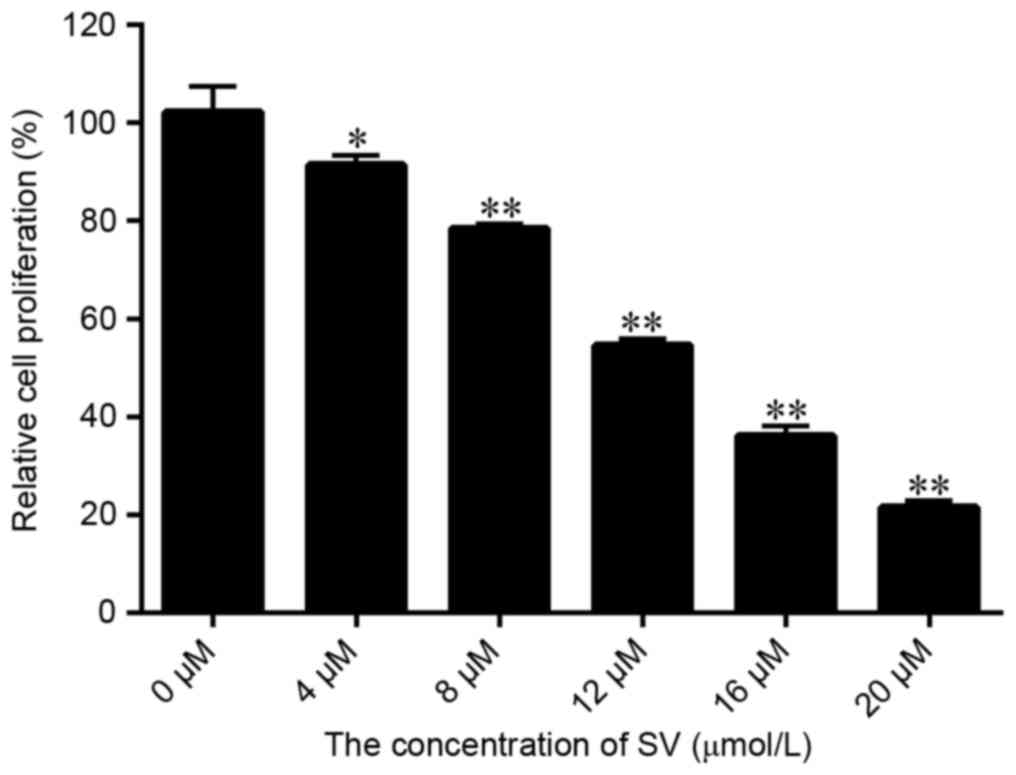
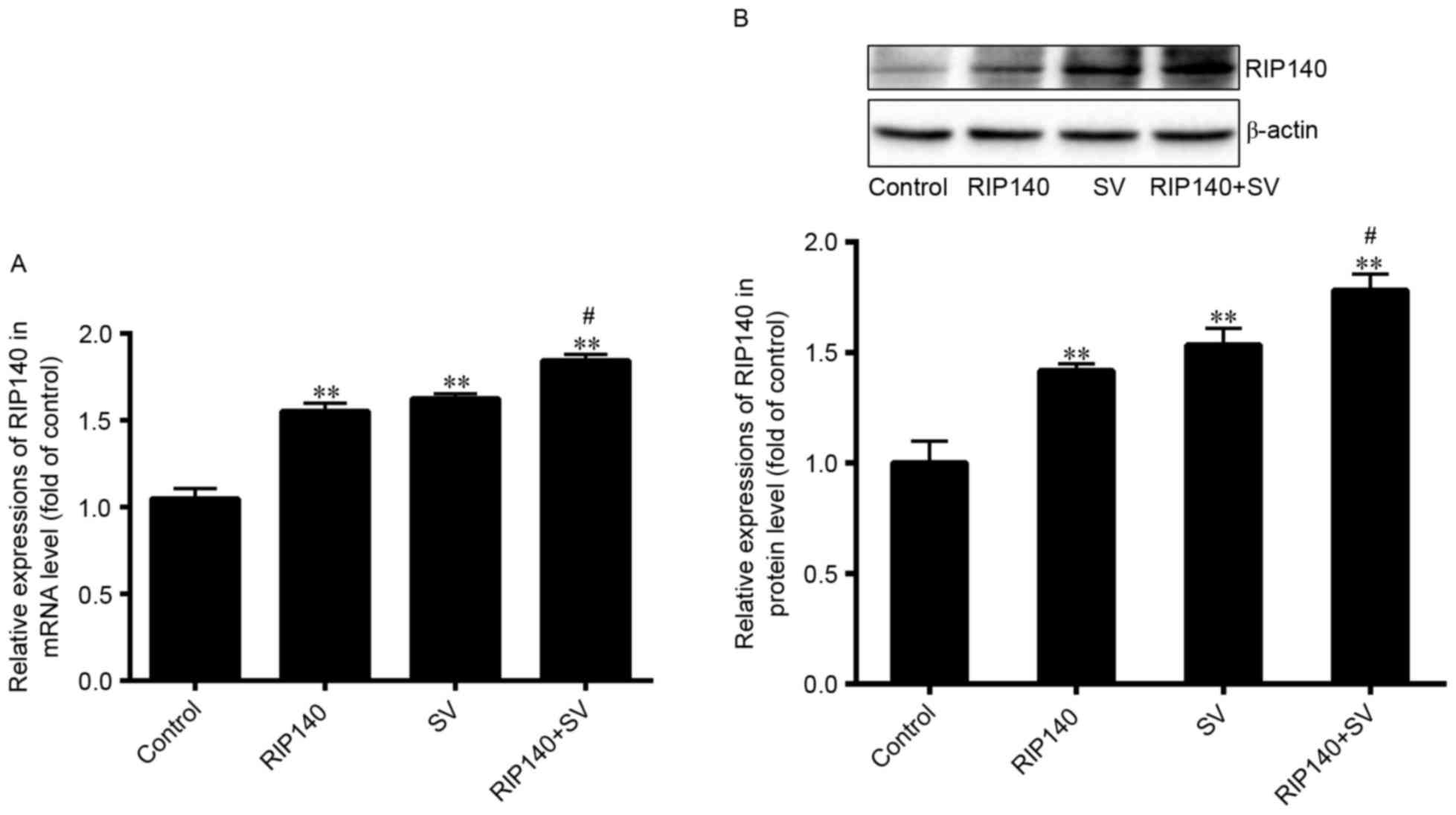
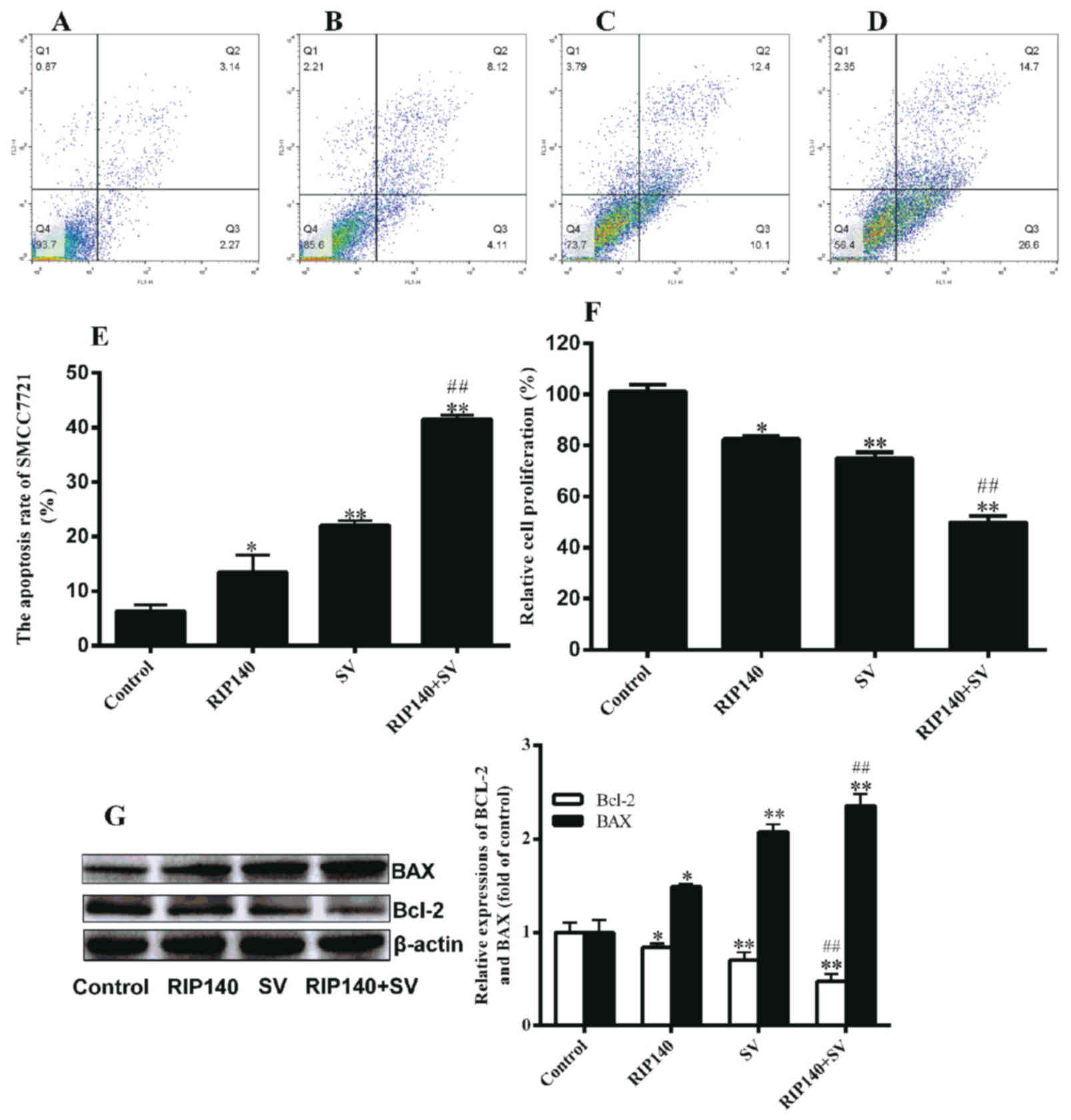
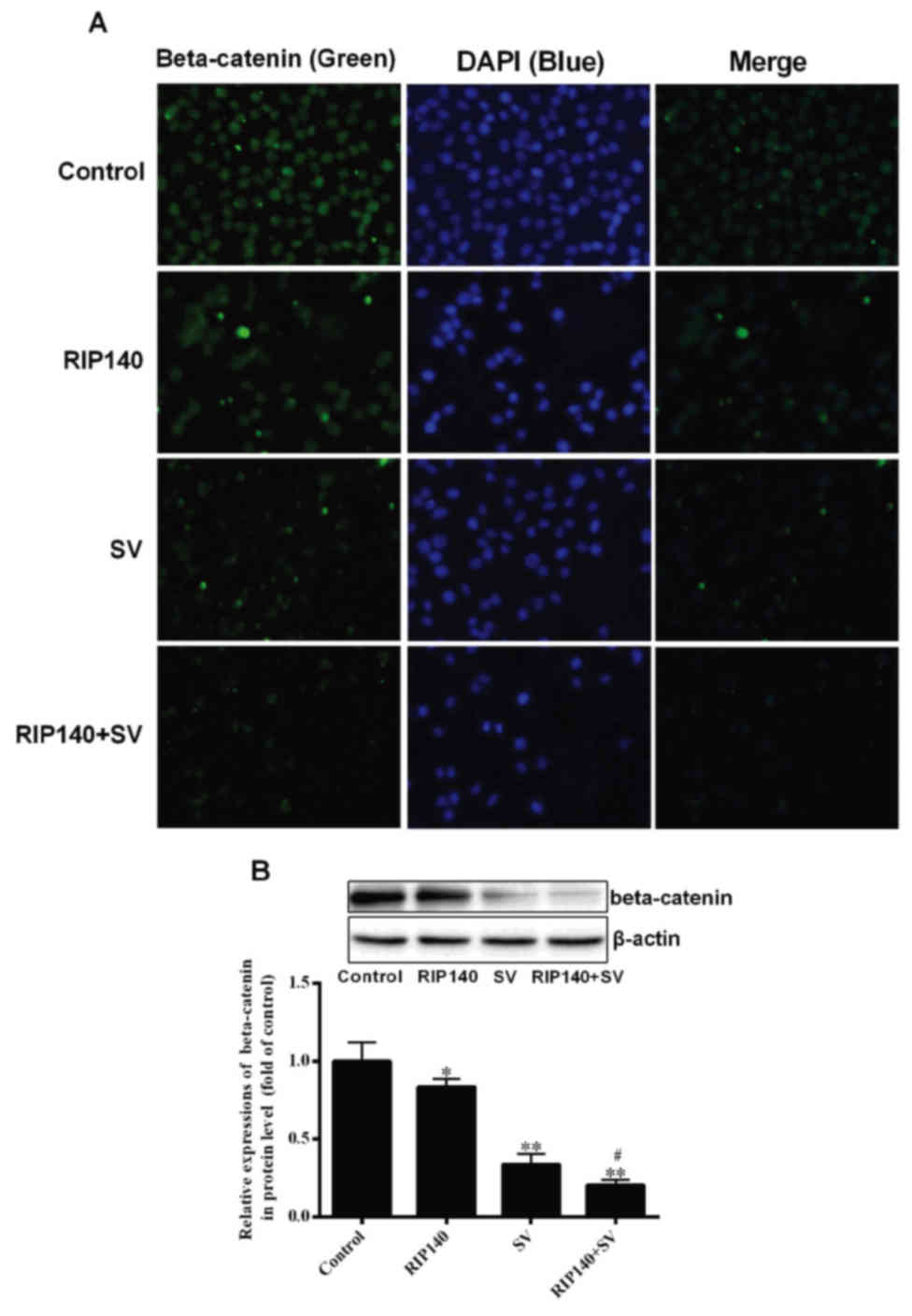
|
1
|
Istvan ES and Deisenhofer J: Structural
mechanism for statin inhibition of HMG-CoA reductase. Science.
292:1160–1164. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lee Y, Lee KH, Lee GK, Lee SH, Lim KY, Joo
J, Go YJ, Lee JS and Han JY: Randomized phase II study of afatinib
plus simvastatin versus afatinib alone in previously treated
patients with advanced nonadenocarcinomatous non-small cell lung
cancer. Cancer Res Treat. 49:1001–1011. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhou YY, Zhu GQ, Wang Y, Zheng JN, Ruan
LY, Cheng Z, Hu B, Fu SW and Zheng MH: Systematic review with
network meta-analysis: Statins and risk of hepatocellular
carcinoma. Oncotarget. 7:21753–21762. 2016.PubMed/NCBI
|
|
4
|
Demierre MF, Higgins PD, Gruber SB, Hawk E
and Lippman SM: Statins and cancer prevention. Nat Rev Cancer.
5:930–942. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Blanc-Brude OP, Mesri M, Wall NR, Plescia
J, Dohi T and Altieri DC: Therapeutic targeting of the survivin
pathway in cancer: Initiation of mitochondrial apoptosis and
suppression of tumor-associated angiogenesis. Clin Cancer Res.
9:2683–2692. 2003.PubMed/NCBI
|
|
6
|
Li G, Zheng J, Xu B, Ling J, Qiu W and
Wang Y: Simvastatin inhibits tumor angiogenesis in
HER2-overexpressing human colorectal cancer. Biomed Pharmacother.
85:418–424. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang Z, Zhang Y, Wang W, Hua Y, Liu L,
Shen S and Peng B: Thrombocytopenia and the outcomes of hepatectomy
for hepatocellular carcinoma: A meta-analysis. J Surg Res.
210:99–107. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ferreira-Silva GÁ, Lages CC, Sartorelli P,
Hasegawa FR, Soares MG and Ionta M: Casearin D inhibits ERK
phosphorylation and induces downregulation of cyclin D1 in HepG2
cells. Toxicol In Vitro. 38:27–32. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu WH, Lee YM, Lam KK, Chen YF, Wang JJ,
Yen MH and Cheng PY: The role of receptor-interacting protein 140
in the accumulation of fat in ovariectomised rats. Obes Surg.
21:935–940. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mejhert N, Laurencikiene J, Pettersson AT,
Kaaman M, Stenson BM, Rydén M and Dahlman I: Role of
receptor-interacting protein 140 in human fat cells. BMC Endocr
Disord. 10:12010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Catalán V, Gómez-Ambrosi J, Lizanzu A,
Rodríguez A, Silva C, Rotellar F, Gil MJ, Cienfuegos JA, Salvador J
and Frühbeck G: RIP140 gene and protein expression levels are
downregulated in visceral adipose tissue in human morbid obesity.
Obes Surg. 19:771–776. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lapierre M, Docquier A, Castet-Nicolas A,
Gitenay D, Jalaguier S, Teyssier C and Cavaillès V: The emerging
role of the transcriptional coregulator RIP140 in solid tumors.
Biochim Biophys Acta. 1856:144–150. 2015.PubMed/NCBI
|
|
13
|
Aziz MH, Chen X, Zhang Q, DeFrain C,
Osland J, Luo Y, Shi X and Yuan R: Suppressing NRIP1 inhibits
growth of breast cancer cells in vitro and in vivo. Oncotarget.
6:39714–39724. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Huang CR, Jin ZX, Dong L, Tong XP, Yue S,
Kawanami T, Sawaki T, Sakai T, Miki M, Iwao H, et al: Cisplatin
augments FAS-mediated apoptosis through lipid rafts. Anticancer
Res. 30:2065–2071. 2010.PubMed/NCBI
|
|
15
|
Zhang D, Wang Y, Dai Y, Wang J, Suo T, Pan
H and Liu H, Shen S and Liu H: Downregulation of RIP140 in
hepatocellular carcinoma promoted the growth and migration of the
cancer cells. Tumour Biol. 36:2077–2085. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jafari N, Zargar SJ, Yassa N and Delnavazi
MR: Induction of apoptosis and cell cycle arrest by dorema glabrum
root extracts in a gastric adenocarcinoma (AGS) cell line. Asian
Pac J Cancer Prev. 17:5189–5193. 2016.PubMed/NCBI
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hu S, Ding Y, Gong J and Yan N:
Sphingomyelin synthase 2 affects CD14-associated induction of NF-κB
by lipopolysaccharides in acute lung injury in mice. Mol Med Rep.
14:3301–3306. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Berwick DC, Javaheri B, Wetzel A,
Hopkinson M, Nixon-Abell J, Grannò S, Pitsillides AA and Harvey K:
Pathogenic LRRK2 variants are gain-of-function mutations that
enhance LRRK2-mediated repression of β-catenin signaling. Mol
Neurodegener. 12:92017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sohda T, Iwata K, Hirano G, Sakurai K,
Yokoyama K, Morihara D, Takeyama Y, Irie M, Shakado S and Sakisaka
S: 3-Hydroxyl-3-methylglutaryl-coenzyme A reductase is up regulated
in hepatocellular carcinoma associated with paraneoplastic
hypercholesterolemia. Med Mol Morphol. 46:239–242. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Furuya Y, Sekine Y, Kato H, Miyazawa Y,
Koike H and Suzuki K: Low-density lipoprotein receptors play an
important role in the inhibition of prostate cancer cell
proliferation by statins. Prostate Int. 4:56–60. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Huang X, Ma J, Xu J, Su Q and Zhao J:
Simvastatin induces growth inhibition and apoptosis in HepG2 and
Huh7 hepatocellular carcinoma cells via upregulation of Notch1
expression. Mol Med Rep. 11:2334–2340. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lee SJ, Hwang JW, Yim H, Yim HJ, Woo SU,
Suh SJ, Hyun JJ, Jung SW, Koo JS, Kim JH, et al: Synergistic effect
of simvastatin plus NS398 on inhibition of proliferation and
survival in hepatocellular carcinoma cell line. J Gastroenterol
Hepatol. 29:1299–1307. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Higashi T, Hayashi H, Kitano Y, Yamamura
K, Kaida T, Arima K, Taki K, Nakagawa S, Okabe H, Nitta H, et al:
Statin attenuates cell proliferative ability via TAZ (WWTR1) in
hepatocellular carcinoma. Med Oncol. 33:1232016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kim W, Yoon JH, Kim JR, Jang IJ, Bang YJ,
Kim YJ and Lee HS: Synergistic anti-tumor efficacy of lovastatin
and protein kinase C-beta inhibitor in hepatocellular carcinoma.
Cancer Chemother Pharmacol. 64:497–507. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lei JJ, Peng RJ, Kuang BH, Yuan ZY, Qin T,
Liu WS, Guo YM, Han HQ, Lian YF, Deng CC, et al: NOP14 suppresses
breast cancer progression by inhibiting NRIP1/Wnt/β-catenin
pathway. Oncotarget. 6:25701–25714. 2015. View Article : Google Scholar : PubMed/NCBI
|