Introduction
Laryngeal squamous cell carcinoma (LSCC) is the
second most common type of head and neck squamous cell cancer
(HNSCC), representing ~2.4% of all cancer cases and 2.1% of all
cancer-associated mortalities worldwide in June 2009 (1). The latest Chinese cancer statistics
indicated that an ~26,400 novel cancer cases and 14,500 cancer
mortalities occurred in China in 2015 (2). Despite significant progress in surgery,
radiotherapy and chemotherapy over the last few decades, there has
been no improvement in the 5-year survival status of patients with
laryngeal cancer (LC), which has remained steady at 70–80%
(3). It was observed that the 3-year
disease-free survival (DFS) rate post-surgical intervention was
71.2%, while there was a significant decrease in survival rates for
patients with recurrent/metastatic (R/M) LC (4). One of the reasons for the poor survival
rates in glottis LSCC (GLSCC) may be due to inadequate tumor
profiling using conventional histopathology (4). Thus, it is necessary to elucidate the
molecular basis of LC and to detect prognostic biomarkers, which
may enable clinicians to improve the management of patients with
LSCC.
Epidermal growth factor receptor (EGFR) is a
transmembrane glycoprotein. This receptor binds with different
types of ligands, which induce receptor homo- and
heterodimerization, leading to intrinsic tyrosine kinase
activation, and resulting in cell proliferation, apoptosis,
differentiation and survival (5). A
total of <90% of patients with HNSCC express high levels of
EGFR, which is associated with poor survival (6). EGFR inhibitors, including cetuximab,
have been approved by the Food Drug Administration as monotherapy
for R/M HNSCC (7). Several studies
have explored the association between EGFR and prognosis. However,
the role of EGFR overexpression remains controversial in HNSCCs,
including LSCC (8–10).
Hepatocyte growth factor (HGF) receptor (c-Met)
encodes a transmembrane tyrosine kinase. It is known to stimulate
cell motility, dissociation of epithelial sheets, invasion of
cellular matrix and induction of angiogenesis (11). This receptor was found to be
overexpressed in the majority of types of solid tumors (12).
The incidences of overexpression of c-Met in lung,
colorectal cancer and renal cell carcinoma are 13.7, 50.0 and
44.8%, respectively, and are strongly associated with poor
prognosis (13–15). Overexpression of c-Met has been
observed in 50–80% of HNSCC cases (16–18), with
a mutation rate of 14% (19). Studies
have demonstrated that overexpression of c-Met is associated with
tumor progression, and is an important treatment target of HNSCC
(16,20,21).
However, the association between c-Met expression and survival
status in glottic LSCC (GLSCC) has been rarely studied (22).
Since there is heterogeneity in results when cancer
samples from different sites in the head and neck are used
(23–26), analysis was restricted to only GLSCC
samples. Association of c-Met and EGFR with clinical factors and
survival status has not yet been studied in GLSCC. To the best of
our knowledge, this is the first study that investigates the role
of EGFR and c-Met in GLSCC. The expression of EGFR and c-Met was
assessed in patients with GLSCC, and the expression of EGFR and
c-Met was compared with clinical parameters and DFS and overall
survival (OS) status. The present study found that high expression
of c-Met or EGFR were associated with poor survival and are
important predictors for prognosis of patients with GLSCC.
Materials and methods
Ethics
The experimental protocol was established, according
to the ethical guidelines of the Declaration of Helsinki and was
approved by the Human Ethics and Protocol Review Committee of the
Third Military Medical University (Chongqing, China). Written
informed consent was obtained from individual patients in the
study. All patients or guardians, subsequent to reading, filled in
and signed the consent form and agreed to be involved in the
present study.
Patients
The present study included 71 male patients with a
diagnosis of GLSCC, only male patients were included as the
incidence of GLSCC tends to be higher in males than females
(2). These patients were treated at
the Third Affiliated Hospital, Third Military Medical University,
Chongqing University (Chongqing, China) between December 2006 and
December 2011. The median age was 60 years old (range, 39–79
years). The Tumor staging and grading was determined according to
the American Joint Committee on Cancer tumor-node-metastasis
classification system of 2002 (27)
and histological grade was based on the World Health Organization
system (Table I). Primary treatment
for all 71 patients was surgery, which included 44 cases with
post-operation radiotherapy, 7 cases with post-operation concurrent
chemoradiotherapy and 20 patients without any post-surgery
treatment. There were 39 recurrent cases. The primary endpoint of
the study was DFS, and the secondary endpoint was OS.
 | Table I.Association between
clinicopathological factors and EGFR and c-Met expression. |
Table I.
Association between
clinicopathological factors and EGFR and c-Met expression.
|
|
| EGFR expression,
n | c-Met expression,
n |
|---|
|
|
|
|
|
|---|
| Factors | Number, n (%) | Low | High |
P-valuea | Low | High |
P-valuea |
|---|
| Age |
|
|
| 0.262 |
|
| 0.034a |
| ≤50
years | 11 (15.5) | 7 | 4 |
| 9 | 2 |
|
| >50
years | 60 (84.5) | 24 | 36 |
| 25 | 35 |
|
| Smoking |
|
|
| 0.841 |
|
| 0.634 |
| No | 13 (18.3) | 6 | 7 |
| 7 | 6 |
|
|
Yes | 58 (81.7) | 25 | 33 |
| 27 | 31 |
|
| Alcohol use |
|
|
| 0.144 |
|
| 0.327 |
| No | 46 (64.8) | 23 | 23 |
| 24 | 22 |
|
|
Yes | 25 (35.2) | 8 | 17 |
| 10 | 15 |
|
| Tstage |
|
|
| 0.200 |
|
| 0.060 |
| T1 | 12 (16.9) | 5 | 7 |
| 9 | 3 |
|
| T2 | 34 (47.9) | 11 | 23 |
| 12 | 22 |
|
| T3 | 18 (25.2) | 11 | 7 |
| 8 | 10 |
|
| T4 | 7 (10.0) | 4 | 3 |
| 5 | 2 |
|
| N stage |
|
|
| 0.368 |
|
| 1.000 |
| N0 | 65 (91.5) | 27 | 38 |
| 32 | 33 |
|
| N1 | 5 (7.0) | 3 | 2 |
| 2 | 3 |
|
| N2 | 1 (1.5) | 1 | 0 |
| 0 | 1 |
|
| Clinical stage |
|
|
| 0.194 |
|
| 0.082 |
| I | 10 (14.1) | 3 | 7 |
| 7 | 3 |
|
| II | 30 (42.2) | 10 | 20 |
| 10 | 20 |
|
|
III | 21 (29.6) | 12 | 9 |
| 10 | 11 |
|
| IV | 10 (14.1) | 6 | 4 |
| 7 | 3 |
|
| Histological
grade |
|
|
| 0.525 |
|
| 0.213 |
| G1 | 26 (36.7) | 9 | 17 |
| 16 | 10 |
|
| G2 | 41 (57.7) | 20 | 21 |
| 17 | 24 |
|
| G3 | 4 (5.6) | 2 | 2 |
| 1 | 3 |
|
| Recurrence |
|
|
| 0.001a |
|
|
<0.001a |
|
Yes | 39 (55.0) | 10 | 29 |
| 9 | 30 |
|
| No | 32 (45.0) | 21 | 11 |
| 25 | 7 |
|
Immunohistochemistry (IHC)
All tissue samples were fixed in 4%
formaldehyde solution at 4°C for 24 h, dehydrated with 70,
80 and 95% alcohol, each for 5 min, followed by 100% alcohol 3
times for 5 min, embedded in paraffin and cut into
3-µm-thick sections for IHC. Human c-Met polyclonal antibody
(catalog no. ZA-0636; Zhong Shan Golden Bridge Biological
Technology, Beijing, China) and human EGFR monoclonal antibody
(catalog no. ZA-0505; Zhong Shan Golden Bridge Biological
Technology) primary antibodies were pre-diluted by the supplier.
Immunostaining was achieved by SPlink Detection kit
(Biotin-Streptavidin HRP Detection System; catalog no. SP-9001;
Zhong Shan Golden Bridge Biological Technology) according to the
manufacturer's protocol. Sections were dewaxed in xylene 2 times (5
min each) and rehydrated using a descending alcohol series (100%
alcohol 2 times for 3 min each, followed by once with 95, 70 and
50% alcohol for 3 min each). Subsequently, endogenous peroxidase
activity was blocked by incubation with 0.3% hydrogen peroxide in
methanol at room temperature for 10 min to block endogenous
peroxidase activity. Sections were incubated with the
aforementioned primary antibody overnight at 4°C and later
sequentially incubated with biotin-labeled goat anti-rabbit IgG and
HRP-conjugated streptavidin, pre-diluted by the supplier
(Biotin-Streptavidin HRP Detection Systems; catalog no. SP-9001;
Zhong Shan Golden Bridge Biological Technology) at room temperature
for 1 h. The peroxidase reaction was visualized using
3,3′-Diaminobenzidine (DAB) substrate solution (0.05% DAB, 0.015%
H2O2, PBS) for 5 min at room temperature and
the sections were counterstained with hematoxylin. Negative
controls were assessed by replacing the primary antibody with
PBS.
The expression of EGFR and c-Met was evaluated using
immunostaining. The slides were examined by two independent
pathologists (Department of Pathology, Institute of Surgical
Research, Daping Hospital, Third Military Medical University,
Chongqing, China), who had no prior knowledge of the clinical and
pathological parameters. The intensity of staining was classified
into four grades: No staining, -; definite but weak staining, +;
moderate staining, ++; and strong staining, +++ (28). This method has been used and validated
previously (29). The proportion of
positive cells was counted in five microscopic fields at ×400
magnification (range, 0–100%). The percentage of cells with
different staining intensities was determined by visual assessment
with light microscopy. The H-score was calculated using the formula
1× (% of weak staining cells) + 2× (% of moderate staining cells) +
3× (% of strong staining cells; range, 0–300) (30).
Statistical analysis
Statistical analyses were performed using SPSS
software version 16.0 (SPSS, Inc., Chicago, IL, USA). The
χ2 test and Fisher's exact test were used to analyze the
association between expression of c-Met and EGFR, and
clinicopathological factors. Univariate and multivariate Cox
proportional hazards regression models were used to find prognostic
factors of DFS and OS. The likelihood ratio test was used to
determine if one covariate entered into the regression models is
significant. Survival analysis was performed by the Kaplan-Meier
method, and the log-rank test was used to compare the survival
curves. P<0.05 was considered to indicate a statistically
significant difference.
Results
Association between
clinicopathological parameters and expression of EGFR and c-Met
proteins in patients with GLSCC
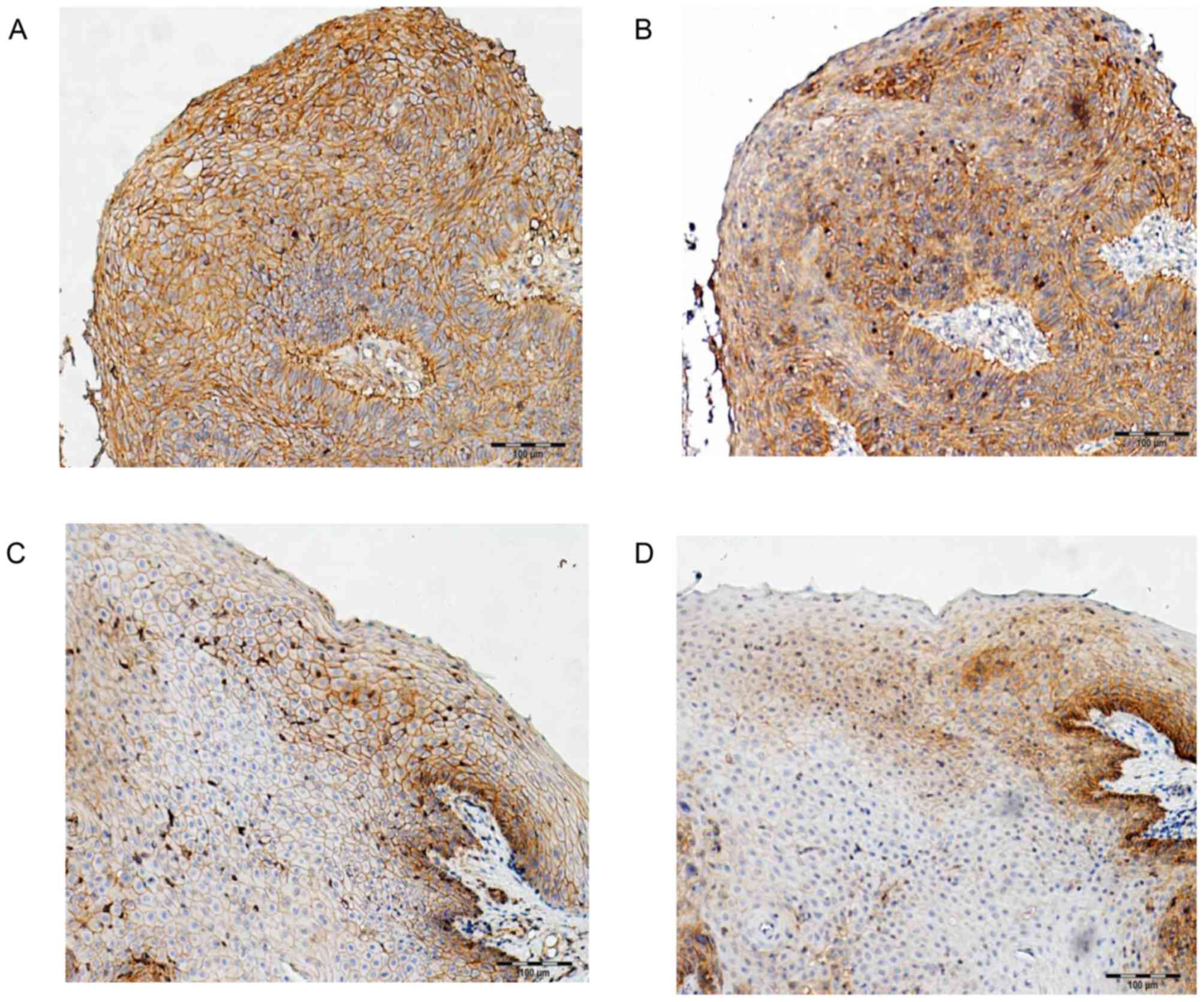
Positive expression (either complete membranous
staining or cytoplasmic staining near the cell membrane) of c-Met
and EGFR was observed in 69.0 (49/71) and 91.5% (65/71) cases,
respectively (Fig. 1). The median
values of H-score of c-Met and EGFR expression were 160 (range,
0–270) and 240 (range, 0–270), respectively. When the H-score in
individual subjects was greater than the median value, it was
considered to be indicative of a high expression of c-Met and
EGFR.
Table I presents the
association between c-Met and EGFR expression and
clinicopathological factors. High expression of EGFR or c-Met was
significantly associated with tumor recurrence in GLSCC (P=0.001
and P<0.001, respectively). Factors, including old age, advanced
T stages and tumor recurrence, were significantly associated with
high expression of c-Met (P=0.034, P=0.06 and P<0.001,
respectively). High expression of EGFR was strongly associated with
only tumor recurrence (P=0.001) and not with other factors. In
addition, 22 patients showed reduced expression of EGFR and c-Met,
while high expression of EGFR and c-Met was detected in 28 patients
(Table II). These findings suggested
that there was a significant positive association between the
expression of EGFR and that of c-Met (P=0.001).
 | Table II.Association between EGFR expression
and c-Met expression. |
Table II.
Association between EGFR expression
and c-Met expression.
|
| EGFR, n |
|
|---|
|
|
|
|
|---|
| Expression | Low | High | P-value |
|---|
| c-Met |
|
| 0.001a |
|
Low | 22 | 12 |
|
|
High | 9 | 28 |
|
Survival analysis
All patients were followed up until mortality or
until December 31, 2014. During the follow-up period, 20 patients
succumbed due to GLSCC-associated disease, 6 patients were lost to
follow-up. A total of 45 patients remain alive as of the last
follow-up. The median follow-up time was 43 months (range, 12–96).
The median DFS and OS times of all patients were 42.4 and 81.8
months, respectively. The DFS and OS for 1, 2 and 3-years were
75.9, 60.1 and 54.2%, and 95.22, 84.91 and 77.01%, respectively
(Fig. 2A and B). Kaplan-Meier
survival analysis for DFS revealed that patients with low levels of
EGFR expression had a longer DFS time compared with patients with
high expression, and the 2-year DFS rates were 69.5 and 51.7%,
respectively, in these two groups of patients (log-rank
χ2=9.708; P=0.002; Fig.
2C). Similarly, the DFS time was longer in patients with low
expression of c-Met compared with patients with high expression,
and the 2-year DFS rates were 73.5 and 47.3%, respectively
(log-rank χ2=19.526; P<0.001; Fig. 2D). In addition, the 2-year OS rate was
89.8% in patients with low levels of EGFR vs. 79.4% in patients
with high expression (log-rank χ2=7.066; P=0.008;
Fig. 2E), and the 2-year OS rate was
94.7% in patients with low level of c-Met vs. 73.1% in patients
with high expression (log-rank χ2=12.805, P<0.001;
Fig. 2F).
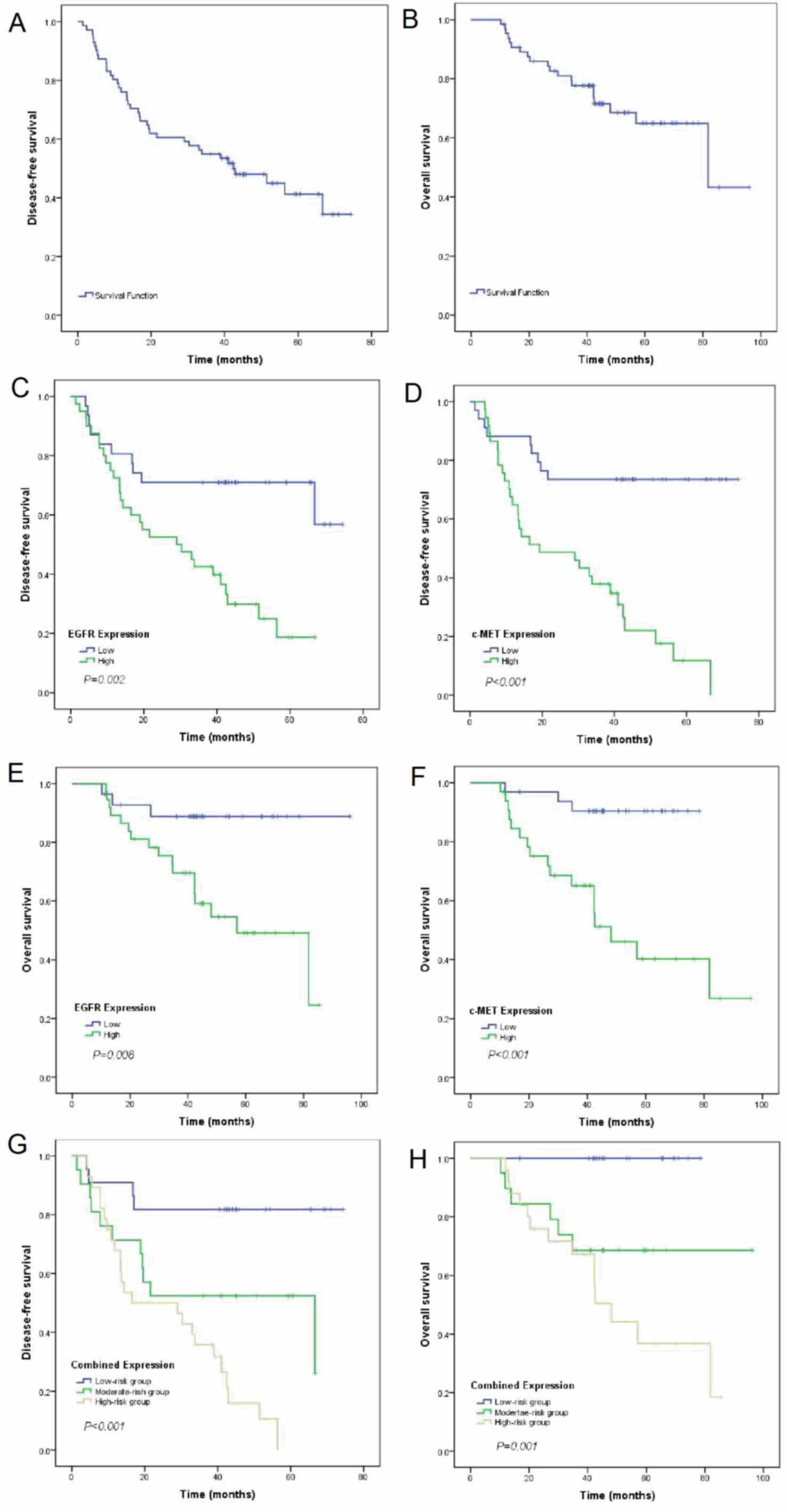 | Figure 2.KM curves demonstrated different
prognosis between subgroups. (A) Survival analysis of whole
population for DFS, (B) Survival analysis of whole population for
OS, (C) KM curves of DFS for EGFR low and high expression
subgroups, (D) KM curves of FDS for c-MET low and high expression
subgroups, (E) KM curves of OS for EGFR low and high expression
subgroups, (F) KM curves of OS for c-MET low and high expression
subgroups, (G) KM curves of DFS for low, moderate and high risk
subgroups, (H) KM curves of OS for low, moderate and high risk
subgroups. EGFR, epidermal growth factor receptor; DFS,
disease-free survival; OS, overall survival; KM, Kaplan-Meier. |
The patients were categorized into three subgroups:
A low-risk group, comprising of patients with low EGFR and c-Met
expression; a high-risk group, comprising patients with high EGFR
and c-Met expression; and a moderate-risk group, comprised of
patients with only one highly-expressed protein (EGFR or c-Met).
The 2-year DFS rates in low-, moderate- and high-risk groups were
81.8, 51.0 and 47.8%, respectively (log-rank, χ2=20.214;
P<0.001; Fig. 2G). The 2-year OS
rates in low-, moderate- and high-risk groups were 100.0, 80.4 and
73.5%, respectively (log-rank, χ2=13.570; P=0.001;
Fig. 2H).
Univariate and multivariable analysis
of prognostic factors for DFS and OS
Univariate Cox regression analysis showed a
significant association of expression of EGFR- and c-Met- with DFS
and OS (Tables III and IV). The risk of disease progression was
significantly increased in patients with high expression of c-Met
or EGFR when compared with those with low expression (HR=4.785,
P<0.001; HR=3.028, P=0.003, respectively). Additionally, the
risk of mortality was significantly increased in patients with high
expression of c-Met or EGFR when compared with those with low
expression (HR=6.976, P=0.002; HR=4.554, P=0.016, respectively).
Among all the other factors, only lymph node metastasis increased
the risk of disease recurrence (HR=3.246, P=0.016).
 | Table III.Univariate/multivariable analyses of
prognosis factors for disease-free survival. |
Table III.
Univariate/multivariable analyses of
prognosis factors for disease-free survival.
|
| Univariate | Multivariable |
|---|
|
|
|
|
|---|
| Factors | 95%CI | HR |
P-valuea | 95%CI | HR | P-value |
|---|
| Age (≤50 vs.
>50) | 0.911–9.664 | 2.967 | 0.071 | – | – | – |
| Smoking (no vs.
yes) | 0.343–1.528 | 0.724 | 0.396 | – | – | – |
| Drinking (no vs.
yes) | 0.633–2.091 | 1.151 | 0.645 | – | – | – |
| T stage (T1/2 vs.
T3/4) | 0.480–1.795 | 0.929 | 0.826 | – | – | – |
| N stage (N1/2 vs.
N0) | 1.251–8.426 | 3.246 | 0.016a | 0.993–19.773 | 4.432 | 0.051 |
| Clinical stage
(I/II vs. III/IV) | 0.572–2.041 | 1.080 | 0.812 | – | – | – |
| Histological grade
(G1 vs. G2/3) | 0.357–1.269 | 0.674 | 0.222 | 0.172–0.818 | 0.376 | 0.014 |
| EGFR
expression(high vs. low) | 1.458–6.286 | 3.028 | 0.003a | 0.631–3.650 | 1.518 | 0.352 |
| c-Met
expression(high vs. low) | 2.239–10.225 | 4.785 |
<0.001a | 1.695–9.678 | 4.050 | 0.002 |
 | Table IV.Univariate/multivariable analyses of
prognosis factors for overall survival. |
Table IV.
Univariate/multivariable analyses of
prognosis factors for overall survival.
|
| Univariate | Multivariable |
|---|
|
|
|
|
|---|
| Factors | 95%CI | HR |
P-valuea | 95%CI | HR | P-value |
|---|
| Age (≤50 vs. >50
years) | 0.417–7.838 | 1.809 | 0.428 | – | – | – |
| Smoking (no vs.
yes) | 0.159–1.029 | 0.405 | 0.058 | 0.163–1.654 | 0.520 | 0.268 |
| Drinking (no vs.
yes) | 0.351–2.188 | 0.876 | 0.777 | – | – | – |
| T stage (T1/2 vs.
T3/4) | 0.554–3.322 | 1.357 | 0.504 | – | – | – |
| N stage (N1/2 vs.
N0) | 0.602–11.572 | 2.640 | 0.198 | – | – | – |
| Clinical stage
(I/II vs. III/IV) | 0.784–4.756 | 1.895 | 0.156 | – | – | – |
| Histological grade
(G1 vs. G2/3) | 0.288–1.682 | 0.697 | 0.421 | – | – | – |
| EGFR expression
(high vs. low) | 1.333–15.562 | 4.554 | 0.016a | 0.538–11.909 | 2.532 | 0.240 |
| c-Met expression
(high vs. low) | 2.026–24.026 | 6.976 | 0.002 | 1.780–45.921 | 9.040 | 0.008 |
The multivariate Cox proportional hazards model
analysis revealed that c-Met expression is an independent
prognostic factor for DFS and OS (HR=4.050, P=0.002; HR=9.040,
P=0.008, respectively; Tables III
and IV). By contrast, EGFR was not
observed to be an independent prognostic factor for DFS and OS
(P=0.352 and P=0.240, respectively). Histological grade was an
independent prognostic factor for DFS and N stage had borderline
significance (HR=0.376, P=0.014; HR=4.432, P=0.051, respectively;
Table III).
Unlike results of c-Met, the assessment of the
combined expression of c-Met and EGFR did not further improve
prognostic capacity of c-Met for DFS and OS (likelihood ratio test,
P=0.119 and P=0.160, respectively; Table
V). These findings demonstrated that combined assessment of
EGFR and c-Met expression did not have any more prognostic value
superimposed effect in prognostic assessment.
 | Table V.Compared role of c-Met and combined
assessment with both c-Met as well as EGFR for prognosis. |
Table V.
Compared role of c-Met and combined
assessment with both c-Met as well as EGFR for prognosis.
|
| HR (95% CI) | −2 Log
likelihood | χ2 | P-value |
|---|
|
|
|
|
|
|
|---|
| Expression | DFS | OS | DFS | OS | DFS | OS | DFS | OS |
|---|
| c-Met | 4.785
(2.239–10.225) | 6.976
(2.026–24.026) | 277.754 | 135.494 | 2.426 | 1.978 | 0.119 | 0.160 |
| Combination |
|
|
|
|
|
|
|
|
|
c-Met | 3.816
(1.695–8.592) | 4.941
(1.330–18.360) | 275.328 | 133.516 |
|
|
|
|
|
EGFR | 1.832
(0.832–4.032) | 2.372
(0.644–8.740) |
|
|
|
|
|
|
Discussion
Tumor recurrence or metastasis following surgery is
one of the main factors affecting prognosis (31). The known biomarkers of HNSCC, EGFR,
cyclin D1, B-cell lymphoma 2, cyclin-dependent kinase inhibitor
p27, vascular endothelial growth factor and p53 (32), are limited in their ability to predict
prognosis, mainly due to the heterogeneity of cancers at different
head and neck sites (32,33). This is evident from the inconsistent
studies on the association of EGFR and c-Met with prognosis of
HNSCC (Table VI). The present study
indicated that high expression of c-Met and EGFR significantly
reduced DFS and OS. These findings indicated that abnormal
expression of c-Met or EGFR proteins serve as potential biological
markers for GLSCC, and may have an improved predictive value when
compared with clinicopathological factors.
 | Table VI.Studies on c-Met-and EGFR-expression
in HNSCC. |
Table VI.
Studies on c-Met-and EGFR-expression
in HNSCC.
| Gene | Author, year | Tumor sites | Method | Positive
expression, n | Association between
c-Met/EGFR expression and clinicopathological factors and
survival | (Refs. no.) |
|---|
| EGFR | Almadori et
al (2010) | Larynx | IHC | 23/67 | Metastases-free
survival (P=0.0001); OS (P=0.0002) | (8) |
|
| Kontic et al
(2015) | Larynx | IHC | 127/185 | Histopathological
grade (P<0.001); stage (P<0.001); metastasis (P<0.001);
relapse (P<0.001); survival (P<0.001). | (9) |
|
| Young et al
(2011) | Multiple | IHC | 81/93 | Failure-free
survival (P=0.35); OS (P=0.22) | (10) |
|
| Carballeira et
al (2014) | Lip | IHC | 50/55 | Tumor ulceration
(P=0.022); tumor thickness (P=0.002); tumor width (P=0.021). | (17) |
|
| Wei et al
(2008) | Larynx | IHC | 35/40 | There was a good
agreement between the primary tumors and the paired metastases
regarding EGFR expression (P<0.05) | (34) |
|
| Ma et al
(2014) | Multiple | IHC | 30/43 | HPV infection
(P=0.009); 3-year OS (P=0.037) | (36) |
|
| Cao et al
(2013) | Nasopharynx | IHC | 102/127 | Primary lesion
stage (P=0.001); clinical stage (P=0.002); relapse (P=0.015); DFS
(P=0.013); OS (P= 0.015). | (37) |
|
| Won et al
(2012) | Oropharynx/Oral
cavity | IHC | 78/121 | EGFR expression was
higher in oral cavity cancers compared with oropharyngeal cancers
(P=0.005) | (51) |
|
| Jiang et al
(2009) | Larynx | IHC | 31/75 | DFS (P=0.199); OS
(P=0.293). | (52) |
| c-Met | Choe et al
(2012) | Multiple | IHC | 34/82 | Lymph node
metastasis (P<0.05); primary location of the tumor
(P<0.05) | (16) |
|
| Baschnagel et
al (2014) | Multiple | IHC | 100/107 | Locoregional
control (P=0.031); distant metastasis (P=0.005); DFS (P<0.001);
OS (P<0.001) |
(18) |
|
| Kim et al
(2010) | Oral | IHC | 33/61 | Lymph node
metastasis (P=0.005), tumor classification (P=0.004); recurrence
(P=0.018); survival (P=0.003) | (20) |
|
| Zhang et al
(2014) | Larynx | IHC | 33/52 | TNM stage
(P<0.05); lymph node metastasis (P<0.05) | (42) |
|
| Luan et al
(2014) | Nasopharynx | IHC | 74/106 | TNM stage
(P<0.01); cervical lymph node metastasis (P<0.05) | (43) |
|
| Lim et al
(2012) | Oral tongue | IHC | 39/71 | Neck metastasis
(P<0.05); >4 mm depth of tumor invasion (P<0.05); survival
rates (P<0.05) | (44) |
|
| Zhao et al
(2011) | Oral | IHC | 44/86 | DFS (P=0.010); OS
(P=0.010) | (53) |
|
| Kim et al
(2006) | Hypopharynx | IHC | 28/40 | Lymph node
metastasis (P<0.05) | (54) |
EGFR is a member of the ErbB family, which promotes
cell proliferation, invasion, metastasis and survival (5). Similar to findings from a previous study
(34), the present study also
revealed that >90% of patients with GLSCC expressed EGFR
protein. However, EGFR was not demonstrated to be an independent
prognostic factor of DFS and OS. The present findings are in
contrast to previous studies, which have observed that EGFR
overexpression, increases risk of recurrence and mortality in
patients with LSCC (35–37). The reason for such discrepancies in
findings may be attributed to small sample size (38), difference in scoring methods and
studies conducted on HNSCC rather than only GLSCC samples.
Additionally, the present study was a retrospective one.
Furthermore, a meta-analysis revealed that EGFR is most appropriate
as an independent predictor of DFS in oropharyngeal carcinoma and
not in laryngeal cancer (33). This
indicated that EGFR may not be a suitable prognostic marker for all
types of head and neck cancers. However, considering the
aforementioned study limitations, it may be prudent to study this
in a larger sample size in a prospective study.
c-Met, with a molecular weight of 190 kDa, consists
of an extracellular α-chain and transmembrane β-chain with tyrosine
kinase activity (39). It has been
observed that alterations of the c-Met gene in the form of
amplification, deletion, mutation and overexpression are associated
with tumor cell proliferation, migration, invasion and angiogenesis
(40). Alterations of c-Met has been
revealed to be associated with poor prognosis of numerous tumors,
including breast, colorectal, liver, lung cancer and HNSCC
(20,41). However, such an association has not
been studied specifically in GLSCC. The present study demonstrated
an association between high expression of c-Met and recurrence and
mortality of patients with GLSCC. It was also observed that high
expression of c-Met is frequent in GLSCC, and was directly
associated with the relapse, age and T-stage, which are factors
linked with poor prognosis (42).
Previous studies have revealed associations between c-Met
expression and lymph node metastasis (16,43–45). Since
the present study consisted of very few patients (n=6) with nodal
involvement, such an association could not be detected. It was
observed that c-Met was an independent predictor of DFS and OS,
which suggested that IHC evaluation of c-Met in primary tumors may
contribute to identifying those patients with relapse and reduced
chances of survival. Therefore, in the future, increased c-Met
expression in patients with LSCC should be considered indicative of
the requirement for good treatment modalities with consistent
follow-up. In addition, univariate and multivariate analysis
indicated that high expression of c-Met protein was significantly
associated with a poorer prognosis when compared with that of EGFR
protein. Thus, c-Met may perform a crucial function in the
prognosis of LSCC; however, analysis in large numbers of LSCC
samples is required.
c-Met and EGFR are frequently co-expressed in
tumors, and act in synchrony to activate downstream signaling
pathways, including Ras-Raf-extracellular signal-regulated kinase,
signal transducer and activator of transcription 3 and
phosphoinositide 3-kinase/Akt-mechanistic target of rapamycin
cascades, to promote tumor progression (46). c-Met may be activated following EGFR
activation, in the absence of HGF (47). c-Met activation by HGF was also shown
to confer resistance to irreversible EGFR inhibitors (48,49). The
possible reason for this may be that the downstream signaling
pathways maybe activated by c-Met, which is independent of EGFRs,
leading to EGFR inhibitor-resistance (49). Benedettini et al (50) also demonstrated that lung cancer cells
with low response to EGFR inhibitors, including gefitinib and
erlotinib, exhibited high levels of c-Met. c-Met inhibitors may be
used to circumvent the problem of drug-resistance to EGFR therapies
(51). Thus, combined therapy with
c-Met as well as EGFR inhibitors may improve the control of tumor
cell proliferation (51). Based on
these findings, the association between c-Met and EGFR was
analyzed, and it was revealed that there was a significant positive
association between c-Met and EGFR expression. In addition,
subgroup analysis revealed that the DFS and OS times were extended
within the subgroups of low EGFR and c-Met expression compared with
at least one highly-expressed protein. However, combination of
c-MET and EGFR did not provide more prognostic information,
compared with c-MET alone for DFS and OS. This may be due to the
fact that EGFR was not an independent prognostic factor of DFS and
OS. Therefore, additional studies with a larger sample size are
required to investigate the combined role of EGFR and c-Met.
To conclude, c-Met and EGFR are important predictors
of survival in patients with GLSCC. Therefore, IHC analysis of
primary tumors with the biological marker c-Met may provide greater
potential to identify the prognosis. However, evaluation of c-Met
and EGFR expression status should be performed on a larger sample
population to obtain more reliable and consistent results. A more
accurate prediction of outcomes with specific therapies,
particularly molecular-targeted therapies, remains a worthy area of
investigation.
References
|
1
|
Rudolph E, Dyckhoff G, Becher H, Dietz A
and Ramroth H: Effects of tumour stage, comorbidity and therapy on
survival of laryngeal cancer patients: A systematic review and a
meta-analysis. Eur Arch Otorhinolaryngol. 268:165–179. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Carvalho AL, Nishimoto IN, Califano JA and
Kowalski LP: Trends in incidence and prognosis for head and neck
cancer in the United States: A site-specific analysis of the SEER
database. Int J Cancer. 114:806–816. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhang SY, Lu ZM, Luo XN, Chen LS, Ge PJ,
Song XH, Chen SH and Wu YL: Retrospective analysis of prognostic
factors in 205 patients with laryngeal squamous cell carcinoma who
underwent surgical treatment. PLoS One. 8:e601572013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Schlessinger J: Common and distinct
elements in cellular signaling via EGF and FGF receptors. Science.
306:1506–1507. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Herchenhorn D and Ferreira CG: Targeting
epidermal growth factor receptor to optimize chemoradiotherapy in
locally advanced head and neck cancer: Has biology been taken into
account. J Clin Oncol. 29:e283–e287. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Specenier P and Vermorken JB: Cetuximab in
the treatment of squamous cell carcinoma of the head and neck.
Expert Rev Anticancer Ther. 11:511–524. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Almadori G, Bussu F, Gessi M, Ferrandina
G, Scambia G, Lauriola L, Paludetti G and Ranelletti FO: Prognostic
significance and clinical relevance of the expression of the HER
family of type I receptor tyrosine kinases in human laryngeal
squamous cell carcinoma. Eur J Cancer. 46:1144–1152. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kontić M, Milovanović J, Čolović Z, Poljak
NK, Šundov Ž, Sučić A and Pešutić-Pisac V: Epidermal growth factor
receptor (EGFR) expression in patients with laryngeal squamous cell
carcinoma. Eur Arch Otorhinolaryngol. 272:401–405. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Young RJ, Rischin D, Fisher R, McArthur
GA, Fox SB, Peters LJ, Corry J, Lim A, Waldeck K and Solomon B:
Relationship between epidermal growth factor receptor status,
p16(INK4A), and outcome in head and neck squamous cell carcinoma.
Cancer Epidemiol Biomarkers Prev. 20:1230–1237. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Maroun CR and Rowlands T: The Met receptor
tyrosine kinase: A key player in oncogenesis and drug resistance.
Pharmacol Ther. 142:316–338. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Graveel CR, Tolbert D and Woude GF Vande:
MET: A critical player in tumorigenesis and therapeutic target.
Cold Spring Harb Perspect Biol. 5:pii: a0092092013. View Article : Google Scholar
|
|
13
|
Park S, Choi YL, Sung CO, An J, Seo J, Ahn
MJ, Ahn JS, Park K, Shin YK, Erkin OC, et al: High MET copy number
and MET overexpression: Poor outcome in non-small cell lung cancer
patients. Histol Histopathol. 27:197–207. 2012.PubMed/NCBI
|
|
14
|
Lee HE, Kim MA, Lee HS, Jung EJ, Yang HK,
Lee BL, Bang YJ and Kim WH: MET in gastric carcinomas: Comparison
between protein expression and gene copy number and impact on
clinical outcome. Br J Cancer. 107:325–333. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Albiges L, Guegan J, Le Formal A, Verkarre
V, Rioux-Leclercq N, Sibony M, Bernhard JC, Camparo P, Merabet Z,
Molinie V, et al: MET is a potential target across all papillary
renal cell carcinomas: Result from a large molecular study of pRCC
with CGH array and matching gene expression array. Clin Cancer Res.
20:3411–3421. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Choe JY, Yun JY, Nam SJ and Kim JE:
Expression of c-Met Is different along the location and associated
with lymph node metastasis of head and neck carcinoma. Korean J
Pathol. 46:515–522. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Carballeira A, Ginarte M, Diniz-Freitas M,
Fernández-Campos I, Gude F, Fraga M, Antúnez JR and
García-Caballero T: Immunohistochemical evaluation of EGFR
expression in lip squamous cell carcinoma. Correlation with
clinicopathological characteristics. Histol Histopathol.
29:641–648. 2014.PubMed/NCBI
|
|
18
|
Baschnagel AM, Williams L, Hanna A, Chen
PY, Krauss DJ, Pruetz BL, Akervall J and Wilson GD: c-Met
expression is a marker of poor prognosis in patients with locally
advanced head and neck squamous cell carcinoma treated with
chemoradiation. Int J Radiat Oncol Biol Phys. 88:701–707. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ghadjar P, Blank-Liss W, Simcock M, Hegyi
I, Beer KT, Moch H, Aebersold DM and Zimmer Y: MET
Y1253D-activating point mutation and development of distant
metastasis in advanced head and neck cancers. Clin Exp Metastasis.
26:809–815. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kim CH, Koh YW, Han JH, Kim JW, Lee JS,
Baek SJ, Hwang HS and Choi EC: c-Met expression as an indicator of
survival outcome in patients with oral tongue carcinoma. Head Neck.
32:1655–1664. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Seiwert TY, Jagadeeswaran R, Faoro L,
Janamanchi V, Nallasura V, El Dinali M, Yala S, Kanteti R, Cohen
EE, Lingen MW, et al: The MET receptor tyrosine kinase is a
potential novel therapeutic target for head and neck squamous cell
carcinoma. Cancer Res. 69:3021–3031. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Luo J, Zha S, Gage WR, Dunn TA, Hicks JL,
Bennett CJ, Ewing CM, Platz EA, Ferdinandusse S, Wanders RJ, et al:
α-methylacyl-CoA racemase: A new molecular marker for prostate
cancer. Cancer Res. 62:2220–2226. 2002.PubMed/NCBI
|
|
23
|
Sawatsubashi M, Sasatomi E, Mizokami H,
Tokunaga O and Shin T: Expression of c-Met in laryngeal carcinoma.
Virchows Arch. 432:331–335. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bosch FX, Ritter D, Enders C,
Flechtenmacher C, Abel U, Dietz A, Hergenhahn M and Weidauer H:
Head and neck tumor sites differ in prevalence, and spectrum of p53
alterations but these have limited prognostic value. Int J Cancer.
111:530–538. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Götte K: Intratumoral genomic
heterogeneity in head and neck cancer. Laryngorhinootologie.
83:122–123. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Betts GN, Eustace A, Patiar S, Valentine
HR, Irlam J, Ramachandran A, Merve A, Homer JJ, Möller-Levet C,
Buffa FM, et al: Prospective technical validation and assessment of
intra-tumour heterogeneity of a low density array hypoxia gene
profile in head and neck squamous cell carcinoma. Eur J Cancer.
49:156–65. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Moertel S, Ackermann H, Baghi M, Eckardt
A, Wagenblast J, Stöver T and Hambek M: Heterogeneity of primary
site biopsies in head and neck squamous cell carcinoma. Anticancer
Res. 31:665–669. 2011.PubMed/NCBI
|
|
28
|
Greene FL, Page DL, Fleming ID, et al:
AJCC Cancer Staging Manual. 6th. New York, NY: Springer; 2002,
View Article : Google Scholar
|
|
29
|
Carlsson J, Nordgren H, Sjöström J, Wester
K, Villman K, Bengtsson NO, Ostenstad B, Lundqvist H and Blomqvist
C: HER2 expression in breast cancer primary tumours and
corresponding metastases. Original data and literature review. Br J
Cancer. 90:2344–2348. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kim CH, Moon SK, Bae JH, Lee JH, Han JH,
Kim K and Choi EC: Expression of hepatocyte growth factor and c Met
in hypopharyngeal squamous cell carcinoma. Acta Otolaryngol.
126:88–94. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Patel M, Lu L, Zander DS, Sreerama L, Coco
D and Moreb JS: ALDH1A1 and ALDH3A1 expression in lung cancers:
Correlation with histologic type and potential precursors. Lung
Cancer. 59:340–349. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mazières J, Brugger W, Cappuzzo F, Middel
P, Frosch A, Bara I, Klingelschmitt G and Klughammer B: Evaluation
of EGFR protein expression by immunohistochemistry using H-score
and the magnification rule: Re-analysis of the SATURN study. Lung
Cancer. 82:231–237. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Schaaij-Visser TB, Brakenhoff RH, Leemans
CR, Heck AJ and Slijper M: Protein biomarker discovery for head and
neck cancer. J Proteomics. 73:1790–1803. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kim KY, Mcshane LM and Conley BA:
Designing biomarker studies for head and neck cancer. Head Neck.
36:1069–1075. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Keren S, Shoude Z, Lu Z and Beibei Y: Role
of EGFR as a prognostic factor for survival in head and neck
cancer: A meta-analysis. Tumor Biol. 35:2285–2295. 2014. View Article : Google Scholar
|
|
36
|
Wei Q, Sheng L, Shui Y, Hu Q, Nordgren H
and Carlsson J: EGFR, HER2, and HER3 expression in laryngeal
primary tumors and corresponding metastases. Ann Surg Oncol.
15:1193–1201. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Farhadieh RD, Salardini A, Rees CG,
Russell PJ, Yang JL and Smee R: Protein expression of epidermal
growth factor receptor in laryngeal squamous cell carcinoma index
tumors correlates with diagnosis of second primary tumors of the
upper aero-digestive tract. Ann Surg Oncol. 16:2888–2894. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ma L, Wang D, Wufuer A, Wu R, Zhang S and
Wang R: Relationship between human papilloma virus infection and
expression of p16 and EGFR in head and neck squamous cell carcinoma
and their prognostic significance. Zhonghua Zhong Liu Za Zhi.
36:23–28. 2014.(In Chinese). PubMed/NCBI
|
|
39
|
Cao XJ, Hao JF, Yang XH, Xie P, Liu LP,
Yao CP and Xu J: Prognostic value of expression of EGFR and nm23
for locoregionally advanced nasopharyngeal carcinoma. Med Oncol.
29:263–271. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Marioni G, Blandamura S, Loreggian L,
Koussis H, Lionello M, Giacomelli L, Fasanaro E, Lovato A and
Staffieri A: Laryngeal carcinoma prognosis after postoperative
radiotherapy correlates with CD105 expression, but not with
angiogenin or EGFR expression. Eur Arch Otorhinolaryngol.
268:1779–1787. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Bhardwaj V, Cascone T, Cortez MA, Amini A,
Evans J, Komaki RU, Heymach JV and Welsh JW: Modulation of c-Met
signaling and cellular sensitivity to radiation: Potential
implications for therapy. Cancer. 119:1768–1775. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Han CB, Ma JT, Li F, Zhao JZ, Jing W, Zhou
Y and Zou HW: EGFR and KRAS mutations and altered c-Met gene copy
numbers in primary non-small cell lung cancer and associated stage
N2 lymph node-metastasis. Cancer Lett. 314:63–72. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Goetsch L, Caussanel V and Corvaia N:
Biological significance and targeting of c-Met tyrosine kinase
receptor in cancer. Front Biosci (Landmark Ed). 18:454–473. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zhang Y, Zhao J and Zhang Q: The
expression and clinical significance of OPN and C-met in laryngeal
carcinoma. Lin Chung Er Bi Yan Hou Tou Jing Wai Ke Za Zhi.
28:256–258. 2014.(In Chinese). PubMed/NCBI
|
|
45
|
Luan T and Yu Y: Increased hepatocyte
growth factor and c-Met receptor expression in nasopharyngeal
carcinoma. Int J Clin Exp Med. 7:5583–5587. 2014.PubMed/NCBI
|
|
46
|
Lim YC, Han JH, Kang HJ, Kim YS, Lee BH,
Choi EC and Kim CH: Overexpression of c-Met promotes invasion and
metastasis of small oral tongue carcinoma. Oral Oncol.
48:1114–1119. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Zhao D, Wang SH, Feng Y, Hua CG, Zhao J
and Tang XF: Intratumoral c-Met expression is associated with
vascular endothelial growth factor C expression, lymphangiogenesis,
and lymph node metastasis in oral squamous cell carcinoma:
Implications for use as a prognostic marker. Hum Pathol.
42:1514–1523. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Ishibe S, Karihaloo A, Ma H, Zhang J,
Marlier A, Mitobe M, Togawa A, Schmitt R, Czyczk J, Kashgarian M,
et al: Met and the epidermal growth factor receptor act
cooperatively to regulate final nephron number and maintain
collecting duct morphology. Development. 136:337–345. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Saunders VC, Lafitte M, Adrados I, Quereda
V, Feurstein D, Ling Y, Fallahi M, Rosenberg LH and Duckett DR:
Identification of an EGFRvIII-JNK2-HGF/c-Met-signaling axis
required for intercellular crosstalk and glioblastoma multiforme
cell invasion. Mol Pharmacol. 88:962–969. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Puri N and Salgia R: Synergism of EGFR and
c-Met pathways, cross-talk and inhibition, in non-small cell lung
cancer. J Carcinog. 7:92008. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yamada T, Matsumoto K, Wang W, Li Q,
Nishioka Y, Sekido Y, Sone S and Yano S: Hepatocyte growth factor
reduces susceptibility to an irreversible epidermal growth factor
receptor inhibitor in EGFR-T790M mutant lung cancer. Clin Cancer
Res. 16:174–183. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Benedettini E, Sholl LM, Peyton M, Reilly
J, Ware C, Davis L, Vena N, Bailey D, Yeap BY, Fiorentino M, et al:
Met activation in non-small cell lung cancer is associated with de
novo resistance to EGFR inhibitors and the development of brain
metastasis. Am J Pathol. 177:415–423. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Lau PC and Chan AT: Novel therapeutic
target for head and neck squamous cell carcinoma: HGF-MET signaling
pathway. Anticancer Drugs. 22:665–673. 2011. View Article : Google Scholar : PubMed/NCBI
|