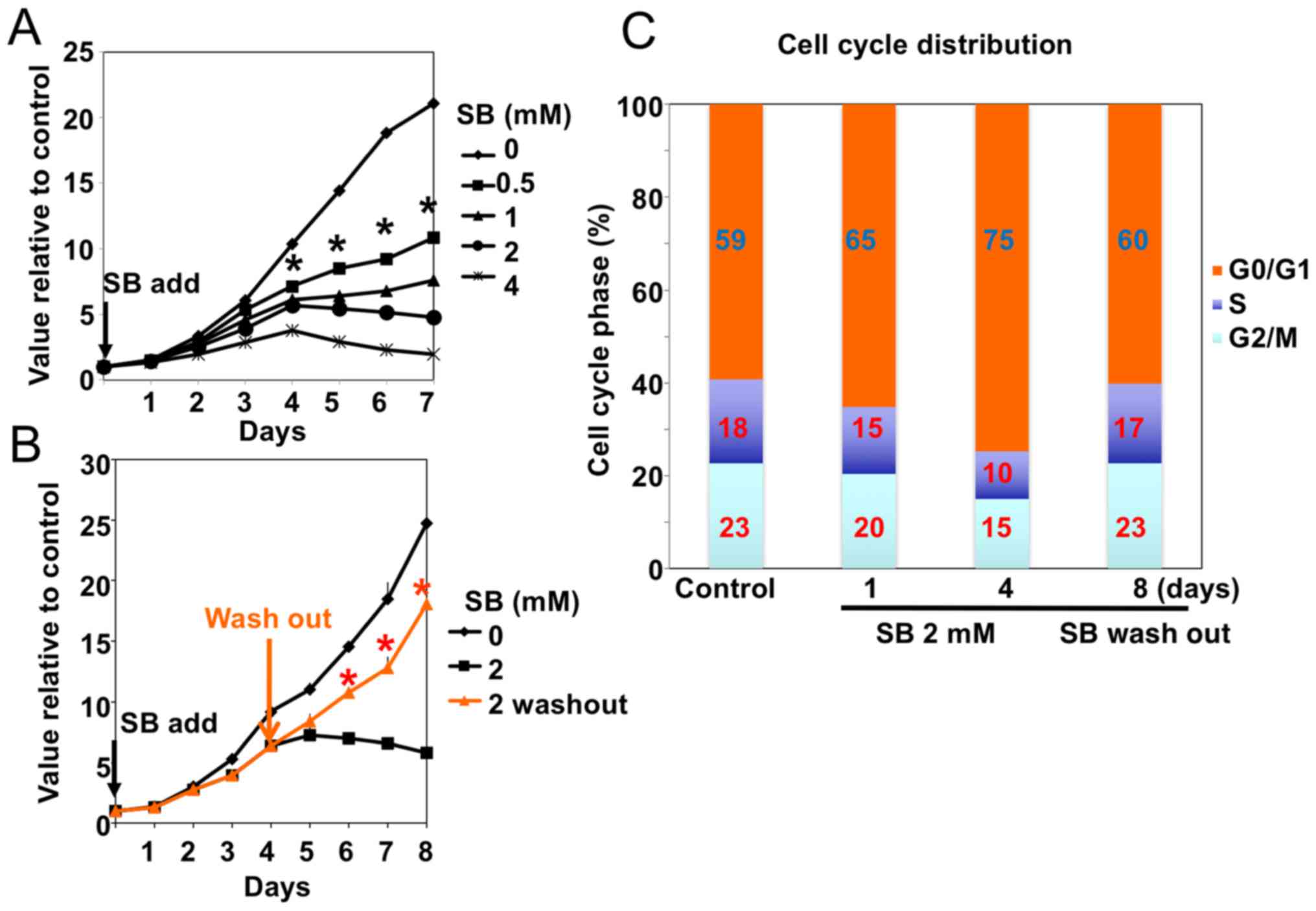
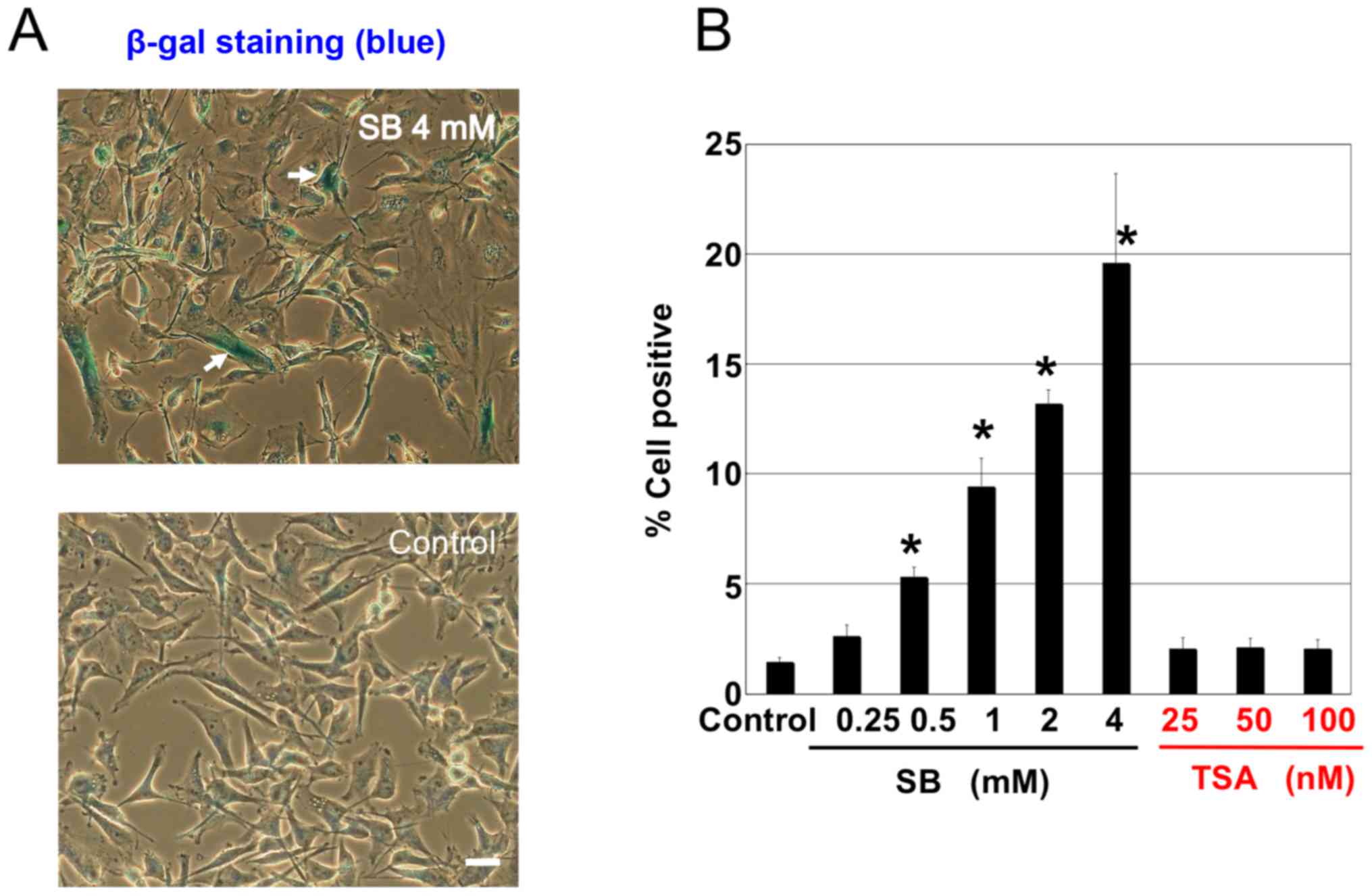
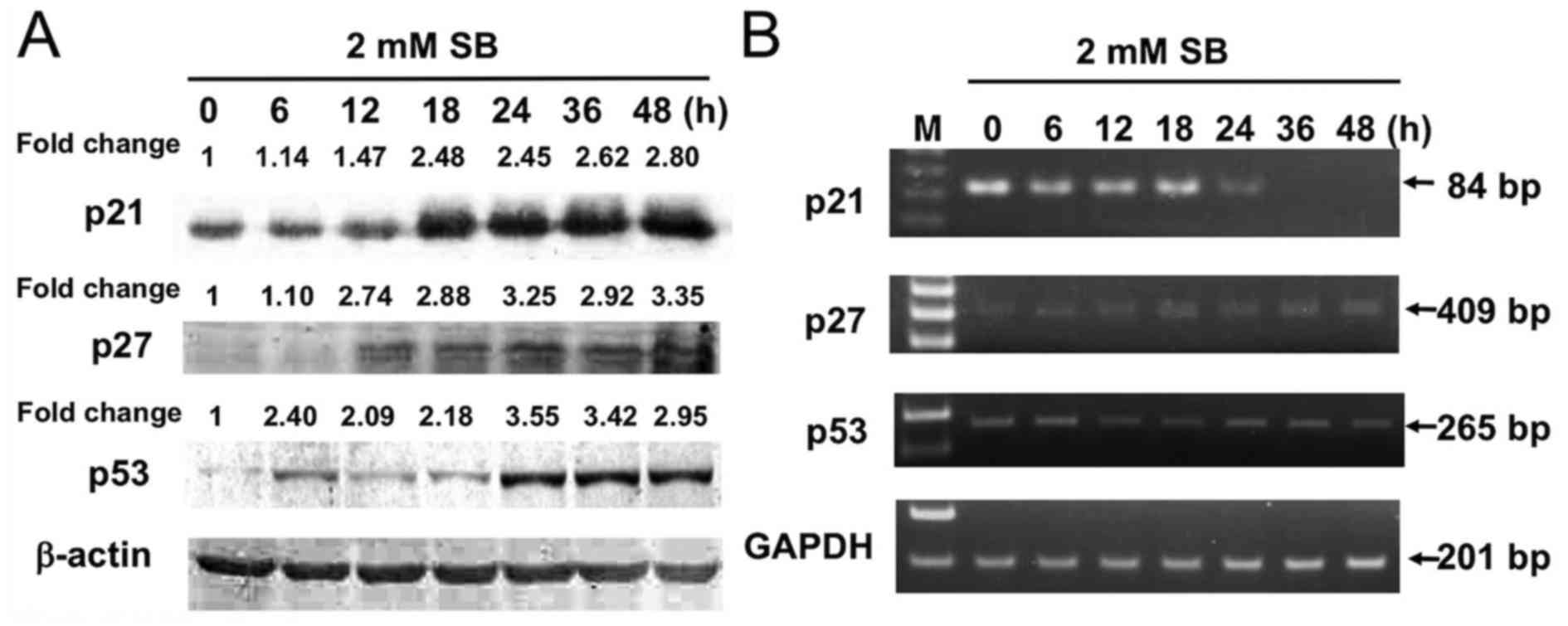
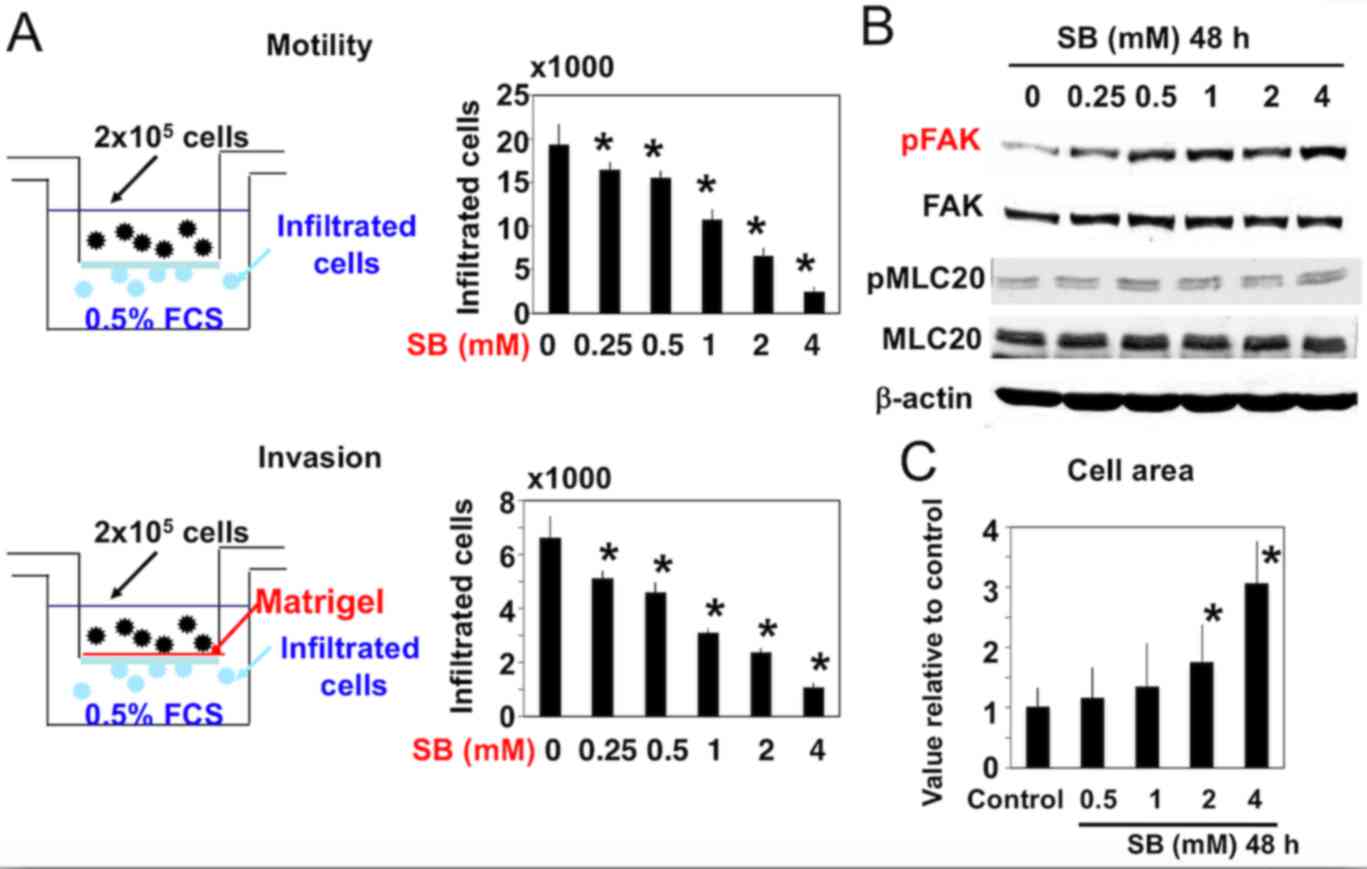
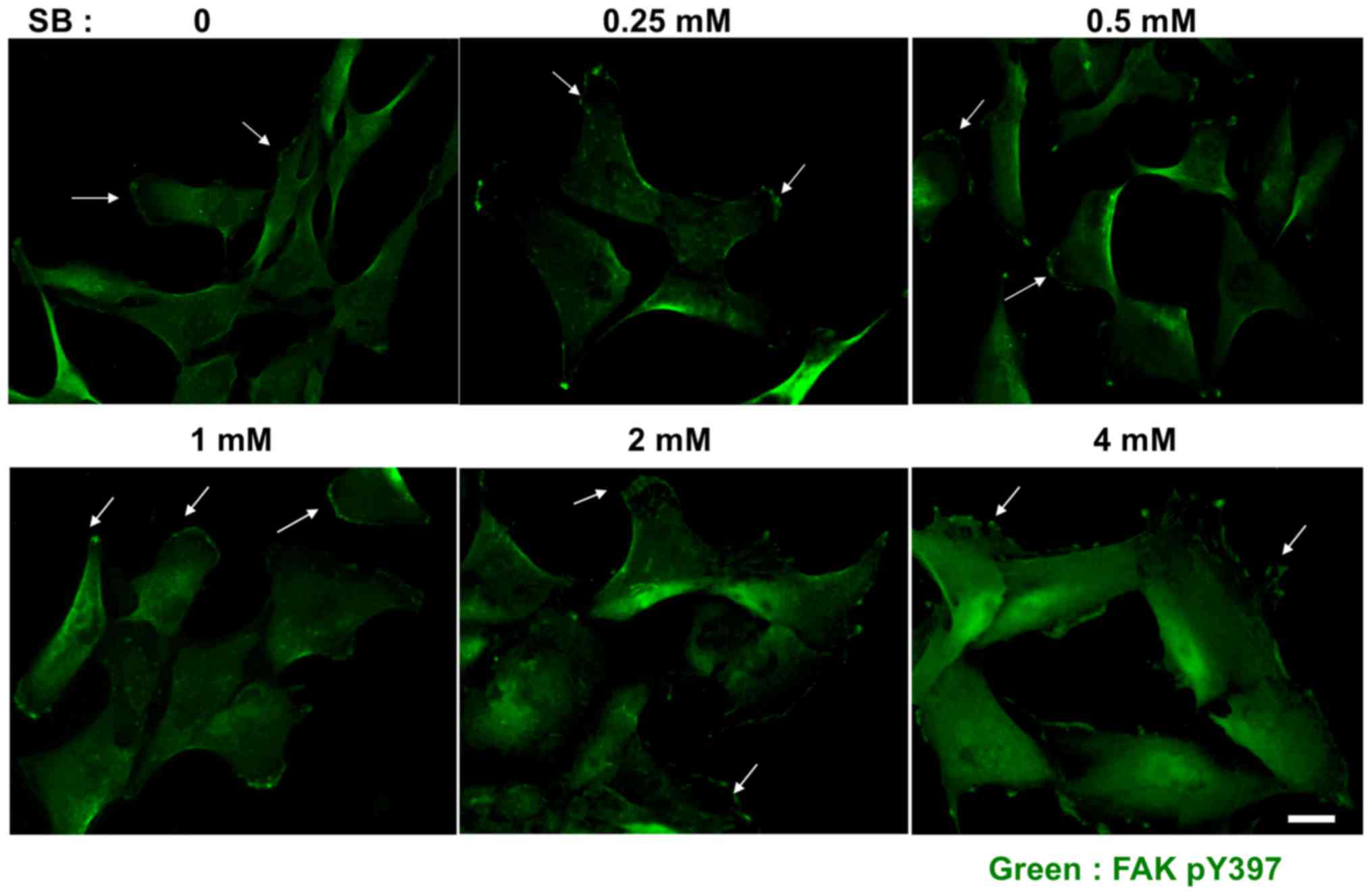
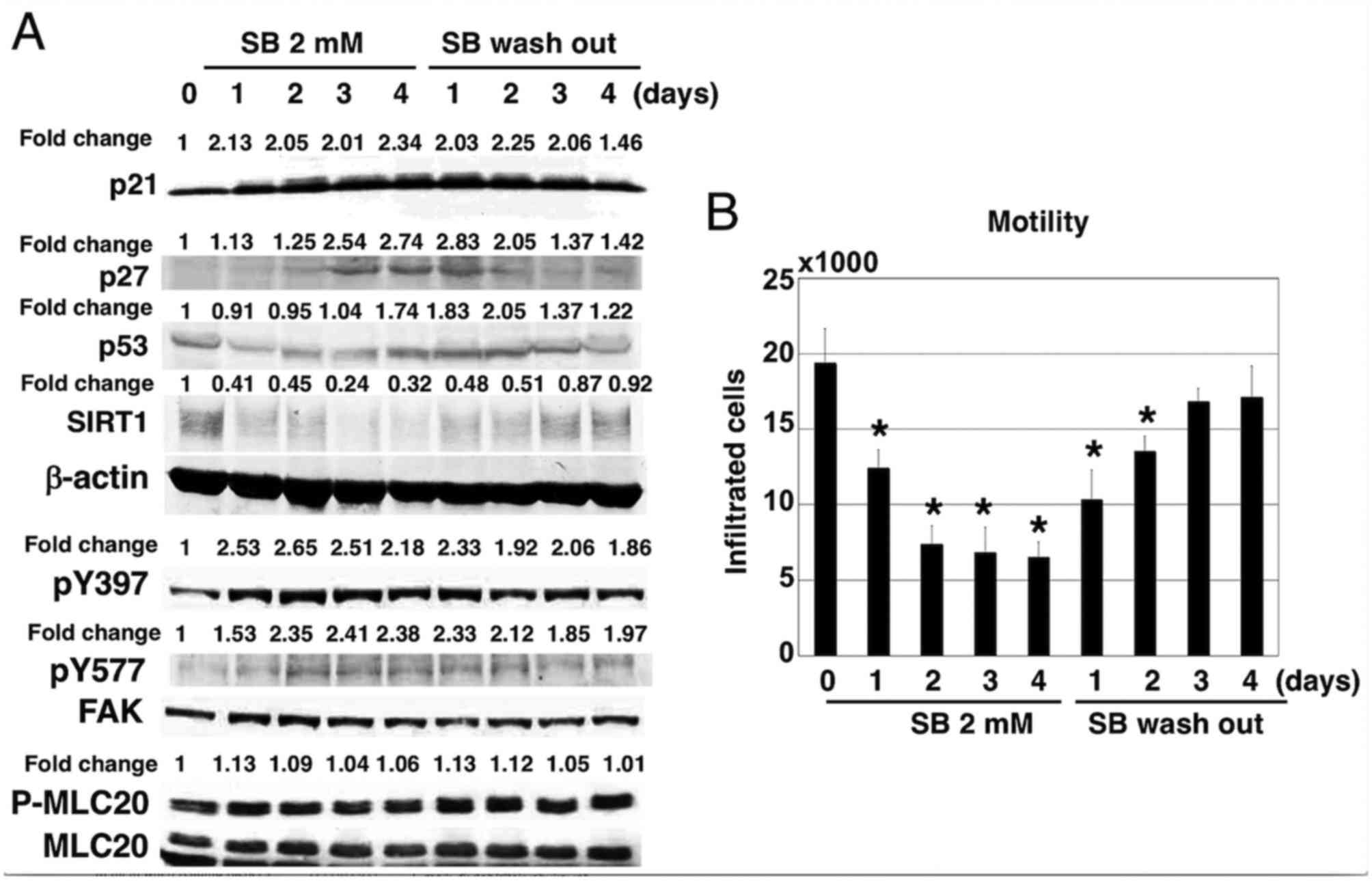
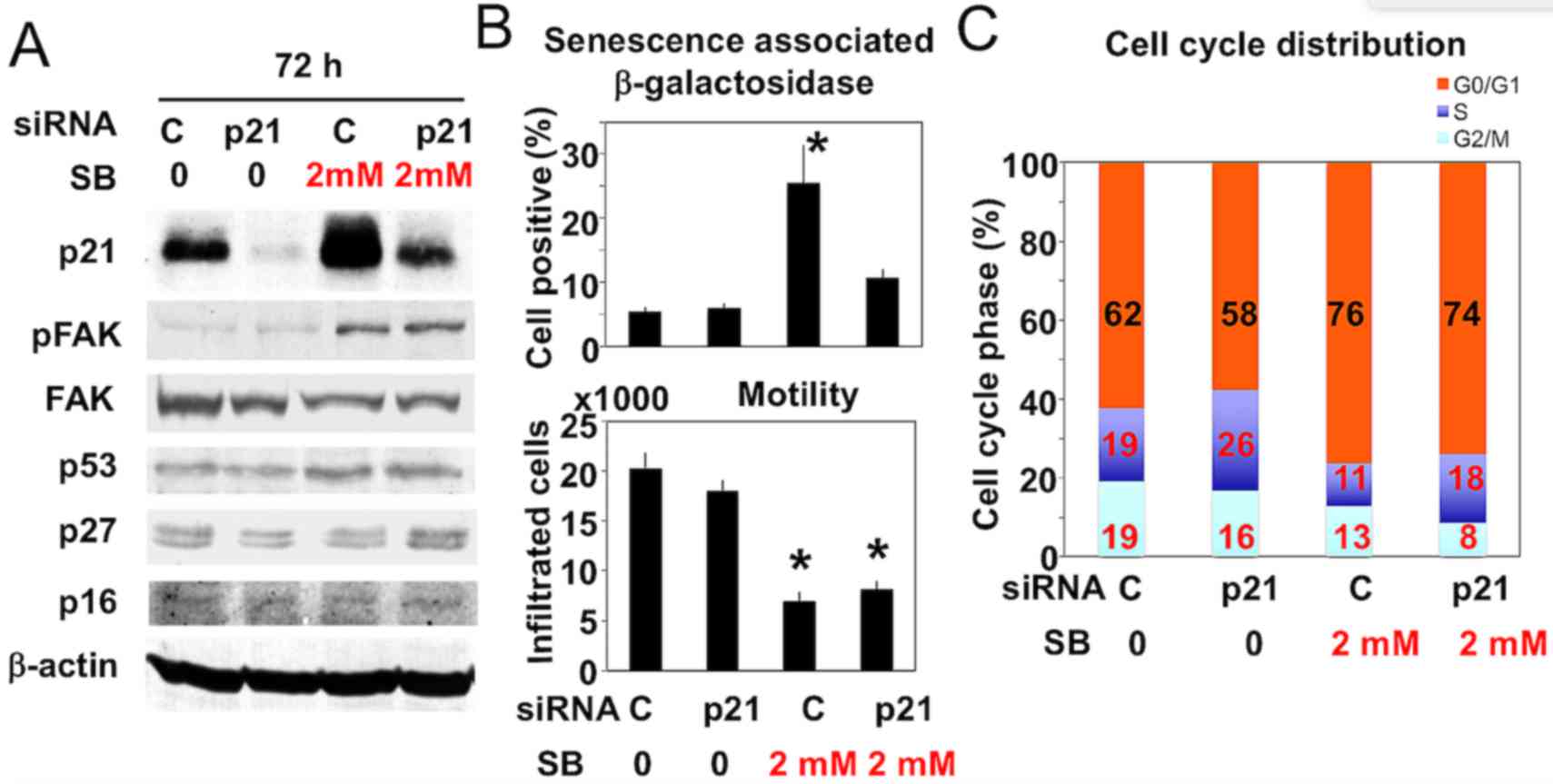
|
1
|
Stupp R, Mason WP, van den Bent MJ, Weller
M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn
U, et al: Radiotherapy plus concomitant and adjuvant temozolomide
for glioblastoma. N Engl J Med. 352:987–996. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Stupp R, Hegi ME, Mason WP, van den Bent
MJ, Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A, Fisher B,
Belanger K, et al: Effects of radiotherapy with concomitant and
adjuvant temozolomide versus radiotherapy alone on survival in
glioblastoma in a randomised phase III study: 5-year analysis of
the EORTC-NCIC trial. Lancet Oncol. 10:459–466. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gzell C, Back M, Wheeler H, Bailey D and
Foote M: Radiotherapy in Glioblastoma: The Past, the present and
the future. Clin Oncol (R Coll Radiol). 29:15–25. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nakagawa H, Yamada M, Kanayama T,
Tsuruzono K, Miyawaki Y, Tokiyoshi K, Hagiwara Y and Hayakawa T:
Myelin basic protein in the cerebrospinal fluid of patients with
brain tumors. Neurosurgery. 34:825–833. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Blumenthal DT, Gorlia T, Gilbert MR, Kim
MM, Nabors L Burt, Mason WP, Hegi ME, Zhang P, Golfinopoulos V,
Perry JR, et al: Is more better? The impact of extended adjuvant
temozolomide in newly diagnosed glioblastoma: A secondary analysis
of EORTC and NRG Oncology/RTOG. Neuro Oncol. 19:1119–1126. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Heerdt BG, Houston MA and Angenlicht LH:
Potentiation by specific short-chain fatty acids of differentiation
and apoptosis in human colonic carcinoma cell lines. Cancer Res.
54:3288–3293. 1994.PubMed/NCBI
|
|
7
|
Hague A, Elder DJ, Hicks DJ and Paraskeva
C: Apoptosis in colorectal tumour cells: Induction by the short
chain fatty acids butyrate, propionate and acetate and by the bile
salt deoxycholate. Int J Cancer. 60:400–406. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Farrow B, Rychahou P, O'Connor KL and
Evers BM: Butyrate inhibits pancreatic cancer invasion. J
Gastrointest Surg. 7:864–870. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang XM, Li J and Evers BM: Inhibition of
proliferation, invasion and adhesion of liver cancer cells by
5-azacytidine and butyrate. Anticancer Res. 19:2901–2906.
1999.PubMed/NCBI
|
|
10
|
Engelhard HH, Duncan HA, Kim S, Criswell
PS and Van Eldik L: Therapeutic effect of sodium butyrate on glioma
cells in vitro and in the rat C6 glioma model. Neurosurgery.
48:616–625. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hague A, Manning AM, Hanlon KA, Huschtscha
LI, Hart D and Paraskeva C: Sodium butyrate induces apoptosis in
human colonic tumour cell lines in a p53-independent pathway:
Implications for the possible role of dietary fibre in the
prevention of large-bowel cancer. Int J Cancer. 55:498–505. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Medina V, Edmonds B, Young GP, James R,
Appleton S and Zalewski PD: Induction of caspase-3 protease
activity and apoptosis by butyrate and trichostatin A (inhibitors
of histone deacetylase): Dependence on protein synthesis and
synergy with a mitochondrial/cytochrome c-dependent pathway. Cancer
Res. 57:3697–3707. 1997.PubMed/NCBI
|
|
13
|
Nakagawa H, Yoshioka K, Miyahara E,
Fukushima Y, Tamura M and Itoh K: Intrathecal administration of
Y-27632, a specific Rho-associated kinase inhibitor, for rat
neoplastic meningitis. Mol Cancer Res. 3:425–433. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Naka N, Takenaka S, Araki N, Miwa T,
Hashimoto N, Yoshioka K, Joyama S, Hamada K, Tsukamoto Y, Tomita Y,
et al: Synovial sarcoma is a stem cell malignancy. Stem Cells.
28:1119–1131. 2010.PubMed/NCBI
|
|
15
|
Park CM, Park MJ, Kwak HJ, Moon SI, Yoo
DH, Lee HC, Park IC, Rhee CH and Hong SI: Induction of p53-mediated
apoptosis and recovery of chemosensitivity through p53 transduction
in human glioblastoma cells by cisplatin. Int J Oncol. 28:119–125.
2006.PubMed/NCBI
|
|
16
|
Itoh K, Yoshioka K, Akedo H, Uehata M,
Ishizaki T and Narumiya S: An essential part for Rho-associated
kinase in the transcellular invasion of tumor cells. Nat Med.
5:221–225. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yoshioka K, Nakamori S and Itoh K:
Overexpression of small GTP-binding protein RhoA promotes invasion
of tumor cells. Cancer Res. 59:2004–2010. 1999.PubMed/NCBI
|
|
18
|
Yoshioka K, Foletta V, Bernard O and Itoh
K: A role for LIM-kinase in cancer invasion. Proc Natl Acad Sci
USA. 100:pp. 7247–7252. 2003; View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yui Y, Itoh K, Yoshioka K, Naka N,
Watanabe M, Hiraumi Y, Matsubara H, Watanabe K, Sano K, Nakahata T
and Adachi S: Mesenchymal mode of migration participates in
pulmonary metastasis of mouse osteosarcoma LM8. Clin Exp
Metastasis. 27:619–630. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Karsy M, Guan J, Cohen AL, Jensen RL and
Colman H: New molecular considerations for Glioma: IDH, ATRX, BRAF,
TERT, H3 K27M. Curr Neurol Neurosci Rep. 17:192017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Pant K, Yadav AK, Gupta P, Islam R, Saraya
A and Venugopal SK: Butyrate induces ROS-mediated apoptosis by
modulating miR-22/SIRT-1 pathway in hepatic cancer cells. Redox
Biol. 12:340–349. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Han R, Sun Q, Wu L, Zeng P and Zhao G:
Sodium butyrate upregulates miR-203 expression to exert
Anti-proliferation effect on colorectal cancer cells. Cell Physiol
Biochem. 39:1916–1929. 2016. View Article : Google Scholar
|
|
23
|
Jeon HY, Kim JK, Ham SW, Oh SY, Kim J,
Park JB, Lee JY, Kim SC and Kim H: Irradiation induces glioblastoma
cell senescence and senescence-associated secretory phenotype.
Tumor Biol. 37:5857–5867. 2016. View Article : Google Scholar
|
|
24
|
Morelli MB, Amantini C, Nabissi M,
Cardinali C, Santoni M, Bermadini G, Santoni A and Santoni G:
Axitinib induces senescence-associated cell death and necrosis in
glioma cell lines: The proteasome inhibitor, bortezomib,
potentiates axitinib-induced cytotoxicity in a p21(Waf/Cip1)
dependent manner. Oncotarget. 8:3380–3395. 2017.PubMed/NCBI
|
|
25
|
Itoh K, Yoshioka K and Nakagawa H: Sodium
butyrate induced cellular senescence and inhibited invasion of
cancer cells with distinct mechanism. Cancer Res. 73 8
Suppl:S26242013. View Article : Google Scholar
|