Introduction
Esophageal carcinoma (EC) is a common upper
gastrointestinal tumor, with the third incidence and the fourth
mortality in China (1). Despite
improvements in diagnosis and treatments of esophageal cancer
patients, overall 5-year survival rates were still very low (−40%)
(1,2).
Esophagectomy is the standard strategy for resectable EC.
Unfortunately locoregional recurrence [especially lymphatic
metastasis (LNM)]was the main failure for these patients (3,4).
Multimodality treatment has become increasingly used
for EC, which proved to have good outcomes (5). Neoadjuvant chemoradiotherapy was proved
to be a better strategy for local advanced patients (6,7).
Postoperative radiotherapy (PRT) was only recommended for positive
margin patients in NCCN guidelines (8). However, in most area of China, surgery
was still the first choice of EC patients by now. Postoperative
chemoradiotherapy was controversial and had been studied for many
years. Several Meta analyses concluded that postoperative
chemoradiotherapy significantly decreased postoperative mortality,
local recurrence and distant metastasis rates, with no increased
postoperative complications for patients with resectable esophageal
carcinoma (9–11).
The lymphatic drainage of esophagus is great
complexity, which not only runs through transversely to the
adjacent lymph nodes, but also vertically to distant nodes
(12,13). Early EC with the submucosa
infiltrated, therefore, can be found widely metastasis, even
skipping metastasis (14,15). Because of the complexity lymphatic
drainage of esophageal carcinoma, it is difficult to determine the
target of postoperative volume. After surgery, the anatomy of
esophagus and lymphatic drainage changed. Therefore, the
postoperative target volume is different from that of non-operative
radiotherapy. Many target plans have been used in past decades
(16,17–21),
without a standard criterion.
In order to find out the failure pattern after
surgery, several retrospective studies had been investigated and
advices were concluded for clinic target delineation (22–25).
However, these researches were focused on the patients without PRT.
Few papers were found to investigate the pattern of recurrence
after PRT using a specific target. In this study, recurrence
pattern after radical surgery was investigated by retrospective
analysis of TEC patients with and without PRT aimed to find an
appropriate plan for PRT.
Patients and methods
Patients
The present study was a retrospective investigation
to assess recurrence rates in patients that have undergone a
radical esophagectomy for thoracic EC, with or without PRT. From
January 2012 to December 2015, patients with recurrence who
underwent radical esophagectomy for thoracic esophageal carcinoma
were collected at the First Affiliated Hospital of Anhui Medical
University. Tumor locations were based on the Japanese
classification system (26). Clinic
pathological characteristics (tumor invasion, node, metastasis and
stage) were based on the tumor-node-metastasis (TNM) classification
(7th edition), by the International Union against Cancer (27). Lymphatic station was classified based
on American Thoracic Society/International Association for the
Study of Lung Cancer (IASLC) lymph node station nomenclature
(28).
Case selection
Inclusion criteria: Thoracic CT, abdominal
B-ultrasound, and other images were made to exclude distant
metastasis in all patients before surgery; All patients were proved
to be TEC by pathologic examination after surgery; All patients
were proved to have postoperative recurrence for the first time by
the following ways: clinical, cytology or pathology, and
B-ultrasound, CT or MRI, and other imaging ways (i.e., PET/CT).
Exclusion criteria: Patients with unknown or unclear pathological
characterization; patients without sufficient evidence to support
clinic or pathological recurrence; patients with unknown lymph node
station; patients associated with other tumor; patients with
unknown target or target not accordance with the principle which
discussed below for PRT.
The diagnosis of recurrence
The diagnosis of neck/supraclavicular LNM was mainly
based on physical examination, B-ultrasound, CT/MRI and fine needle
aspiration. The diagnosis of mediastinal LNM was mainly based on
CT, MRI, or PET/CT. The diagnosis of celiac LNM was mainly based on
B-ultrasound, CT or PET/CT. The short diameter >10 mm (5 mm for
lymph nodes of tracheoesophageal groove) or fusion of lymph nodes
or whatever size of lymph node combined with hoarseness or cough
was considered as mediastinal LNM in CT/MRI image, while HUVmax
value of lymph nodes >4.0 in PET/CT (29). The diagnosis of anastomotic recurrence
was based on esophagoscopy. The diagnosis of hematological
recurrence was based on mageological diagnosis according to
different positions.
PRT
Patients with pT3-4 or pN(+) were carried out PRT in
Radiation Oncology Department of my hospital. The time interval was
3–12 months. The most common scheme was 50 Gy/25 fractions, 2
Gy/day, and 5–6 fractions/week. Dose of two patients was <50 Gy
and dose of five patients was >50 Gy, 2 Gy/fraction. Conformal
computed tomography-based planning and a linear accelerator were
used to delivery external beam radiation therapy for these
patients. The clinical target volume (CTV) included tumor bed and
lymphatic drainage regions at high risk. The principle of CTV was
as follows: Upper TEC: The tumor bed with a 3 cm enlargement
superiorly and inferiorly, station 1, station 2, station 4, station
5 and station 7; Middle TEC: The tumor bed with a 3 cm enlargement
superiorly and inferiorly, station 2, station 4, station 5 and
station 7; Lower TEC: The tumor bed with a 3 cm enlargement
superiorly and inferiorly, station 7, station 8, and cardia and
left gastric lymph nodes. The planning target volume (PTV) was
defined as the CTV plus a 0.5–0.8 cm margin.
Follow-up
In the first 2 years, patients were followed-up
every 3 months after surgery and every 6 months thereafter.
Re-examinations included chest enhanced CT scans, abdominal and
cervical ultrasound screening. When necessary, cervical or
abdominal CT, PET/CT, endoscopy, and fine needle aspiration
according to specific symptoms.
Statistical analysis
Statistical analysis was performed using the
statistical package SPSS (version 19.0 for Windows, IBM SPSS,
Armonk, NY, USA). Chi-square and Fisher's test were used in
qualitative variables. Student's t-test was used for continuous
variables. P<0.05 was considered to indicate a statistically
significant difference.
Results
Characterization of patients
A total of 244 patients were collected for the
present study, clinical and pathological characteristics were shown
in Table I. Proportion of pT3-4 and
pN(+) patients with PRT was more than that of patients without PRT
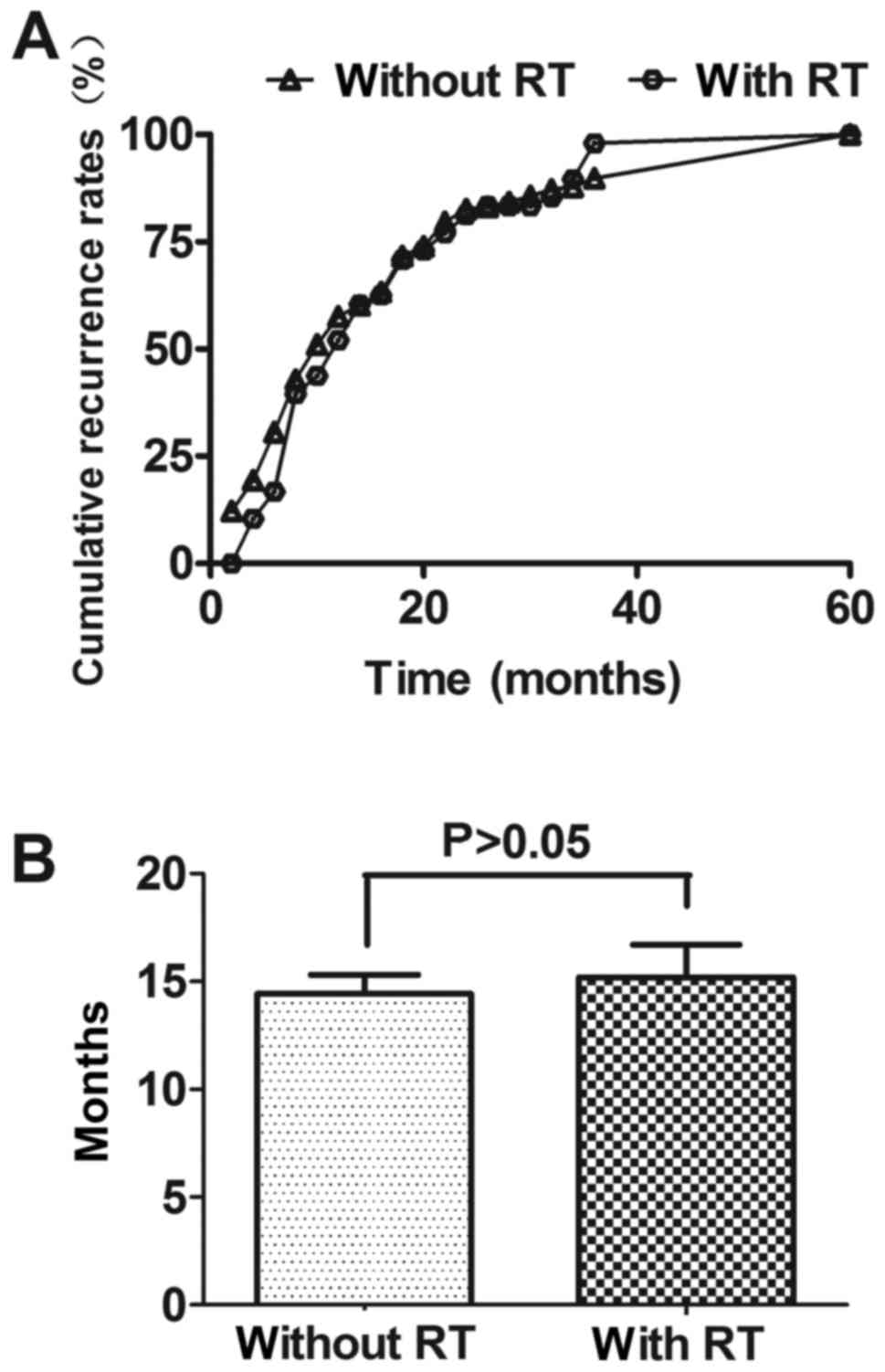
(P=0.04; <0.01, respectively). Among 196 patients treated
without PRT, the mean recurrence time (MT) was 14.22±0.88 months
(1.0–61.0 months) after operation, while MT was 15.37±1.53 months
(3.0–54.0 months) for the other 48 patients who treated with PRT.
The recurrence time of these two groups had no significant
difference. The recurrence time was shown in Fig. 1.
 | Table I.Clinical and pathological
characteristics of patients (n=244). |
Table I.
Clinical and pathological
characteristics of patients (n=244).
| Parameters | Without PRT
(n=196) | With PRT (n=48) | P-value |
|---|
| Age (years) |
|
| 0.94 |
| ≤60 | 87 | 21 |
|
|
>60 | 109 | 27 |
|
| Sex |
|
| 0.57 |
|
Male | 165 | 42 |
|
|
Female | 31 | 6 |
|
| T stage |
|
| 0.04 |
|
pT1-2 | 81 | 12 |
|
|
pT3-4 | 115 | 36 |
|
| N stage |
|
| 0.01 |
| pN
(−) | 108 | 11 |
|
| pN
(+) | 88 | 37 |
|
|
Differentiation |
|
| 0.04 |
|
Poor | 53 | 22 |
|
|
Moderate | 96 | 19 |
|
|
Well | 47 | 7 |
|
| Tumor location |
|
| 0.11 |
|
Upper | 20 | 1 |
|
|
Middle | 114 | 29 |
|
|
Lower | 62 | 18 |
|
| Type of tumor |
|
| 0.76 |
|
SCC | 183 | 46 |
|
|
Others | 13 | 2 |
|
| Chemotherapy |
|
| – |
| 0–2
cycles | – | 31 |
|
| ≥3
cycles | – | 17 |
|
|
Lymphadenectomy |
|
| 0.36 |
|
Two-field | 147 | 39 |
|
|
Three-field | 49 | 9 |
|
| Anastomotic
site |
|
| 0.60 |
|
Neck | 29 | 9 |
|
| Upon
aortic arch | 137 | 34 |
|
| Below
aortic arch | 30 | 5 |
|
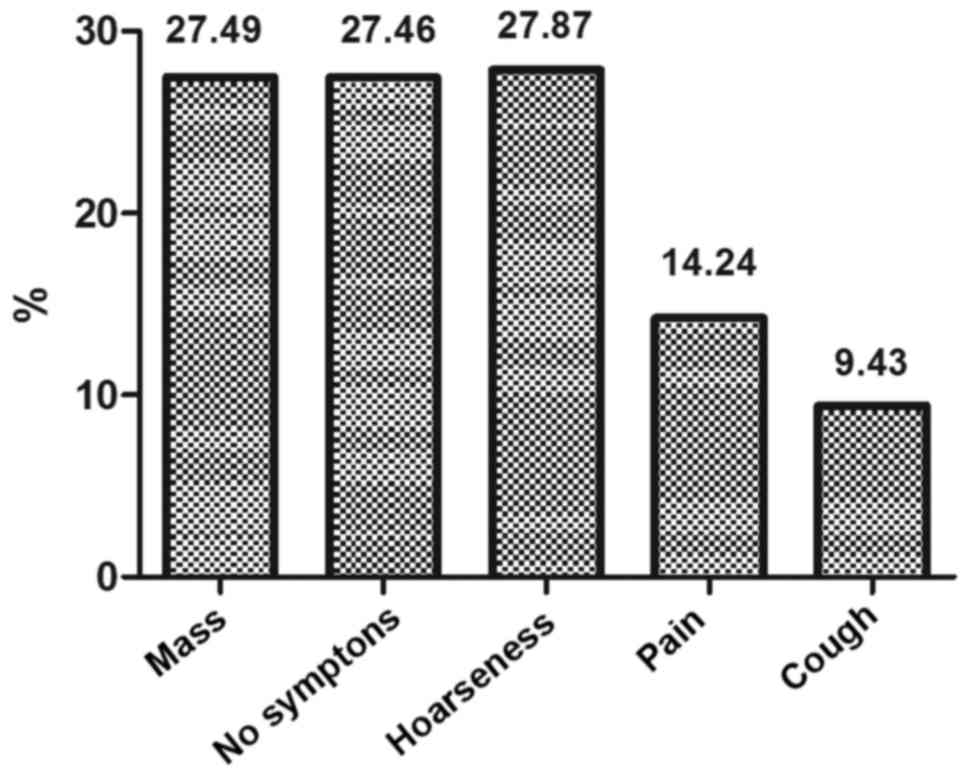
The common symptoms for those patients were mass
found in the lower neck and supraclavicular regions (27.5%), hoarse
voice (27.9%), and local pain (14.2%), while a large number (27.5%)
were found with no symptom by using imaging examinations (i.e.,
chest CT/MRI, abdominal B-ultrasound, PET/CT). The symptoms for
these patients were shown in Fig.
2.
Pattern of recurrence after radical
surgery
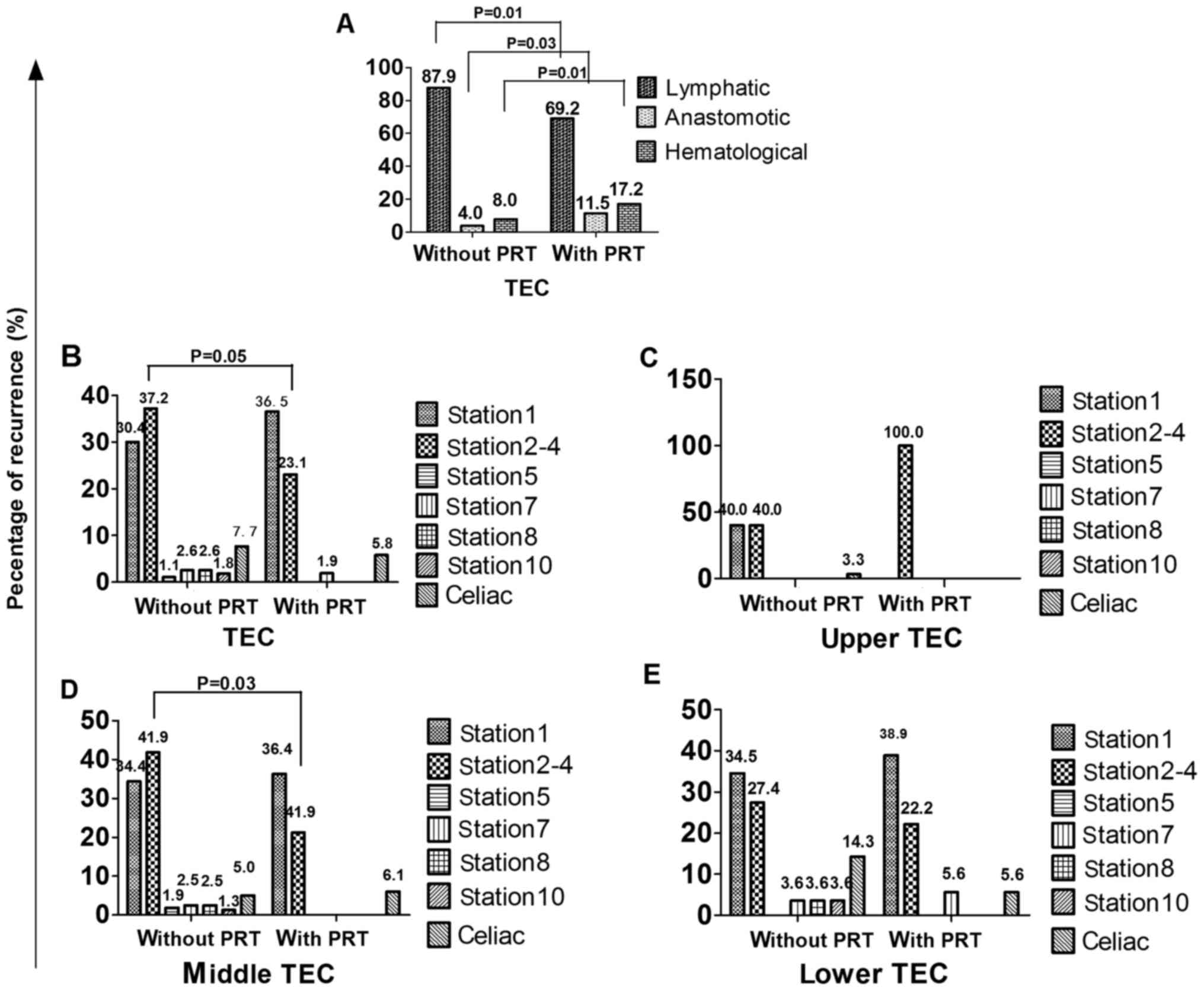
Distribution
Among the 249 patients, a total of 332 sites (274
for patients without PRT, 52 for patients with PRT) of recurrence
were found and the distribution of sites was shown in Table II. The patterns of initial recurrence
included lymphatic recurrence, anastomotic recurrence and
hematological recurrence for patients without and with PRT, as
shown in Fig. 3A. The lymphatic
recurrence ratio was 87.9% (241/274) vs. 69.2% (36/52), the
difference had statistical significance (P=0.01). The anastomotic
recurrence ratio was 4.0% (11/274) vs. 11.5% (6/52), the difference
had statistical significance (P=0.03). The hematological recurrence
ratio was 8.0% (22/274) vs. 17.2% (10/52), the difference had
statistical significance (P=0.01).
 | Table II.Location of recurrence patterns in
patients. |
Table II.
Location of recurrence patterns in
patients.
|
| Upper | Middle | Lower | Total |
|---|
|
|
|
|
|
|
|---|
| Location | Without PRT | With PRT | Without PRT | With PRT | Without PRT | With PRT | Without PRT | With PRT |
|---|
|
Supraclavicular |
|
|
|
|
|
|
|
|
| Station
1 | 12 | 0 | 55 | 12 | 29 | 7 | 96 | 19 |
| Mediastinal |
|
|
|
|
|
|
|
|
| Station
2–4 | 12 | 1 | 67 | 7 | 23 | 4 | 102 | 12 |
| Station
5 | 0 | 0 | 3 | 0 | 0 | 0 | 3 | 0 |
| Station
7 | 0 | 0 | 4 | 0 | 3 | 1 | 7 | 1 |
| Station
8 | 0 | 0 | 4 | 0 | 3 | 0 | 7 | 0 |
| Station
10 | 0 | 0 | 2 | 0 | 3 | 0 | 5 | 0 |
| Celiac | 1 | 0 | 8 | 2 | 12 | 1 | 21 | 3 |
| Anastomotic | 2 | 0 | 5 | 4 | 4 | 2 | 11 | 6 |
| Hematological | 3 | 0 | 12 | 8 | 7 | 3 | 22 | 11 |
| Total | 30 | 1 | 160 | 33 | 84 | 18 | 274 | 52 |
Fig. 3B shows the
lymphatic recurrence ratio in different regions. The recurrence
ratios of station 1, station 2–4, station 5, station 7, station 8,
station 10 and celiac regions were 30.0% (96/274) vs. 36.5%
(19/52), 37.2% (102/274) vs. 23.1% (12/52), 1.1% (3/274) vs. 0.0%
(0/52), 2.6% (7/274) vs. 1.9% (1/52), 2.6% (7/274) vs. 0.0% (0/52),
1.8% (5/274) vs. 0.0% (0/52), 7.7% (21/274) vs. 5.8% (3/52),
respectively for patients without and with PRT. The difference of
recurrence ratio in each region for patients without and with PRT
had no statistical significance (P>0.05).
Fig. 3C-E shows the
lymphatic recurrence ratios in different regions of patients
without and with PRT in different locations. For upper TEC, the
differences of recurrence ratio in each region for patients without
and with PRT had no statistical significance (P>0.05). For
middle TEC, the difference of recurrence ratio in station 2–4 for
patients without and with PRT had statistical significance
(P=0.03), while other regions had no statistical significance
(P>0.05). For lower TEC, the differences of recurrence ratio in
each region for patients without and with PRT had also no
statistical significance (P>0.05).
Relation between recurrence and target
for radiotherapy patients
We defined the recurrence in the primary target as
infield recurrence, while the others as outfield recurrence. Among
53 patients with PRT, recurrence ratio of infield was 25.0%
(13/52). Univariate analysis showed that the location of tumor was
the influence factor for infield recurrence. The upper TEC had a
higher infield recurrence (100.0%), while the middle and lower TEC
had a higher outfield recurrence (67.7, 88.9%, respectively). The
differences in different locations had a statistical significance
(P=0.02). The pattern of infield and outfield recurrences was shown
in Table III.
 | Table III.Univariate analysis for infield and
outfield recurrence for patients with PRT. |
Table III.
Univariate analysis for infield and
outfield recurrence for patients with PRT.
| Parameters | Infield (n=13) | Outfield
(n=39) | P-value |
|---|
| Age (years) |
|
| 0.18 |
|
≤60 | 10 | 19 |
|
|
>60 | 3 | 20 |
|
| Sex |
|
| 0.82 |
|
Male | 12 | 33 |
|
|
Female | 1 | 6 |
|
| T stage |
|
| 1.0 |
|
pT1-2 | 4 | 11 |
|
|
pT3-4 | 9 | 28 |
|
| N stage |
|
| 1.00 |
|
pN(−) | 11 | 31 |
|
|
pN(+) | 2 | 8 |
|
|
Differentiation |
|
| 0.11 |
|
Poor | 3 | 19 |
|
|
Moderate | 9 | 14 |
|
|
Well | 1 | 6 |
|
| Tumor location |
|
| 0.01 |
|
Upper | 1 | 0 |
|
|
Middle | 11 | 22 |
|
|
Lower | 1 | 17 |
|
| Type of tumor |
|
| 1.00 |
|
SCC | 13 | 37 |
|
|
Others | 0 | 2 |
|
| Chemotherapy |
|
| 1.0 |
| 0–2
cycles | 9 | 25 |
|
| ≥3
cycles | 4 | 14 |
|
|
Lymphadenectomy |
|
| 0.06 |
|
Two-field | 8 | 35 |
|
|
Three-field | 5 | 4 |
|
| Anastomotic
site |
|
| 0.90 |
|
Neck | 3 | 7 |
|
| Upon
aortic arch | 9 | 28 |
|
| Below
aortic arch | 1 | 4 |
|
Discussion
In the present study, we found that there was no
statistical difference for recurrence time of patients with and
without PRT, but the MT for patients with PRT was over one month
longer than that for patients without PRT. What is more, the
patients with PRT were usually advanced TEC [i.e., pT3/4, pN (+)].
Therefore, local advanced TEC may benefit from PRT, which had been
studied by many researches (9–11,30). We noticed that a few patients recurred
in <3 months after radical surgery, which may due to
micrometastasis that cannot be found using common clinic
examinations before surgery and was not cleaned up completely.
Prenzel et al (31) found that
15% of patients with pT1N0M0 carcinoma of the esophagus and even
those with submucosal infiltration show nodal micrometastasis.
Methods should be used to early discovery the removed or unremoved
micrometastasis (32,33).
Lymphatic recurrence was the most common pattern for
patients without PRT, 87.9% in the present study. Other researches
also have the same results (3,22,23). Taken in this sense, PRT should be a
useful way to reduce recurrence, as sub-clinic lesions or lymphatic
micrometastasis can be further controlled. However, the lymphatic
system of esophagus presents great complexity. The esophageal wall
has a rich network of lymphatic drainage from layer of muscularis
mucosa to tunica adventitia. Early EC can be found widely
metastasis, even skipping metastasis. At present, two-field
lymphadenectomy and three-field lymphadenectomy were the common
procedure (in our hospital, two-field lymphadenectomy was the
main). Because of the limitation of the operation itself, the upper
mediastinum and supraclavicular lymph nodes are difficult to clean
up, and they are most likely to become sub-clinical lesions invaded
regions. Moreover, when patients are operated via left thoracic
incision, we could not clear up the upper paraesophageal nodes and
the nodes located in the cervicothoracic junction, due to the
occlusion of the aortic arch, left lock artery and subclavian
artery. As a result, the lower neck, supraclavicular and the upper
mediastinum regions have a high recurrence. In the present study,
the recurrence ratios of station 1 and station 2–4 for patients
without PRT were 30.0 and 37.2%, totally 67.2%. The lower
mediastinum and celiac lymph nodes can be easily cleaned up, so the
recurrence ratios of station 5, station 7, station 8, station 10
and celiac regions were very low (totally 15.7%). The results were
similar to other research (24,34). From
what has been discussed above, we may draw that lower neck,
supraclavicular regions and upper mediastinum regions (station 1–4)
are the hot spots in PRT.
Then we need to think about whether there are
differences in postoperative target for different locations of TEC
patients. As we know, lymphatic of TEC can drain to different
regions, but usually there is one predominant region of drainage.
The upper lymphatic of TEC drains upward mainly, the middle
lymphatic can drain upward and downward and the lower lymphatic
mainly drains downward (35). Because
lymph nodes of the middle, lower mediastinum and celiac regions can
be easily and thoroughly cleaned up, the CTV should be different
from that of patients without esophagectomy. In this study, the
lymphatic recurrence ratios in the station 1 and station 2–4 were
totally 80.0, 76.3, 61.9%, respectively, for upper, middle and
lower TEC. The lymphatic recurrence ratios in station 5–10 were
totally 0.0, 8.1 and 10.7%, respectively. The lymphatic recurrence
ratios in celiac regions were 3.3, 5.0 and 14.3%, respectively. The
recurrence rates in station 5–10 for all TEC and celiac regions for
upper and middle TEC were low, so more evidences should be
collected to explain whether these regions should be included in
CTV of PRT. Many researches (24,35,36) also
suggested that PRT target should include supraclavicular and
mediastinal regions for all TEC, while celiac lymph node regions
should also be included for lower TEC.
In order to further find out the suitable CTV for
PRT, we analyzed the pattern of recurrence for patients treated
with PRT using the target mentioned above. The lymphatic recurrence
ratio decreased (69.2 vs. 87.9%; P=0.01), while the hematological
recurrence ratio increased compared to patients without PRT (17.2
vs. 8.0%; P=0.01). It indicated that PRT should decrease lymphatic
recurrence. For upper TEC, CTV including station 1 and station 2–4
has been an accepted standard. In the present study, only one
patient (2.1%) was collected and recurred in station 2–4 (infield),
indicating that lower recurrence rate after PRT. The negative
statistical results of recurrence ratio in station 1 and station
2–4 may due to the limited PRT patients. For middle TEC (68.8%),
the recurrence was mostly in station1 (outfield, 36.4%) and station
2–4 (infield, 21.2%). However the recurrence ratio of station 2–4
decreased compared to patients without PRT (P=0.03), while the
recurrence ratio in other regions have no statistic difference for
patients with and without PRT. For lower TEC (37.5%), the
recurrence was also mostly in station1 (38.9%) and station 2–4
(22.2%), which were both outfield. The recurrence ratios in station
7, station 8 and celiac regions (infield) had no statistic
difference for patients with and without PRT. Chen et al
(37) found that lower TEC had a wide
range of celiac lymph nodes metastasis, 34.4% in nodes along the
left gastric artery, 21.7% in nodes along left cardiac, 17.8% in
nodes along the lesser curvature, 9.4% in nodes along right cardiac
and 6.1% in nodes along the common hepatic artery at time of
surgery. However nodes along the left gastric artery, left cardiac
and lesser curvature usually can be cleaned up. Oppedijk et
al (38) found that radiotherapy
did not decrease the celiac lymph nodes metastasis. For these
reasons, we also have no adequate evidence to irradiate the lower
mediastinum and celiac regions for middle and lower TEC, which may
increase complications or radiotherapy-related death.
For infield recurrences, we might think about
whether 50 Gy of biological effective dose (BED) was enough for
sub-clinic or micrometastasis tumor. Moon et al (39) suggested that total radiation dose
should be at least 50 Gy in PRT alone. Also, researches (3,22,38,40) showed
that the anastomotic recurrence ratio was low (4.0% in my study),
we should not include it into CTV. In the past, we usually focused
on the tumor bed for PRT of TEC, however, the anatomical structure
of the primary esophagus changed after surgery. The recurrence
ratio in primary tumor bed was also very low, ranging from 3.6 to
8.8% (41–43), so primary tumor bed was not indeed for
PRT.
In conclusion, for patients treated with standard
esophagectomy, the lower neck, supraclavicular regions and upper
mediastinal regions (station 1, 2 and 4) should be included in the
CTV of PRT, while lower mediastinal regions and celiac regions may
not be included in CTV. More evidence are needed to find out a
suitable BED for PRT. Because this was a retrospective study,
potential bias may exist (i.e., more local recurrence patients
collected in my department for further treatments). Furthermore,
there are many limitations in the present study, such as patients
with and without PRT were different [i.e., patients with PRT were
usually pT3-4, pN (+)]; Chemotherapy were not very clear in
patients without PRT and the used CTV of PRT was still not accepted
in some research centers. As a result, we cannot evaluate the
efficacy of PRT and chemotherapy. In future, multicentric,
perspective, large-scale trials should be conducted to find out a
more suitable modality for PRT of EC.
References
|
1
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–122. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen MF, Chen PT, Lu MS, Lee CP and Chen
WC: Survival benefit of surgery to patients with esophageal
squamous cell carcinoma. Sci Rep. 7:461392017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Liu Q, Cai XW, Wu B, Zhu ZF, Chen HQ and
Fu XL: Patterns of failure after radical surgery among patients
with thoracic esophageal squamous cell carcinoma: Implications for
the clinical target volume design of postoperative radiotherapy.
PLoS One. 9:e972252014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hsu PK, Chen HS, Huang CS, Liu CC, Hsieh
CC, Hsu HS, Wu YC and Wu SC: Patterns of recurrence after
oesophagectomy and postoperative chemoradiotherapy versus surgery
alone for oesophageal squamous cell carcinoma. Br J Surg.
104:90–97. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Markar SR, Noordman BJ, Mackenzie H,
Findlay JM, Boshier PR, Ni M, Steyerberg EW, van der Gaast A,
Hulshof MCCM, Maynard N, et al: Multimodality treatment for
esophageal adenocarcinoma: Multi-center propensity-score matched
study. Ann Oncol. 28:519–527. 2017.PubMed/NCBI
|
|
6
|
Kumaran D, John S, Isiah R and Das S:
Management of locally advanced carcinoma oesophagus with
radiation/chemoradiation: Single institute experience. J
Gastrointest Cancer. 47:313–317. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Huang Y, Wang H, Luo G, Zhang Y, Wang L
and Li K: A systematic review and network meta-analysis of
neoadjuvant therapy combined with surgery for patients with
resectable esophageal squamous cell carcinoma. Int J Surg.
38:41–47. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
National Comprehensive Cancer Network, .
Esophageal Cancer Clinical Practice Guidelines in Oncology.
http://www.nccn.org/professionals/physician_gls/f_guidelines.aspJuly
1–2016
|
|
9
|
Thallinger CM, Kiesewetter B, Raderer M
and Hejna M: Pre- and postoperative treatment modalities for
esophageal squamous cell carcinoma. Anticancer Res. 32:4609–4627.
2012.PubMed/NCBI
|
|
10
|
Zhu Y, Li M, Kong L and Yu J:
Postoperative radiation in esophageal squamous cell carcinoma and
target volume delineation. Onco Targets Ther. 9:4187–4196. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ku GY and Ilson DH: Adjuvant
(postoperative) therapy for esophageal cancer. Thorac Surg Clin.
23:525–533. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nishimaki T, Tanaka O, Suzuki T, Aizawa K,
Hatakeyama K and Muto T: Patterns of lymphatic spread in thoracic
esophageal. Cancer. 74:4–11. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Herbella FA, Del Grande JC and Colleoni R;
Japanese Society for Disease of the Esophagus, : Anatomical
analysis of the mediastinal lymph nodes of normal Brazilian
subjects according to the classification of the Japanese Society
for Diseases of the Esophagus. Surg Today. 33:249–253. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gotohda N, Nishimura M, Yoshida J, Nagai K
and Tanaka N: The pattern of lymphatic metastases in squamous cell
carcinoma of the esophagus. Hepatogastroenterology. 52:105–107.
2005.PubMed/NCBI
|
|
15
|
Sgourakis G, Gockel I, Lyros O, Hansen T,
Mildenberger P and Lang H: Detection of lymph node metastases in
esophageal cancer. Expert Rev Anticancer Ther. 11:601–612. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ténière P, Hay JM, Fingerhut A and Fagniez
PL: Postoperative radiation therapy does not increase survival
after curative resection for squamous cell carcinoma of the middle
and lower esophagus as shown by a multicenter controlled trial.
French University Association for Surgical Research. Surg Gynecol
Obstet. 173:123–130. 1991.PubMed/NCBI
|
|
17
|
Rice TW, Adelstein DJ, Chidel MA, Rybicki
LA, DeCamp MM, Murthy SC and Blackstone EH: Benefit of
postoperative adjuvant chemoradiotherapy in locoregionally advanced
esophageal carcinoma. J Thorac Cardiovasc Surg. 126:1590–1596.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yu E, Tai P, Younus J, Malthaner R, Truong
P, Stitt L, Rodrigues G, Ash R, Dar R, Yaremko B, et al:
Postoperative extended-volume external-beam radiation therapy in
high-risk esophageal cancer patients: A prospective experience.
Curr Oncol. 16:48–54. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Qiao XY, Wang W, Zhou ZG, Gao XS and Chang
JY: Comparison of efficacy of regional and extensive clinical
target volumes in postoperative radiotherapy for esophageal
squamous cell carcinoma. Int J Radiat Oncol Biol Phys. 70:396–402.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang W, Liu X, Xiao Z, Wang L, Zhang H,
Chen D, Zhou Z, Feng Q, Hui Z, Liang J, et al: Efficacy of
intensity-modulated radiotherapy for resected thoracic esophageal
squamous cell carcinoma. Thorac Cancer. 6:597–604. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xu X, Xie HY, Zhou D, Huang RH, Bai YR,
Yuan J and Ye M: Comparison and prognostic analysis of adjuvant
radiotherapy versus salvage radiotherapy for treatment of radically
resected locally advanced esophageal squamous cell carcinoma.
Biomed Res Int. 2016:85486942016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Nakagawa S, Kanda T, Kosugi S, Ohashi M,
Suzuki T and Hatakeyama K: Recurrence pattern of squamous cell
carcinoma of the thoracic esophagus after extended radical
esophagectomy with three-field lymphadenectomy. J Am Coll Surg.
198:205–211. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ninomiya I, Okamoto K, Tsukada T,
Kinoshita J, Oyama K, Fushida S, Osugi H and Ohta T: Recurrence
patterns and risk factors following thoracoscopic esophagectomy
with radical lymph node dissection for thoracic esophageal squamous
cell carcinoma. Mol Clin Oncol. 4:278–284. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wu SG, Dai MM, He ZY, Sun JY, Lin HX, Lin
H and Li Q: Patterns of regional lymph noed recurrence after
radical surgery for thoracic esophageal squamous cell carcinoma.
Ann Thorac Surg. 101:551–557. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sugiyama M, Morita M, Yoshida R, Ando K,
Egashira A, Takefumi O, Saeki H, Oki E, Kakeji Y, Sakaguchi Y and
Maehara Y: Patterns and time of recurrence after complete resection
of esophageal cancer. Surg Today. 42:752–758. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Japanese Esophageal Society, . Japanese
Classification of Esophageal Cancer, 11th Edition: Part I.
Esophagus. 14:1–36. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sobin LH, Gospodarowicz MK and Wittekind
C: TNM classification of malignant tumors. 7th. Wiley-Blackwell;
Oxford: 2010
|
|
28
|
Rusch VW, Asamura H, Watanabe H, Giroux
DJ, Rami-Porta R and Goldstraw P; Members of IASLC Staging
Committee, : The IASLC lung cancer staging project: A proposal for
a new international lymph node map in the forthcoming seventh
edition of the TNM classification for lung cancer. J Thorac Oncol.
4:568–577. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ela Bella AJ, Zhang YR, Fan W, Luo KJ,
Rong TH, Lin P, Yang H and Fu JH: Maximum standardized uptake value
on PET/CT in preoperative assessment of lymph node metastasis from
thoracic esophageal squamous cell carcinoma. Chin J Cancer.
33:211–217. 2014.PubMed/NCBI
|
|
30
|
Wong AT, Shao M, Rineer J, Lee A, Schwartz
D and Schreiber D: The impact of adjuvant postoperative radiation
therapy and chemotherapy on survival after esophagectomy for
esophageal carcinoma. Ann Surg. 265:1146–1151. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Prenzel KL, Hölscher AH, Drebber U,
Agavonova M, Gutschow CA and Bollschweiler E: Prognostic impact of
nodal micrometastasis in early esophageal cancer. Eur J Surg Oncol.
38:314–318. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mumtaz WR, Hegde V and Yadav N:
Micrometastasis detection using special stains in nodal tissues of
oral squamous cell carcinoma-a histochemical study. J Clin Diagn
Res. 10:ZC23–ZC26. 2016.PubMed/NCBI
|
|
33
|
Zhou Z, Qutaish M, Han Z, Schur RM, Liu Y,
Wilson DL and Lu ZR: MRI detection of breast cancer micrometastases
with a fibronectin-targeting contrast agent. Nat Commun.
6:79842015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Cai WJ and Xin PL: Pattern of relapse in
surgical treated patients with thoracic esophageal squamous cell
carcinoma and its possible impact on target delineation for
postoperative radiotherapy. Radiother Oncol. 96:104–107. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Riquet M, Saab M, Le Pimpec Barthes F and
Hidden G: Lymphatic drainage of the esophagus in the adult. Surg
Radiol Anat. 15:209–211. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang WC, Wang QF, Xiao ZF, Yang LH and
Liu XY: Patterns of failure after complete resection of thoracic
esophageal squamous cell carcinoma: Implications for postoperative
radiation therapy volumes. Chin J Radiat Oncol Oncol Radiat Oncol.
21:38–41. 2012.(In Chinese).
|
|
37
|
Chen J, Liu S, Pan J, Zheng X, Zhu K, Zhu
J, Xiao J and Ying M: The pattern and prevalence of lymphatic
spread in thoracic oesophageal squamous cell carcinoma. Eur J
Cardiothorac Surg. 36:480–486. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Oppedijk V, van der Gaast A, van Lanschot
JJ, van Hagen P, van Os R, van Rij CM, van der Sangen MJ, Beukema
JC, Rütten H, Spruit PH, et al: Patterns of recurrence after
surgery alone versus preoperative chemoradiotherapy and surgery in
the CROSS trials. J Clin Oncol. 32:385–391. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Moon S, Kim H, Chie E, Kim J and Park C:
Positive impact of radiation dose on disease free survival and
locoregional control in postoperative radiotherapy for squamous
cell carcinoma of esophagus. Dis Esophagus. 22:298–304. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Li CL, Zhang FL, Wang YD, Han C, Sun GG,
Liu Q, Cheng YJ, Jing SW and Yang CR: Characteristics of recurrence
after radical esophagectomy with two-field lymph node dissection
for thoracic esophageal cancer. Oncol Lett. 5:355–359. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kim KH, Chang JS, Cha JH, Lee IJ, Kim DJ,
Cho BC, Park KR and Lee CG: Optimal adjuvant treatment for
curatively resected thoracic esophageal squamous cell carcinoma: A
radiotherapy perspective. Cancer Res Treat. 49:168–177. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Yamashita K, Watanabe M, Mine S, Kurogochi
T, Okamura A, Hayami M and Imamura Y: Patterns and outcomes of
recurrent esophageal cancer after curative esophagectomy. World J
Surg. 41:2337–2344. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wang X, Luo Y, Li M, Yan H, Sun M and Fan
T: Recurrence pattern of squamous cell carcinoma in the midthoracic
esophagus: Implications for the clinical target volume design of
postoperative radiotherapy. Onco Targets Ther. 9:6021–6027. 2016.
View Article : Google Scholar : PubMed/NCBI
|