Introduction
Prostate cancer (PCa) was the second most frequently
diagnosed malignant tumor and the fifth leading cause of cancer
mortality in men worldwide in 2012 (1), and its development and progression have
been associated with obesity through extensive previous studies
[reviewed in (2)]. A wide range of
epidemiological studies have demonstrated that obesity was
positively associated with more aggressive PCa, with higher
pathological grade (3), increased
recurrence rate (4) and increased
cancer-specific mortality (5),
whereas the exact molecular mechanisms that contribute to PCa
progression remain unclear.
Excessive adipose tissue deposition in obese
patients, along with its paracrine and endocrine impact on tumor
microenvironment, was suggested to exert reciprocal interactions
with cancer cells and contribute to cancer progression (6,7). In
particular, adipokines, a group of cytokines secreted by
adipocytes, together with their associated pro-inflammatory
cytokines, were demonstrated to exert multifaceted effects on
cancer (8). The prostate is
surrounded by marked amounts of adipose tissues, and, accordingly,
the progression of PCa is more likely to be affected by the
aberrant secretion and signaling of adipokines and cytokines in
obese patients in comparison with non-obese patients (9,10). Leptin
and adiponectin are two primary adipokines that have been studied
previously: It was demonstrated that increased serum leptin levels
were positively associated with tumor development and progression,
whereas adiponectin inhibited the cancer growth (11). The downstream pathways and regulatory
mechanisms were complicated, involving apoptosis, cell cycle,
angiogenesis and migration (11).
Notably, a previous study identified that adipocytes
cultivated with cancer cells exhibited an altered phenotype, with
delipidation in cells, decreased adipocyte markers and
overexpression of pro-inflammatory cytokines (12). This study considered that the
cancer-associated adipocytes may be an important factor that
contributed to cancer progression. However, the function of these
cancer-associated adipocytes in cancer development and progression,
involving the crosstalk of adipocytes and cancer cells, remains
largely unknown.
To elucidate the effect of obesity on PCa growth and
progression, a high-fat diet (HFD) was used to induce obesity in a
transgenic adenocarcinoma of mouse prostate (TRAMP) animal model,
which closely resembled the natural progression of PCa in obese
human patients (13). Through a
series of in vivo and in vitro experiments, the
hypothesis that HFD-induced obesity may promote the PCa progression
via altered serum adipokines and cytokines was tested. The trends
in serum adipokine and cytokine alterations, which were jointly
affected by HFD-induced obesity and PCa progression were also
investigated.
Materials and methods
Animals and diets
All of the animal studies were approved by the
Institutional Animal Care and Use Committee from Department of
Laboratory Animal Science, Fudan University (Shanghai, China). A
total of 108 TRAMP mice were obtained from Jackson Laboratory (Bar
Harbor, ME, USA), bred and maintained under pathogen-free
conditions at the Department of Laboratory Animal Science, Fudan
University. Each mouse was kept and raised separately in a cage,
with cork dust bedding and a 12-h light/12-h dark cycle, with an
environmental temperature of 22–26°C and humidity of 50–60%. All
male TRAMP mice were selected by genotyping as previously described
(14), and then randomly assigned to
two groups, where the mice were fed on a control diet (CD) or an
HFD ad libitum at 5 weeks of age. CD (16% calories from fat)
and HFD (40% calories from fat) was supplied by Puluteng
Biotechnology, Co., Ltd. (http://www.slaccas.com; Shanghai, China; Table I).
 | Table I.Nutrient and energy compositions of
the control and high-fat diets. |
Table I.
Nutrient and energy compositions of
the control and high-fat diets.
| Component | Control diet | High-fat diet |
|---|
| Nutrients (g) |
|
|
| Fat | 7 | 20 |
|
Carbohydratesa | 64 | 45 |
|
Protein | 20 | 22 |
| Energy (%) |
|
|
| Fat | 16 | 40 |
|
Carbohydrates | 64 | 40 |
|
Protein | 20 | 20 |
To evaluate the effect of HFD on the development and
progression of prostate cancer, TRAMP mice were sacrificed at 20,
24 and 28 weeks of the age according to previous studies (CD-fed
TRAMP: 20-week, 24-week, 28-week, three groups, n=12 mice/group;
HFD-fed TRAMP: 20-week, 24-week, 28-week, three groups, n=24
mice/group) (15,16).
Systemic evaluation, serum and tissue
preparation
All TRAMP mice were not fed overnight, and received
weight and blood glucose examinations prior to sacrifice. At the
time of sacrifice, each TRAMP mouse was anaesthetized with an
intraperitoneal injection of pentobarbital (50 mg/kg), scanned by
GE eXplore Locus micro-computed tomography (CT) scanner (GE
Healthcare BioSciences, Pittsburg, PA, USA) for systemic
evaluation, and euthanized by asphyxiation with CO2.
During euthanasia, the mice were kept in a 3 l-cage, then 100%
CO2 was introduced at a flow rate of 0.75 l per min. The
final concentration of CO2 reached was ~75%. The
mortality of the mice was confirmed by checking breathing and the
dilation of the pupil. Subsequently, blood was harvested from the
portal vein using a 1-ml syringe, and the serum was collected and
stored at −80°C for future analysis. The tumor, genitourinary
tract, epididymal fat, liver and lung were removed from the TRAMP
mice, weighed and fixed for histopathological analysis. The
epididymal fat pad was dissected between the distal major blood
vessel and the epididymis from each side.
Histopathological analysis
Prostate and other prepared tissues were fixed in
10% buffered formalin, processed in an alcohol (80, 90 and
100%)-xylene series and embedded in paraffin (at 56°C for 4 h). A
series of 2-µm sections were prepared, and stained with hematoxylin
(0.5%) for 10 min and eosin (0.5%) for 2 min (H&E) at 26°C. A
light microscope was used at a magnification of ×40 and ×200. For
evaluation of tumor differentiation, local invasion and detection
of distant metastasis, these sections were additionally analyzed
and cross-checked by two pathologists from Huashan Hospital, Fudan
University (Shanghai, China). The differentiation of prostate
cancer was identified based on Gleason score and categorized into
three groups, including well-differentiated (Gleason score, <7),
moderately differentiated (Gleason score, 7) and poorly
differentiated (Gleason score, >7) (17).
Serological analysis
Concentrations (in pg/ml) of different adipokines
and associated cytokines in mouse sera were measured by commercial
ProcartaPlex Multiplex Immunoassays (eBioscience; Thermo Fisher
Scientific, Inc., Waltham, MA, USA), which use the Luminex
technology with Multi-Analyte Profiling beads and enable the
simultaneous detection and quantification of multiple cytokines in
one sample. The equipment was used according to the manufacturer's
protocol. A total of 30 µl serum from each mouse was examined
following the manufacturer's protocol. The adipokines and cytokines
investigated included leptin, adiponectin, interleukin (IL)-4,
IL-5, IL-6, IL12-p70, interferon (IFN)-γ, tumor necrosis factor
(TNF)-α, CC chemokine ligand (CCL)2, CCL3, CCL4, CCL5, CCL7, CCL11,
CXC chemokine ligand (CXCL)1, CXCL2 and CXCL10.
Cell culture and culture medium
preparation
DU145 cells, an androgen-independent human prostate
cancer cell line, were purchased from the Type Culture Collection
of the Chinese Academy of Sciences (Shanghai, China). The DU145
cells were cultured in Ham's F-12K (Kaighn's) medium (Gibco; Thermo
Fisher Scientific, Inc.), supplemented with 10% fetal bovine serum
(Gibco; Thermo Fisher Scientific, Inc.) and 1% penicillin and
streptomycin combination. Cells were incubated at 37°C in a
humidified atmosphere of 5% CO2.
In the series of in vitro experiments, cells
were treated with three different culture media: i) Serum-free
medium (SFM); ii) HFD-sera-conditioned medium (HFD-CM) which
contained SFM plus 2.5% mixed sera from all experimental 28-week
HFD-fed TRAMP mice; and iii) CD-sera-conditioned medium (CD-CM)
which contained SFM plus 2.5% mixed sera from all experimental
28-week CD-fed TRAMP mice.
Cell Counting Kit-8 (CCK-8)
proliferation assay
Cell proliferation was determined using a CCK-8
assay (Dojindo Molecular Technologies, Inc., Kumamoto, Japan).
Exponential phase DU145 cells were seeded in 96-well plates (10,000
cells/well) in culture medium at 37°C overnight, and then the
medium was replaced by SFM for 18 h. Subsequently, cells were
treated with SFM, CD-CM or HFD-CM, and incubated for 0, 24, 48, 72
and 96 h. Each group was tested in triplicate in three replicate
wells. CCK-8 solution (10 µl) was added to each well at each time
point as mentioned above. Following incubation at 37°C for another
2.5 h, the optical density (OD) values were determined at a
wavelength of 450 nm using a microplate reader BioTek, ELx800
(BioTek Instruments, Winooski, VT, USA) at each time point.
Wound-healing assay
Cell migration was studied by conducting a
wound-healing assay. DU145 cells were seeded on 6-well plates
(50,000 cells/well) in culture medium. After 24 h, culture medium
was replaced by SFM for 18 h. A wound was generated by scratching
down the cells in the center of the well with a sterile pipette
tip, and the detached cells were washed away with PBS. Then, the
cells were incubated in the following experimental conditions for
96 h: SFM, CD-CM and HFD-CM. Images were captured using Nikon
TE2000 inverted microscope (magnification, ×40) connected to a
Nikon Coolpix 4500 camera (Nikon Corporation, Tokyo, Japan) at 0,
24, 48, 72 and 96 h. The distance between the scratched regions
were additionally measured with Spot Imaging 5.2 software
(Diagnostic Instruments, Sterling Heights, MI, USA).
Matrigel-Transwell invasion assay
Cell invasion was studied by conducting a
Matrigel-Transwell assay with BD Biocoat Matrigel Chambers
(Discovery Labware; BD Biosciences, Franklin Lakes, NJ, USA). A
total of ~120,000 DU145 cells were suspended and spread to the
upper chamber under serum-free conditions, and the lower chamber of
the well was filled with SFM, CD-CM or HFD-CM as a chemoattractant.
After 24 h, inserted cells were fixed in 95% ethanol (at 26°C for
30 min) and stained with 1.0% crystal violet (at 26°C for 10 min).
The images of inserts were captured using a Nikon TE2000 inverted
microscope connected to a Nikon Coolpix 4500 camera (Nikon
Corporation). Subsequently, cells were counterstained with 33%
acetic acid, and the OD of the counterstained solution was
additionally examined using a microplate reader at a wavelength of
570 nm.
Statistical analysis
Results are presented as the mean ± standard error
of the mean. Cell experiments were conducted in triplicate. The
TRAMP mouse sample size was determined by a previous study
(14) that characterized the mouse
model, and all mice were randomized into different groups in a
blinded manner. Analyses were conducted with an unpaired two-tailed
Student's t-test, χ2 test, Fisher's exact test or
two-way analysis of variance followed by Sidak's/Tukey's multiple
comparisons test, using GraphPad Prism (version 6.0c; GraphPad
Software, La Jolla, CA, USA). P<0.05 was considered to indicate
a statistically significant difference.
Results
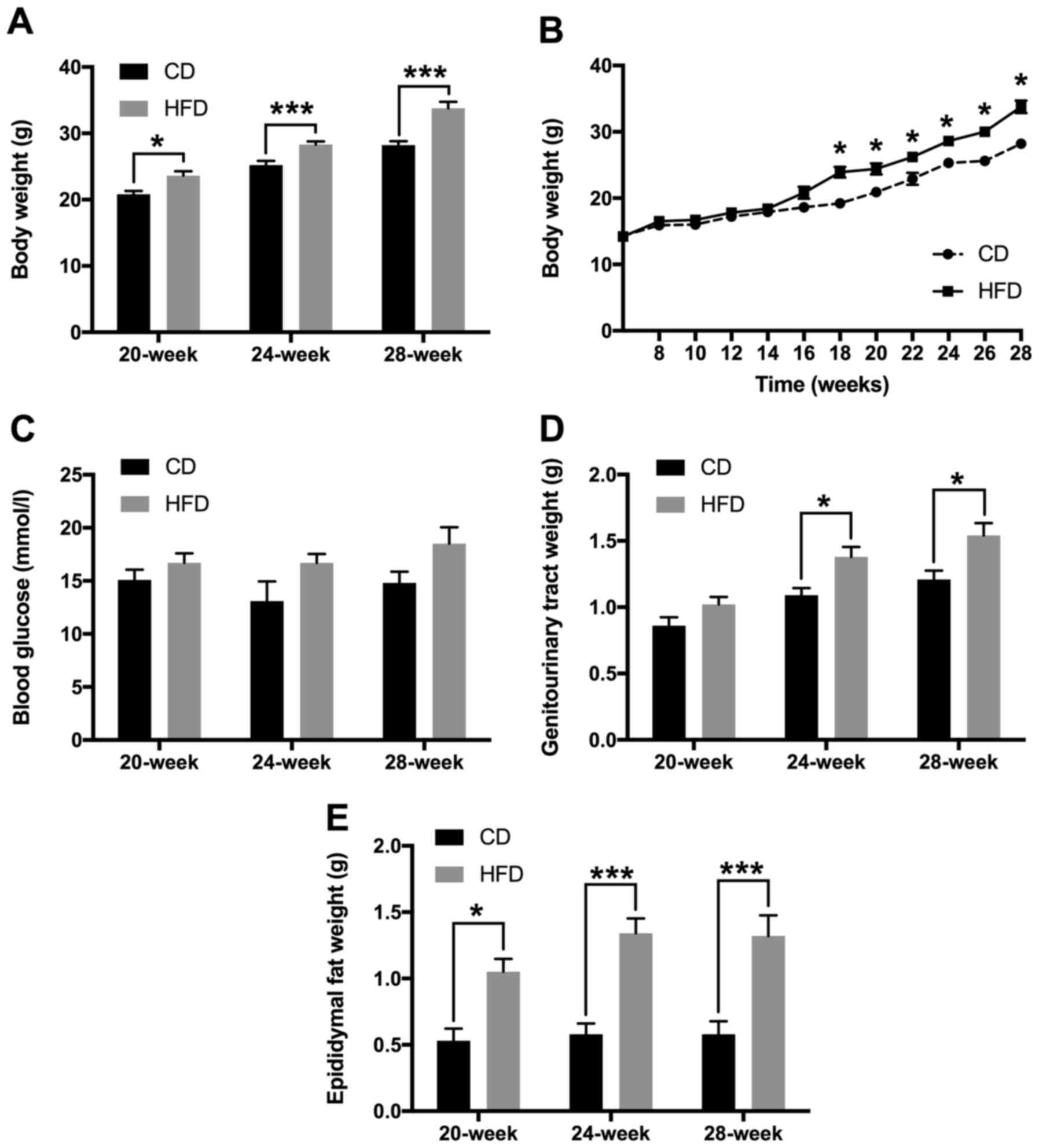
HFD feeding increases body weight and
fat deposition in TRAMP mice
HFD feeding increased the body weight of TRAMP mice
compared with CD feeding. The differences in mean body weight
between HFD-fed and CD-fed TRAMP mice were all of statistical
significance in the 20-week (23.6±3.4 vs. 20.8±1.8 g; P<0.05),
24-week (28.3±2.4 vs. 25.2±2.3 g; P<0.001) and 28-week groups
(33.8±4.7 vs. 28.2±2.1 g; P<0.001) (Fig. 1A). This difference appeared to be
time-dependent (Fig. 1B). Blood
glucose levels of the mice following fasting overnight did not
differ markedly between CD-fed and HFD-fed groups (Fig. 1C). As expected, HFD-fed TRAMP mice
presented with increased genitourinary tract weight compared CD-fed
mice in the 24- and 28-week groups (Fig.
1D). Additionally, epididymal fat weight was also increased in
HFD-fed mice in all comparisons (Fig.
1E).
HFD feeding promotes tumor progression
in TRAMP mice
Micro-CT scanning, optic observation and
histopathological studies of dissected tissues were applied to
evaluate the tumor development and progression. Prostate cancer was
detected in all TRAMP mice from the CD-fed or HFD-fed groups. In
the older TRAMP mice, PCa tended to be more advanced and
aggressive, irrespective of the CD or HFD. In addition, HFD-fed
mice possessed PCa with poorer differentiation as compared with
CD-fed mice at the same age (Fig.
2A). An increased number of events of local invasion and
metastasis were detected in HFD-fed mice compared with their CD-fed
counterparts (Fig. 2B and C). The
H&E staining sections also revealed that the PCa of HFD-fed
mice was poorly differentiated compared with CD-fed mice at the
same age (Fig. 2D). Extracapsular
invasion, seminal vesical invasion and metastasis to
retroperitoneal lymph nodes or lung were common in the 28-week
HFD-fed TRAMP mice in the present study (Fig. 2E and F).
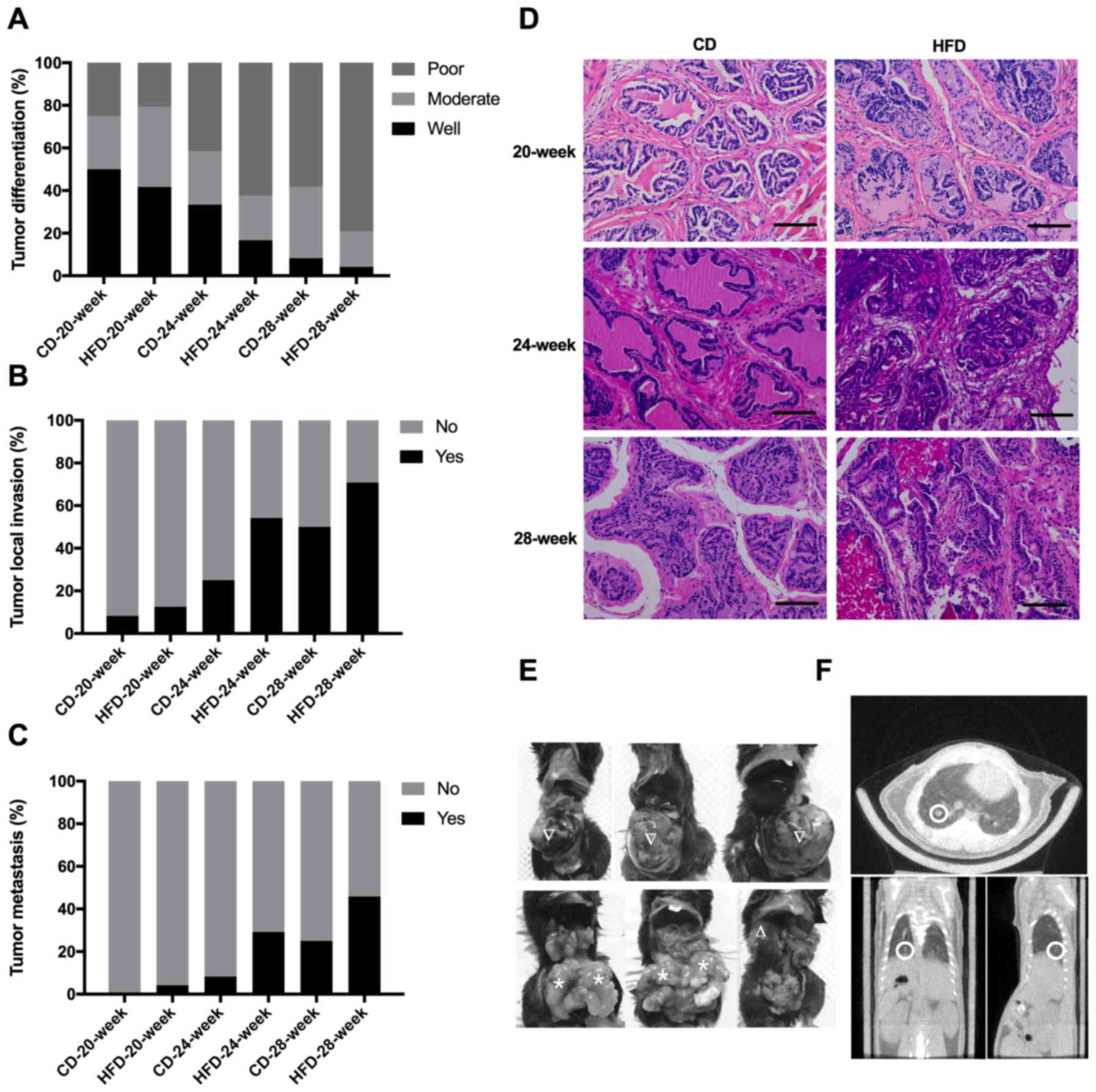 | Figure 2.TRAMP mice fed on an HFD exhibited
more advanced pathological carcinogenic lesions, increased rate of
local invasion and metastasis of prostate cancer. (A) TRAMP mice
fed on an HFD or a CD until 20, 24 or 28 weeks were sacrificed, and
each mouse prostate was dissected, fixed, sectioned and stained
with hematoxylin and eosin. The tumor differentiation was
additionally classified into poor, moderate and well according to
histopathological studies of stained sections. (B) The local
invasion of prostate cancer was determined by histopathological
studies of dissected prostate and surrounding tissues. (C) The
metastasis of prostate cancer was determined by micro-CT scanning
outcomes and histopathological studies of dissected liver, lung and
other organs. (D) Representative pathological images of mice
prostate in different groups (magnification, ×200; scale bar, 100
µm). (E) Representative images of prostate cancer mass (∇),
bilateral seminal vesical invasion (*), and retroperitoneal lymph
node enlargement (∆) in the 28-week HFD-fed TRAMP mice group. (F)
Representative micro-CT images of lung metastasis from a 28-week
HFD-fed TRAMP mouse (demarcated by a white circle). Micro-CT,
micro-computed tomography; HFD, high-fat diet; CD, control diet;
TRAMP, transgenic adenocarcinoma of mouse prostate. |
HFD induces adipokine and cytokine
alterations in mouse sera
As a great variety of adipokines and cytokines that
are associated with tumor development and progression may be
altered by HFD feeding, multiple immunoassays were applied for the
detection of cytokines in TRAMP mouse sera. The comparisons of
cytokine levels depended on three different ages of mice: The
20-week group, where the majority of the CD-fed and HFD-fed mice
experienced organ-confined PCa, the 28-week group, where half of
the mice experienced local invasion and metastasis of PCa, and the
24-week group, with intermediate cancer status. A time-dependent
trend in changes in adipokines and cytokines was detected. HFD
feeding increased leptin levels >2-fold in all age groups, and
the increase appeared to be even more marked in older mice
(Fig. 3A). CCL3 levels were increased
in the CD-fed mice in the 24-week group compared with the HFD-fed
mice at the same time, whereas in the 28-week group, CCL3 levels
were increased in the HFD-fed mice compared with the CD-fed mice
(Fig. 3J). Similarly, levels of CCL4
and CCL5 were increased in the CD-fed mice in the 24-week group;
however, this difference was diminished and the levels were
slightly increased in the HFD-fed mice in the 28-week group
(Fig. 3K and L). CXCL10 levels were
significantly increased in HFD-fed mice compared with CD-fed
counterparts in the 28-week group (Fig.
3Q). The present study failed to detect any difference between
CD-fed and HFD-fed mice in levels of adiponectin, IL-4, IL-5, IL-6,
IL-12p70, IFN-γ, TNF-α, CCL2, CCL7, CCL11, CXCL1 and CXCL2
(Fig. 3).
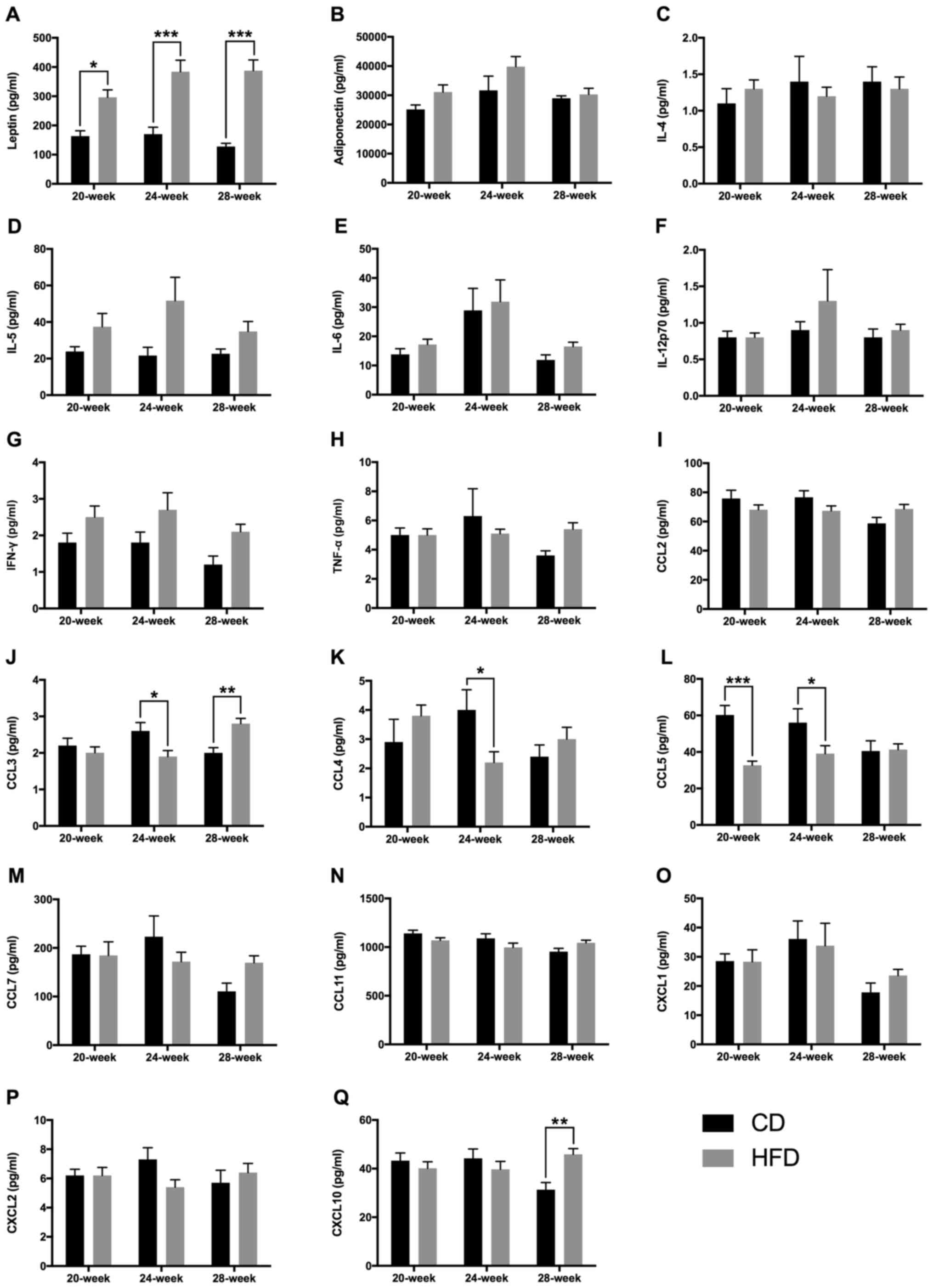 | Figure 3.High-fat diet-induced adipokine and
cytokine alterations in TRAMP mouse sera. TRAMP mice fed on an HFD
or CD were sacrificed at 20, 24 and 28 weeks. Mouse sera were
collected, and serum adipokines and cytokines were examined using
ProcartaPlex Multiplex Immunoassays. The adipokines and cytokines
under examination included (A) leptin, (B) adiponectin, (C) IL-4,
(D) IL-5, (E) IL-6, (F) IL-12p70, (G) IFN-γ, (H) TNF-α, (I) CCL2,
(J) CCL3, (K) CCL4, (L) CCL5, (M), CCL7, (N) CCL11, (O) CXCL1, (P)
CXCL2 and (Q) CXCL10. Results are the mean ± standard error of the
mean. *P<0.05, **P<0.01 and ***P<0.001. IL, interleukin;
IFN-γ, interferon γ; TNF-α, tumor necrosis factor α; CCL, CC
chemokine ligand; CXCL, CXC chemokine ligand; HFD, high-fat diet;
CD, control diet; TRAMP, transgenic adenocarcinoma of mouse
prostate. |
HFD-CM promotes proliferation,
migration and invasion of DU145 cells
A series of in vitro studies were conducted
to determine the proliferative, migratory and invasive abilities of
DU145 cells cultured in SFM, CD-CM and HFD-CM. CD-CM and HFD-CM
stimulated the proliferation of DU145 cells compared with SFM after
72 h of culture, and the increase in the HFD-CM group was more
marked compared with that of the CD-CM counterparts (Fig. 4A). As for cell migration, cells
cultured in HFD-CM exhibited a more marked wound closure speed
compared with cells cultured in CD-CM and SFM following 48 h of
culture (Fig. 4B and C). In addition,
HFD-CM also promoted significantly different cell invasion
abilities in the Transwell-Matrigel assay compared with CD-CM and
SFM (Fig. 4D and E).
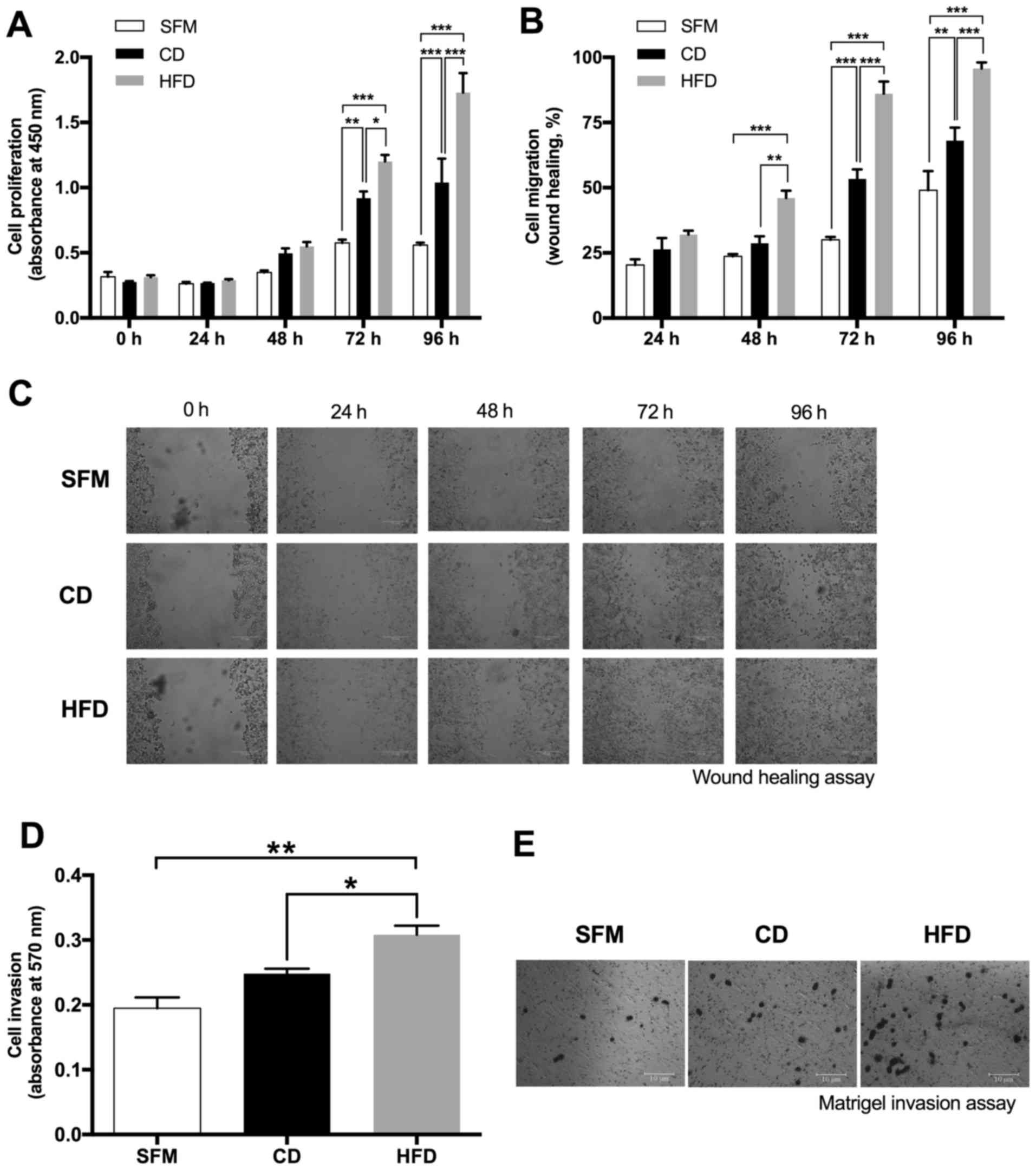 | Figure 4.Sera from high-fat diet-fed TRAMP mice
promoted proliferation, migration and invasion of prostate cancer
cells. (A) DU145 cells were treated with SFM, CM containing 2.5%
mixed sera from CD-fed TRAMP mice or CM containing 2.5% mixed sera
from HFD-fed TRAMP mice. Cell viability was determined using a Cell
Counting Kit-8 assay. (B) Effects of SFM, CD-CM and HFD-CM culture
on the migration of DU145 cells as determined using a wound-healing
assay. (C) Representative images of the wound-healing experiment
demonstrating the migration of DU145 cells cultured with SFM, CD-CM
and HFD-CM, respectively. (D) Invasion of DU145 cells was
determined using a Matrigel-Transwell invasion assay. SFM,
CD-sera-CM or HFD-sera-CM was added to the lower chamber of the
Transwell as the chemoattractant. (E) Representative images of the
Matrigel-Transwell experiment, demonstrating the invasive abilities
of DU145 cells cultured with SFM, CD-CM and HFD-CM, respectively.
Results are presented as the mean ± standard error of the mean
(n=3). *P<0.05, **P<0.01 and ***P<0.001. SFM, serum-free
medium; CD, control diet; HFD, high-fat diet; CM, conditioned
media; TRAMP, transgenic adenocarcinoma of mouse prostate. |
Discussion
A positive association between obesity and PCa
incidence and aggressiveness has been demonstrated previously
(18). However, owing to the
complexity and variability of molecular biological networks,
elucidating the exact role of obesity in PCa progression is
challenging.
High dietary fat, comprising a large part of western
diets, induces obesity by increasing adipose tissue deposition via
adipocyte hypertrophy and hyperplasia (6). In turn, the excess fat deposition tends
to additionally alter the function of the adipocytes, and
accordingly affects cancer progression (6). Several studies were conducted in murine
xenograft models, implicating that diet-induced obesity was
associated with PCa progression (19,20).
Additionally, genetically engineered animal models were of even
greater utility, owing to their normal immune system function, and
orthotopic and spontaneous development of tumors. The first study
indicating a promoting role of HFD feeding in PCa development in
TRAMP mice was conducted in 2010 (21). Subsequently, Cho et al
(20) revealed that an HFD containing
lard increased PCa development and progression, and decreased
survival rates in TRAMP mice. Consistent with previous studies, the
results of the present study indicated that HFD feeding increased
body weight and visceral fat deposition (e.g., increased epididymal
fat) in TRAMP mice. TRAMP mice fed on an HFD presented with poorer
differentiation, increased rates of local invasion and distant
metastasis of PCa at the age of 20, 24 and 28 weeks. Taken
together, these results confirmed that HFD-induced obesity
stimulates the progression of PCa. Conversely, Bonorden et
al (22) failed to detect any
association between HFD and tumor development in TRAMP mice. In
that study, HFD-fed mice were divided into obesity-resistant,
overweight and obesity-prone groups according to their body weight.
These subgroups afforded novel perspectives on the effects of the
response of the body to HFD feeding, and additional bias, which
requires future validation and exploration.
In addition to triacylglycerol storage, it was
established that white adipose tissue also functions in appetite
control, immune and metabolism regulation by secreting and
affecting a large number of adipokines and cytokines (6). Multiple previous studies indicated that
these adipokines and cytokines were critical in regulating tumor
growth and spread, the results of which are as follows. Xu et
al (14) identified that
pro-inflammatory cytokines, including IL-1α, IL-1β, IL-6 and TNF-α,
were increased in HFD-fed mice in comparison with CD-fed mice,
indicating that these cytokines might contribute to PCa
progression. Price et al (9)
demonstrated that PCa cells exposed to sera from obese C57BL/6
mice, containing increased levels of leptin and pro-inflammatory
cytokines and decreased levels of insulin-like growth
factor-binding protein 1 and tissue inhibitor of metalloproteinase
1, promoted cell proliferation, invasion, migration and
epithelial-mesenchymal transition. Previously, cancer-associated
adipocytes have gained attention for their dysmorphisms and altered
functions when located adjacent to cancer cells; they appear to
form reciprocal signaling pathways with cancer cells and promote a
more aggressive cancer phenotype (23,24). Cho
et al (20) conducted a series
of in vitro studies and identified that adipose
tissue-conditioned medium from HFD-fed mice promoted the
proliferation, migration of PCa cells and angiogenesis. Similarly,
the results of the present study revealed that the HFD-CM promoted
proliferation, migration and invasion in DU145 cells compared with
CD-CM or SFM. Owing to the limited quantity of mouse sera, the
experiments were not extended to more cell lines or more serum
concentrations.
Notably, the present study initiated comprehensive
comparisons of different serum adipokine and cytokine levels among
CD-fed and HFD-fed TRAMP mice at the age of 20, 24 and 28 weeks. As
PCa developed and progressed spontaneously in TRAMP mice, the
present study may reveal the complex adipokine and cytokine
alterations affected jointly by obesity and cancer progression,
which best imitated the actual trends of cytokine changes in obese
patients with cancer. In the analysis, leptin levels were
significantly increased in all three HFD feeding groups. However,
the trends in CCL3, CCL4, CCL5 and CXCL10 appeared to be
complicated, where a notable reversal in the changing trends was
detected. Specifically, the levels of these adipokines were
generally increased in CD-fed mouse sera in the 20-week or 24-week
group compared with the HFD-fed mouse sera, whereas levels became
increased in the HFD-fed mouse sera compared with the CD-fed mouse
sera in the 28-week group. These trends could implicate the joint
effects of HFD-induced excess adipose tissue deposition and tumor
progression.
In the present study, five adipokines, including
leptin, CCL3, CCL4, CCL5 and CXCL10, were identified to be involved
in the stimulation of PCa progression by HFD. Leptin, associated
with levels of adipose tissue in human body, was increased in obese
patients (11). Its functions
primarily involve satiety regulation and energy expenditure, and
also serve a role in reproduction, hematogenesis and carcinogenesis
(11). Kato et al (10) identified that leptin exposure
increased the cell migration and invasion and maintenance of the
mesenchymal phenotype of ovarian cancer cells via the activation of
Janus kinase/signal transducer and activator of transcription,
phosphoinositide 3-kinase/protein kinase B and RhoA/RhoA kinase
pathways. Increased concentrations of CCL3, also known as
macrophage inflammatory protein-1α, an inflammatory chemokine, were
measured in the omentum and liver of obese patients, trafficking
pro-inflammatory cells and driving the development and progression
of oesophagogastric adenocarcinoma (25). Vielma et al (26) identified that CCL5, IL-6 and IL-10
secretions from spleen cells were stimulated by adipocytes and
additionally enhanced by premalignant lesion cell supernatant,
which provides evidence that obesity-associated inflammation, with
the inflammatory environment of premalignant cells may promote the
development and progression of cancer.
The results of the present study suggested that HFD
induced excessive adipose tissue deposition and obesity in TRAMP
mice, which additionally promoted development and progression of
PCa. Concurrently, it was identified that DU145 cells cultured in
HFD-CM, with altered adipokine and cytokine levels, increased the
proliferative, migratory and invasive abilities of DU145 cells
compared with the counterparts in CD-CM. On the basis of these
results, it is hypothesized that leptin, CCL3, CCL4, CCL5 and
CXCL10 levels, which were demonstrated to exhibit significantly
different levels of a cocktail of cytokines in obesity-related and
malignancy-associated sera between HFD- and CD-fed mice, and may
contribute together to cancer progression.
The aim of the present pilot study was to explore
the function of adipokine and cytokine alterations in
obesity-stimulated PCa progression. The application of HFD-induced
obesity and spontaneous development of PCa in TRAMP mice mimicked
the natural course of PCa in patients with obesity, and accordingly
the adipokine and cytokine alterations identified in the present
study may contribute to the reciprocal effect of adipocytes and
cancer cells on cancer progression. Further studies are warranted
to identify the specific cytokines and their associated signaling
pathways in mediating the aggressiveness of cancer cells.
Consequently, these cytokines and signaling pathways may be
targeted to reverse the negative effects of obesity on cancer
progression.
To conclude, the results of the present study
demonstrated that HFD-fed TRAMP mice presented with increased body
weight, excess fat deposition, poorer differentiation, increased
local invasion and metastasis rates of PCa. Concurrently, HFD
feeding time-dependently altered the levels of various adipokines
and cytokines in TRAMP mice, and in vitro studies indicated
that HFD-CM promoted the proliferative, migratory and invasive
abilities of prostate cancer cells. These results suggested that
the circulating adipokine and cytokine alterations in response to
excess adipose tissue deposition induced by HFD feeding contributed
to PCa progression.
Acknowledgements
The present study was sponsored by Shanghai Sailing
Program (grant no. 17YF1401700), National Natural Science
Foundation of China (grant no. 81272835), Outstanding Talents
Cultivation Project of Shanghai Medical System (grant no.
XBR2013092), Talents Development Foundation of Shanghai (grant no.
201302) and Scientific Research Innovation Project of Shanghai
Education Committee (grant no. 14ZZ010).
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Parikesit D, Mochtar CA, Umbas R and Hamid
AR: The impact of obesity towards prostate diseases. Prostate Int.
4:1–6. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hu MB, Liu SH, Jiang HW, Bai PD and Ding
Q: Obesity affects the biopsy-mediated detection of prostate
cancer, particularly high-grade prostate cancer: A dose-response
meta-analysis of 29,464 patients. PLoS One. 9:e1066772014.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hu MB, Xu H, Bai PD, Jiang HW and Ding Q:
Obesity has multifaceted impact on biochemical recurrence of
prostate cancer: A dose-response meta-analysis of 36,927 patients.
Med Oncol. 31:8292014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Calle EE, Rodriguez C, Walker-Thurmond K
and Thun MJ: Overweight, obesity and mortality from cancer in a
prospectively studied cohort of U.S. adults. N Engl J Med.
348:1625–1638. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Toren P, Mora BC and Venkateswaran V:
Diet, obesity and cancer progression: Are adipocytes the link?
Lipid Insights. 6:37–45. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pérez-Hernández AI, Catalán V,
Gómez-Ambrosi J, Rodríguez A and Frühbeck G: Mechanisms linking
excess adiposity and carcinogenesis promotion. Front Endocrinol
(Lausanne). 5:652014.PubMed/NCBI
|
|
8
|
Andò S, Barone I, Giordano C, Bonofiglio D
and Catalano S: The multifaceted mechanism of leptin signaling
within tumor microenvironment in driving breast cancer growth and
progression. Front Oncol. 4:3402014.PubMed/NCBI
|
|
9
|
Price RS, Cavazos DA, De Angel RE,
Hursting SD and deGraffenried LA: Obesity-related systemic factors
promote an invasive phenotype in prostate cancer cells. Prostate
Cancer Prostatic Dis. 15:135–143. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kato S, Abarzua-Catalan L, Trigo C,
Delpiano A, Sanhueza C, García K, Ibañez C, Hormazábal K, Diaz D,
Brañes J, et al: Leptin stimulates migration and invasion and
maintains cancer stem-like properties in ovarian cancer cells: An
explanation for poor outcomes in obese women. Oncotarget.
6:21100–21119. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vansaun MN: Molecular pathways:
Adiponectin and leptin signaling in cancer. Clin Cancer Res.
19:1926–1932. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dirat B, Bochet L, Dabek M, Daviaud D,
Dauvillier S, Majed B, Wang YY, Meulle A, Salles B, Le Gonidec S,
et al: Cancer-associated adipocytes exhibit an activated phenotype
and contribute to breast cancer invasion. Cancer Res. 71:2455–2465.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Nguewa PA and Calvo A: Use of transgenic
mice as models for prostate cancer chemoprevention. Curr Mol Med.
10:705–718. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Xu H, Hu MB, Bai PD, Zhu WH, Liu SH, Hou
JY, Xiong ZQ, Ding Q and Jiang HW: Proinflammatory cytokines in
prostate cancer development and progression promoted by high-fat
diet. Biomed Res Int. 2015:2497412015.PubMed/NCBI
|
|
15
|
Foster BA, Gingrich JR, Kwon ED, Madias C
and Greenberg NM: Characterization of prostatic epithelial cell
lines derived from transgenic adenocarcinoma of the mouse prostate
(TRAMP) model. Cancer Res. 57:3325–3330. 1997.PubMed/NCBI
|
|
16
|
Gingrich JR, Barrios RJ, Morton RA, Boyce
BF, DeMayo FJ, Finegold MJ, Angelopoulou R, Rosen JM and Greenberg
NM: Metastatic prostate cancer in a transgenic mouse. Cancer Res.
56:4096–4102. 1996.PubMed/NCBI
|
|
17
|
D'Amico AV, Whittington R, Malkowicz SB,
Schultz D, Blank K, Broderick GA, Tomaszewski JE, Renshaw AA,
Kaplan I, Beard CJ and Wein A: Biochemical outcome after radical
prostatectomy, external beam radiation therapy, or interstitial
radiation therapy for clinically localized prostate cancer. JAMA.
280:969–974. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Center MM, Jemal A, Lortet-Tieulent J,
Ward E, Ferlay J, Brawley O and Bray F: International variation in
prostate cancer incidence and mortality rates. Eur Urol.
61:1079–1092. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mavropoulos JC, Buschemeyer WC III, Tewari
AK, Rokhfeld D, Pollak M, Zhao Y, Febbo PG, Cohen P, Hwang D, Devi
G, et al: The effects of varying dietary carbohydrate and fat
content on survival in a murine LNCaP prostate cancer xenograft
model. Cancer Prev Res (Phila). 2:557–565. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cho HJ, Kwon GT, Park H, Song H, Lee KW,
Kim JI and Park JH: A high-fat diet containing lard accelerates
prostate cancer progression and reduces survival rate in mice:
Possible contribution of adipose tissue-derived cytokines.
Nutrients. 7:2539–2561. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Llaverias G, Danilo C, Wang Y, Witkiewicz
AK, Daumer K, Lisanti MP and Frank PG: A Western-type diet
accelerates tumor progression in an autochthonous mouse model of
prostate cancer. Am J Pathol. 177:3180–3191. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bonorden MJL, Grossmann ME, Ewing SA, et
al: Growth and Progression of TRAMP Prostate Tumors in Relationship
to Diet and Obesity. Prostate Cancer. 2012:543970–5440132012.
View Article : Google Scholar
|
|
23
|
Wang YY, Lehuédé C, Laurent V, Dirat B,
Dauvillier S, Bochet L, Le Gonidec S, Escourrou G, Valet P and
Muller C: Adipose tissue and breast epithelial cells: A dangerous
dynamic duo in breast cancer. Cancer Lett. 324:142–151. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ribeiro R, Monteiro C, Cunha V, Oliveira
MJ, Freitas M, Fraga A, Príncipe P, Lobato C, Lobo F, Morais A, et
al: Human periprostatic adipose tissue promotes prostate cancer
aggressiveness in vitro. J Exp Clin Cancer Res. 31:322012.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Conroy MJ, Galvin KC, Kavanagh ME, Mongan
AM, Doyle SL, Gilmartin N, O'Farrelly C, Reynolds JV and Lysaght J:
CCR1 antagonism attenuates T cell trafficking to omentum and liver
in obesity-associated cancer. Immunol Cell Biol. 94:531–537. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Vielma SA, Klein RL, Levingston CA and
Young MR: Premalignant lesions skew spleen cell responses to immune
modulation by adipocytes. Anticancer Res. 33:1809–1818.
2013.PubMed/NCBI
|