Introduction
Lung cancer accounts for ~14% of incident cases of
all cancers, and was the most common cause of cancer-associated
mortality in the USA in 2013 (1).
Metastasis is the process in which cancer cells in the primary site
spread to a different location in the body and form a new tumor;
this involves entry of tumor cells into circulatory systems (blood
and lymph) from the original site, attachment to distant organs,
cloning and angiogenesis (2).
Metastasis is a common event in cancer pathology, and represents
the primary cause of cancer-associated mortality. In lung cancer,
metastasis is regulated by numerous factors and involves a number
of steps and stages (1–3). Improved understanding of the molecular
mechanisms of metastasis in lung cancer may lead to more effective
clinical control of lung cancer.
MicroRNAs (miRNAs/miRs) are a class of endogenous,
non-coding, single-stranded RNAs with a length of 20–25
nucleotides. By interacting with the 3′-untranslated regions
(3′-UTRs) of target mRNAs, miRNAs silence protein expression
post-transcriptionally in two ways: Degradation of the mRNAs, or
inhibition of mRNA translation into proteins. miRNAs serve
important roles in numerous cellular processes, including
proliferation, differentiation and apoptosis (4–7), and in
the development and progression of lung cancer (8–10).
Our previous study (11) identified that miR-339-5p was
downregulated in non-small cell lung cancer (NSCLC) cells and
tissue samples, and that its expression was inversely associated
with tumor-node-metastasis (TNM) stage. Furthermore, its
upregulation could inhibit NSCLC cell migration and invasion in
vitro. Bioinformatics analyses also demonstrated that the
B-cell CLL/lymphoma 6 (BCL6) and valosin-containing protein
genes may be potential targets of miR-339-5p (11). The present study aimed to confirm that
BCL6 is the target gene of miR-339-5p, and that miR-339-5p
represses the epithelial-to-mesenchymal transition (EMT) of NSCLC
cells via BCL6 regulation. EMTs are vital for morphogenesis
during embryonic development and are also implicated in the
conversion of early stage tumors into invasive malignancies
(2,12). During the EMT process, epithelial
tumor cells lose their expression of E-cadherin, which is an
important epithelial cell-cell adhesion molecule, and begin to
exhibit motility and invasiveness (12). In addition, the relative expression of
miR-339-5p in peripheral blood and its association with disease
progression were investigated.
Materials and methods
Cell culture
The NSCLC A549, H1299, H810, H358 and H1975 cell
lines were provided by the Department of Respiratory Diseases
(purchased from the American Type Culture Collection and maintained
at the Chinese People's Liberation Army General Hospital, Beijing,
China). These cells were grown in RPMI-1640 medium containing 10%
fetal bovine serum (both from Gibco; Thermo Fisher Scientific,
Inc., Waltham, MA, USA), in a humidified atmosphere of 95% air and
5% CO2 at 37°C.
NSCLC tissue specimensand peripheral
blood
The present study was approved by the Ethics
Committee of the 309th Hospital of Chinese PLA (Beijing, China). A
total of 60 surgical NSCLC tissue specimens or biopsy specimens and
pairing peripheral blood samples were obtained from the 309th
Hospital of Chinese PLA. The baseline clinical features of the
patient are shown in Table I.
 | Table I.Baseline clinical features of
patients. |
Table I.
Baseline clinical features of
patients.
| Clinical
features | Case no. |
|---|
| Age (years) |
|
|
≥60 | 41 |
|
<60 | 19 |
| Gender |
|
|
Female | 31 |
|
Male | 29 |
| Smoking
history |
|
|
Yes | 21 |
| No | 39 |
| Pathological
type |
|
|
Adenocarcinoma | 41 |
|
Squamous cell carcinoma | 10 |
| Mixed
type | 9 |
| Clinical stage |
|
| I | 10 |
| II | 17 |
|
III | 21 |
| IV | 12 |
| Lymph node
metastases |
|
| N0 | 12 |
| N1 | 11 |
| N2 | 7 |
| N3 | 30 |
| Distant
metastases |
|
| M0 | 38 |
| M1 | 12 |
All patients signed informed consent forms. A total
of 32 patients underwent complete surgical resection of the primary
tumor. Following surgical resection or biopsy, the peripheral blood
samples were obtained. After 6 months, the peripheral blood samples
were obtained again. Patients who had received chemotherapy or
radiation therapy prior to surgery, or who had rheumatic disease,
acute infection, human immunodeficiency virus infection or other
types of cancer, were excluded from the present study. The clinical
staging was performed according to 7th American Joint Committee on
Cancer/Union for International Cancer Control TNM classification
(13).
Isolation of total RNA and reverse
transcription (RT)-quantitative polymerase chain reaction (qPCR)
analysis
RNA from the NSCLC cells and fresh tissue samples
was extracted using the mirVana™ RNA Isolation kit (catalog no.
AM1560; Ambion®; Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol. RNA from peripheral blood
cells was isolated with the miRNeasy™ Mini kit (catalog no. 217004;
Qiagen, Inc., Valencia, CA, USA). The RNA quality and quantity were
determined by spectrophotometry (ND-1000; NanoDrop Technologies;
Thermo Fisher Scientific, Inc.).
RNA samples from lung cancer cells, tissue samples
or peripheral blood cells were subjected to RT reactions using the
TaqMan MicroRNA Reverse Transcription kit (catalog no. 4366596;
Thermo Fisher Scientific, Inc.). Primers were: miRNA-339-5p
(catalog no. 4427975-ID002257) and U6, (catalog no.
44279975-ID001973) (Thermo Fisher Scientific, Inc.). The
thermocycling conditions were as follows: 16°C for 30 min and 42°C
for 30 min, 85°C for 5 min and 4°C for 5 min. The cDNA was then
amplified by qPCR using TaqMan assays with the aforementioned
primers and TaqMan Universal Master Mix (catalog no. 4440040;
Thermo Fisher Scientific, Inc.) in three independent experiments
according to the manufacturer's protocol. The thermocycling
conditions were as follows: 50°C for 2 min and 95°C for 10 min, for
40 cycles, and each cycle contained two steps, 95°C for 15 sec,
then 60°C for 60 sec). PCR was performed in triplicate for each
individual sample. U6 small nuclear RNA was used as an internal
control. The miRNA expression data are presented as the fold
difference relative to U6 based on the 2−ΔΔCq method
(14), where ΔCq = CqmiRNA-CqU6.
Transient miRNA transfection
A549 and H1299 cells (1×106) were seeded
and cultured overnight in 6-well plates. Subsequently, the cells
were transfected with 20 pmol miR-339-5p mimic (Shanghai GenePharma
Co., Ltd., Shanghai, China) or 20 pmol 2′-O methylated
single-stranded miR-339-5p antisense oligonucleotides (inhibitor;
Shanghai GenePharma Co., Ltd.) using Lipofectamine 2000®
(Invitrogen; Thermo Fisher Scientific, Inc.) according to the
manufacturer's protocol. The miRNA mimics consisted of small
double-stranded RNA oligonucleotides with the following sequence:
positive-sense strand, 5′-UCCCUGUCCUCCAGGAGCUCACG-3′; and antisense
strand, 5′-UGAGCUCCUGGAGGACAGGGAUU-3′. The inhibitor sequence was
5′-CGUGAGCUCCUGGAGGACAGGGA-3′. Negative control RNAs (Shanghai
GenePharma Co., Ltd.) were used to eliminate potential
nonsequence-specific effects, and the sequences used were not
homologous to any sequence in the human genome. The sequences were,
5′-UUCUCCGAACGUGUCACGUTT-3′ (positive-sense strand) and
5′-ACGUGACACGUUCGUAGAATT-3′ (antisense strand; a negative control
for the miRNA mimic) and 5′-CAGUACUUUUGUGUAGUACAA-3′ (a negative
control for the mRNA antisense transfection).
Cell invasion assay
Transwell inserts (24-well insert; pore size, 8 µm;
Corning Incorporated, Corning, NY, USA) were used to explore the
effects of miR-339-5p on A549 and H1299 cell invasion in
vitro. The inserts were initially coated with 100 µl pre-cooled
serum-free RPMI-1640 and Matrigel (1:10; BD Biosciences, Franklin
Lakes, NJ, USA) and allowed to solidify at 37°C in 5%
CO2 for 3 h. Subsequently, 5×104 cells were
resuspended in 100 µl erum-free RPMI-1640 and placed in the top
portion of each Transwell chamber. The lower chamber was filled
with RPMI-1640 medium containing 10% fetal calf serum (HyClone; GE
Healthcare Life Sciences, Little Chalfont, UK) as a
chemoattractant, and the plates were then incubated at 37°C in 5%
CO2 for 24 h. The cells on the upper surface of the
membrane were removed using cotton swabs and PBS, and the cells on
the lower surface of the insert were fixed in 75% methanol and
stained with 0.1% crystal violet at room temperature for 15 min.
Images of fiverandom fields of view for each insert were captured
under a light microscope at ×200 magnification. The cells in the
images were counted, and the data were summarized as the mean ±
standard deviation. The assays were conducted in duplicate for
three independent experiments.
Plasmid construction
The BCL6 3′-UTR was amplified with the
following primers: BCL6-3′-UTR-long non-coding (lnc)-up,
5′-CGGAATTCAGCATGGAGTGTTGATGCTTTCGTCTCCA-3′; and
BCL6-3′-UTR-lnc-down,
5′-GCTCTAGACCAGTTTGACTTTTCAACATTTTATTCTTATA-3′. The PCR product was
inserted between the XbaI and EcoRI sites,
immediately downstream of the stop codon of luciferase gene in
pGL3M, which was obtained from the pGL3-control vector (Promega
Corporation, Madison, WI, USA) by inserting PstI,
EcoRI and NdeI sites (15). To introduce mutations into the seed
sequences in the BCL6 3′-UTR, recombinant PCR was performed
using the aforementioned primers and the following primers:
BCL6-3′-UTR mutated (mut)-down,
5′-AAAGTGATCCGGAGTAGTTATAACACAAGCATGACGCAGA-3′; BCL6-3′-UTR
mut-up, 5′-GCGTCATGCTTGTGTTATAACTACTCCGCATCACTTTTTT-3′. The
mut-down and mut-up primers were respectively used with lnc-up and
lnc-down primers for two PCR. Then the two products were purified
and mixed. Subsequently, lnc-up and lnc-down primers were used to
amplify the mixture. Finally, the mutation for the seed sequence
was gained.
Luciferase reporter assay
pGL3-BCL6 3′-UTR and pGL3-BCL6
3′-UTR-mut were co-transfected with 20 pmol miRNA-339-5p mimics or
negative controls into A549 and H1299 cells using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.). Luciferase assay (Promega Corporation) was
performed by detecting firefly luciferase activity with normalizing
to Renilla luciferase activity after 48 h of transfection as
previous study (16). All experiments
were performed in triplicate and repeated three times.
Western blot analysis
Following transfection of the cells with miR-339-5P
mimics, inhibitors or controls, western blot analyses were
performed, as previously described (15), to detect the levels of BCL6, as well
as the EMT-associated markers neural (N)-cadherin and epithelial
(E)-cadherin, using GAPDH as the loading control. The primary
antibodies were BCL-6 (D-8; catalog no. sc-7388), GAPDH (0411;
catalog no. sc-47724) and NFkBp65 (C-20; catalog no. sc-372) (all
from Santa Cruz Biotechnology, Inc., Dallas, TX, USA. Each
experiment was repeated three times.
Recovery of BCL6 expression
The expression vector for the BCL6 gene was
generated by cloning the PCR product into a pGL3M vector using the
following primers: 5′-CGGAATTCAGGCATTGGACTCAAGATGG-3′; and
5′-GCTCTAGAAACAAAAACATTCCCCCACA-3′. For transfection, cells were
seeded in 6-well plates and then transfected with the indicated
plasmids using Lipofectamine 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocol.
Statistical analysis
All statistical analyses were performed using SPSS
v13.0 (SPSS, Inc., Chicago, IL, USA). Differences between variables
were assessed by Student's t-test. A Spearman's correlation
analysis was used to determine the correlation between miR-339-5p
expression levels in the tissues and in the paired peripheral blood
samples. P<0.05 was considered to indicate a statistically
significant difference.
Results
miR-339-5p expression in NSCLC cells
and patients with NSCLC
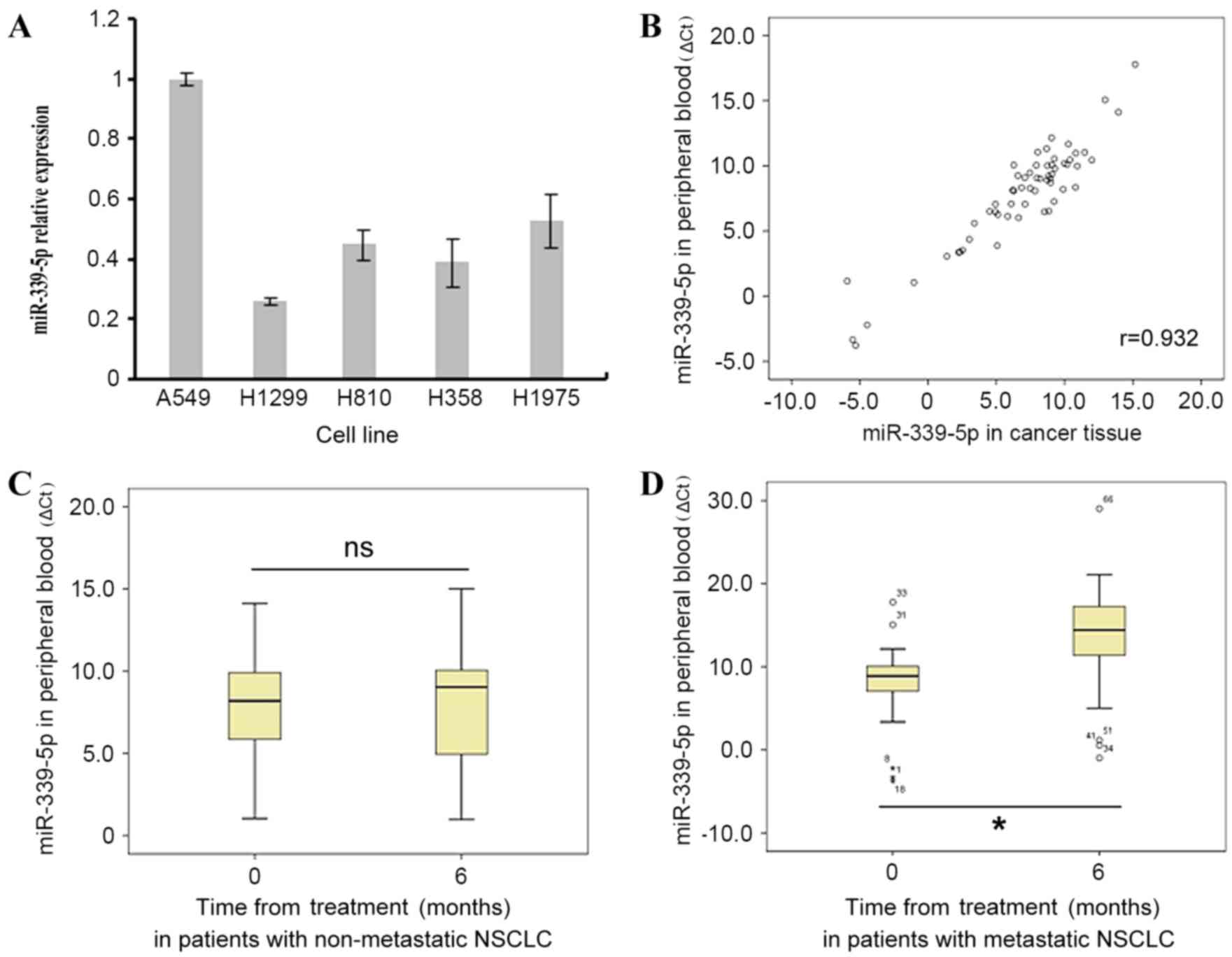
To verify the expression of miR-339-5p in NSCLC,
five typical NSCLC cell lines were used. As demonstrated in
Fig. 1A, miR-339-5p was expressed in
all of the cells, and its expression was highest in A549 cells and
lowest in H1299 cells; these two cells lines were then used for
further study.
The levels of miR-339-5p were also measured in the
peripheral blood samples of 60 patients at the time of their
admission to hospital and 6 months later, in order to determine the
association between the expression of miR-339-5p in peripheral
blood cells and NSCLC metastasis. Spearman's correlation analysis
demonstrated that miR-339-5p levels in peripheral blood samples
were positively correlated to the levels in tumor tissues (r=0.932;
Fig. 1B), indicating that peripheral
miR-339-5p was a good marker for additional study. At 6 months
after admission, the peripheral miR-339-5p expression levels in the
patients (n=27) with non-new metastatic cancer was not
significantly different from that at the time of admission
(P>0.05; Fig. 1C). Conversely,
peripheral miR-339-5p expression in the patients (n=33) with new
metastatic cancer was increased significantly after 6 months
(P<0.05; Fig. 1D) compared with
that at the time of hospital admission.
miR-339-5p inhibits A549 and H1299
cell invasion
A gain-of-function assay was adopted in A549 and
H1299 cell lines to reveal the effect of miR-339-5p on invasion, by
transfecting miR-339-5p mimics into the two cell lines. RT-qPCR
confirmed a significant increase in the expression of miR-339-5p in
cells transfected with the mimic compared with cells transfected
with the negative control, and this increase was concomitant with a
significant decrease in the number of invading cells (P<0.01;
Fig. 2A and B). For the
loss-of-function assay, an inhibitor of miR-339-5p was transfected
into A549 cells. Subsequently, the expression of miR-339-5p was
significantly decreased, and this was concomitant with a
significant increase in the number of invading cells, as compared
with cells transfected with the scramble control (P<0.01;
Fig. 2C and D). These results
indicate that miR-339-5p has an inhibitory effect on cell
invasion.
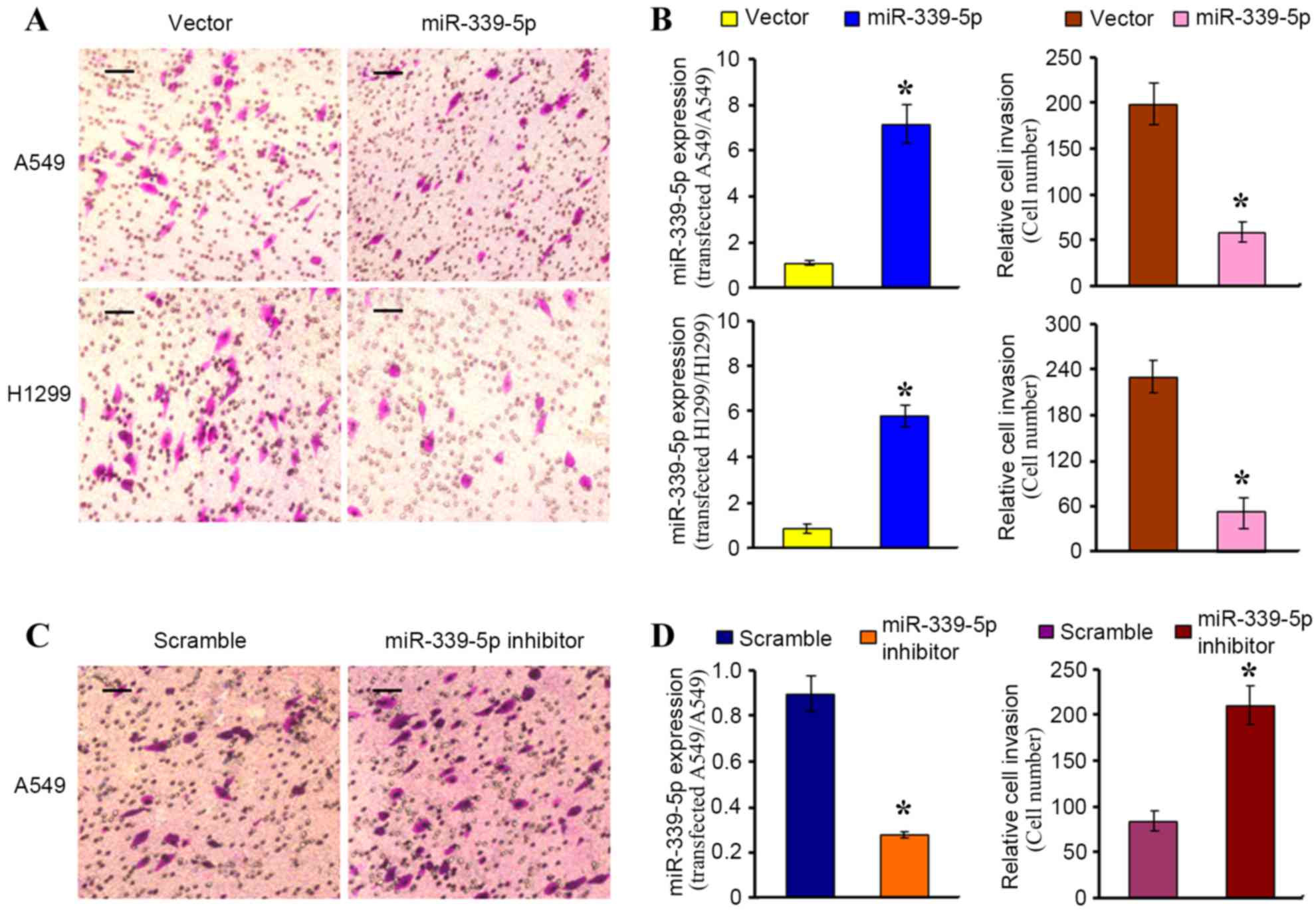 | Figure 2.Inhibition of cell invasion by
miR-339-5p in non-small cell lung cancer cells. A549 or H1299 cells
were transfected with (A and B) miR-339-5p mimics or (C and D)
miR-339-5p inhibitors, or their respective controls. Cell invasion
was evaluated using Matrigel-coated Transwell chambers, and invaded
cells were fixed and stained with 0.1% crystal violet. (A and C)
Representative images of the invaded cells are shown (scale bar;
100 µM). (B and D) Histograms demonstrate the miR-339-5p mRNA
expression levels in the transfected cells (left), and the
corresponding relative cell invasion (right); *P<0.05 vs.
control (vector or scramble). (A and B) Followingtransfection with
mimics and vector (mimics control), the expression of
microRNA339-5p of A549 and H1299 increased 7 and 6 times
respectively, so the number of migrating cells decreased (198_+5
vs. 56_+4, 230-+3, vs. 53+-2, P<0.05). (C and D)
Followingtransfection with inhibitor and scramble (inhibitor
control), the expression of microRNA339-5p of A549 decreased 2.8
times, so the number of migrating cells increased (95_+3 vs.
210_+2, P<0.05). All values presented are the mean ± standard
deviation of triplicate measurements. Experiments were repeated
three times. This is one of the results. miR, microRNA. |
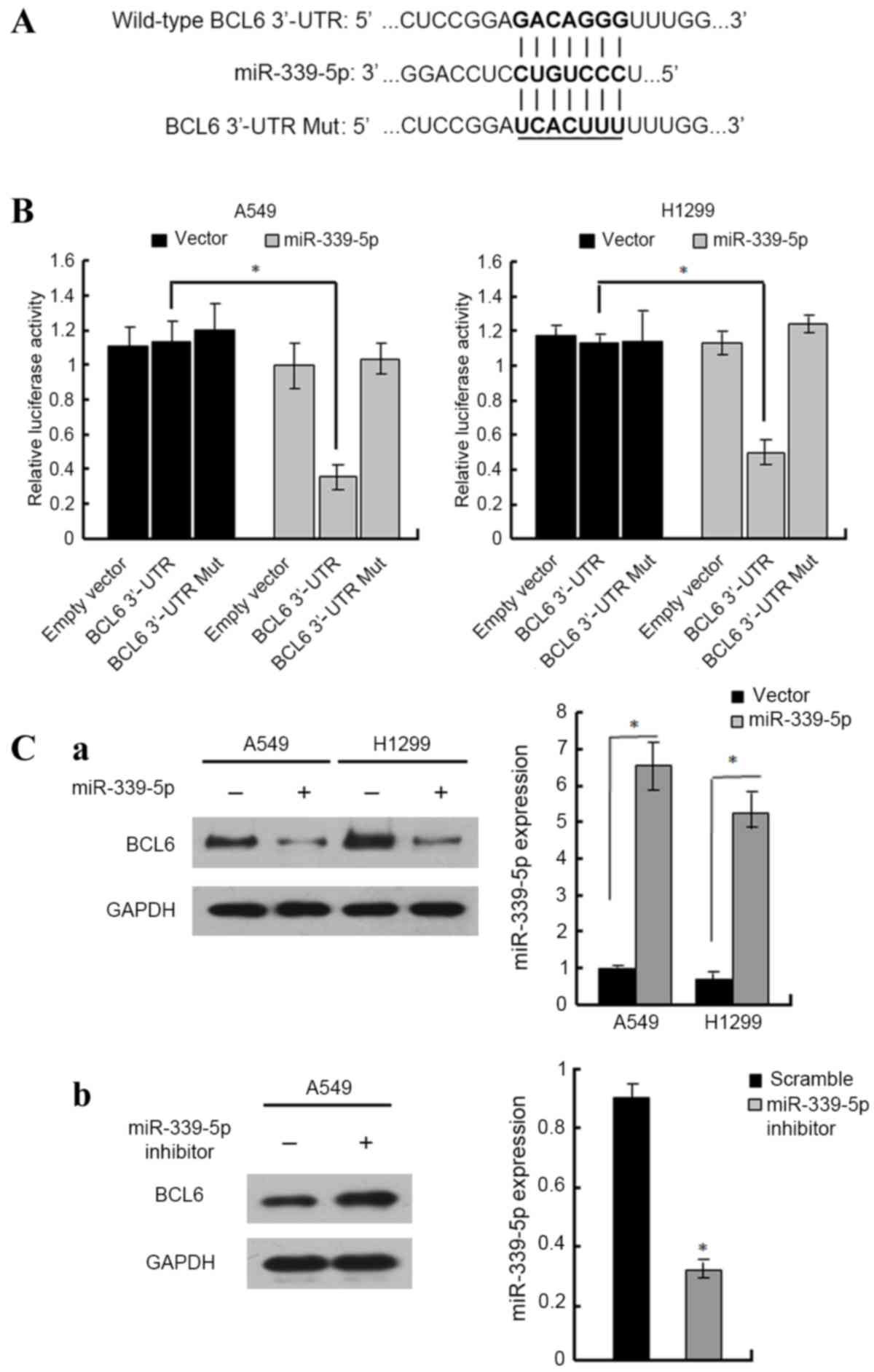
miR-339-5p inhibits BCL6 expression by
targeting its 3′-UTR
In our previous study, target prediction programs,
TargetScan and miRanda, determined that BCL6 was a potential
target gene of miR-339-5p (11). To
confirm this, Dual-Luciferase assays were performed in A549 and
H1299 cell lines transfected with reporter vectors containing the
wild-type or mutated forms of the BCL6 3′-UTR (Fig. 3A). The results demonstrated that
transfection with miR-339-5p mimics decreased the luciferase
activity in A549 and H1299 cells transfected with reporter vector
containing the wild-type BCL6 3′-UTR, whereas miR-339-5p did
not affect the luciferase activity in cells when the BCL6
3′-UTR was mutated (Fig. 3B). Western
blot analysis demonstrated that miR-339-5p mimic transfection
inhibited BCL6expression in A549 and H1299 cells compared with the
controls (Fig. 3C-a). By contrast,
transfection with the miR-339-5p inhibitor increased BCL6 levels in
A549 cells (Fig. 3C-b). These results
suggest that miR-339-5p inhibits BCL6 expression by directly
targeting its 3′-UTR in NSCLC cells.
miR-339-5p regulates the EMT by
inhibiting BCL6
The EMT is one of the most important mechanisms
underlying the metastatic ability of cancer cells. To determine
whether miR-339-5p inhibited NSCLC cells from migrating and
invading via regulation of EMT, the effects of miR-339-5p on
EMT-associated markers were examined in A549 cells. The
overexpression of miR-339-5p significantly inhibited the expression
levels of BCL6 and the mesenchymal marker N-cadherin, while
increasing the expression of the epithelial marker E-cadherin. In
addition, recovery of BCL6 expression in miR-339-5p-overexpressing
cells could attenuate the effects of miR-339-5p overexpression on
the regulation of EMT markers (Fig.
4A). Concordantly, downregulation of miR-339-5p by transfection
with the miR-339-5p inhibitor led to increased levels of BCL6 and
N-cadherin, and decreased levels of E-cadherin (Fig. 4B). These results suggest that
miR-339-5p may regulate EMT, and that BCL6 is essential in this
process. To verify this, miR-339-5p was overexpressed in cells in
which BCL6 was knocked down, revealing that miR-339-5p
overexpression failed to affect the levels of EMT markers when BCL6
was knocked down (Fig. 4C).
Therefore, it was concluded that miR-339-5p regulates EMT via
BCL6.
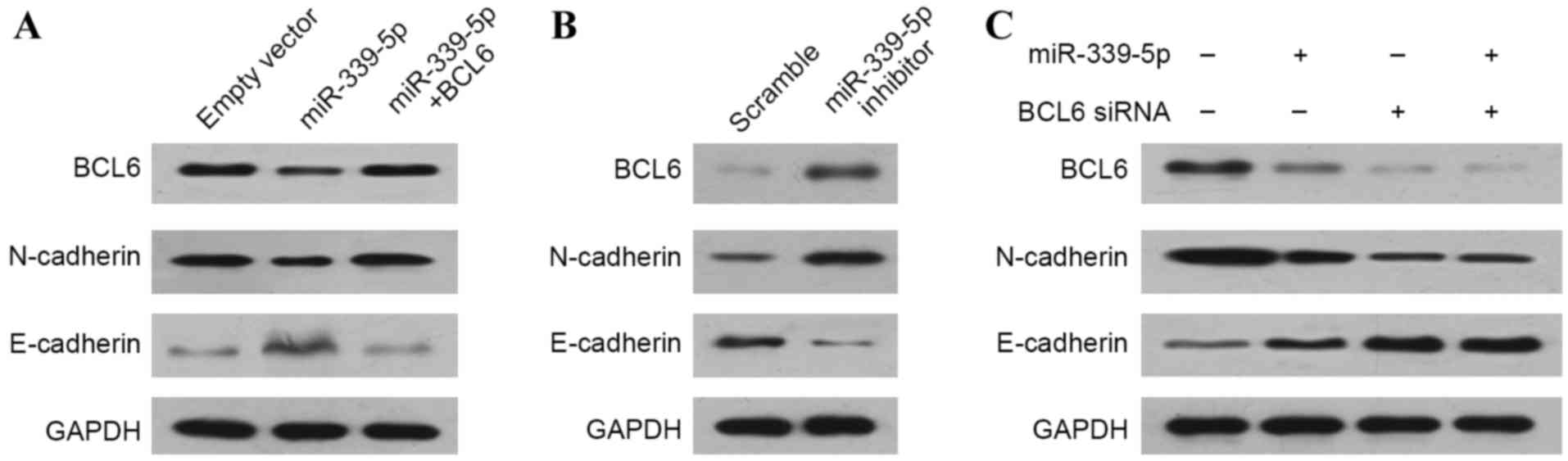 | Figure 4.miR-339-5p regulates the
epithelial-mesenchymal transition by inhibiting BCL6. Western blot
analyses were performed on A549 cells. A549 cells transfected with
(A) miR-339-5p mimics, miR-339-5p mimics plus BCL6, (B) miR-339-5p
inhibitor, (C) BCL6 siRNA, or miR-339-5p mimics plus BCL6 siRNA,
were cultured in regular medium. miR, microRNA; BCL6, B-cell
CLL/lymphoma 6; siRNA, small interfering RNA; N-cadherin, neural
cadherin; E-cadherin, epithelial cadherin. |
Discussion
Previously, multiple studies have demonstrated that
miRNAs are associated with cancer metastasis. The molecular
mechanisms underlying this association may involve various aspects,
as follows.
Firstly, miRNAs can regulate the metastasis of
cancer cells by regulating the expression of oncogenes or tumor
suppressor genes. Asangani et al (17) identified that miR-21 stimulated
invasion and migration in colon cancer cells by inhibiting
programmed cell death protein 4, which has been confirmed to be
tumor suppressor gene. Additionally, Bourguignon et al
(18) verified this result in breast
cancer cells. Furthermore, a study by Li et al (19) demonstrated that miR-34a inhibited cell
migration and invasion by the downregulation of proto-oncogene
C-met expression in human hepatocellular carcinoma cells.
Secondly, miRNAs can regulate metastasis by
regulating the expression of tumor metastasis-associated genes,
such as sex-determining region Y-box 4, radixin (RDX) and
Ras homolog family member A (RHOA). In breast cancer cells,
miR-31 inhibited cell invasion and migration by inhibiting Frizzled
class receptor 3, integrin subunit α 5, matrix metallopeptidase 16,
RDX and RHOA genes, which may promote metastasis
(20). Furthermore, miR-373 and
miR-520c, which are downregulated in prostate cancer cells and
tumors isolated from prostatectomy compared with in their benign
counterparts, may suppress cluster of differentiation 44
translation and enhance the invasion of cancer cells in
vitro (21).
Thirdly, miRNAs may regulate metastasis by
regulating the angiogenesis of tumor cells. Tumor growth depends on
the formation of new blood vessels to facilitate blood perfusion
throughout tumor tissues. A previous study showed that the
downregulation of miR-130a caused the overexpression of the
Homeobox A5 protein, which downregulated the expression of the
angiogenesis-promoting factors vascular endothelial growth factor
receptor and hypoxia-inducible factor-1, resulting in the
inhibition of tumor metastasis (22,23).
Fourthly, miRNAs may regulate metastasis by
inhibiting DNA methylation-associated enzymes, which alter the DNA
methylation status of tumor cells. Fabbri et al (24) revealed that the miR-29 family reverted
aberrant methylation in lung cancer by targeting DNA
methyltransferases 3A and B.
Finally, miRNAs may regulate metastasis via the
regulation of EMT. EMT involves the alteration from an epithelial
to a fibroblast or mesenchymal cell phenotype, with cells gaining
migratory ability. Numerous studies have demonstrated that EMT is
important for the progress of tumor metastasis, and various miRNAs
are involved in EMT regulation (25–28). The
miR-200 family (which includes miR-200a, miR-200b, miR-200c,
miR-141 and miR-429) are important regulators of EMT. Studies have
identified that the miR-200 family could inhibit EMT and cancer
cell migration by directly targeting E-cadherin transcriptional
repressors, including zinc finger E-box-binding homeobox (ZEB)1,
ZEB2, (also known as SIP1) and transcription factor 8 (29–31).
Our previous study suggested that miR-339-5p
inhibited NSCLC cell migration and invasion; in patients with
NSCLC, the expression of miR-339-5p was decreased in cancer tissues
compared with normal lung tissues. Furthermore, statistical
analysis demonstrated that miR-339-5p expression was inversely
correlated with TNM stage and lymph node metastasis (11). In the present study, BCL6 was
verified as the direct target gene of miR-339-5p, and miR-339-5p
was shown to regulate EMT through BCL6.
BCL6 is a 95-kDa nuclear protein, which is encoded
by a gene located on chromosome 3q27 and acts as transcriptional
repressor; it was originally characterized as a regulator of
B-lymphocyte growth and development. BCL6 has been implicated in
the development and growth of B-cell lymphoma (32–35).
Previously, numerous studies have demonstrated that BCL6 is
associated with cancer metastasis. In breast cancer cell lines, Wu
et al (36) suggested that
miR-339-5p may inhibit the expression of BCL6 mRNA, which
was associated with suppressed migration and invasion of cells.
Zhao et al (37) identified
that the downregulation of miR-127 expression may promote
metastasis and proliferation, and that BCL6 was a target of
miR-127. Pinto et al (38)
demonstrated that, in metastatic lymph nodes, the expression of
BCL6 protein was significantly lower compared with that in the
corresponding primary breast cancer. Hirata et al (39) revealed that maintenance of BCL6
function was important for the regulation of progression in gastric
cancer. In the present study, BCL6 was confirmed to be a
target gene of miR-339-5p and was demonstrated to regulate the
invasiveness of NSCLC cells in vitro. Additionally, it was
verified that the relative expression of peripheral miR-339-5p
increases in patients with metastatic NSCLC.
In conclusion, the present study provides evidence
that miR-339-5p may inhibit lung cancer cell invasion and migration
by regulating EMT through BCL6 in vitro, and that miR-339-5p
expression is associated with cancer metastasis, indicating that
miR-339-5p may be an important biomarker for lung cancer
metastasis.
References
|
1
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clinc. 64:9–29. 2014. View Article : Google Scholar
|
|
2
|
Steeq PS: Tumor metastasis: Mechanistic
insights and clinical challenges. Nat Med. 12:895–904. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fidler IJ: The pathogenesis of cancer
metastasis: The ‘seed and soil’ hypothesis revisited. Nat Rev
Cancer. 3:453–458. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Croce CM and Calin GA: miRNAs, cancer, and
stem cell division. Cell. 122:6–7. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Iorio MV and Croce CM: MicroRNAs in
cancer: Small molecules with a huge impact. J Clin Oncol.
27:5848–5856. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mendell JT: MicroRNAs: Critical regulators
of development, cellular physiology and malignancy. Cell Cycle.
4:1179–1184. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yanaihara N, Caplen N, Bowman E, Seike M,
Kumamoto K, Yi M, Stephens RM, Okamoto A, Yokota J, Tanaka T, et
al: Unique microRNA molecular profiles in lung cancer diagnosis and
prognosis. Cancer Cell. 9:189–198. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gibborns DL, Lin W, Creiqhton CJ, Rizvi
ZH, Gregory PA, Goodall GJ, Thilaganathan N, Du L, Zhang Y,
Pertsemlidis A and Kurie JM: Contextual extracellular cues promote
tumor cell EMT and metastasis by regulating miR-200 family
expression. Genes Dev. 23:2140–2151. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ceppi P, Mudduluru G, Kumarswamy R, Rapa
I, Scagliotti GV, Papotti M and Allgayer H: Loss of miR-200c
expression induces an aggressive, invasive, and chemoresistant
phenotype in non-small cell lung cancer. Mol Cancer Res.
8:1207–1216. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li Y, Zhao W, Bao P, Li C, Ma XQ, Li Y and
Chen LA: miR-339-5p inhibits cell migration and invasion in
vitro and may be associated with the tumor-node-metastasis
staging and lymph node metastasis of non-small cell lung cancer.
Oncology Lett. 8:719–725. 2014.
|
|
12
|
Kang Y and Massagué J:
Epithelial-mesenchymal transitions: Twist in development and
metastasis. Cell. 118:277–279. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sobin LH, Gospodrowicz MK and Wittekind C:
International Union against Cancer: TNM Classification of Malignant
Tumours. 7th edition. Wiley-Blackwell; New York, NY: 2011
|
|
14
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhang X, Zhu J, Xing R, Tie Y, Fu H, Zheng
X and Yu B: miR-513a-3p sensitizes human lung adenocarcinoma cells
to chemotherapy by targeting GSTP1. Lung Cancer. 77:488–494. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fan Z, Cui H, Xu X, Lin Z, Zhang X, Kang
L, Han B, Meng J, Yan Z, Yan X and Jiao S: miR-125a suppresses
tumor growth, invasion and metastasis in cervical cancer by
targeting STAT3. Oncotarget. 6:25266–25280. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Asangani IA, Rasheed SA, Nikolova DA,
Leupold JH, Colburn NH, Post S and Allgayer H: MicroRNA-21 (miR-21)
post-transcriptionally downregulates tumor suppressor Pdcd4 and
stimulates invasion, intravasation and metastasis in colorectal
cancer. Oncogene. 27:2128–2136. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bourguignon LY, Spevak CC, Wong G, Xia W
and Gilad E: Hyaluronan-CD44 interaction with protein kinase
C(epsilon) promotes oncogenic signaling by the stem cell marker
Nanog and the production of microRNA-2, leading to down-regulation
of the tumor suppressor protein PDCD4, anti-apoptosis, and
chemotherapy resistance in breast tumor cells. J Biol Chem.
284:26533–26546. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li N, Fu H, Tie Y, Hu Z, Kong W, Wu Y and
Zheng X: miR-34a inhibits migration and invasion by down-regulation
of c-Met expression in human hepatocellular carcinoma cells. Cancer
Lett. 275:44–53. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Valastyan S, Reinhardt F, Benaich N,
Calogrias D, Szász AM, Wang ZC, Brock JE, Richardson AL and
Weinberg RA: A pleiotropically acting microRNA, miR-31, inhibits
breast cancer metastasis. Cell. 137:1032–1046. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yang K, Handorean AM and Iczkowski KA:
MicroRNAs 373 and 520c are downregulated in prostate cancer,
suppress CD44 translation and enhance invasion of prostate cancer
cells in vitro. Int J Clin Exp Pathol. 2:361–369. 2009.PubMed/NCBI
|
|
22
|
Rhoads K, Arderiu G, Charboneau A, Hansen
SL, Hoffman W and Boudreau N: A role for Hox A5 in regulating
angiogenesis and vascular patterning. Lymphat Res Biol. 3:240–252.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chen Y and Gorski DH: Regulation of
angiogenesis through a microRNA (miR-130a) that down-regulates
antiangiogenic homeobox genes GAX and HOXA5. Blood. 111:1217–1226.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Fabbri M, Garzon R, Cimmino A, Liu Z,
Zanesi N, Callegari E, Liu S, Alder H, Costinean S,
Fernandez-Cymering C, et al: MicroRNA-29 family reverts aberrant
methylation in lung cancer by targeting DNA methyltransferases 3A
and 3B. Proc Natl Acad Sci USA. 104:15805–15810. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bracken CP, Gregory PA, Khew-Goodall Y and
Goodall GJ: The role of microRNAs in metastasis and
epithelial-mesenchymal transition. Cell Mol Life Sci. 66:1682–1699.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gregory PA, Bracken CP, Bert AG and
Goodall GJ: MicroRNAs as regulators of epithelial-mesenchymal
transition. Cell Cycle. 7:3112–3118. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Polyak K and Weinberg RA: Transitions
between epithelial and mesenchymal states: Acquisition of malignant
and stem cell traits. Nat Rev Cancer. 9:265–273. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ma L and Weinberg RA: Micromanagers of
malignancy: Role of microRNAs in regulating metastasis. Trends
Genet. 24:448–456. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Park SM, Gaur AB, Lengvel E and Peter ME:
The miR-200 family determines the epithelial phenotype of cancer
cells by targeting the E-cadherin repressors ZEB1 and ZEB2. Genes
Dev. 22:894–907. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hurteau GJ, Carlson JA, Spivack SD and
Brock GJ: Overexpression of the microRNA hsa-miR-200c leads to
reduced expression of transcription factor 8 and increased
expression of E-cadherin. Cancer Res. 67:7972–7976. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gregory PA, Bert AG, Paterson EL, Barry
SC, Tsykin A, Farshid G, Vadas MA, Khew-Goodall Y and Goodall GJ:
The miR-200 family and miR-205 regulate epithelial to mesenchymal
transition by targeting ZEB1 and SIP1. Nat Cell Biol. 10:593–601.
2008. View
Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chang CC, Ye BH, Chaganti RS and
Dalla-Favera R: BCL-6, a POZ/zinc-finger protein, is a
sequence-specific transcriptional repressor. Proc Natl Acad Sci
USA. 93:6947–6952. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Polo JM, Dell'Oso T, Ranuncolo SM,
Cerchietti L, Beck D, Da Silva GF, Prive GG, Licht JD and Melnick
A: Specific peptide interference reveals BCL6 transcriptional and
oncogenic mechanisms in B-cell lymphoma cells. Nat Med.
10:1329–1335. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
34
|
Phan RT, Saito M, Basso K, Niu H and
Dalla-Favera R: BCL6 interacts with the transcription factor Miz-1
to suppress the cyclin-dependent kinase inhibitor p21 and cell
cycle arrest in germinal center B cells. Nat Immunol. 6:1054–1060.
2005. View
Article : Google Scholar : PubMed/NCBI
|
|
35
|
Jardin F, Ruminy P, Bastard C and Tilly H:
The BCL6 proto-oncogene: A leading role during germinal center
development and lymphomagenesis. Pathol Biol (Paris). 55:73–83.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wu ZS, Wu Q, Wang CQ, Wang XN, Wang Y,
Zhao JJ, Mao SS, Zhang GH, Zhang N and Xu XC: miR-339-5p inhibits
breast cancer cell migration and invasion in vitro and may be a
potential biomarker for breast cancer prognosis. BMC Cancer.
10:5422010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhao X, Duan Z, Liu X, Wang B, Wang X, He
J, Yao Z and Yang J: MicroRNA-127 is downregulated by Tudor-SN
protein and contributes to metastasis and proliferation in breast
cancer cell line MDA-MB-231. Anat Rec (Hoboken). 296:1842–1849.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Pinto AE, André S, Silva G, Vieira S,
Santos AC, Dias S and Soares J: BCL-6 oncoprotein in breast cancer:
Loss of expression in disease progression. Pathobiology.
76:235–242. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hirata Y, Ogasawara N, Sasaki M, Mizushima
T, Shimura T, Mizoshita T, Mori Y, Kubota E, Wada T, Tanida S, et
al: BCL6 degradation caused by the interaction with the C-terminus
of pro-HB-EGF induces cyclin D2 expression in gastric cancers. Br J
Cancer. 100:1320–1329. 2009. View Article : Google Scholar : PubMed/NCBI
|