Introduction
Malignant melanoma is the most aggressive skin
cancer and has poor prognosis. Its incidence and mortality have
increased rapidly worldwide and in Asia. In China, the estimated
new cancer cases and deaths of melanoma in 2015 were 8,000 and
3,200 respectively (1). The overall
survival (OS) rate at 5 years for patients with stage IV disease is
<5% (2).
Somatic genetic aberrations have provided a
framework for developing targeted therapy in advanced cancer. One
of the most validated treatments in this area is B rapidly
accelerated fibrosarcoma (BRAF) inhibition. BRAF mutations are
present in 50% (3) of cases of
Caucasian melanoma. The most common mutation is B rapidly
accelerated fibrosarcoma (BRAFV600E) (4–6). BRAF
inhibitors, such as vemurafenib and dabrafenib, lead to tumor
regression in 60–80% of patients with melanoma (7). However, in Chinese patients with
melanoma, the incidence of somatic mutations within the BRAF genes
is 25.5% (8). Melanoma has diverse
clinicopathological characteristics, especially in BRAF mutation
frequencies in different ethnic groups and pathologic subtypes.
Tumor surgical or biopsy tissue is the standard
material used to determine the presence of somatic mutations before
the start of targeted therapy. However, mutation status is unstable
and often changes (9). It is
difficult to obtain tumor tissue from rebiopsies owing to
discomfort and high risk and cost for patients. A liquid biopsy
from blood samples, included the circulating tumor DNA (ctDNA)
obtained from cell-free DNA (cfDNA), cellular tumor cells (CTCs)
and others, overcome the invasive nature and heterogeneity
(10). The concordance between ctDNA
for BRAFV600E and BRAFV600E status in tissue
has been shown to be ~70%. CtDNA for BRAFV600E has a
sensitivity of 38–79% and a specificity of 40–100% (9,11–13). BRAFV600E mutated DNA was
detected in CTCs of 77% of melanoma patients with recorded mutated
tumor tissues (14). In colorectal
cancer, a report before showed that among 23 matched CTCs and ctDNA
samples, the concordance was 73.9% for BRAF mutations (15). But the disadvantage of CTCs is that
the repetitive rate and the success rate of PCR in CTCs were really
low in the detection of BRAF mutations (14,16). So
the higher concentrations of ctDNA, which is correlated with tumor
burden, can be a great alternative to rebiopsy. CtDNA can provide
the genetic landscape of all cancerous lesions (primary and
metastases) as well as offering the opportunity to systematically
track genomic evolution which is used for evaluating response after
treatment and monitoring disease recurrence. However, there is no
reports associated with BRAFV600E in ctDNA and Chinese
melanoma patients.
3D digital PCR (dPCR) is a third-generation dPCR
technique that minimizes the use of DNA template. It can be used
for true real-time quantitative detection and can detect mutation
frequencies as low as 0.005–0.01% (17). In the present study we use 3D dPCR to
detect ctDNA with BRAFV600E in 58 Chinese patients with
melanoma and also determine whether the levels of ctDNA with
BRAFV600E at baseline before BRAF inhibitor therapy
correlate with treatment response and survival.
Materials and methods
Patients and sample collection
Paired tissues and blood samples from 58 patients
with melanoma who were hospitalized between 2012 and 2015 in the
Renal Cancer and Melanoma Department of Beijing Cancer Hospital
were used in our study. Written consent was obtained from the
patients or patients' parent/carer. All tissue samples were
confirmed to be positive for melanoma using hematoxylin and eosin
(H&E) staining. Of all the patients, the mean age is 43 years,
ranged from 17 to 74 years. We obtained the following information
from the patients' clinical histories, as recorded by their
oncologists: disease stage, tissue mutation, Eastern Cooperative
Oncology Group (ECOG) performance status, and lactate dehydrogenase
(LDH) values. Blood samples were collected before BRAF inhibitor
treatment and tumor response was evaluated in accordance with the
Response Evaluation Criteria in Solid Tumors (RECIST version 1.1).
The latest follow-up date was October 1, 2016. The present study
was approved by the Medical Ethics Committee of the Beijing Cancer
Hospital and Institute and was conducted according to the
Declaration of Helsinki Principles.
Sample collection and cfDNA
extraction
Blood samples from patients with melanoma were
collected in ethylene diamine tetraacetic acid (EDTA) vacutainer
tubes and stored at 4°C within 24 h before isolation. After
centrifugation at 2,500 rpm for 10 min and a further centrifugation
at 1,200 rpm for 15 min, plasma samples were collected. CfDNA was
extracted from 2 ml plasma stored at −80°C using the QIAamp
Circulating Nucleic Acid kit (Qiagen China Co., Ltd.) according to
the manufacturer's protocols. CfDNA was eluted in 30 µl of Buffer
AVE (Qiagen China Co., Ltd.).
CfDNA detection using 3D digital
polymerase chain reaction (dPCR)
The TaqMan® BRAF SNP Genotyping assay
(cat. no. 4465804; Life; Thermo Fisher Scientific, Inc.) was used
for dPCR detection. Each 15 µl dPCR reaction contained 0.375 µl
BRAF assay, 7.5 µl QuantStudio™ 3D Digital PCR Master Mix 2X (Life;
Thermo Fisher Scientific, Inc.), 6.125 µl nuclease-free water
(Life, Thermo Fisher Scientific, Inc.), and 1 µl cfDNA. The dPCR
reactions were then loaded onto a QuantStudio™ 3D Digital PCR Chip
v2 (Life; Thermo Fisher Scientific, Inc.). The chips were preformed
using QuantStudio™ 3D Digital PCR System (Life; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocols. The
dPCR cycling was performed as follows: Initial denaturation at 96°C
for 10 min, followed by 39 cycles of annealing at 60°C for 2 min,
denaturation at 98°C for 30 sec, and elongation at 60°C for 2 min.
The levels of cfDNA were determined using QuantStudio™ 3D
AnalysisSuite™ Software (Life; Thermo Fisher Scientific, Inc.) by
analyzing the numbers and proportions of positive droplets.
Statistical analyses
Statistical analysis was performed using SPSS 20.0
for Windows (IBM Corp, Armonk, NY, USA). Qualitative data are
described as percentages. Quantitative data are described as means
± standard deviations. Pearson's chi-square tests, Fisher's exact
tests, and Mann-Whitney U tests were used to assess correlations
between the qualitative variables. Survival time was assessed using
disease-free survival (DFS), progression-free survival (PFS), and
OS. DFS and PFS were calculated separately as the duration from the
time of initial surgery to the diagnosis of a recurrence and the
time of targeted therapy to the diagnosis of progression. OS time
was assessed for prognostic analysis. We used Kaplan-Meier and
log-rank tests to analyze survival differences between groups.
Univariate Cox regression analyses were performed to examine
correlations between ctDNA levels and clinicopathologic measures
and PFS. These tests were bilateral and the significance level was
defined as 0.05, so that P<0.05 was considered to indicate a
statistically significant difference.
Results
Concordance between
BRAFV600E in ctDNA (3D dPCR) and in tumor tissue
(PCR)
We used the maximum concentration of
BRAFV600E in ctDNA from 4 healthy individuals
(0.097–0.133 copies/ml) to establish our criteria and divided the
58 patients with melanoma into two groups: undetectable type (UT),
and mutant type (MT). 43 (74.1%) of 58 plasma samples had the
BRAFV600E mutation, while 13 (25.9%) did not. A total of
50 tissue samples had the BRAFV600E mutation, as
assessed using PCR. The concordance and sensitivity of
BRAFV600E between ctDNA and tumor tissue were 70.2% and
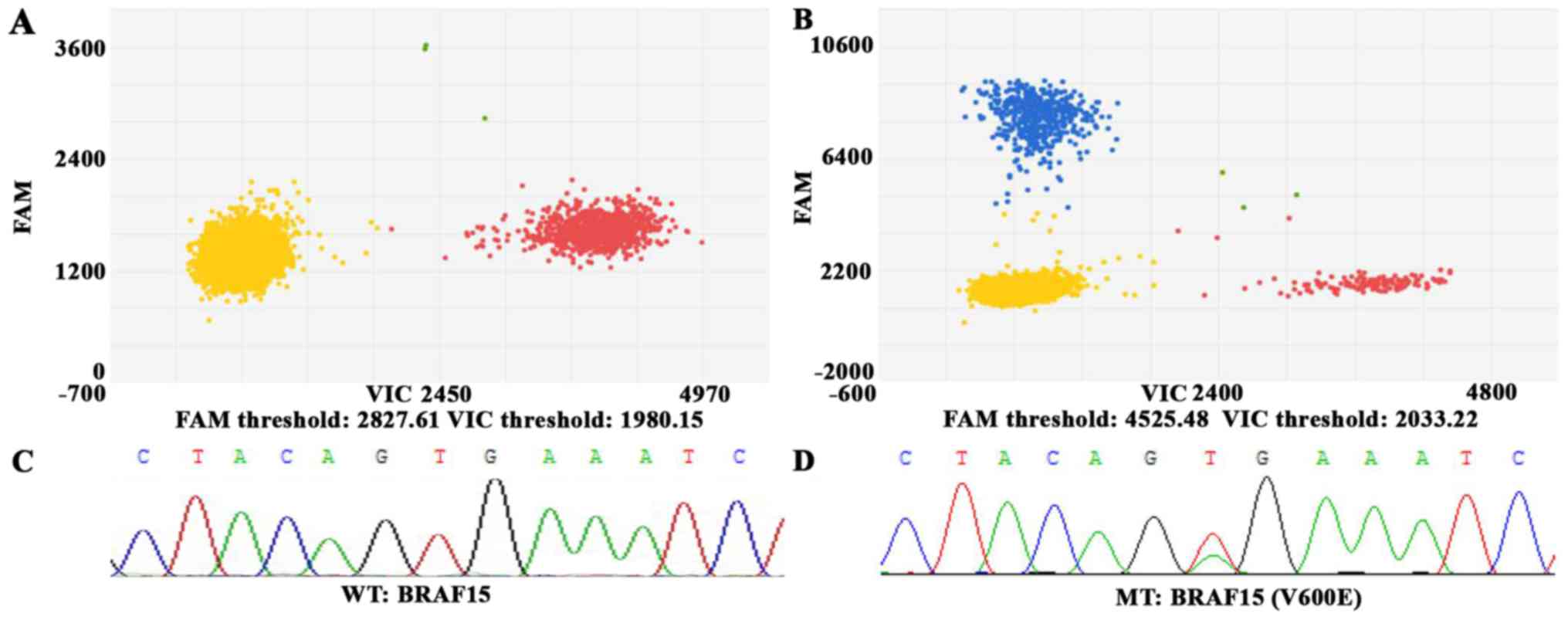
76%, respectively. The results are presented in Table I. Fluorescent amplitude plots of 3D
dPCR in ctDNA samples and Sanger Sequencing in tumor tissues from
melanoma patients are presented in Fig.
1. The plot of 3D dPCR contains the negative droplets, positive
droplets for wild-type, mutant type and both. And the plot of
Sanger Sequencing shows the location and variation of nucleotides
in BRAF gene.
 | Table I.BRAFV600E in ctDNA and
tumor tissue. |
Table I.
BRAFV600E in ctDNA and
tumor tissue.
|
| Tumor tissue |
|---|
|
|
|
|---|
| BRAFV600E
mutation | WT | MT | NA | Total |
|---|
| ctDNA |
|
|
|
|
| UT | 2 | 12 | 0 | 14 |
| MT | 5 | 38 | 1 | 44 |
|
Total | 7 | 50 | 1 | 58 |
Clinical correlations between
BRAFV600E in ctDNA and clinical characteristics of
patients
The presence of the BRAFV600E mutation in
ctDNA was correlated with LDH concentration (P=0.04) and ECOG
performance status (P=0.04). The levels of BRAFV600E
mutation in ctDNA levels were markedly higher in patients with high
LDH levels compared to those with normal LDH levels [UT: ctDNA
median, 0.08 copies/ml (0–0.094 copies/ml); LDH median, 197 IU/l
(125–269 IU/l); MT: ctDNA median, 0.37 copies/ml (0.149–456.9
copies/ml); LDH median, 206 IU/l (129–1371 IU/l)]. Worse ECOG
performance status was associated with higher BRAFV600E
mutation levels in plasma. There were no significant associations
between BRAFV600E mutation in ctDNA and other clinical
characteristics, such as sex, age, subtype, stage, thickness, or
ulcer. The details of our findings are presented in Table II.
 | Table II.Correlation between
BRAFV600E in ctDNA and characteristics of patients. |
Table II.
Correlation between
BRAFV600E in ctDNA and characteristics of patients.
|
|
BRAFV600E mutation of
ctDNA |
|---|
|
|
|
|---|
| Clinical
charateristics | UT (n=15,%) | MT (n=43,%) | P-value |
|---|
| Sex |
|
|
|
|
Male | 5 (26.3) | 14 (73.7) | 1.00 |
|
Female | 10 (25.6) | 29 (74.4) |
|
| Age, years |
|
|
|
|
>60 | 1 (14.3) | 6 (85.7) | 0.46 |
|
≤60 | 14 (27.5) | 37 (72.5) |
|
| Subtype |
|
|
|
| Acral +
Mucosal | 3 (20) | 12 (80) | 0.68 |
|
+Uveal |
|
|
|
| CSD +
non-CSD | 10 (30.3) | 23 (69.7) |
|
|
Unknown | 2 (25) | 8 (75) |
|
| Stage |
|
|
|
|
I–III | 8 (29.6) | 19 (70.4) | 0.54 |
| IV | 7 (22.6) | 24 (77.4) |
|
| Thickness, mm |
|
|
|
|
<4 | 3 (37.5) | 5 (62.5) | 0.76 |
| ≥4 | 3 (27.3) | 8 (72.7) |
|
| Ulcer |
|
|
|
|
With | 5 (29.4) | 12 (70.6) | 1.00 |
|
Without | 6 (26.1) | 17 (73.9) |
|
| LDH |
|
|
|
| <240
IU/L | 12 (36.4) | 21 (63.6) | 0.04 |
| ≥240
IU/L | 1 (6.7) | 14 (93.3) |
|
| ECOG |
|
|
|
| 0 | 12 (42.9) | 16 (57.1) | 0.04 |
| 1 | 3 (12) | 23 (88) |
|
| 2 | 0 (0) | 4 (100) |
|
Clinical correlations between
BRAFV600E in ctDNA and clinical benefits after targeted
therapy
A total of 30 out of 58 patients with melanoma were
treated with BRAF inhibitors. This accounted for 20% (6/30), 66.7%
(20/30), and 13.3% (4/30) of patients with first-, second-, and
third-line targeted therapy, respectively. The median follow-up
time was 34.9 months (range, 30.3–39.6 months). There was no
correlation between BRAFV600E in ctDNA and response,
PFS, or OS after targeted therapy with BRAF inhibitors. The subset
of patients undergoing targeted therapy included 13 patients with
partial responses (PR), 13 with stable disease (SD), and 1
progressive disease (PD), as assessed using RECIST criteria. We
also had 3 unassessable patients (Table
III). Objective response rate (ORR), PFS, and OS stratified by
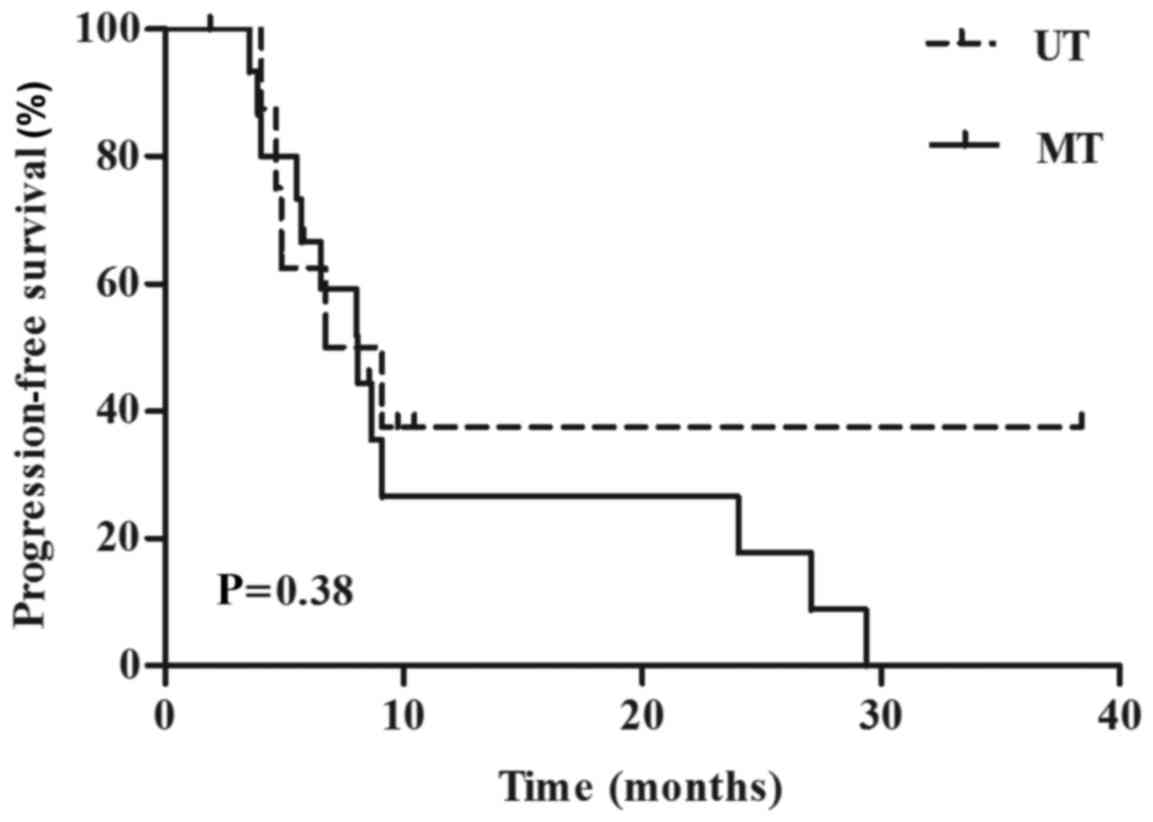
BRAFV600E in ctDNA were 30.0% vs. 56.7% (P=0.3)
(Table III), 8.1 months vs. 6.7
months (P=0.38) (Fig. 2), and 65.6
months vs. 42.3 months (P=0.52), respectively for the UT and MT
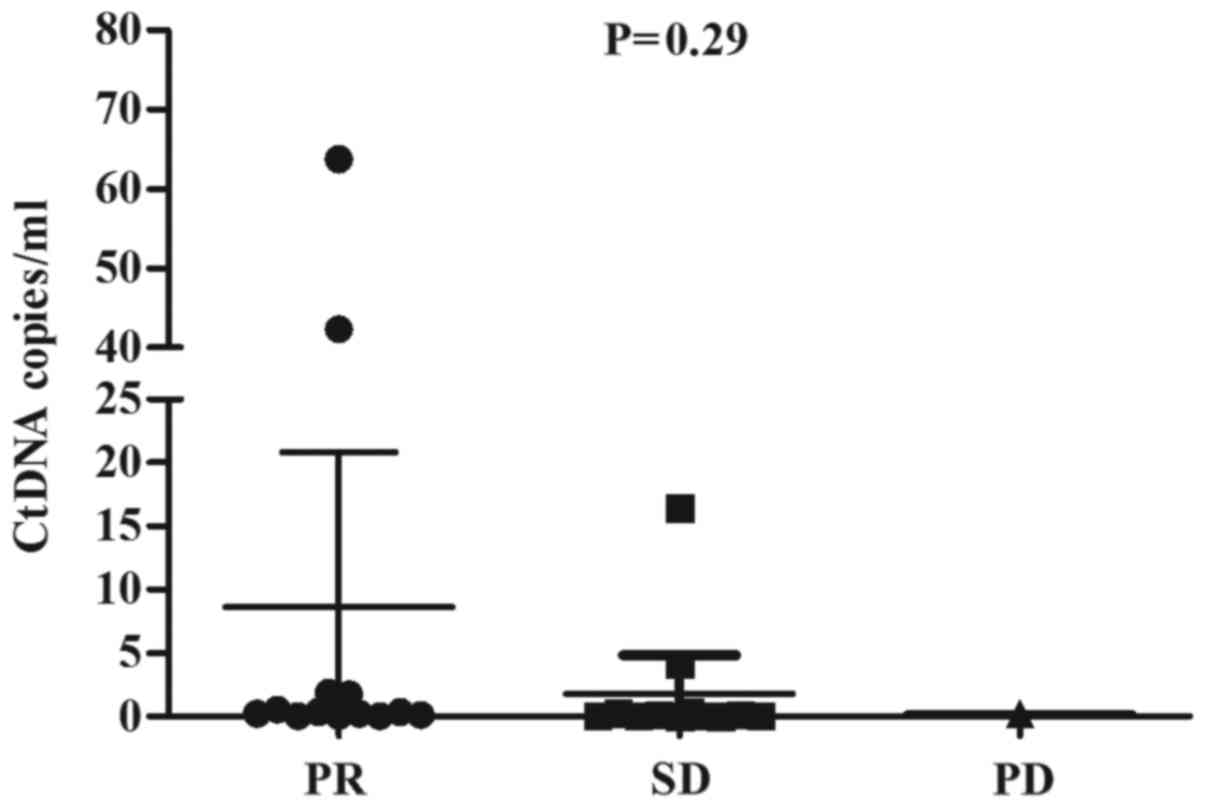
groups. Quantitative and qualitative data of response are presented
in Fig. 3 and Table III, respectively.
 | Table III.Associations between baseline ctDNA
concentrations and response to targeted treatment. |
Table III.
Associations between baseline ctDNA
concentrations and response to targeted treatment.
|
BRAFV600E mutation | PR | SD | PD | Total | P-value |
|---|
| UT | 3 | 6 | 0 | 9 | 0.30 |
| MT | 10 | 7 | 1 | 18 |
|
| Total | 13 | 13 | 1 | 27 |
|
Discussion
In the present study we used 3D dPCR to detect ctDNA
with BRAFV600E in 58 Chinese patients with melanoma and
statistically analyzed the correlations between ctDNA level and
clinical characteristics, treatment response, and survival. The aim
of our study is to know whether ctDNA is a great alternative to
biopsy and whether 3D dPCR is a suitable detection method for
ctDNA. Furthermore, we want to explore if BRAFV600E
ctDNA is a candidated biomarker in patients with melanoma or even a
predictive marker after treatment of BRAF inhibitors.
In prior reports, the concordance between
BRAFV600E in ctDNA and BRAFV600E in tissue
samples was ~70% and the sensitivity was 38–79% (9,11–13). The concordance and sensitivity between
ctDNA and tumor tissue in our study were 70.2 and 76%, which were
higher than those of prior reports. The main reason for this
observation is that our study uniquely used 3D dPCR, which is
highly sensitive, quantitative, and real-time. Unlike other
detection methods, such as Beads Emulsion Amplification Magnetics
(BEAMing), Amplification Refractory Mutation System (ARMS), and
Next Generation Sequencing (NGS) (9,11–13), each reaction of 3D dPCR has 20,000
wells and each well is isolated from its neighbors. This ensures
the high sensitivity and accuracy of 3D dPCR. In addition, 3D dPCR
is simple to perform. In fact we were able to obtain our results
within 2 h. The use of 3D dPCR prevents mistakes due to complex
operation and calculation errors. Although BRAFV600E in
ctDNA was identified by 3D dPCR, which is the most sensitive among
all of the detection tools use thus far, 29.8% of the patients had
plasmic BRAF statuses that were distinct from those of their tumor
tissues. This may have been owing to the mechanisms underlying the
transfer of ctDNA into the blood, which are as yet not completely
clear. It is also known that the levels of ctDNA vary at different
times of the day. In addition, the heterogeneity of the tumors may
have led to differences between the ctDNA and the tissue. CtDNA
derived from apoptotic or necrotic cell debris of primary tumors,
metastatic tumors, or CTCs may better reflect the entire picture of
the tumor when compared to using one site to obtain tumor tissue
(18). Finally, genetic alterations
may appear after different treatments (13). This is important, as ~80% of the
patients with melanoma in this study had not been treated with the
first-line therapy.
Our results are in agreement with those of previous
studies that have shown that BRAFV600E mutant ctDNA
correlates with tumor burden (19).
Patients with lower or undetectable amounts of BRAFV600E
mutant ctDNA tend to be those with less disease burden as measured
by LDH, RECIST sum of diameters, and ECOG performance (12). LDH is the only blood-based biomarker
incorporated into the staging system, as elevated levels of LDH are
associated with significantly decreased survival (19). Both BRAFV600E in ctDNA and
LDH in the blood are good prognostic markers and are thought to be
useful in the follow-up of patients with melanoma (20). LDH is neither sensitive nor specific,
and other studies have shown that ctDNA is more consistent and
informative than LDH (21,22). In addition, ctDNA is significantly
more accurate for tracking disease status than traditional serum
markers, such as carcinoembryonic antigen (CEA) and cancer antigen
15-3 (CA15-3), which are used to diagnose colorectal and breast
cancer, respectively (23,24). In the future, we will monitor the
dynamic status of ctDNA and compare the results thus obtained with
those of LDH in a large scale study. Here, 18 patients that had
surgery before in our study collected their blood samples during
the period between the metastatic lesions determined and the
first-line therapy. Of note, DFS stratified by BRAFV600E
in ctDNA was 26.4 months vs. 9.1 months (P=0.013) for UT and MT,
respectively. This indicates that patients with longer DFS before
recurrences tended to have lower levels of BRAFV600E
mutant ctDNA. Some experts in the breast cancer field have also
found that level of ctDNA are reduced or even disappeared after
surgery, although some patients have remaining ctDNA (25). This highlights a new direction for
ctDNA research. Specifically, it will be interesting to determine
whether ctDNA detected after surgery identifies patients at risk
for recurrence. This may then guide adjuvant therapy decisions for
individual patients.
Another objective of our study was to determine
whether BRAFV600E in ctDNA can be used as criteria to
select patients for BRAF-targeted therapy. Some previous reports
have shown that low baseline ctDNA is a good predictor of response
to treatment, longer PFS, and even OS in targeted therapy (12,13,20).
Although there was no statistical significance between
BRAFV600E in ctDNA and clinical benefit, patients with
wild-type BRAF ctDNA have longer PFS and OS than mutant type which
supports the findings of previous reports. Three factors may
explain the reason of no statistical significance. First, the major
factors influenced the effect of BRAF inhibitor is unknown. A
decrease of BRAFV600E in ctDNA indicated response to
therapy (13,26). Also the increased concentration of
mutant copies observed following disease progression (26) and the state of a secondary resistance
to the treatment may associated with effect of therapy (13). The effect of BRAF inhibitors would be
affected by many factors such as a secondary resistance, so
baseline ctDNA of BRAFV600E maybe not the best
predictive marker to target therapy, but the variation of this
mutation or other gene associated with resistance. Second, it will
take time for the PFS and OS to mature. Since only 75% of the
patients have reached their endpoints, mortality only accounts for
43.1% of the outcomes. Finally, our study was carried out on a
small scale. In fact, there was only one patient who did not have a
response after targeted therapy. Therefore, it remains to determine
the realistic relationship between clinical benefit and
BRAFV600E in ctDNA.
Due to the limitation of our study, we hope a large
scale research in the future which includes multiple plasma samples
during the treatment and progression. CtDNA from different periods
can reflect different conditions of disease and predict clinical
benefits of treatment. In addition, experts can make comparison
between ctDNA and traditional biomarkers.
In conclusion, our data confirm that 3D dPCR is
suitable for ctDNA detection and that BRAFV600E ctDNA is
a non-invasive prognostic marker in patients with melanoma.
Acknowledgements
The present study was supported by grants from
National Natural Science Foundation of China (grant no. 81672696)
and Beijing Municipal Natural Science Foundation (grant no.
7152033).
References
|
1
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chi Z, Li S, Sheng X, Si L, Cui C, Han M
and Guo J: Clinical presentation, histology, and prognoses of
malignant melanoma in ethnic Chinese: A study of 522 consecutive
cases. BMC cancer. 11:852011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cancer Genome Atlas Network, . Genomic
classification of cutaneous melanoma. Cell. 161:1681–1696. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Flaherty KT, Puzanov I, Kim KB, Ribas A,
McArthur GA, Sosman JA, O'Dwyer PJ, Lee RJ, Grippo JF, Nolop K and
Chapman PB: Inhibition of mutated, activated BRAF in metastatic
melanoma. N Engl J Med. 363:809–819. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hauschild A, Grob JJ, Demidov LV, Jouary
T, Gutzmer R, Millward M, Rutkowski P, Blank CU, Miller WH Jr,
Kaempgen E, et al: Dabrafenib in BRAF-mutated metastatic melanoma:
A multicentre, open-label, phase 3 randomised controlled trial.
Lancet. 380:358–365. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sosman JA, Kim KB, Schuchter L, Gonzalez
R, Pavlick AC, Weber JS, McArthur GA, Hutson TE, Moschos SJ,
Flaherty KT, et al: Survival in BRAF V600-mutant advanced melanoma
treated with vemurafenib. N Engl J Med. 366:707–714. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Coit DG, Thompson JA, Algazi A, Andtbacka
R, Bichakjian CK, Carson WE III, Daniels GA, DiMaio D, Fields RC,
Fleming MD, et al: NCCN guidelines insights: Melanoma, version
3.2016. J Natl Compr Canc Netw. 14:945–958. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Si L, Kong Y, Xu X, Flaherty KT, Sheng X,
Cui C, Chi Z, Li S, Mao L and Guo J: Prevalence of BRAF V600E
mutation in Chinese melanoma patients: Large scale analysis of BRAF
and NRAS mutations in a 432-case cohort. Eur J Cancer. 48:94–100.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Molina-Vila MA, de-Las-Casas CM,
Bertran-Alamillo J, Jordana-Ariza N, González-Cao M and Rosell R:
cfDNA analysis from blood in melanoma. Ann Transl Med.
3:3092015.PubMed/NCBI
|
|
10
|
Crowley E, Di Nicolantonio F, Loupakis F
and Bardelli A: Liquid biopsy: Monitoring cancer-genetics in the
blood. Nat Rev Clin Oncol. 10:472–484. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gonzalez-Cao M, Mayo-de-Las-Casas C,
Molina-Vila MA, De Mattos-Arruda L, Muñoz-Couselo E, Manzano JL,
Cortes J, Berros JP, Drozdowskyj A, Sanmamed M, et al: BRAF
mutation analysis in circulating free tumor DNA of melanoma
patients treated with BRAF inhibitors. Melanoma Res. 25:486–495.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Santiago-Walker A, Gagnon R, Mazumdar J,
Casey M, Long GV, Schadendorf D, Flaherty K, Kefford R, Hauschild
A, Hwu P, et al: Correlation of BRAF mutation status in
circulating-free DNA and tumor and association with clinical
outcome across four BRAFi and MEKi clinical trials. Clin Cancer
Res. 22:567–574. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gray ES, Rizos H, Reid AL, Boyd SC,
Pereira MR, Lo J, Tembe V, Freeman J, Lee JH, Scolyer RA, et al:
Circulating tumor DNA to monitor treatment response and detect
acquired resistance in patients with metastatic melanoma.
Oncotarget. 6:42008–42018. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Reid AL, Freeman JB, Millward M, Ziman M
and Gray ES: Detection of BRAF-V600E and V600K in melanoma
circulating tumour cells by droplet digital PCR. Clin Biochem.
48:999–1002. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sakaizawa K, Goto Y, Kiniwa Y, Uchiyama A,
Harada K, Shimada S, Saida T, Ferrone S, Takata M, Uhara H and
Okuyama R: Mutation analysis of BRAF and KIT in circulating
melanoma cells at the single cell level. Br J Cancer. 106:939–946.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kidess-Sigal E, Liu HE, Triboulet MM, Che
J, Ramani VC, Visser BC, Poultsides GA, Longacre TA, Marziali A,
Vysotskaia V, et al: Enumeration and targeted analysis of KRAS,
BRAF and PIK3CA mutations in CTCs captured by a label-free
platform: Comparison to ctDNA and tissue in metastatic colorectal
cancer. Oncotarget. 7:85349–85364. 2016.PubMed/NCBI
|
|
17
|
Jennings LJ, George D, Czech J, Yu M and
Joseph L: Detection and quantification of BCR-ABL1 fusion
transcripts by droplet digital PCR. J Mol Diagn. 16:174–179. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lawrence MS, Stojanov P, Polak P, Kryukov
GV, Cibulskis K, Sivachenko A, Carter SL, Stewart C, Mermel CH,
Roberts SA, et al: Mutational heterogeneity in cancer and the
search for new cancer-associated genes. Nature. 499:214–218. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Balch CM, Gershenwald JE, Soong SJ,
Thompson JF, Atkins MB, Byrd DR, Buzaid AC, Cochran AJ, Coit DG,
Ding S, et al: Final version of 2009 AJCC melanoma staging and
classification. J Clin Oncol. 27:6199–6206. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Knol AC, Vallée A, Herbreteau G, Nguyen
JM, Varey E, Gaultier A, Théoleyre S, Saint-Jean M, Peuvrel L,
Brocard A, et al: Clinical significance of BRAF mutation status in
circulating tumor DNA of metastatic melanoma patients at baseline.
Exp Dermatol. 25:783–788. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tsao SC, Weiss J, Hudson C, Christophi C,
Cebon J, Behren A and Dobrovic A: Monitoring response to therapy in
melanoma by quantifying circulating tumour DNA with droplet digital
PCR for BRAF and NRAS mutations. Sci Rep. 5:111982015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chang GA, Tadepalli JS, Shao Y, Zhang Y,
Weiss S, Robinson E, Spittle C, Furtado M, Shelton DN,
Karlin-Neumann G, et al: Sensitivity of plasma BRAFmutant and
NRASmutant cell-free DNA assays to detect metastatic melanoma in
patients with low RECIST scores and non-RECIST disease progression.
Mol Oncol. 10:157–165. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bettegowda C, Sausen M, Leary RJ, Kinde I,
Wang Y, Agrawal N, Bartlett BR, Wang H, Luber B, Alani RM, et al:
Detection of circulating tumor DNA in early- and late-stage human
malignancies. Sci Transl Med. 6:224ra242014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Dawson SJ, Tsui DW, Murtaza M, Biggs H,
Rueda OM, Chin SF, Dunning MJ, Gale D, Forshew T, Mahler-Araujo B,
et al: Analysis of circulating tumor DNA to monitor metastatic
breast cancer. N Engl J Med. 368:1199–1209. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Beaver JA, Jelovac D, Balukrishna S,
Cochran R, Croessmann S, Zabransky DJ, Wong HY, Toro PV, Cidado J,
Blair BG, et al: Detection of cancer DNA in plasma of patients with
early-stage breast cancer. Clin Cancer Res. 20:2643–2650. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sanmamed MF, Fernández-Landázuri S,
Rodríguez C, Zárate R, Lozano MD, Zubiri L, Perez-Gracia JL,
Martín-Algarra S and González A: Quantitative cell-free circulating
BRAFV600E mutation analysis by use of droplet digital PCR in the
follow-up of patients with melanoma being treated with BRAF
inhibitors. Clin Chem. 61:297–304. 2015. View Article : Google Scholar : PubMed/NCBI
|