Introduction
Non-small cell lung cancer (NSCLC) accounts for
between 80 and 85% of cases of lung cancer, a leading cause of
mortality worldwide (1). Despite
advances in the diagnosis and treatment of lung cancer over the
last few decades, the majority of patients are diagnosed only at
advanced stages, and the overall prognosis of patients with NSCLC
remains poor (2). Therefore, it is
crucial to understand the underlying mechanisms of disease
progression and to identify new therapeutic targets for NSCLC.
MicroRNAs (miRNAs or miRs) are a class of small
non-coding RNAs of between 18 and 25 nucleotides in length that
bind to the 3′-untranslated region of mRNAs, thus resulting in mRNA
degradation or in decreased gene expression at the
post-transcriptional level (3).
Emerging evidence has revealed that miRNAs contribute to the
regulation of diverse cellular functions and that deregulation of
miRNA expression is often associated with a variety of disorders
including human malignancies (4,5). miR-145
has been identified as a tumor suppressor that is downregulated in
various types of cancer, including prostate (6), ovarian (7), bladder (8)
and colon (9–11) cancer. miR-145 has also been reported
to be involved in the progression of NSCLC (12–14).
However, the molecular mechanism underlying the effects of miR-145
on the biological progression of NSCLC is not well understood.
In the present study, the effects of miR-145 on the
proliferation, invasion, migration and apoptosis of NSCLC A549
cells were investigated. The results indicated that increased
miR-145 expression inhibited the proliferation, invasion and
migration of, and induced morphological changes in, NSCLC A549
cells. Furthermore, the upregulation of miR-145 activated the
caspase-3 cascade. These results suggest that miR-145 serves an
important function in NSCLC progression and thus may be a promising
target for the treatment of NSCLC.
Materials and methods
Reagents
The human NSCLC cell line A549 was purchased from
the American Type Culture Collection (Manassas, VA, USA). RPMI-1640
medium, fetal bovine serum (FBS), PBS, bovine serum albumin (BSA),
dimethylsulfoxide and Cell Counting Kit-8 (CCK-8) were purchased
from Beijing Transgen Biotech Co., Ltd. (Beijing, China).
Antibodies against matrix metalloproteinase (MMP)-2, MMP-9, B-cell
lymphoma 2 (Bcl-2), Bcl-2-associated X protein (Bax), caspase-3,
poly (ADP-ribose) polymerase (PARP), β-actin and horseradish
peroxidase (HRP)-conjugated secondary antibodies were purchased
from Cell Signaling Technology, Inc. (Danvers, MA, USA).
Lipofectamine® 2000 and Opti-MEM were purchased from
Invitrogen; Thermo Fisher Scientific, Inc. (Waltham, MA, USA).
AnnexinV/fluorescein isothiocyanate (FITC) kit and Matrigel
invasion chambers were purchased from BD Biosciences (San Jose, CA,
USA). The Transwell invasion chambers were purchased from Costar
(Corning Life Sciences, Cambridge, MA, USA). Crystal violet
staining solution was purchased from Beyotime Institute of
Biotechnology (Haimen, China). Scrambled sequence and miR-145 mimic
were purchased from Biotend (Shanghai, China).
Cell culture and transfection
The A549 cells were grown in RPMI-1640 medium
supplemented with 10% FBS, 100 U/ml penicillin and 100 µg/ml
streptomycin, and were cultured at 37°C in a humidified atmosphere
containing 5% CO2. All cells used in the present study
were subjected to <20 passages.
In the exponential phase, A549 cells were seeded at
a density of 5×105 cells/well in a 6-well plate for 24 h
before transfection. Lipofectamine 2000 was used for the
transfection of scrambled sequence: Forward,
5′-UUCUCCGAACGUGUCACGUTT-3′ and reverse,
5′-ACGUGACACGUUCGGAGAATT-3′; or miR-145 mimic, forward,
5′-GUCCAGUUUUCCCAGGAAUCCCU-3′ and reverse,
5′-GGAUUCCUGGGAAAACUGGACUU-3′, according to the manufacturer's
protocol. A final concentration of 100 nM miR-145 mimics and 100 nM
negative control miRNA was used for transfection. At 24 or 48 h,
the transfected cells were collected and used in the subsequent
experiments.
Cell proliferation assay
Cell proliferation was detected using the CCK-8
assay. Briefly, A549 cells were transfected with scrambled sequence
or miR-145 mimic. Transfected and non-transfected cells were
incubated in 96-well plates at a density of 4×103
cells/well. At 24, 48 and 72 h post-transfection, CCK-8 solution
was added (10 µl/well) and cells were incubated at 37°C for 2 h.
Absorbance at 450 nm was measured using a Universal Microplate
Reader EL800 (Bio-Tek instruments, Inc., Vermont, MA, USA).
Boyden chamber transwell assays
For the invasion assays, 5×104 cells were
plated in a 8.0 µm pore size Matrigel invasion chamber without
serum and the lower chamber contained RPMI-1640 supplemented with
20% FBS that acted as a chemoattractant. At 24 h, the non-invading
cells were removed with cotton swabs. The invasive cells located on
the lower side of the chamber were fixed in 4% formaldehydeat room
temperature for 15 min, stained with 0.2% crystal violet staining
solution at room temperature for 30 min and counted under a
phase-contrast microscope (Olympus Corporation, Tokyo, Japan) in
three random fields (magnification, ×100).
Wound healing assays
The migration of the A549 cells was evaluated using
wound-healing assays. Cells (5×105 cells cells/well)
from the three groups (CON, non-transfected control; NC,
non-specific negative control; and MIMIC, miR-145 mimic) seeded in
a 6-well culture plate to form a confluent monolayer and were then
wounded using 100 µl pipette tips. The scratch wounds were
observed, and images were captured at 0 and 24 h, under a
phase-contrast microscope (magnification, ×40).
Hoechst 33258 staining of the A549
cells
At 48 h post-transfection, A549 cells from the three
groups (CON, non-transfected control; NC, non-specific negative
control; and MIMIC, miR-145 mimic) were seeded in 6-well plates and
fixed with 4% paraformaldehyde for 10 min at 4°C. Subsequently,
cells were washed three times with ice-cold PBS and stained with 10
mg/l Hoechst 33258 for 10 min at 25°C in the dark. Nuclei were
observed under a fluorescence microscope (Olympus Corporation,
Tokyo, Japan) (magnification, ×200).
Cell apoptosis analysis
The cell apoptosis assays were performed using an
Annexin-V/FITC kit. Cell apoptosis was detected using flow
cytometry at 48 h after transfection. The transfected cells were
collected and washed with ice-cold PBS prior to being stained with
Annexin-V/FITC and propidium iodide (PI) solution for 15 min in the
dark. The proportion of the apoptotic cells was determined using a
flow cytometer (BD Biosciences, San Jose, CA, USA) and data were
analyzed using FlowJo software (version 10; Tree Star, Inc.,
Ashland, OR, USA). All experiments were performed three times.
Western blot analysis
At 48 h post-transfection, cells from the three
groups (CON, non-transfected control; NC, non-specific negative
control; and MIMIC, miR-145 mimic) were collected and lysed in
radioimmunoprecipitation assay buffer containing
phenylmethanesulfonyl fluoride and phosphatase inhibitor cocktail
(Applygen Technologies Inc., Beijing, China). Each sample was
centrifuged at 17,105.6 × g for 10 min at 4°C to remove cell debris
and the supernatant was collected for immune blotting. Protein
concentrations were calculated using BSA. Equal quantities of
proteins (40 µg) were loaded and separated by SDS-PAGE (10% gels)
for 2 h at 100 V under reducing conditions. The proteins were
transferred onto polyvinylidene difluoride membranes in a
Tris-glycine transfer buffer. Subsequent to blocking with 5% skim
milk in Tris-buffered saline containing 0.1% Tween-20 (TBST) at
room temperature for 2 h, the membrane was incubated with primary
antibodies at 4°C overnight. The primary antibodies were: Anti-Bax
(cat. no. 2774), anti-Bcl-2 (cat. no. 2872), anti-caspase-3 (cat.
no. 9662), anti-PARP (cat. no. 9542), anti-MMP-2 (cat. no. 4022),
anti-MMP-9 (cat. no. 3852) and β-actin (cat. no. 8457; dilution of
all, 1:1,000). The membranes were washed with Tris-buffered saline
with Tween-20 (TBST) three times and incubated with secondary
HRP-conjugated antibodies (Anti-rabbit IgG; cat. no. 7074;
dilution, 1:5,000) at 25°C for 2 h. Membranes were washed with TBST
three more times and immune reactive protein bands were detected by
an enhanced chemiluminescence kit (Beijing Trans gen Biotech Co.,
Ltd., Beijing, China). Image Quant TL 7.0 software (GE Healthcare,
Chicago, IL, USA) was employed to quantify protein expression
levels.
Statistical analysis
Data were analyzed using SPSS software (version
19.0; IBM Corp., Armonk, NY, USA). Data are expressed as the mean ±
standard deviation. Statistical analysis was performed using
independent two-sample t-tests, or by one-way analysis of variance
(ANOVA) with Tukey's post-hoc test for ≥3 groups. P<0.05 was
considered to indicate a statistically significant difference.
Results
Upregulation of miR-145 expression
inhibits the proliferation of A549 cells
A549 cells were transfected with scrambled sequence
or miR-145 mimic, and their proliferative ability was assessed
using a CCK-8 assay. At 24, 48 and 72 h post-transfection, the
optical density values of the miR-145 mimic group, the
non-transfected group, and non-specific negative control were
0.335±0.012, 0.426±0.002 and 0.349±0.048; 0.704±0.008, 0.928±0.070
and 0.903±0.032; 1.558±0.049, 2.028±0.209 and 1.987±0.155,
respectively (Fig. 1A). Therefore,
ectopic miR-145 expression significantly decreased the
proliferation of A549 cells (P<0.05).
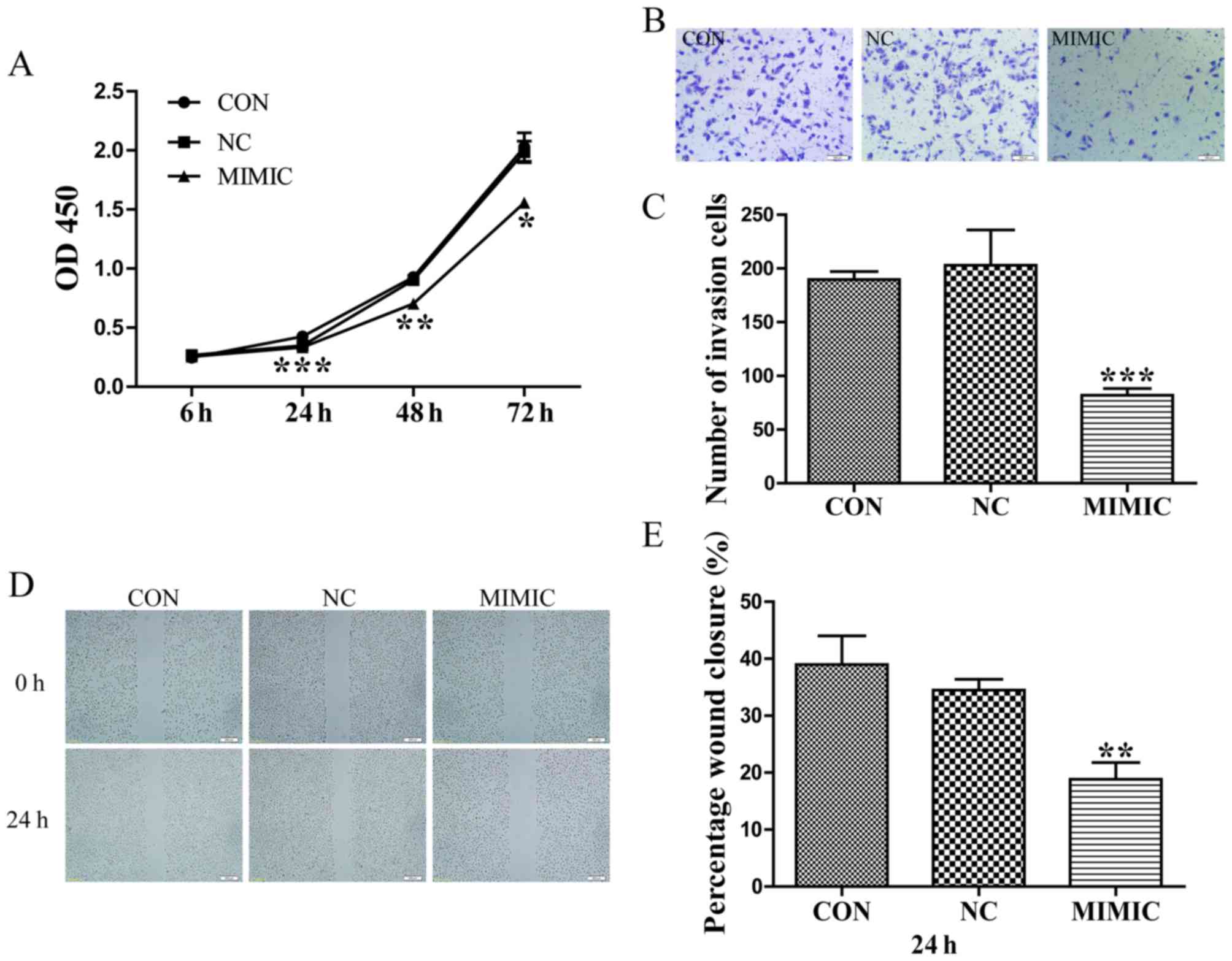 | Figure 1.Increased miR-145 expression inhibits
the proliferation, invasion and migration of A549 cells. (A)
Increased miR-145 expression significantly inhibited the
proliferation of A549 cells (*P<0.05, **P<0.01, ***P<0.001
vs. CON). (B) Effects of ectopic expression of miR-145 on the
migration of A549 cells. (C) Abnormal miR-145 expression
significantly repressed the number ofinvasive A549 cells (scale
bar, 100 µm; magnification, ×100; ***P<0.001 vs. CON). (D) Wound
healing assay. Gap length of theinitial (0 h) and the residual gap
length of 24 h after wounding were analyzed from photomicrographs.
(E) miR-145 over expression significantly suppressed the percentage
of wound closure in A549 cells (magnification, ×40) (**P<0.01
vs. CON). miR-145, microRNA-145; CON, non-transfected group; NC,
non-specific negative control; and MIMIC, miR-145 mimic; OD,
optical density. |
Abnormal miR-145 expression suppresses
the invasion and migration of A549 cells
Boyden chamber Transwell assays were employed to
investigate the effect of miR-145 expression on the invasive
ability of A549 cells. Typical micrographs of the Transwell filters
are presented in Fig. 1B. The
invasive cell count (Fig. 1B and C)
indicated that invasive capacity was significantly decreased in the
miR-145 mimic group compared with that in the non-transfected group
(P<0.001). The invasive cell count of the non-transfected group
compared with the non-specific negative control demonstrated no
significant difference (P>0.05). Additionally, the migratory
ability of A549 cells in response to miR-145 treatment was
evaluated using a wound healing assays, which identified that
treatment with an miR-145 mimic decreased the percentage of wound
closure compared with the non-transfected group, 24 h
post-transfection (P<0.01; Fig. 1D and
E). No difference in wound closure was observed between the
non-specific negative control and the non-transfected group
(P>0.05; Fig. 1E).
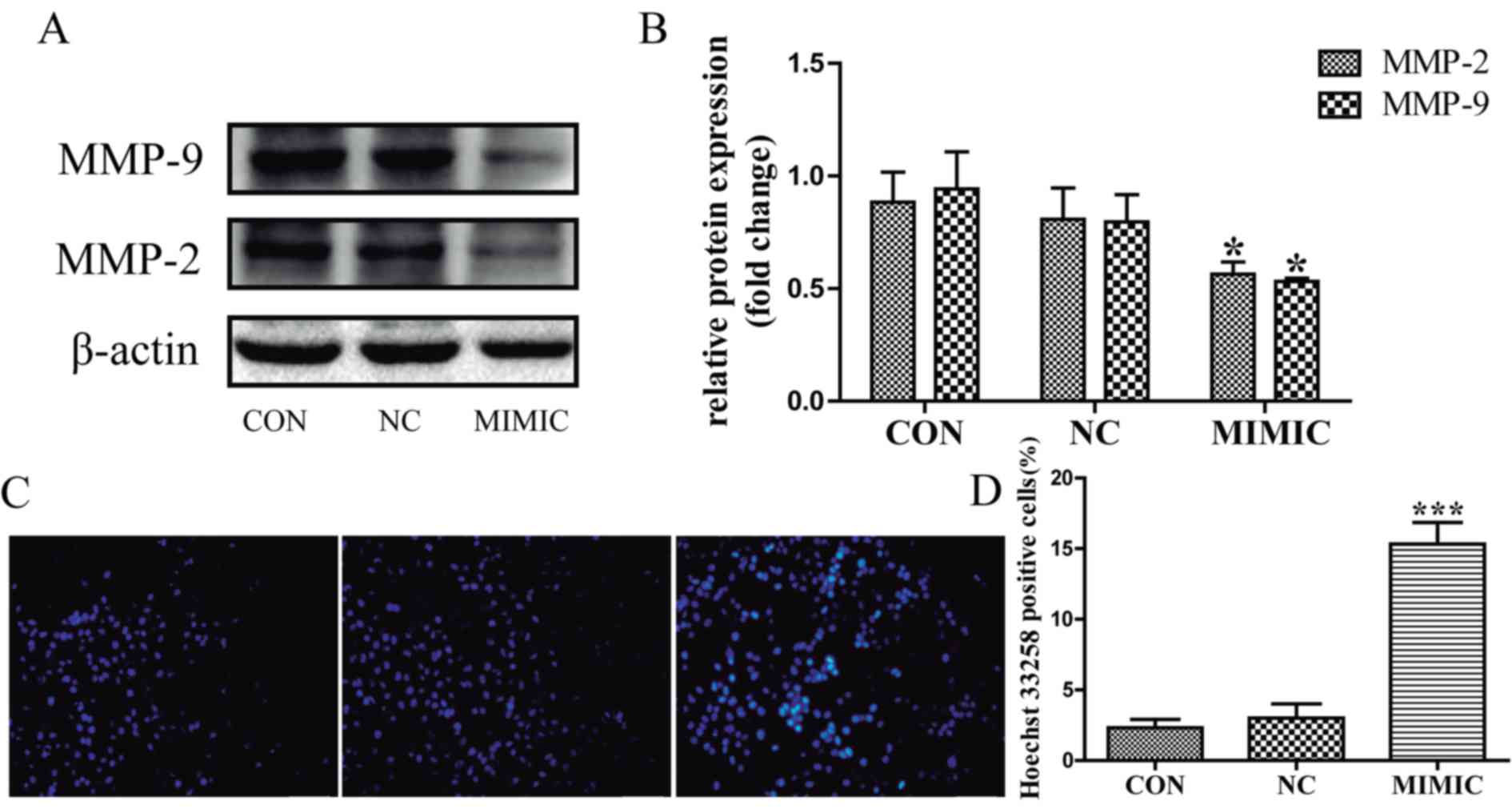
MMP-2 and MMP-9 expression is associated with tumor
invasion and metastasis (15–17). Therefore, the present study
investigated the expression levels of MMP-2 and MMP-9 in response
to miR-145 treatment in A549 cells using western blot analysis
(Fig. 2A). MMP-2 and MMP-9 expression
was significantly decreased in the miR-145 mimic group compared
with that in the non-transfected group (P<0.05; Fig. 2B). There was no significant difference
between the non-transfected group and the non-specific negative
control in the expression of MMP-2 and MMP-9 (P>0.05; Fig. 2B). These results indicate that
increased miR-145 expression inhibits the invasion and migration of
A549 cells possibly by repressing the expression of MMP-2 and
MMP-9.
Ectopic miR-145 expression promoted
the apoptosis of A549 cells
Apoptotic cells were detected in A549 cells using
the fluorescent DNA-binding dye Hoechst 33258, which stains the
nuclei of normal cells blue and those of apoptotic cells bright
blue or even white (Fig. 2C). The
results indicated that the number of apoptotic cells was increased
in the miR-145 mimic group compared with that in the
non-transfected group (P<0.001; Fig.
2D), there was no significant difference in the number of
apoptotic cells between the non-transfected group and the
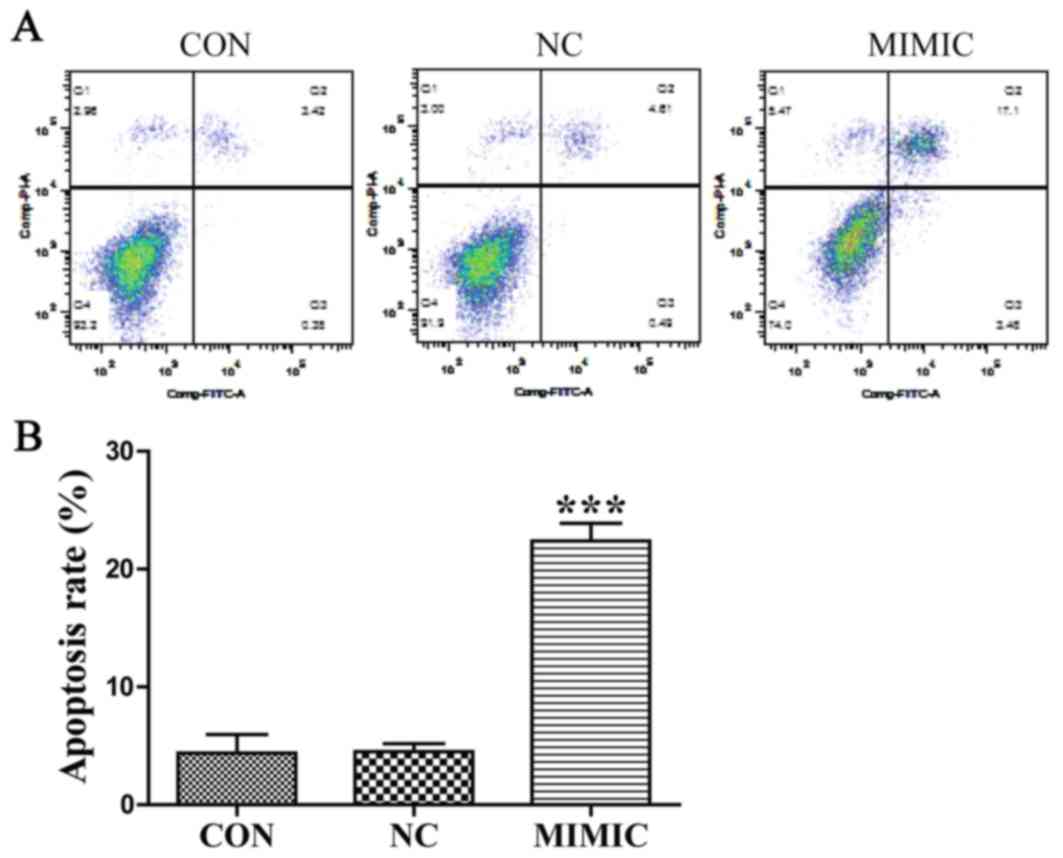
non-specific negative control (P>0.05). Next, flow cytometric
analysis using Annexin V-FITC/PI double staining depicted apoptosis
in A549 cells (Fig. 3A). Cell
apoptosis of ectopic miR-145 expression was increased and the
apoptotic index was significantly higher when compared with the
non-transfected group. The results indicated that ectopic miR-145
expression induced apoptosis of A549 cells (P<0.001; Fig. 3B). There was no significant difference
between the non-transfected group and the non-specific negative
control (P>0.05). Collectively, these results indicate that
deregulation of miR-145 expression promoted the apoptosis of A549
cells.
In order to elucidate the potential molecular
mechanism underlying the effect of miR-145 on apoptosis of A549
cells, the expression of Bax, Bcl-2, procaspase-3 and PARP was
examined using western blot analysis. miR-145 treatment increased
the expression of Bax, but led to a marked decrease in the
expression level of Bcl-2. Thus, the Bax/Bcl-2 ratio was increased
in the miR-145 mimic group compared with the non-transfected group
(P<0.01; Fig. 4A and B).
Furthermore, miR-145 over expression promoted the cleavage of
procaspase-3 and PARP (P<0.05; Fig. 4C
and D). No difference in the Bax/Bcl-2 ratio, or the cleavage
of procaspase-3 or PARP were observed between the non-transfected
group and the non-specific negative control group (P>0.05).
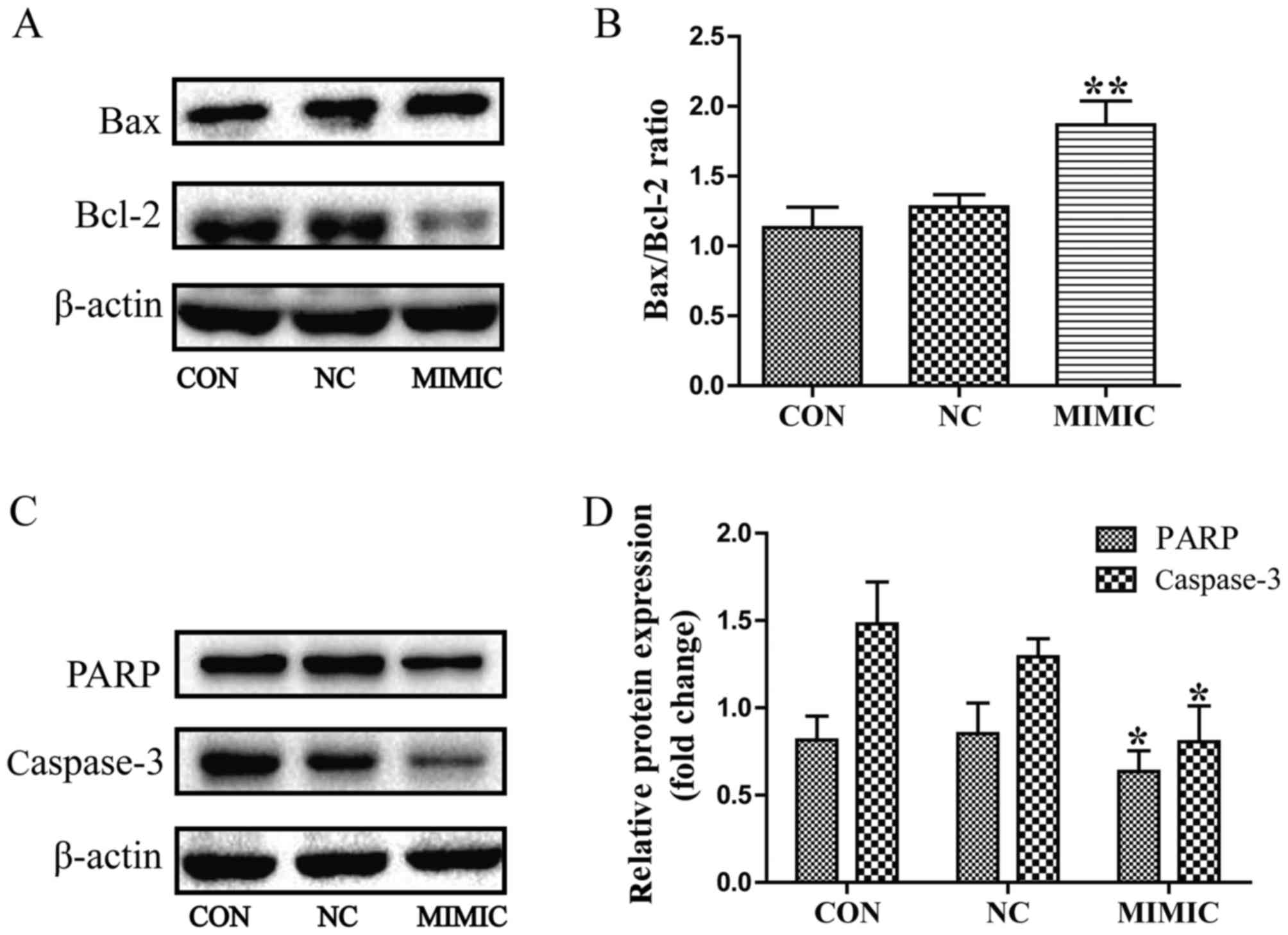 | Figure 4.Deregulated miR-145 expression
increases the Bax/Bcl-2 ratio and promoted the cleavage of PARP and
caspase-3 in A549 cells, as assessed using western blot analysis.
(A) miR-145 over expression increased the expression of Bax, but
decreased the expression level of Bcl-2. (B) Quantification of the
protein bands in (A). Thus, the Bax/Bcl-2 ratio was increased in
the MIMIC group (**P<0.01 vs. CON). (C and D) miR-145 over
expression promoted the cleavage of caspase-3 and PARP in A549
cells (*P<0.05 vs. CON). (D) Quantification of the protein bands
PARP and Caspase-3. miR-145, microRNA-145; CON, non-transfected
group; NC, non-specific negative control; MIMIC, miR-145 mimic
group; Bcl-2, B-cell lymphoma 2; Bax, Bcl-2-associated X protein;
PARP, poly(ADP-ribose) polymerase. |
Discussion
NSCLC accounts for ~80% of all types of lung cancer
and is a major cause of mortality worldwide (18). Despite advances in the medical
approaches for the treatment of NSCLC, there has not been any
improvement in the prognosis of patients with NSCLC over the last
few decades (19). The majority of
patients with NSCLC are diagnosed at advanced stages of the disease
(20), which suggests that NSCLC
cells have invasive and metastatic characters. Therefore, effective
molecular targets for the treatment of NSCLC are required.
miRNAs have been implicated in several aspects of
cancer biology, including cell proliferation, apoptosis, invasion
and migration (21). Additionally,
downregulation of miR-145 expression has been identified in various
types of human cancer, including prostate (6), hepatocellular (22), lung (23), gallbladder (24), thyroid (25), gastric (26) and kidney (27) cancer. However, a limited number of
studies have reported miR-145 as an oncogenic regulator (28–30) and
that miRNAs serve crucial functions in carcinogenesis and tumor
progression in NSCLC (31). miR-145
has been described as a tumor suppressor in lung cancer (32), which is consistent with the results of
the present study. miR-145 may regulate the expression of thyroid
cell differentiation markers, decrease cell proliferation, and
induce cell cycle arrest and apoptosis, which has been demonstrated
in various types of cancer (25,33). A
decrease in miR-145 expression may regulate a number of
tumor-associated targets, including c-Myc, signal transducer and
activator of transcription 1and yes-associated protein, thus
promoting tumorigenesis (34,35). However, the exact molecular mechanism
of the action of miR-145 on the tumor biology of NSCLC remains
controversial.
In the present study, miR-145 over expression
inhibited the proliferation, invasion and migration of, and induced
apoptosis in, A549 NSCLC cells. Previous studies have demonstrated
that upregulation of miR-145 expression prevents glioma stem cells
(GSCs) from digesting Matrigel and migrate through the pores thus
decreasing the migratory ability of GSCs. Additionally, it was
demonstrated that the miR-145-ATP-binding cassette transporter
G2-MMP-2/9 pathway regulated the migration and invasion in GSCs
(36). The results of the present
study indicated that ectopic miR-145 expression may decrease the
expression of MMP-2, MMP-9 and Bcl-2, and increase the expression
of Bax, thus increasing the Bax/Bcl-2 ratio. Previous studies have
demonstrated that miR-145 induces increased levels of pro-apoptotic
proteins including caspase-3, caspase-9 and PARP in glioma cells
(37). Consistent with those studies,
the results of the present study also confirmed that miR-145 over
expression induced apoptosis in NSCLC cells. Caspase-3 is an
executioner in caspase cascades, which are crucial steps in
apoptosis-induced cell death (38).
Caspase-3 activation induces the cleavage of specific substrates
such as PARP, which are pivotal for the occurrence of apoptosis
(39,40). Furthermore, changes in the ratio of
Bcl-2 family proteins are associated with an imbalance in
mitochondrial homeostasis, which leads to apoptosis (41,42).
Increased levels of pro-apoptotic Bax and/or decreased levels of
anti-apoptotic Bcl-2 lead to loss of mitochondrial membrane
potential, which is a critical process in the initiation of
apoptosis (43).
However, there are several limitations to the
present study. First, the effects of miR-145 on the cellular
proliferation, invasion and migration in lung cancer was evaluated
in vitro, therefore additional in vivo studies are
required. Secondly, the downstream proteins and the targets of
miR-145 in NSCLC cells require further investigation. Additionally,
the association between miR-145 and cellular pathways requires
investigation in future studies.
Taken together, the results of the present study
indicate that abnormal miR-145 expression is able to suppress the
proliferation and malignant phenotype of NSCLC A549 cells possibly
by decreasing the expression of MMP-2 and MMP-9, the Bax/Bcl-2
ratio and the activity of the caspase-3 cascade. Therefore, miR-145
may be a promising therapeutic target for patients with NSCLC.
Acknowledgements
The present study was supported by the Natural
Science Foundation of Jiangxi Province (grant no. 20132BAB205052)
and the Graduate Innovation Foundation of Nanchang University
(grant no. cx2016357).
Glossary
Abbreviations
Abbreviations:
|
NSCLC
|
non-small cell lung cancer
|
|
miR
|
microRNA
|
|
FBS
|
fetal bovine serum
|
|
HRP
|
horseradish peroxidase
|
|
SD
|
standard deviation
|
References
|
1
|
Ferlay J, Shin HR, Bray F, Forman D,
Mathers C and Parkin DM: Estimates of worldwide burden of cancer in
2008: GLOBOCAN 2008. Int J Cancer. 127:2893–2917. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jemal A, Siegel R, Ward E, Hao Y, Xu J and
Thun MJ: Cancer statistics, 2009. CA Cancer J Clin. 59:225–249.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ambros V: The functions of animal
microRNAs. Nature. 431:350–355. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Guz M, Rivero-Müller A, Okoń E,
Stenzel-Bembenek A, Polberg K, Słomka M and Stepulak A:
MicroRNAs-role in lung cancer. Disease Markers. 2014:2181692014.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bartel DP: MicroRNAs: Target recognition
and regulatory functions. Cell. 136:215–233. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Porkka KP, Pfeiffer MJ, Waltering KK,
Vessella RL, Tammela TL and Visakorpi T: MicroRNA expression
profiling in prostate cancer. Cancer Res. 67:6130–6135. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nam EJ, Yoon H, Kim SW, Kim H, Kim YT, Kim
JH, Kim JW and Kim S: MicroRNA expression profiles in serous
ovarian carcinoma. Clin Cancer Res. 14:2690–2695. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ichimi T, Enokida H, Okuno Y, Kunimoto R,
Chiyomaru T, Kawamoto K, Kawahara K, Toki K, Kawakami K, Nishiyama
K, et al: Identification of novel microRNA targets based on
microRNA signatures in bladder cancer. Int J Cancer. 125:345–352.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Akao Y, Nakagawa Y and Naoe T:
MicroRNA-143 and −145 in colon cancer. DNA Cell Biol. 26:311–320.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sachdeva M, Zhu S, Wu F, Wu H, Walia V,
Kumar S, Elble R, Watabe K and Mo YY: p53 represses c-Myc through
induction of the tumor suppressor miR-145. Proc Natl Acad Sci USA.
106:pp. 3207–3212. 2009; View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Slaby O, Svoboda M, Fabian P, Smerdova T,
Knoflickova D, Bednarikova M, Nenutil R and Vyzula R: Altered
expression of miR-21, miR-31, miR-143 and miR-145 is related to
clinicopathologic features of colorectal cancer. Oncology.
72:397–402. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Campayo M, Navarro A, Viñolas N, Diaz T,
Tejero R, Gimferrer JM, Molins L, Cabanas ML, Ramirez J, Monzo M
and Marrades R: Low miR-145 and high miR-367 are associated with
unfavourable prognosis in resected nonsmall cell lung cancer. Eur
Respir J. 41:1172–1178. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cho WC, Chow AS and Au JS: MiR-145
inhibits cell proliferation of human lung adenocarcinoma by
targeting EGFR and NUDT1. RNA Biol. 8:125–131. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cho WC, Chow AS and Au JS: Restoration of
tumour suppressor hsa-miR-145 inhibits cancer cell growth in lung
adenocarcinoma patients with epidermal growth factor receptor
mutation. Eur J cancer. 45:2197–2206. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li H, Zhang K, Liu LH, Ouyang Y, Bu J, Guo
HB and Xiao T: A systematic review of matrix metalloproteinase 9 as
a biomarker of survival in patients with osteosarcoma. Tumour Biol.
35:5487–5491. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang J, Shi Q, Yuan TX, Song QL, Zhang Y,
Wei Q, Zhou L, Luo J, Zuo G, Tang M, et al: Matrix
metalloproteinase 9 (MMP-9) in osteosarcoma: Review and
meta-analysis. Clin Chim Acta. 433:225–231. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Shang HS, Chang JB, Lin JH, Lin JP, Hsu
SC, Liu CM, Liu JY, Wu PP, Lu HF, Au MK and Chung JG: Deguelin
inhibits the migration and invasion of U-2 OS human osteosarcoma
cells via the inhibition of matrix metalloproteinase-2/-9 in vitro.
Molecules. 19:16588–16608. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li W, Wang Y, Zhang Q, Tang L, Liu X, Dai
Y, Xiao L, Huang S, Chen L, Guo Z, et al: MicroRNA-486 as a
biomarker for early diagnosis and recurrence of non-small cell lung
cancer. PLoS One. 10:e01342202015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Verdecchia A, Francisci S, Brenner H,
Gatta G, Micheli A, Mangone L and Kunkler I; EUROCARE-4 Working
Group, : Recent cancer survival in Europe: A 2000-02 period
analysis of EUROCARE-4 data. Lancet Oncol. 8:784–796. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Molina JR, Yang P, Cassivi SD, Schild SE
and Adjei AA: Non-small cell lung cancer: Epidemiology, risk
factors, treatment, and survivorship. Mayo Clin Proc. 83:pp.
584–594. 2008; View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Angulo M, Lecuona E and Sznajder JI: Role
of MicroRNAs in lung disease. Arch Bronconeumol. 48:325–330. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yang XW, Zhang LJ, Huang XH, Chen LZ, Su
Q, Zeng WT, Li W and Wang Q: miR-145 suppresses cell invasion in
hepatocellular carcinoma cells: miR-145 targets ADAM17. Hepatol
Res. 44:551–559. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ling DJ, Chen ZS, Zhang YD, Liao QD, Feng
JX, Zhang XY and Shi TS: MicroRNA-145 inhibits lung cancer cell
metastasis. Mol Med Rep. 11:3108–3114. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Letelier P, García P, Leal P, Álvarez H,
Ili C, López J, Castillo J, Brebi P and Roa JC: miR-1 and miR-145
act as tumor suppressor microRNAs in gallbladder cancer. Int J Clin
Exp Pathol. 7:1849–1867. 2014.PubMed/NCBI
|
|
25
|
Boufraqech M, Zhang L, Jain M, Patel D,
Ellis R, Xiong Y, He M, Nilubol N, Merino MJ and Kebebew E: miR-145
suppresses thyroid cancer growth and metastasis and targets AKT3.
Endocr Relat Cancer. 21:517–531. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gao P, Xing AY, Zhou GY, Zhang TG, Zhang
JP, Gao C, Li H and Shi DB: The molecular mechanism of microRNA-145
to suppress invasion-metastasis cascade in gastric cancer.
Oncogene. 32:491–501. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Papadopoulos EI, Yousef GM and Scorilas A:
Cytotoxic activity of sunitinib and everolimus in Caki-1 renal
cancer cells is accompanied by modulations in the expression of
apoptosis-related microRNA clusters and BCL2 family genes. Biomed
Pharmacother. 70:33–40. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yuan W, Sui C, Liu Q, Tang W, An H and Ma
J: Up-regulation of microRNA-145 associates with lymph node
metastasis in colorectal cancer. PLoS One. 9:e1020172014.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li Z, Gu X, Fang Y, Xiang J and Chen Z:
microRNA expression profiles in human colorectal cancers with brain
metastases. Oncol Lett. 3:346–350. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Arndt GM, Dossey L, Cullen LM, Lai A,
Druker R, Eisbacher M, Zhang C, Tran N, Fan H, Retzlaff K, et al:
Characterization of global microRNA expression reveals oncogenic
potential of miR-145 in metastatic colorectal cancer. BMC Cancer.
9:3742009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Du L and Pertsemlidis A: microRNA
regulation of cell viability and drug sensitivity in lung cancer.
Expert Opin Biol Ther. 12:1221–1239. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lu Y, Govindan R, Wang L, Liu PY, Goodgame
B, Wen W, Sezhiyan A, Pfeifer J, Li YF, Hua X, et al: MicroRNA
profiling and prediction of recurrence/relapse-free survival in
stage I lung cancer. Carcinogenesis. 33:1046–1054. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Xu Q, Liu LZ, Qian X, Chen Q, Jiang Y, Li
D, Lai L and Jiang BH: MiR-145 directly targets p70S6K1 in cancer
cells to inhibit tumor growth and angiogenesis. Nucleic Acids Res.
40:761–774. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chen Z, Zeng H, Guo Y, Liu P, Pan H, Deng
A and Hu J: miRNA-145 inhibits non-small cell lung cancer cell
proliferation by targeting c-Myc. J Exp Clin Cancer Res.
29:1512010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gregersen LH, Jacobsen AB, Frankel LB, Wen
J, Krogh A and Lund AH: MicroRNA-145 targets YES and STAT1 in colon
cancer cells. PLoS One. 5:e88362010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Shi L, Wang Z, Sun G, Wan Y, Guo J and Fu
X: miR-145 inhibits migration and invasion of glioma stem cells by
targeting ABCG2. Neuromolecular Med. 16:517–528. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Rani SB, Rathod SS, Karthik S, Kaur N,
Muzumdar D and Shiras AS: MiR-145 functions as a tumor-suppressive
RNA by targeting Sox9 and adducin 3 in human glioma cells. Neuro
Oncol. 15:1302–1316. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ding L, Wu JP, Xu G, Zhu B, Zeng QM, Li DF
and Lu W: Lentiviral-mediated RNAi targeting caspase-3 inhibits
apoptosis induced by serum deprivation in rat endplate chondrocytes
in vitro. Braz J Med Biol Res. 47:445–451. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Norbury CJ and Zhivotovsky B: DNA
damage-induced apoptosis. Oncogene. 23:2797–2808. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Soldani C and Scovassi AI:
Poly(ADP-ribose) polymerase-1 cleavage during apoptosis: An update.
Apoptosis. 7:321–328. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Orrenius S: Mitochondrial regulation of
apoptotic cell death. Toxicol Lett. 149:19–23. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Scorrano L and Korsmeyer SJ: Mechanisms of
cytochrome c release by proapoptotic BCL-2 family members. Biochem
Biophys Res Commun. 304:437–444. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Meeran SM and Katiyar SK: Grape seed
proanthocyanidins promote apoptosis in human epidermoid carcinoma
A431 cells through alterations in Cdki-Cdk-cyclin cascade, and
caspase-3 activation via loss of mitochondrial membrane potential.
Exp Dermatol. 16:405–415. 2007. View Article : Google Scholar : PubMed/NCBI
|