Introduction
Hepatocellular carcinoma (HCC) is one of the most
life-threatening types of cancer in the world, with >50,000 new
patients being diagnosed every year (1). The majority of patients with HCC are
asymptomatic at the early stage and, due to delayed diagnosis
numerous patients with HCC are unable to undergo radical resection
or transplantation. Therefore, there is an urgent requirement to
examine other potential therapeutics to enhance the outcome in
patients with HCC. Radiation therapy (RT) is commonly used in
patients with HCC who are unable to undergo resection or
transplantation (2). With the
progress of three-dimensional conformal RT and stereotactic body
RT, RT has shown promising results for HCC in clinical trials
(3). However, its effectiveness in
larger tumours, particularly in HCC, is limited by radiosensitivity
due to radiation resistance, and radiation-induced liver disease
(RILD) also limits the efficiency of radiation therapy (4,5).
Therefore, an innovative approach to manage RILD and radiation
resistance is likely to be of significant therapeutic benefit to
patients with HCC and clinical trial treatments. The use of
radiosensitizers is a reasonable method to enhance the
radiosensitivity of RT for HCC.
Among various radiosensitizers investigated for
radiotherapeutic applications, gold nanoparticles (GNPs) have been
investigated more extensively due to their high X-ray absorption
coefficient, and their tuneable size, unique surface chemistry,
electronic properties and low osmolality (6,7). GNPs have
unique physiochemical surface properties, which allow them to be
coated with various antibodies, peptides, proteins, aptamers and
other biomolecules, which facilitates specific targeting to cancer
cells and increased GNP accumulation in the tumour, leading to an
enhanced radiation effect. Using GNPs to enhance the dose of X-ray
radiation absorbed by tumours has attracted increasing attention
(8–10).
Specific delivery can be accomplished by conjugating
GNPs to antibodies or ligands, which target overexpressed proteins
on cancer cell surfaces (11).
Cetuximab (C225), a targeting agent, is a chimeric human-murine
monoclonal antibody, which binds to the extracellular domain of
epidermal growth factor receptor (EGFR) (12). EGFR is an attractive target, which is
overexpressed in a number of human malignancies, including HCC
(13). It was hypothesized that
EGFR-targeted GNPs may enhance the cytotoxic effects of radiation
therapy, and concentrate the effect on targeted tumour cells. EGFR
targeting has been achieved using several immunoconjugates,
including C225 conjugated to GNPs, in several cancer cell lines and
cancer models (14–16). C225-conjugated GNPs are highly stable
in serum. However, few reports are available on the
radiosensitivity of HCC treatment with antibody-functionalised
GNPs, and the exact mechanism by which radiosensitization occurs
remains to be elucidated. To investigate this, the present study
synthesized GNPs coated with C225 (C225-GNPs) to investigate their
radiosensitivity on an EGFR-overexpressing HCC cell line
(SMCC7721). The results demonstrated that the C225-GNPs offer
potential as radiosensitizers for HCC therapy.
Materials and methods
Cell culture
The SMCC7721 cells were cultured in RPMI 1640 medium
(Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA)
supplemented with 10% heat-inactivated foetal calf serum (Gibco;
Thermo Fisher Scientific, Inc.) and 1% penicillin-streptomycin
(Gibco; Thermo Fisher Scientific, Inc.) at 37°C under a humidified
atmosphere containing 5% CO2 and maintained in an
exponential growth state. The cells were passaged and harvested
once every 3 days using 0.25% trypsin.
Preparation and characterization of
C225-GNPs
The synthesis of naked GNPs with a 20-nm diameter
was accomplished using a sodium citrate reduction method. The
preparation method has been described previously (17). The size of the GNPs was determined by
transmission electron microscopy (TEM; JEM-100CX II; JEOL, Ltd.,
Tokyo, Japan) images. The method used for synthesizing the GNP-C225
conjugates was previously performed by El-Sayed et al
(18). Briefly, a solution of GNPs
(50 µg/ml) was diluted with HEPES solution (pH 8.0). The C225 (500
µl) was diluted to a final volume of 5 ml with HEPES solution, and
the mixture was added to 10 ml of the GNP solution; the 15-ml final
volume solution was stirred vigorously at room temperature for 2 h.
The mixture was then centrifuged at 1,500 × g for 30 min at 4°C to
separate the desired GNP-antibody from unconjugated antibody. The
conjugates formed a loose pellet at the bottom of the centrifuge
tube and were carefully collected. The pellet was then diluted in
10 ml PBS (pH 7.8) and vortexed for 10 min. The gold concentration
of the conjugates was determined from the absorbance obtained by
UV-visible spectrometry (Tianjin Gangdong Science and Technology
Development Co., Ltd., Tianjin, China) absorbance at 500 nm (A500).
The GNPs were characterized by TEM following drop-coating 100 µl of
the sample on a carbon-coated copper grid. Dynamic light scattering
spectroscopy (DLS; HORIBA Jobin Yvon, Edison, NJ, USA) allowed for
determination of the hydrodynamic diameter of colloidal particles
and conjugates, which was the diameter of a sphere with the same
Brownian motion as the analysed particle.
Determining the number of C225
molecules bound to a GNP
The number of C225 molecules bound to GNP was
calculated by ELISA (17). Briefly,
100 µl of the standards sample was added to an ELISA plate
according to its sequence, and 200 µl C225-GNP solution was added
to the same micropore. C225 solution (10 µl, 2 µg/µl) was added to
the microplate as a control. All the samples were disposed with 10
µg/µl horseradish peroxidase (HRP)-labelled goat anti-mouse-IgG
(1:2,000; cat no. KGAA36; Nanjing KeyGen Biotech Co., Ltd.,
Nanjing, China) and incubated for 1 h at room temperature. The
C225-GNP conjugates were centrifuged at 1,500 × g at 4°C for 15 min
to remove the unconjugated HRP. Subsequently, tetramethyl benzidine
was reacted with HRP for 15 min, and 2 mol/l sulphuric acid was
added to terminate the reaction. The number of C225 antibodies
bound to C225-GNPs was determined by UV-visible spectrometry at 450
nm, which was compared with the standard curve of the
HRP-anti-IgG/C225. The number of C225 antibodies bound per GNP was
calculated from the total number of C225 antibodies in the solution
divided by the total number of GNPs in the solution (19).
Nanoparticle cytotoxicity assay
Cell Counting Kit-8 (CCK8; Nanjing KeyGen Biotech
Co., Ltd.) assays were used to determine the cytotoxicity of C225,
GNPs and C225-GNPs. The assays were performed according to the
manufacturer's protocol to assess cell viability. The cells were
cultured at a density of 3×103 cells per well in
flat-bottomed 96-well plates. The C225, GNPs and C225-GNPs were
diluted to various concentrations in 1X PBS (Ph 7.4), and then
added into the wells and incubated for 24 h at 37°C, followed by
exposure to 10 µl CCK8, which was added to each well for 2 h. The
absorbance was measured at 490 nm using a microplate reader
(DG3022; Bio-Rad Laboratories, Inc., Hercules, CA, USA) and the 50%
inhibition concentration (IC50) value was estimated.
Cell uptake assay
The SMCC7721 cells were treated with GNPs or
C225-GNPs (the concentrations of GNPs and C225-GNPs was 1/5 of the
IC50 for 24 h) and were centrifuged with 500 × g at 37°C
for 10 min and fixed in 2.5% glutaraldehyde for 4 h at room
temperature, followed by rinsing with PBS twice. The cells were
then gradually dehydrated with 70, 80 and 90% acetone solutions,
and embedded in epoxy resin at 60°C for 48 h. Ultra-thin sections
(70–100 nm) were cut with an ultramicrotome and stained with 5%
uranyl acetate in 50% ethanol, followed by 2% aqueous lead citrate.
Finally, the ultra-thin sections were imaged by TEM at 200 KV.
Flame atomic absorption spectroscopy (FAAS; SpectrAA 140; Agilent
Technologies, Inc., Santa Clara, CA, USA) was used to measure the
gold concentrations of the two groups. Briefly, the SMCC7721 cells
were incubated with GNPs or C225-GNPs for 2 h, the medium was
removed and the cells were washed three times with PBS to remove
excess nanoparticles. The cells were collected and gold
concentrations in the samples were measured by FAAS. The number of
GNPs within the cells was calculated according to the particle
diameter.
Clonogenic assay
To assess clonogenic survival, the SMCC7721 cells
were pretreated with C225, GNPs or C225-GNPs for 24 h, following
which the drugs were removed and the cells were exposed to 0, 1, 2,
4, 6 or 8 Gy X-ray radiation from a medical linear accelerator
(Varan linear accelerator; Unique Medical Systems, Palo Alto, CA,
USA) and incubated for 12 days at 37°C. The visible colonies with
>50 cells were counted and fixed with methanol followed by
trypan blue staining. The cell survival curve was estimated
according to a multitarget single-hit model: y=1-[1-exp (-k *
x)]^N, (y=probability of survival; k=a dose that causes a mean of
one hit per cell, x=number of hits per cell, N=number of targets,
and D0=a dose that causes a mean of one-hit per cell) was
calculated. The sensitization enhancement ratio (SER) was
determined by the ratio of radiation dose resulting in 50% survival
of the cells.
Apoptotic assay
The cells were treated with GNPs, C225 or C225-GNPs
for 24 h followed by X-ray irradiation (2 Gy). Apoptosis was
detected in 5×105 cells washed with PBS by staining with
5 µl Annexin V-APC and 5 µl 7-AAD (Nanjing KeyGen Biotech Co.,
Ltd..). The stained cells were analysed using a flow cytometer.
Western blot analysis
The cells were treated with C225, GNPs or C225-GNPs
for 24 h followed by X-ray irradiation (2 Gy). The cells were lysed
in lysis buffer [20 mM HEPES-NaOH (pH 7.4), 2 mM EGTA, 50 mM
glycerophosphate, 1% Triton X-100, 10% glycerol, 1 mM PMSF, 10
µg/ml leupeptin, 10 µg/ml aprotinin and 10 µg/ml pepstatin]. The
cells were lysed using RIPA buffer (cat no. P0013; Beyotime
Institute of Biotechnology, Shanghai, China), protein lysates (20
µg) were resolved using 10% SDS-PAGE. Protein was quantified using
a micro BCA kit (cat.no. 23235; Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol and analyzed using an
acrylamide gel and then transferred onto a nitrocellulose membrane.
The membrane was probed with specific antibodies against B-cell
lymphoma 2 (Bcl-2; 1:500; cat no. SAB1306605), Bcl-2-associated X
protein (Bax; 1:500; cat no. SAB2108447), caspase-3 (1:500; cat no.
C5737), glucose-regulated protein 78 (GRP78; 1:800; cat no. G9043),
inositol-requiring enzyme (IRE1α; 1:800; cat no. I6785) and
PRKR-like endoplasmic reticulum kinase (PERK; 1:800; cat no.
P7704), all from Sigma; Merck Millipore (Darmstadt, Germany)
diluted with TBST [10 mM Tris-HCl (pH 7.4), 0.1 M NaCl and 0.1%
Tween-20] containing 5% non-fat skim milk overnight at 4°C.
Following washing, the membranes were subsequently incubated with
HRP-conjugated secondary antibodies (IgG; cat. no. KGAA36; Nanjing
KeyGen Biotech Co., Ltd.), followed by application of enhanced
chemiluminescence kits. β-actin was used as an internal
control.
Xenograft assay
Four-week-old specific pathogen-free athymic (T-cell
deficient) nude mice were purchased from the Model Animal Research
Center (Nanjing, China). For the generation of hepatic tumour
models, single-cell suspensions (2×106 cells in 0.1 ml
HBSS) were injected into the right subcutaneous armpit of each nude
mouse. The mice were randomly distributed into four groups (n=5)
following tumour cell implantation. C225-GNPs or GNPs at a gold
concentration of 220 µg/ml were injected into the tumour of each
mouse or 20 mg/kg of C225. For X-ray irradiation, the mice were
exposed to 2.5 Gy of X-rays operating at 6 MV every 3 days. The
total dose and dose rate for each mouse was 20 Gy and 1.23 Gy/min,
respectively. Following X-ray irradiation, the mice were housed in
micro-isolator cages until the time of sacrifice. The room
temperature was maintained between 24 and 26°C, and the relative
humidity was maintained between 60 and 70%. The institutional
laboratory housing provided a 12-h light/dark cycle and nude mice
were allowed ad libitum access to food and water. The
tumours were measured every 3 days with callipers, and the
diameters were recorded. Tumour volume was calculated by the
formula a2b/2, where a and b are the two maximum
diameters. The care and use of animals in the present study was
approved by the Animal Laboratory of Southeast University (Nanjing,
China) and the Use Committee, and conformed to international
guidelines on the ethical use of animals. All efforts were made to
minimize the number of experimental animals and their
suffering.
Statistical analysis
The data are expressed as the mean ± standard
deviation. Significance was evaluated by one-way analysis of
variance or Student's t-test, as appropriate. Statistical
comparisons of slopes were made using the F-test. All statistical
analysis was performed using GraphPad Prism 6 (GraphPad Software,
Inc., La Jolla, CA, USA). P<0.05 was considered to indicate a
statistically significant difference.
Results
Characterization of C225-GNPs
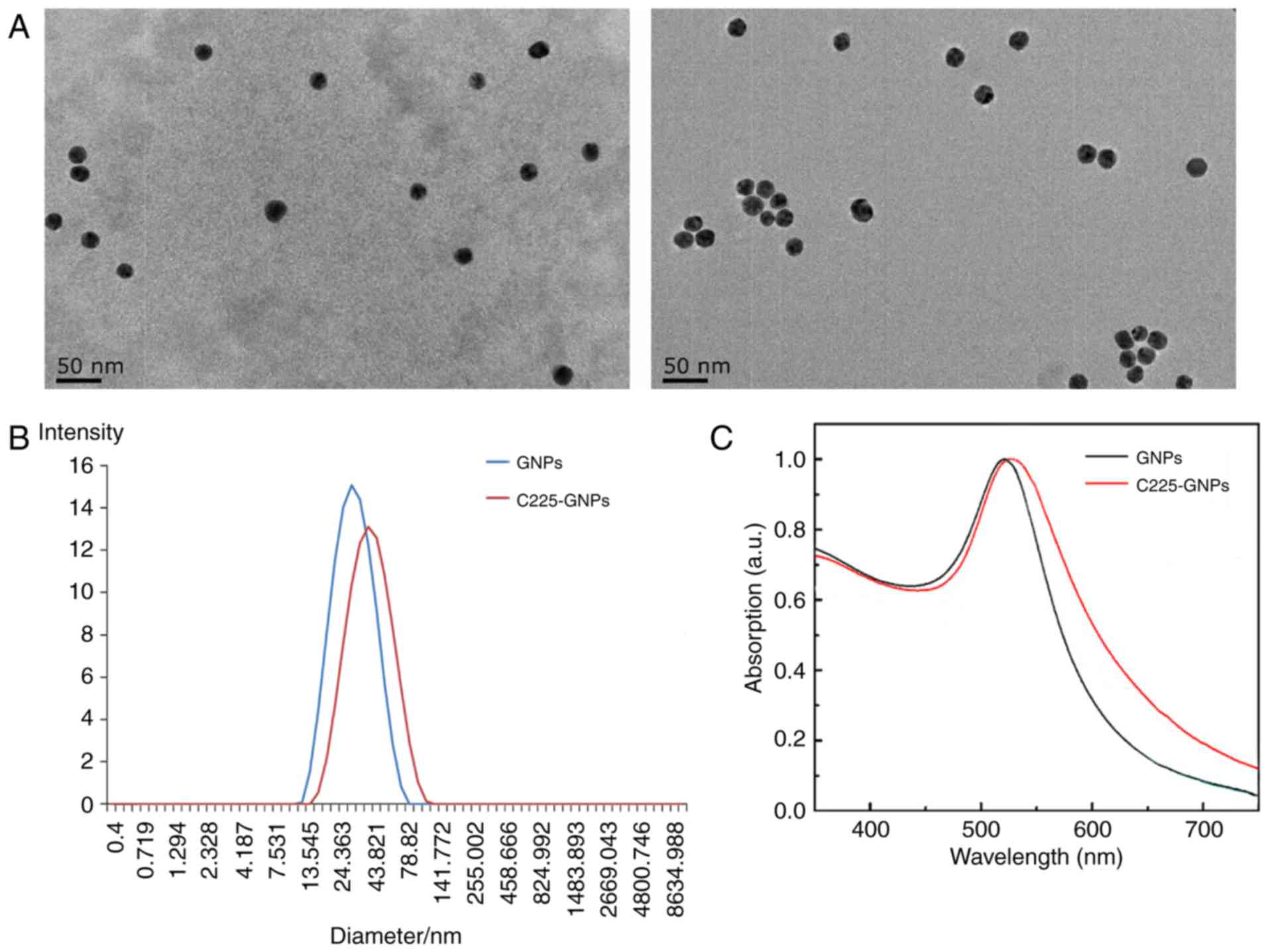
Following coating of the GNPs with C225, the
nanoparticle diameters observed by TEM imaging ranged between 15
and 26 nm (mean diameter of 21.7 nm), which were the same as the
uncoated GNPs (~20.6 nm). However, in contrast to the uncoated
GNPs, C225-GNPs exhibited a thin white ring structure on the
surface (Fig. 1A) when stained with
2% phosphotungstic acid. The average particle size produced was
24.5±0.9 nm for uncoated GNPs, and 41.1±4.4 nm for C225-GNPs, as
determined by DLS (Fig. 1B). The
UV-Vis spectra (Fig. 1C) showed a
marginal redshift in the particles, indicating C225 attachment to
the surface of the GNPs.
Quantity of C225 conjugated to
GNPs
Gradient dilutions of goat anti-mouse IgG coated
with HRP were incubated with C225-GNPs, and the absorbance (OD) was
measured when the substrate was added. The standard curve for
HRP-anti-IgG/C225 was calculated following determining the highest
OD value, and the quantity of C225 in 200 µl of C225-GNP solution
was 2.83 µg; the number of C225 molecules
(A)=0.65×10−6/(152×103)×6.02×1023
(the molecular weight of C225 is 152 kDa). There was 10 µg of gold
nanoparticles in 200 µl of C225-GNPs solution (50 µg/ml),
therefore, the number of gold atoms
(B)=10×10−6/196.966=5.08×10−8 mol=50.8 nmol.
The number of gold atoms (NA) contained in each gold
nanometre was calculated using the following formula:
NA=(59 nm−3) (π/6)
(DMS)3 (20),
NA=59×3.14/6×203=247013.3 A). The number of each
nano-bound C225 antibody was A/(B ×
6.02×1023/NA)=94.65.
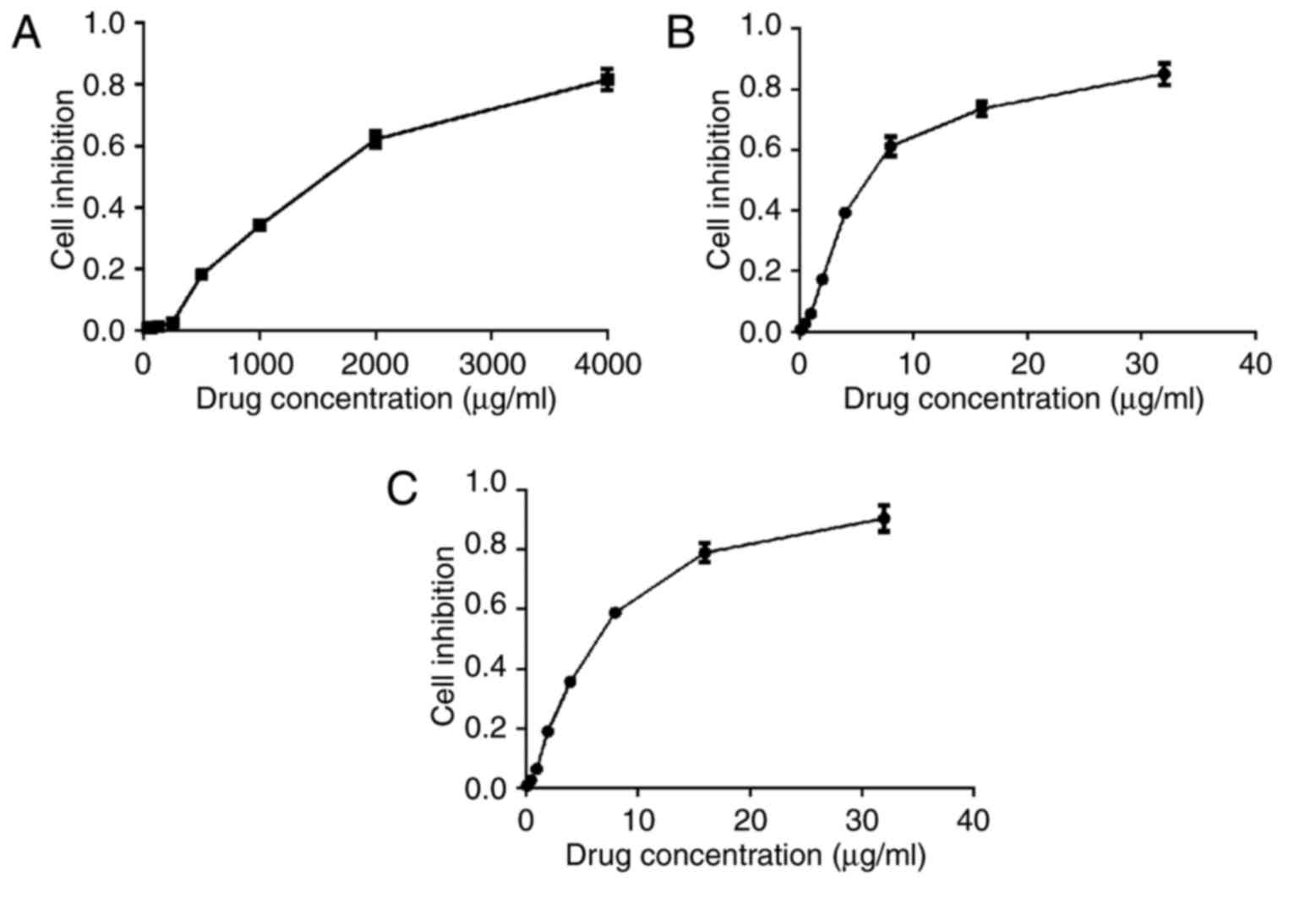
Cell cytotoxicity of C225-GNPs
As shown in Fig.
2A-C., the cell viability of the three groups decreased with an
increasing concentration of drug. There was no significant
difference in cell inhibition rate between GNP-treated cells and
C225-GNP-treated cells at the same drug concentration (P>0.05).
The IC50 values of C225, GNPs and C225-GNPs in SMCC7721
cells were 1,404, 6.14 and 4.11 µg/ml, respectively. One-fifth of
the IC50 value of each drug was used as the experimental
concentration in the follow-up assay.
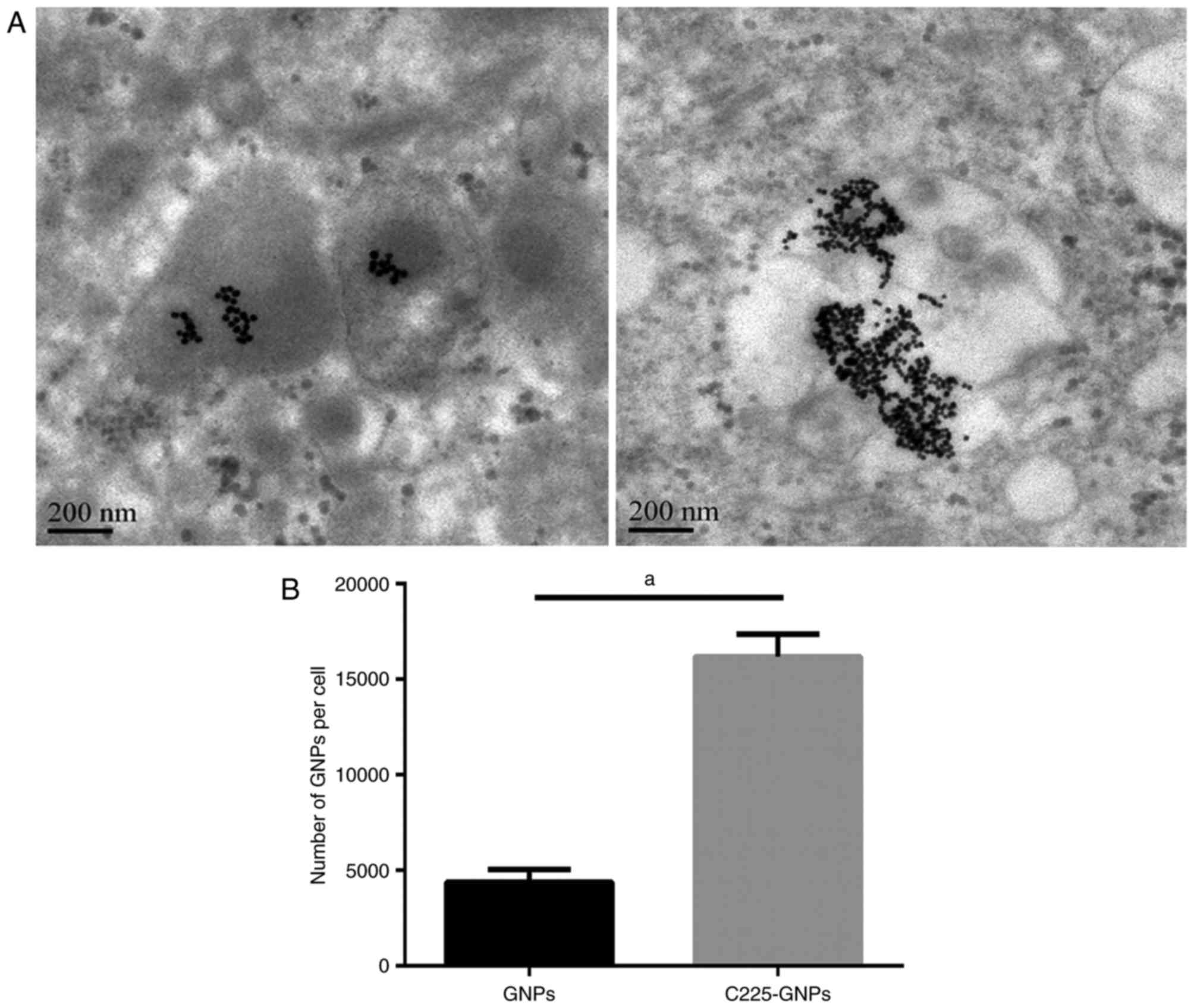
Cell uptake and nanoparticle
distribution
To understand the intracellular uptake by human
pancreatic cancer cell lines expressing EGFR, SMCC7721 cells were
treated with C225-GNPs or with nonspecific isotype control GNPs.
Following treatment of the cells with GNPs or C225-GNPs for 24 h,
TEM was used to observe the distribution and number of GNPs in
cells. The GNPs appeared as dark spots within the cells, and the
majority were located around the ER under the TEM (Fig. 3A). The images further confirmed that
the number of C225-GNPs uptaken by the SMCC7721 cells was markedly
higher, compared with that of GNPs. FAAS was used to quantitatively
determine the quantity of internalised gold in the SMCC7721 cells
of the GNP and C225-GNP groups. The quantity of gold was then
translated to the number of GNPs per single cell and to the total
surface area per single cell. The results showed that more gold was
taken up by cells with C225-GNPs, whereas the GNPs exhibited poor
uptake (P<0.01; Fig. 3B).
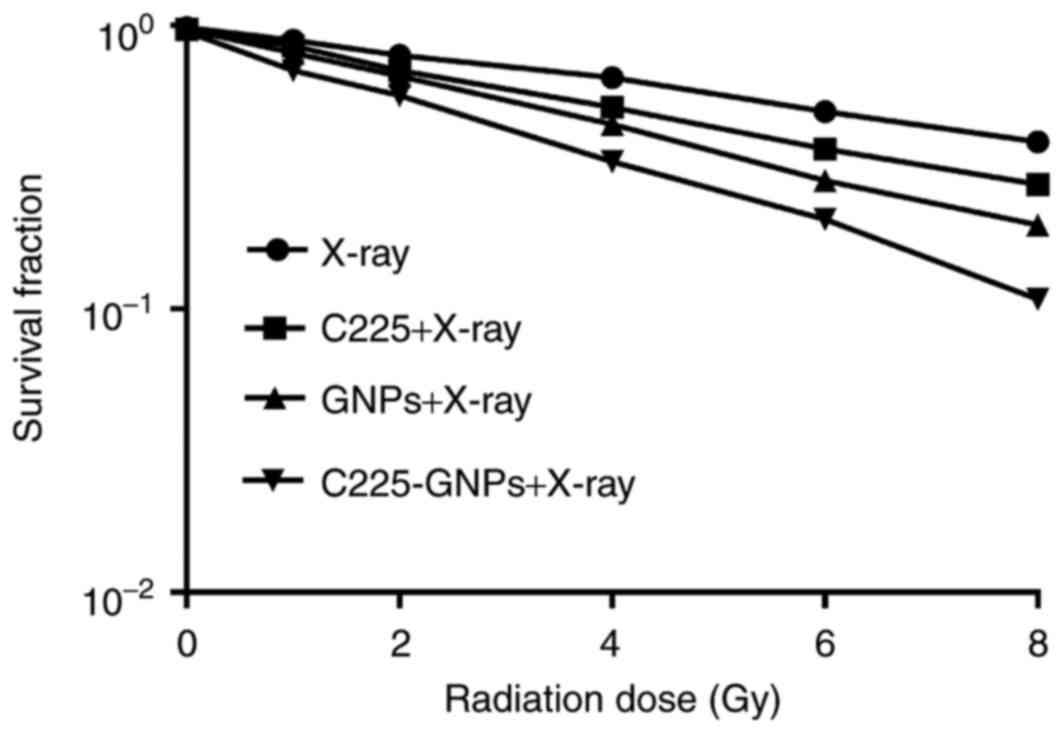
Radiosensitivity of SMCC7721 cells to
C225-GNPs
The clonogenic cell survival assay was accurately
used to determine the enhancement of radiosensitivity by C225, GNPs
and C225-GNPs. The survival curves in Fig. 4 show the radiation enhancement effects
of the three groups on cell survival fractions combined with
different radiation doses of 6 MV X-ray irradiation. There was a
significant separation of the curves compared with that of
radiation alone (P<0.05). The curves showed that C225-GNPs
induced more radiosensitivity than GNPs, according to the fitting
cell survival curve obtained by D0; the SERs of C225, GNPs and
C225-GNPs were 1.35, 1.71 and 2.00, respectively.
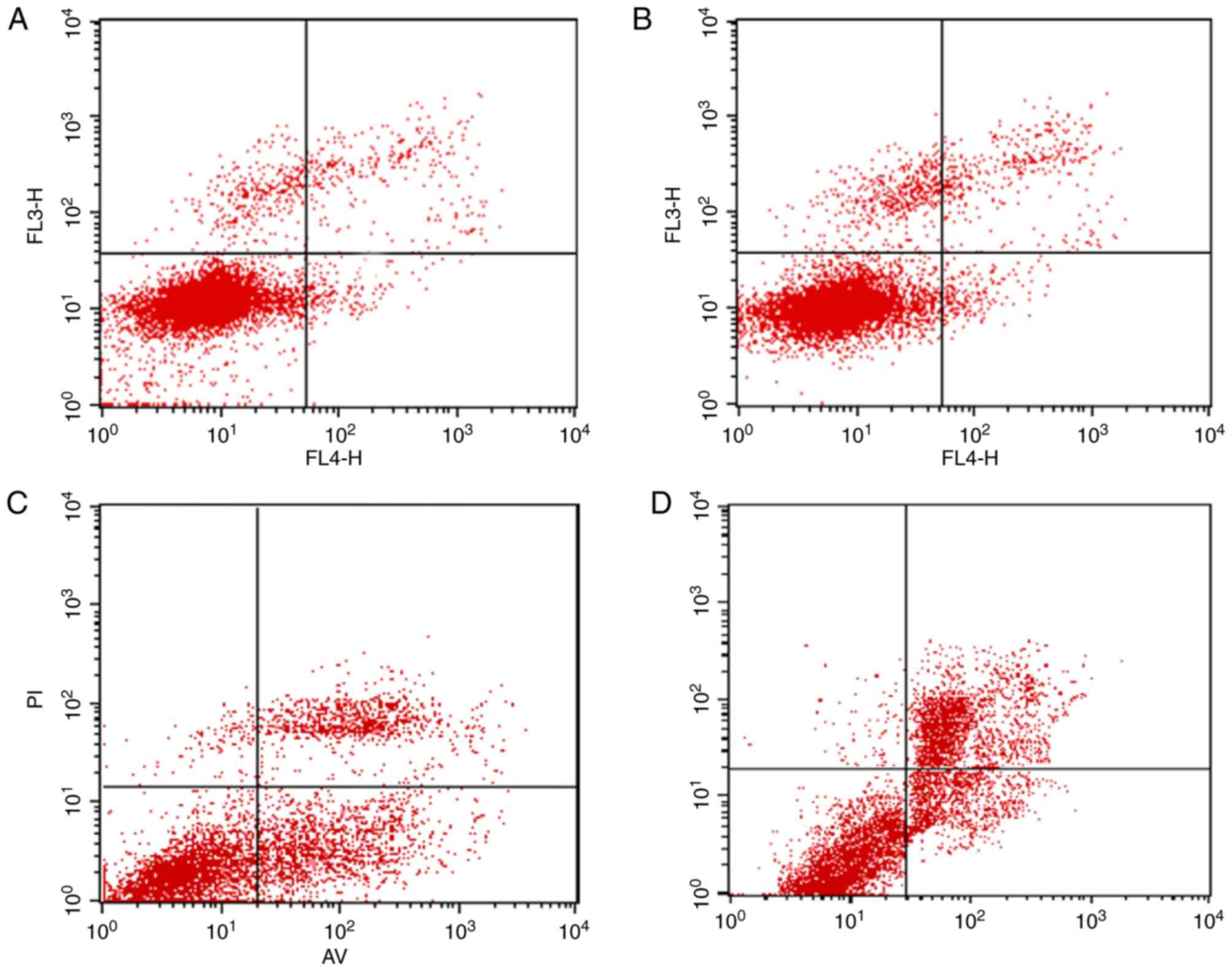
Cell apoptosis assay
The annexin V-FITC/AAD double staining procedure was
used to calculate the modes of cell death. The percentage of
apoptotic cell death, as shown in Fig.
5, is presented as the sum of the percentage of early apoptotic
cells and late apoptotic cells. The apoptotic rate in the control
group induced by 2 Gy X-ray only was 2.07±0.42%, and the percentage
of apoptotic cells in the C225 and GNP groups, which were exposed
to 2 Gy X-ray and drug treated were 5.46±0.60 and 6.46±0.82%,
respectively. However, a significant increase in the percentage of
apoptosis was detected in the C225-GNP group (13.11±1.20%),
compared with that in the C225 and GNP groups, which indicated that
the C225-GNPs induced cellular apoptosis more efficiently
(P<0.05).
Western blot analysis
The expression levels of Bax, Bcl-2, and caspase-3
in cells treated with X-ray alone, C225/X-ray, GNPs/X-ray, or
C225-GNPs/X-ray were measured using western blot analysis (Fig. 6A). The expression levels of Bax and
caspase-3 were upregulated, whereas the expression of Bcl-2 was
downregulated in cells treated with GNPs/X-ray or C225-GNPs/X-ray.
These results indicated that GNPs induced a higher expression of
intracellular apoptotic molecules and significantly inhibited the
expression of anti-apoptotic proteins, compared with the cells
treated with radiation only. The expression of the ERS proteins
PERK, GRP78 and IREα, were also measured in the four groups. The
results showed that the expression of PERK was downregulated in the
cells treated with X-ray alone, C225/X-ray, GNPs/X-ray and
C225-GNPs/X-ray, whereas the expression levels of GRP78 and IREα
were upregulated (Fig. 6B).
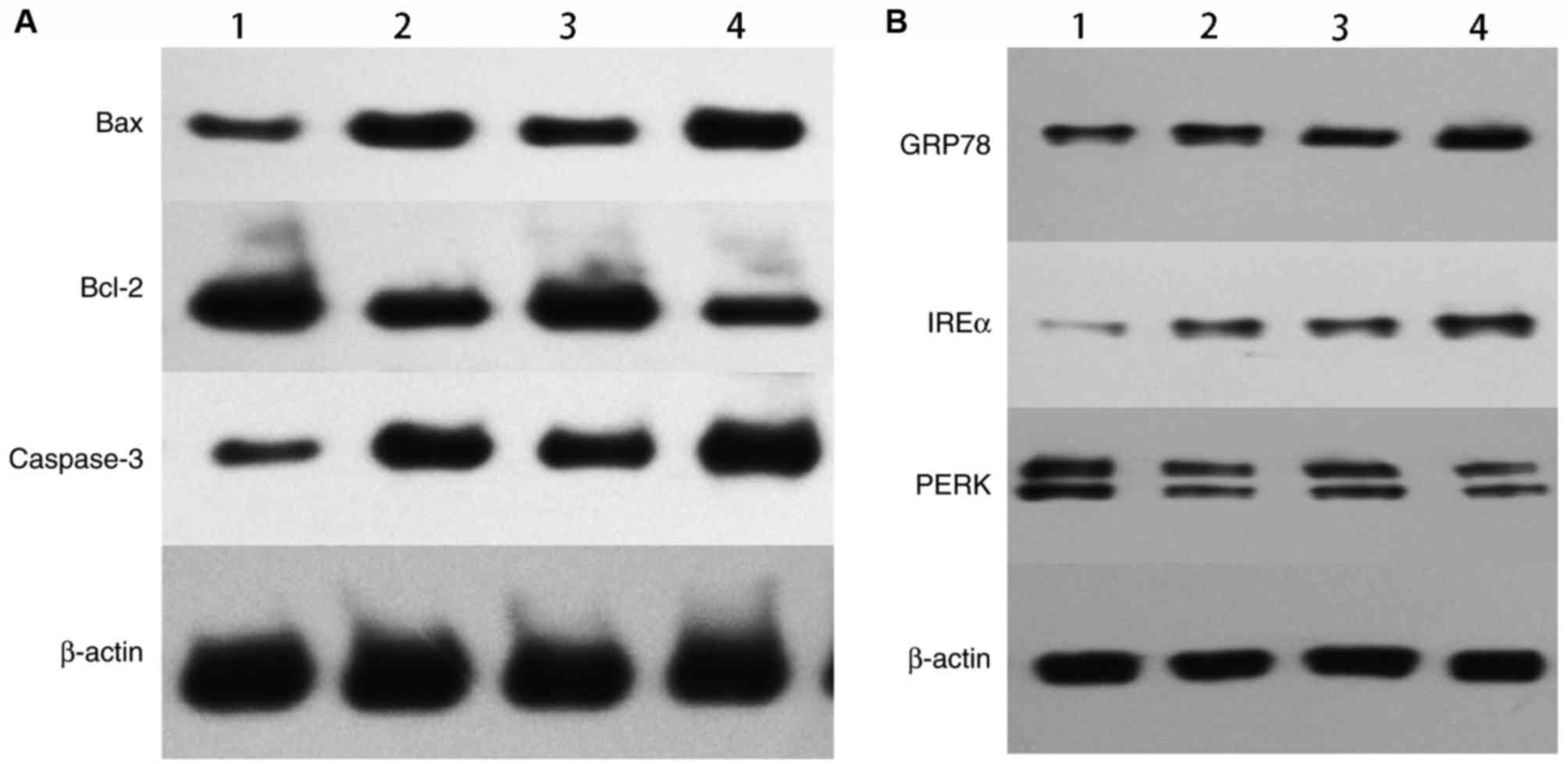 | Figure 6.Protein expression levels of Bax,
Bcl-2, caspase-3, GRP78, IREα, PERK were detected by western blot
analysis. (A) Expression of apoptotic proteins. (B) Expression of
ERS proteins. 1, X-ray; 2, C225/X-ray; 3, GNPs/X-ray; 4,
C225-GNPs/X-ray. Drug concentrations of C225, C225-GNPs and GNPs
were one fifth of each 50% inhibition concentration and the
radiation dose was 2 Gy. GNPs, gold nanoparticles; C225, cetuximab;
Bcl-2, B-cell lymphoma 2; Bax, Bcl-2-associated X protein; GRP78,
glucose-regulated protein 78; IRE1α, inositol-requiring enzyme α;
PERK, PRKR-like endoplasmic reticulum kinase. |
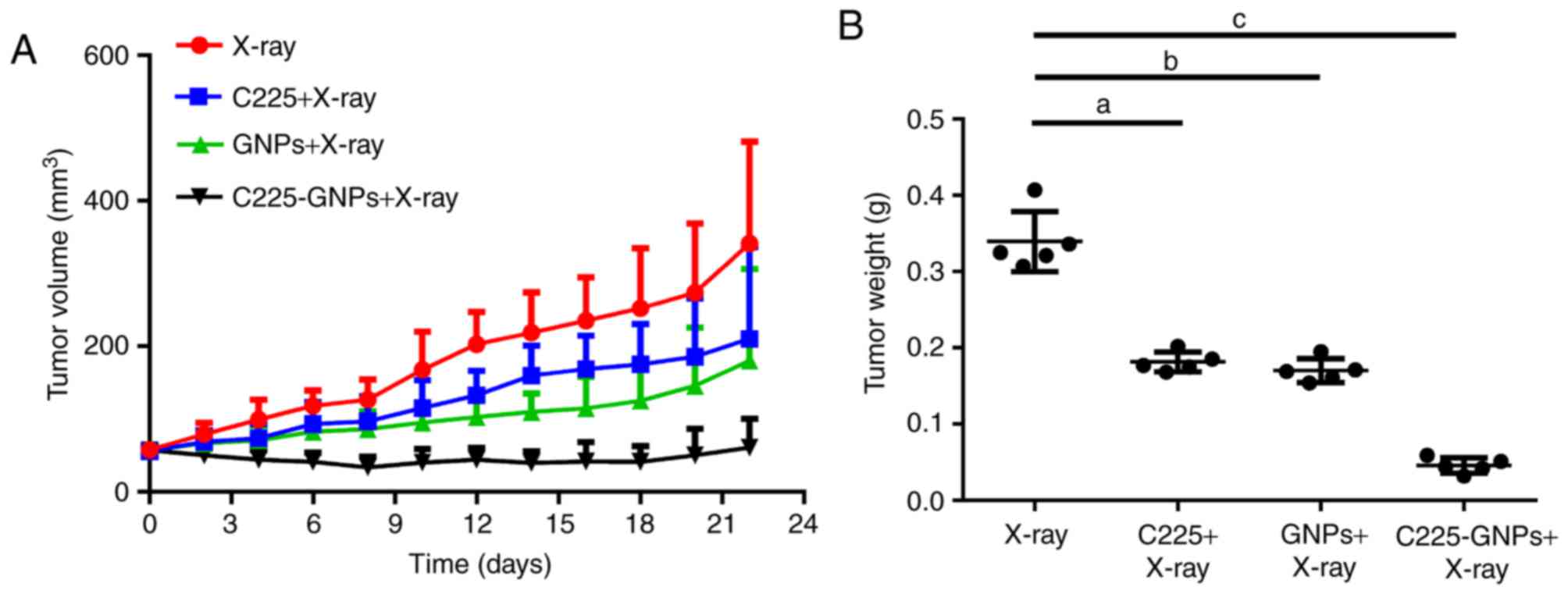
In vivo evaluation of the
radiosensitization properties of C225-GNPs
The average initial tumour volumes prior to
treatment were 57.75±11.29 mm3 (X-ray only), 56.88±10.82
mm3 (C225/X-ray), 55.67±9.91 mm3 (GNPs/X-ray)
and 57.44±8.95 mm3 (C225-GNPs/X-ray). There were no
significant differences in these tumour volumes (P<0.05). Tumour
volumes and tumour weights following X-ray and drug treatments are
shown in Fig. 7. Following exposure
to 20 Gy X-ray radiation, tumour volumes at experimental
termination (day 21) were significantly lower for mice treated with
C225-GNPs/X-ray, compared with X-ray only, C225/X-ray or GNPs/X-ray
(Fig. 7A). Furthermore, the use of
C225-GNPs/X-ray resulted in significantly slower tumour weight
growth, compared with the mice treated with GNPs/X-ray, C225/X-ray
or X-ray only (P<0.01). At the end of the experiment, the tumour
weights of mice treated by X-ray only, GNPs/X-ray, C225/X-ray and
C225-GNPs/X-ray were 0.339±0.039, 0.181±0.013, 0.17±0.016 and
0.046±0.01 g, respectively (Fig.
7B).
Discussion
The targeted delivery of nanomaterials is an
essential area of investigation for nanomedicine. High atomic
number materials, including GNPs, are ideal radiosensitizing agents
due to their distinguished absorption of photons and release of
secondary energy in the form of X-rays into surrounding tissue. One
of the major advantages of GNPs is their formation of stable bonds
with various antibodies, peptides and proteins, which allows more
GNPs to accumulate in the tumour and leads to further enhancement
of the radiation effect on the tumour. The radiosensitivity of
targeted-GNPs to tumour cells leading to increased cell death at KV
or MV X-ray doses has been realised in various cancer cell lines
and animal models (10,14,16–18).
However, few reports are available on HCC with targeted-GNPs
regarding radiosensitivity, and the exact radiosensitization
mechanism induced by GNPs remains to be elucidated.
The present study aimed to assess the effects of
GNP-mediated radiosensitization towards HCC cells in vitro
and in vivo, as tumour xenografts in athymic mice. To
deliver more GNPs to cancer cells and to specifically
radiosensitize them while minimizing side-effects, the present
study selected EGFR-overexpressing SMCC7721 cells and
EGFR-targeting C225 for delivering GNPs to SMCC7721 cells in
vitro and in vivo. When the C225-GNPs conjugates were
synthetized, ELISA-based assays were used to quantify the number of
C225 antibodies coated per GNP. The results showed there was an
average of 94.65 molecules of C225 bound per GNP. As expected, the
cell uptake assay showed that more GNPs conjugates than uncoated
GNPs were accumulated in the cancer cell cytoplasm (P<0.01),
suggesting that C225 significantly increased GNP uptake. The TEM
images showed that the majority of the GNPs were distributed in the
cytoplasm of cells. This was consistent with a previous report
describing the targeting of human pancreatic cancer cell lines with
C225-coated GNPs (14). The
clonogenic cell survival assay is the gold standard for
radiosensitization. The results of the present study showed that
the C225, GNPs or C225-GNPs enhanced the radiosensitization of
SMCC7721 cells to X-ray, and the SER of the C225-GNP-treated group
was the highest due to increased gold uptake by cells. The cell
apoptotic rate was also markedly enhanced by C225-GNPs combined
with X-ray.
Endothelial network stress (ERS) is a novel pathway
of apoptosis, which has been well investigated in previous studies
(21–23). It has a cytoprotective function via
activating the unfolded protein response (UPR). However, if the
cellular damage is too severe or if the stress is excessive, the
UPR stimulates the pro-apoptotic cascade and leads to cell death.
It is suggested that severe induction of the UPR by overloading the
ER capacity enhances cellular sensitivity to radiation or
chemotherapeutic agents. The role of the Bcl-2 family in the ERS
pathway has been reported in previous years (24). The Bcl-2 family is located in
mitochondria but also in the ER and affects its homeostasis.
Bcl-2/Bcl-extra large can inhibit ERS-induced apoptosis, and the
deletion of Bax and Bcl-2-antagonist/killer 1 can protect
ERS-induced apoptosis. The ERS-induced activation of caspase-12,
which is located in the ER epicardium, activates the caspase-9
zymogen. The activated caspase-9 cleaves the caspase-3 zymogen and
eventually leads to the apoptotic cascade.
Cancer cells treated with acriflavine or tunicamycin
have been reported to be sensitized to ionizing radiation through
activation of the UPR (25,26). In the present study, GNPs accumulated
in the cytoplasm and the expression levels of ERS-related proteins
GRP78 and IREα were upregulated. It was also found that the
expression of pro-apoptotic proteins Bax and caspase-3 was
upregulated, whereas the anti-apoptotic protein Bcl-2 was
downregulated in cells. These results suggested that GNPs acted as
an ER-inducer and enhanced X-ray-induced apoptosis. These results
are supported by findings from previous studies that indicated that
GNPs induced ERS and UPR signalling-dependent apoptosis in cancer
cells (27). Yasui et al
(28), also suggested that GNPs
radiosensitized cells by enhancing apoptosis and impairing DNA
repair capacity via ERS induction.
Finally, the radiosensitization efficacy of
C225-GNPs was demonstrated in vivo in an aggressive
orthotopic model of HCC. The in vivo data in the present
study clearly indicated that the GNP-based targeted drug delivery
system showed the most significant radiation enhancement effect in
this xenograft model.
In conclusion, C225-GNP nanoconjugates were
successfully synthesized in the present study, and it was shown
that the nanoparticles significantly improved the efficacy of
radiotherapy in vitro and in vivo, and briefly
discussed the mechanism of radiosensitization in SWCC7721 cells.
The results suggested that C225-GNPs may be used as a potential
radiosensitizer for treating malignant tumours overexpressing EGFR
in radiotherapy.
Acknowledgements
The present study was supported by the Natural
Science Foundation of Jiangsu Province (grant no. BK20141084), the
General Topics of Nanjing Medical Technology Development Project
(grant nos. YKK15141 and YKK15142) and the Key Topics of Nanjing
Medical Technology Development Project (grant no. ZKX13019).
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2017. CA Cancer J Clin. 67:7–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Dhir M, Melin AA, Douaiher J, Lin C, Zhen
WK, Hussain SM, Geschwind JF, Doyle MB, Abou-Alfa GK and Are C: A
review and update of treatment options and controversies in the
management of hepatocellular carcinoma. Ann Surg. 263:1112–1125.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Schlachterman A, Craft WW Jr, Hilgenfeldt
E, Mitra A and Cabrera R: Current and future treatments for
hepatocellular carcinoma. World J Gastroenterol. 21:8478–8491.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tanguturi SK, Wo JY, Zhu AX, Dawson LA and
Hong TS: Radiation therapy for liver tumors: Ready for inclusion in
guidelines? Oncologist. 19:868–879. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tang WY, Chau SP, Tsang WP, Kong SK and
Kwok TT: The role of Raf-1 in radiation resistance of human
hepatocellular carcinoma Hep G2 cells. Oncol Rep. 12:1349–1354.
2004.PubMed/NCBI
|
|
6
|
Kim D, Park S, Lee JH, Jeong YY and Jon S:
Antibiofouling polymer-coated gold nanoparticles as a contrast
agent for in vivo X-ray computed tomography imaging. J Am Chem Soc.
129:7661–7665. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Daniel MC and Astruc D: Gold
nanoparticles: Assembly, supramolecular chemistry,
quantum-size-related properties, and applications toward biology,
catalysis, and nanotechnology. Chem Rev. 104:293–346. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Berrezoug A, Dib ASA and Belbachir AH:
Enhanced X-ray absorption by using gold nanoparticles in a
biological tissue. Radioprotection. 50:281–285. 2015. View Article : Google Scholar
|
|
9
|
Mesbahi A: A review on gold nanoparticles
radiosensitization effect in radiation therapy of cancer. Rep Pract
Oncol Radiother. 15:176–180. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhu CD, Zheng Q, Wang LX, Xu HF, Tong JL,
Zhang QA, Wan T and Wu JQ: Synthesis of novel galactose
functionalized gold nanoparticles and its radiosensitizing
mechanism. J Nanobiotechnol. 13:672015. View Article : Google Scholar
|
|
11
|
Yao CP, Zhang LW, Wang J, He Y, Xin J,
Wang S, Xu H and Zhang Z: Gold nanoparticle mediated phototherapy
for cancer. J Nanomater. 2016:Article ID 54971362016. View Article : Google Scholar
|
|
12
|
Martinelli E, De Palma R, Orditura M, De
Vita F and Ciardiello F: Anti-epidermal growth factor receptor
monoclonal antibodies in cancer therapy. Clin Exp Immunol. 158:1–9.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sogawa C, Tsuji AB, Yoshida C, Inubushi M,
Furukawa T, Koizumi M, Akahori Y, Ukai Y, Kurosawa G, Kurosawa Y
and Saga T: Novel human monoclonal antibody against epidermal
growth factor receptor as an imaging probe for hepatocellular
carcinoma. Nucl Med Commun. 33:719–725. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Khan JA, Kudgus RA, Szabolcs A, Dutta S,
Wang E, Cao S, Curran GL, Shah V, Curley S, Mukhopadhyay D, et al:
Designing nanoconjugates to effectively target pancreatic cancer
cells in vitro and in vivo. PLoS One. 6:e203472011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Dreifuss T, Betzer O, Shilo M, Popovtzer
A, Motiei M and Popovtzer R: A challenge for theranostics: Is the
optimal particle for therapy also optimal for diagnostics?
Nanoscale. 7:15175–15184. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Popovtzer A, Mizrachi A, Motiei M,
Bragilovski D, Lubimov L, Levi M, Hilly O, Ben-Aharon I and
Popovtzer R: Actively targeted gold nanoparticles as novel
radiosensitizer agents: An in vivo head and neck cancer model.
Nanoscale. 8:2678–2685. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Day ES, Bickford LR, Slater JH, Riggall
NS, Drezek RA and West JL: Antibody-conjugated gold-gold sulfide
nanoparticles as multifunctional agents for imaging and therapy of
breast cancer. Int J Nanomedicine. 5:445–454. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
El-Sayed IH, Huang X and El-Sayed MA:
Surface plasmon resonance scattering and absorption of anti-EGFR
antibody conjugated gold nanoparticles in cancer diagnostics:
Applications in oral cancer. Nano Lett. 5:829–834. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Averitt RD, Westcott SL and Halas NJ:
Linear optical properties of gold nanoshells. J Opt Soc Am B.
16:1824–1832. 1999. View Article : Google Scholar
|
|
20
|
Alvarez MM, Khoury JT, Schaaff TG,
Shafigullin MN, Vezmar I and Whetten RL: Optical absorption spectra
of nanocrystal gold molecules. J Phys Chem B. 101:3706–3712. 1997.
View Article : Google Scholar
|
|
21
|
Ma YJ and Hendershot LM: The role of the
unfolded protein response in tumour development: Friend or foe? Nat
Rev Cancer. 4:966–977. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dejeans N, Barroso K, Fernandez-Zapico ME,
Samali A and Chevet E: Novel roles of the unfolded protein response
in the control of tumor development and aggressiveness. Semin
Cancer Biol. 33:67–73. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Galmiche A, Sauzay C, Chevet E and Pluquet
O: Role of the unfolded protein response in tumor cell
characteristics and cancer outcome. Curr Opin Oncol. 29:41–47.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kuwana T and Newmeyer DD: Bcl-2-family
proteins and the role of mitochondria in apoptosis. Curr Opin Cell
Biol. 15:691–699. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Contessa JN, Bhojani MS, Freeze HH,
Rehemtulla A and Lawrence TS: Inhibition of N-linked glycosylation
disrupts receptor tyrosine kinase signaling in tumor cells. Cancer
Res. 68:3803–3809. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lim MJ, Ahn JY, Han Y, Yu CH, Kim MH, Lee
SLO, Lim DS and Song JY: Acriflavine enhances radiosensitivity of
colon cancer cells through endoplasmic reticulum stress-mediated
apoptosis. Int J Biochem Cell Biol. 44:1214–1222. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tsai YY, Huang YH, Chao YL, Hu KY, Chin
LT, Chou SH, Hour AL, Yao YD, Tu CS, Liang YJ, et al:
Identification of the nanogold particle-induced endoplasmic
reticulum stress by omic techniques and systems biology analysis.
ACS Nano. 5:9354–9369. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yasui H, Takeuchi R, Nagane M, Meike S,
Nakamura Y, Yamamori T, Ikenaka Y, Kon Y, Murotani H, Oishi M, et
al: Radiosensitization of tumor cells through endoplasmic reticulum
stress induced by PEGylated nanogel containing gold nanoparticles.
Cancer Lett. 347:151–158. 2014. View Article : Google Scholar : PubMed/NCBI
|