Introduction
The prognosis of patients with glioblastoma (GB)
remains poor despite the use of multidisciplinary treatments, which
consist of maximal surgical resection, radiotherapy and
concomitant/adjuvant chemotherapy with temozolomide (1). The median time-to-recurrence following
standard therapy is ~7 months (2).
Once tumor recurrence has occurred (generally within 1 year),
further clinical deterioration may occur, along with expansive
tumor growth, as identified by contrast-enhanced imaging (3) and the dissemination of the tumor into
the cerebrospinal fluid (4). Patients
with GB typically succumb within 2 years of diagnosis, despite
multiple types of therapy available to treat GB (5). Therefore, in order to combat this
devastating disease, novel targeted therapies to treat patients
with GB are strongly required. In addition, since GB appeared in
the brain, therapies are required to minimize adverse effects on
the neuronal cells to keep host brain function.
Sodium butyrate (SB), a short chain (C-4) saturated
fatty acid, is present in the human bowel at increased
concentrations (~2 mM) as a food metabolite (6). It has been reported that SB exerts an
anti-tumor effect as a histone deacetylase (HDAC) inhibitor in
colon (6,7), pancreas (8) and liver cancer (9) as well as in glioma (10); however, its precise mechanism of
action remains to be fully elucidated (10–12). The
present study focused on the underlying mechanism of SB regarding
its effects on human GB cell proliferation, motility and
invasion.
In the present study, SB induced dose-dependent
growth inhibitory effects in vitro with G1/S phase arrest
and stabilization of p21 expression. SB also increased
senescence-associated (SA) β-galactosidase (gal) levels and
exhibited a significant inhibitory effect on tumor cell invasion
in vitro. All these biological effects of SB were reversible
and were attenuated following SB withdrawal.
Materials and methods
Materials and reagents
SB (cat no. 193-01152; reagent grade), trichostatin
A (TSA; cat. no. 203-17561), methotrexate (cat. no. 139-13571) and
N-acetyl-L-cysteine (NAC; cat. no. 015-05132; ROS scavenger) were
purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan).
Anti-focal adhesion kinase (FAK) polyclonal antibody (cat no.
sc-1688) and antibodies against p16 (cat no. sc166760), p21 (cat
no. sc-166630), p27 (cat no. sc-1641), p53 (cat no. sc-47698),
SIRT1 (cat no. sc-74504) MLC20 (cat no. sc-9449) and β-actin (cat
no. sc-47778) were purchased from Santa Cruz Biotechnology, Inc.
(Dallas, TX, USA). Anti-phospho (p)-tyrosine pY397-FAK (cat no.
44-624G) and pY577-FAK (cat no. 44-614G) polyclonal antibodies were
purchased from Thermo Fisher Scientific, Inc. (Waltham, MA, USA).
Anti-p-myosin light chain 20 (p-MLC20) antibody was provided by Dr
Fumio Matsumura (Rutgers University, New Brunswick, NJ, USA). All
other chemicals used were commercial products from Wako Pure
Chemical Industries, Ltd. SB (200 mM stock) and TSA (1 mM stock)
were dissolved in phosphate-buffered saline (PBS) and diluted in
the cell media prior to use.
Cell culture
The human GB cell line A172 was obtained from the
American Type Culture Collection (Manassas, VA, USA). Cells were
maintained in Dulbecco's modified Eagle's medium (DMEM)
supplemented with 10% fetal bovine serum (FBS; Thermo Fisher
Scientific, Inc.) at 37°C in a humidified incubator with 5%
CO2.
Cell proliferation assay and cell
cycle analysis
Cell proliferation was analyzed following a
previously described protocol (13).
Briefly, A172 cells were seeded at a density of 5×103
cells/well onto 6-well plates and cultured in DMEM with 10% FBS at
37°C for 24 h. Cells were then treated with SB (0, 0.5, 1, 2 and 4
mM) for 7 days. Following 4 days treatment, a subset of A172 cells
treated with 2 mM SB were washed with PBS and cultured for a
further 4 days in DMEM with 10% FBS without SB. For all cells, the
culture medium was replenished every 2 d and cell numbers were
recorded every 24 h using a WST-8 assay (cat no. 341-07761; Wako
Pure Chemical Industries, Ltd.). Cell cycle analysis was performed
using a BD FACSCalibur flow cytometer (BD Biosciences, Franklin
Lakes, NJ, USA), according to the manufacturer's protocol. Briefly,
A172 cells were cultured with indicated concentrations of SB, 10,
100 nM TSA or 0.1, 1 mM methotrexate for 4 d, then collected and
resuspended in 300 µl of PBS. After, 700 µl of 100% ethanol was
added and kept on ice for 30 min to fix the cells. The fixed cells
were resuspended in PBS supplemented with 100 µg/ml of RNase A and
50 µg/ml of propidium iodide (PI) at room temperature for 1 h. The
PI stained cells were analyzed according to standard FACS protocol
for cell cycle analysis with BD FACStation (v6.1, BD Biosciences,
Franklin Lakes, NJ, USA).
Western blot analysis
Cell lysates were prepared with Laemmli's sample
buffer (1 µl/104 cells) and 10 µg/lane [determined using
bicinchoninic acid protein assay kit (cat no. T9300A; Takara
Biotechnology Co., Ltd., Dalian, China) according to the
manufacturers protocol] was loaded onto 7.5–12.5% SDS
polyacrylamide gel, and western blot analysis was performed as
previously described (13) on A172
cells treated with 2 mM SB, prior to and following SB washout.
Proteins were electrically transferred to nitrocellulose membranes,
then the membranes were incubated with the following primary
antibodies: FAK, p16, p21, p27, p53, SIRT1, MLC20 and β-actin (all
at a dilution of 1:1,000), and pY397FAK, pY-577FAK and p-MLC20 (all
1:100) at 4°C overnight. The membranes were then incubated with
were alkaline phosphatase (AP)-conjugated anti-rabbit IgG
(dilution, 1:7,500; cat. no. S3731; Promega Corporation, Madison,
WI, USA) or AP-conjugated anti-mouse IgG (dilution, 1:7,500; cat.
no. S3721; Promega Corporation) secondary antibodies for 30 min at
room temperature. Final detection was performed using Western Blue
stabilized substrate for alkaline phosphatase (cat. no. S3841,
Promega Corporation). Band intensity was analyzed using ImageJ
1.38e software (National Institutes of Health, Bethesda, MD,
USA).
RNA isolation and reverse
transcription polymerase chain reaction (RT-PCR)
Total RNA was extracted and purified using TRIzol
reagent (Invitrogen; Thermo Fisher Scientific, Inc.), according to
the manufacturer's protocol. A total of 1 µg RNA was subsequently
used as a template for reverse transcription using a High-Capacity
cDNA Reverse Transcription kit (Applied Biosystems; Thermo Fisher
Scientific, Inc.) following the manufacturer's protocol. PCR was
performed using Taq DNA Polymerase (Promega Corporation) with the
following gene-specific primers: p21, forward,
5′-TTATGAAATTCACCCCCTTT-3′; and reverse,
5′-GCCCCTTCAAAGTGCCATCT-3′; p27, forward,
5′-AGATGTCAAACGTGCGAGTG-3′; and reverse,
5′-TCAGTCTTTGGGTCCACCAA-3′; p53, forward,
5′-GGGCCCACTTCACCGTACTA-3′; and reverse,
5′-TCTACCTAACCAGCTGCCCA-3′; and GAPDH, forward,
5′-AAAGGGAAGCTGACAGGGAT-3′ and reverse, 5′-GTGGGTATCACCGAGGAAGT-3′.
GAPDH functioned as the reference gene. Thermocycling conditions
were 95°C for 30 sec for denaturation, 60–65°C for 30 sec for
annealing and 68°C for 1 min for extension and cycle numbers were
30–35 cycles.
Immunofluorescence microscopy
Immunofluorescence analysis was performed following
a previously described protocol (13). Briefly, A172 cells were cultured with
0–4 mM SB on type I collagen-coated 2-well chamber slides (BD
Biosciences). After 48 h, the cells were fixed with 1%
paraformaldehyde at room temperature for 1 h in PBS and then
permeabilized with 0.2% TritonX-100 in PBS. Non-specific blocking
was performed in 0.1% bovine serum albumin (Wako Pure Chemical
Industries, Ltd.) at room temperature for 1 h. Cells were incubated
with primary antibody, Anti-p-tyrosine pY397-FAK (dilution, 1:100)
at 4°C overnight, followed by incubation with Alexa 488 anti-rabbit
immunoglobulin G (dilution, 1:1,000; cat no. A-11008; Molecular
Probes; Thermo Fisher Scientific, Inc.) secondary antibody, at room
temperature for 1 h in dark room. Images were obtained using the
Olympus BX50 fluorescence microscope (magnification, ×40; numerical
aperture, 0.6; Olympus Corporation, Tokyo, Japan). Images were
analyzed and processed for presentation by adjusting brightness and
contrast using ImageJ 1.38 software (National Institutes of Health,
Bethesda, MD, USA).
Effects of SB on cell motility and
invasion
A172 cells were pretreated with 0, 0.25, 0.5, 1, 2
and 4 mM of SB for 48 h. The cell motility assay was performed for
16 h using 24-well Bio-Coat cell migration chambers (BD
Biosciences) using 0.5% FBS as a chemoattractant, following a
previously described protocol (13).
The cell invasion assay was performed using Matrigel-coated
polyethylene terephthalate membranes for 16 h. Cells at a density
of 2×105 cells/well were inserted into the upper
chamber. The migrating cells on the lower side of the filter were
fixed with 70% ethanol at room temperature for 1 h, prior to being
stained with Giemsa solution (Wako Pure Chemical Industries, Ltd.)
at room temperature for 10 min and counted using Olympus IX70 phase
contrast microscopy with a ×10 objective lens. For cell area
analysis, cell images were captured using Olympus IX70 phase
contrast microscopy with a ×10 objective lens. Cell periphery was
circled then the inner area was calculated with ImageJ 1.38e
software. A minimum of 100 cells/each experiment were measured.
β-gal staining assay
The β-gal staining assay was performed using a
Senescence β-Galactosidase Staining kit (cat. no. 9860; Cell
Signaling Technology, Inc., Danvers, MA, USA). Briefly, A172 cells
were cultured with 0, 0.25, 0.5, 1, 2 or 4 mM SB, or 25, 50 or 100
nM TSA at 37°C for 4 days in 35 mm plastic culture plates, and
fixed with 10% formaldehyde at room temperature for 1 h. Plates
were rinsed twice with PBS, subsequently the β-gal staining
solution containing X-gal as a substrate was added and plates were
incubated at 37°C overnight in a dry incubator. β-gal-positive
cells were then counted using Olympus IX70 phase contrast
microscopy with ×10 objective lens and the percentage of positive
cells was calculated.
Proteasome activity assay
The proteasome activity assay was performed using a
Proteasome Activity assay kit (cat. no. ab107921; Abcam, Tokyo,
Japan) following the manufacturer's protocol. A172 cells were
treated with 2 mM SB for 48 h. The chymotrypsin-like function of
A172 cell lysate for a 7-Amino-4-methyl-coumarin-tagged peptide
substrate (proteasome substrate Succ-LLVY-AMC) was compared with
that of the positive control Jurkat cell lysate in the presence of
the proteasome inhibitor MG132 (2 µM).
Transient RNA interference
Small interfering (si)RNA against human p21 (CDKN1A,
SignalSilence p21 Waf1/Cip1 siRNA) was obtained from Cell Signaling
Technology, Inc. A siRNA-control was designed for Photinus Pyralis
GL3 luciferase as previously described and obtained from Hokkaido
System Science Co., Ltd. (Sapporo, Japan) (14). This gene is not present in mammalian
cell transcripts and therefore the luciferase siRNA-control
minimizes the off-targeting effect (14). A172 cells were transfected with
siRNA-p21 or siRNA-control at a final concentration of 50 nmol/l
using Lipofectamine 2000 (Invitrogen; Thermo Fisher Scientific,
Inc.). Cell lysates were prepared for western blotting 72 h after
transfection, performed as aforementioned. Transfected cells
underwent staining for β-gal. Cell motility and invasion assays, as
well as cell cycle analyses were also performed.
Statistical analysis
Data are expressed as the mean ± standard deviation.
Differences were determined by analysis of variance with
Bonferroni's test as a post-hoc test in JMP software (version 11;
SAS Institute, Inc., Cary, NC, USA). P<0.01 was considered to
indicate a statistically significant difference.
Results
Effect of SB on the proliferation of
A172 human glioblastoma cells
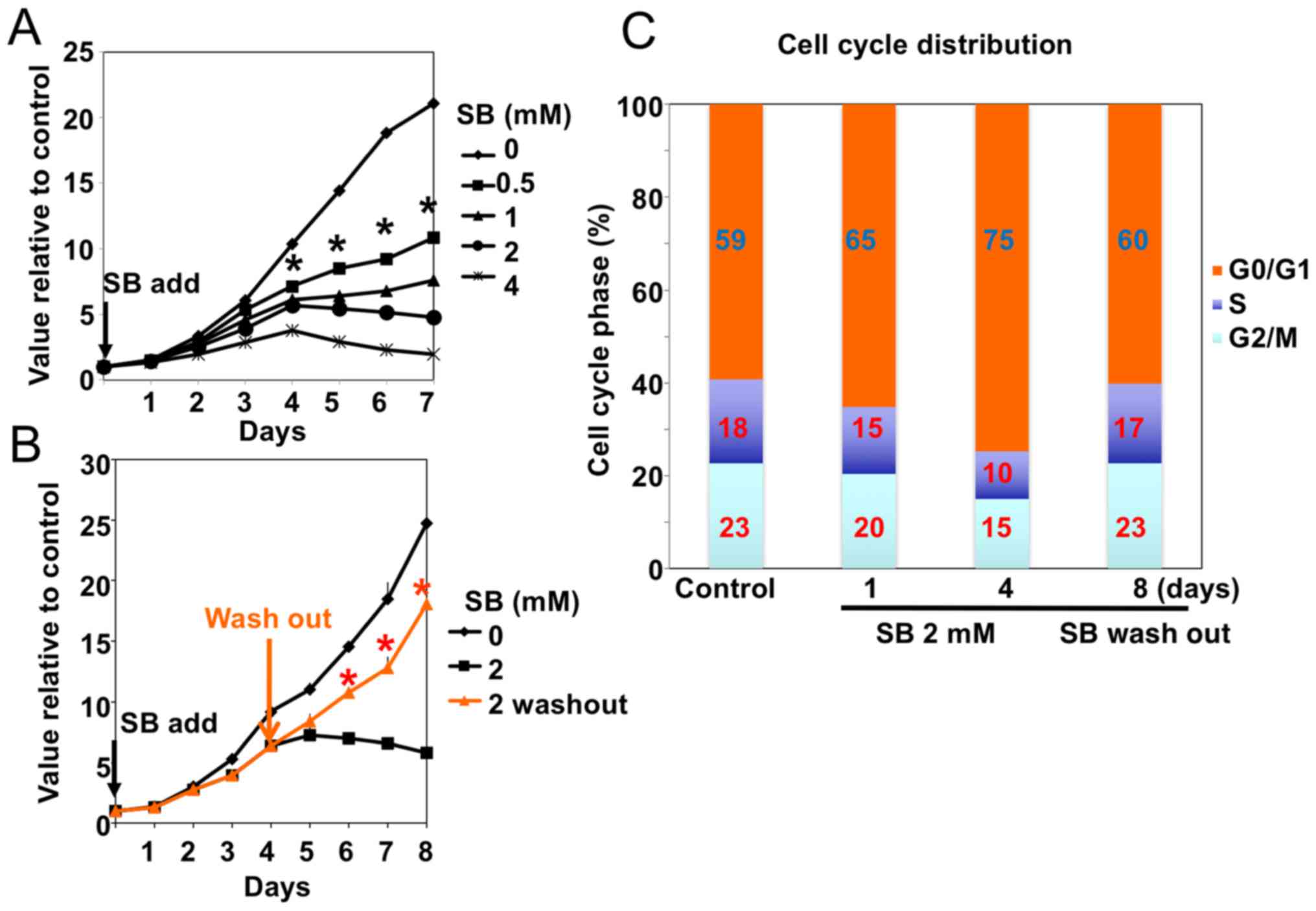
Physiological concentrations of SB (0.5–4 mM)
inhibited the proliferation of the human GB A172 cells in a
dose-dependent manner (Fig. 1A). In
addition, cell cycle analysis identified an increase in the
percentage of cells in the G0/G1-phase following SB treatment,
suggesting G1/S arrest. This inhibitory effect of SB on A172 cell
proliferation and cell cycle progression was reversible and
attenuated following wash-out of the drug [Fig. 1B (orange line) and Fig. 1C]. No sub-G1 apoptotic population was
observed following FACS analysis of the A172 cells treated with SB.
By contrast, FACS analysis identified apoptotic A172 cells
following the treatment of cells with the HDAC inhibitor TSA or the
chemotherapy drug methotrexate (data not shown).
Effect of SB and the HDAC inhibitor
TSA on SA β-gal staining
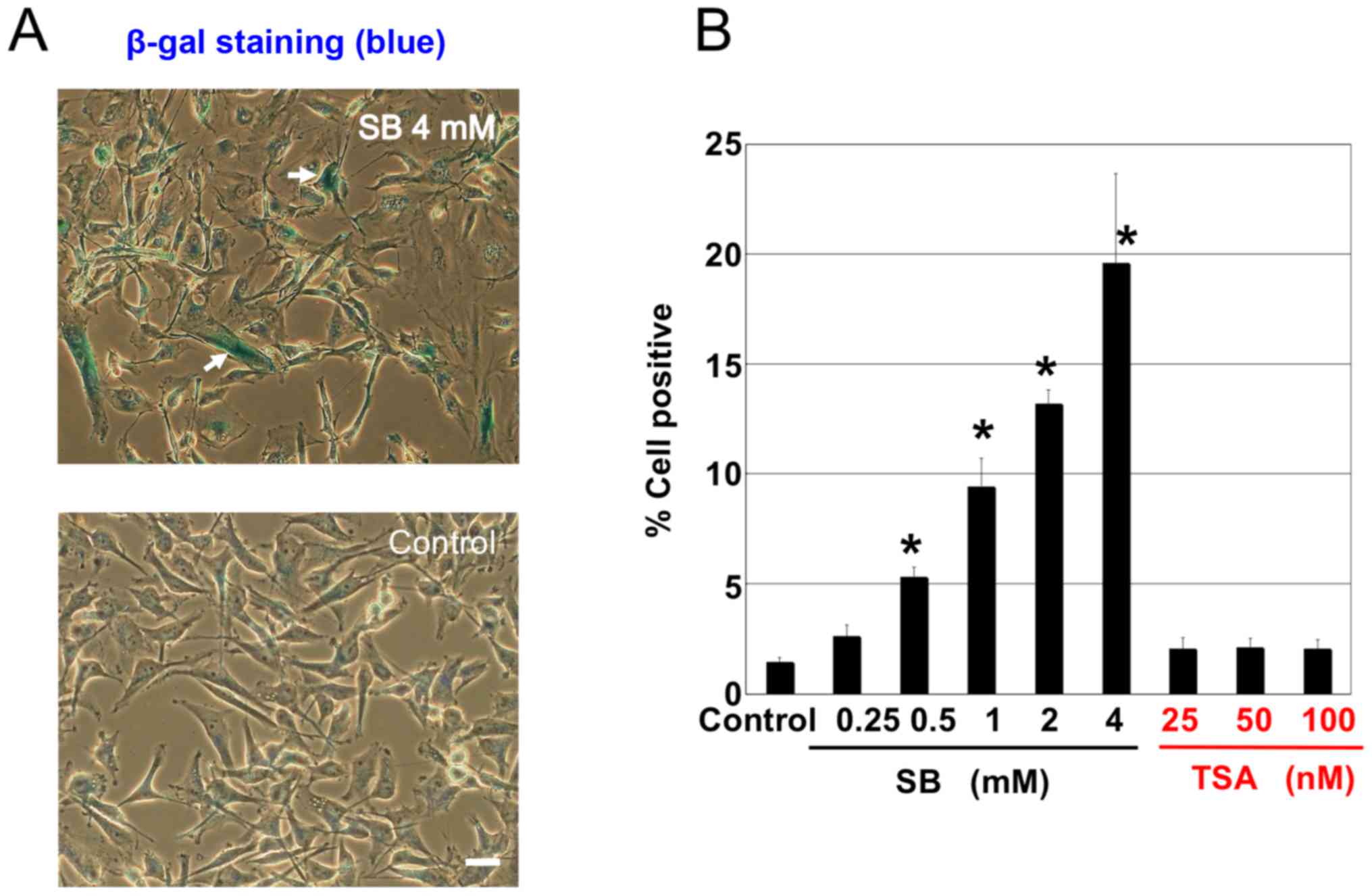
SB also induced positive staining for SA β-gal in
A172 cells (Fig. 2A). This positive
staining for β-gal, indicating cellular senescence, was SB
dose-dependent (Fig. 2B). However,
the HDAC inhibitor TSA did not induce any positive staining for
β-gal (Fig. 2B). To elucidate the
mechanism associated with this cell cycle arrest, the expression of
cell cycle regulator proteins was assessed.
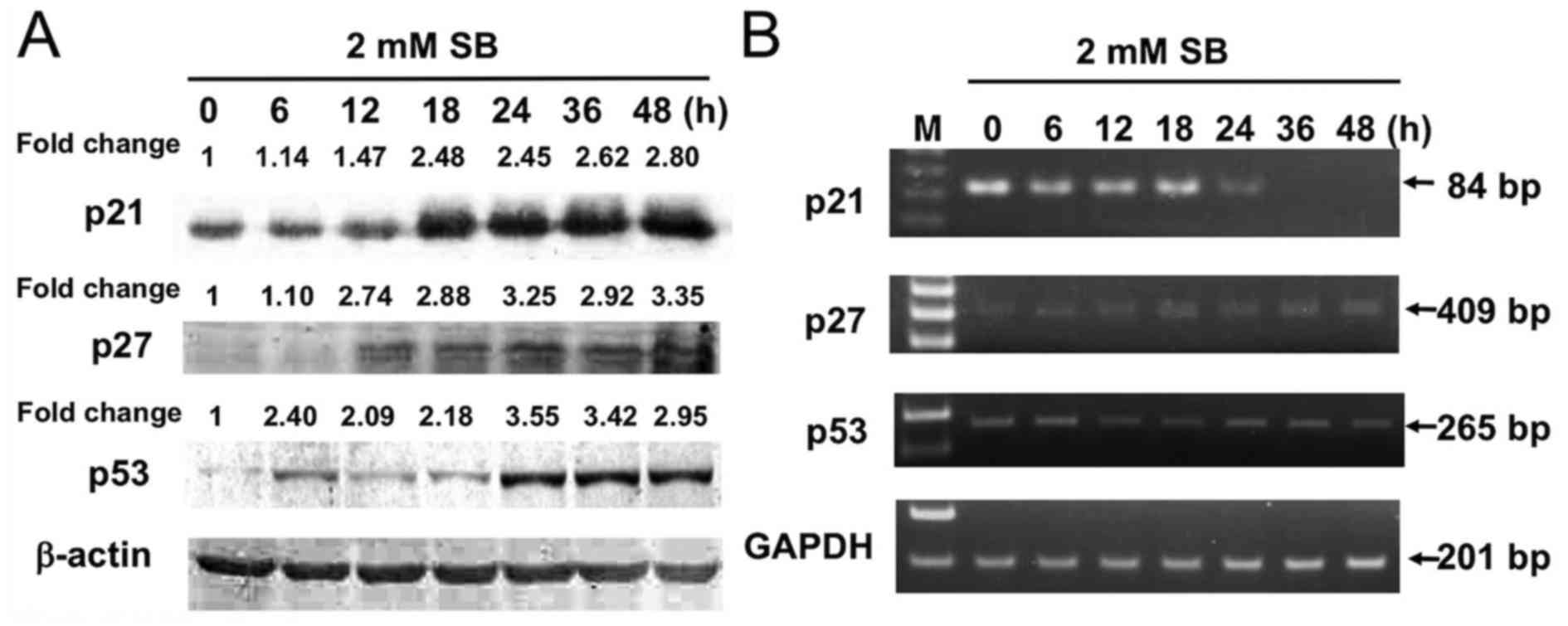
SB increased the p21 protein
level
A172 cells treated with 2 mM SB exhibited elevated
levels of p21, p27 and p53 and this increase in expression was
time-dependent (Fig. 3A); however,
levels of p21 mRNA were decreased 24 h after treatment with SB
(Fig. 3B). Since A172 cells harbor
wild-type p53 (15), it was deduced
that the p53-p21 axis functioned in the cells. The results of the
present study suggest that 24 h treatment with SB stabilizes the
expression of the three cell cycle regulator proteins p21, p27 and
p53 in A172 cells. Although levels of p21 mRNA decreased, the
levels of p27 and p53 mRNA were unaltered. Therefore, it is likely
that the post-translational protein stabilization of p21, p27 and
p53 induced by SB treatment is the primary mechanism responsible
for the results of the present study. The present study therefore
assessed the potential inhibitory activity of SB against the
proteasome compared with that of MG132, a specific proteasome
inhibitor. SB did not exhibit any direct inhibitory effect on the
proteasome in this proteasome activity assay (data not shown).
Effect of SB on the motility, invasion
and morphology of A172 cells
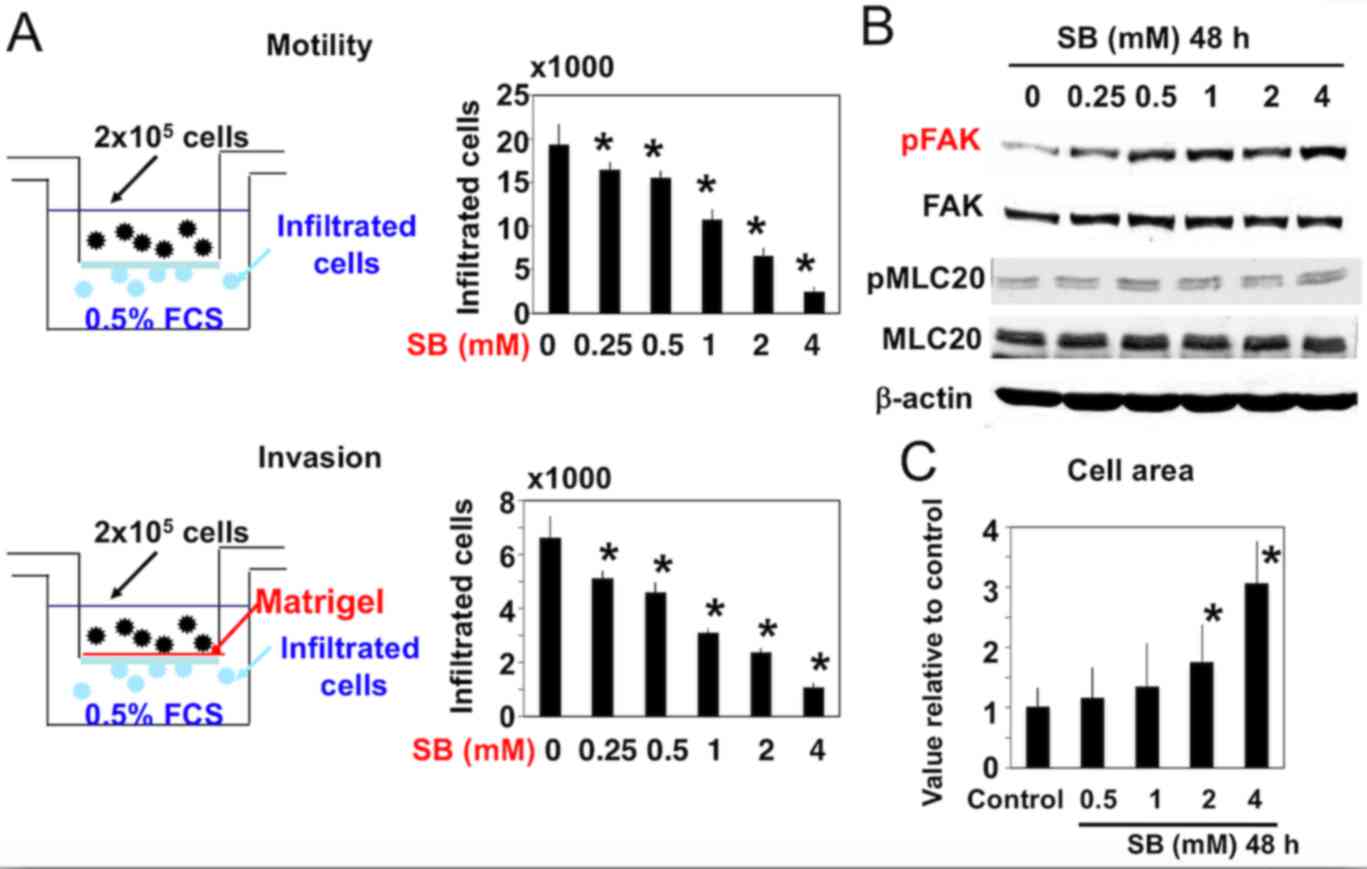
SB also inhibited cellular motility and invasion in
a dose-dependent manner and affected the morphology of A172 cells
(Fig. 4). Increased cell area and
adherence to the extracellular matrix was observed following
treatment with SB (Fig. 4C). Since 48
h SB treatment did not affect cellular proliferation or apoptosis
(Fig. 1), it could be concluded that
the decreased number of infiltrated cells was due to a decrease in
cell motility following SB treatment and not due to a decrease in
total cell number. In addition, the phosphorylation of FAK (Y397
site) was increased, while that of MLC20 (S19 site) remained
unaltered (Fig. 4B).
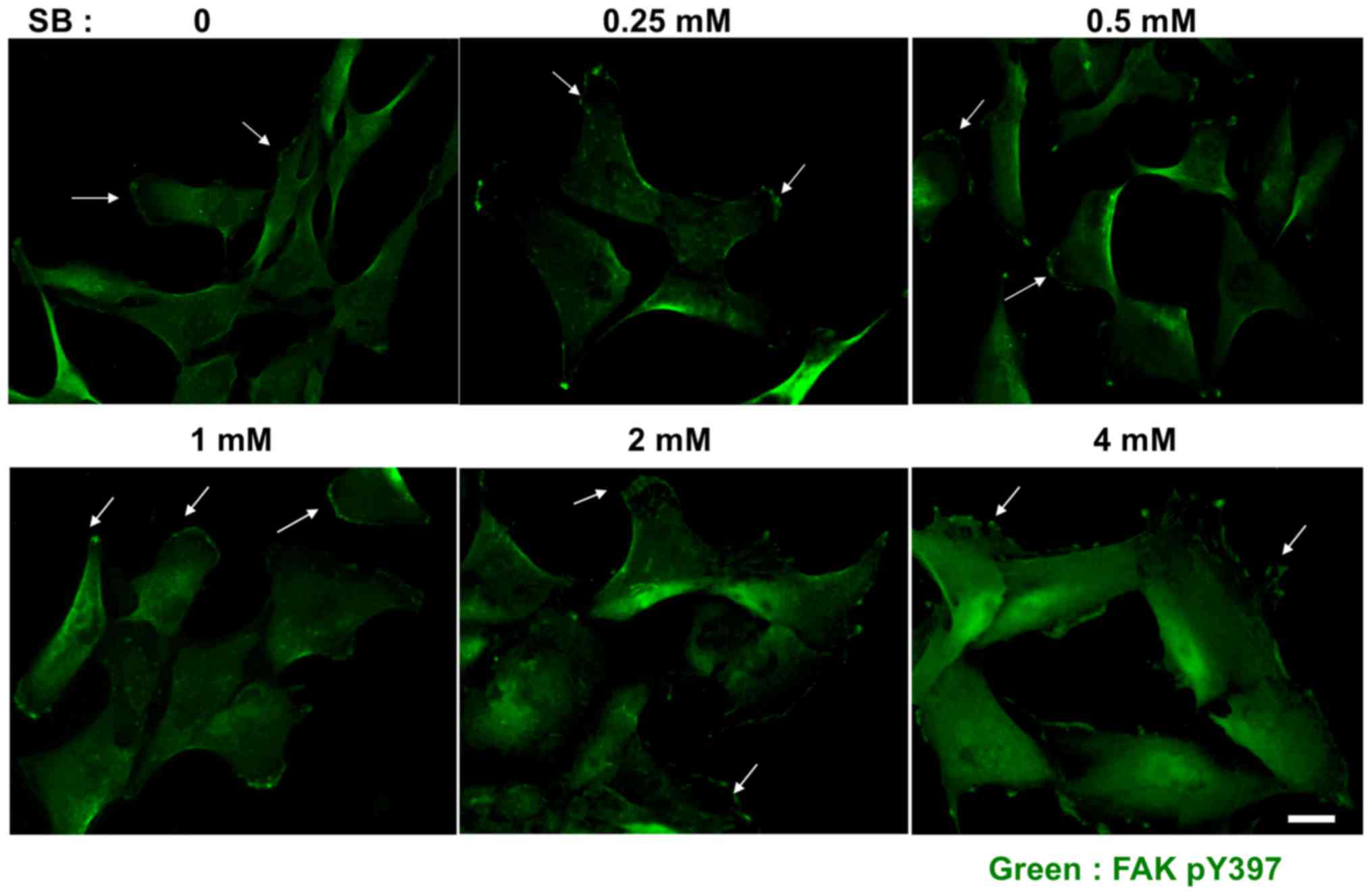
The results of the immunofluorescence analysis
indicated that SB dose-dependently induced an increase in the
amount of focal adhesions at the cell peripheries (white arrows,
stained with anti-pY397-FAK antibodies; Fig. 5) and increased cell spreading area on
the substratum. The effects of SB on the expression and
phosphorylation of FAK were reversible and were attenuated
following the washing out of SB from the cell culture (Fig. 6A). Cellular motility also recovered
3–4 days following SB wash out (Fig.
6B).
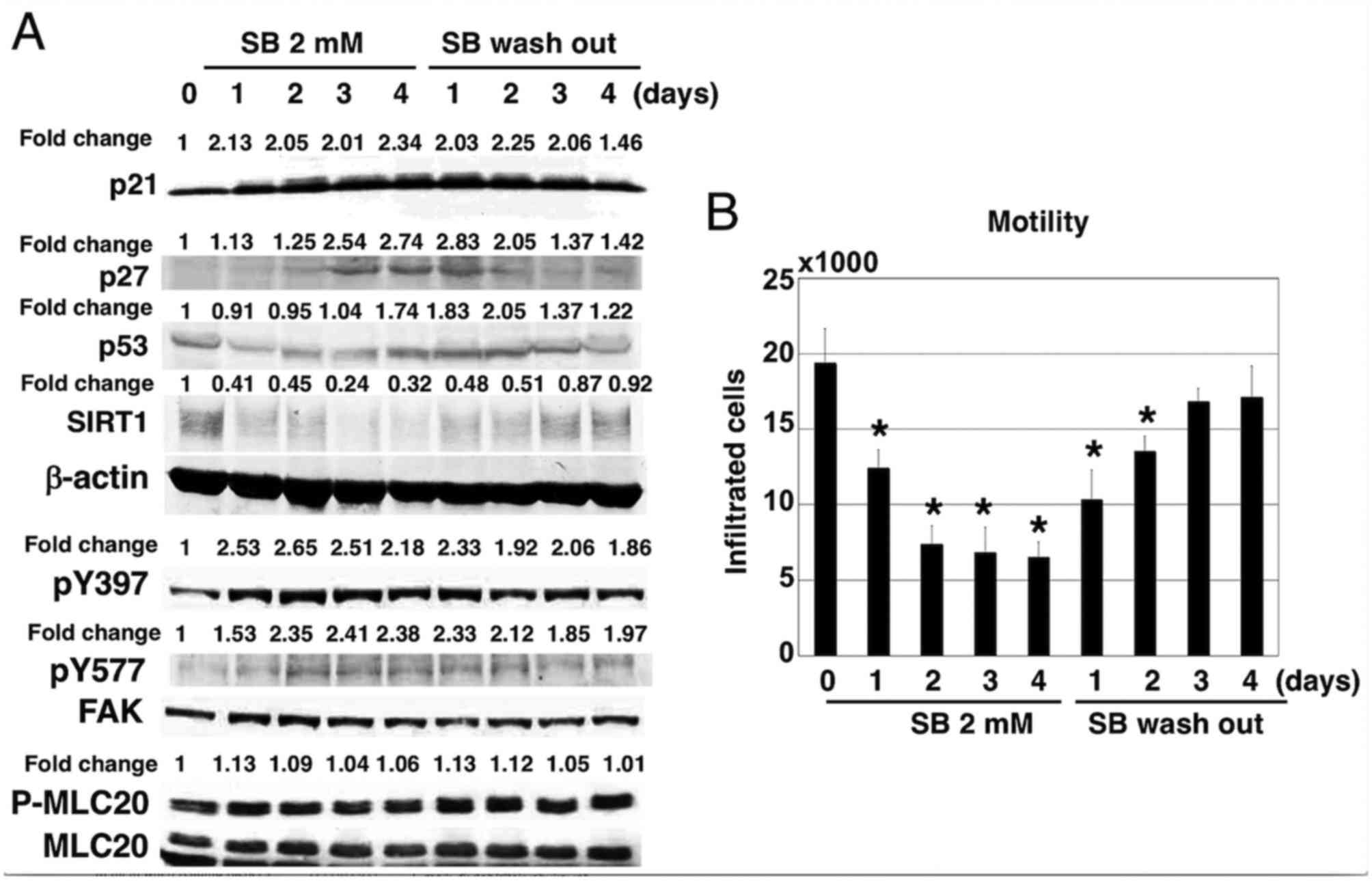 | Figure 6.Reversibility of the effects of SB in
A172 cells. (A) At 4 days after treatment with 2 mM SB, A172 cells
were washed in media without SB and cultured for 4 additional days.
The effects on the expression of p21 (FK506 binding protein like),
p27 (cyclin dependent kinase inhibitor 1B), p53, SIRT1, pY397/pY577
FAK and p-MLC20 were determined by western blotting. (B) Cell
motility was measured in A172 cells treated as in (A). Results are
presented as the mean ± standard deviation (n=6). *P<0.01 vs.
control. SB, sodium butyrate; SIRT, sirtuin; FAK, focal adhesion
kinase; p-, phosphorylated; MLC, myosin light chain. |
p21 involved SB induced A172 cellular
senescence but not involved cellular motility
The present study focused on the underlying
mechanism of SB regarding its effects on cellular senescence and
motility. Knockdown of p21 expression was achieved by transfection
with siRNA specific for the human p21 gene CDKN1A. The inhibition
of p21 expression was confirmed by western blotting and assessed
following the treatment of cells with 2 mM SB (Fig. 7A). Knockdown of p21 expression did not
affect the expression levels of p53, p27 and p16 (Fig. 7A). In contrast, knockdown of p21
expression partially reversed the inhibition of proliferation and
positive SA β-gal staining, but cell motility remained inhibited
(Fig. 7B). Knockdown of p21
expression increased S phase in the control and SB treated cells
using cell cycle analysis (Fig. 7C).
However, the increase in FAK phosphorylation induced by 2 mM SB was
unaffected following knockdown of p21 (Fig. 7A). These results suggest that
upregulation of p21 is implicated in the cellular senescence, but
not the inhibition of cell motility, that is induced by SB.
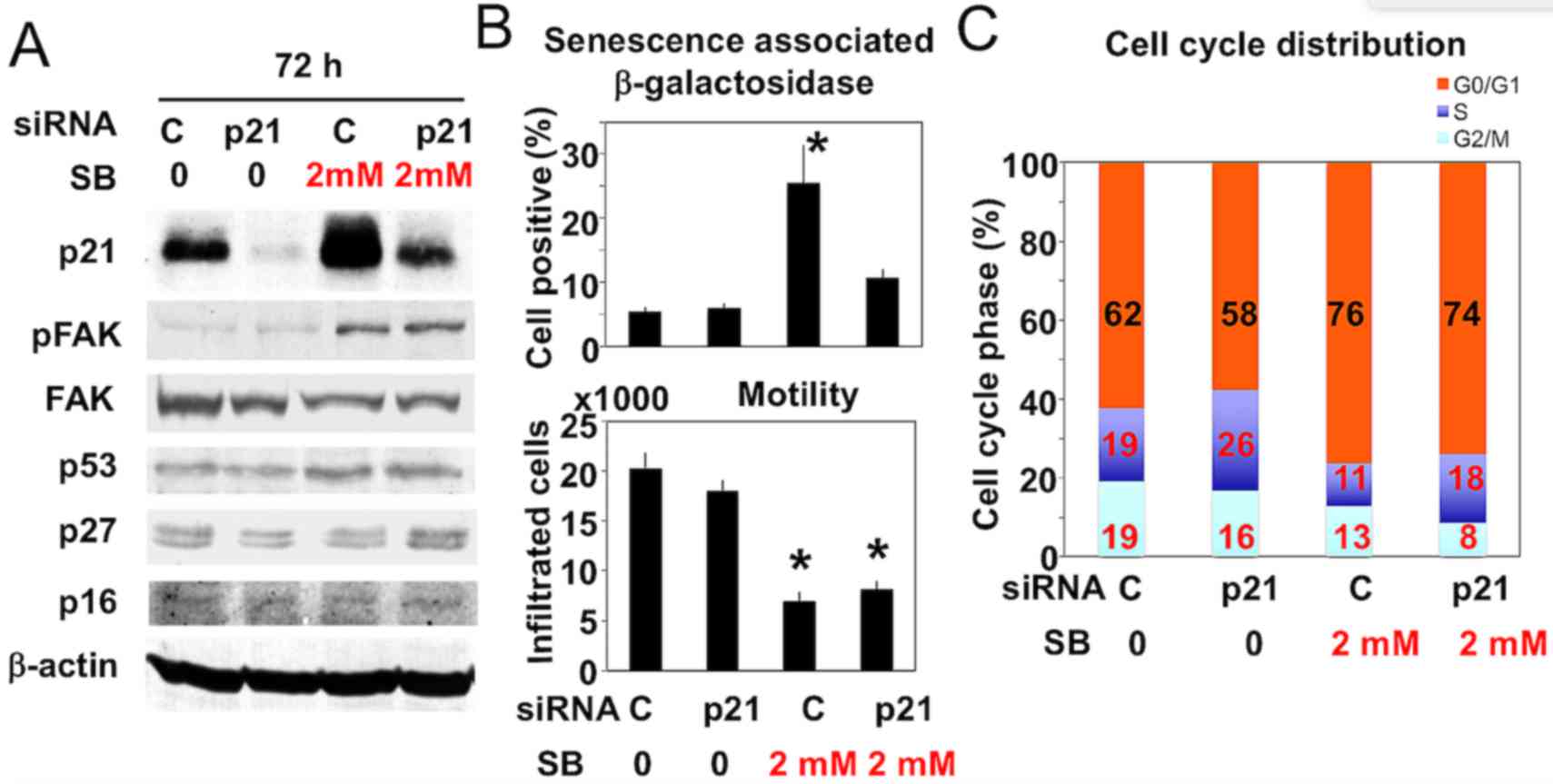 | Figure 7.Effect of SB on the levels of p21
(FK506 binding protein like), p53, p27 (cyclin dependent kinase
inhibitor 1B), p16 and pFAK in p21 knockdown and control A172
cells. (A) Knockdown of p21 was achieved by transfection with
specific siRNA against human p21 for 72 h. The effect of 2 mM SB on
the levels of p21 (FK506 binding protein like), p53, p27 (cyclin
dependent kinase inhibitor 1B), p16 and FAK phosphorylation in
control and p21-knockdown cells was determined using western blot
analysis. (B) β-galactosidase positive staining (n=4; upper panel);
and cell motility (lower panel; n=6) were measured in the A172
cells in (A). Results are presented as the mean ± standard
deviation. *P<0.01 vs. control. (C) Cell cycle analysis of the
A172 cells in (A) using a BD FACScalibur system. SB, sodium
butyrate; p-, phosphorylated; FAK, focal adhesion kinase; si, small
interfering; C, control. |
Discussion
The results of the present study indicated that SB
reversibly inhibited A172 cell proliferation and induced cellular
senescence by upregulating and stabilizing the cell cycle regulator
proteins p21, p27 and p53. SB also reversibly inhibited cellular
motility and invasion. Downregulating p21 using specific siRNA
partially reversed the proliferation-inhibiting effect of SB on
cell cycle analysis and β-gal staining. By contrast, cell motility,
cell spreading and the phosphorylation of FAK remained decreased
following p21 knockdown in A172 cells.
SB exhibits HDAC-inhibiting activity. However, TSA
(a typical HDAC inhibitor) did not promote cellular senescence or
reversibly inhibit cellular proliferation in A172 cells, and did
not induce cell apoptosis. Previous studies by our group used
similar assays to evaluated the effect of SB in various other tumor
cell lines, including human fibrosarcoma (HT-1080), human breast
cancer (MDA-MB-231; unpublished) and rat mammary cancer (Walker
256; manuscript in preparation). In all cases, SB reversibly
inhibited cell proliferation, increased cell adhesion, and
inhibited cell motility and invasion. Furthermore, the effect of SB
on normal primary cultured cells was assessed and it was identified
that SB did not affect cell proliferation much. Therefore, SB may
potentially be used as an anti-cancer therapy, although continuous
treatment may be required. Using an osmotic pump to maintain the
drug concentration, as reported previously (13), the intrathecal administration of SB
into rat cerebrospinal space was performed in a previous study by
our group, which inhibited the invasion of Walker 256 cells (a
syngeneic rat mammary cancer cell line) into the brain parenchyma
and increased the survival rate of rats (manuscript in
preparation).
The mechanism corresponding to the inhibition of
motility induced by SB is currently under investigation. It has
been reported that cancer motility and invasion is primarily
regulated by the Rho family of small GTP-binding proteins
(including RhoA and cell division cycle 42) and Rho-associated
kinase (ROCK), and that the downstream molecular motor activity of
myosin, determined by the phosphorylation of MLC20, is crucial
(16). The specific ROCK inhibitor
Y-27632 inhibits the motility, invasion and adhesion of hepatoma
(16,17) and breast cancer cells (13,18). Our
group has also demonstrated that the invasive phenotype of murine
osteosarcoma cells is positively associated with increased FAK
levels (19). In the present study,
the effects of SB were unusual, particularly regarding the
increases in FAK phosphorylation, cell spreading and cell area
associated with decreased cell motility. Therefore, novel molecular
targets of SB within tumor cells should be evaluated. SB induced
multiple metabolic changes in A172 cells; therefore, metabolic
pathways associated with isocitrate dehydrogenase 1 and branched
chain aminotransferase, which have been implicated as critical
targets in oncogenesis and progression of malignant glioma
(20), may be associated with the
effect of SB on glioma cell motility. Further studies by our group
aim to analyze the metabolome alterations induced by SB.
Previous studies have focused on the effect of
butyrate on microRNAs (miRs). Pant et al (21) reported that miR-22 and SB inhibited
sirtuin 1 (SIRT-1) expression, enhanced reactive oxygen species
(ROS) release in hepatic cancer cells and induced cellular
apoptosis. Similarly, the present study demonstrated that treatment
with SB reversibly inhibited the expression of SIRT-1 in A172
cells; however, an ROS scavenger (N-acetyl-L-cysteine (NAC)) did
not affect SB-induced A172 cell senescence (data not shown). Han
et al (22) reported that SB
induced miR-203 expression and that the miR-203 target gene neural
precursor cell expressed developmentally downregulated 9 was
associated with the SB-induced inhibition of colony formation and
invasion in colorectal cancer cells. Further studies into the
targeting activity of SB besides its HDAC-inhibiting function are
required.
It was previously reported that irradiation induced
GB cells to senescence (23) and that
anti-vascular endothelial growth factor A-targeting therapy induced
senescence-associated cell death (24). p21 protein (FK506 binding protein
like)-dependent cytotoxicity was suggested in these cases (23,24).
Therefore, the present study hypothesized that SB-induces
senescence via a similar pathway.
In conclusion, SB inhibited A172 cell proliferation,
induced cellular senescence and inhibited cell invasion,
potentially via a mechanism associated with upregulation of p21. It
may therefore be developed as a novel treatment for GB.
Acknowledgements
The authors acknowledged that the preliminary work
of the present study was reported at the American Association for
Cancer Research (AACR) annual meeting on April 9, 2013, and
appeared in an AACR article as the meeting abstract (25). The authors would like to thank Dr
Kiyoko Yoshioka (Osaka Medical Center for Cancer and Cardiovascular
Diseases, Osaka, Japan) for her technical assistance. The present
study was supported by the Japan Society for the Promotion of
Science (grant no. 15K15560 to KI).
References
|
1
|
Stupp R, Mason WP, van den Bent MJ, Weller
M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn
U, et al: Radiotherapy plus concomitant and adjuvant temozolomide
for glioblastoma. N Engl J Med. 352:987–996. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Stupp R, Hegi ME, Mason WP, van den Bent
MJ, Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A, Fisher B,
Belanger K, et al: Effects of radiotherapy with concomitant and
adjuvant temozolomide versus radiotherapy alone on survival in
glioblastoma in a randomised phase III study: 5-year analysis of
the EORTC-NCIC trial. Lancet Oncol. 10:459–466. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gzell C, Back M, Wheeler H, Bailey D and
Foote M: Radiotherapy in Glioblastoma: The Past, the present and
the future. Clin Oncol (R Coll Radiol). 29:15–25. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nakagawa H, Yamada M, Kanayama T,
Tsuruzono K, Miyawaki Y, Tokiyoshi K, Hagiwara Y and Hayakawa T:
Myelin basic protein in the cerebrospinal fluid of patients with
brain tumors. Neurosurgery. 34:825–833. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Blumenthal DT, Gorlia T, Gilbert MR, Kim
MM, Nabors L Burt, Mason WP, Hegi ME, Zhang P, Golfinopoulos V,
Perry JR, et al: Is more better? The impact of extended adjuvant
temozolomide in newly diagnosed glioblastoma: A secondary analysis
of EORTC and NRG Oncology/RTOG. Neuro Oncol. 19:1119–1126. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Heerdt BG, Houston MA and Angenlicht LH:
Potentiation by specific short-chain fatty acids of differentiation
and apoptosis in human colonic carcinoma cell lines. Cancer Res.
54:3288–3293. 1994.PubMed/NCBI
|
|
7
|
Hague A, Elder DJ, Hicks DJ and Paraskeva
C: Apoptosis in colorectal tumour cells: Induction by the short
chain fatty acids butyrate, propionate and acetate and by the bile
salt deoxycholate. Int J Cancer. 60:400–406. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Farrow B, Rychahou P, O'Connor KL and
Evers BM: Butyrate inhibits pancreatic cancer invasion. J
Gastrointest Surg. 7:864–870. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang XM, Li J and Evers BM: Inhibition of
proliferation, invasion and adhesion of liver cancer cells by
5-azacytidine and butyrate. Anticancer Res. 19:2901–2906.
1999.PubMed/NCBI
|
|
10
|
Engelhard HH, Duncan HA, Kim S, Criswell
PS and Van Eldik L: Therapeutic effect of sodium butyrate on glioma
cells in vitro and in the rat C6 glioma model. Neurosurgery.
48:616–625. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hague A, Manning AM, Hanlon KA, Huschtscha
LI, Hart D and Paraskeva C: Sodium butyrate induces apoptosis in
human colonic tumour cell lines in a p53-independent pathway:
Implications for the possible role of dietary fibre in the
prevention of large-bowel cancer. Int J Cancer. 55:498–505. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Medina V, Edmonds B, Young GP, James R,
Appleton S and Zalewski PD: Induction of caspase-3 protease
activity and apoptosis by butyrate and trichostatin A (inhibitors
of histone deacetylase): Dependence on protein synthesis and
synergy with a mitochondrial/cytochrome c-dependent pathway. Cancer
Res. 57:3697–3707. 1997.PubMed/NCBI
|
|
13
|
Nakagawa H, Yoshioka K, Miyahara E,
Fukushima Y, Tamura M and Itoh K: Intrathecal administration of
Y-27632, a specific Rho-associated kinase inhibitor, for rat
neoplastic meningitis. Mol Cancer Res. 3:425–433. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Naka N, Takenaka S, Araki N, Miwa T,
Hashimoto N, Yoshioka K, Joyama S, Hamada K, Tsukamoto Y, Tomita Y,
et al: Synovial sarcoma is a stem cell malignancy. Stem Cells.
28:1119–1131. 2010.PubMed/NCBI
|
|
15
|
Park CM, Park MJ, Kwak HJ, Moon SI, Yoo
DH, Lee HC, Park IC, Rhee CH and Hong SI: Induction of p53-mediated
apoptosis and recovery of chemosensitivity through p53 transduction
in human glioblastoma cells by cisplatin. Int J Oncol. 28:119–125.
2006.PubMed/NCBI
|
|
16
|
Itoh K, Yoshioka K, Akedo H, Uehata M,
Ishizaki T and Narumiya S: An essential part for Rho-associated
kinase in the transcellular invasion of tumor cells. Nat Med.
5:221–225. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yoshioka K, Nakamori S and Itoh K:
Overexpression of small GTP-binding protein RhoA promotes invasion
of tumor cells. Cancer Res. 59:2004–2010. 1999.PubMed/NCBI
|
|
18
|
Yoshioka K, Foletta V, Bernard O and Itoh
K: A role for LIM-kinase in cancer invasion. Proc Natl Acad Sci
USA. 100:pp. 7247–7252. 2003; View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yui Y, Itoh K, Yoshioka K, Naka N,
Watanabe M, Hiraumi Y, Matsubara H, Watanabe K, Sano K, Nakahata T
and Adachi S: Mesenchymal mode of migration participates in
pulmonary metastasis of mouse osteosarcoma LM8. Clin Exp
Metastasis. 27:619–630. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Karsy M, Guan J, Cohen AL, Jensen RL and
Colman H: New molecular considerations for Glioma: IDH, ATRX, BRAF,
TERT, H3 K27M. Curr Neurol Neurosci Rep. 17:192017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Pant K, Yadav AK, Gupta P, Islam R, Saraya
A and Venugopal SK: Butyrate induces ROS-mediated apoptosis by
modulating miR-22/SIRT-1 pathway in hepatic cancer cells. Redox
Biol. 12:340–349. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Han R, Sun Q, Wu L, Zeng P and Zhao G:
Sodium butyrate upregulates miR-203 expression to exert
Anti-proliferation effect on colorectal cancer cells. Cell Physiol
Biochem. 39:1916–1929. 2016. View Article : Google Scholar
|
|
23
|
Jeon HY, Kim JK, Ham SW, Oh SY, Kim J,
Park JB, Lee JY, Kim SC and Kim H: Irradiation induces glioblastoma
cell senescence and senescence-associated secretory phenotype.
Tumor Biol. 37:5857–5867. 2016. View Article : Google Scholar
|
|
24
|
Morelli MB, Amantini C, Nabissi M,
Cardinali C, Santoni M, Bermadini G, Santoni A and Santoni G:
Axitinib induces senescence-associated cell death and necrosis in
glioma cell lines: The proteasome inhibitor, bortezomib,
potentiates axitinib-induced cytotoxicity in a p21(Waf/Cip1)
dependent manner. Oncotarget. 8:3380–3395. 2017.PubMed/NCBI
|
|
25
|
Itoh K, Yoshioka K and Nakagawa H: Sodium
butyrate induced cellular senescence and inhibited invasion of
cancer cells with distinct mechanism. Cancer Res. 73 8
Suppl:S26242013. View Article : Google Scholar
|