tumor cell proliferation (29). G-secretase inhibitors may be used to
block Notch signaling and have previously been applied in clinical
studies (30,31). However, one of the major challenges is
to eliminate unwanted toxicity associated with γ-secretase
inhibitors (32); thus, alternative
Notch inhibitors must be identified.
XN, a product of hops, may inhibit cell growth and
induce apoptosis in numerous types of human cancer, including
breast cancer (19–23,33).
Previous studies into the biological activity of XN have revealed
their antiproliferative activity and possible antitumor activity
(34). Guerreiro et al
(33) suggested that XN may modulate
alkaline phosphatase isoenzymes in MCF-7 breast cancer cells, and
that alkaline phosphatase loss was associated with increased cell
proliferation. In 2012, Cho et al (35) reported that XN induced cancer cell
specific apoptosis in MCF-7 human breast cancer cells. These
results raise the possibility that XN may be a potential
therapeutic agent in human breast cancer. There are currently few
studies that address this question directly and this appears to be
an area worthy of further investigation.
Notch1 acts as an oncogene in breast cancer;
therefore, inhibition of Notch1 expression may lead to
inhibition of cell growth and apoptotic cell death in breast cancer
cells (10,33). The present study demonstrated that
proliferation inhibition and apoptosis induction by XN are specific
to cancer cells with a constitutively inhibited Notch 1
signaling pathway; DAPT was used as a positive control. An MTT
assay and light microscopy (Fig. 2A and
B) indicated that XN inhibited cell proliferation in breast
cancer cell lines. Proliferation associated proteins, including
c-Myc and survivin, expression levels were determined (Fig. 2C). MD-MBA-231 cells, invasive breast
cancer cells, were used to assess the ability to modulate cell
migration through Boyden chamber porous membranes. As presented in
Fig. 2D, cells treated with XN
prevented cell migration to the lower chamber in a Boyden chamber
assay, which was significantly different compared with the control.
Activation of the EGFR induced signaling pathway correlated with
cancer metastasis in various tumors, including breast carcinoma
(36). Dai et al (37) reported that Notch and EGFR signaling
pathways are positively associated with human breast cancer. The
Notch signaling pathway inhibitor may also inhibit EGFR expression.
Furthermore, additional factors, including MIF, which inhibits cell
migration and XN, increased MIF expression.
The results of the present study indicate that MCF-7
and MDA-MB-231 cells were inhibited in the
G0/G1 phase of the cell cycle, and underwent
induced apoptosis when treated with XN. Downstream target genes of
the Notch signaling pathway are also involved in cell cycle arrest
and apoptotic pathway (14,15,18).
Cyclin D1 and CDK4 phosphorylate key cell cycle proteins,
controlling the G1-S cell cycle phases; p21 is an
ubiquitous inhibitor of kinase CDK, which guides cell cycle arrest
(38–40). The Hes/Hey genes are Notch
target genes, which are basic helix-loop-helix repressors (4). A number of additional genes, including
p21 and cyclin D1, have been suggested to be direct targets of the
Notch signaling pathway (13,14).
In breast cancer, the nuclear antigen Ki-67 has
previously been applied widely for comparison of cell proliferation
between tumor samples in immunohistochemical assessment (41,42). The
immunohistochemical evaluations revealed that XN decreased Notch 1,
Ki-67 and survivin expression levels, and increased caspase-3
expression. The present study demonstrated that XN may inhibit
breast tumor growth and promote apoptosis (Fig. 5).
In conclusion, the present study revealed that XN
possesses antiproliferative, anti-metastatic and pro-apoptotic
effects in breast cancer cells. It was also revealed that XN
inhibits tumor growth using an in vivo tumor growth assay.
The results of the present study demonstrated that XN may be a
promising chemopreventive candidate for breast cancer treatment via
inhibition of the Notch signaling pathway. These results suggest
that XN requires further study into its potential therapeutic role
in breast cancer treatment.
Acknowledgements
The present study was supported by the Science and
Technology Support Projects of Gansu Province, China (grant no.
2010GS05414). The authors would like to thank Dr Yumen Tuopu,
Science Development and Technology Co., Ltd., for providing XN, and
Dr. Diane Hayward (The Johns Hopkins University, Baltimore, MD,
USA) for providing the 23A plasmid, which contained four copies of
the CBF1-binding elements in pGL2 Luciferase Reporter Vectors.
References
|
1
|
Guo H, Wu F, Wang Y, Yan C and Su W:
Overexpressed ubiquitin ligase Cullin7 in breast cancer promotes
cell proliferation and invasion via down-regulating p53. Biochem
Biophys Res Commun. 450:1370–1376. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Keyaerts M, Xavier C, Heemskerk J,
Devoogdt N, Everaert H, Ackaert C, Vanhoeij M, Duhoux FP, Gevaert
T, Simon P, et al: Phase I study of 68Ga-HER2-Nanobody for PET/CT
assessment of HER2-expression in breast carcinoma. J Nucl Med.
57:27–33. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Al-Hussaini H, Subramanyam D, Reedijk M
and Sridhar SS: Notch signaling pathway as a therapeutic target in
breast cancer. Mol Cancer Ther. 10:9–15. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yuan X, Zhang M, Wu H, Xu H, Han N, Chu Q,
Yu S, Chen Y and Wu K: Expression of Notch1 correlates with breast
cancer progression and prognosis. PLoS One. 10:e01316892015.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Roy M, Pear WS and Aster JC: The
multifaceted role of Notch in cancer. Curr Opin Genet Dev.
17:52–59. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Palmer WH and Deng WM: Ligand-independent
mechanisms of Notch activity. Trends Cell Biol. 25:697–707. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Artavanis-Tsakonas S, Rand MD and Lake RJ:
Notch signaling: Cell fate control and signal integration in
development. Science. 284:770–776. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kopan R and Ilagan MX: The canonical Notch
signaling pathway: Unfolding the activation mechanism. Cell.
137:216–233. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Radtke F and Raj K: The role of Notch in
tumorigenesis: Oncogene or tumour suppressor. Nat Rev Cancer.
3:756–767. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Robinson DR, Kalyana-Sundaram S, Wu YM,
Shankar S, Cao X, Ateeq B, Asangani IA, Iyer M, Maher CA, Grasso
CS, et al: Functionally recurrent rearrangements of the MAST kinase
and Notch gene families in breast cancer. Nat Med. 17:1646–1651.
2011. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liao WR, Hsieh RH, Hsu KW, Wu MZ, Tseng
MJ, Mai RT, Wu Lee YH and Yeh TS: The CBF1-independent Notch1
signal pathway activates human c-myc expression partially via
transcription factor YY1. Carcinogenesis. 28:1867–1876. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xia J, Li Y, Yang Q, Mei C, Chen Z, Bao B,
Ahmad A, Miele L, Sarkar FH and Wang Z: Arsenic trioxide inhibits
cell growth and induces apoptosis through inactivation of Notch
signaling pathway in breast cancer. In J Mol Sci. 13:9627–9641.
2012. View Article : Google Scholar
|
|
13
|
Rangarajan A, Talora C, Okuyama R, Nicolas
M, Mammucari C, Oh H, Aster JC, Krishna S, Metzger D, Chambon P, et
al: Notch signaling is a direct determinant of keratinocyte growth
arrest and entry into differentiation. EMBO J. 20:3427–3436. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ling H and Jolicoeur P: Notch-1 signaling
promotes the cyclinD1-dependent generation of mammary
tumor-initiating cells that can revert to bi-potential progenitors
from which they arise. Oncogene. 32:3410–3419. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Palomero TL, Lim WK, Odom DT, Sulis ML,
Real PJ, Margolin A, Barnes KC, O'Neil J, Neuberg D, Weng AP, et
al: Notch1 directly regulates c-MYC and activates a
feed-forward-loop transcriptional network promoting leukemic cell
growth. Proc Natl Acad Sci USA. 103:pp. 18261–18266. 2006;
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ramdass B, Maliekal TT, Lakshmi S, Rehman
M, Rema P, Nair P, Mukherjee G, Reddy BK, Krishna S and
Radhakrishna Pillai M: Coexpression of Notch1 and NF-kappaB
signaling pathway components in human cervical cancer progression.
Gynecol Onco. 104:352–361. 2007. View Article : Google Scholar
|
|
17
|
Xie M, He CS, Wei SH and Zhang L: Notch-1
contributes to epidermal growth factor receptor tyrosine kinase
inhibitor acquired resistance in non-small cell lung cancer in
vitro and in vivo. Eur J Cancer. 49:3559–3572. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sionov RV, Kfir-Erenfeld S, Spokoini R and
Yefenof E: A role for bcl-2 in Notch1-dependent transcription in
thymic lymphoma cells. Adv Hematol. 2012:4352412012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Colgate EC, Miranda CL, Stevens JF, Bray
TM and Ho E: Xanthohumol, a prenylflavonoid derived from hops
induces apoptosis and inhibits NF-kappaB activation in prostate
epithelial cells. Cancer Lett. 246:201–209. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gerhauser C, Alt A, Heiss E, Gamal-Eldeen
A, Klimo K, Knauft J, Neumann I, Scherf HR, Frank N, Bartsch H and
Becker H: Cancer chemopreventive activity of Xanthohumol, a natural
product derived from hop. Mol Cancer Ther. 1:959–969.
2002.PubMed/NCBI
|
|
21
|
Gonçalves P, Araújo JR, Pinho MJ and
Martel F: In vitro studies on the inhibition of colon cancer by
butyrate and polyphenolic compounds. Nutr Cancer. 63:282–294. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Nozawa H: Xanthohumol, the chalcone from
beer hops (Humulus lupulus L.), is the ligand for farnesoid X
receptor and ameliorates lipid and glucose metabolism in KK-A(y)
mice. Biochem Biophys Res Commun. 336:754–761. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Pan L, Becker H and Gerhäuser C:
Xanthohumol induces apoptosis in cultured 40–16 human colon cancer
cells by activation of the death receptor- and mitochondrial
pathway. Mol Nutr Food Res. 49:837–843. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shi J, Chen J, Serradji N, Xu X, Zhou H,
Ma Y, Sun Z, Jiang P, Du Y, Yang J, et al: PMS1077 sensitizes TNF-α
induced apoptosis in human prostate cancer cells by blocking NF-κB
signaling pathway. PLoS One. 8:e611322013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Liu P, Zhao L, Xu X, Liu F, Zhang W, Zhou
C, Chen J, Pan Y, Du Y, Yang J and Wang Q: N6-substituted adenosine
analogues, a novel class of JAK2 inhibitors, potently block STAT3
signaling in human cancer cells. Cancer Lett. 354:43–57. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang X, Samadi AK, Roby KF, Timmermann B
and Cohen MS: Inhibition of cell growth and induction of apoptosis
in ovarian carcinoma cell lines CaOV3 and SKOV3 by natural
withanolide Withaferin A. Gynecol Oncol. 124:606–612. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Pei J and Wang B: Notch-1 promotes breast
cancer cells proliferation by regulating LncRNA GAS5. Int J Clin
Exp Med. 8:14464–14471. 2015.PubMed/NCBI
|
|
28
|
Farnie G and Clarke RB: Breast stem cells
and cancer. Ernst Schering Found Symp Proc. pp. 1–153. 2006;
|
|
29
|
Rasul S, Balasubramanian R, Filipović A,
Slade MJ, Yagüe E and Coombes RC: Inhibition of gamma-secretase
induces G2/M arrest and triggers apoptosis in breast cancer cells.
Br J Cancer. 100:1879–1888. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shih IeM and Wang TL: Notch signaling,
gamma-secretase inhibitors and cancer therapy. Cancer Res.
67:1879–1882. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Beel AJ and Sanders CR: Substrate
specificity of gamma-secretase and other intramembrane proteases.
Cell Mol Life Sci. 65:1311–1334. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
van Es JH, van Gijn ME, Riccio O, van den
Born M, Vooijs M, Begthel H, Cozijnsen M, Robine S, Winton DJ,
Radtke F and Clevers H: Notch/gamma-secretase inhibition turns
proliferative cells in intestinal crypts and adenomas into goblet
cells. Nature. 435:959–963. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Guerreiro S, Monteiro R, Martins MJ,
Calhau C, Azevedo I and Soares R: Distinct modulation of alkaline
phosphatase isoenzymes by 17beta-estradiol and xanthohumol in
breast cancer MCF-7 cells. Clin Biochem. 40:268–273. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mendes V, Monteiro R, Pestana D, Teixeira
D, Calhau C and Azevedo I: Xanthohumol influences preadipocyte
differentiation: Implication of antiproliferative and apoptotic
effects. J Agric Food Chem. 56:11631–11637. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Cho MY, Park SY, Park S, Lee YR, Han GD
and Kim JA: Geranyl derivative of phloroacetophenone induces cancer
cell-specific apoptosis through Bax-mediated mitochondrial pathway
in MCF-7 human breast cancer cells. Biol Pharm Bull. 35:98–104.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hsieh CY, Tsai PC, Tseng CH, Chen YL,
Chang LS and Lin SR: Inhibition of EGF/EGFR activation with
naphtho[1,2-b]furan-4,5-dione blocks migration and invasion of
MDA-MB-231 cells. Toxicol In Vitro. 27:1–10. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Dai J, Ma D, Zang S, Guo D, Qu X, Ye J and
Ji C: Cross-talk between Notch and EGFR signaling in human breast
cancer cells. Cancer Invest. 27:533–540. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lange C, Huttner WB and Calegari F:
Cdk4/cyclinD1 overexpression in neural stem cells shortens G1,
delays neurogenesis, and promotes the generation and expansion of
basal progenitors. Cel Stem Cell. 5:320–331. 2009. View Article : Google Scholar
|
|
39
|
Waga S, Hannon GJ, Beach D and Stillman B:
The p21 inhibitor of cyclin-dependent kinases controls DNA
replication by interaction with PCNA. Nature. 369:574–578. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Xiong Y, Hannon GJ, Zhang H, Casso D,
Kobayashi R and Beach D: p21 is a universal inhibitor of cyclin
kinases. Nature. 366:701–704. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Dowsett M, Nielsen TO, A'Hern R, Bartlett
J, Coombes RC, Cuzick J, Ellis M, Henry NL, Hugh JC, Lively T, et
al: Assessment of Ki67 in breast cancer: Recommendations from the
International Ki67 in Breast Cancer working group. J Natl Cancer
Inst. 103:1656–1664. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Basu S, Combe K, Kwiatkowski F,
Caldefie-Chézet F, Penault-Llorca F, Bignon YJ and Vasson MP:
Cellular expression of cyclooxygenase, aromatase, adipokines,
inflammation and cell proliferation markers in breast cancer
specimen. PLoS One. 10:e01384432015. View Article : Google Scholar : PubMed/NCBI
|