Introduction
Pulmonary metastases from sarcomas form
well-circumscribed nodules. The incidence of cystic pulmonary
metastasis caused by a soft tissue tumor is extremely rare. When a
pulmonary cyst is not accompanied by nodules, it may not be readily
identified as a pulmonary metastasis (1). Previously, seven cases with pulmonary
cystic metastases from an epithelioid sarcoma have been reported in
the English literature (1–7). We report the case of an epithelioid
sarcoma of the left forearm, with cystic metastasis to the lung in
a 33-year-old man presenting with left spontaneous pneumothorax
during repeated chemotherapy treatment; we also reviewed the
literature. The clinical behavior of this rare presentation of
epithelioid sarcoma is discussed.
This patient and his families were informed that the
data from his case would be submitted for publication, and provided
consent.
Case report
A 33-year old man presented with sudden dyspnea and
chest pain on the left side. The patient had undergone wide
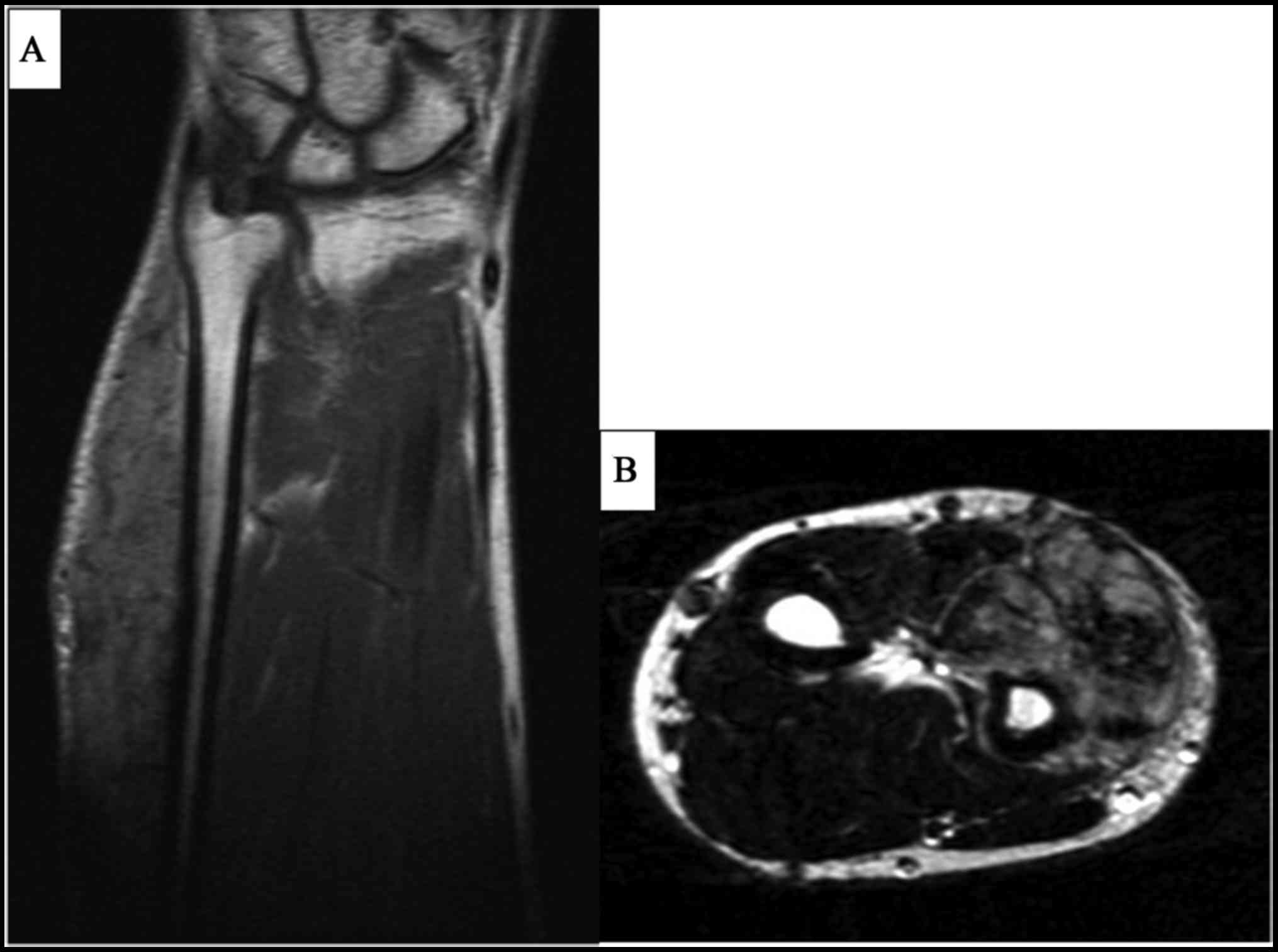
resection of an epithelioid sarcoma in the left forearm (Fig. 1) and pre- and post-operative
chemotherapy treatment 6 years prior. Four years ago, he complained
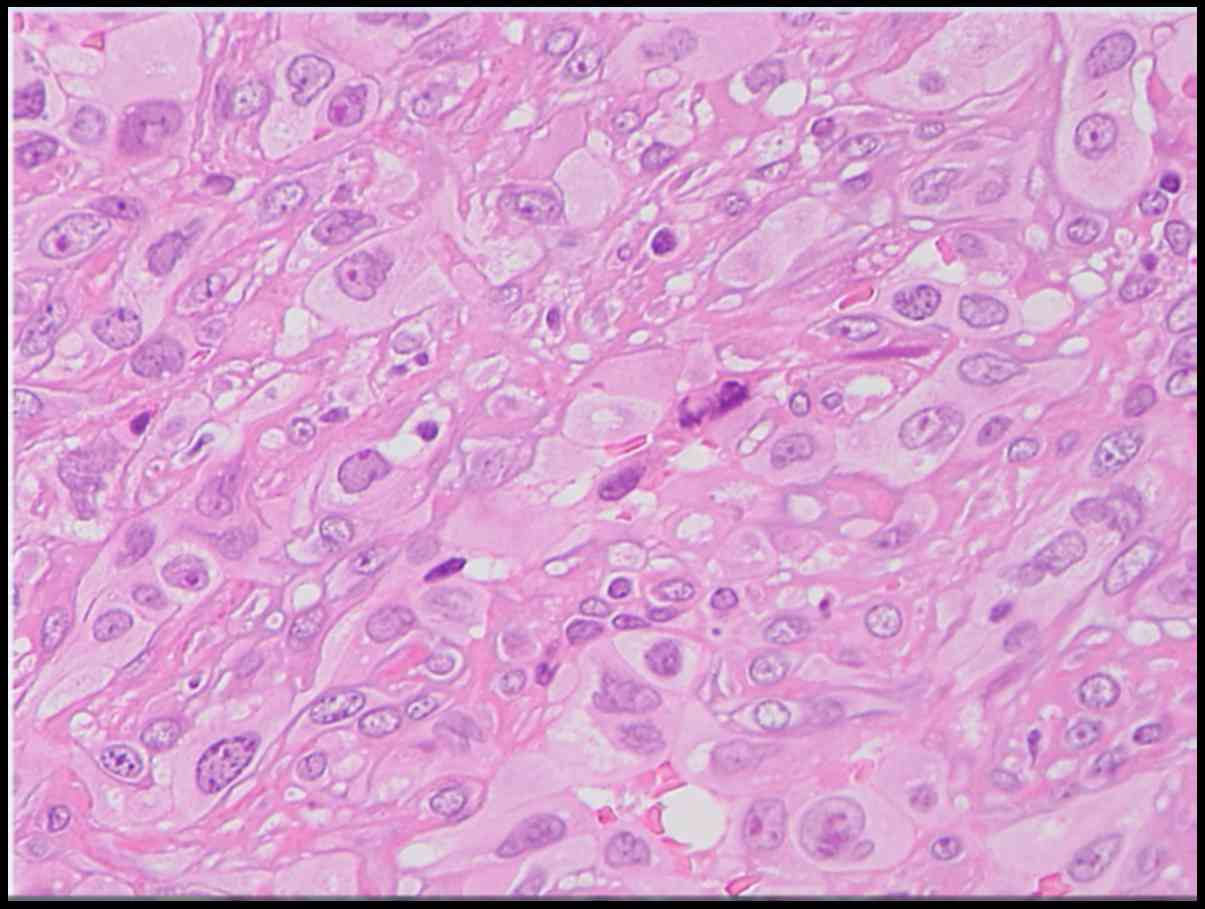
of continuous bloody sputum. A solid nodule of sarcomatous
metastasis appeared in the right lung, and histological examination
via bronchoscopy confirmed the pulmonary metastases from the
epithelioid sarcoma (Fig. 2).
Following histological confirmation, he started receiving repeated
chemotherapy treatments, composed of doxorubicin (DOX; 60
mg/m2)/ifosfamide (IFM; 10 g/m2), ifosfamide
(IFM; 15 g/m2), docetaxel (DTX; 75
mg/m2)/gemcitabine (GEM; 900 mg/m2), and
methotrexate (MTX; 10 g/m2). Despite repeated
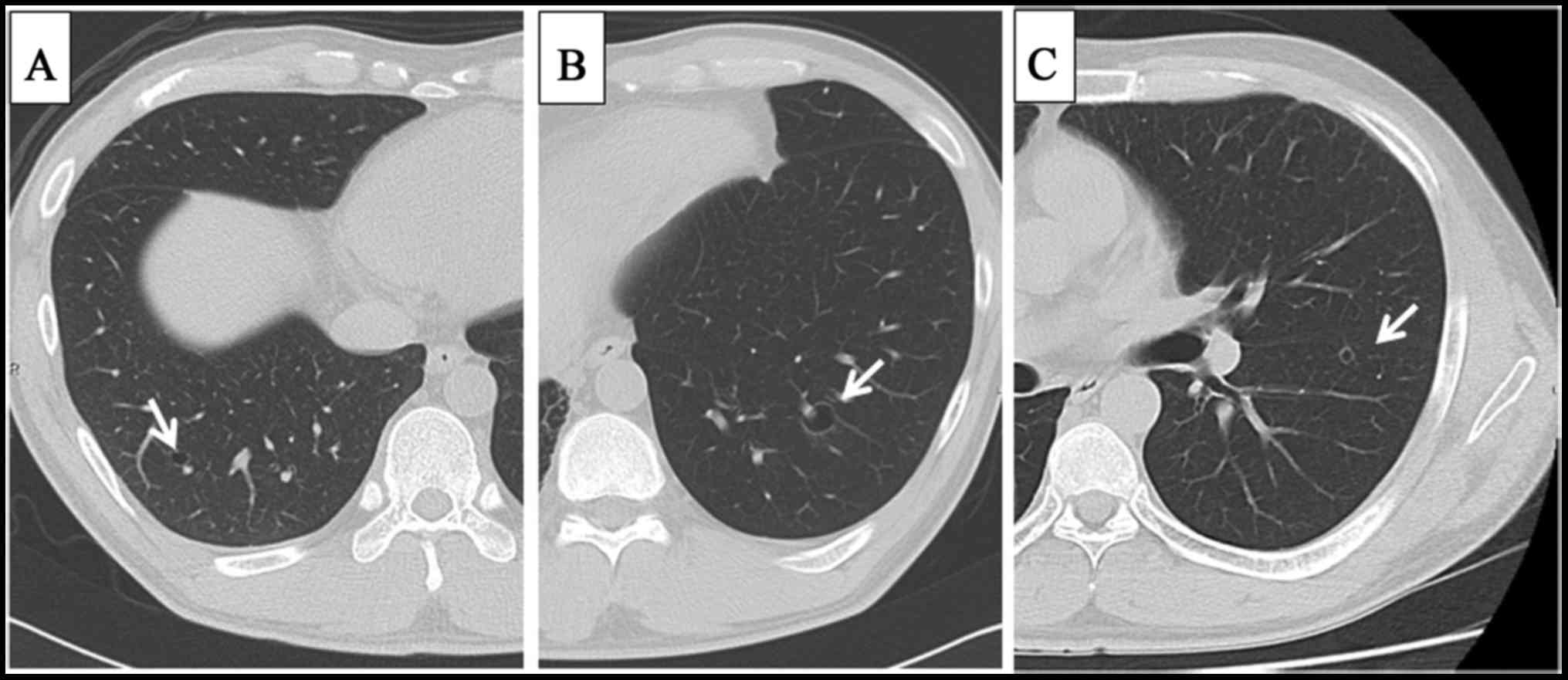
chemotherapy treatments, multiple small, thin-walled cystic
metastases in bilateral lungs appeared on computed tomography (CT)
examination (Fig. 3), and the number
and size of cystic metastases increased in 4 years.
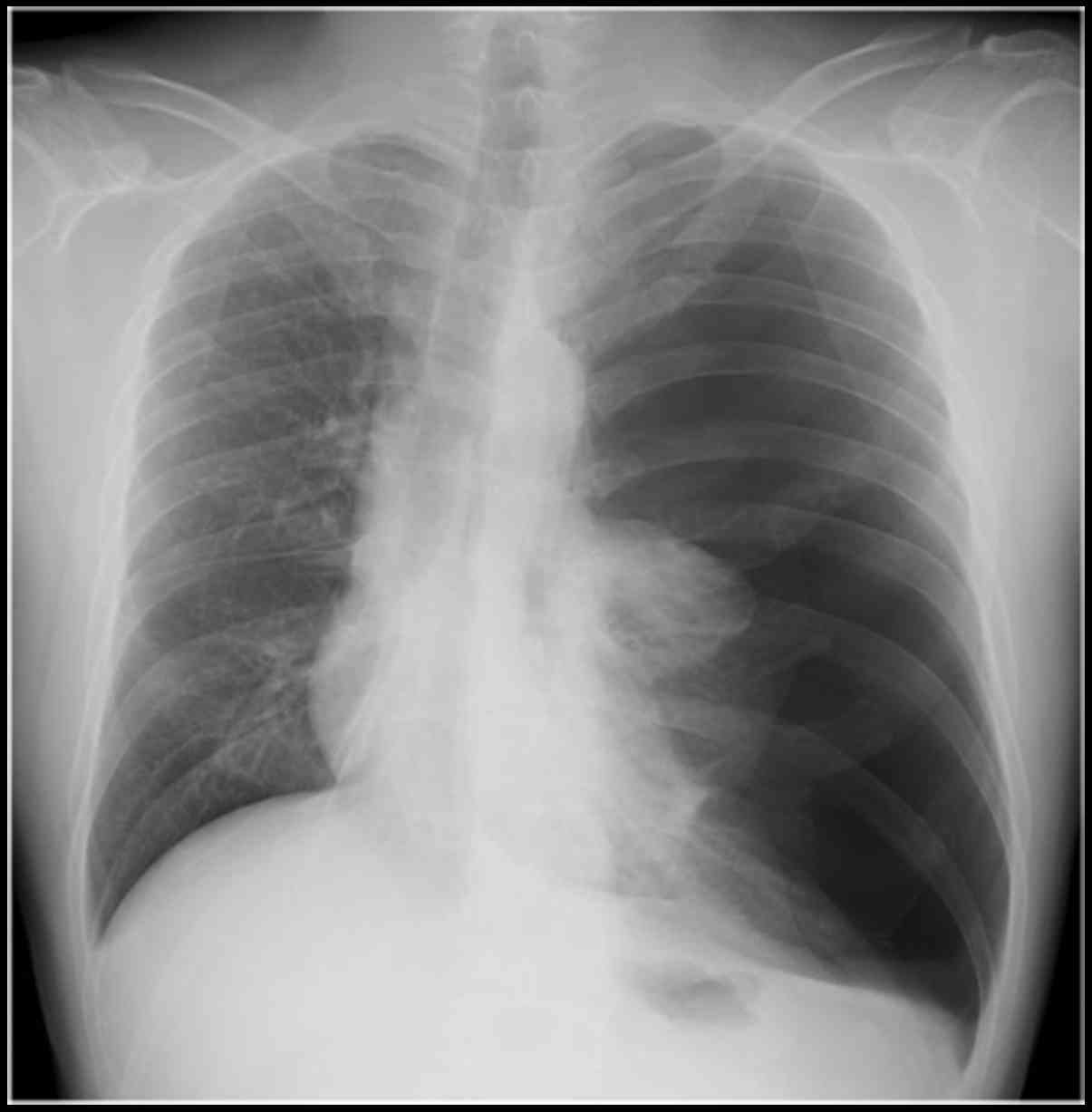
Plain radiography and CT displayed spontaneous
pneumothorax in the left lung (Fig.
4). Chest tube placement and pleurodesis in the right lung were
performed. Complete pulmonary expansion was achieved. Thereafter,
he began receiving chemotherapy again, and 2 years have passed
since the recovery from pneumothorax. At the last follow-up, the
patient was living with multiple cystic pulmonary metastases, and
he developed bone metastases in the rib, sacrum, and left femur;
cystic pulmonary metastases gradually appeared, and radiotherapy
was administered to each bone metastasis.
Discussion
Epithelioid sarcoma is a rare malignant tumor with
an incidence of 0.6–1.0% among sarcomas (8,9), and has
an extremely poor prognosis. Metastases develop in 40–50% of
patients, usually after repeated recurrences, and frequently
involve the lung and regional lymph node (10). Pulmonary metastases from sarcomas
usually appear on CT as small, solid, rounded peripheral nodules.
Pulmonary cystic metastasis from sarcoma is extremely rare. Only
seven previous studies in English on cystic pulmonary metastases
from epithelioid sarcoma have been described (1–7).
In our patient, the cystic lesion was not evident on
chest radiograpy, but was well visible with CT. CT has been shown
to be more sensitive in the detection of small metastatic foci.
Small cystic pulmonary lesions are also best visualized with CT.
Cystic change associated with primary lung cancer is relatively
frequent. Cystic pulmonary metastases usually form a thin smooth
wall similar to an emphysematous bulla or a pneumatocele (2). The accurate differential diagnosis of a
cavity metastasis from a benign bulla and primary lung cancer is
critically important. Metastatic tumor nodules are easily
recognizable when many solid tumor nodules are also present. When
few or no solid lesions are seen, however, cavity tumor nodules
must be differentiated from primary lung cancer, primary benign
cystic pulmonary disease (2,11,12).
Ultimately, the detailed pathological diagnosis is mandatory to
determine a cystic change in sarcoma.
An additional complication of pulmonary metastases
is the occurrence of spontaneous pneumothorax. Spontaneous
pneumothorax associated with metastatic tumor is a rare condition,
despite that sarcomatous pulmonary metastases are relatively
common. According to the previous paper by Hoag et al
(13), the incidence of pneumothorax
was between 5 to 10 per 100,000 per year, and the prevalence of
spontaneous pneumothorax in sarcoma was calculated to be 1.9%.
Additionally, Hoag et al (13)
reviewed 126 cases with pneumothorax secondary to sarcomatous
metastases, and the common histologies were found to be osteogenic
sarcoma (31.4%), angiosarcoma (18.3%), synovial sarcoma (8.5%), and
epithelioid sarcoma (1.3%). Transpleural rupture of subpleural
cysts may result in the development of pneumothoraces (14,15).
On reviewing the previously reported seven cases of
cystic pulmonary metastases from epithelioid sarcoma (Table I), most cases were found to have
multiple metastases in bilateral lungs, and pneumothorax was
reported to be a frequent complication (85.7%). Four cases with
cystic pulmonary metastases presented with bilateral pneumothorax,
two cases presented with unilateral pneumothorax, and only one
presented without pneumothorax.
 | Table I.Previous case reports of cystic
pulmonary metastases from epithelioid sarcoma. |
Table I.
Previous case reports of cystic
pulmonary metastases from epithelioid sarcoma.
|
|
|
|
| Lung metastases |
|
|
|
|
|---|
|
|
|
|
|
|
|
|
|
|
|---|
| Case | Author, year | Age/sex | Primary lesion | Lung | Number | Initial symptom | Pneumothorax | Treatment for
lung | Metastases at other
sites | Follow-up | Outcome | (Refs.) |
|---|
| 1 | Hasegawa et
al, 1999 | 29/F | Forearm | Bilateral | Multiple | Pneumothorax | Bilateral | Thoracotomy | Upper arm | 7 years | Alive | (1) |
| 2 | Chan et al,
2003 | 42/M | Thigh | Bilateral | Multiple | Identification by
CT | Bilateral | Thoracotomy Mesh
pleurodesis | None | N/A | Alive | (2) |
| 3 | Kikuchi et al,
2006 | 39/F | Hand | Bilateral | Multiple | Pneumothorax | Bilateral | Thoracotomy | None | 38 months | Dead | (3) |
| 4 | Choi et al,
2008 | 35/M | Scaip | Bilateral | Multiple | Identification by
CT | None | N/A | None | N/A | N/A | (4) |
| 5 | Liu et al,
2011 | 30/M | Hand | N/A | N/A | Identification by
CT | Right | None | Arm, Shoulder,
Scalp | N/A | N/A | (5) |
| 6 | Chong and
Casserly | 31/M | Forearm | Bilateral | Multiple | Pleuritis | Bilateral | Pleurodesis | None | N/A | Dead | (6) |
| 7 | Jeon et al,
2016 | 42/M | Perianal area | Bilateral | Multiple | Pleural effusion | Right | Thoracotomy | Inguinal lymph
node | 3 months | Dead | (7) |
| 8 | The present case | 33M | Forearm | Bilateral | Multiple | Identification by
CT | Right | Pleurodesis | Rib, pelvis,
femur | 24 months | Alive |
|
Regarding the treatment of pneumothorax, Chan et
al (2) reported a case of
recurrent spontaneous pneumothorax requiring surgery for the cystic
lesions. Traweek et al (14)
proposed that these lesions must be surgically removed from the
patients. On reviewing 153 cases of spontaneous
pneumothorax-complicated sarcoma by Hoag et al (13), chest tube placement was performed most
commonly (73.2%), followed by thoracic surgery (36.1%), pleurodesis
(21.6%), and aspiration (13.4%).
In the present case, pleurodesis was successfully
achieved. In the previous seven cases with pneumothorax of
epithelioid sarcoma (Table I),
thoracotomies were required in four cases, and pleurodesis in two
cases. Considering the treatment of pneumothorax associated with
cystic metastases from epithelioid sarcoma, chest tube placement
must be performed immediately, and then thoracic surgery should be
performed if all lung metastases are locally resectable. In
patients with multiple lung metastases, it may be advisable to
start systemic chemotherapy promptly at first in pleurodesis. If
pleurodesis is not effective, thoracic surgery is considerable.
Although, lung metastasis occasionally undergoes a
cystic change from a solid nodule due to central necrosis in the
natural cancer course, in the present case, the patient continued
to receive repeated chemotherapies of DOX/IFM, IFM, DTX/GEM, and
MTX. It is suggested that chemotherapeutic agents are likely to be
effective for microscopic lung metastases and responsible for the
discharge of necrotic materials inside, resulting in cystic changes
in the lung. Moreover, the necrosis of a peripherally located
pulmonary metastasis in response to chemotherapy is likely to be
responsible for pneumothorax (14).
After spontaneous pneumothorax, Hoag et al
(13) reported that the prognoses of
sarcomas after pneumothorax is generally poor, with 50% mortality
between 4 and 5 months, and 75% with 1-year mortality; furthermore,
less than 10% cases survived more than 2 years. However, from the
review of previous reports concerning pneumothorax from epithelioid
sarcomas, tumor progression with cystic pulmonary metastases of
epithelioid sarcoma may be less aggressive. Only one case (7) resulted in death within 3 months due to
massive pleural effusion from the first visit. Besides this case,
most patients survive for several years even if a pneumothorax due
to multiple pulmonary metastases is found. In the present case, the
patient was alive 24 months after the development of a spontaneous
pneumothorax.
At the follow-up, lung metastasis from sarcoma is
common. Close lung follow-up is required by the orthopedic
oncologist. Pulmonary metastasis from sarcomas is generally
well-circumscribed and solid nodules. However, whether the lung
nodule is cystic or non-cystic, careful attention must be paid
every three months at least when the nodule size and numbers of are
increasing in the lung.
In conclusion, pulmonary metastases forming small,
solid, rounded peripheral nodules from soft tissue sarcoma are very
common, but cystic pulmonary metastases from epithelioid sarcomas
are extremely rare. Additionally, pneumothorax formation is a very
common complication associated with cystic pulmonary metastases.
For orthopedic oncologists, close observation is mandatory to gain
awareness of this serious complication during follow-up in the
advanced stage of epithelioid sarcoma. Tumor progression after
pneumothorax appears to be less aggressive compared to that for
pneumothorax of other sarcoma types.
Acknowledgements
We would like to thank Yuko Kuwae for pathological
examinations and interpretations.
References
|
1
|
Hasegawa S, Inui K, Kamakari K, Kotoura Y,
Suzuki K and Fukumoto M: Pulmonary cysts as the sole metastatic
manifestation of soft tissue sarcoma: Case report and consideration
of the pathogenesis. Chest. 116:263–265. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chan DP, Griffith JF, Lee TW, Chow LT and
Yim AP: Cystic pulmonary metastases from epithelioid cell sarcoma.
Ann Thorac Surg. 75:1652–1654. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kikuchi E, Kinoshita I, Yamazaki K, Itoh
T, Shimizu T, Shimizu H and Nishimura M: Epithelioid sarcoma
presenting as pulmonary cysts with cancer antigen 125 expression.
Respirology. 11:826–829. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Choi SY, Kim YH, Kwon JB, Suh JH, Shin OR
and Hong SH: Epithelioid sarcoma metastatic to the lung as
pulmonary cysts without other metastatic manifestation. J Thorac
Oncol. 3:532–533. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu Y, Ma X, Zang D, Zhou C and Zhang J:
Epithelioid sarcoma with osteoporosis and pneumothorax. Eur J
Dermatol. 21:296–297. 2011.PubMed/NCBI
|
|
6
|
Chong SG and Casserly BP: An unusual cause
of spontaneous recurrent pneumothoraces. Br J Hosp Med (Lond).
73:110–111. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jeon SY, Yhim HY and Lee NR: Epithelioid
sarcoma with spontaneous pneumothorax and massive pleural effusion.
Korean J Intern Med. 31:191–193. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ross HM, Lewis JJ, Woodruff JM and Brennan
MF: Epithelioid sarcoma: Clinical behavior and prognostic factors
of survival. Ann Surg Oncol. 4:491–495. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Livi L, Shah N, Paiar F, Fisher C, Judson
I, Moskovic E, Thomas M and Harmer C: Treatment of epithelioid
sarcoma at the royal marsden hospital. Sarcoma. 7:149–152. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Oda Y, Cin PD and Laskin WB: Epithelioid
sarcoma. Fletcher CDM, Bridge JA, Hogendoorn PCW and Mertens F:
World Health Organization Classification of Tumors of Soft Tissue
and Bone, Lyon, International Agency for Research on Cancer. 1–217.
2013.
|
|
11
|
Corrin B, Liebow AA and Friedman PJ:
Pulmonary lymphangiomyomatosis. A review. Am J Pathol. 79:348–382.
1975.PubMed/NCBI
|
|
12
|
Mark EJ: Mesenchymal cystic hamartoma of
the lung. N Engl J Med. 315:1255–1259. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hoag JB, Sherman M, Fasihuddin Q and Lund
ME: A comprehensive review of spontaneous pneumothorax complicating
sarcoma. Chest. 138:510–518. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Traweek T, Rotter AJ, Swartz W and Azumi
N: Cystic pulmonary metastatic sarcoma. Cancer. 65:1805–1811. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Somasekharan Nair KK, Zabell AS, Vo KL and
Shaikh MA: Pneumothorax: A classical presentation of metastatic
scalp angiosarcoma. Ann Thorac Surg. 94:e77–e78. 2012. View Article : Google Scholar : PubMed/NCBI
|