Introduction
Lung cancer is the most common cause of
cancer-associated mortality worldwide. Non-small cell lung cancer
(NSCLC) accounts for >85% of lung cancer cases, with squamous
cell carcinoma and adenocarcinoma being the predominant subtypes
(1). Patients with early-stage NSCLC
typically undergo surgical therapy as it is currently the most
effective form of treatment; however, 75% of patients with NSCLC
present with an advanced disease stage at the time of diagnosis
(2). Despite developments in
diagnostic and therapeutic techniques, overall prognosis is
unsatisfactory as the overall 5-year survival rate is ~15%
(3). The tumor-node-metastasis (TNM)
staging system (4), according to
histopathologic findings, lacks sufficient predictive value as
significant differences in survival are often observed for the same
TNM stage. Therefore, a combination of a number of biomarkers
including p53, p21, Ki-67, KRAS, and cyclin D1, to more accurately
distinguish patients with NSCLC with poor survival would be
valuable (5,6).
Signal transduction and activators of transcription
factor (STAT)3, a member of the STAT family, is frequently regarded
as an oncogene (7). STAT3 can be
activated via phosphorylation events within the Janus kinase-STAT
and/or Ras-mitogen-activated protein kinase signaling pathways,
producing tyrosine or serine phosphorylated STAT3 (p-STAT3)
(8). p-STAT3 is able to increase the
expression levels of certain target genes, including vascular
endothelial growth factor and mucin 1 (MUC1) (9,10). STAT3
may serve as one of the oncogenic critical factors in NSCLC.
MUC1 is a transmembrane glycoprotein (11). In normal tissues, it is present on the
apical surface of normal glandular epithelial cells (12). In certain types of cancer tissue, MUC1
expression is upregulated and presented on the whole cell surface
(13). MUC1 may serve an important
function in tumor development and progression and is associated
with a poor prognosis in certain types of cancer.
A limited number of studies have previously
demonstrated the prognostic value of STAT3 and MUC1 in NSCLC.
Therefore, the present study investigated the association between
STAT3 expression, MUC1 expression and the clinical features of
patients with NSCLC. Furthermore, the present study also
investigated the potential prognostic value of using STAT3 and MUC1
expression levels to predict the survival rate of patients with
NSCLC using univariate and multivariate analysis.
Materials and methods
Patients
The present study was approved by the Shandong
University Ethics Committee (Shandong, China). In total, 106
consecutive patients with NSCLC who attended the Department of
Thoracic Surgery, Jinan Central Hospital Affiliated to Shandong
University (Shandong, China) and the Department of Thoracic Surgery
East Ward, Provincial Hospital Affiliated to Shandong University
(Shandong, China) between September 2011 and December 2011 were
enrolled into this retrospective study. A total of 98 patients
remained subsequent to use of the following inclusion criteria: i)
Patients underwent a radical excision (lobectomy or pneumonectomy
with regional lymph node dissection) and received confirmation of
squamous cell carcinoma or adenocarcinoma via pathological analysis
following surgery; ii) patients were diagnosed with stage I–IIIa
NSCLC, according to the TNM staging system outlined by the
International Union Against Cancer (2009); iii) patients accepted
no pre-surgical radiotherapy, or chemotherapy and had no surgical
contraindications; and iv) follow-up notes were well preserved.
Patients who had incomplete or lost follow-up were excluded. The
ages of the patients enrolled in this study ranged from 47 to 78
years (mean, 63.7 years). A total of 55 patients were men and 43
were women. A total of 52 patients had adenocarcinoma and 46 had
squamous cell carcinoma. A total of 68 patients had lymph node
metastasis and 30 patients did not have lymph node metastasis. A
total of 24 patients had stage I disease, 52 patients had stage II
disease and 22 patients had stage IIIa disease. Table I outlines patient clinicopathological
characteristics.
 | Table I.Association between STAT3, p-STAT3 and
MUC1 expression and clinical features of 98 patients with
NSCLC. |
Table I.
Association between STAT3, p-STAT3 and
MUC1 expression and clinical features of 98 patients with
NSCLC.
|
|
| STAT3 | p-STAT3 | MUC1 |
|---|
|
|
|
|
|
|
|---|
| Clinical
features | No. of patients | −, n | +, n | P-value | −, n | +, n | P-value | −, n | +, n | P-value |
|---|
| Total | 98 | 16 | 82 |
| 47 | 51 |
| 37 | 61 |
|
| Sex |
|
|
| 0.596a |
|
| 0.158 |
|
| 0.835 |
| Male | 55 | 8 | 47 |
| 30 | 25 |
| 20 | 35 |
|
|
Female | 43 | 8 | 35 |
| 17 | 26 |
| 17 | 26 |
|
| Age, years |
|
|
| 0.176a |
|
| 0.842 |
|
| 0.682 |
|
<60 | 45 | 10 | 35 |
| 21 | 24 |
| 17 | 28 |
|
| ≥60 | 53 | 6 | 47 |
| 26 | 27 |
| 20 | 33 |
|
| Tumor location |
|
|
| 1.000a |
|
| 0.539 |
|
| 0.397 |
|
Central | 39 | 6 | 33 |
| 17 | 22 |
| 17 | 22 |
|
|
Peripheral | 59 | 10 | 49 |
| 30 | 29 |
| 20 | 39 |
|
| Histological
type |
|
|
| 1.000a |
|
| 0.005 |
|
| 0.036 |
|
SCC | 52 | 9 | 43 |
| 32 | 20 |
| 25 | 27 |
|
|
ADC | 46 | 7 | 39 |
| 15 | 31 |
| 12 | 34 |
|
|
Differentiation |
|
|
|
>0.05a |
|
|
>0.05a |
|
|
>0.05a |
|
Well | 15 | 1 | 14 |
| 10 | 5 |
| 8 | 7 |
|
|
Moderately | 51 | 12 | 39 |
| 23 | 28 |
| 16 | 35 |
|
|
Poorly | 32 | 3 | 29 |
| 14 | 18 |
| 13 | 19 |
|
| pT |
|
|
|
>0.05a |
|
|
>0.05a |
|
|
<0.05a |
|
pT1 | 11 | 2 | 9 |
| 6 | 5 |
| 8 | 3 |
|
|
pT2 | 73 | 14 | 59 |
| 35 | 38 |
| 26 | 47 |
|
|
pT3 | 14 | 0 | 14 |
| 6 | 8 |
| 3 | 11 |
|
| pN |
|
|
| 0.08a |
|
| 0.001a |
|
| 0.001a |
| − | 30 | 8 | 22 |
| 23 | 7 |
| 19 | 11 |
|
| + | 68 | 8 | 60 |
| 24 | 44 |
| 18 | 50 |
|
| pTNM |
|
|
|
<0.01a |
|
|
<0.05a |
|
|
<0.01a |
| pI | 24 | 8 | 16 |
| 17 | 7 |
| 17 | 7 |
|
|
pII | 52 | 8 | 44 |
| 23 | 29 |
| 15 | 37 |
|
|
pIIIa | 22 | 0 | 22 |
| 7 | 15 |
| 4 | 17 |
|
Immunohistochemical (IHC)
analysis
IHC staining for STAT3, p-STAT3 and MUC1 protein
were detected using the streptavidin-peroxidase method. The
experimental specimens were included in the 98 cancer tissue
specimens obtained from the 98 patients with NSCLC. Adjacent
non-tumorous lung tissues were used as the control tissues. The
experimental specimens were fixed in 10% neutral buffered formalin
(cat no. M004; Shanghai GeFan Biotechnology Company, Ltd.,
Shanghai, China) immediately following surgery at room temperature
for no more than 24 h. Each 4-µm-thick section was deparaffinised,
rehydrated and incubated with fresh 0.3% H2O2
in methanol for 30 min at room temperature to block endogenous
peroxidase activity. Following rehydration through a graded series
of ethanol concentrations (95% 5 min, 80% 5 min, 75% 5 min) at room
temperature, the sections were autoclaved in 10 mM citrate buffer
(pH 6.0) at 120°C for 3 min and then cooled to 30°C. Following
rinsing with 0.1 M phosphate-buffered saline (PBS; pH 7.4),
nonspecific binding sites were blocked by incubation with 10%
normal goat serum (cat no. WE0387-KPO; Beijing Baiao Lai Bo
Technology Co., Ltd., Beijing, China, dilution, 1:100) for 30 min
at room temperature. Sections were then incubated at 4°C overnight
with the primary rabbit antibody against human STAT3 (cat no.
CAU29097; 1:100; Spring Bioscience Corporation, Pleasanton, CA,
USA), rabbit antibody against human p-STAT3 (cat no. sc-7993
tyr705; 1:100; Santa Cruz Biotechnology, Inc., Dallas, TX, USA,
dilution 1:100) and rabbit antibody against human MUC1 (cat no.
bs-1018R; 1:500; Bo Ao Sen Biotechnology Co., Ltd., Beijing, China)
in PBS containing 1% bovine serum albumin. The sections were then
washed with PBS and incubated with biotinylated peroxidase
labelling sheep anti-rabbit immunoglobulin G (cat no. A100970;
1:400; Nanjing Long Kwai Biological Technology Co., Ltd., Nanjing,
China) for 30 min at room temperature, and incubated with
streptavidin-biotin peroxidase complex solution for 30 min at room
temperature. The chromogen, 3,3′-diaminobenzidine
tetrahydrochloride, was applied as a 0.02% solution containing
0.005% H2O2 in 50 mM ammonium acetate-citrate
acid buffer (pH 6.0). Finally, the sections were lightly
counterstained with Mayer's haematoxylin for 30 min and mounted at
room temperature. Specimens were visualized using the Envision
System (Dako; Agilent Technologies, Inc., Santa Clara, CA, USA) and
an Olympus optical microscope (magnification, ×200). STAT3, p-STAT3
and MUC1 expression levels were measured using the IHC scoring
system as described previously (14).
Briefly, a score of 3 indicated that >50% of the cells exhibited
mild to moderate staining intensity, or >20% of cells exhibited
strong staining intensity; a score of 2 indicated that 20–50% of
cells demonstrated mild to moderate staining intensity, or 20% of
cells exhibited strong staining intensity; a score of 1 indicated
that <20% of cells demonstrated mild to moderate staining
intensity; and a score of 0 indicated that no staining was present.
A score of ≥2 demonstrated positive expression.
Follow-up
In total, 74 patients received chemotherapy, 25
patients received postsurgical radiotherapy and 20 patients
received epidermal growth factor receptor-tyrosine kinase inhibitor
therapy. Patients were examined every 3 or 4 months during the
first 3 years. During each follow-up visit the patient underwent a
thorough physical examination, chest computed tomography (CT),
brain CT or magnetic resonance imaging, abdomen ultrasonography or
CT. In total, 6 patients underwent positron emission tomography
combined with CT examination. The location and time of tumor
relapse were recorded. Patients who succumbed to mortality due to
the tumor were included in the prognostic analysis.
Statistical analysis
Enumeration data were analyzed using the
χ2 test or Fisher's exact probability test. Spearman's
rank correlation was performed to analyze correlations between
STAT3, p-STAT3 and MUC1 expression. Univariate analysis was
conducted using the Kaplan-Meier estimator curve method, and the
log-rank test was used to calculate the survival rates. Cox
regression multivariate analysis was performed in order to
determine prognostic factors. All statistical data were analyzed
using SPSS (version 13; SPSS, Inc., Chicago, IL, USA). P<0.05
was considered to indicate a statistically significant
difference.
Results
Association between STAT3, p-STAT3 and
MUC1 expression levels and clinical characteristics
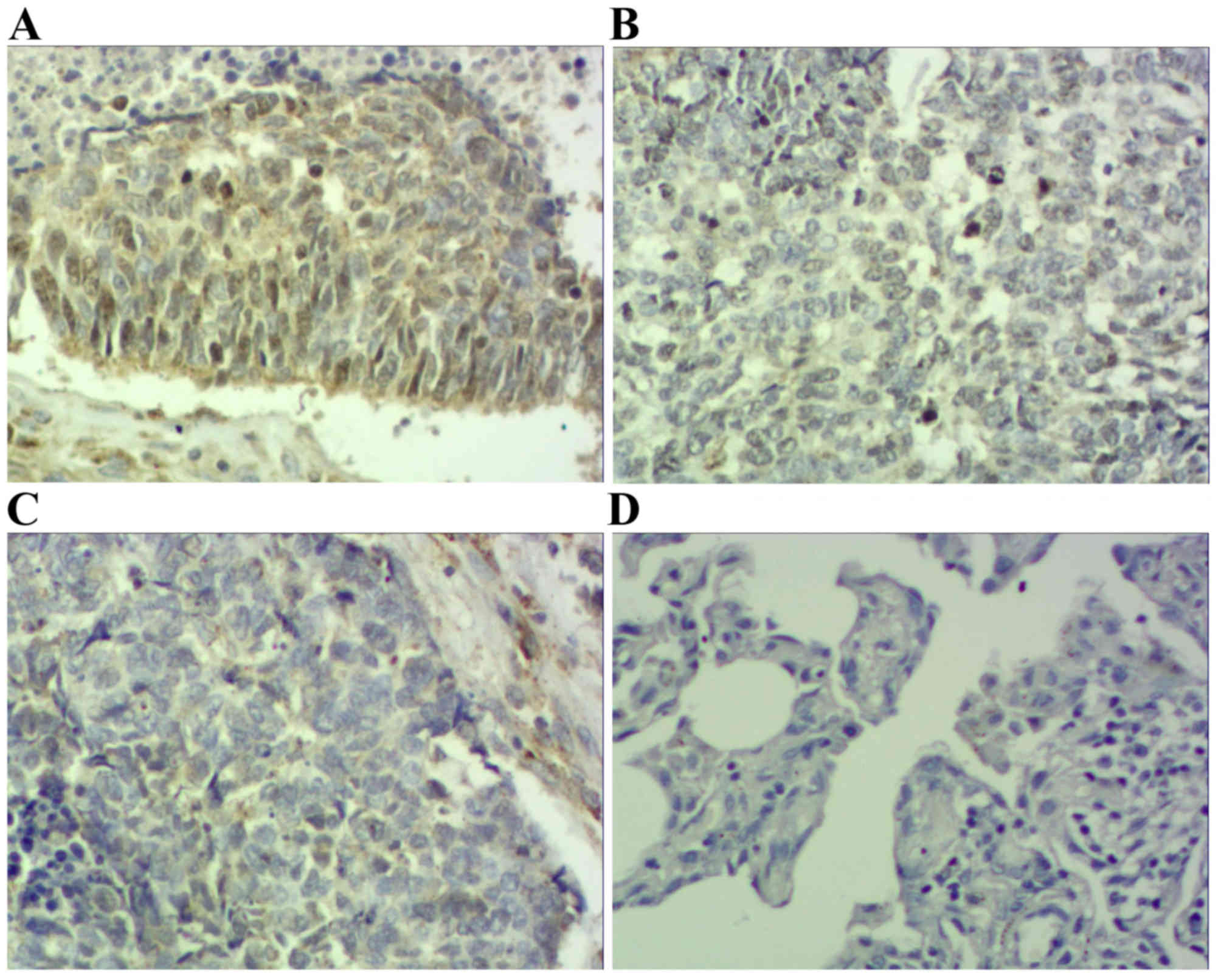
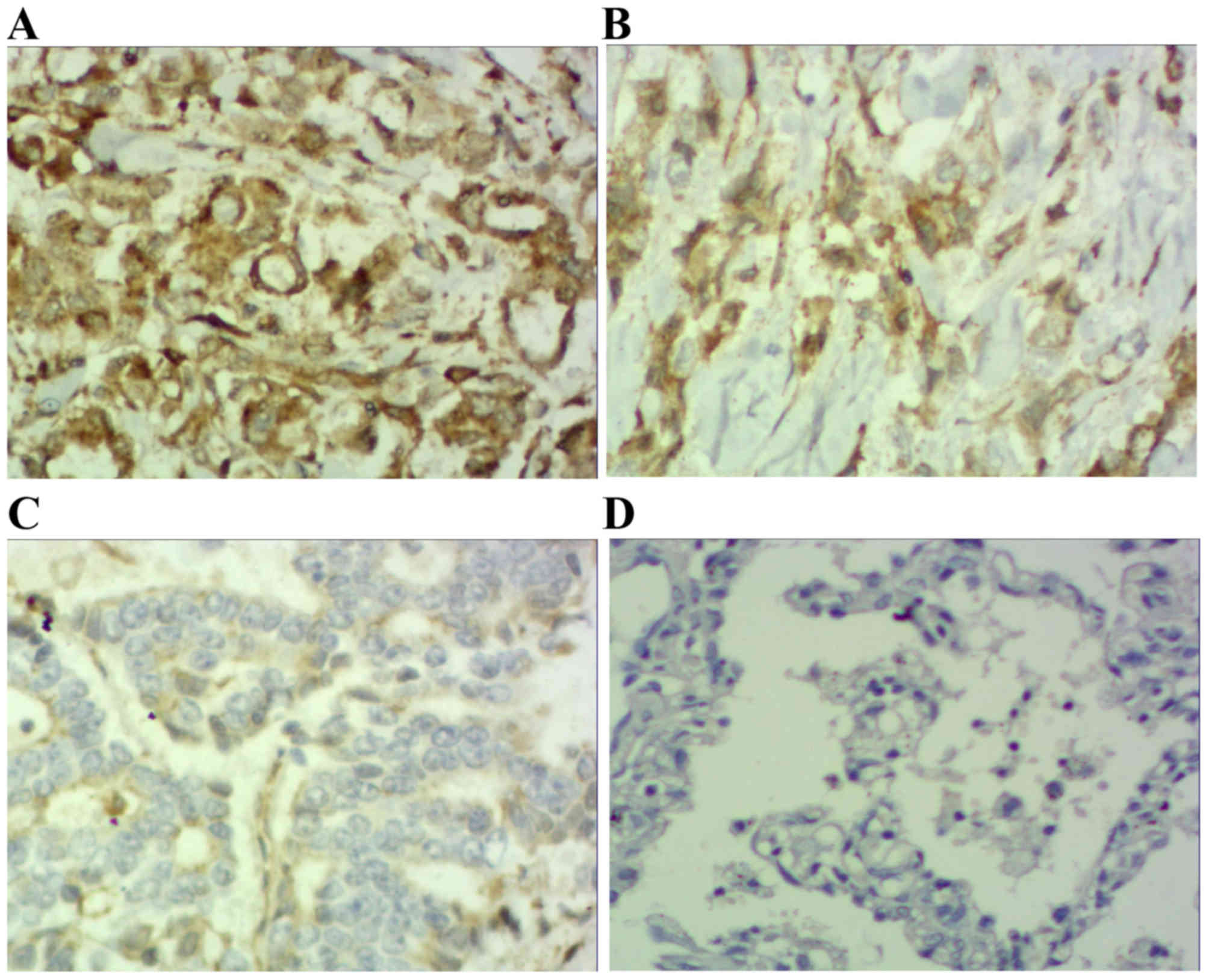
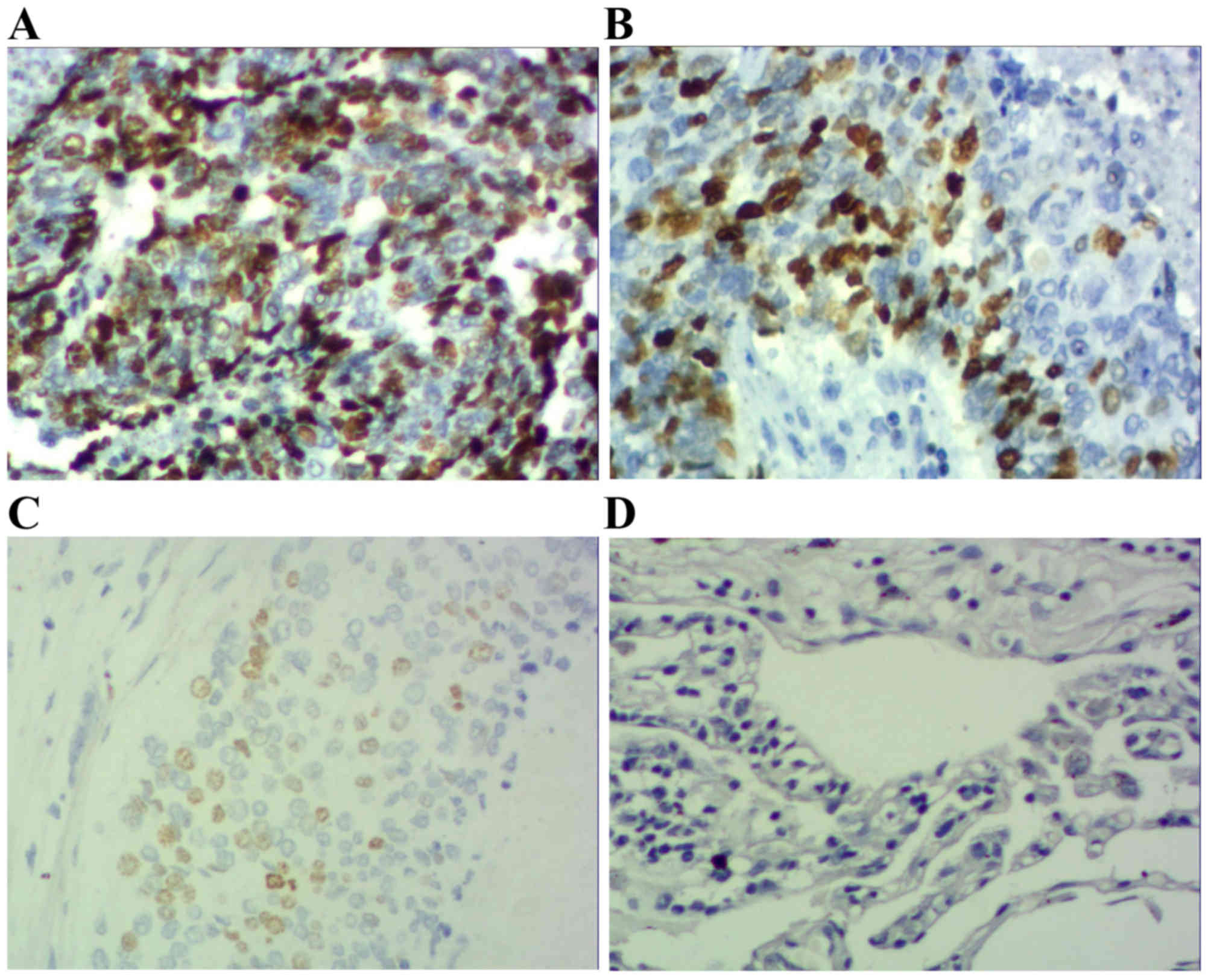
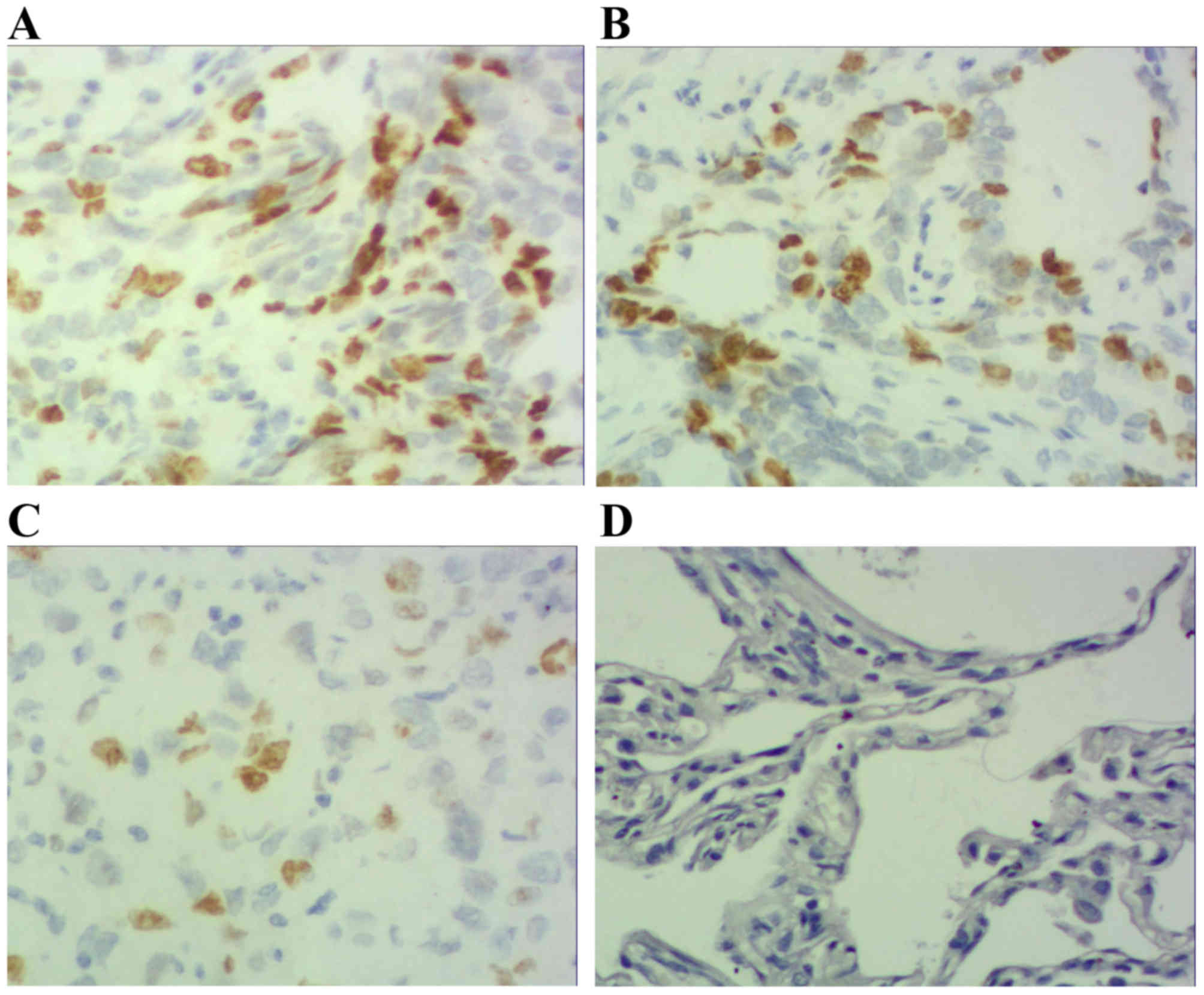
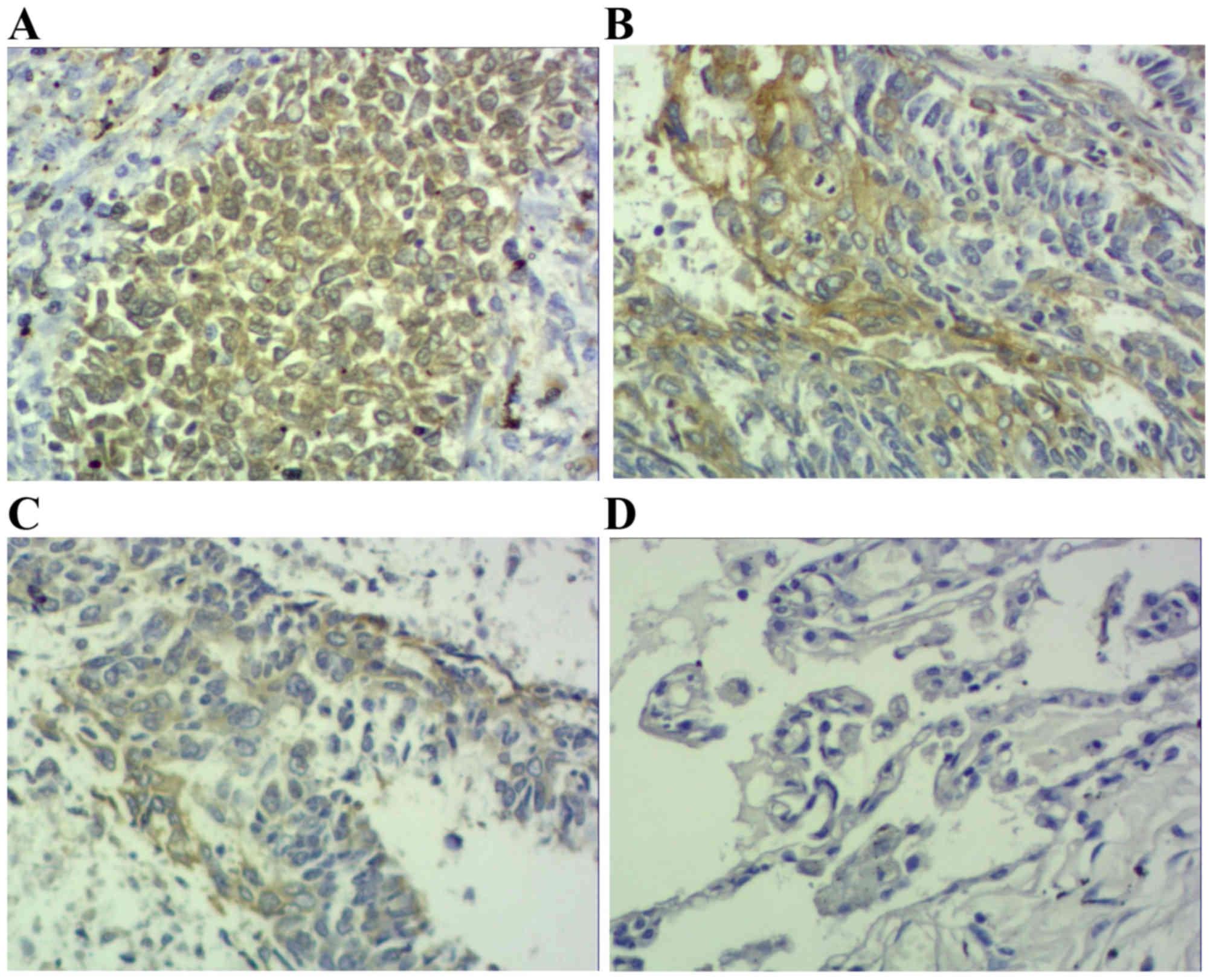
Results demonstrated that positive STAT3 expression
was located within the cytoplasm and nucleus (Figs. 1 and 2),
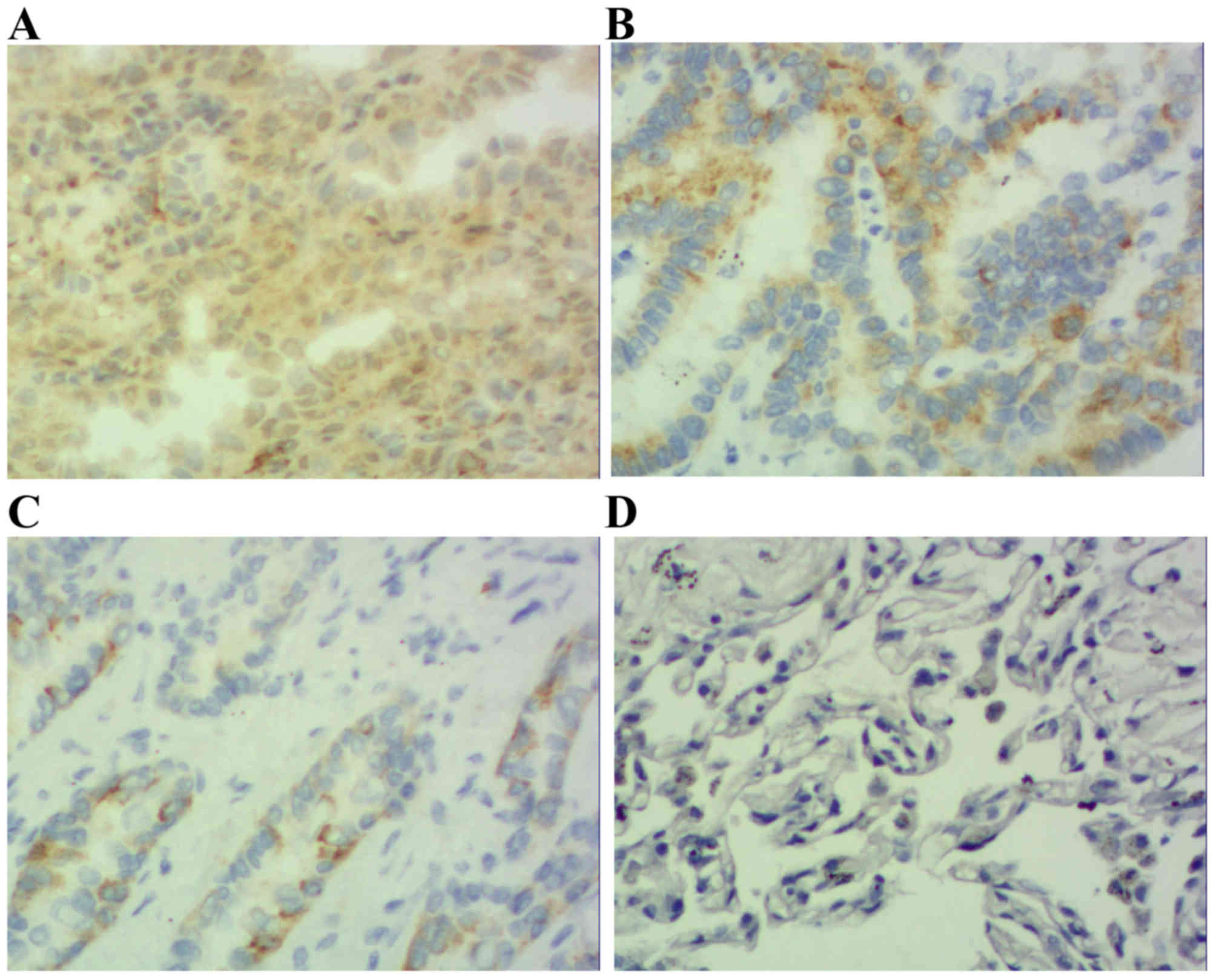
positive p-STAT3 expression was located within the nucleus
(Figs. 3 and 4) and positive MUC1 expression was located
within the cytoplasm (Figs. 5 and
6). STAT3 and p-STAT3 expression were
identified in 83.7% (82/98) and 52.0% (51/98) of cases,
respectively. MUC1 protein expression was identified in 62.2%
(61/98) of cases. Table I
demonstrates that increasing STAT3 expression was significantly
associated with increasing pTNM stage (pI, 66.7% vs. pII, 84.6% vs.
pIIIa, 100.0%; P<0.01), whereas p-STAT3 expression was
significantly associated with pathological type (squamous cell
carcinoma 38.5% vs. adenocarcinoma 67.4%; P<0.01), pathological
lymph node (pN-23.3% vs. pN+ 64.7%.; P<0.01) and pTNM stage (pI,
29.2% vs. pII, 55.8% vs. pIIIa, 68.2%; P<0.05) MUC1 expression
was associated with pathological type (squamous cell carcinoma
52.0% vs. adenocarcinoma 74.0%; P<0.05), pathological tumor (pT1
27.3% vs. pT2 64.3% vs. pT3 78.6%; P<0.05), pathological lymph
node (pN-36.7% vs. pN+73.5%; P<0.01) and pTNM stage (pI, 29.2%
vs. pII, 71.1% vs. pIIIa, 81.8%; P<0.01). Table II demonstrates that STAT3 expression
was positively correlated with p-STAT3 expression (P<0.05), and
that p-STAT3 expression was positively correlated with MUC1
expression (P<0.01). There was no correlation between STAT3 and
MUC1 expression (P>0.05).
 | Table II.Association between variables in the
NSCLC tissue group. |
Table II.
Association between variables in the
NSCLC tissue group.
| Variable | rs |
P-valuea |
|---|
| STAT3 and
p-STAT3 | 0.239 | 0.018 |
| STAT3 and MUC1 | 0.059 | 0.562 |
| p-STAT3 and
MUC1 | 0.306 | 0.002 |
Associations between STAT3, p-STAT3
and MUC1 expression levels and prognosis
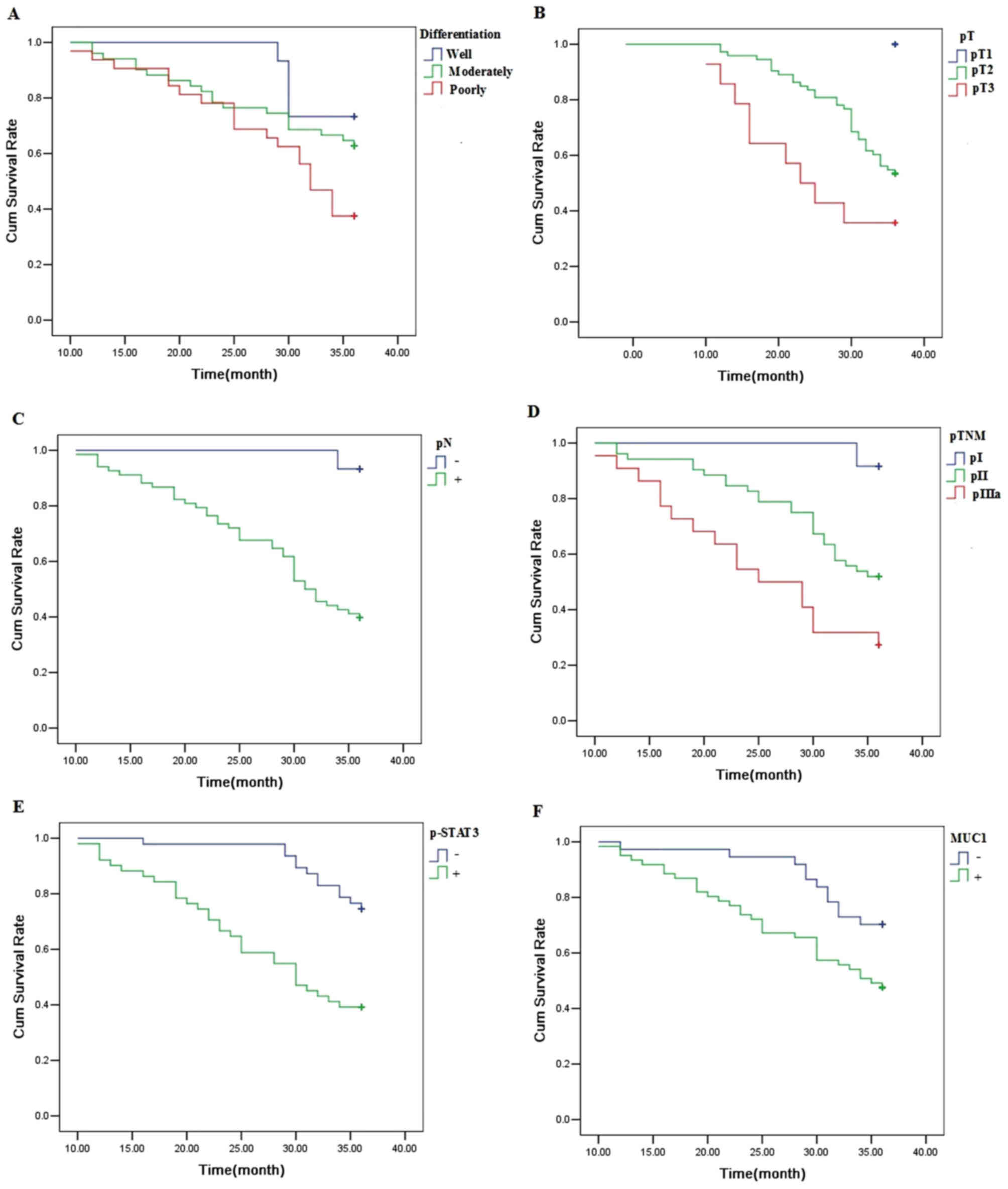
The 3-year survival rate for 98 patients with NSCLC
was 56.1%. A univariate analysis was conducted using the log-rank
test, and the 3-year survival rate was significantly associated
with the degree of differentiation (P<0.05), pT (P<0.01), pN
(P<0.01), pTNM stage (P<0.01), p-STAT3 expression (P<0.01)
and MUC1 expression (P<0.05) (Fig.
7; Table III). Cox multivariate
regression demonstrated that pN and p-STAT3 expression were
independent factors for the 3-year survival rate (Table IV).
 | Table III.Univariate analysis with regard to
the 3-year survival rate. |
Table III.
Univariate analysis with regard to
the 3-year survival rate.
|
|
| 3-year survival
rate |
|---|
|
|
|
|
|---|
| Clinical
features | Patients, n | Patients, n | Survival rate,
% | P-value |
|---|
| Total | 98 | 55 | 56.1 |
|
| Sex |
|
|
| 0.149 |
|
Male | 55 | 34 | 61.9 |
|
|
Female | 43 | 21 | 44.2 |
|
| Age, years |
|
|
| 0.866 |
|
<60 | 45 | 25 | 55.6 |
|
|
≥60 | 53 | 30 | 56.6 |
|
| Tumor location |
|
|
| 0.261 |
|
Central | 39 | 19 | 48.7 |
|
|
Peripheral | 59 | 36 | 61.0 |
|
| Histological
type |
|
|
| 0.060 |
|
SCC | 52 | 33 | 63.5 |
|
|
ADC | 46 | 22 | 47.8 |
|
|
Differentiation |
|
|
| 0.033 |
|
Well | 15 | 11 | 72.7 |
|
|
Moderately | 51 | 32 | 62.7 |
|
|
Poorly | 32 | 12 | 37.5 |
|
| pT |
|
|
| 0.002 |
|
pT1 | 11 | 11 | 100 |
|
|
pT2 | 73 | 39 | 53.4 |
|
|
pT3 | 14 | 5 | 35.7 |
|
| pN |
|
|
| 0.001 |
| − | 30 | 28 | 93.3 |
|
| + | 68 | 27 | 39.7 |
|
| pTNM |
|
|
| 0.001 |
| pI | 24 | 22 | 91.7 |
|
|
pII | 52 | 27 | 51.9 |
|
|
pIIIa | 22 | 6 | 27.3 |
|
| Chemotherapy |
|
|
| 0.636 |
| No | 24 | 14 | 58.3 |
|
|
Yes | 74 | 41 | 55.4 |
|
| Radiotherapy |
|
|
| 0.939 |
| No | 73 | 42 | 57.5 |
|
|
Yes | 25 | 13 | 52.0 |
|
| EGFR-TKI
therapy |
|
|
| 0.152 |
| No | 78 | 47 | 60.3 |
|
|
Yes | 20 | 8 | 40.0 |
|
| STAT3 |
|
|
| 0.278 |
| − | 16 | 11 | 68.8 |
|
| + | 82 | 44 | 53.7 |
|
| p-STAT3 |
|
|
| 0.001 |
| − | 47 | 35 | 74.5 |
|
| + | 51 | 20 | 39.2 |
|
| MUC1 |
|
|
| 0.019 |
| − | 37 | 26 | 70.3 |
|
| + | 61 | 29 | 47.5 |
|
 | Table IV.Results of cox regression
multivariate 3-year survival analysis. |
Table IV.
Results of cox regression
multivariate 3-year survival analysis.
| Variable | B | SE | Wald | P-value | HR | 95% CI |
|---|
| Sex | 0.350 | 0.651 | 0.289 | 0.591 | 1.419 | 0.396–5.078 |
| Age, years | −0.423 | 0.404 | 1.095 | 0.295 | 0.655 | 0.297–1.446 |
| Tumor location | 0.234 | 0.491 | 0.228 | 0.633 | 1.264 | 0.483–3.307 |
| Histological
type | 0.356 | 0.523 | 0.464 | 0.496 | 1.428 | 0.512–3.977 |
|
Differentiation | 0.420 | 0.323 | 1.692 | 0.193 | 1.522 | 0.808–2.867 |
| pT | 0.798 | 0.483 | 2.735 | 0.098 | 2.221 | 0.863–5.721 |
| pN | 1.996 | 0.848 | 5.544 | 0.018 | 7.362 | 1.397–38.794 |
| pTNM | 0.360 | 0.459 | 0.617 | 0.432 | 1.434 | 0.584–3.522 |
| Chemotherapy | −0.570 | 0.466 | 1.495 | 0.221 | 0.566 | 0.227–1.410 |
| Radiotherapy | 0.046 | 0.402 | 0.013 | 0.910 | 1.047 | 0.476–2.301 |
| EGFR-TKI
therapy | −0.204 | 0.422 | 0.234 | 0.629 | 0.815 | 0.357–1.864 |
| STAT3 | −0.333 | 0.573 | 0.337 | 0.562 | 0.717 | 0.233–2.206 |
| p-STAT3 | 0.898 | 0.417 | 4.634 | 0.031 | 2.454 | 1.084–5.556 |
| MUC1 | −0.276 | 0.423 | 0.290 | 0.514 | 0.759 | 0.331–1.738 |
Discussion
STAT3 is regarded as a primary mediator of
tumorigenesis and serves an important function in the
proliferation, apoptosis and hyperplasia of tumor cells (15). Constitutively activated STAT3 has been
identified in certain types of cancer, including NSCLC. In a study
by Yin et al (16), a total of
76 patients with NSCLC were enrolled in the study, and the results
demonstrated that STAT3 expression detected by IHC was associated
with lymph node metastasis, tumor differentiation and clinical
staging. Ai et al (17) used
IHC to detect STAT3 expression in a total of 65 patients with NSCLC
and demonstrated that increased STAT3 expression was associated
with tumor differentiation. In the present study, 83.7% of tumor
specimens exhibited STAT3 expression. STAT3 expression was
significantly associated with pTNM, and STAT3 expression in
advanced-stage patients was significantly increased compared with
that in early-stage patients. These results suggest that increased
STAT3 expression may be a frequent event in patients with NSCLC. A
previous study demonstrated that constitutively activated STAT3 was
enrolled in the janus tyrosine kinase/STAT signaling pathway of
NSCLC, and results demonstrated that 22–65% of patients with NSCLC
exhibited positive p-STAT3 expression (18). Wang et al (19) reported that the expression of p-STAT3
in NSCLC was significantly increased compared with that in
paracancerous tissue, and it was associated with smoking and the
size of the tumor. Xu and Lu (20)
summarized 17 trials using meta-analysis and identified that
p-STAT3 expression was associated with differentiation of NSCLC. In
the present study, 52.0% of NSCLC tumor specimens demonstrated
p-STAT3 expression which was associated with pathological type, pN
and pTNM. The expression levels of p-STAT3 in the adenocarcinoma
group (67.4%) were significantly increased compared with that of
the squamous cell carcinoma group (38.5%; P<0.01). P-STAT3
expression within the lymph node metastasis group (64.7%) was
significantly increased compared with that of the group lacking
lymph node metastasis (23.3%; P<0.01). Furthermore, p-STAT3
expression in the advanced-stage group was increased compared with
that of the early-stage group (pI, 29.2% vs. pII, 55.8% vs. pIIIa,
68.2%; P<0.05). The present study demonstrated that STAT3
activation increases metastasis of NSCLC. Results from the present
study demonstrated that the 3-year survival rate of patients with
NSCLC was 56.1%, and it was significantly associated with the
degree of differentiation, pT, pN, pTNM stage and p-STAT3
expression. Additionally, pN and p-STAT3 expression were relevant
independent factors for a poor prognosis.
MUC1 expression is associated with invasion,
metastasis and poor survival in certain types of cancer. Previous
studies demonstrated that increased MUC1 expression was present in
breast cancer (21,22). In gastric cancer, increased MUC1
expression was identified in primary and metastatic cancer
(23,24). Furthermore, increased MUC1 expression
was associated with lymph node metastasis in oral, liver and
pancreatic cancer (25–27). In specific types of cancer, including
renal clear cell carcinoma and thyroid cancer, it was reported that
increased MUC1 expression was also associated with a shorter
metastasis-free survival time (28,29).
Collectively, these studies demonstrate a marked association
between increased MUC1 expression and cancer invasion/metastasis.
Few studies have reported the clinicopathological characteristics
of MUC1 in patients with NSCLC. Situ et al (30) demonstrated that in patients with
NSCLC, MUC1 was more frequently expressed in adenocarcinoma
compared with that in squamous cell carcinoma. Furthermore, it was
also demonstrated that in patients with stage IB NSCLC, MUC1
expression was associated with being an independent prognostic
factor for survival rates. Demirag et al (31) identified that increased MUC1
expression was present in lung adenosquamous cancer, and was
significantly associated with disease progression. Results from the
present study demonstrated that MUC1 expression occurred in 62.2%
of patients with NSCLC. Patients within the adenocarcinoma group
demonstrated increased MUC1 expression compared with the squamous
cell carcinoma group. Furthermore, MUC1 expression was also
associated with pT, pN and pTNM. The present study demonstrated
that increased MUC1 expression was associated with a decreased
3-year survival rate; however, MUC1 expression was not an
independent factor for survival rates.
Previous studies demonstrated that STAT3 may serve
an important function in tumor progression by mediating MUC1
expression. For example, Ahmad et al (32) identified that STAT3 was able to
activate MUC1 transcription through the MUC1 carboxyl-terminal
receptor subunit (MUC1-C) in breast cancer cells. MUC1-C and STAT3
may serve numerous functions in cancer cell survival in an
autoinductive regulatory loop. Gao et al (33) demonstrated that MUC1 overexpression in
NSCLC cell lines was associated with high levels of activated
STAT3. Therefore, STAT transcription factors may stimulate the MUC1
promoter at the mRNA and protein level. The present study
demonstrated that p-STAT3 expression was associated with positive
MUC1 expression, and that p-STAT3 and MUC1 expression were
correlated with pT, pN and pTNM. Results from the present study
demonstrated that p-STAT3 and MUC1 were synergistically involved in
the metastasis of NSCLC.
To conclude, STAT3 expression was associated with
pTNM, and the expression of p-STAT3 was associated with
pathological type, pN and pTNM. MUC1 expression was associated with
pathological type, pT, pN and pTNM in patients with NSCLC.
Furthermore, the 3-year survival rate was correlated with
differentiation, pT, pN, pTNM stage, p-STAT3 and MUC1 expression,
with pN and p-STAT3 serving as relevant independent factors.
Additionally, p-STAT3 expression and MUC1 expression exhibited a
significant positive correlation in NSCLC tissue. Collectively, the
results suggest that p-STAT3 and MUC1 may serve as essential
biomarkers for tumor invasion and metastasis in NSCLC.
Acknowledgements
The present study was funded by The Second Group of
Jinan Science and Technology Development Program (grant no.
201602204).
References
|
1
|
Li Y, Wei S, Wang J, Hong L, Cui L and
Wang C: Analysis of the factors associated with abnormal
coagulation and prognosis in patients with non-small cell lung
cancer. Zhongguo Fei Ai Za Zhi. 17:789–796. 2014.(In Chinese).
PubMed/NCBI
|
|
2
|
Sun W, Song L, Ai T, Zhang Y, Gao Y and
Cui J: Prognostic value of MET, cyclin D1 and MET gene copy number
in non-small cell lung cancer. J Biomed Res. 27:220–230.
2013.PubMed/NCBI
|
|
3
|
Molina JR, Yang P, Cassivi SD, Schild SE
and Adjei AA: Non-small cell lung cancer: Epidemiology, risk
factors, treatment, and survivorship. Mayo Clin Proc. 83:pp.
584–594. 2008; View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mirsadraee S, Oswal D, Alizadeh Y, Caulo A
and van Beek E Jr: The 7th lung cancer TNM classification and
staging system: Review of the changes and implications. World J
Radiol. 28:128–134. 2012.
|
|
5
|
Tan Z, Yang C, Zhang X, Zheng P and Shen
W: Expression of glucose transporter 1 and prognosis in non-small
cell lung cancer: A pooled analysis of 1665 patients. Oncotarget.
8:60954–60961. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhou J, Yu Y, Pei Y, Cao C, Ding C, Wang
D, Sun L and Niu G: A potential prognostic biomarker SPC24 promotes
tumorigenesis and metastasis in lung cancer. Oncotarget.
8:65469–65480. 2017.PubMed/NCBI
|
|
7
|
Geiger JL, Grandis JR and Bauman JE: The
STAT3 pathway as a therapeutic target in head and neck cancer:
Barriers and innovations. Oral Oncol. 56:84–92. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Banerjee K and Resat H: Constitutive
activation of STAT3 in breast cancer cells: A review. Int J Cancer.
138:2570–2578. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang H, Byfield G, Jiang Y, Smith GW,
McCloskey M and Hartnett ME: VEGF-mediated STAT3 activation
inhibits retinal vascularization by down-regulating local
erythropoietin expression. Am J Pathol. 180:1243–1253. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kondo S, Yoshizaki T, Wakisaka N, Horikawa
T, Murono S, Jang KL, Joab I, Furukawa M and Pagano JS: MUC1
induced by Epstein-Barr virus latent membrane protein 1 causes
dissociation of the cell-matrix interaction and cellular
invasiveness via STAT signaling. J Virol. 81:1554–1562. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gendler SJ, Lancaster CA,
Taylor-Papadimitriou J, Duhig T, Peat N, Burchell J, Pemberton L,
Lalani EN and Wilson D: Molecular cloning and expression of human
tumor-associated polymorphic epithelial mucin. J Biol Chem.
265:15286–15293. 1990.PubMed/NCBI
|
|
12
|
Jarrard JA, Linnoila RI, Lee H, Steinberg
SM, Witschi H and Szabo E: MUC1 is a novel marker for the type II
pneumocyte ineage during lung carcinogenesis. Cancer Res.
58:5582–5589. 1998.PubMed/NCBI
|
|
13
|
Ho SB, Niehans GA, Lyftogt C, Yan PS,
Cherwitz DL, Gum ET, Dahiya R and Kim YS: Heterogeneity of mucin
gene expression in normal and neoplastic tissues. Cancer Res.
53:641–651. 1993.PubMed/NCBI
|
|
14
|
Mizoguchi M, Betensky RA, Batchelor TT,
Bernay DC, Louis DN and Nutt CL: Activation of STAT3, MAPK, and AKT
in malignant astrocytic gliomas: Correlation with EGFR status,
tumor grade, and survival. J Neuropathol Exp Neurol. 65:1181–1188.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhou W, Bi X, Gao G and Sun L: miRNA-133b
and miRNA-135a induce apoptosis via the JAK2/STAT3 signaling
pathway in human renal carcinoma cells. Biomed Pharmacother.
84:722–729. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yin Z, Zhang Y, Li Y, Lv T, Liu J and Wang
X: Prognostic significance of STAT3 expression and its correlation
with chemoresistance of non-small cell lung cancer cells. Acta
Histochem. 114:151–158. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ai T, Wang Z, Zhang M, Zhang L, Wang N, Li
W and Song L: Expression and prognostic relevance of STAT3 and
cyclin D1 in non-small cell lung cancer. Int J Biol Markers.
27:e132–e138. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yu Y, Zhao Q, Wang Z and Liu XY: Activated
STAT3 correlates with prognosis of non-small cell lung cancer and
indicates new anticancer strategies. Cancer Chemother Pharmacol.
75:917–922. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang RJ, Zhang JZ and Wang P: Expression
of pSTAT3 in non-small cell lung cancer and its clinical
significance. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 28:288–290.
2012.(In Chinese). PubMed/NCBI
|
|
20
|
Xu YH and Lu S: A meta-analysis of STAT3
and phospho-STAT3 expression and survival of patients with
non-small-cell lung cancer. Eur J Surg Oncol. 40:311–317. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lavrsen K, Madsen CB, Rasch MG, Woetmann
A, Ødum N, Mandel U, Clausen H, Pedersen AE and Wandall HH:
Aberrantly glycosylated MUC1 is expressed on the surface of breast
cancer cells and a target for antibody-dependent cell-mediated
cytotoxicity. Glycoconj J. 30:227–236. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yang E, Hu XF and Xing PX: Advances of
MUC1 as a target for breast cancer immunotherapy. Histol
Histopathol. 22:905–922. 2007.PubMed/NCBI
|
|
23
|
Yonezawa S, Kitajima S, Higashi M, Osako
M, Horinouchi M, Yokoyama S, Kitamoto S, Yamada N, Tamura Y,
Shimizu T, et al: A novel anti-MUC1 antibody against the MUC1
cytoplasmic tail domain: Use in sensitive identification of poorly
differentiated cells in adenocarcinoma of the stomach. Gastric
Cancer. 15:370–381. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang XT, Kong FB, Mai W, Li L and Pang LM:
MUC1 immunohistochemical expression as a prognostic factor in
gastric cancer: Meta-analysis. Dis Markers. 2016:94215712016.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang K, Tang W, Qu X, Guo Q, Inagaki Y,
Seyama Y, Abe H, Gai R, Kokudo N, Sugawara Y, et al: KL-6 mucin in
metastatic liver cancer tissues from primary colorectal carcinoma.
Hepatogastroenterology. 56:960–963. 2009.PubMed/NCBI
|
|
26
|
Mizumoto M, Honjo G, Kobashi Y, Awane M
and Matsusue S: Molecular profile of apomucin and p53 protein as
predictors of malignancy in intraductal papillary mucinous
neoplasms of the pancreas. Hepatogastroenterology. 58:1791–1795.
2011.PubMed/NCBI
|
|
27
|
Hamada T, Nomura M, Kamikawa Y, Yamada N,
Batra SK, Yonezawa S and Sugihara K: DF3 epitope expression on MUC1
mucin is associated with tumor aggressiveness, subsequent lymph
node metastasis, and poor prognosis in patients with oral squamous
cell carcinoma. Cancer. 118:5251–5264. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Leroy X, Zerimech F, Zini L, Copin MC,
Buisine MP, Gosselin B, Aubert JP and Porchet N: MUC1 expression is
correlated with nuclear grade and tumor progression in pT1 renal
clear cell carcinoma. Am J Clin Pathol. 118:47–51. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kaira K, Murakami H, Serizawa M, Koh Y,
Abe M, Ohde Y, Takahashi T, Kondo H, Nakajima T and Yamamoto N:
MUC1 expression in thymic epithelial tumors: MUC1 may be useful
marker as differential diagnosis between type B3 thymoma and thymic
carcinoma. Virchows Arch. 458:615–620. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Situ D, Wang J, Ma Y, Zhu Z, Hu Y, Long H
and Rong T: Expression and prognostic relevance of MUC1 in stage IB
non-small cell lung cancer. Med Oncol. 28 Suppl 1:S596–S604. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Demirag F, Cakir E, Bayiz H and Eren
Yazici U: MUC1 and bcl-2 expression in preinvasive lesions and
adenosquamous carcinoma of the lung. Acta Chir Belg. 113:19–24.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ahmad R, Rajabi H, Kosugi M, Joshi MD,
Alam M, Vasir B, Kawano T, Kharbanda S and Kufe D: MUC1-C
oncoprotein promotes STAT3 activation in an autoinductive
regulatory loop. Sci Signal. 4:ra92011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gao J, McConnell MJ, Yu B, Li J, Balko JM,
Black EP, Johnson JO, Lloyd MC, Altiok S and Haura EB: MUC1 is a
downstream target of STAT3 and regulates lung cancer cell survival
and invasion. Int J Oncol. 35:337–345. 2009.PubMed/NCBI
|