Introduction
Breast cancer is one of the most common malignancies
in females, particularly in developed countries, including those of
Europe and the USA. Each year, ~1 million females will be diagnosed
with breast cancer globally (1). The
incidence of breast cancer has steadily increased over previous
years, with patients being affected at a much younger age, which
poses a serious health threat with a large global impact (2). In 2014, 232,670 females were diagnosed
with breast cancer and 40,000 fatalities resulted from it in the
USA (3). Although the incidence of
breast cancer is lower in China, it is increasing annually in urban
and economically developed areas (4).
With the development of methods for the early detection of breast
cancer and novel agents for chemotherapy, endocrinotherapy and
targeted therapy, early-stage breast cancer has been well
controlled. However, the treatment of advanced-stage breast cancer
has been less successful. The National Comprehensive Cancer Network
guidelines define locally advanced breast cancer (LABC) as
Tumor-Node-Metastasis (TNM) stage IIIA [T0-2, N2, M0; T3, N1-2,
M0], stage IIIB (T4, N0-2, M0) or IIIC (T0-4, N3, M0) (5). Surgical resection is the most effective
method for treating operable LABC (clinical stage T3, N1, M0). For
inoperable LABC, preoperative neoadjuvant chemotherapy with
anthracycline alone or combined with taxane is the standard
treatment. If patients are human epidermal growth factor receptor 2
(HER2)-positive, trastuzumab is used simultaneously. If the
patients are responsive to chemotherapy, surgical resection,
radiotherapy or endocrinotherapy may follow. For patients with
disease progression during preoperative chemotherapy, palliative
breast irradiation is advised to enhance local control. Subsequent
systemic therapy is also standard. However, if patients are not
responsive to any of these treatments, intra-arterial
interventional therapy (IAIT) may serve as a safe and effective
alternative for the management of inoperable LABC. The present
study aimed to evaluate the safety and efficacy of intra-arterial
interventional therapy (IAIT) in the treatment of LABC.
Patients and methods
Patients
The inclusion criteria of the present study were as
follows: Patients with pathologically-proven inoperable LABC who
were not suitable for mastectomy following primary treatment. The
exclusion criteria were as follows: Patients who had severe
coagulation mechanism abnormality or had severe cardiac, renal or
liver failure. A total of 7 patients were enrolled in the present
study between February 2009 and September 2016 (Table I). All patients volunteered to
participate in the program and provided written informed consent.
Overall, 1 patient did not tolerate neoadjuvant chemotherapy, 2
were partially responsive to neoadjuvant chemotherapy but were
unsuitable for mastectomy, 2 presented with disease progression, 1
exhibited stable disease, and a 90-year-old patient was in a poor
physical condition and was unable to tolerate neoadjuvant
chemotherapy. All the patients received IAIT-centered systemic
treatment. All IAIT was performed by senior doctors in the
Department of Interventional Radiology at the People's Liberation
Army Army General Hospital (Beijing, China).
 | Table I.Patient characteristics, treatments
and evaluations. |
Table I.
Patient characteristics, treatments
and evaluations.
| Case no. | Age, years | Size, cm | LN, cm | Stage | Biopsy | NCT, n | IAIT, n | Pathology | Response | PFS time, months | OS time, months |
|---|
| 1 | 83 | 15×15 | 0 | cT4N0M0 | Neuroendocrine
carcinoma | 0 | 3 | Neuroendocrine
carcinoma | Partial | 88 | Alive |
|
|
|
|
|
| ER(weakly+),
PR(weakly+), HER2(−) |
|
| LN(1/18), ER(+; 90%),
PR(+; 90%), HER2(−), Ki-67(+; 5%) |
|
|
|
| 2 | 90 | 16×15 | 2 | cT4N1M0 | Invasive ductal
carcinoma | 1 | 4 | Invasive ductal
carcinoma | Partial | 8 | 8 |
|
|
|
|
|
| ER(−), PR(−),
HER2(−), Ki-67(+; 70%) |
|
| LN(17/17), ER(−),
PR(−), HER2(−), Ki-67(+; 30–60%) |
| (succumbed to other
causes) |
|
| 3 | 46 | 20×20 | 5 | cT4N2M0 | Invasive
carcinoma | 5 | 2 | Invasive
carcinoma | Partial | 26 | Alive |
|
|
|
|
|
| ER(+; 90%), PR(+;
3–5%), HER2(−), Ki-67(+; 40%) |
|
| LN(8/8), ER(+; 90%),
PR(+; 5–10%), HER2(−), Ki-67(+; 10%) |
|
|
|
| 4 | 59 | 13×10 | 1 | cT4N1M0 | Invasive ductal
carcinoma | 7 | 3 | Invasive ductal
carcinoma | Partial | 6 | 9 |
|
|
|
|
|
| ER(−), PR(−),
HER2(−), Ki-67(+; 85%) |
|
| LN(1/19), ER(−),
PR(−), HER2(−), Ki-67(+; 50%) |
|
|
|
| 5 | 42 | 6×6 | 0 | cT4N0M0 | Invasive
micropapillary carcinoma unclear | 2 | 2 | Invasive
micropapillary carcinoma | Partial | 35 | Alive |
|
|
|
|
|
|
|
|
| LN(4/10), ER(−),
PR(−), HER2(−), Ki-67(+; 70%) |
|
|
|
|
| 6 | 34 | 10×9 | 1 | cT4N1M0 | Invasive ductal
carcinoma | 5 | 1 | Invasive ductal
carcinoma | SD | 9 | 11 |
|
|
|
|
|
| ER(−), PR(−),
HER2(−), Ki-67(+; 40%) |
|
| LN(0/21), ER(−),
PR(−), HER2(−), Ki-67(+; 40%) |
|
|
|
| 7 | 59 | 8×6 | 2 | cT4N2M0 | Invasive
carcinomaunclear | 4 | 1 | Invasive lobular
carcinoma | Partial | 15 | Alive |
|
|
|
|
|
|
|
|
| LN(12/17), ER(+;
85%), PR(+; 1%), HER2(−), Ki-67(+; 5%) |
|
|
|
IAIT
All 7 patients were cannulated via the femoral
artery ipsilateral to the lesion. Subsequently, a 4F fixed-curve
catheter (Cobra catheter; Cook Medical, Inc., Bloomington, IN, USA)
was advanced into the internal mammary artery ipsilateral to the
lesion and into the subclavian artery. Digital subtraction
angiography (DSA) was performed to determine the blood supply to
the tumor. IAIT with or without embolization was subsequently
administered to the patients every 3 weeks on average. The regimens
for IAIT were different. During the early years our breast surgeons
posed the idea of IAIT, while the IAIT was performed by doctors in
the interventional radiology department in People's Liberation Army
Army General Hospital. So what regimen should be used was almost
decided by them. And the regimen was the most often used for solid
tumors in their practice, which proved effective. In recent years,
with the deep understanding and treatment of breast cancer,
especially with the worldwide accept of NCCN guidelines,
anthracycline combined with or without taxane become the standard
treatment. So now the doctors in the interventional radiology
department accept our advice to use anthracycline and taxane based
regimens for IAIT.
Due to space limitations, only four representative
cases were listed below.
Case 1
An 83-year-old woman visited the Department of
Breast Surgery, People's Liberation Army Army General Hospital in
February 2009 due to a tumor with a walnut-like appearance on the
left breast that had become apparent 6 years previously. The tumor
was not painful and therefore the patient had not consulted a
doctor or undergone any prior treatment. After 4.5 years, the tumor
had grown larger and became ulcerated with bleeding. The patient
attended a local hospital in order to attenuate the bleeding, but
no other treatments were provided. By the time the patient attended
the People's Liberation Army Army General Hospital, the tumor was
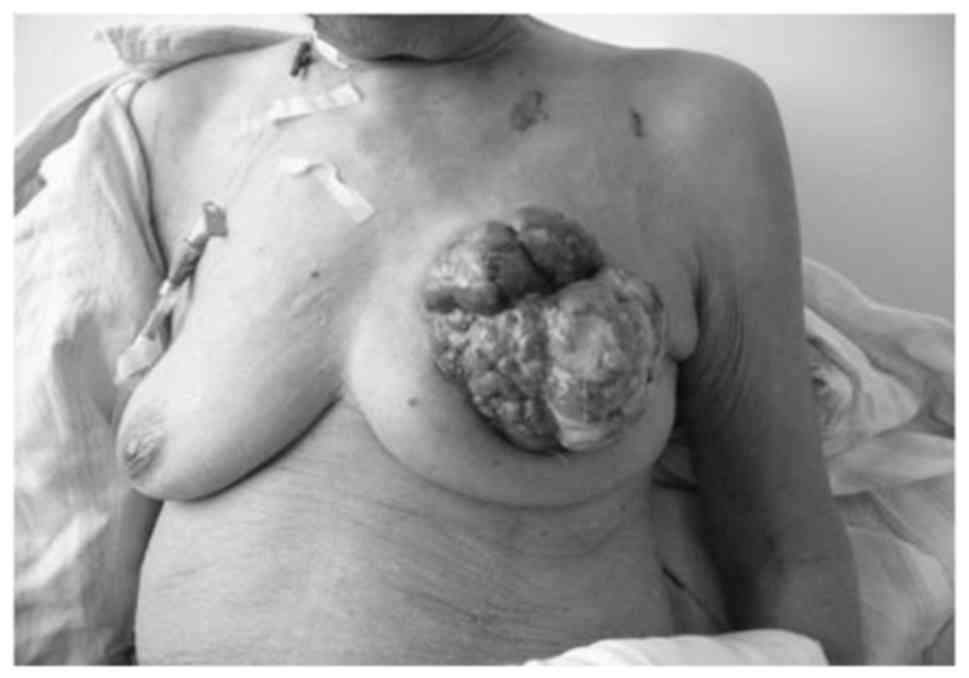
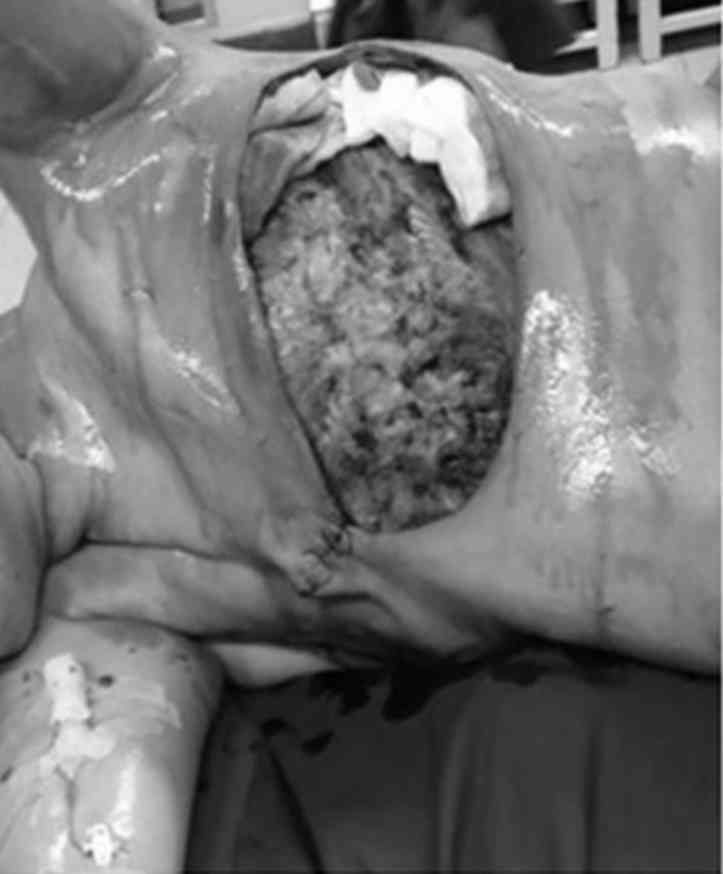
15×15 cm in diameter with a cauliflower-like appearance (Fig. 1). Skin ulceration, necrosis, bleeding
and effusion were observed, along with an unpleasant odor. The
tumor was fixed to the chest wall and was not movable. The patient
presented with severe anemia and heart failure, with a hemoglobin
level of 32 g/l (normal range: 110–150 g/l) and a hematocrit of
0.11 (normal range: 0.37–0.47). Anemia and heart failure were
corrected through blood transfusion, wound dressing was conducted
daily in order to improve the local condition and antibiotics were
administered in order to control the lung infection. Biopsy
revealed a neuroendocrine carcinoma and immunohistochemistry
revealed the following: weakly positive estrogen receptor (ER) and
progesterone receptor (PR) expression, and HER2-negative. The
patient was administered IAIT (30 mg pirarubicin, 30 mg
hydroxycamptothecin and 250 mg fluorouracil) via the left internal
thoracic artery and the subscapular artery. Simultaneous
intra-arterial embolization with selective absorbable gelatin
sponge particles was conducted. Following the first cycle of IAIT,
the tumor decreased to a diameter of 8×7 cm in diameter. During the
second and third cycles of IAIT, 20 mg pirarubicin, 15 mg
hydroxycamptothecin and 500 mg fluorouracil were administered.
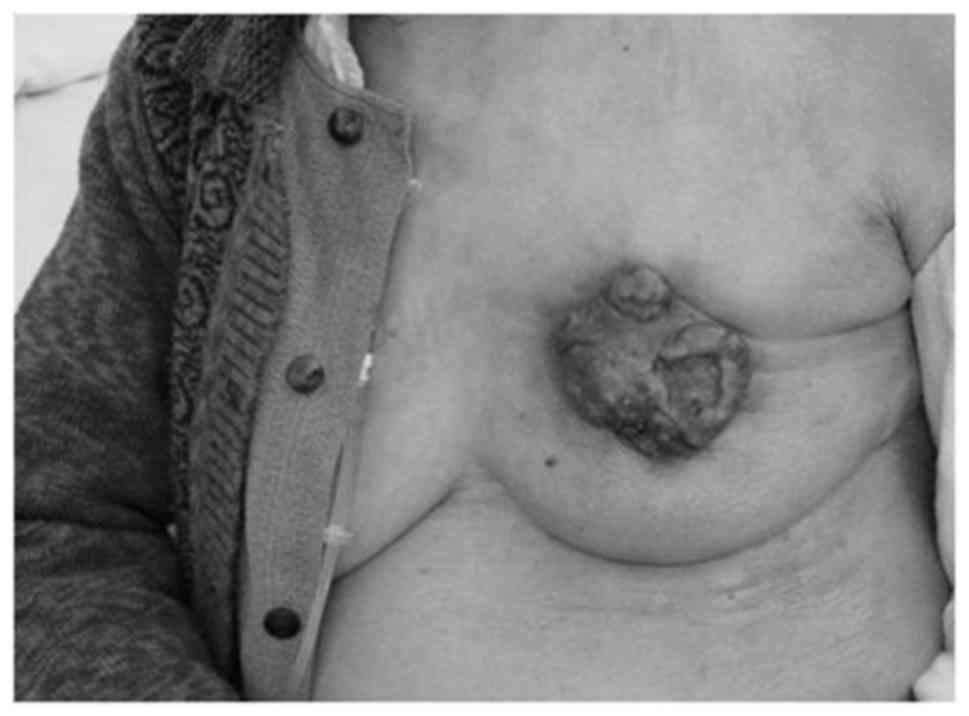
Following 3 cycles of IAIT, the tumor measured 5×4 cm in diameter
(Fig. 2). In May 2009, the patient
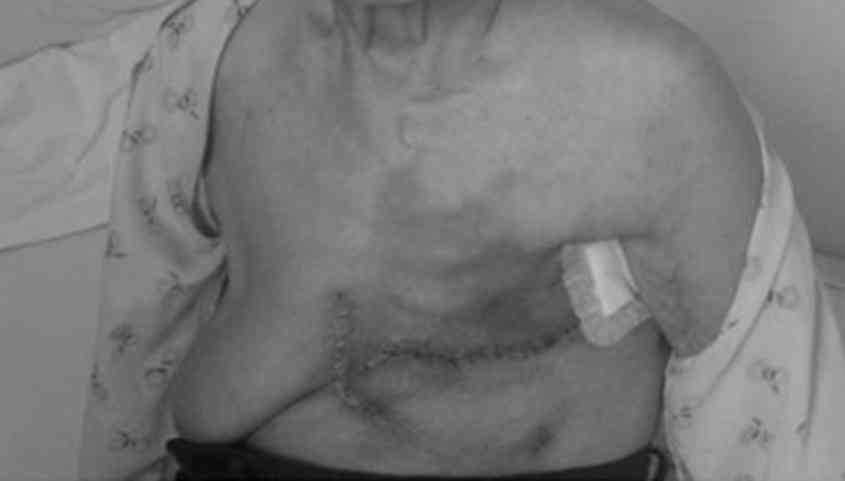
underwent a modified radical mastectomy. Pathology identified
neuroendocrine carcinoma and carcinoma emboli in the vessels. One
axillary lymph node exhibited invasion by the carcinoma (1/18).
Immunohistochemistry revealed that the tumor was ER-positive (90%),
PR-positive (90%), HER2-negative and Ki-67-positive (5%). The
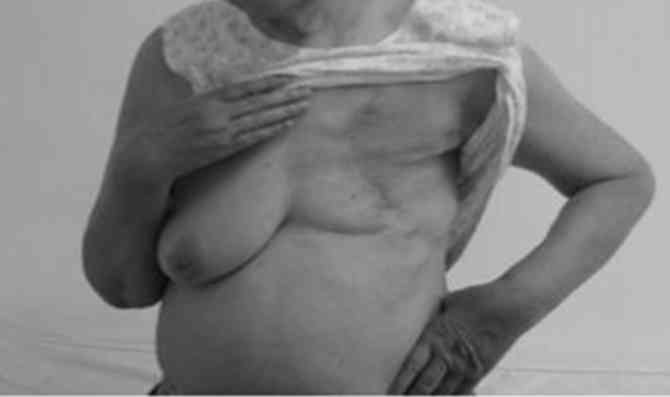
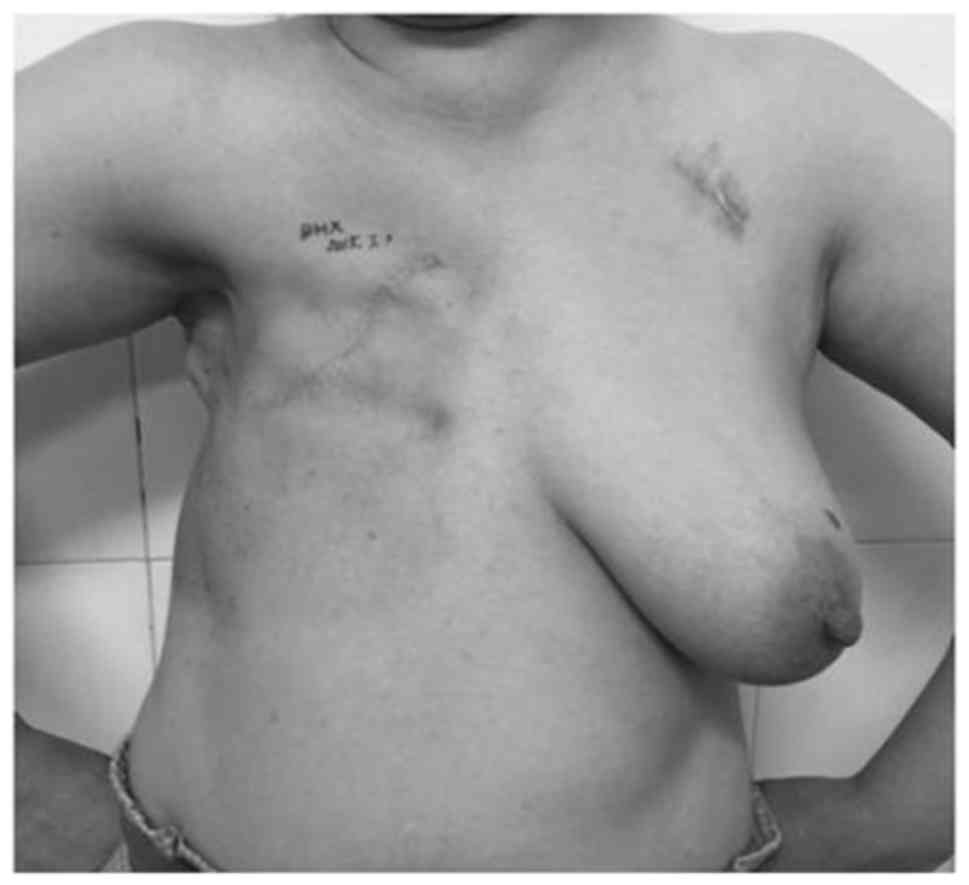
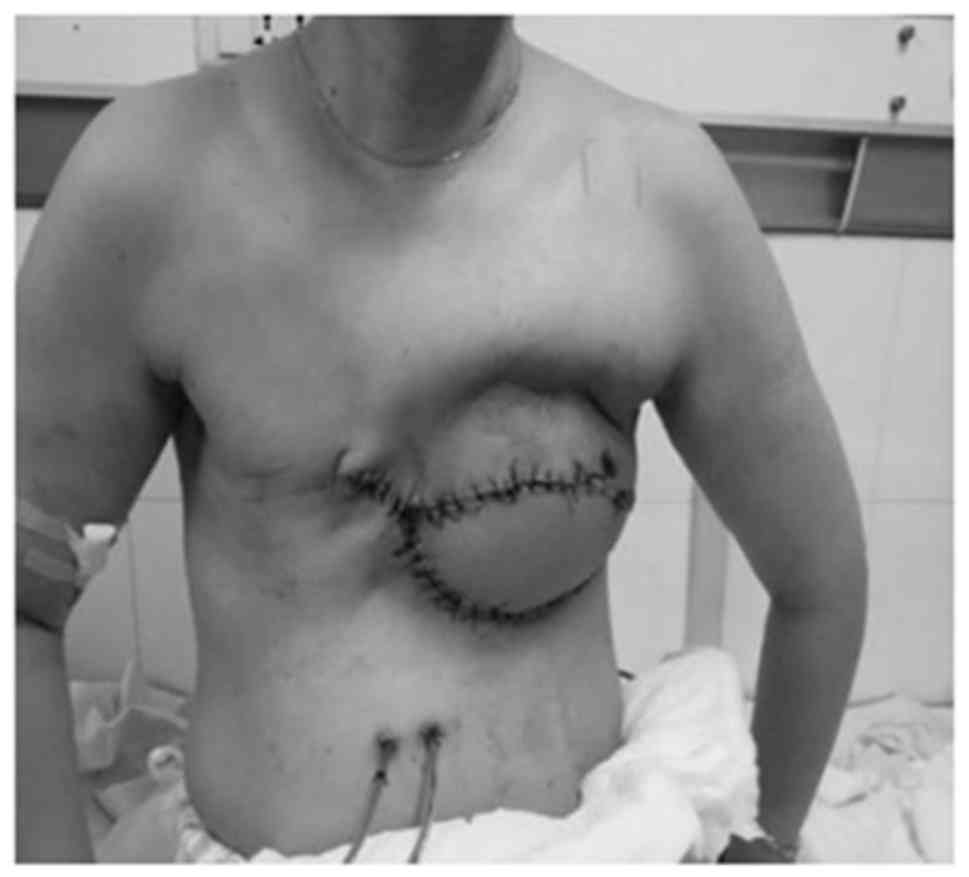
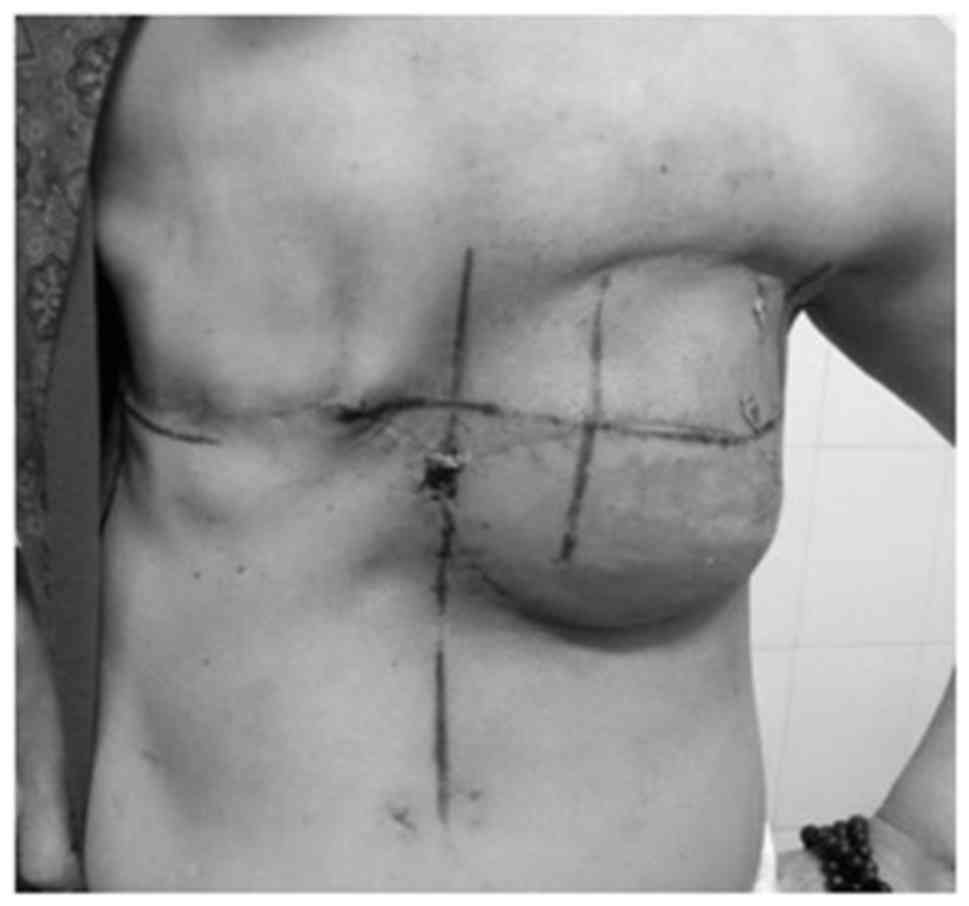
patient recovered from the surgery without complications (Fig. 3) and accepted endocrinotherapy
(exemestane 25 mg once daily) for 6 months afterwards, but refused
any subsequent treatment. The patient is now 90 years old and
remains healthy.
Case 2
A 90-year-old woman attended the Department of
Breast Surgery, People's Liberation Army Army General Hospital in
January 2013. In April 2012, the patient found a 3-cm tumor of the
left breast. The tumor was hard and painful when palpated. The
patient was diagnosed with breast cancer at another hospital
clinically without a biopsy being performed, and due to an advanced
age, did not receive any treatment. The tumor grew to ~6 cm in
diameter and the surrounding skin became red, and the patient
subsequently attended the same hospital again in December 2012. A
tumor biopsy revealed invasive ductal carcinoma and
immunohistochemistry revealed that the tumor was ER-negative,
PR-negative, HER2-negative and Ki-67-positive (70%). Administration
of paclitaxel liposome neoadjuvant chemotherapy (90 mg on days 1, 8
and 15) was planned, however, due to serious side effects,
including nausea, vomiting and fever, the patient did not complete
the first cycle. When the patient attended the People's Liberation
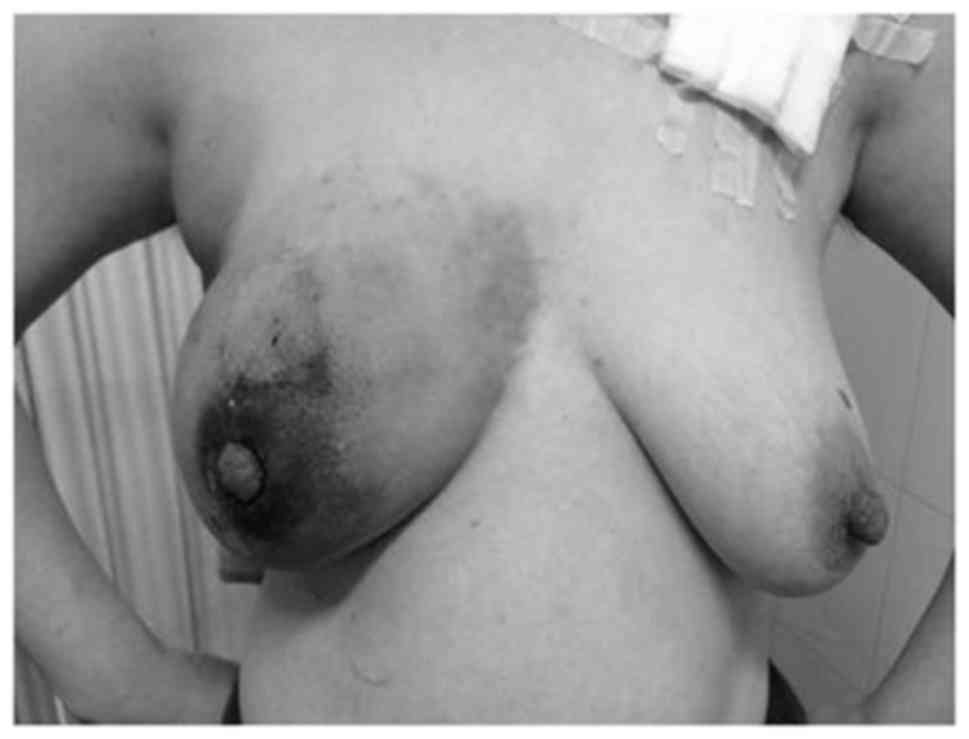
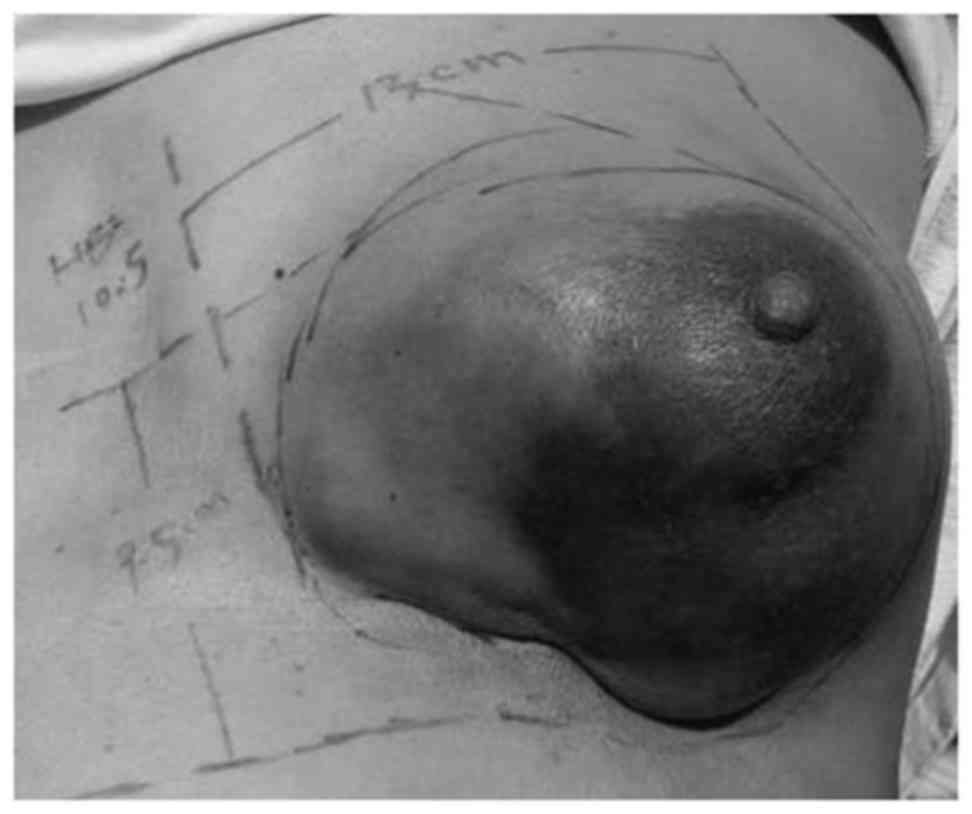
Army General Hospital, the tumor was 6 cm in diameter with a 16×15
cm are of surrounding red skin (Fig.
4). The tumor was hard and stiff without a distinct boundary,
and it was fixed to the chest wall and immovable. The patient was
administered IAIT (30 mg pirarubicin and 120 mg paclitaxel) twice
via the left internal thoracic artery and the subscapular artery.
Subsequently, 2 cycles of IAIT with 40 mg epirubicin and 150 mg
paclitaxel, and intra-arterial embolization with selective
absorbable gelatin sponge particles, was performed. Following 4
cycles of IAIT, the tumor diameter decreased to ~4 cm and the skin
returned to an almost normal color. The tumor became soft and
movable with a clear boundary. The patient tolerated IAIT well and
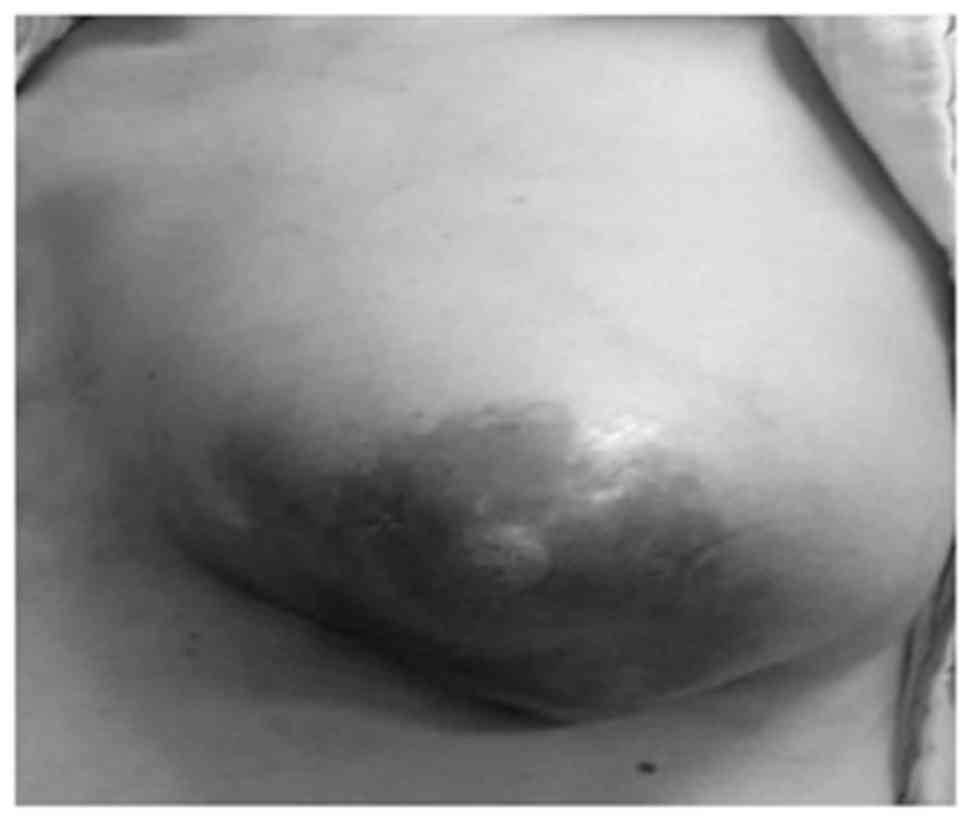
no serious side effects were observed. In May 2013, a modified
radical mastectomy was performed. Pathology revealed aninvasive
ductal carcinoma that was ER-negative, PR-negative, HER2-negative
and Ki-67-positive (30–60%). Carcinoma emboli were observed in the
vessels. The axillary lymph nodes (10/10) and the subclavian lymph
nodes (7/7) exhibited invasion by the carcinoma. The patient
recovered normally without complications (Fig. 5), but succumbed to another disease in
September 2013.
Case 3
A 46-year-old woman visited the Department of Breast
Surgery, People's Liberation Army Army General Hospital in March
2014. In 2012, the patient suffered an injury to the right breast
and subsequently noticed that there was a 4-cm lump above the
nipple. In February 2013, the patient attempted to reduce the size
of the lump by covering it with Chinese herbs, but itcontinued to
grow. The patient subsequently attempted treatment with traditional
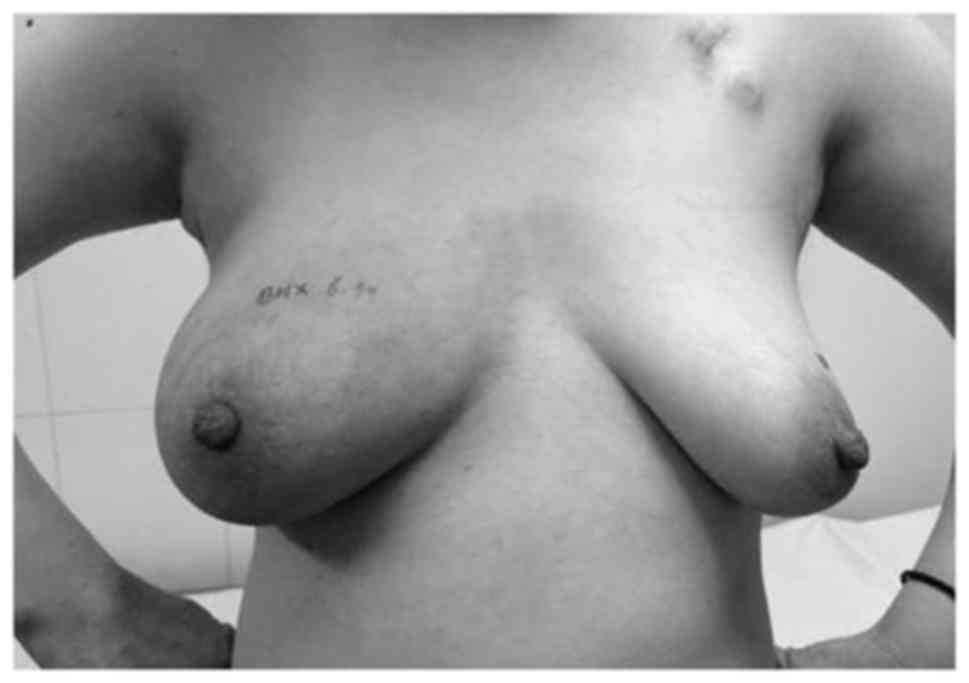
Chinese medicines, but the lump continued to grow rapidly to a
diameter of 20×20 cm and the skin became darker (Fig. 6). Upon examination, a 5-cm axillary
lymph node was observed and biopsy revealed an invasive carcinoma
that was ER-positive (90%), PR-positive (3–5%), Ki-67-positive
(40%) and HER2-negative. The patient received 5 cycles of a
doxorubicin, epirubicin and cyclophosphamide neoadjuvant
chemotherapy regimen and the tumor became softer, with a decreased
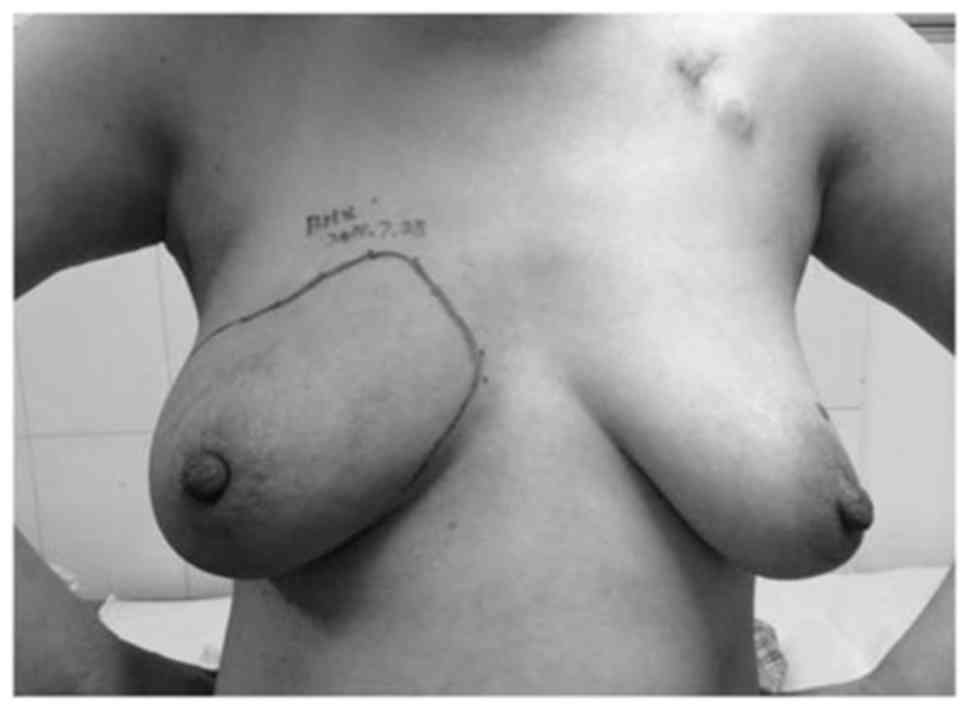
diameter of 10×10 cm, without any palpable axillary lymph nodes.
The previously darkened skin became lighter, began to peel and
eventually returned to an almost normal color (Fig. 7). IAIT with 40 mg pirarubicin, 30 mg
hydroxycamptothecin and 1,000 mg fluorouracil was administered via
the left internal thoracic artery. IAIT and embolization were
conducted simultaneously during the second treatment cycle and the
tumor continued to become softer and smaller (Fig. 8). In August 2014, the patient
underwent a modified radical mastectomy and partial
pectoralis-major removal. Residual invasive carcinoma was observed
in the pathological sample and 8/8 axillary lymph nodes exhibited
invasion. Immunohistochemistry revealed that the tumor was
ER-positive (90%), PR-positive (5–10%), Ki-67-positive (10%) and
HER2-negative. The patient recovered normally without any
complications. Subsequently, 6 cycles of gemcitabine (1,500 mg on
days 1,8) and capecitabine (2,000 mg twice daily on days 1–14) in
combination with goserelin (3.6 mg once per month) were
administered, and radiotherapy (50 Gy/5 weeks) followed and then
endocrinotherapy (exemestane 25 mg once daily) are ongoing
(Fig. 9).
Case 4
A 58-year-old woman visited the Department of Breast
Surgery, People's Liberation Army Army General Hospital in
September 2014. In September 2003, the patient had been diagnosed
with right invasive breast cancer and underwent a modified radical
mastectomy. Immunohistochemistry revealed that the tumor was
ER-negative, PR-negative, Her-2-negative and Ki-67-positive (40%),
and no lymph nodes exhibited invasion (0/25). The patient received
one cycle of pirubicin (6 mg on days 1,8) and fluorouracil (750 mg
on days 1,8), three cycles of cyclophosphamide (800 mg on days
1,8), epirubicin (90 mg on day 1) and fluorouracil (750 mg on days
1,8) and one cycle of epirubicin (90 mg on day 1) and paclitaxel
(270 mg on day 1) chemotherapy. In 2010, the patient found a tumor
in the left breast, but did not obtain a diagnosis or receive any
treatment. In 2013, the tumor grew larger, red, swollen, painful
and ulcerated. Biopsy revealed a diagnosis of invasive ductal
carcinoma and immunohistochemistry revealed that the tumor was
ER-negative, ERβ-positive (70%), Her-2-negative and Ki-67-positive
(85%). Since December 2013, the patient underwent 1 cycle of
capecitabine (1,500 mg twice daily on days 1–14) followed by 2
cycles of capecitabine (1,500 mg twice daily on days 1–14) and
docetaxel (120 mg on day 1), but the tumor grew larger. Following
one cycle of adriamycin liposome (60 mg on day 1), two cycles of
adriamycin liposome (40 mg on day 1) and cyclophosphamide (1,000 mg
on day 1), one and a half of gemcitabine (1,500 mg on days 1,8) and
carboplatin (600 mg on day 1) chemotherapy the tumor continued to
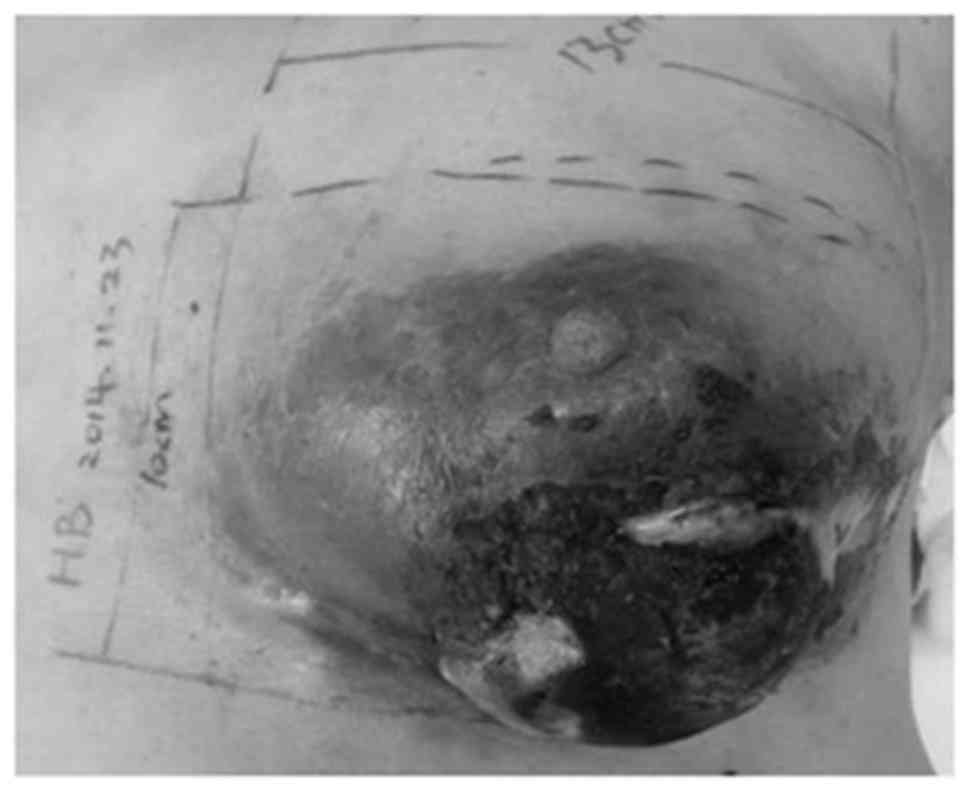
grow. When the patient visited the Department of Breast Surgery,
People's Liberation Army Army General Hospital, the tumor was
13×9.5 cm in diameter (Fig. 10).
Biopsy revealed a diagnosis of invasive ductal carcinoma and
immunohistochemistry demonstrated that the tumor was ER-negative,
PR-negative, Ki-67-positive (85%) and HER2-negative. On September
23, 2014, DSA demonstrated that the blood supply to the tumor was
from the left internal thoracic artery and a branch of the left
axillary artery. Subsequently, 40 mg lobaplatin, 30 mg
hydroxycamptothecin and 40 mg epirubicin was administered via the
left internal thoracic artery and a branch of the left axillary
artery. The ratio of medication administered via the two vessels
was 4:1. Intra-arterial embolization with 1.5-mm selective
absorbable gelatin sponge particles was conducted via the left
internal thoracic artery. Following the first cycle of IAIT, the
tumor became slightly softer and smaller (Fig. 11). For the second cycle of IAIT, the
treatment was composed of 150 mg etoposide and 40 mg navelbine. A
total of 80% of the medication was administered via the left
internal thoracic artery, while the remaining 20% was administered
via a branch of the left axillary artery. Intra-arterial
embolization was also performed and the tumor continued to become
softer and smaller. For the third cycle of IAIT, 135 mg etoposide,
30 mg navelbine and 30 mg epirubicin was administered via the left
internal thoracic artery and the left external thoracic artery.
Following the third cycle of IAIT, the lower half of the tumor
became necrotic and fell off (Fig.
12). On September 25 2014, the patient underwent a modified
radical mastectomy with removal of a portion of the
pectoralis-major (Fig. 13). Pedicled
latissimusdorsi was harvested to repair a defect of the chest wall.
The patient recovered normally one week later (Fig. 14). Pathology revealed a grade III
invasive ductal carcinoma that was ER-negative, PR-negative,
Ki-67-positive (50%) and HER2-negative. One axillary lymph node
(1/15) and no subclavian lymph nodes (0/4) were invaded by the
carcinoma. Following 1 cycle of adjuvant chemotherapy with
etoposide (140 mg on days 1 to 5) and navelbine (50 mg on days
1,8), a gynecological tumor was identified and the uterus and
bilateral uterine adnexa were removed on January 13, 2015.
Pathology revealed serous carcinoma of the right fallopian tube.
Local radiotherapy (50 Gy/5 weeks) was performed between January 29
and March 6, 2015. The radiation range covered the chest wall,
subclavicular and supraclavicular area (Fig. 15). Another cycle of adjuvant
chemotherapy with etoposide (120 mg on days 1 to 5) and navelbine
(40 mg on days 1,8) was completed in April 2015. Dermis and pleural
fluid invasion of the carcinoma was identified in May, but despite
treatment with paclitaxel liposome, the patient succumbed to
gynecological tumor in June 2015.
Patient evaluation
Liver, kidney, bone marrow and cardiac function was
assessed prior to and following IAIT. Clinical observations
included the size, color and mobility of the tumor 3 weeks after
IAIT. If the size of the tumor decreased notably and the involved
skin and muscle improved markedly, the patients underwent surgical
resection. Otherwise, the patients underwent another cycle of IAIT.
Efficacy of the IAIT was assessed using the UICC criteria (6). A complete response (CR) was defined as
the complete disappearance of all clinical evidence of the tumor
and enlarged lymph nodes by clinical evaluation, mammogram and
ultrasound. A partial response was defined as a ≥50% decrease in
the two longest perpendicular diameters of the tumor mass and local
lymph nodes. Mild response was defined as a ≤50% decrease in the
two longest perpendicular diameters of the tumor mass and local
lymph nodes. Stable disease (SD) was defined as no change in the
two longest perpendicular diameters of the tumor mass and local
lymph nodes. Progressive disease (PD) was defined as the appearance
of any new lesions not previously identified or an increase in the
two longest perpendicular diameters of the tumor mass and local
lymph nodes.
Results
All 7 patients were female and aged between 34 and
90 (mean age, 59). The largest observed lesion was 20 cm in
diameter. Overall, 5 patients exhibited palpable lymph nodes and 6
had received ≥1 cycle of neoadjuvant chemotherapy. None of the
patients exhibited any distant metastatic disease, but mild
myelosuppression occurred in 2 patients. No clear side effects,
including cardiac failure and normal skin necrosis, were observed
at any point. PR occurred in 6 patients and SD occurred in 1
patient. All the patients underwent surgical resection following
IAIT. A total of 4 of the patients were diagnosed with
triple-negative breast cancer. Progression-free survival (PFS) time
ranged between 6 and 88 months. In total, 1of the patients
succumbed as a result of another disease 8 months after receiving
IAIT, another succumbed from carcinoma of the right fallopian tube
and multiple organ metastases 9 months after IAIT and another
survived for 11 months and died of heart disease. The other 4
patients remain alive and no local recurrence or metastasis has
been observed.
Discussion
Breast cancer is the most frequently occurring type
of cancer among females, the leading cause of cancer-associated
mortality and one of the main causes of morbidity and mortality for
females worldwide (7). LABC includes
large breast tumors, tumors involving the skin over the breast or
the underlying chest wall muscles, and breast cancer with extensive
local lymph node involvement. LABC also includes inflammatory
breast cancer, a rapidly proliferating cancer type that results in
a red and swollen breast appearance. LABC accounts for 5–15% of new
breast cancer cases in the USA and for 40–60% of new cases in
non-industrialized countries (8).
LABC is generally treated with combined-modality therapy that
includes systemic chemotherapy, surgery, radiotherapy,
endocrinotherapy and targeted therapy (9).
Neoadjuvant chemotherapy is of paramount importance
in the treatment of breast cancer and is considered to reduce tumor
size, thereby rendering breast-conserving surgery possible in LABC
(10–12). Anthracycline combined with taxane is
the standard treatment for LABC and 84% of the previously
inoperable cases may become resectable upon its use (13). A pathological CR (PCR) is easy to
obtain in early-stage breast cancer patients. However, in LABC,
only a small proportion of patients may achieve PCR due to the
large tumor size (14). Additionally,
re-administration of anthracycline to patients with
anthracycline-pretreated breast cancer may be associated with
reduced efficacy and significant cardiotoxicity (15), making LABC treatment a challenge to
clinical physicians and surgeons. The present study indicates that
IAIT is an option for the management of older patients with LABC,
particularly those with cardiac dysfunction. In the present study,
the eldest patient was 90 years old and was unable to tolerate even
1 cycle of paclitaxel liposome neoadjuvant chemotherapy. However,
the patientwas able to complete four cycles of IAIT without any
observable side effects.
The breast is a solid organ with a definite blood
supply from the internal thoracic artery, lateral thoracic artery,
intercostal arteries and subscapular artery (16). IAIT is not a standard therapy, but is
occasionally performed with the aim of downstaging or for local
control. There are a number of studies regarding the use of IAIT
(17–20). Shimamoto et al (20) reported that the local response rate of
redistributed subclavian arterial infusion chemotherapy for LABC
was 77.3%, and that the response rate was higher (83.3%) if the
first treatment was infusion chemotherapy. In the present study,
the response rate was 85.7% (6/7). IAIT provides a higher plasma
concentration, better clinical results and fewer side effects than
traditional systemic chemotherapy (21,22). A
previous study demonstrated that IAIT is effective and safe for the
treatment of locally advanced breast cancer even in elderly
patients (19). In the present study,
none of the patients experienced any serious side effects,
including normal skin necrosis, liver, kidney or cardiac failure,
and all of the patients completed IAIT.
One study observed that, compared with that of
early-stage breast cancer, the overall survival (OS) rate of LABC
was lower, and the risk of local recurrence and metastasis was
markedly higher, with a 10-year survival rate of ~30% (23). Hoehne et al (24) reported that the 5-year OS rate for
stage IIIC LABC was 71.9%. In China, the 5-year OS rate for stage
IIB and III LABC was reported as 71.3% (25). In the present study, the OS data are
not complete as a number of patients remain alive, but the current
longest OS time is 88 months.
Endocrinotherapy is also an effective treatment for
LABC, particularly for patients with the lumina A subtype. However,
if the tumor burden is heavy (diameter >10 cm), good control may
not be achieved in a short time period. If the tumor is not
responsive to endocrinotherapy and metastasizes to other organs,
the outcome is generally poor. The principle of the treatment of
LABC is to rapidly control and decrease the tumor size, reduce the
tumor burden, gain opportunity for surgical resection, decrease
drug resistance and increase therapeutic efficacy. For large lumina
A LABC, IAIT may be used to rapidly downstage and decrease tumor
burden. In the present study, case 1 is a good example of such
large lumina A patient, as this patient needs to rapidly reduce the
tumor size. The patient was 83 years old with severe anemia and
heart failure. Chemotherapy and surgical resection was not suitable
due to the size of the tumor and the poor physical condition of the
patient. As a result of IAIT, the tumor size was rapidly decreased,
thereby enabling surgical resection. The PFS of this patient is
currently 88 months and the patient is now 90 years old. For older
patients with large tumors who are not suitable for neoadjuvant
chemotherapy, local IAIT with or without embolization is a safe and
effective method without any clear side effects.
Timing is extremely important in the management of
LABC. Due to the fact that a number of patients experience
bleeding, anemia and hypoproteinemia, there may not be much
opportunity to attempt alternative neoadjuvant chemotherapy
regimens. Particularly with regard to precision medicine, the
manner in which to precisely identify target patients and make good
treatment choices has become a challenge. Improved understanding of
tumor subtypes and genetic tests may inform clinicians that certain
patients may not respond to neoadjuvant chemotherapy, thereby
indicating the use of IAIT to control the disease.
The present study has a number of limitations. To
begin with, there were not enough cases for statistical analysis to
be possible. Additionally, the present study was retrospective and
therefore was not strictly a case-control study. Furthermore, the
drugs used for IAIT were not uniform due to differences in tumor
subtypes. However, all the tumors were large and the clinical
effects were clear, thereby permitting the conclusion that IAIT is
a safe and effective approach for the treatment of LABC.
References
|
1
|
Igene H: Global health inequalities and
breast cancer: An impending public health problem for developing
countries. Breast J. 14:428–434. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Veronesi U, Boyle P, Goldhirsch A,
Orecchia R and Viale G: Breast cancer. Lancet. 365:1727–1741. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ziegler RG, Anderson WF and Gail MH:
Increasing breast cancer incidence in China: The numbers add up. J
Natl Cancer Inst. 100:1339–1341. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gradishar WJ, Anderson BO, Balassanian R,
Blair SL, Burstein HJ, Cyr A, Elias AD, Farrar WB, Forero A,
Giordano SH, et al: Breast Cancer Version 2. 2015. J Natl Compr
Canc Netw. 13:448–475. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Miller AB, Hoogstraten B, Staquet M and
Winkler A: Reporting results of cancer treatment. Cancer.
47:207–214. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Toriola AT and Colditz GA: Trends in
breast cancer incidence and mortality in the United States:
Implications of prevention. Breast Cancer Res Treat. 138:665–673.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Winer EP, Mrrow M, Osborne CK and Harris
JR: Malignant tumors of the breastDe Vita T, Hellman JS and
Rosenberg SA: Cancer, Principle and Practice of Oncology. 6th.
Philadelphia: Lippincott Williams & Wilkins; pp. 16972001
|
|
9
|
Kaufmann M, von Minckwitz G, Bear HD,
Buzdar A, McGale P, Bonnefoi H, Colleoni M, Denkert C, Eiermann W,
Jackesz R, et al: Recommendations from an international expert
panel on the use of neoadjuvant (primary) systemic treatment of
operable breast cancer: New perspectives 2006. Ann Oncol.
18:1927–1934. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Uzzan B, Nicolas P, Cucherat M and Perret
GY: Microvessel density as a prognostic factor in women with breast
cancer: A systematic review of the literature and meta-analysis.
Cancer Res. 64:2941–2955. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Schwartz GF and Hortobagyi GN: Proceedings
of the consensus conference on neoadjuvant chemotherapy in
carcinoma of the breast, April 26–28, 2003, Philadelphia,
Pennsylvania. Cancer. 100:2512–2532. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bevers TB, Anderson BO, Bonaccio E, Buys
S, Daly MB, Dempsey PJ, Farrar WB, Fleming I, Garber JE, Harris RE,
et al: NCCN clinical practice guidelines in oncology: Breast cancer
screening and diagnosis. J Natl Compr Canc Netw. 7:1060–1096. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Alvarez RH, Booser DJ, Cristofanilli M,
Sahin AA, Strom EA, Guerra L, Kau SW, Gonzalez-Angulo AM,
Hortobagyi GN and Valero V: Phase 2 trial of primary systemic
therapy with doxorubicin and docetaxel followed by surgery,
radiotherapy, and adjuvant chemotherapy with cyclophosphamide,
methotrexate, and 5-fluorouracil based on clinical and pathologic
response in patients with stage IIB to III breast cancer: Long-term
results from the University of Texas M. D. Anderson Cancer Center
Study ID97-099. Cancer. 116:1210–1217. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Nahleh Z, Sivasubramaniam D, Dhaliwal S,
Sundarajan V and Komrokji R: Residual cancer burden in locally
advanced breast cancer: A superior tool. Curr Oncol. 15:271–278.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pacilio C, Morabito A, Nuzzo F, Gravina A,
Labonia V, Landi G, Rossi E, De Maio E, Di Maio M, D'Aiuto G, et
al: Is epirubicin effective in first-line chemotherapy of
metastatic breast cancer (MBC) after an epirubicin-containing
adjuvant treatment? A single center phase III trial. Br J Cancer.
94:1233–1236. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bilbao JI, Rebollo J, Longo JM, Mansilla
F, Muñoz-Galindo L and Vieitez JM: Neoadjuvant intra-arterial
chemotherapy in inflammatory carcinoma of the breast. Br J Radiol.
65:248–251. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Murakami M, Kuroda Y, Nishimura S, Sano A,
Okamoto Y, Taniguchi T, Nakajima T, Kobashi Y and Matsusue S:
Intraarterial infusion chemotherapy and radiotherapy with or
without surgery for patients with locally advanced or recurrent
breast cancer. Am J Clin Oncol. 24:185–191. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kitagawa K, Yamakado K, Nakatsuka A,
Tanaka N, Matsumura K, Takeda K and Kawarada Y: Preoperative
transcatheter arterial infusion chemotherapy for locally advanced
breast cancer (stage IIIb) for down-staging and increase of
resectability. Eur J Radiol. 43:31–36. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pacetti P, Mambrini A, Paolucci R,
Sanguinetti F, Palmieri B, Della Seta R, Muttini MP, Fiorentini G
and Cantore M: Intra-arterial chemotherapy: A safe treatment for
elderly patients with locally advanced breast cancer. In Vivo.
20:761–764. 2006.PubMed/NCBI
|
|
20
|
Shimamoto H, Takizawa K, Ogawa Y,
Yoshimatsu M, Yagihashi K, Okazaki H, Kanemaki Y, Nakajima Y, Ohta
T, Ogata H and Fukuda M: Clinical efficacy and value of
redistributed subclavian arterial infusion chemotherapy for locally
advanced breast cancer. Jpn J Radiol. 29:236–243. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Fiorentini G, Tsetis D, Bernardeschi P,
Varveris C, Rossi S, Kalogeraki A, Athanasakis E, Dentico P,
Kanellos P, Biancalani M, et al: First-line intraarterial
chemotherapy (IAC) with epirubicin and mitoxantrone in locally
advanced breast cancer. Anticancer Res. 23:4339–4345.
2003.PubMed/NCBI
|
|
22
|
Takatsuka Y, Yayoi E, Kobayashi T, Aikawa
T and Kotsuma Y: Neoadjuvantintraarterial chemotherapy in locally
advanced breast cancer: A prospective randomized study. Osaka
Breast Cancer Study Group. Jpn J Clin Oncol. 24:20–25.
1994.PubMed/NCBI
|
|
23
|
Mathew J, Asgeirsson KS, Cheung KL, Chan
S, Dahda A and Robertson JF: Neoadjuvant chemotherapy for locally
advanced breast cancer: A review of the literature and future
directions. Eur J Surg Oncol. 35:113–122. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hoehne F, Chen S, Mabry H and Giuliano AE:
An update on prognosis in breast cancer patients with extensive
axillary disease. Breast J. 14:76–80. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Huang O, Chen C, Wu J, Chen S, Chen X, Liu
G, Hu Z, Lu J, Wu J, Shao Z, et al: Retrospective analysis of 119
Chinese noninflammatory locally advanced breast cancer cases
treated with intravenous combination of vinorelbine and epirubicin
as a neoadjuvant chemotherapy: A median follow-up of 63.4 months.
BMC Cancer. 9:3752009. View Article : Google Scholar : PubMed/NCBI
|